The Effect of Three Gums on the Retrogradation of Indica Rice Starch
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Retrograded Starch by Autoclaving
2.3. Retrograded Starch Content
3. Results
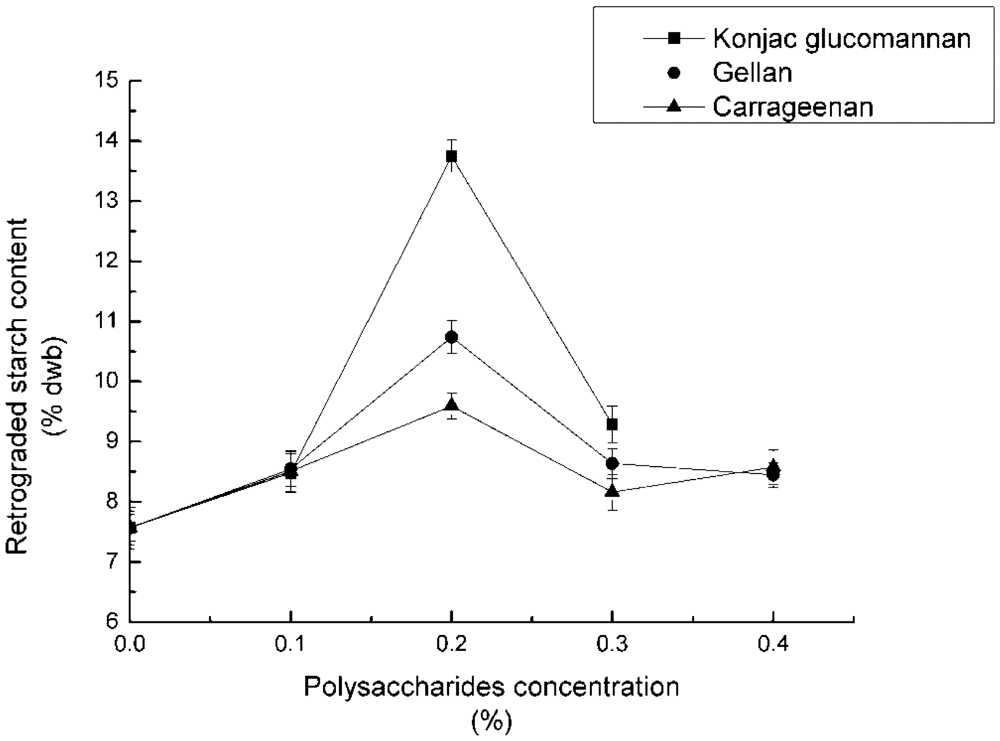
3.1. Effect of Concentration of Polysaccharides on RS3 Formation
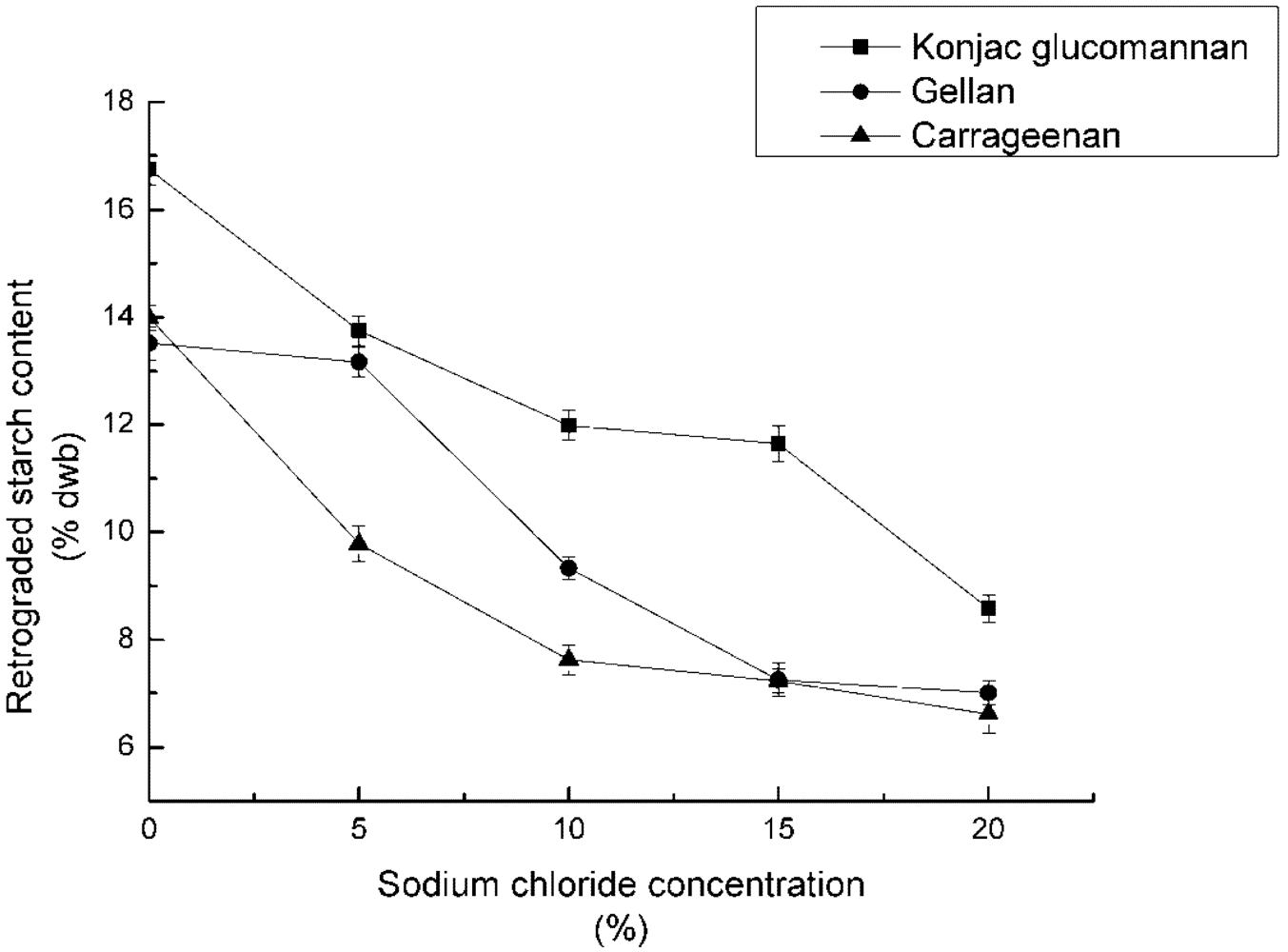
3.2. Effect of Concentration of Sodium Chloride on RS3 Formation
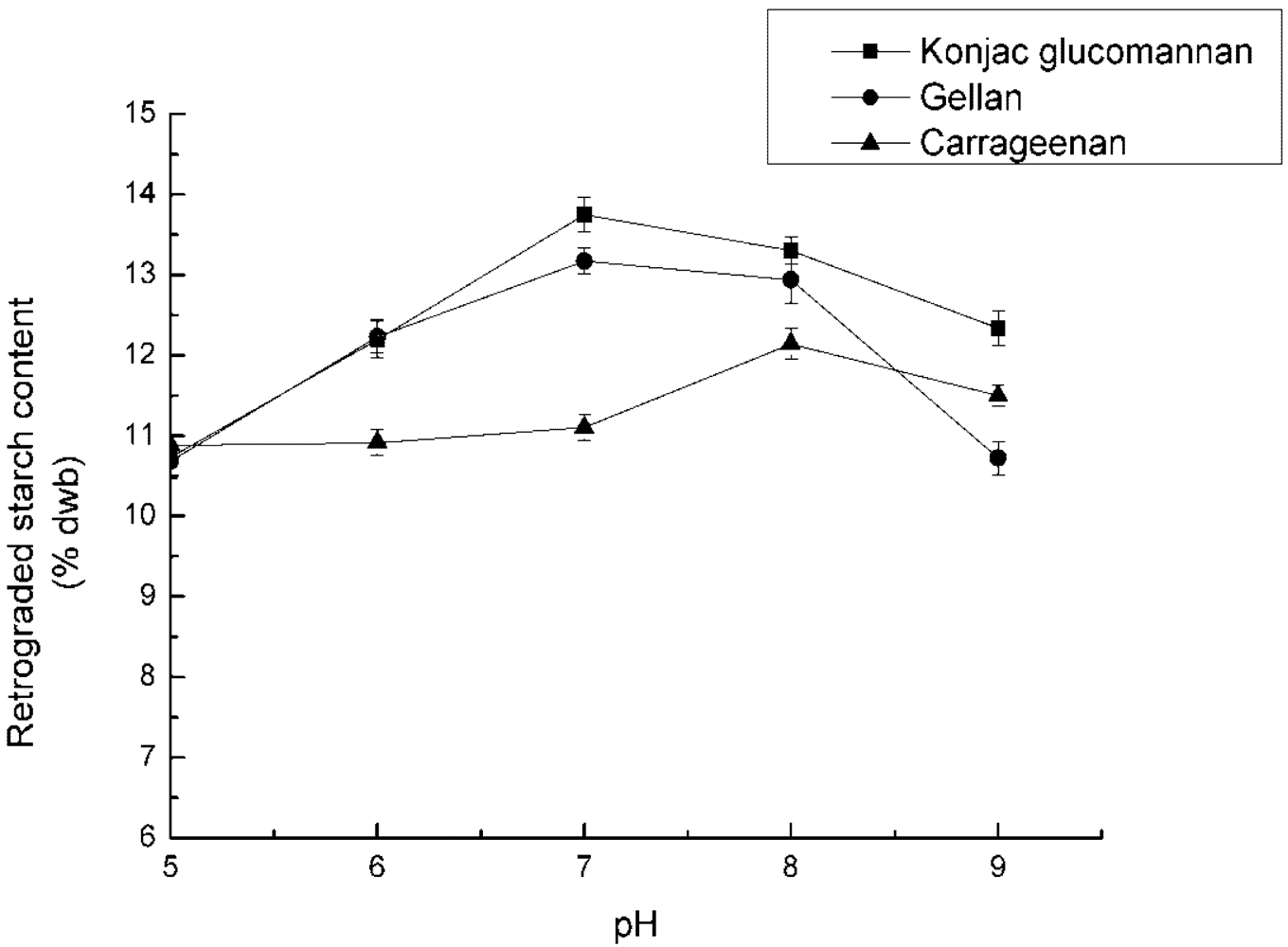
3.3. Effect of PH on RS3 Formation
3.4. Effect of Autoclaving Time on RS3 Formation

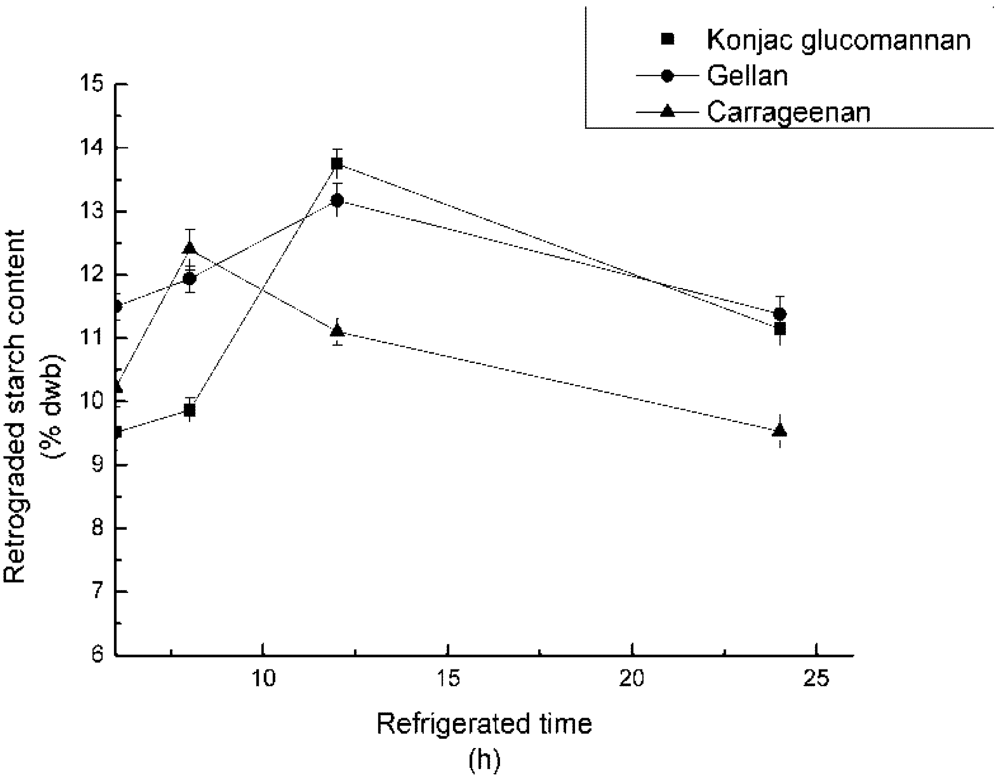
3.5. Effect of Refrigerated Time on RS3 Formation
4. Discussion
4.1. Effect of Concentration of Polysaccharides on RS3 Formation
4.2. Effect of Concentration of Sodium Chloride on RS3 Formation
4.3. Effect of pH on RS3 Formation
4.4. Effect of Autoclaving Time on RS3 Formation
4.5. Effect of Refrigerated Time on RS3 Formation
5. Conclusions
Acknowledgments
References
- Englyst, H.N.; Kingman, S.; Cummings, J. Classification and measurement of nutritionally important starch fractions. Eur. J. Clin. Nutr. 1992, 46, S33–S50. [Google Scholar]
- Jenkins, D.J.A.; Vuksan, V.; Kendall, C.W.C.; Wursch, P.; Jeffcoat, R.; Waring, S.; Mehling, C.C.; Vidgen, E.; Augustin, L.S.A.; Wong, E. Physiological effects of resistant starches on fecal bulk, short chain fatty acids, blood lipids and glycemic index. J. Am. Coll. Nutr. 1998, 17, 609–616. [Google Scholar]
- Sajilata, M.; Singhal, R.S.; Kulkarni, P.R. Resistant starch-a review. Compr. Rev. Food Sci. Food Saf. 2006, 5, 1–17. [Google Scholar] [CrossRef]
- Sanz, T.; Salvador, A.; Fiszman, S. Resistant starch (RS) in battered fried products: Functionality and high-fiber benefit. Food Hydrocoll. 2008, 22, 543–549. [Google Scholar] [CrossRef]
- Haralampu, S. Resistant starch—a review of the physical properties and biological impact of RS3. Carbohydr. Polym. 2000, 41, 285–292. [Google Scholar] [CrossRef]
- Hsein-Chih, H.W.; Sarko, A. The double-helical molecular structure of crystalline B-amylose. Carbohydr. Res. 1978, 61, 7–25. [Google Scholar] [CrossRef]
- Miles, M.J.; Morris, V.J.; Orford, P.D.; Ring, S.G. The roles of amylose and amylopectin in the gelation and retrogradation of starch. Carbohydr. Res. 1985, 135, 271–281. [Google Scholar] [CrossRef]
- Orford, P.D.; Ring, S.G.; Carroll, V.; Miles, M.J.; Morris, V.J. The effect of concentration and botanical source on the gelation and retrogradation of starch. J. Sci. Food Agric. 1987, 39, 169–177. [Google Scholar] [CrossRef]
- Gudmundsson, M. Retrogradation of starch and the role of its components. Thermochim. Acta 1994, 246, 329–341. [Google Scholar] [CrossRef]
- Fredriksson, H.; Silverio, J.; Andersson, R.; Eliasson, A.C.; Åman, P. The influence of amylose and amylopectin characteristics on gelatinization and retrogradation properties of different starches. Carbohydr. Polym. 1998, 35, 119–134. [Google Scholar] [CrossRef]
- Wunderlich, B. Macromolecular Physics: Crystal Nucleation, Growth, Annealing; Academic Press: Waltham, MA, USA, 1976. [Google Scholar]
- Eerlingen, R.; Crombez, M.; Delcour, J. Enzyme-resistant starch. I. Quantitative and qualitative influence of incubation time and temperature of autoclaved starch on resistant starch formation. Cereal Chem. 1993, 70, 339–339. [Google Scholar]
- Vandeputte, G.; Vermeylen, R.; Geeroms, J.; Delcour, J. Rice starches. III. Structural aspects provide insight in amylopectin retrogradation properties and gel texture. J. Cereal Sci. 2003, 38, 61–68. [Google Scholar] [CrossRef]
- Silverio, J.; Fredriksson, H.; Andersson, R.; Eliasson, A.C.; Åman, P. The effect of temperature cycling on the amylopectin retrogradation of starches with different amylopectin unit-chain length distribution. Carbohydr. Polym. 2000, 42, 175–184. [Google Scholar] [CrossRef]
- Feng, J.; Liu, W.-X.; Lin, Y.-L.; Liu, J.-Z. Advance and application prospect in starch anti-retrogradation. Food Sci. 2011, 32, 335–339. [Google Scholar]
- Huang, M.; Kennedy, J.; Li, B.; Xu, X.; Xie, B. Characters of rice starch gel modified by gellan, carrageenan, and glucomannan: A texture profile analysis study. Carbohydr. Polym. 2007, 69, 411–418. [Google Scholar] [CrossRef]
- Yang, X.-Q.; Shen, Y.-Y. The physicochemical properties, special functions, rheological properties of glucomannan and their application in different food industries. Beverage Fast Frozen Food Ind. 2002, 8, 29–33. [Google Scholar]
- Alloncle, M.; Lefebvre, J.; Llamas, G.; Doublier, J. A rheological characterization of cereal starch-galactomannan mixtures. Cereal Chem. 1989, 66, 90–93. [Google Scholar]
- Escarpa, A.; Gonzalez, M.; Morales, M.; Saura-Calixto, F. An approach to the influence of nutrients and other food constituents on resistant starch formation. Food Chem. 1997, 60, 527–532. [Google Scholar] [CrossRef]
- Charoenrein, S.; Tatirat, O.; Rengsutthi, K.; Thongngam, M. Effect of konjac glucomannan on syneresis, textural properties and the microstructure of frozen rice starch gels. Carbohydr. Polym. 2011, 83, 291–296. [Google Scholar]
- Chaisawang, M.; Suphantharika, M. Pasting and rheological properties of native and anionic tapioca starches as modified by guar gum and xanthan gum. Food Hydrocoll. 2006, 20, 641–649. [Google Scholar] [CrossRef]
- Funami, T.; Kataoka, Y.; Omoto, T.; Goto, Y.; Asai, I.; Nishinari, K. Effects of non-ionic polysaccharides on the gelatinization and retrogradation behavior of wheat starch. Food Hydrocoll. 2005, 19, 1–13. [Google Scholar] [CrossRef]
- Khanna, S.; Tester, R. Influence of purified konjac glucomannan on the gelatinisation and retrogradation properties of maize and potato starches. Food Hydrocoll. 2006, 20, 567–576. [Google Scholar] [CrossRef]
- García-Diz, L.; Manas, E.; Saura-Calixto, F. Analysis of resistant starch: A method for foods and food products. Food Chem. 1996, 56, 445–449. [Google Scholar] [CrossRef]
- Aparicio-Saguilán, A.; Flores-Huicochea, E.; Tovar, J.; García-Suárez, F.; Gutiérrez-Meraz, F.; Bello-Pérez, L.A. Resistant starch-rich powders prepared by autoclaving of native and lintnerized banana starch: Partial characterization. Starch 2005, 57, 405–412. [Google Scholar] [CrossRef]
- Viturawong, Y.; Achayuthakan, P.; Suphantharika, M. Gelatinization and rheological properties of rice starch/xanthan mixtures: Effects of molecular weight of xanthan and different salts. Food Chem. 2008, 111, 106–114. [Google Scholar] [CrossRef]
- Xiangqing, Y.; Yueyu, S. The physicochemical properties, special functions, rheological properties of glucomannan and their application in different food industries. Beverage Fast Frozen Food Ind. 2002, 8, 29–33. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Song, R.; Huang, M.; Li, B.; Zhou, B. The Effect of Three Gums on the Retrogradation of Indica Rice Starch. Nutrients 2012, 4, 425-435. https://doi.org/10.3390/nu4060425
Song R, Huang M, Li B, Zhou B. The Effect of Three Gums on the Retrogradation of Indica Rice Starch. Nutrients. 2012; 4(6):425-435. https://doi.org/10.3390/nu4060425
Chicago/Turabian StyleSong, Rukun, Min Huang, Bin Li, and Bin Zhou. 2012. "The Effect of Three Gums on the Retrogradation of Indica Rice Starch" Nutrients 4, no. 6: 425-435. https://doi.org/10.3390/nu4060425
APA StyleSong, R., Huang, M., Li, B., & Zhou, B. (2012). The Effect of Three Gums on the Retrogradation of Indica Rice Starch. Nutrients, 4(6), 425-435. https://doi.org/10.3390/nu4060425



