How Important Are Dietary Habits Compared to Other Factors for Sleep Quality?—An Analysis Using Data from a Specific Region in Japan
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Procedure and Participants
2.2. Measures
2.2.1. Proxies for Sleep Quality
2.2.2. Key Focused Factors Influencing Sleep Quality
- Stress: In the sleep diary employed in the present study, six stress-related items were included in the questionnaires administered immediately before bedtime and upon awakening: “I feel relaxed,” “I feel irritated or angry,” “I feel motivated to do things,” “I can concentrate,” “I feel anxious or worried,” and “I feel depressed.” Participants evaluated each item using a four-point scale. Although the Japanese phrasing of these items differs slightly, their content corresponds closely to several stress response items from the Japanese Occupational Stress Questionnaire [63,64].
- Bedtime conditions: As variables related to bedtime conditions, the analysis utilized items from the post-awakening section of the sleep diary, including bedtime, time in bed, and the presence or absence of caffeine intake, alcohol consumption, and ICT device use before bedtime. Bedtime was converted from the 24 h format recorded in the diary into hourly values relative to midnight (24:00) for analytical purposes.
- Weather conditions: Weather data were obtained from the historical daily records for Ebetsu City published on the Japan Meteorological Agency’s website [65]. The analysis incorporated daily values for precipitation, average temperature, diurnal temperature range (maximum temperature minus minimum temperature), average wind speed, and sunshine duration on each day of going to bed. Humidity and atmospheric pressure were not included in the published daily data.
- Physical characteristics: The variables related to physical condition included age, body mass index (BMI), and systolic blood pressure. While BMI and systolic blood pressure were measured during both the summer and winter observation periods, age was recorded at only a single time point. Nonetheless, as the observation period for all study participants was less than one year, any change in age during the study would be limited to a maximum of one year.
- Exercise habits: Regarding exercise habits, self-reported data on the monthly amount of physical activity for each type of exercise were available at two time points: summer and winter. The types of exercise were categorized into four groups: (i) light walking such as strolling, (ii) brisk walking such as walking for exercise, (iii) light to moderate activities such as golf or gardening, and (iv) vigorous activities such as tennis or jogging. The questionnaire asked participants to report the frequency per month and the duration per session for each type of exercise, from which the monthly amount of activity was calculated. For all types of exercise, the distribution of monthly activity amounts was right-skewed with a median of 0 h/month. Therefore, for each exercise type, a dummy variable was created, taking the value of 1 if the activity amount was greater than zero, and was used in the analysis.
- Dietary habits: Dietary habit variables were derived from responses to a FFQ administered during the winter phase of the observational study. Since the FFQ inquired about dietary habits over the past year, the responses can also be considered reflective of dietary patterns at the time of the summer sleep diary recordings. Based on the reported intake frequency and portion size for approximately 130 food items, the average daily intake was calculated for each of 12 food group categories (cereals, potatoes, beans, green and yellow vegetables, other vegetables, fruits, mushrooms, seaweeds, seafood, meat, eggs, dairy products). These values were then standardized per 1000 kcal of daily energy intake and used in the analysis.
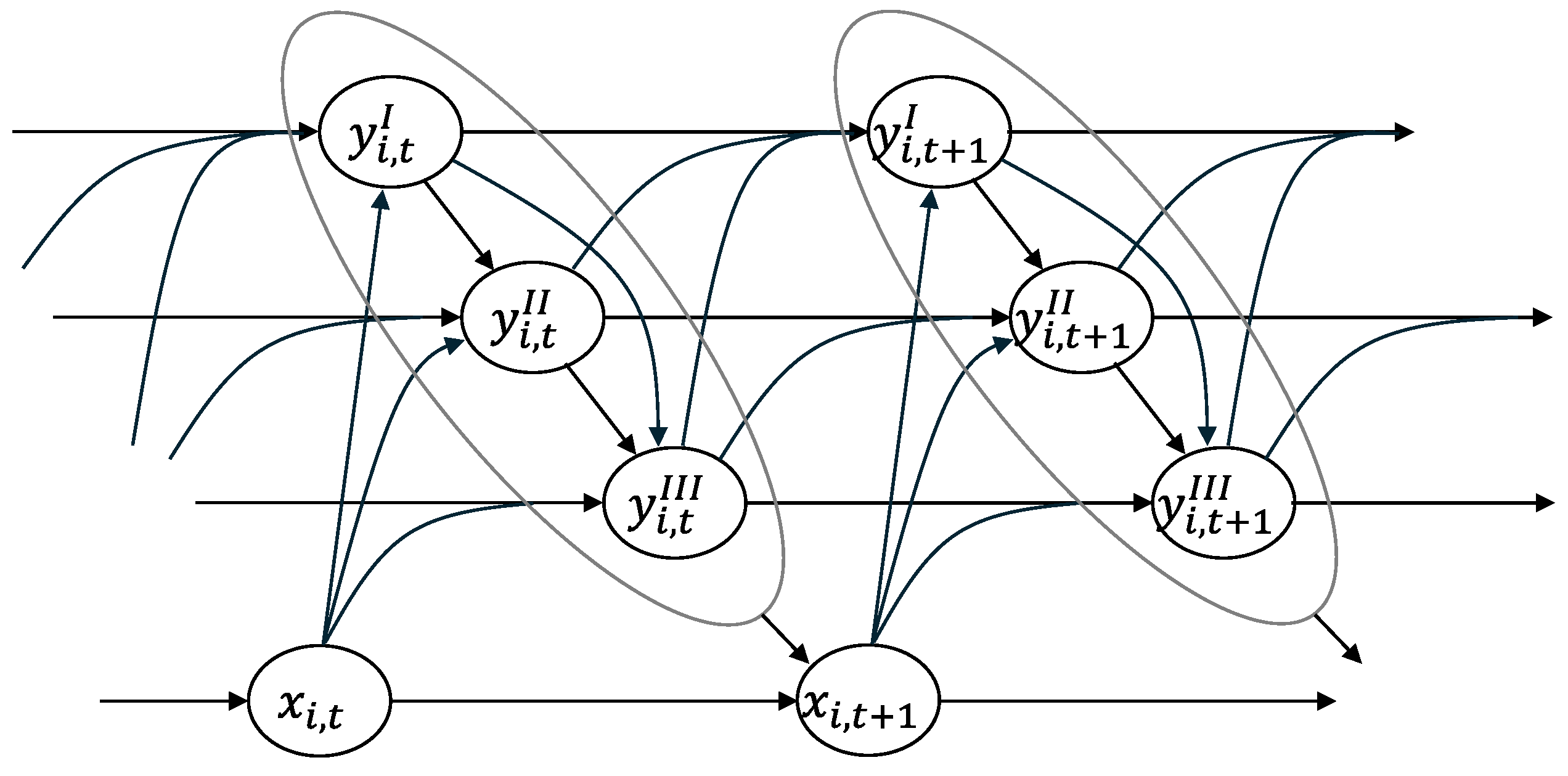
2.3. Analyses
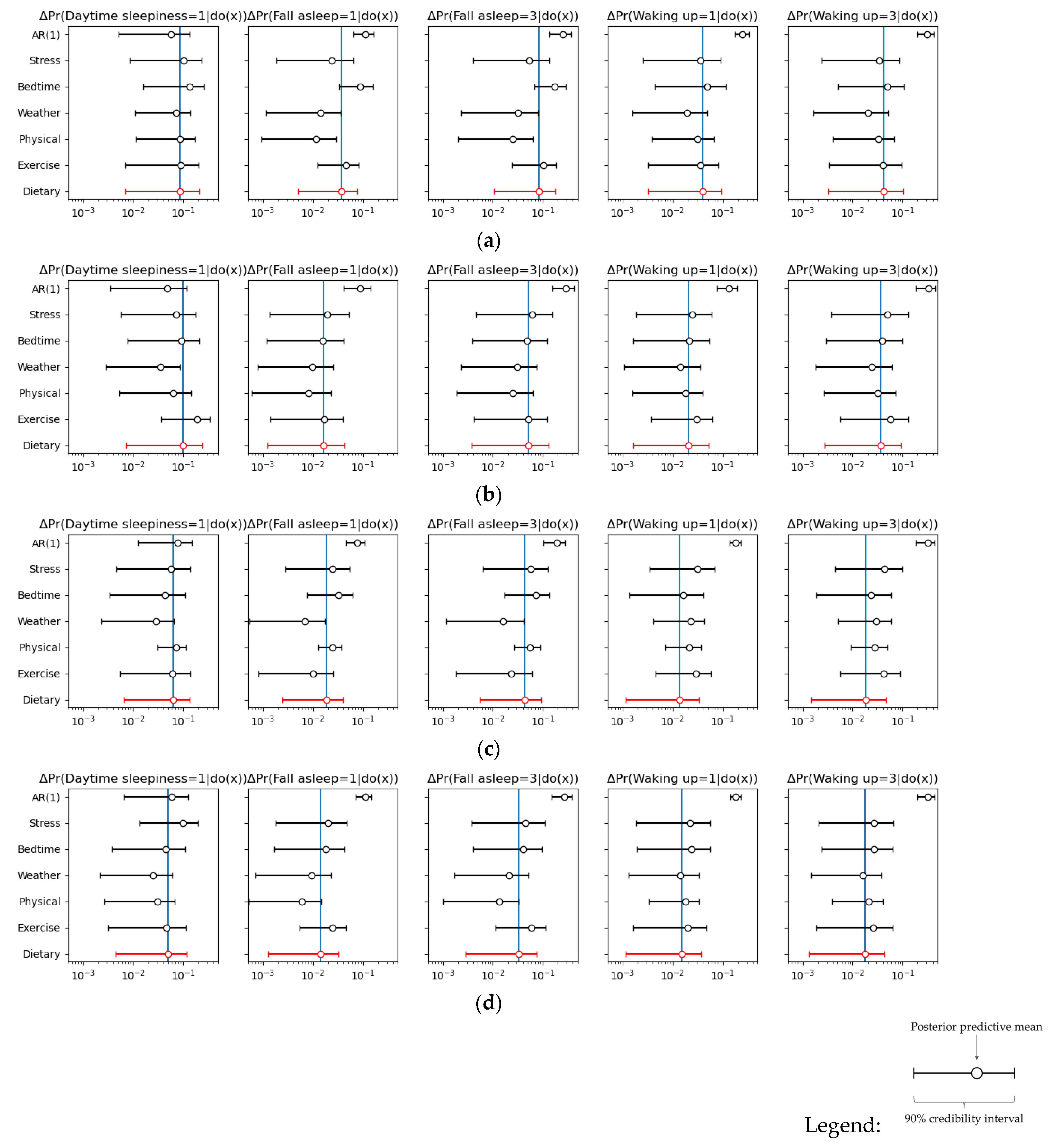
3. Results
4. Discussion
4.1. Discussion of the Analysis Results
4.2. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DAG | Directed acyclic graph |
| DMPM | Dynamic multivariate panel model |
| FFQ | Food frequency questionnaire |
| PSQI | Pittsburgh Sleep Quality Index |
Appendix A
| Variable | Description |
|---|---|
| (Outcome variables) | |
| Daytime sleepiness | Indicating daytime sleepiness (1 = Yes, 0 = No) |
| Fall asleep | 1 = Poor sleep onset, 2 = Slight delay in sleep onset, 3 = Good sleep onset |
| Waking up | 1 = Difficult awakening, 2 = Average, 3 = Refreshing awakening |
| (Stress) | |
| Relax | The intensity of each of the following states—relaxation, irritability, motivation, concentration, worry, and depressed mood—was assessed both before bedtime and after waking using a four-point scale: 1 = Strongly agree, 2 = Agree, 3 = Disagree, 4 = Strongly disagree |
| Irritable | |
| Motivated | |
| Concentration | |
| Worried | |
| Feel down | |
| (Bedtime conditions) | |
| Caffeine | Indicating caffeine intake before bedtime (1 = Yes, 0 = No) |
| Alcohol | Indicating alcohol intake before bedtime (1 = Yes, 0 = No) |
| ICT | Indicating use of ICT devices before bedtime (1 = Yes, 0 = No) |
| Bed time | Bedtime (measured in hourly units from 12:00 AM as the reference point) |
| Time in bed | Time in bed (in hours) |
| (Weather conditions) | |
| Rain | Precipitation (mm) |
| Temperature | Average temperature (°C) |
| ΔTemperature | Diurnal temperature range (°C) |
| Wind | Average wind speed (m/s) |
| Sun light | Sunshine duration (hours) |
| (Physical characteristics) | |
| Age | Age |
| BMI | Body mass index |
| SBP | Systolic blood pressure |
| (Exercise habits) | |
| Exercise 1 | Indicating a habit of light physical activity such as walking (1 = Yes, 0 = No) |
| Exercise 2 | Indicating a habit of moderate physical activity such as brisk walking (1 = Yes, 0 = No) |
| Exercise 3 | Indicating a habit of moderate physical activity such as golf or gardening (1 = Yes, 0 = No) |
| Exercise 4 | Indicating a habit of vigorous physical activity such as tennis or jogging (1 = Yes, 0 = No) |
| (Dietary habits) | |
| Cereals/Potatoes/Beans/GY vegetables/Other vegetables/Fruits/Mushrooms/Seaweeds/Seafood/Meat/Eggs/Dairy | Intake per 1000 kcal of daily energy consumption (g/kcal) |
| Variable | Males, Summer (n = 1118) | Males, Winter (n = 1110) | Females, Summer (n = 2893) | Females, Winter (n = 2923) | ||||
|---|---|---|---|---|---|---|---|---|
| Mean | sd | Mean | sd | Mean | sd | Mean | sd | |
| (Outcome variables) | ||||||||
| Daytime sleepiness | 0.42 | 0.49 | 0.38 | 0.49 | 0.48 | 0.50 | 0.43 | 0.49 |
| Fall asleep | 2.64 | 0.58 | 2.67 | 0.54 | 2.55 | 0.62 | 2.55 | 0.62 |
| Waking up | 2.21 | 0.63 | 2.20 | 0.62 | 2.14 | 0.65 | 2.12 | 0.61 |
| (Stress) | ||||||||
| Relax (before) | 3.25 | 0.82 | 3.26 | 0.82 | 3.15 | 0.86 | 3.18 | 0.83 |
| Relax (after) | 3.02 | 0.86 | 3.05 | 0.89 | 2.85 | 0.93 | 2.86 | 0.92 |
| Irritable (before) | 1.36 | 0.64 | 1.40 | 0.66 | 1.40 | 0.68 | 1.43 | 0.68 |
| Irritable (after) | 1.36 | 0.63 | 1.42 | 0.64 | 1.39 | 0.66 | 1.43 | 0.67 |
| Motivated (before) | 2.62 | 0.90 | 2.59 | 0.97 | 2.36 | 0.89 | 2.36 | 0.89 |
| Motivated (after) | 2.85 | 0.82 | 2.80 | 0.89 | 2.71 | 0.86 | 2.71 | 0.85 |
| Concentration (before) | 2.58 | 0.88 | 2.56 | 0.93 | 2.30 | 0.85 | 2.32 | 0.86 |
| Concentration (after) | 2.70 | 0.84 | 2.74 | 0.86 | 2.53 | 0.84 | 2.58 | 0.85 |
| Worried (before) | 1.69 | 0.87 | 1.73 | 0.87 | 1.90 | 0.96 | 1.91 | 0.95 |
| Worried (after) | 1.67 | 0.85 | 1.73 | 0.86 | 1.86 | 0.92 | 1.91 | 0.94 |
| Feel down (before) | 1.40 | 0.68 | 1.49 | 0.72 | 1.53 | 0.79 | 1.54 | 0.78 |
| Feel down (after) | 1.42 | 0.69 | 1.47 | 0.69 | 1.52 | 0.77 | 1.53 | 0.75 |
| (Bedtime conditions) | ||||||||
| Caffeine | 0.15 | 0.36 | 0.16 | 0.37 | 0.15 | 0.36 | 0.17 | 0.37 |
| Alcohol | 0.44 | 0.50 | 0.45 | 0.50 | 0.25 | 0.44 | 0.24 | 0.43 |
| ICT | 0.59 | 0.49 | 0.61 | 0.49 | 0.64 | 0.48 | 0.64 | 0.48 |
| Bed time | −0.39 | 1.75 | −0.37 | 1.77 | −0.34 | 1.63 | −0.26 | 1.58 |
| Time in bed | 6.80 | 1.59 | 6.99 | 1.54 | 6.88 | 1.64 | 7.10 | 1.75 |
| (Weather conditions) | ||||||||
| Rain | 2.21 | 5.47 | 1.83 | 3.84 | 2.15 | 5.52 | 1.76 | 3.51 |
| Temperature | 18.27 | 2.93 | 0.00 | 5.16 | 18.34 | 3.05 | 0.61 | 5.17 |
| ΔTemperature | 10.07 | 3.51 | 8.82 | 3.57 | 10.51 | 3.73 | 8.71 | 3.55 |
| Wind | 3.29 | 1.41 | 3.03 | 1.47 | 3.28 | 1.42 | 3.05 | 1.44 |
| Sun light | 6.03 | 4.44 | 3.08 | 2.29 | 6.63 | 4.55 | 2.99 | 2.19 |
| (Physical characteristics) | ||||||||
| Age | 53.94 | 12.43 | 54.11 | 12.40 | 49.82 | 10.97 | 50.04 | 11.06 |
| BMI | 23.73 | 3.59 | 23.66 | 3.14 | 21.46 | 3.36 | 21.55 | 3.37 |
| SBP | 128.04 | 17.87 | 129.51 | 17.54 | 114.47 | 16.87 | 112.75 | 16.52 |
| (Exercise habits) | ||||||||
| Exercise 1 | 0.40 | 0.49 | 0.47 | 0.50 | 0.38 | 0.49 | 0.42 | 0.49 |
| Exercise 2 | 0.31 | 0.46 | 0.42 | 0.49 | 0.30 | 0.46 | 0.31 | 0.46 |
| Exercise 3 | 0.47 | 0.50 | 0.37 | 0.48 | 0.39 | 0.49 | 0.31 | 0.46 |
| Exercise 4 | 0.31 | 0.46 | 0.23 | 0.42 | 0.14 | 0.35 | 0.16 | 0.37 |
| (Dietary habits) | ||||||||
| Cereals | 183.28 | 32.38 | 182.47 | 32.71 | 176.14 | 12.40 | 175.99 | 12.64 |
| Potatoes | 19.62 | 4.52 | 19.58 | 4.53 | 23.16 | 1.83 | 23.15 | 1.85 |
| Beans | 40.66 | 23.36 | 40.36 | 22.99 | 43.94 | 21.16 | 44.23 | 21.77 |
| GY vegetables | 80.02 | 28.35 | 80.37 | 29.80 | 88.00 | 18.36 | 88.23 | 18.56 |
| Other vegetables | 89.58 | 19.92 | 89.42 | 19.99 | 98.95 | 10.71 | 98.88 | 10.84 |
| Fruits | 63.97 | 12.60 | 64.28 | 12.77 | 88.02 | 7.54 | 88.16 | 7.78 |
| Mushrooms | 7.60 | 2.62 | 7.59 | 2.62 | 12.15 | 6.67 | 12.20 | 6.69 |
| Seaweeds | 5.94 | 1.88 | 5.95 | 1.90 | 5.06 | 1.50 | 5.08 | 1.54 |
| Seafood | 47.71 | 4.79 | 47.72 | 4.71 | 45.24 | 3.29 | 45.16 | 3.23 |
| Meat | 35.23 | 6.98 | 35.11 | 6.79 | 39.06 | 11.27 | 39.14 | 11.39 |
| Eggs | 16.64 | 1.72 | 16.70 | 1.75 | 19.52 | 5.76 | 19.61 | 5.89 |
| Dairy | 81.33 | 41.43 | 81.41 | 42.06 | 94.79 | 26.43 | 94.94 | 27.42 |
Appendix B
References
- Moussa-Chamari, I.; Farooq, A.; Romdhani, M.; Washif, J.A.; Bakare, U.; Helmy, M.; Al-Horani, R.A.; Salamh, P.; Robin, N.; Hue, O. The relationship between quality of life, sleep quality, mental health, and physical activity in an international sample of college students: A structural equation modeling approach. Front. Public Health 2024, 12, 1397924. [Google Scholar] [CrossRef]
- Chen, B.; Alexandros, N.V.; Li, Y. Good sleep and health. Lancet Healthy Longev. 2023, 4, e295. [Google Scholar] [CrossRef]
- Arora, T.; Grey, I.; Östlundh, L.; Alamoodi, A.; Omar, O.M.; Lam, K.H.; Grandner, M. A systematic review and meta-analysis to assess the relationship between sleep duration/quality, mental toughness and resilience amongst healthy individuals. Sleep Med. Rev. 2022, 62, 101593. [Google Scholar] [CrossRef]
- Carpi, M.; Cianfarani, C.; Vestri, A. Sleep Quality and Its Associations with Physical and Mental Health-Related Quality of Life among University Students: A Cross-Sectional Study. Int. J. Environ. Res. Public Health 2022, 19, 2874. [Google Scholar] [CrossRef]
- Scott, A.J.; Webb, T.L.; James, M.M.; Rowse, G.; Weich, S. Improving sleep quality leads to better mental health: A meta-analysis of randomised controlled trials. Sleep Med. Rev. 2021, 60, 101556. [Google Scholar] [CrossRef]
- Clement-Carbonell, V.; Portilla-Tamarit, I.; Rubio-Aparicio, M.; Madrid-Valero, J.J. Sleep Quality, Mental and Physical Health: A Differential Relationship. Int. J. Environ. Res. Public Health 2021, 18, 460. [Google Scholar] [CrossRef]
- Sella, E.; Miola, L.; Toffalini, E.; Borella, E. The relationship between sleep quality and quality of life in aging: A systematic review and meta-analysis. Health Psychol. Rev. 2021, 17, 169–191. [Google Scholar] [CrossRef] [PubMed]
- Altena, E.; Ellis, J.G. How Sleep Affects Daytime Functioning: The Latest Insights from Different Patient and Age Groups. Brain Sci. 2021, 11, 1163. [Google Scholar] [CrossRef]
- Mazzotti, D.R.; Guindalini, C.; Moraes, W.A.; Andersen, M.L.; Cendoroglo, M.S.; Ramos, L.R.; Tufik, S. Human longevity is associated with regular sleep patterns, maintenance of slow wave sleep, and favorable lipid profile. Front. Aging Neurosci. 2014, 6, 134. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.; Cheung, J. The effect of mediterranean diet and chrononutrition on sleep quality: A scoping review. Nutr. J. 2025, 24, 31. [Google Scholar] [CrossRef] [PubMed]
- Godos, J.; Grosso, G.; Castellano, S.; Galvano, F.; Caraci, F.; Ferri, R. Association between diet and sleep quality: A systematic review. Sleep Med. Rev. 2021, 57, 101430. [Google Scholar] [CrossRef] [PubMed]
- Binks, H.; Vincent, G.E.; Gupta, C.; Irwin, C.; Khalesi, S. Effects of Diet on Sleep: A Narrative Review. Nutrients 2020, 12, 936. [Google Scholar] [CrossRef]
- Harvard Health Publishing. Could What We Eat Improve our Sleep? Available online: https://www.health.harvard.edu/blog/could-what-we-eat-improve-our-sleep-2021030922112 (accessed on 23 July 2025).
- St-Onge, M.P.; Mikic, A.; Pietrolungo, C.E. Effects of Diet on Sleep Quality. Adv. Nutr. 2016, 7, 938–949. [Google Scholar] [CrossRef]
- Alruwaili, N.W.; Alqahtani, N.; Alanazi, M.H.; Alanazi, B.S.; Aljrbua, M.S.; Gatar, O.M. The effect of nutrition and physical activity on sleep quality among adults: A scoping review. Sleep Sci. Pract. 2023, 7, 8. [Google Scholar] [CrossRef]
- Wilson, K.; St-Onge, M.P.; Tasali, E. Diet Composition and Objectively Assessed Sleep Quality: A Narrative Review. J. Acad. Nutr. Diet. 2022, 122, 1182–1195. [Google Scholar] [CrossRef]
- Sandri, E.; Broccolo, A.; Piredda, M. Socio-Demographic, Nutritional, and Lifestyle Factors Influencing Perceived Sleep Quality in Spain, with a Particular Focus on Women and Young People. Nutrients 2025, 17, 1065. [Google Scholar] [CrossRef]
- Çakir, B.; Nişancı Kılınç, F.; Özata Uyar, G.; Özenir, Ç.; Merve Ekici, E.; Karaismailoğlu, E. The relationship between sleep duration, sleep quality and dietary intake in adults. Sleep Biol. Rhythm. 2020, 18, 49–57. [Google Scholar] [CrossRef]
- Nisar, M.; Mohammad, R.M.; Arshad, A.; Hashmi, I.; Yousuf, S.M.; Baig, S. Influence of Dietary Intake on Sleeping Patterns of Medical Students. Cureus 2019, 11, e4106. [Google Scholar] [CrossRef]
- Zuraikat, F.M.; Wood, R.A.; Barragán, R.; St-Onge, M.P. Sleep and Diet: Mounting Evidence of a Cyclical Relationship. Annu. Rev. Nutr. 2021, 41, 309–332. [Google Scholar] [CrossRef]
- Zuraikat, F.M.; Makarem, N.; Liao, M.; St-Onge, M.P.; Aggarwal, B. Measures of Poor Sleep Quality Are Associated With Higher Energy Intake and Poor Diet Quality in a Diverse Sample of Women From the Go Red for Women Strategically Focused Research Network. J. Am. Heart Assoc. 2020, 9, e014587. [Google Scholar] [CrossRef]
- Conti, F. Dietary Protocols to Promote and Improve Restful Sleep: A Narrative Review. Nutr. Rev. 2025. ahead of print. [Google Scholar] [CrossRef]
- Pearl, J. Causality: Models, Reasoning, and Inference, 2nd ed.; Cambridge University Press: New York, NY, USA, 2009. [Google Scholar]
- Huang, Y.; Yang, L.; Liu, Y.; Zhang, S. Effects of perceived stress on college students’ sleep quality: A moderated chain mediation model. BMC Psychol. 2024, 12, 476. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Xiang, S.; Li, X.; Tang, Y.; Hu, Q. The impact of stress on sleep quality: A mediation analysis based on longitudinal data. Front. Psychol. 2024, 15, 1431234. [Google Scholar] [CrossRef]
- Zhang, J.; Li, X.; Tang, Z.; Xiang, S.; Tang, Y.; Hu, W.; Tan, C.; Wang, X. Effects of stress on sleep quality: Multiple mediating effects of rumination and social anxiety. Psicol. Reflex. Crít. 2024, 37, 10. [Google Scholar] [CrossRef]
- Narmandakh, A.; Oldehinkel, A.J.; Masselink, M.; de Jonge, P.; Roest, A.M. Affect, worry, and sleep: Between- and within-subject associations in a diary study. J. Affect. Disord. Rep. 2021, 4, 100134. [Google Scholar] [CrossRef]
- Martire, V.L.; Caruso, D.; Palagini, L.; Zoccoli, G.; Bastianini, S. Stress & sleep: A relationship lasting a lifetime. Neurosci. Biobehav. Rev. 2020, 117, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Gu, S.; Wang, Z.; Li, H.; Xu, X.; Zhu, H.; Deng, S.; Ma, X.; Feng, G.; Wang, F.; et al. Relationship Between Stressful Life Events and Sleep Quality: Rumination as a Mediator and Resilience as a Moderator. Front. Psychiatry 2019, 10, 348. [Google Scholar] [CrossRef] [PubMed]
- Kalmbach, D.A.; Anderson, J.R.; Drake, C.L. The impact of stress on sleep: Pathogenic sleep reactivity as a vulnerability to insomnia and circadian disorders. J. Sleep Res. 2018, 27, e12710. [Google Scholar] [CrossRef]
- Obayashi, K.; Saeki, K.; Iwamoto, J.; Okamoto, N.; Tomioka, K.; Nezu, S.; Ikada, Y.; Kurumatani, N. Effect of exposure to evening light on sleep initiation in the elderly: A longitudinal analysis for repeated measurements in home settings. Chronobiol. Int. 2014, 31, 461–467. [Google Scholar] [CrossRef]
- Xu, Y.X.; Zhang, J.H.; Tao, F.B.; Sun, Y. Association between exposure to light at night (LAN) and sleep problems: A systematic review and meta-analysis of observational studies. Sci. Total Environ. 2023, 857, 159303. [Google Scholar] [CrossRef]
- Okamoto-Mizuno, K.; Tsuzuki, K. Effects of season on sleep and skin temperature in the elderly. Int. J. Biometeorol. 2010, 54, 401–409. [Google Scholar] [CrossRef]
- Saeki, K.; Obayashi, K.; Tone, N.; Kurumatani, N. A warmer indoor environment in the evening and shorter sleep onset latency in winter: The HEIJO-KYO study. Physiol. Behav. 2015, 149, 29–34. [Google Scholar] [CrossRef]
- Czeisler, C.A.; Gooley, J.J. Sleep and circadian rhythms in humans. Cold Spring Harb. Symp. Quant. Biol. 2007, 72, 579–597. [Google Scholar] [CrossRef]
- Obayashi, K.; Saeki, K.; Iwamoto, J.; Okamoto, N.; Tomioka, K.; Nezu, S.; Ikada, Y.; Kurumatani, N. Positive effect of daylight exposure on nocturnal urinary melatonin excretion in the elderly: A cross-sectional analysis of the HEIJO-KYO study. J. Clin. Endocrinol. Metab. 2012, 97, 4166–4173. [Google Scholar] [CrossRef]
- Cavaillès, C.; Stone, K.L.; Leng, Y.; Peltz, C.; Yaffe, K. Objective sleep quality in diverse older adults: A cross-sectional study on the importance of race and ethnicity and sex. BMC Med. 2025, 23, 391. [Google Scholar] [CrossRef]
- Li, S.; Yang, B.; Shang, S.; Jiang, W. Associations of BMI, sleep quality, and sleep duration trajectories with new-onset diabetes mellitus in the elderly. Obesity 2025, 33, 1595–1605. [Google Scholar] [CrossRef]
- Kim, Y.; Mattos, M.K.; Esquivel, J.H.; Davis, E.M.; Logan, J. Sleep and blood pressure variability: A systematic literature review. Heart Lung 2024, 68, 323–336. [Google Scholar] [CrossRef] [PubMed]
- Al Haddad, N.; Costanian, C.; Zibara, V.; Bilen, Y.; Kilani, H.; Tohme, F.; Bahous, S.A. The association between sleep disturbances and blood pressure variability: A review of the literature. J. Clin. Sleep. Med. 2023, 19, 1533–1544. [Google Scholar] [CrossRef] [PubMed]
- Amiri, S. Body mass index and sleep disturbances: A systematic review and meta-analysis. Postep. Psychiatr. Neurol. 2023, 32, 96–109. [Google Scholar] [CrossRef] [PubMed]
- Casagrande, M.; Forte, G.; Favieri, F.; Corbo, I. Sleep Quality and Aging: A Systematic Review on Healthy Older People, Mild Cognitive Impairment and Alzheimer’s Disease. Int. J. Environ. Res. Public Health 2022, 19, 8457. [Google Scholar] [CrossRef]
- Li, L.; Nakamura, T.; Hayano, J.; Yamamoto, Y. Age and gender differences in objective sleep properties using large-scale body acceleration data in a Japanese population. Sci. Rep. 2021, 11, 9970. [Google Scholar] [CrossRef]
- Garfield, V. The Association Between Body Mass Index (BMI) and Sleep Duration: Where Are We after nearly Two Decades of Epidemiological Research? Int. J. Environ. Res. Public Health 2019, 16, 4327. [Google Scholar] [CrossRef]
- Madrid-Valero, J.J.; Martínez-Selva, J.M.; Ribeiro do Couto, B.; Sánchez-Romera, J.F.; Ordoñana, J.R. Age and gender effects on the prevalence of poor sleep quality in the adult population. Gac. Sanit. 2017, 31, 18–22. [Google Scholar] [CrossRef] [PubMed]
- Kline, C.E.; Hillman, C.H.; Bloodgood Sheppard, B.; Tennant, B.; Conroy, D.E.; Macko, R.F.; Marquez, D.X.; Petruzzello, S.J.; Powell, K.E.; Erickson, K.I. Physical activity and sleep: An updated umbrella review of the 2018 Physical Activity Guidelines Advisory Committee report. Sleep Med. Rev. 2021, 58, 101489. [Google Scholar] [CrossRef]
- Haupt, S.; Eckstein, M.L.; Wolf, A.; Zimmer, R.T.; Wachsmuth, N.B.; Moser, O. Eat, Train, Sleep—Retreat? Hormonal Interactions of Intermittent Fasting, Exercise and Circadian Rhythm. Biomolecules 2021, 11, 516. [Google Scholar] [CrossRef]
- Matsumoto, T.; Tabara, Y.; Murase, K.; Takahashi, Y.; Setoh, K.; Kawaguchi, T.; Muro, S.; Kadotani, H.; Kosugi, S.; Sekine, A.; et al. Combined association of clinical and lifestyle factors with non-restorative sleep: The Nagahama Study. PLoS ONE 2017, 12, e0171849. [Google Scholar] [CrossRef] [PubMed]
- Kubitz, K.A.; Landers, D.M.; Petruzzello, S.J.; Han, M. The effects of acute and chronic exercise on sleep. A Meta-Analytic review. Sports Med. 1996, 21, 277–291. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Alén, M.; Wiklund, P.; Partinen, M.; Cheng, S. Effects of aerobic exercise on home-based sleep among overweight and obese men with chronic insomnia symptoms: A randomized controlled trial. Sleep Med. 2016, 25, 113–121. [Google Scholar] [CrossRef]
- Aritake-Okada, S.; Tanabe, K.; Mochizuki, Y.; Ochiai, R.; Hibi, M.; Kozuma, K.; Katsuragi, Y.; Ganeko, M.; Takeda, N.; Uchida, S. Diurnal repeated exercise promotes slow-wave activity and fast-sigma power during sleep with increase in body temperature: A human crossover trial. J. Appl. Physiol 2019, 127, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Kovacevic, A.; Mavros, Y.; Heisz, J.J.; Fiatarone Singh, M.A. The effect of resistance exercise on sleep: A systematic review of randomized controlled trials. Sleep Med. Rev. 2018, 39, 52–68. [Google Scholar] [CrossRef]
- Helske, J.; Tikka, S. Estimating causal effects from panel data with dynamic multivariate panel models. Adv. Life Course Res. 2024, 60, 100617. [Google Scholar] [CrossRef]
- Tsubono, Y.; Takamori, S.; Kobayashi, M.; Takahashi, T.; Iwase, Y.; Iitoi, Y.; Akabane, M.; Yamaguchi, M.; Tsugane, S. A data-based approach for designing a semiquantitative food frequency questionnaire for a population-based prospective study in Japan. J. Epidemiol. 1996, 6, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Kagami-Katsuyama, H.; Sato-Ueshima, M.; Satoh, K.; Tousen, Y.; Takimoto, H.; Maeda-Yamamoto, M.; Nishihira, J. The relationship between mental and physical minor health complaints and the intake of dietary nutrients. Nutrients 2023, 15, 865. [Google Scholar] [CrossRef]
- Nelson, K.L.; Davis, J.E.; Corbett, C.F. Sleep quality: An evolutionary concept analysis. Nurs. Forum 2022, 57, 144–151. [Google Scholar] [CrossRef]
- Ohayon, M.; Wickwire, E.M.; Hirshkowitz, M.; Albert, S.M.; Avidan, A.; Daly, F.J.; Dauvilliers, Y.; Ferri, R.; Fung, C.; Gozal, D.; et al. National Sleep Foundation’s sleep quality recommendations: First report. Sleep Health 2017, 3, 6–19. [Google Scholar] [CrossRef]
- Meyer, N.; Harvey, A.G.; Lockley, S.W.; Dijk, D.J. Circadian rhythms and disorders of the timing of sleep. Lancet 2022, 400, 1061–1078. [Google Scholar] [CrossRef]
- Pérez-Carbonell, L.; Mignot, E.; Leschziner, G.; Dauvilliers, Y. Understanding and approaching excessive daytime sleepiness. Lancet 2022, 400, 1033–1046. [Google Scholar] [CrossRef] [PubMed]
- Buysse, D.J.; Reynolds, C.F., 3rd; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef] [PubMed]
- Mollayeva, T.; Thurairajah, P.; Burton, K.; Mollayeva, S.; Shapiro, C.M.; Colantonio, A. The Pittsburgh sleep quality index as a screening tool for sleep dysfunction in clinical and non-clinical samples: A systematic review and meta-analysis. Sleep Med. Rev. 2016, 25, 52–73. [Google Scholar] [CrossRef]
- Doi, Y.; Minowa, M.; Uchiyama, M.; Okawa, M.; Kim, K.; Shibui, K.; Kamei, Y. Psychometric assessment of subjective sleep quality using the Japanese version of the Pittsburgh Sleep Quality Index (PSQI-J) in psychiatric disordered and control subjects. Psychiatry Res. 2000, 97, 165–172. [Google Scholar] [CrossRef]
- Shimomitsu, T.; Haratani, T.; Nakamura, K.; Kawakami, N.; Hayashi, T.; Hiro, H. The Final Development of the Brief Job Stress Questionnaire Mainly Used for Assessment of the Individuals. In Ministry of Labor Sponsored Grant for the Prevention of Work-related Illness: The 1999 Report; Kato, M., Ed.; Tokyo Medical University: Tokyo, Japan, 2000; pp. 126–164. (In Japanese) [Google Scholar]
- Inoue, A.; Kawakami, N.; Shimomitsu, T.; Tsutsumi, A.; Haratani, T.; Yoshikawa, T.; Shimazu, A.; Odagiri, Y. Development of a short questionnaire to measure an extended set of job demands, job resources, and positive health outcomes: The New Brief Job Stress Questionnaire. Ind. Health 2014, 52, 175–189. [Google Scholar] [CrossRef] [PubMed]
- Japan Meteorological Agency. Available online: https://www.data.jma.go.jp/stats/etrn/index.php (accessed on 20 July 2025).
- Viswanath, V.K.; Hartogenesis, W.; Dilchert, S.; Pandya, L.; Hecht, F.M.; Mason, A.E.; Wang, E.J.; Smarr, B.L. Five million nights: Temporal dynamics in human sleep phenotypes. NPJ Digit. Med. 2024, 7, 150. [Google Scholar] [CrossRef]
- Ekuni, R.; Souza, B.M.N.; Cogo-Moreira, H.; Lourenço, F.C.; Pompeia, S. Bidirectional longitudinal dynamics of self-reported total sleep time and perceived stress: Establishing potential causal relationships. Sleep Health 2022, 8, 406–409. [Google Scholar] [CrossRef]
- Zhou, Z.; Birditt, K.S.; Leger, K.A.; Fingerman, K.L. Daily worry, rumination, and sleep in late life. J. Psychosom. Res. 2024, 179, 111622. [Google Scholar] [CrossRef]
- Blaxton, J.M.; Bergeman, C.S.; Whitehead, B.R.; Braun, M.E.; Payne, J.D. Relationships Among Nightly Sleep Quality, Daily Stress, and Daily Affect. J. Gerontol. Ser. B 2017, 72, 363–372. [Google Scholar] [CrossRef]
- Tikka, S.; Helske, J. dynamite: An R Package for Dynamic Multivariate Panel Models. arXiv 2023, arXiv:2302.01607. [Google Scholar] [CrossRef]
- Imaeda, N.; Goto, C.; Sasakabe, T.; Mikami, H.; Oze, I.; Hosono, A.; Naito, M.; Miyagawa, N.; Ozaki, E.; Ikezaki, H.; et al. Reproducibility and validity of food group intake in a short food frequency questionnaire for the middle-aged Japanese population. Environ. Health Prev. Med. 2021, 26, 28. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Liu, X. A chain mediating model of the impact of physical exercise on sleep quality. BMC Public Health 2025, 25, 1493. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhang, Q.; Zhu, P.; Dong, Y.; Tan, L.; Liu, P.; Yi, Z. A chain mediation model for physical exercise and sleep quality. Sci. Rep. 2025, 15, 30527. [Google Scholar] [CrossRef]
- Popoola, A.A.; Frediani, J.K.; Hartman, T.J.; Paynabar, K. Mitigating Underreported Error in Food Frequency Questionnaire Data Using a Supervised Machine Learning Method and Error Adjustment Algorithm. BMC Med. Inform. Decis. Mak. 2023, 23, 178. [Google Scholar] [CrossRef] [PubMed]
- Scott, R.Q.; Rodríguez, A.J. Improving Quality of Sleep in Healthy Adults. Curr. Pulmonol. Rep. 2023, 12, 46–55. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hazama, M.; Kagami-Katsuyama, H.; Ito, N.; Maeda-Yamamoto, M.; Nishihira, J. How Important Are Dietary Habits Compared to Other Factors for Sleep Quality?—An Analysis Using Data from a Specific Region in Japan. Nutrients 2025, 17, 2787. https://doi.org/10.3390/nu17172787
Hazama M, Kagami-Katsuyama H, Ito N, Maeda-Yamamoto M, Nishihira J. How Important Are Dietary Habits Compared to Other Factors for Sleep Quality?—An Analysis Using Data from a Specific Region in Japan. Nutrients. 2025; 17(17):2787. https://doi.org/10.3390/nu17172787
Chicago/Turabian StyleHazama, Makoto, Hiroyo Kagami-Katsuyama, Naohito Ito, Mari Maeda-Yamamoto, and Jun Nishihira. 2025. "How Important Are Dietary Habits Compared to Other Factors for Sleep Quality?—An Analysis Using Data from a Specific Region in Japan" Nutrients 17, no. 17: 2787. https://doi.org/10.3390/nu17172787
APA StyleHazama, M., Kagami-Katsuyama, H., Ito, N., Maeda-Yamamoto, M., & Nishihira, J. (2025). How Important Are Dietary Habits Compared to Other Factors for Sleep Quality?—An Analysis Using Data from a Specific Region in Japan. Nutrients, 17(17), 2787. https://doi.org/10.3390/nu17172787






