Probiotics in Inflammatory Bowel Diseases and Associated Conditions
Abstract
:1. Introduction
2. Ulcerative Colitis
2.1. Treatment of Active Inflammation in Ulcerative Colitis
- probiotics (Yakult™) + 5-ASA had similar effectiveness to placebo + 5-ASA for induction of remission [28]: probiotic 40%, placebo 30%, OR 0.64 (95% CI 0.10 to 4.10);
- probiotics (VSL#3™) + balsalazide had similar effectiveness to placebo + balsalazide for induction of remission [30]: probiotic 80%, placebo 70%, OR 0.58 (95% CI 0.18 to 1.91).
- probiotics (E. coli Nissle 1917) + steroids had similar effectiveness to mesalazine + steroids for induction of remission [31]: probiotic 68.4%, mesalazine 74.6%, OR 1.35 (95% CI 0.6 to 3.04).
| Participants (# Treated) | Trial Design | Probiotic (Strains) | Dosing (CFU/day) | Trial Length (Weeks) | References |
|---|---|---|---|---|---|
| 20 (10) | EBRPC | Blend Probiotic (Yakult™) | 1 × 1010 | 12 | [28] |
| 16 (8) | DBRPC | Single Probiotic + Prebioticmn (B. longum) | 4 × 1011 | 4 | [29] |
| 90 (30) | R | Blend Probiotic (VSL#3™) | 9 × 1011 | 8 | [30] |
| 102 (52) | DBRDD | Single strain (E. coli Nissle) | 1 × 1011 | 12 | [31] |
| 29 (14) | DBRPC | Blend Probiotic (VSL#3™) | 1 × 1011/kg | 52 | [34] |
| 120 (80) | R | Probiotic ± Prebiotic (B. longum) | 2 × 109 | 4 | [35] |
| 90 (70) | DBRPC | Single strain (E. coli Nissle) | 1–4 × 109 | 2–8 | [36] |
| 147 (77) | DBRPC | Blend Probiotic (VSL#3™) | 7.2 × 1012 | 12 | [37] |
2.2. Probiotics as Maintenance Therapy in Ulcerative Colitis
| Participants (# Treated) | Trial Design | Probiotic (Strains) | Dosing (CFU/day) | Trial Length (Months) | References |
|---|---|---|---|---|---|
| 103 (50) | DBRDD | Single strain (E. coli Nissle) | 5 × 1010 | 3 | [44] |
| 83 (39) | DBRDD | Single strain (E. coli Nissle) | 1 × 1011 | 12 | [31] |
| 21 (11) | R | Blend (Yakult®) | 1 × 1010 | 12 | [42] |
| 327 (162) | DBRDD | Single strain (E. coli Nissle) | 5 × 109 | 12 | [40] |
| 187 (127) | R | Single strain (L. rhamnosus GG) | 1.8 × 1010 | 12 | [41] |
| 29 (14) | DBRPC | Blend Probiotic (VSL#3™) | 1 × 1011/kg | 12 | [34] |
2.3. Pouchitis
2.3.1. Probiotics as Treatment of Pouchitis
2.3.2. Prevention of Initial Post-Operative Onset of Pouchitis
2.3.3. Maintenance of Pouchitis Remission (Table 3)
| Participants (# Treated) | Trial Design | Probiotic (Strains) | Dosing (CFU/day) | Trial Length (Months) | References |
|---|---|---|---|---|---|
| 40 (20) | DBRPC | Blend (VSL#3™) | 9 × 1011 | 12 | [55] |
| 31 (16) | R | Blend (VSL#3™) | 9 × 1011 | 12 | [57] |
| 40 (20) | DBRPC | Blend (VSL#3™) | 1.8 × 1012 | 9 | [58] |
| 36 (20) | DBRPC | Blend (VSL#3™) | 9 × 1011 | 12 | [51] |
4. Crohn’s Disease
4.1. Treatment of Active Crohn’s Disease Inflammation
4.2. Probiotics as Maintenance Therapy for Crohn’s Disease
| Participants (# Treated) | Trial Design | Probiotic (Strains) | Dosing (CFU/day) | Trial Length (Months) | References |
|---|---|---|---|---|---|
| 28 (16) | DBRPC | Single strain (E. coli Nissle 1917) | 5 × 1010 | 12 | [64] |
| 32 (16) | R | Single strain (S. boulardii) | N/A | 6 | [65] |
| 11 (5) | DBRPC | Single strain (L. rhamnosus strain GG) | 2 × 109 | 6 | [66] |
| 75 (39) | DBRPC | Single strain (L. rhamnosus strain GG) | 2 × 1010 | 24 | [67] |
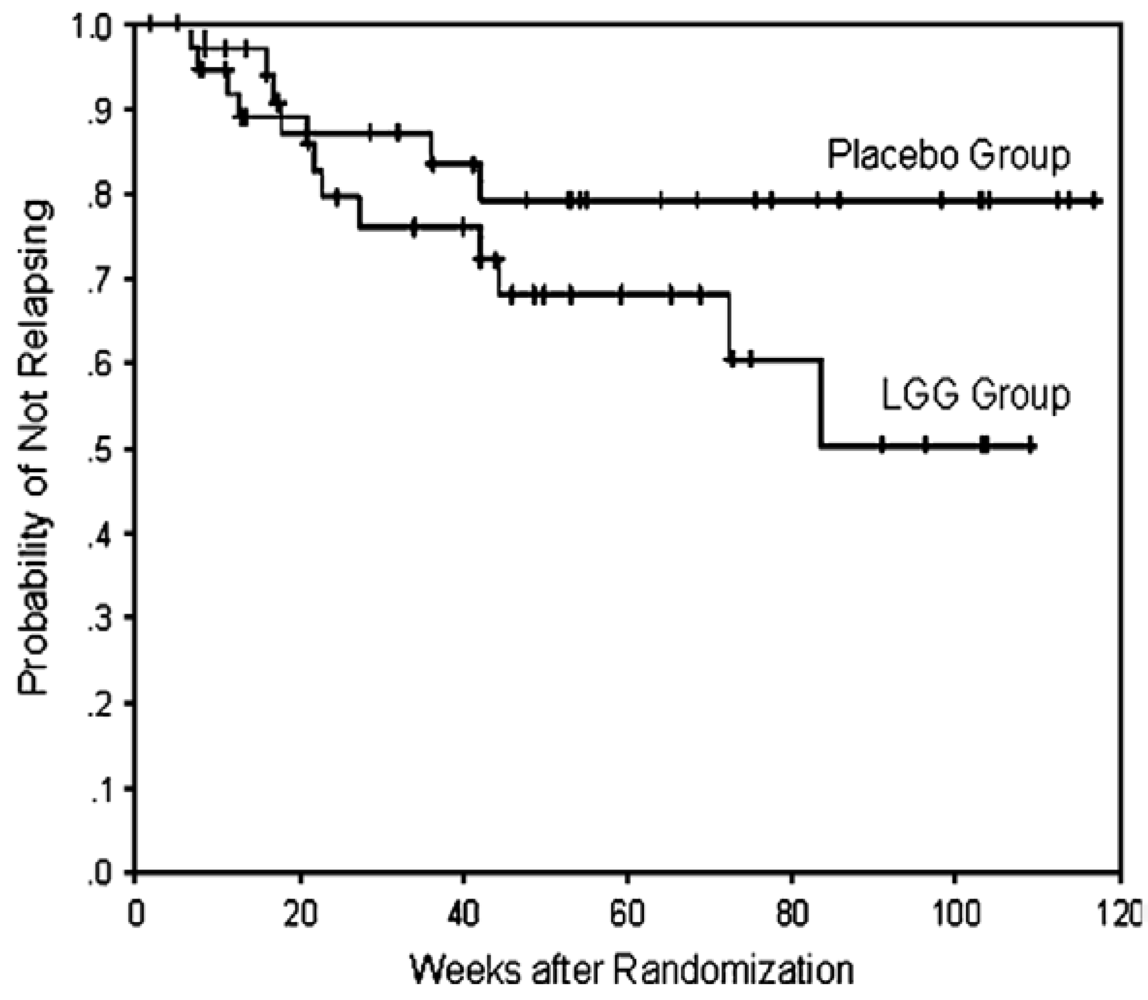
4.3. Probiotics for Prevention of Post-Operative Crohn’s Disease Recurrence
| Participants(# Treated) | Probiotic (Strains) | Dosing (CFU/day) | Trial Length (Months) | References |
|---|---|---|---|---|
| 45 (23) | Single Strain (L. rhamnosus GG) | 1.2 × 109 | 12 | [70] |
| 98 (48) | Single Strain (L. johnsonii LA1) | 4 × 109 | 6 | [71] |
| 70 (34) | Single Strain (L. johnsonii strain LA1) | 1010 | 3 | [72] |
| 30 (2) | Blend Probiotics + Prebiotics (P. pentoseceus, L. raffinolactis, L. paracasei susp paracasei 19 and L. plantarum 2362) | 1010 | 24 | [73] |
| 120 (58) | Blend Probiotics (VSL#3™) | 1.8 × 1012 | 3 | [74] |
5. Associated Conditions
5.1. Arthralgia
5.2. Spondylarthropathy
5.3. Sclerosing Cholangitis
6. Summary
Conflict of Interest
References
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T.; et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–65. [Google Scholar]
- Bousvaros, A.; Antonioli, D.A.; Colletti, R.B.; Dubinsky, M.C.; Glickman, J.N.; Gold, B.D.; Griffiths, A.M.; Jevon, G.P.; Higuchi, L.M.; Hyams, J.S.; Kirschner, B.S.; Kugathasan, S.; Baldassano, R.N.; Russo, P.A. Differentiating ulcerative colitis from Crohn disease in children and young adults: report of a working group of the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition and the Crohn’s and Colitis Foundation of America. J. Pediatr. Gastroenterol. Nutr. 2007, 44, 653–674. [Google Scholar] [PubMed]
- Benchimol, E.I.; Guttmann, A.; Griffiths, A.M.; Rabeneck, L.; Mack, D.R.; Brill, H.; Howard, J.; Guan, J.; To, T. Increasing incidence of pediatric inflammatory bowel disease in Ontario, Canada: evidence from health administrative data. Gut 2009, 58, 1490–1497. [Google Scholar]
- Barrett, J.C.; Hansoul, S.; Nicolae, D.L.; Cho, J.H.; Duerr, R.R.; Rioux, J.D.; Brant, S.R.; Silverberg, M.S.; Taylor, K.D.; Barmada, M.M.; et al. Genome-wide association defines more that 30 distinct susceptibility loci for Crohn’s disease. Nat. Genet. 2008, 40, 955–962. [Google Scholar] [PubMed]
- Imielinski, M.; Baldassano, R.N.; Griffiths, A.; Russell, R.K.; Annese, V.; Dubinsky, M.; Kugathasan, S.; Bradfield, J.P.; Walters, T.D.; Sleiman, P.; et al. Common variants at five new loci associated with early-onset inflammatory bowel disease. Nat. Genet. 2009, 41, 1335–1340. [Google Scholar]
- McGovern, D.P.; Gardet, A.; Torkvist, L.; Goyette, P.; Essers, J.; Taylor, K.D.; Neale, B.M.; Ong, R.T.; Lagace, C.; Li, C.; et al. Genome-wide association identifies multiple ulcerative colitis susceptibility loci. Nat. Genet. 2010, 42, 332–337. [Google Scholar]
- O’Sullivan, M.; O’Morain, C. Nutrition in inflammatory bowel disease. Best. Prac. Res. Clin. Gastroenterol. 2006, 20, 561–573. [Google Scholar]
- D’Souza, S.; Levy, E.; Mack, D.; Israel, D.; Lambrette, P.; Ghadirian, P.; Deslandres, C.; Morgan, K.; Seidman, E.G.; Amre, D.K. Dietary patterns and risk for Crohn’s disease in children. Inflamm. Bowel. Dis. 2008, 14, 367–373. [Google Scholar]
- Mahid, S.S.; Minor, K.S.; Soto, R.E.; Hornung, C.A.; Galandiuk, S. Smoking and inflammatory bowel disease: a meta-analysis. Mayo Clin. Proc. 2006, 81, 1462–1471. [Google Scholar]
- Jones, D.T.; Osterman, M.T.; Bewtra, M.; Lewis, J.D. Passive smoking and inflammatory bowel disease: a meta-analysis. Am. J. Gastroenterol. 2008, 103, 2382–2393. [Google Scholar]
- Wang, T.T.; Dabbas, B.; Laperriere, D.; Bitton, A.J.; Soualhine, H.; Tavera-Mendoza, L.E.; Dionne, S.; Servant, M.J.; Bitton, A.; Seidman, E.G.; et al. Direct and indirect induction by 1,25-dihydroxyvitamin D3 of the NOD2/CARD15-defensin β2 innate immune pathway defective in Crohn disease. J. Biol. Chem. 2010, 285, 2227–2231. [Google Scholar]
- Kong, J.; Zhang, Z.; Musch, M.W.; Ning, G.; Sun, J.; Hart, J.; Bissonnette, M.; Li, Y.C. Novel role of the vitamin D receptor in maintaining the integrity of the intestinal mucosal barrier. Am. J. Physiol. Gastrointest. Liver Physiol. 2008, 294, G208–G218. [Google Scholar]
- Bernstein, C.N.; Rawsthorne, P.; Cheang, M.; Blanchard, J.F. A population-based case control study of potential risk factors for IBD. Am. J. Gastroenterol. 2006, 101, 993–1002. [Google Scholar]
- Radon, K.; Windstetter, D.; Poluda, A.L.; Mueller, B.; von Mutius, E.; Koletzko, S. Chronic Autoimmune Disease and Animal Contact Study Group. Contact with farm animals in early life and juvenile inflammatory bowel disease: A case-control study. Pediatrics 2007, 120, 354–361. [Google Scholar] [PubMed]
- Pinsk, V.; Lemberg, D.A.; Grewal, K.; Barker, C.C.; Schreiber, R.A.; Jacobson, K. Inflammatory bowel disease in the south Asian pediatric population of British Columbia. Am. J. Gastroenterol. 2007, 102, 1077–1083. [Google Scholar]
- Sartor, R.B. Mechanisms of disease: pathogenesis of Crohn’s disease and ulcerative colitis. Nat. Clin. Pract. Gastroenterol. Hepatol. 2006, 3, 390–407. [Google Scholar]
- Clavel, T.; Haller, D. Bacteria- and host-derived mechanisms to control intestinal epithelial cell homeostasis: implications for chronic inflammation. Inflamm. Bowel. Dis. 2007, 13, 1153–1164. [Google Scholar]
- Garrett, W.S.; Lord, G.M.; Punit, S.; Lugo-Villarino, G.; Mazmanian, S.K.; Ito, S.; Glickman, J.N.; Glimcher, L.H. Communicable ulcerative colitis induced by T-bet deficiency in the innate immune system. Gut 2007, 131, 33–45. [Google Scholar]
- Dubinsky, M.C.; Kugathasan, S.; Mei, L.; Picornall, Y.; Nebel, J.; Wrobel, I.; Quiros, A.; Silber, G.; Wahbeh, G.; Katzir, L.; et al. Increased immune reactivity predicts aggressive complicating Crohn’s disease in children. Clin. Gastroenterol. Hepatol. 2008, 6, 1105–1111. [Google Scholar] [CrossRef] [PubMed]
- Szajewska, H.; Setty, M.; Mrukowicz, J.; Guandalini, S. Probiotics in gastrointestinal diseases in children: hard and not-so-hard evidence of efficacy. J. Pediatric. Gastroenterol. Nutr. 2006, 42, 454–475. [Google Scholar]
- Li, F.X.; Verhoef, M.J.; Best, A.; Otley, A.; Hilsden, R.J. Why patients with inflammatory bowel disease use or do not use complementary and alternative medicine: a Canadian national survey. Can. J. Gastroenterol. 2005, 19, 567–573. [Google Scholar]
- Quattropani, C.; Ausfeld, B.; Straumann, A.; Heer, P.; Selbold, F. Complementary alternative medicine in patients with inflammatory bowel disease: use and attitudes. Scand. J. Gastroenterol. 2003, 38, 277–282. [Google Scholar]
- Heuschkel, R.; Afzal, N.; Wuerth, A.; Zurakowski, D.; Leichtner, A.; Kemper, K.; Tolia, V.; Bousvaros, A. Complementary medicine use in children and young adults with inflammatory bowel disease. Am. J. Gastroenterol. 2002, 97, 382–388. [Google Scholar]
- Day, A.S.; Whitten, K.E.; Bohane, T.D. Use of complementary and alternative medicines in children and adolescents with inflammatory bowel disease. J. Paediatr. Child Health 2004, 40, 681–684. [Google Scholar] [CrossRef] [PubMed]
- Hilsden, R.J.; Verhoef, M.J.; Best, A.; Pocobelli, G. Complementary and alternative medicine use in Canadian patients with inflammatory bowel disease: results of a national survey. Am. J. Gastroenterol. 2003, 98, 1563–1568. [Google Scholar]
- Joos, S.; Rosemann, T.; Szecsenyi, J.; Hahn, E.G.; Willich, S.N.; Brinkhaus, B. Use of complementary and alternative medicine in Germany—a survey of patients with inflammatory bowel disease. BMC Complement. Altern. Med. 2006, 6, 19. [Google Scholar] [CrossRef] [PubMed]
- Wong, A.P.; Clark, A.L.; Garnett, E.A.; Acree, M.; Cohen, S.A.; Ferry, G.D.; Heyman, M.B. Use of complementary medicine in pediatric patients with inflammatory bowel disease: results from a multicenter survey. J. Pediatr. Gastroenterol. Nutr. 2008, 44, 55–60. [Google Scholar]
- Kato, K.; Mizuno, S.; Umesaki, Y.; Ishii, Y.; Sugitani, M.; Imaoka, A.; Otsuka, M.; Hasunuma, O.; Kurihara, R.; Iwasaki, A.; et al. Randomized placebo-controlled trial assessing the effect of bifidobacteria-fermented milk on active ulcerative colitis. Aliment. Pharmacol. Ther. 2004, 20, 1133–1141. [Google Scholar]
- Furrie, E.; Macfarlane, S.; Kennedy, A.; Cummings, J.H.; Walsh, S.V.; O’Neill, D.A.; Macfarlane, G.T. Synbiotic therapy (Bifidobacterium longum/Synergy 1) initiates resolution of inflammation in patients with active ulcerative colitis: a randomized controlled pilot trial. Gut 2005, 54, 242–249. [Google Scholar]
- Tursi, A.; Brandimarte, G.; Giorgetti, G.M.; Forti, G.; Modeo, M.E.; Gigliobianco, A. Low-dose balsalazide plus a high-potency probiotic preparation is more effective than balsalazide alone or mesalazine in the treatment of acute-mild-to-moderate ulcerative colitis. Med. Sci. Monit. 2004, 10, Pl126–Pl131. [Google Scholar]
- Rembacken, B.J.; Snelling, A.M.; Hawkey, P.M.; Chalmers, D.M.; Axon, A.T. Non-pathogenic Escherichia coli versus mesalazine for the treatment of ulcerative colitis: a randomized trial. Lancet 1999, 354, 635–639. [Google Scholar]
- Mallon, P.T.; McKay, D.; Kirk, S.J.; Gardiner, K. Probiotics for induction of remission in ulcerative colitis. Cochrane Database Syst. Rev. 2007, 4, CD005573. [Google Scholar]
- Hanauer, S.B.; Sandborn, W.J.; Kornbluth, A.; Katz, S.; Safdi, M.; Woogen, S.; Regalli, G.; Yeh, C.; Smith-Hall, N.; Ajayi, F. Delayed-release oral mesalamine at 4.8 g/day (800 mg tablet) for the treatment of moderately active ulcerative colitis: the Ascend II trial. Am. J. Gastroenterol. 2005, 100, 2478–2485. [Google Scholar] [CrossRef] [PubMed]
- Miele, E.; Pascarella, F.; Giannetti, E.; Quaglietta, L.; Baldassano, R.N.; Staiano, A. Effect of a probiotic preparation (VSL#3) on induction and maintenance of remission in children with ulcerative colitis. Am. J. Gastroenterol. 2009, 104, 437–443. [Google Scholar]
- Fujimori, S.; Gudis, K.; Mitsui, K.; Seo, T.; Yonezawa, M.; Tanaka, S.; Tatsuguchi, A.; Sakamoto, C. A randomized controlled trial on the efficacy of synbiotic versus probiotic or prebiotic treatment to improve the quality of life in patients with ulcerative colitis. Nutrition 2009, 25, 520–525. [Google Scholar] [CrossRef] [PubMed]
- Sood, A.; Midha, V.; Makharia, G.K.; Ahuja, V.; Singal, D.; Goswami, P.; Tandon, R.K. The probiotic preparation, VSL#3 induces remission in patients with mild-to-moderately active ulcerative colitis. Clin. Gastroenterol. Hepatol. 2009, 7, 1202–1209. [Google Scholar]
- Matthes, H.; Krummenerl, T.; Giensch, M.; Wolff, C.; Schulze, J. Clinical trial: probiotic treatment of acute distal ulcerative colitis with rectally administered Escherichia coli Nissle 1917 (EcN). BMC Complement. Altern. Med. 2010, 10, 13. [Google Scholar] [CrossRef] [PubMed]
- Hyams, J.; Markowitz, J.; Lerer, T.; Griffiths, A.; Mack, D.; Bousvaros, A.; Otley, A.; Evans, J.; Pfefferkorn, M.; Rosh, J.; et al. The Natural History of Corticosteroid Therapy for Ulcerative Colitis in Children. Clin. Gastroenterol. Hepatol. 2006, 4, 1118–1123. [Google Scholar]
- Hanauer, S.B. Medical therapy for ulcerative colitis 2004. Gastroenterology 2004, 126, 1582–1592. [Google Scholar]
- Kruis, W.; Fric, P.; Pokrotnieks, J.; Lukas, M.; Fixa, B.; Kascak, M.; Kamm, M.A.; Weismueller, J.; Beglinger, C.; Stolte, M.; et al. Maintaining remission of ulcerative colitis with the probiotic Escherichia coli Nissle 1917 is as effective as with standard mesalazine. Gut 2004, 53, 1617–1623. [Google Scholar] [CrossRef] [PubMed]
- Zocco, M.A.; dal Verme, L.Z.; Cremonini, F.; Piscaglia, A.A.; Nista, E.C.; Candelli, M.; Novi, M.; Rigante, D.; Cazzato, I.A.; Ojetti, V.; et al. Efficacy of Lactobacillus GG in maintaining remission of ulcerative colitis. Aliment. Pharmacol. Ther. 2006, 23, 1567–1574. [Google Scholar]
- Ishikawa, H.; Akedo, I.; Umesmaki, Y.; Tanaka, R.; Imaoka, A.; Otani, T. Randomized controlled trial of the effect of Bifidobacteria-fermented milk on ulcerative colitis. J. Am. Coll. Nutr. 2003, 22, 56–63. [Google Scholar]
- Sandborn, W.J. Treatment of ulcerative colitis with oral mesalamine: advances in drug formulation, efficacy expectations and dose response, compliance, and chemoprevention. Rev. Gastroenterol. Disord. 2006, 6, 97–105. [Google Scholar] [PubMed]
- Kruis, W.; Schutz, E.; Fric, P.; Fixa, B.; Judmaier, G.; Stolte, M. Double-blind comparison of an oral Escherichia coli preparation and mesalazine in maintaining remission of ulcerative colitis. Aliment. Pharmacol. Ther. 1997, 11, 853–858. [Google Scholar] [PubMed]
- Elahi, B.; Nikfar, S.; Derakhshani, S.; Vafaie, M.; Abdollahi, M. On the benefit of probiotics in the management of pouchitis in patients underwent ileal pouch anal anastomosis: A meta-analysis of controlled clinical trials. Dig. Dis. Sci. 2008, 53, 1278–1284. [Google Scholar]
- Pardi, D.S.; D’Haens, G.; Shen, B.; Campbell, S.; Gionchetti, P. Clinical guidelines for the management of pouchitis. Inflamm. Bowel. Dis. 2009, 15, 1424–1431. [Google Scholar]
- Kuisma, J.; Mentula, S.; Jarvinen, H.; Kahri, A.; Saxelin, M.; Farkkila, M. Effect of Lactobacillus rhamnosus GG on ileal pouch inflammation and microbial flora. Aliment. Pharmacol. Ther. 2003, 17, 509–515. [Google Scholar]
- Sandborn, W.J.; Tremaine, W.J.; Batts, K.P.; Pemberton, J.H.; Philips, S.F. Pouchitis following ileal pouch-anal anastomosis: a pouchitis disease activity index. Mayo Clin. Proc. 1994, 69, 409–415. [Google Scholar]
- Laake, K.O.; Bjorneklett, A.; Aamodt, G.; Aabakken, L.; Jacobsen, M.; Bakka, A.; Vatn, M.H. Outcome of four weeks’ intervention with probiotics on symptoms and endoscopic appearance after surgical reconstruction with a J-configuration ileal-pouch-anal-anastomosis in ulcerative colitis. Scand. J. Gastroenterol. 2005, 40, 43–51. [Google Scholar]
- Gionchetti, P.; Rizzello, F.; Morselli, C.; Poggioli, G.; Tambasco, R.; Calabrese, C.; Brigidi, P.; Vitali, B.; Straforini, G.; Campieri, M. High-dose probiotics for the treatment of active pouchitis. Dis. Colon Rectum 2007, 50, 2075–2078. [Google Scholar]
- Mimura, T.; Rizzello, F.; Helwig, U.; Poggioli, G.; Schreiber, S.; Talbot, I.C.; Nicholls, R.J.; Gionchetti, P.; Campieri, M.; Kamm, M.A. Once daily high dose probiotic therapy (VSL#3) for maintaining remission in recurrent or refractory pouchitis. Gut 2004, 53, 108–114. [Google Scholar]
- Gotteland, M.; Poliak, L.; Cruchet, S.; Brunser, O. Effect of regular ingestion of Saccharomyces boulardii plus inulin or Lactobacillus acidophilus in children colonized with Helicobacter pylori. Acta Paediatr. 2005, 94, 1747–1751. [Google Scholar] [PubMed]
- Gotteland, M.; Brusner, O.; Cruchet, S. Systematic review: Are probiotics useful in controlling gastric colonization by Helicobacter pylori? Aliment. Pharmacol. Ther. 2006, 23, 1077–1086. [Google Scholar] [CrossRef] [PubMed]
- Pantoflickova, D.; Corthesy-Theulaz, I.; Dorta, G.; Stolte, M.; Isler, P.; Rochat, F.; Enslen, M.; Blum, A.L. Favourable effect of regular intake of fermented milk containing Lactobacillus johnsonii on Helicobacter pylori associated gastritis. Aliment. Pharmacol. Ther. 2003, 18, 805–813. [Google Scholar] [CrossRef] [PubMed]
- Gionchetti, P.; Rizzello, F.; Helwig, U.; Venturi, A.; Lammers, K.M.; Brigidi, P.; Vitali, B.; Poggioli, G.; Miglioli, M.; Campieri, M. Prophylaxis of pouchitis onset with probiotic therapy: A double-blind, placebo-controlled trial. Gastoenterology 2003, 124, 1202–1209. [Google Scholar]
- Holubar, S.D.; Cima, R.R.; Sandborn, W.J.; Pardi, D.S. Treatment and prevention of pouchitis after ileal pouch-anal anastomosis for chronic ulcerative colitis. Cochrane Database Syst. Rev. 2010, 6, CD001176. [Google Scholar]
- Pronio, A.; Montesani, C.; Butteroni, C.; Vecchione, S.; Mumolo, G.; Vestri, A.; Vitolo, D.; Boirivant, M. Probiotic administration in patients with ileal pouch-anal anastomosis for ulcerative colitis is associated with expansion of mucosal regulatory cells. Inflamm. Bowel. Dis. 2008, 14, 662–668. [Google Scholar] [PubMed]
- Gionchetti, P.; Rizzello, F.; Venturi, A.; Brigidi, P.; Matteuzzi, D.; Bazzocchi, G.; Poggioli, G.; Miglioli, M.; Campieri, M. Oral bacteriotherapy a maintenance treatment in patients with chronic pouchitis: A double-blind, placebo-controlled trial. Gastroenterology 2000, 119, 305–309. [Google Scholar]
- Shen, B.; Brzezinski, A.; Fazio, V.W.; Remzi, F.H.; Achkar, J.P.; Bennett, A.E.; Sherman, K.; Lashner, B.A. Maintenance therapy with a probiotic in antibiotic-dependent pouchitis: experience in clinical practice. Aliment. Pharmacol. Ther. 2005, 22, 721–728. [Google Scholar]
- Fujimori, S.; Tatsuguchi, A.; Gudis, K.; Kishida, T.; Ehara, A.; Kobayahi, T.; Sekita, Y.; Seo, T.; Sakamoto, C. High dose probiotic and prebiotic co therapy for remission induction of active Crohn’s disease. J. Gastroenterol. Hepatol. 2007, 22, 1199–1204. [Google Scholar]
- Gupta, P.; Andrew, H.; Kirschner, B.S.; Guandalini, S. Is Lactobacillus GG helpful in children with Crohn’s disease? Results of a preliminary, open-label study. J. Pediatr. Gastroenterol. Nutr. 2000, 31, 453–457. [Google Scholar]
- Turner, D.; Griffiths, A.M.; Walters, T.D.; Seah, T.; Markowitz, J.; Pfefferkorn, M.; Keljo, D.; Otley, A.; Leleiko, N.S.; Mack, D.; Hyams, J.; Levine, A. Appraisal of the Pediatric Crohn’s Disease Activity Index (PCDAI) on four prospectively collected datasets: recommended cutoff values and clinimetric properties. Am. J. Gastroenterol. 2010, 105, 2085–2092. [Google Scholar]
- Butterworth, A.D.; Thomas, A.G.; Akobeng, A.K. Probiotics for induction of remission in Crohn’s disease. Cochrane Database Syst. Rev. 2008, 3, CD006634. [Google Scholar]
- Malchow, H. Crohn’s disease and Escherichia coli: A new approach in therapy to maintain remission of colonic Crohn’s disease. J. Clin. Gastroenterol. 1997, 25, 653–658. [Google Scholar]
- Guslandi, M.; Mezzi, G.; Sorghi, M.; Testoni, P.A. Saccharomyces boulardii in maintenance treatment of Crohn’s disease. Dig. Dis. Sci. 2000, 45, 1462–1464. [Google Scholar] [CrossRef] [PubMed]
- Schultz, M.; Timmer, A.; Herfarth, H.H.; Sartor, R.B.; Vanderhoof, J.A.; Rath, H.C. Lactobacillus GG in inducing and maintaining remission of Crohn’s disease. BMC Gastroenterol. 2004, 4, 5. [Google Scholar]
- Bousvaros, A.; Guandalini, S.; Baldassano, R.N.; Botelho, C.; Evans, J.; Ferry, G.D.; Goldin, B.; Hartigan, L.; Kugathasan, S.; Levy, J.; et al. A randomized, double-blind trial of Lactobacillus GG versus placebo in addition to standard maintenance therapy for children with Crohn’s disease. Inflamm. Bowel. Dis. 2005, 11, 833–839. [Google Scholar] [PubMed]
- Rolfe, V.E.; Fortun, P.J.; Hawkey, C.J.; Bath-Hextall, F.J. Probiotics for maintenance of remission in Crohn’s disease. Cochrane Database Syst. Rev. 2006, 4, CD004826. [Google Scholar]
- Akobeng, A.K.; Gardener, E. Oral 5-aminosalicylate acid for maintenance of medically-induced remission in Crohn’s disease. Cochrane Database Syst. Rev. 2005, 1, CD003715. [Google Scholar]
- Prantera, C.; Scribano, M.L.; Falasco, G.; Andreoli, A.; Luzi, C. Ineffectiveness of probiotics in preventing recurrence after curative resection for Crohn’s disease: a randomized controlled trial with Lactobacillus GG. Gut 2002, 51, 405–409. [Google Scholar]
- Marteau, P.; Lemann, M.; Seksik, P.; Laharie, D.; Colombel, J.F.; Bouhnik, Y.; Cadiot, G.; Soule, J.C.; Bourreille, A.; Metman, E.; et al. Ineffectiveness of Lactobacillus johnsonii LA1 for prophylaxis of postoperative recurrence in Crohn’s disease: a randomized, double blind, placebo controlled GETAID trial. Gut 2006, 55, 842–847. [Google Scholar] [PubMed]
- Van Gossum, A.; Dewit, O.; Louis, E.; de Hertogh, G.; Baert, F.; Fontaine, F.; DeVos, M.; Enslen, M.; Paintin, M.; Franchimont, D. Multicenter randomized-controlled clinical trial of probiotics (Lactobacillus johnsonii, LA1) on early endoscopic recurrence of Crohn’s disease after ileo-cecal resection. Inflamm. Bowel. Dis. 2007, 13, 135–142. [Google Scholar] [PubMed]
- Chermesh, I.; Tamir, A.; Reshef, R.; Chowers, Y.; Suissa, A.; Katz, D.; Gelber, M.; Halpern, Z.; Bengmark, S.; Eliakim, R. Failure of Synbiotic 2000 to prevent postoperative recurrence of Crohn’s disease. Dig. Dis. Sci. 2007, 52, 385–389. [Google Scholar]
- Madsen, K.; Backer, J.L.; Leddin, D.; Dieleman, L.A.; Bitton, A.; Feagan, B.; Petrunia, D.M.; Chiba, N.; Enns, R.A.; Fedorak, R. A randomized trial of VSL#3 for the prevention of endoscopic recurrence following surgery for Crohn’s disease. Gastroenterology 2008, 134 (Suppl. 1), A361. [Google Scholar]
- Doherty, G.; Bennett, G.; Patil, S.; Cheifetz, A.; Moss, A.C. Interventions for prevention of post-operative recurrence of Crohn’s disease. Cochrane Database Syst. Rev. 2009, 4, CD006873. [Google Scholar]
- Karimi, O.; Pena, S.; van Bodegraven, A.A. Probiotics (VSL#3) in arthralgia in patients with ulcerative colitis and Crohn’s disease: A pilot study. Drugs Today 2005, 41, 453–459. [Google Scholar]
- Brophy, S.; Burrows, C.L.; Brooks, C.; Gravenor, M.B.; Siebert, S.; Allen, S.J. Internet-based randomized controlled trials for the evaluation of complementary and alternative medicines: probiotics in spondylarthropathy. BMC Musculoskelet. Disord. 2008, 9, 4. [Google Scholar]
- Vleggaar, F.P.; Monkelbaan, J.F.; van Erpecum, K.J. Probiotics in primary sclerosing cholangitis: A randomized placebo-controlled crossover pilot study. Eur. J. Gastroenterol. Hepatol. 2008, 20, 688–692. [Google Scholar]
- Sokol, H.; Pigneur, B.; Watterlot, L.; Lakhdari, O.; Bermudez-Humaran, L.G.; Gratadoux, J.J.; Blugeon, S.; Bidonneau, C.; Furet, J.P.; Corthier, G.; et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn’s disease patients. Proc. Natl. Acad. Sci. USA 2008, 105, 16731–16736. [Google Scholar]
- Raz, I.; Gollop, N.; Polak-Charcon, S.; Schwartz, B. Isolation and characterization of new putative probiotic bacteria from human colonic flora. Br. J. Nutr. 2007, 97, 725–734. [Google Scholar]
- Kajiura, T.; Takeda, T.; Sakata, S.; Sakamoto, M.; Hashimoto, M.; Suzuki, M.; Benno, Y. Change of intestinal microbiota with elemental diet and its impact on therapeutic effects in a murine model of chronic colitis. Dig. Dis. Sci. 2009, 54, 1892–1900. [Google Scholar]
- Braat, H.; Rottiers, P.; Hommes, D.W.; Huyghebaert, N.; Remaut, E.; Ramon, J.P.; van Deventer, S.J.; Neirynck, S.; Peppelenbosch, M.P.; Steidler, L. A phase 1 trial with transgenic bacteria expressing interleukin-10 in Crohn’s disease. Clin. Gastroenterol. Hepatol. 2006, 4, 754–759. [Google Scholar]
- Loos, M.; Remaut, E.; Rottiers, P.; De Creus, A. Genetically engineered Lactococcus lactis secreting murine IL-10 modulates the functions of bone marrow-derived dendritic cells in the presence of LPS. Scand. J. Immunol. 2009, 69, 130–139. [Google Scholar] [PubMed]
- Munoz, P.; Bouza, E.; Cuenca-Estrella, M.; Eiros, J.M.; Perez, M.J.; Sanchez-Somolinos, M.; Rincon, C.; Hortal, J.; Pelaez, T. Saccharomyces cerevisiae fungemia: an emerging infectious disease. Clin. Infect. Dis. 2005, 40, 1625–1634. [Google Scholar] [CrossRef] [PubMed]
- Farina, C.; Arosio, M.; Mangia, M.; Moioli, F. Lactobacillus casei subsp. Rhamnosus sepsis in a patient with ulcerative colitis. J. Clin. Gastroenterol. 2001, 33, 251–252. [Google Scholar] [CrossRef] [PubMed]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mack, D.R. Probiotics in Inflammatory Bowel Diseases and Associated Conditions. Nutrients 2011, 3, 245-264. https://doi.org/10.3390/nu3020245
Mack DR. Probiotics in Inflammatory Bowel Diseases and Associated Conditions. Nutrients. 2011; 3(2):245-264. https://doi.org/10.3390/nu3020245
Chicago/Turabian StyleMack, David R. 2011. "Probiotics in Inflammatory Bowel Diseases and Associated Conditions" Nutrients 3, no. 2: 245-264. https://doi.org/10.3390/nu3020245
APA StyleMack, D. R. (2011). Probiotics in Inflammatory Bowel Diseases and Associated Conditions. Nutrients, 3(2), 245-264. https://doi.org/10.3390/nu3020245




