The Role of Phytonutrients in Skin Health
Abstract
:1. Introduction
2. UV Damage
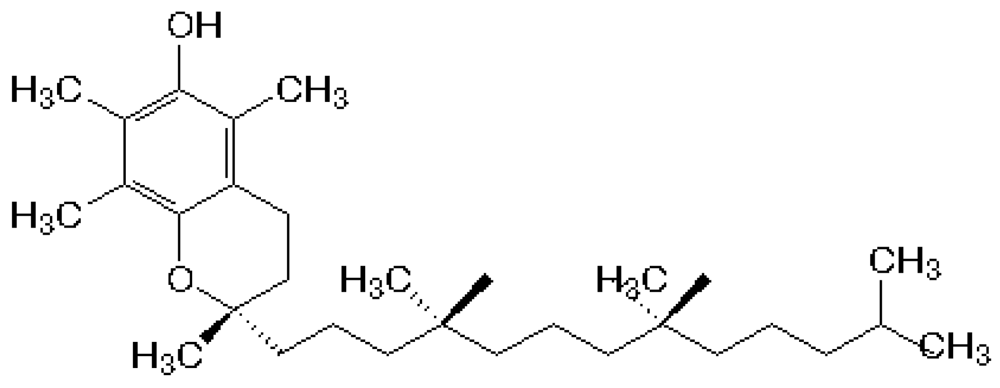
3. α-Tocopherol
| Skin Layer | Concentration | Reference |
|---|---|---|
| Epidermis | 31 ± 3.8 nmol /g tissue | Shindo et al. [20] |
| Dermis | 16.2 ± 1.1 nmol/g tissue | Shindo et al. [20] |
| Stratum corneum | 33 ± 4 nmol/g tissue | Thiele et al. [19] |
| Sebum | 76.5 ± 1.5 nmol/g sebum | Thiele et al. [21] |
3.1. Epidemiologic Studies
3.2. Clinical Trials
3.3. Animal Studies
3.4. In Vitro
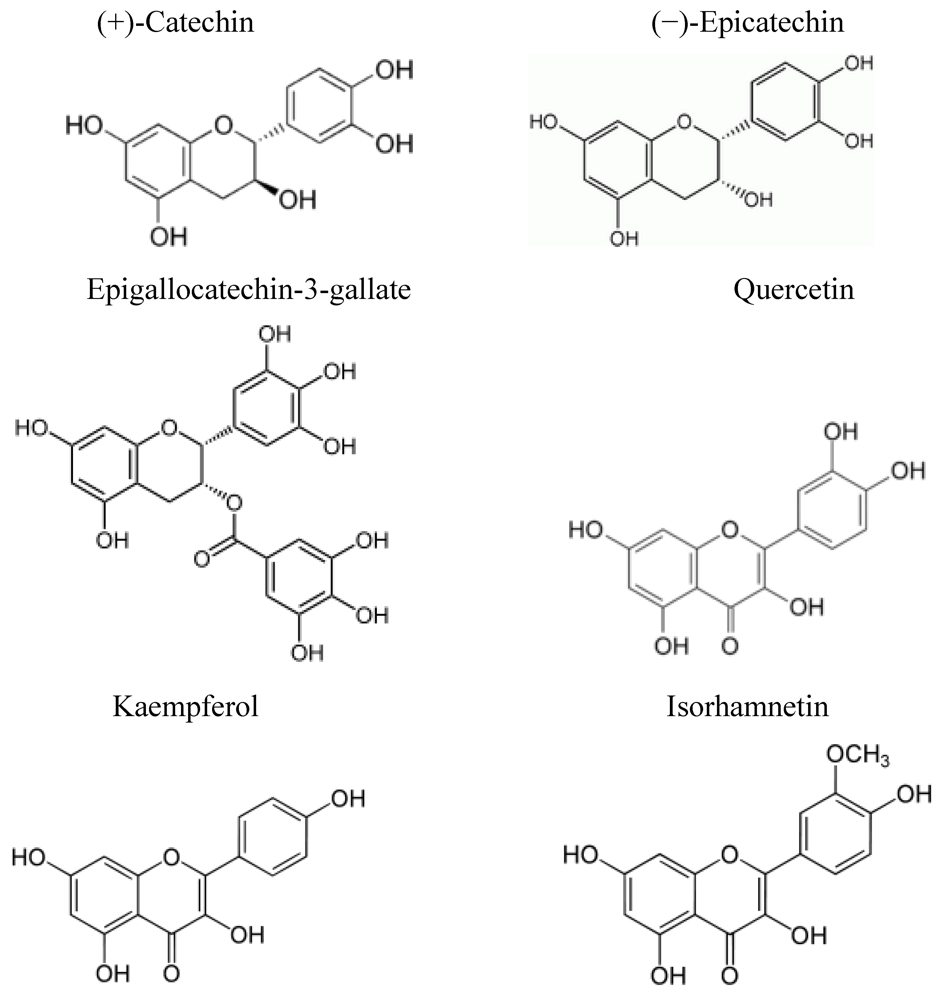
4. Flavonoids
4.1. Epidemiologic Studies
4.2. Clinical Trials
4.3. Animal Studies
4.4. In Vitro Studies
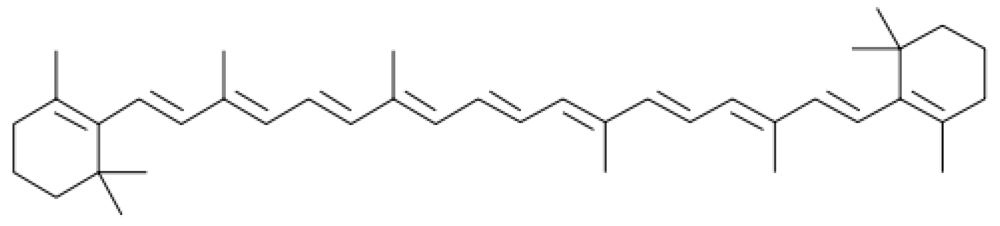
5. Carotenoids
5.1. Epidemiological Studies and Fruit & Vegetable Intakes
5.2. β-Carotene
5.2.1. Epidemiologic Studies
5.2.2. Clinical Trials
5.2.3. Animal Studies
5.2.4. In Vitro Studies
5.3. Lycopene
5.3.1. Epidemiologic Studies
5.3.2. Clinical Trials
5.3.3. In Vitro Studies
5.4. Lutein
5.4.1. Epidemiologic Studies
5.4.2. Clinical Trials
5.4.3. Animal Studies
5.4.4. In Vitro
6. Conclusions and Summary
Acknowledgements
References
- Lippens, S.; Hoste, E.; Vandenabeele, P.; Agostinis, P.; Declercq, W. Cell death in the skin. Apoptosis 2009, 14, 549–569. [Google Scholar]
- Boelsma, E.; Hendriks, H.F.; Roza, L. Nutritional skin care: health effects of micronutrients and fatty acids. Am. J. Clin. Nutr. 2001, 73, 853–864. [Google Scholar]
- Iddamalgoda, A.; Le, Q.T.; Ito, K.; Tanaka, K.; Kojima, H.; Kido, H. Mast cell tryptase and photoaging: possible involvement in the degradation of extra cellular matrix and basement membrane proteins. Arch. Dermatol. Res. 2008, 300, S69–S76. [Google Scholar]
- Mudgil, A.V.; Segal, N.; Andriani, F.; Wang, Y.; Fusenig, N.E.; Garlick, J.A. Ultraviolet B irradiation induces expansion of intraepithelial tumor cells in a tissue model of early cancer progression. J. Invest. Dermatol. 2003, 121, 191–197. [Google Scholar]
- Gloster, H.M., Jr.; Brodland, D.G. The epidemiology of skin cancer. Dermatol. Surg. 1996, 22, 217–226. [Google Scholar]
- Miller, A.J.; Mihm, M.C., Jr. Melanoma. N. Engl. J. Med. 2006, 355, 51–65. [Google Scholar]
- Greenlee, R.T.; Hill-Harmon, M.B.; Murray, T.; Thun, M. Cancer statistics, 2001. CA Cancer J. Clin. 2001, 51, 15–36. [Google Scholar]
- Healthy People 2010, Volume I. Office of Disease Prevention and Health Promotion. U.S. Department of Health and Human Services: Rockville, MD, USA, 2010. Available online: http://www.healthypeople.gov/Document/HTML/Volume1/03Cancer.htm (Accessed on 6 August 2010).
- Silverberg, E.; Boring, C.C.; Squires, T.S. Cancer statistics, 1990. CA. Cancer J. Clin. 1990, 40, 9–26. [Google Scholar]
- National Cancer Institute. SEER Program Cancer Facts and Figures.SEER Cancer Statistics Review 1973-1990; NIH publication no. 93-2789. NIH Publications: Washington, DC, USA, 1993.
- Boelsma, E.; Hendricks, H.F.J.; Roza, L. Nutritional skin care: health effects of micronutrients and fatty acids. Am. J. Clin. Nutr. 2001, 73, 853–864. [Google Scholar]
- Chiu, A.; Kimball, A.B. Topical vitamins, minerals and botanical ingredients as modulators of environmental and chronological skin damage. Br. J. Dermatol. 2003, 149, 681–691. [Google Scholar]
- Bruls, W.A.; Slaper, H.; van der Leun, J.C.; Berrens, L. Transmission of human epidermis and stratum corneum as a function of thickness in the ultraviolet and visible wavelengths. Photochem. Photobiol. 1984, 40, 485–494. [Google Scholar]
- McMillan, D.C.; Talwar, D.; Sattar, N.; Underwood, M.; O'Reilly, D.; McArdle, C. The relationship between reduced vitamin antioxidant concentrations and the systemic inflammatory response in patients with common solid tumours. Clin. Nutr. 2002, 21, 161–164. [Google Scholar]
- Touitou, E.; Godin, B. Skin nonpenetrating sunscreens for cosmetic and pharmaceutical formulations. Clin. Dermatol. 2008, 26, 375–379. [Google Scholar]
- Klotz, L.O.; Pellieux, C.; Briviba, K.; Pierlot, C.; Aubry, J.M.; Sies, H. Mitogen-activated protein kinase (p38-, JNK-, ERK-) activation pattern induced by extracellular and intracellular singlet oxygen and UVA. Eur. J. Biochem. 1999, 260, 917–922. [Google Scholar] [PubMed]
- Zingg, J.M. Modulation of signal transduction by vitamin E. Mol. Aspects Med. 2007, 28, 481–506. [Google Scholar]
- Thiele, J.J.; Ekanayake-Mudiyanselage, S. Vitamin E in human skin: organ-specific physiology and considerations for its use in dermatology. Mol. Aspects Med. 2007, 28, 646–667. [Google Scholar]
- USDA Agricultural Research Service. 1994–1996 Continuing survey of food intakes by individuals and diet and health knowledge survey. USDA: Washington, DC, USA, 1998.
- Rijnkels, J.M.; Moison, R.M.; Podda, E.; van Henegouwen, G.M. Photoprotection by antioxidants against UVB-radiation-induced damage in pig skin organ culture. Radiat. Res. 2003, 159, 210–217. [Google Scholar]
- Thiele, J.J.; Traber, M.G.; Packer, L. Depletion of human stratum corneum vitamin E: an early and sensitive in vivo marker of UV induced photo-oxidation. J. Invest. Dermatol. 1998, 110, 756–761. [Google Scholar] [CrossRef] [PubMed]
- McNaughton, S.A.; Marks, G.C.; Green, A.C. Role of dietary factors in the development of basal cell cancer and squamous cell cancer of the skin. Cancer Epidemiol. Biomarkers Prev. 2005, 14, 1596–1607. [Google Scholar]
- Heinen, M.M.; Hughes, M.C.; Ibiebele, T.I.; Marks, G.C.; Green, A.C.; van der Pols, J.C. Intake of antioxidant nutrients and the risk of skin cancer. Eur. J. Cancer 2007, 43, 2707–2716. [Google Scholar]
- van der Pols, J.C.; Heinen, M.M.; Hughes, M.C.; Ibiebele, T.I.; Marks, G.C.; Green, A.C. Serum antioxidants and skin cancer risk: an 8-year community-based follow-up study. Cancer Epidemiol. Biomarkers Prev. 2009, 18, 1167–1173. [Google Scholar]
- Werninghaus, K.; Meydani, M.; Bhawan, J.; Margolis, R.; Blumberg, J.B.; Gilchrest, B.A. Evaluation of the photoprotective effect of oral vitamin E supplementation. Arch. Dermatol. 1994, 130, 1257–1261. [Google Scholar]
- Fuchs, J.; Kern, H. Modulation of UV-light-induced skin inflammation by D-alpha-tocopherol and L-ascorbic acid: a clinical study using solar simulated radiation. Free Radic. Biol. Med. 1998, 25, 1006–1012. [Google Scholar]
- Eberlein-Konig, B.; Placzek, M.; Przybilla, B. Protective effect against sunburn of combined systemic ascorbic acid (vitamin C) and d-alpha-tocopherol (vitamin E). J. Am. Acad. Dermatol. 1998, 38, 45–48. [Google Scholar]
- Mireles-Rocha, H.; Galindo, I.; Huerta, M.; Trujillo-Hernandez, B.; Elizalde, A.; Cortes-Franco, R. UVB photoprotection with antioxidants: effects of oral therapy with d-alpha-tocopherol and ascorbic acid on the minimal erythema dose. Acta Derm. Venereol. 2002, 82, 21–24. [Google Scholar]
- Placzek, M.; Gaube, S.; Kerkmann, U.; Gilbertz, K.P.; Herzinger, T.; Haen, E.; Przybilla, B. Ultraviolet B-induced DNA damage in human epidermis is modified by the antioxidants ascorbic acid and D-alpha-tocopherol. J. Invest. Dermatol. 2005, 124, 304–307. [Google Scholar]
- Chung, J.H.; Seo, J.Y.; Lee, M.K.; Eun, H.C.; Lee, J.H.; Kang, S.; Fisher, G.J.; Voorhees, J.J. Ultraviolet modulation of human macrophage metalloelastase in human skin in vivo. J. Invest. Dermatol. 2002, 119, 507–512. [Google Scholar] [CrossRef] [PubMed]
- Dreher, F.; Maibach, H. Protective effects of topical antioxidants in humans. Curr. Prob. Dermatol. 2001, 29, 157–164. [Google Scholar]
- Montenegro, L.; Bonina, F.; Rigano, L.; Giogilli, S.; Sirigu, S. Protective effect evaluation of free radical scavengers on UVB induced human cutaneous erythema by skin reflectance spectrophotometry. Int. J. Cosmet. Sci. 1995, 17, 91–103. [Google Scholar]
- Gerrish, K.E.; Gensler, H.L. Prevention of photocarcinogenesis by dietary vitamin E. Nutr. Cancer 1993, 19, 125–133. [Google Scholar]
- Chong, E.W.T.; Wong, T.Y.; Kreis, A.J.; Simpson, J.A.; Guymer, R.H. Dietary antioxidants and primary prevention of age related macular degeneration: systematic review and meta-analysis. BMJ 2007, 335, 755. [Google Scholar]
- Gensler, H.L.; Magdaleno, M. Topical vitamin E inhibition of immunosuppression and tumorigenesis induced by ultraviolet irradiation. Nutr. Cancer 1991, 15, 97–106. [Google Scholar]
- Bissett, D.L.; Chatterjee, R.; Hannon, D.P. Photoprotective effect of superoxide-scavenging antioxidants against ultraviolet radiation-induced chronic skin damage in the hairless mouse. Photodermatol. Photoimmunol. Photomed. 1990, 7, 56–62. [Google Scholar]
- Bissett, D.L.; Chatterjee, R.; Hannon, D.P. Protective effect of a topically applied anti-oxidant plus an anti-inflammatory agent against ultraviolet radiation-induced chronic skin damage in the hairless mouse. J. Soc. Cosmet. Chem. 1992, 43, 85–92. [Google Scholar]
- Bissett, D.L.; Hillebrand, G.G.; Hannon, D.P. The hairless mouse as a model of skin photoaging: its use to evaluate photoprotective materials. Photodermatol 1989, 6, 228–233. [Google Scholar]
- Burke, K.E.; Clive, J.; Combs, G.F., Jr.; Commisso, J.; Keen, C.L.; Nakamura, R.M. Effects of topical and oral vitamin E on pigmentation and skin cancer induced by ultraviolet irradiation in Skh:2 hairless mice. Nutr. Cancer 2000, 38, 87–97. [Google Scholar]
- Chen, W.; Barthelman, M.; Martinez, J.; Alberts, D.; Gensler, H.L. Inhibition of cyclobutane pyrimidine dimer formation in epidermal p53 gene of UV-irradiated mice by alpha-tocopherol. Nutr. Cancer 1997, 29, 205–211. [Google Scholar]
- Gensler, H.L.; Aickin, M.; Peng, Y.M.; Xu, M. Importance of the form of topical vitamin E for prevention of photocarcinogenesis. Nutr. Cancer 1996, 26, 183–191. [Google Scholar]
- Beijersbergen van Henegouwen, G.M.; Junginger, H.E.; de Vries, H. Hydrolysis of RRR-alpha-tocopheryl acetate (vitamin E acetate) in the skin and its UV protecting activity (an in vivo study with the rat). J. Photochem. Photobiol. B 1995, 29, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Stoyanovsky, D.A.; Goldman, R.; Darrow, R.M.; Organisciak, D.T.; Kagan, V.E. Endogenous ascorbate regenerates vitamin E in the retina directly and in combination with exogenous dihydrolipoic acid. Curr. Eye Res. 1995, 14, 181–189. [Google Scholar]
- Evelson, P.; Ordonez, C.P.; Llesuy, S.; Boveris, A. Oxidative stress and in vivo chemiluminescence in mouse skin exposed to UVA radiation. J. Photochem. Photobiol. B 1997, 38, 215–219. [Google Scholar] [PubMed]
- Jurkiewicz, B.A.; Bissett, D.L.; Buettner, G.R. Effect of topically applied tocopherol on ultraviolet radiation-mediated free radical damage in skin. J. Invest. Dermatol. 1995, 104, 484–488. [Google Scholar]
- Khettab, N.; Amory, M.C.; Briand, G.; Bousquet, B.; Combre, A.; Forlot, P.; Barey, M. Photoprotective effect of vitamins A and E on polyamine and oxygenated free radical metabolism in hairless mouse epidermis. Biochimie 1988, 70, 1709–1713. [Google Scholar]
- Lin, J.Y.; Selim, M.A.; Shea, C.R.; Grichnik, J.M.; Omar, M.M.; Monteiro-Riviere, N.A.; Pinnell, S.R. UV photoprotection by combination topical antioxidants vitamin C and vitamin E. J. Am. Acad. Dermatol. 2003, 48, 866–874. [Google Scholar]
- Lopez-Torres, M.; Thiele, J.J.; Shindo, Y.; Han, D.; Packer, L. Topical application of alpha-tocopherol modulates the antioxidant network and diminishes ultraviolet-induced oxidative damage in murine skin. Br. J. Dermatol. 1998, 138, 207–215. [Google Scholar]
- McVean, M.; Liebler, D.C. Inhibition of UVB induced DNA photodamage in mouse epidermis by topically applied alpha-tocopherol. Carcinogenesis 1997, 18, 1617–1622. [Google Scholar]
- McVean, M.; Liebler, D.C. Prevention of DNA photodamage by vitamin E compounds and sunscreens: roles of ultraviolet absorbance and cellular uptake. Mol. Carcinog. 1999, 24, 169–176. [Google Scholar]
- Potapenko, A.; Abijev, G.A.; Pistsov, M.; Roshchupkin, D.I.; Vladimirov Yu, A.; Pliquett, F.; Ermolayev, A.V.; Sarycheva, I.K.; Evstigneeva, R.P. PUVA-induced erythema and changes in mechanoelectrical properties of skin. Inhibition by tocopherols. Arch. Dermatol. Res. 1984, 276, 12–16. [Google Scholar] [CrossRef] [PubMed]
- Record, I.R.; Dreosti, I.E.; Konstantinopoulos, M.; Buckley, R.A. The influence of topical and systemic vitamin E on ultraviolet light-induced skin damage in hairless mice. Nutr. Cancer 1991, 16, 219–225. [Google Scholar]
- Ritter, E.F.; Axelrod, M.; Minn, K.W.; Eades, E.; Rudner, A.M.; Serafin, D.; Klitzman, B. Modulation of ultraviolet light-induced epidermal damage: beneficial effects of tocopherol. Plast. Reconstr. Surg. 1997, 100, 973–980. [Google Scholar]
- Roshchupkin, D.I.; Pistsov, M.Y.; Potapenko, A.Y. Inhibition of ultraviolet light-induced erythema by antioxidants. Arch. Dermatol. Res. 1979, 266, 91–94. [Google Scholar]
- Schoonderwoerd, S.A.; Beijersbergen van Henegouwen, G.M.; Persons, K.C. Effect of alpha-tocopherol and di-butyl-hydroxytoluene (BHT) on UV-A-induced photobinding of 8-methoxypsoralen to Wistar rat epidermal biomacromolecules in vivo. Arch. Toxicol. 1991, 65, 490–494. [Google Scholar] [PubMed]
- Yuen, K.S.; Halliday, G.M. alpha-Tocopherol, an inhibitor of epidermal lipid peroxidation, prevents ultraviolet radiation from suppressing the skin immune system. Photochem. Photobiol. 1997, 65, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Mavon, A.; Raufast, V.; Redoules, D. Skin absorption and metabolism of a new vitamin E prodrug, delta-tocopherol-glucoside: in vitro evaluation in human skin models. J. Control. Release 2004, 100, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, S.; Katoh, E.M.; Tsuzuki, T.; Kobayashi, S. Protective effect of alpha-tocopherol-6-O-phosphate against ultraviolet B-induced damage in cultured mouse skin. J. Invest. Dermatol. 2003, 121, 406–411. [Google Scholar]
- Larsson, P.; Ollinger, K.; Rosdahl, I. Ultraviolet (UV)A- and UVB-induced redox alterations and activation of nuclear factor-kappaB in human melanocytes-protective effects of alpha-tocopherol. Br. J. Dermatol. 2006, 155, 292–300. [Google Scholar]
- Fahlman, B.M.; Krol, E.S. Inhibition of UVA and UVB radiation-induced lipid oxidation by quercetin. J. Agric. Food Chem. 2009, 57, 5301–5305. [Google Scholar]
- Kang, J.H.; Ascherio, A.; Grodstein, F. Fruit and vegetable consumption and cognitive decline in aging women. Ann. Neurol. 2005, 57, 713–720. [Google Scholar]
- Selmi, C.; Mao, T.K.; Keen, C.L.; Schmitz, H.H.; Eric Gershwin, M. The anti-inflammatory properties of cocoa flavanols. J. Cardiovasc. Pharmacol. 2006, 47, S163–S171, discussion S172-S176. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Blumberg, J.B. In vitro activity of almond skin polyphenols for scavenging free radicals and inducing quinone reductase. J. Agric. Food Chem. 2008, 56, 4427–4434. [Google Scholar] [PubMed]
- Li, N.; Jia, X.; Chen, C.Y.; Blumberg, J.B.; Song, Y.; Zhang, W.; Zhang, X.; Ma, G.; Chen, J. Almond consumption reduces oxidative DNA damage and lipid peroxidation in male smokers. J. Nutr. 2007, 137, 2717–2722. [Google Scholar]
- Chen, C.Y.; Milbury, P.E.; Lapsley, K.; Blumberg, J.B. Flavonoids from almond skins are bioavailable and act synergistically with vitamins C and E to enhance hamster and human LDL resistance to oxidation. J. Nutr. 2000, 135, 1366–1373. [Google Scholar]
- Suganuma, M.; Okabe, S.; Oniyama, M.; Tada, Y.; Ito, H.; Fujiki, H. Wide distribution of [3H](-)-epigallocatechin gallate, a cancer preventive tea polyphenol, in mouse tissue. Carcinogenesis 1998, 19, 1771–1776. [Google Scholar] [CrossRef] [PubMed]
- Tardif, J.C.; Cote, B.; Lesperance, J.; Bourassa, M.; Lambert, J.; Doucet, S.; BIolodeau, L.; Nattel, S.; deGuise, P. Propucol and multivitamins in the prevention of restenosis after coronary angioplasty. N. Eng. J. Med. 1997, 337, 365–372. [Google Scholar]
- Fortes, C.; Mastroeni, S.; Melchi, F.; Pilla, M.A.; Antonelli, G.; Camaioni, D.; Alotto, M.; Pasquini, P. A protective effect of the Mediterranean diet for cutaneous melanoma. Int. J. Epidemiol. 2008, 37, 1018–1029. [Google Scholar]
- Purba, M.B.; Kouris-Blazos, A.; Wattanapenpaiboon, N.; Lukito, W.; Rothenberg, E.M.; Steen, B.C.; Wahlqvist, M.L. Skin wrinkling: can food make a difference? J. Am. Coll. Nutr. 2001, 20, 71–80. [Google Scholar] [PubMed]
- Neukam, K.; Stahl, W.; Tronnier, H.; Sies, H.; Heinrich, U. Consumption of flavanol-rich cocoa acutely increases microcirculation in human skin. Eur. J. Nutr. 2007, 46, 53–56. [Google Scholar]
- Greul, A.K.; Grundmann, J.U.; Heinrich, F.; Pfitzner, I.; Bernhardt, J.; Ambach, A.; Bielsalski, H.K.; Gollnick, H. Photoprotection of UV-irradiated human skin: an antioxidative combination of vitamins E and C, carotenoids, selenium and proanthocyanidins. Skin Pharmacol. Appl. Skin Physiol. 2002, 15, 307–315. [Google Scholar] [PubMed]
- Chiu, A.E.; Chan, J.L.; Kern, D.G.; Kohler, S.; Rehmus, W.E.; Kimball, A.B. Double-blinded, placebo-controlled trial of green tea extracts in the clinical and histologic appearance of photoaging skin. Dermatol. Surg. 2005, 31, 855–860. [Google Scholar]
- Camouse, M.M.; Domingo, D.S.; Swain, F.R.; Conrad, E.P.; Matsui, M.S.; Maes, D.; Declercq, L.; Cooper, K.D.; Stevens, S.R.; Baron, E.D. Topical application of green and white tea extracts provides protection from solar-simulated ultraviolet light in human skin. Exp. Dermatol. 2009, 18, 522–526. [Google Scholar]
- Yusef, N.; Irby, C.; Katiyar, S.K.; Elmets, C.A. Photoprotective effects of green tea polyphenol. Photochem. Photoimmunol. Photomed. 2007, 23, 48–56. [Google Scholar]
- Mnich, C.D.; Hoek, K.S.; Virkki, L.V.; Farkas, A.; Dudli, C.; Laine, E.; Urosevic, M.; Dummer, R. Green tea extract reduces induction of p53 and apoptosis in UVB-irradiated human skin independent of transcriptional controls. Exp. Dermatol. 2009, 18, 69–77. [Google Scholar]
- Aquino, R.; Morelli, S.; Tomaino, A.; Pellegrino, M.; Saija, A.; Grumetto, L.; Puglia, C.; Ventura, D.; Bonina, F. Antioxidant and photoprotective activity of a crude extract of Culcitium reflexum H.B.K. leaves and their major flavonoids. J. Ethnopharmacol. 2002, 79, 183–191. [Google Scholar] [PubMed]
- Record, I.R.; Dreosti, I.E. Protection by black tea and green tea against UVB and UVA + B induced skin cancer in hairless mice. Mutat. Res. 1998, 422, 191–199. [Google Scholar]
- Wang, Z.Y.; Huang, M.T.; Ho, C.T.; Chang, R.; Ma, W.; Ferraro, T.; Reuhl, K.R.; Yang, C.S.; Conney, A.H. Inhibitory effect of green tea on the growth of established skin papillomas in mice. Cancer Res. 1992, 52, 6657–6665. [Google Scholar]
- Gensler, H.L.; Timmermann, B.N.; Valcic, S.; Wachter, G.A.; Dorr, R.; Dvorakova, K.; Alberts, D.S. Prevention of photocarcinogenesis by topical administration of pure epigallocatechin gallate isolated from green tea. Nutr. Cancer 1996, 26, 325–335. [Google Scholar]
- Franceschi, C.; Carpri, M.; Monti, D.; Giunta, S.; Oliverieri, F.; Sevini, F.; Panourgia, M.; Invidia, L.; Celani, L.; Scuriti, M.; Cevenini, E.; Castellani, G.C.; Salvioli, S. Inflammaging and anti-inflammaging: a systemic perspective on aging and longevity emerged from studies in humans. Mech. Age. Dev. 2007, 128, 92–105. [Google Scholar] [CrossRef]
- Kahraman, A.; Inal, M.E. Protective effects of quercetin on ultraviolet A light-induced oxidative stress in the blood of rat. J. Appl. Toxicol. 2002, 22, 303–309. [Google Scholar]
- Casagrande, R.; Georgetti, S.R.; Verri, W.A., Jr.; Dorta, D.J.; dos Santos, A.C.; Fonseca, M.J. Protective effect of topical formulations containing quercetin against UVB-induced oxidative stress in hairless mice. J. Photochem. Photobiol. B 2006, 84, 21–27. [Google Scholar]
- Vayalil, P.K.; Mittal, A.; Hara, Y.; Elmets, C.A.; Katiyar, S.K. Green tea polyphenols prevent ultraviolet light-induced oxidative damage and matrix metalloproteinases expression in mouse skin. J. Invest. Dermatol. 2004, 122, 1480–1487. [Google Scholar]
- Garbisa, S.; Sartor, L.; Biggin, S.; Salvato, B.; Benelli, R.; Albini, A. Tumor gelatinases and invasion inhibited by the green tea flavanol epigallocatechin-3-gallate. Cancer 2001, 91, 822–832. [Google Scholar]
- Morley, N.; Clifford, T.; Salter, L.; Campbell, S.; Gould, D.; Curnow, A. The green tea polyphenol (-)-epigallocatechin gallate and green tea can protect human cellular DNA from ultraviolet and visible radiation-induced damage. Photodermatol. Photoimmunol. Photomed. 2005, 21, 15–22. [Google Scholar]
- Gasser, P.; Lati, E.; Peno-Mazzarino, L.; Bouzoud, D.; Allegaert, L.; Bernaert, H. Cocoa polyphenols and their influence on parameters involved in ex vivo skin restructuring. Int. J. Cosmet. Sci. 2008, 30, 339–345. [Google Scholar] [CrossRef] [PubMed]
- van Leeuwen, R.; Boekhoorn, S.; Vingerling, J.R.; Witteman, J.C.M.; Klaver, C.C.W.; Hofman, A.; de Jong, P.T.V.M. Dietary intake of antioxidants and risk of age-related macular degeneration. JAMA 2005, 294, 3101–3107. [Google Scholar]
- Krinsky, N.I.; Johnson, E.J. Carotenoid actions and their relation to health and disease. Mol. Aspects Med. 2005, 26, 459–516. [Google Scholar]
- USDA USDA-NCC. Carotenoid Database for U.S. Foods-1998. USDA: Washington, DC, USA. 2010. Available online: http://www.nal.usda.gov/fnic/foodcomp/Data/car98/car98.html (Accessed on 6 August 2010).
- Stahl, W.; Sies, H. Antioxidant defense: Vitamins E and C and carotenoids. Diabetes 1997, 46, S14–S18. [Google Scholar]
- Cantrell, A.; McGarvey, D.J.; Truscott, T.G.; Rancan, F.; Bohm, F. Singlet oxygen quenching by dietary carotenoids in a model membrane environment. Arch. Biochem. Biophys. 2003, 412, 47–54. [Google Scholar]
- Wertz, K.; Hunziker, P.B.; Seifert, N.; Riss, G.; Neeb, M.; Steiner, G.; Hunziker, W.; Goralczyk, R. beta-Carotene interferes with ultraviolet light A-induced gene expression by multiple pathways. J. Invest. Dermatol. 2005, 124, 428–434. [Google Scholar]
- Peng, Y.-M.; Peng, Y.-S.; Lin, Y. A nonspaonificaton method for the determination of carotenoids, retinoids, and tocopherols in solid human tissues. Cancer Epidemiol. Biomarkers Prev. 1993, 2, 139–144. [Google Scholar] [PubMed]
- Darvin, M.; Gersonde, I.; Meinke, M.; Sterry, W.; Lademann, J. Non-invasive in vivo determination of the carotenoids beta-carotene and lycopene concentrations in the human skin using the Raman spectroscopic method. J. Phys. D: Appl. Phys. 2005, 38, 2696–2700. [Google Scholar] [CrossRef]
- Hata, T.R.; Scholz, T.A.; Ermakov, I.V.; McClane, R.W.; Khachik, F.; Gellerman, W.; Pershing, L.K. Non-invasive Raman spectroscopic detection of carotenoids in human skin. J. Invest. Dermatol. 2000, 115, 441–448. [Google Scholar]
- Richelle, M.; Sabatier, M.; Steiling, H.; Williamson, G. Skin bioavailability of dietary vitamin E, carotenoids, polyphenols, bitamin C, zinc and selenium. Br. J. Nutr. 2006, 96, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Alaluf, S.; Heinrich, U.; Stahl, W.; Tronnier, H.; Wiseman, S. Dietary carotenoids contribute to normal human skin color and UV photosensitivity. J. Nutr. 2002, 132, 399–403. [Google Scholar]
- White, W.S.; Kim, C.I.; Kalkwarf, H.J.; Bustos, P.; Roe, D.A. Ultraviolet light-induced reductions in plasma carotenoid levels. Am. J. Clin. Nutr. 1988, 47, 879–883. [Google Scholar]
- Ribaya-Mercado, J.D.; Garmyn, M.; Gilchrest, B.A.; Russell, R.M. Skin lycopene is destroyed preferentially over beta-carotene during ultraviolet irradiation in humans. J. Nutr. 1995, 125, 1854–1859. [Google Scholar]
- Chung, H.-Y.; Rasmussen, H.M.; Johnson, E.J. Lutein bioavailability is higher from lutein-enriched eggs than from supplements and spinach in men. J. Nutr. 2004, 134, 1887–1893. [Google Scholar]
- Jacques, P.F.; Sulsky, S.I.; Sadowski, J.A.; Philips, J.C.; Rush, D.; Willett, W.C. Comparison of micronutrient intake measured by a dietary questionnaire and biochemical indicators of micronutrient status. Am. J. Clin. Nutr. 1993, 57, 182–189. [Google Scholar]
- Ibiebele, T.I.; van der Pols, J.C.; Hughes, M.C.; Marks, G.C.; Williams, G.M.; Green, A.C. Dietary pattern in association with squamous cell carcinoma of the skin: a prospective study. Am. J. Clin. Nutr. 2007, 85, 1401–1408. [Google Scholar]
- Dorgan, J.F.; Boakye, N.A.; Fears, T.R.; Schleicher, R.L.; Helsel, W.; Anderson, C.; Robinson, J.; Guin, J.D.; Lessin, S.; Ratnasinghe, L.D.; Tangrea, J.A. Serum carotenoids and alpha-tocopherol and risk of nonmelanoma skin cancer. Cancer Epidemiol. Biomarkers Prev. 2004, 13, 1276–1282. [Google Scholar]
- Fung, T.T.; Hunter, D.J.; Spiegelman, D.; Colditz, G.A.; Speizer, F.E.; Willett, W.C. Vitamins and carotenoids intake and the risk of basal cell carcinoma of the skin in women (United States). Cancer Cause. Control 2002, 13, 221–230. [Google Scholar]
- Fung, T.T.; Spiegelman, D.; Egan, K.M.; Giovannucci, E.; Hunter, D.J.; Willett, W.C. Vitamin and carotenoid intake and risk of squamous cell carcinoma of the skin. Int. J. Cancer 2003, 103, 110–115. [Google Scholar]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free. Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar]
- Hunter, D.J.; Colditz, G.A.; Stampfer, M.J.; Rosner, B.; Willett, W.C.; Speizer, F.E. Diet and risk of basal cell carcinoma of the skin in a prospective cohort of women. Ann. Epidemiol. 1992, 2, 231–239. [Google Scholar]
- Sahl, W.J.; Glore, S.; Garrison, P.; Oakleaf, K.; Johnson, S.D. Basal cell carcinoma and lifestyle characteristics. Int. J. Dermatol. 1995, 34, 398–402. [Google Scholar]
- van Dam, R.M.; Huang, Z.; Giovannucci, E.; Rimm, E.B.; Hunter, D.J.; Colditz, G.A.; Stampfer, M.J.; Willett, W.C. Diet and basal cell carcinoma of the skin in a prospective cohort of men. Am. J. Clin. Nutr. 2000, 71, 135–141. [Google Scholar]
- Breslow, R.A.; Alberg, A.J.; Helzlsouer, K.J.; Bush, T.L.; Norkus, E.P.; Morris, J.S.; Spate, V.E.; Comstock, G.W. Serological precursors of cancer: malignant melanoma, basal and squamous cell skin cancer, and prediagnostic levels of retinol, beta- carotene, lycopene, alpha-tocopherol, and selenium. Cancer Epidemiol. Biomarkers Prev. 1995, 4, 837–842. [Google Scholar] [PubMed]
- Davies, T.W.; Treasure, F.P.; Welch, A.A.; Day, N.E. Diet and basal cell skin cancer: results from the EPIC-Norfolk cohort. Br. J. Dermatol. 2002, 146, 1017–1022. [Google Scholar]
- Karagas, M.R.; Greenberg, E.R.; Nierenberg, D.; Stukel, T.A.; Morris, J.S.; Stevens, M.M.; Baron, J.A. Risk of squamous cell carcinoma of the skin in relation to plasma selenium, alpha-tocopherol, beta-carotene, and retinol: a nested case-control study. Cancer Epidemiol. Biomarkers Prev. 1997, 6, 25–29. [Google Scholar] [PubMed]
- Comstock, G.W.; Helzlsouer, K.J.; Bush, T.L. Prediagnostic serum levels of carotenoids and vitamin E as related to subsequent cancer in Washington County, Maryland. Am. J. Clin. Nutr. 1991, 53, 260S–264S. [Google Scholar]
- Dorgan, J.F.; Boakye, N.A.; Fears, T.R.; Schleicher, R.L.; Helsel, W.; Anderson, C.; Robinson, J.; Guin, J.D.; Lessin, S.; Ratnasinghe, L.D.; Tangrea, J.A. Serum carotenoids and alpha-tocopherol and risk of nonmelanoma skin cancer. Cancer Epidemiol. Biomarkers Prev. 2004, 13, 1276–1282. [Google Scholar]
- Fung, T.T.; Spiegelman, D.; Egan, K.M.; Giovannucci, E.; Hunter, D.J.; Willett, W.C. Vitamin and carotenoid intake and risk of squamous cell carcinoma of the skin. Int. J. Cancer 2003, 103, 110–115. [Google Scholar]
- Heinen, M.M.; Hughes, M.C.; Ibiebele, T.I.; Marks, G.C.; BGreen, A.C.; van der Pols, J.C. Intake of antioxidants nutrients and th erisk of skin cancer. Eur. J. Cancer 2007, 43, 2707–2716. [Google Scholar] [CrossRef] [PubMed]
- Mathews-Roth, M.M.; Pathak, M.A.; Parrish, J.; Fitzpatrick, T.B.; Kass, E.H.; Toda, K.; Clemens, W. A clinical trial of the effects of oral beta-carotene on the responses of human skin to solar radiation. J. Invest. Dermatol. 1972, 59, 349–353. [Google Scholar]
- Gollnick, H.P.M.; Hopfenmuller, W.; Hemmes, C.; Chun, S.C.; Schmid, C.; Sundermeier, K.; Biesalski, H.K. Systemic beta-carotene plus topical UV-sunscreen are an optimal protection against harmful effects of natural UV-sunlight: results of the Berlin-Eilath study. Eur. J. Dermatol. 1996, 6, 200–205. [Google Scholar]
- Lee, J.; Jiang, S.; Levien, N.; Watson, R.R. Carotenoid supplementation reduces erythema in human skin after simulated solar radiation exposure. Photochem. Photobiol. 2000, 70, 243–247. [Google Scholar]
- Fuller, C.J.; Faulkner, H.; Bendich, A.; Parker, R.S.; Roe, D.A. Effect of β-carotene supplementation on photosupression of delayed-type hypersensitivity in normal young men. Arch. Intern. Med. 1992, 159, 748–754. [Google Scholar]
- Herraiz, L.A.; Hsieh, W.C.; Parker, R.S.; Swanson, J.E.; Bendich, A.; Roe, D.A. Effect of UV and beta-carotene supplementation on delayed-type hypersensitivity response in healthy older men. J. Am. Coll. Nutr. 1998, 17, 617–624. [Google Scholar]
- Heinrich, U.; Gartner, C.; Wiebusch, M.; Eichler, O.; Sies, H.; Tronnier, H.; Stahl, W. Supplementation with beta-carotene or a similar amount of mixed carotenoids protects humans from UV-induced erythema. J. Nutr. 2003, 133, 98–101. [Google Scholar]
- Kopcke, W.; Krutmann, J. Protection from sunburn with beta-Carotene—a meta-analysis. Photochem. Photobiol. 2008, 84, 284–288. [Google Scholar]
- Albanes, D.; Virtamo, J.; Rautalahti, M.; Haukka, J.; Palmgren, J.; Gref, C.G.; Heinonen, O.P. Serum beta-carotene before and after beta-carotene supplementation. Eur. J.Clin. Nutr. 1992, 46, 15–24. [Google Scholar]
- Omenn, G.S.; Goodman, G.E.; Thornquist, M.D.; Balmes, J.; Cullen, M.R.; Glass, A.; Keogh, J.P.; Meyskens, F.L.; Valais, B.; Williams, J.H.; Barnhardt, S.; Cherniak, M.G.; Brodkin, C.A.; Hammar, S. Risk factors for lung cancer and for intervention effects in CARET, the beta-carotene and retinol efficiency trial. J. Natl. Cancer Inst. 1996, 88, 1550–1559. [Google Scholar]
- Minami, Y.; Kawabata, K.; Kubo, Y.; Arase, S.; Hirasaka, K.; Nikawa, T.; Bando, N.; Kawai, Y.; Terao, J. Peroxidized cholesterol-induced matrix metalloproteinase-9 activation and its suppression by dietary beta-carotene in photoaging of hairless mouse skin. J Nutr. Biochem. 2009, 20, 389–398. [Google Scholar]
- Camera, E.; Mastrofrancesco, A.; Fabbri, C.; Daubrawa, F.; Picardo, M.; Sies, H.; Stahl, W. Astaxanthin, canthaxanthin and beta-carotene differently affect UVA-induced oxidative damage and expression of oxidative stress-responsive enzymes. Exp. Dermatol. 2009, 18, 222–231. [Google Scholar]
- Mein, J.R.; Lian, F.; Wang, X.-D. Biological activity of lycopene metabolites: implications for cancer prevention. Nutr. Rev. 2008, 66, 667–683. [Google Scholar]
- Di Mascio, P.; Kaiser, S.; Sies, H. Lycopene as the most efficient biological carotenoid singlet oxygen quencher. Arch. Biochem. Biophys. 1989, 274, 532–538. [Google Scholar]
- Darvin, M.; Patzelt, A.; Gehse, S.; Schanzer, S.; Benderoth, C.; Sterry, W.; Lademann, J. Cutaneous concentration of lycopene correlates significantly with the roughness of the skin. Eur. J. Pharm. Biopharm. 2008, 69, 943–947. [Google Scholar]
- Lopes, L.B.; VanDeWall, H.; Li, H.T.; Venugopal, V.; Li, H.K.; Naydin, S.; Hosmer, J.; Levendusky, M.; Zheng, H.; Bentley, M.V.L.B.; Levin, R.; Hass, M.A. Topical delivery of lycopene using microemulsions: enhanced skin penetration and tissue antioxidant activity. J. Pharm. Sci. 2010, 99, 1346–1357. [Google Scholar]
- Stahl, W.; Heinrich, U.; Wiseman, S.; Eichler, O.; Sies, H.; Tronnier, H. Dietary tomato paste protects against ultraviolet light-induced erythema in humans. J. Nutr. 2001, 131, 1449–1451. [Google Scholar]
- Aust, O.; Stahl, W.; Sies, H.; Tronnier, H.; Heinrich, U. Supplementation with tomato-based products increases lycopene, phytofluene, and phytoene levels in human serum and protects against UV-light-induced erythema. Int. J. Vitam. Nutr. Res. 2005, 75, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Wertz, K.; Siler, U.; Goralczyk, R. Lycopene: modes of action to promote prostate health. Arch. Biochem. Biophys. 2004, 430, 127–134. [Google Scholar]
- Basu, A.; Imrhan, V. Tomatoes versus lycopene in oxidative stress and carcinogenesis: conclusions from clinical trials. Eur. J. Clin. Nutr. 2007, 61, 295–303. [Google Scholar]
- Wu, W.B.; Chiang, H.S.; Fang, J.Y.; Hung, C.F. Inhibitory effect of lycopene on PDGF-BB-induced signalling and migration in human dermal fibroblasts: a possible target for cancer. Biochem. Soc. Trans. 2007, 35, 1377–1378. [Google Scholar]
- Handelman, G.J.; Nightingale, Z.D.; Lichtenstein, A.H.; Schaefer, E.J.; Blumberg, J.B. Lutein and zeaxanthin concentrations in plasma after dietary supplementation with egg yolk. Am. J. Clin. Nutr. 1999, 70, 247–251. [Google Scholar]
- Mangels, A.R.; Holden, J.M.; Beecher, G.R.; Forman, M.R.; Lanza, E. Carotenoid content of fruits and vegetables: an evaluation of analytic data. J. Am. Diet. Assoc. 1993, 93, 284–296. [Google Scholar]
- Surai, P.F.; MacPherson, A.; Speake, B.K.; Sparks, N.H. Designer egg evaluation in a controlled trial. Eur. J. Clin. Nutr. 2000, 54, 298–305. [Google Scholar]
- Brown, L.; Rimm, E.B.; Seddon, J.M.; Giovanucci, E.L.; Chasen-Taber, L.; Speigelman, D.; Willett, W.C.; Hankinson, S.E. A prospective study of carotenoid intake and risk of cataract extraction in US men. Am. J. Clin. Nutr. 1999, 70, 517–524. [Google Scholar]
- Chasan-Taber, L.; Willett, W.C.; Seddon, J.M.; Stamper, M.J.; Rosner, B.; Colditz, G.A. A prospective study on vitamin supplement intake and cataract extraction among US women. Epidemiology 1999, 10, 679–684. [Google Scholar]
- Seddon, J.M.; Ajani, U.A.; Sperduto, R.D.; Hiller, R.; Blair, N.; Burton, T.C.; Farber, M.D.; Gragoudas, E.S.; Haller, J.; Miller, D.T.; et al. Dietary carotenoids, vitamins A, C, and E, and advanced age-related macular degeneration. Eye Disease Case-Control Study Group. JAMA 1994, 272, 1413–1420. [Google Scholar] [PubMed]
- Mohammedshah, F.D.J.S.; Amann, M.M.; Heimbach, J.M. Dietary intakes of lutein and zeaxanthin and total carotenoids among Americans age 50 and above. FASEB J. 1999, 13, A554. [Google Scholar]
- Nebeling, L.C.; Forman, M.R.; Graubard, B.I.; Snyder, R.A. Changes in carotenoid intake in the United States: the 1987 and 1992 National Health Interview Surveys. J. Am. Diet. Assoc. 1997, 97, 991–996. [Google Scholar]
- Tucker, K.L.; Chen, H.; Wilson, P.W.F.; Schaefer, E.J.; Lammi-Keefe, C.J. Carotenoid intakes, assessed by dietary questionnaire, are associated with plasma carotenoid concentrations in an elderly population. J. Nutr. 1999, 129, 438–445. [Google Scholar] [PubMed]
- Vandenlangenberg, G.M.; Brady, W.E.; Nebeling, L.C.; Block, G.; Forman, M.; Bowen, P.E.; Stacewicz-Sapuntzakis, M.; Mares-Perlman, J.A. Influence of using different sources of carotenoid data in epidemiologic studies. J. Am. Diet. Assoc. 1996, 96, 1271–1275. [Google Scholar]
- Hammond, B.R., Jr.; Johnson, E.J.; Russell, R.M.; Krinsky, N.I.; Yeum, K.J.; Edwards, R.B.; Snodderly, D.M. Dietary modification of human macular pigment density. Invest. Ophthalmol. Vis. Sci. 1997, 38, 1795–1801. [Google Scholar]
- Johnson, E.J.; Chung, H.-Y.; Caldarella, S.M.; Snodderly, D.M. The influence of supplemental lutein and docosahexaenoic acid on serum, lipoproteins, and macular pigmentation. Am. J. Clin. Nutr. 2008, 87, 1521–1529. [Google Scholar] [PubMed]
- Johnson, E.J.; Hammond, R.B.; Yeum, K.-J.; Qin, J.; Wang, X.-D.; Castaneda, C.; Snodderly, D.M.; Russell, R.M. Relation among serum and tissue concentrations of lutein and zeaxanthin and macular pigment density. Am. J. Clin. Nutr. 2000, 71, 1555–1562. [Google Scholar]
- Palombo, P.; Fabrizi, G.; Ruocco, V.; Ruocco, E.; Fluhr, J.; Roberts, R.; Morganti, P. Beneficial long-term effects of combined oral/topical antioxidant treatment with the carotenoids lutein and zeaxanthin on human skin: a double-blind, placebo-control study. Skin Pharmacol. Physiol. 2007, 20, 199–210. [Google Scholar]
- Astner, S.; Wu, A.; Chen, J.; Philips, N.; Rius-Diaz, F.; Parrado, C.; Mihm, M.C.; Goukassian, D.A.; Pathak, M.A.; Gonzalez, S. Dietary lutein/zeaxanthin partially reduces photoaging and photocarcinogenesis in chronically UVB-irradiated Skh-1 hairless mice. Skin Pharmacol. Physiol. 2007, 20, 283–291. [Google Scholar]
- O'Connor, I.; O'Brien, N. Modulation of UVA light-induced oxidative stress by beta-carotene, lutein and astaxanthin in cultured fibroblasts. J. Dermatol. Sci. 1998, 16, 226–230. [Google Scholar]
- Chitchumroonchokchai, C.; Bomser, J.A.; Glamm, J.E.; Failla, M.L. Xanthophylls and alpha-tocopherol decrease UVB-induced lipid peroxidation and stress signaling in human lens epithelial cells. J. Nutr. 2004, 134, 3225–3232. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Evans, J.A.; Johnson, E.J. The Role of Phytonutrients in Skin Health. Nutrients 2010, 2, 903-928. https://doi.org/10.3390/nu2080903
Evans JA, Johnson EJ. The Role of Phytonutrients in Skin Health. Nutrients. 2010; 2(8):903-928. https://doi.org/10.3390/nu2080903
Chicago/Turabian StyleEvans, Julie A., and Elizabeth J. Johnson. 2010. "The Role of Phytonutrients in Skin Health" Nutrients 2, no. 8: 903-928. https://doi.org/10.3390/nu2080903
APA StyleEvans, J. A., & Johnson, E. J. (2010). The Role of Phytonutrients in Skin Health. Nutrients, 2(8), 903-928. https://doi.org/10.3390/nu2080903




