Soy Consumption and the Risk of Type 2 Diabetes and Cardiovascular Diseases: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Sources and Search Strategy
2.2. Eligibility Criteria and Study Selection
2.3. Data Extraction
2.4. Quality Assessment
2.5. Statistical Analysis
3. Results
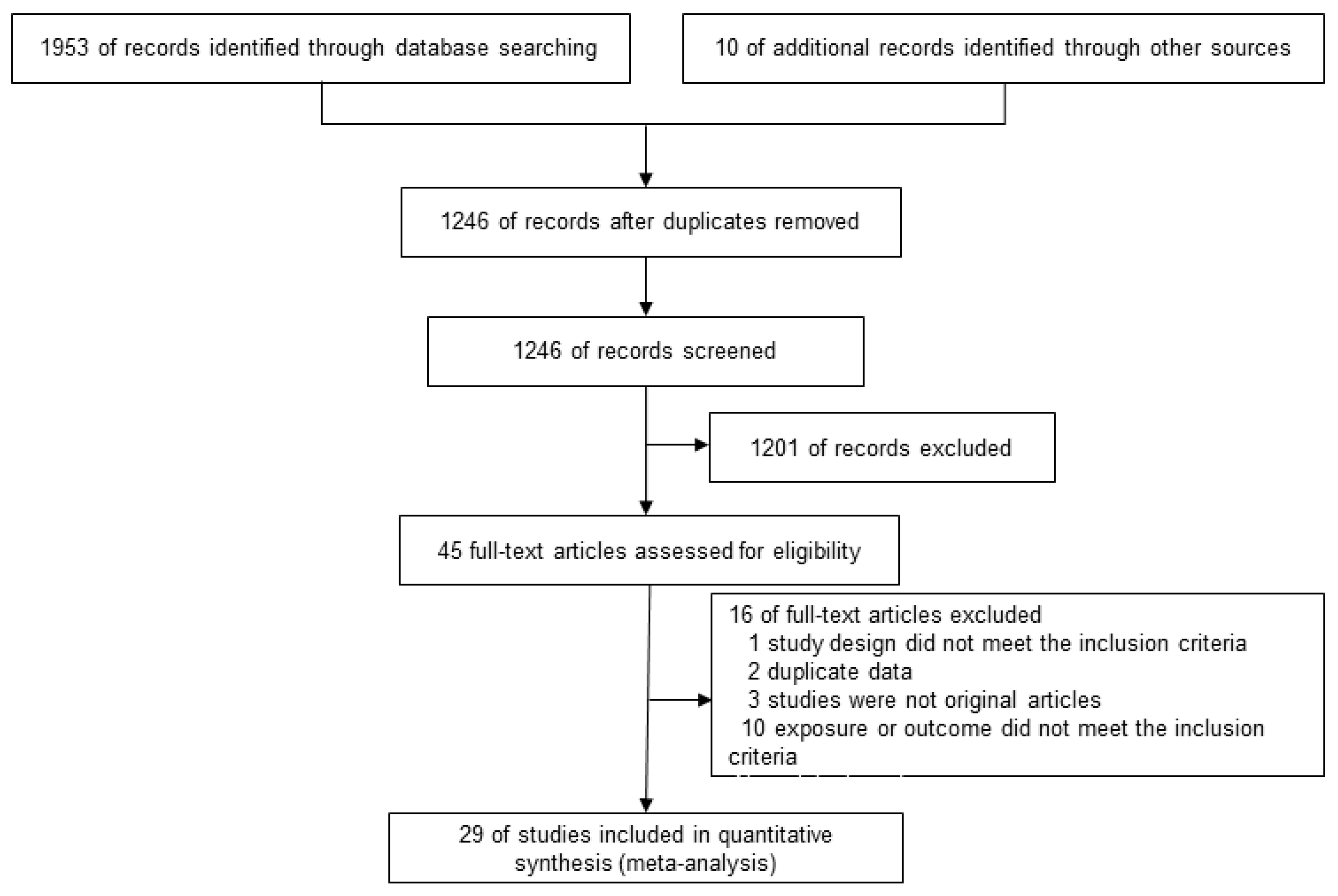
3.1. Literature Search Results
3.2. Study Characteristics and Quality Assessment
3.3. Synthesis with Meta-Analysis
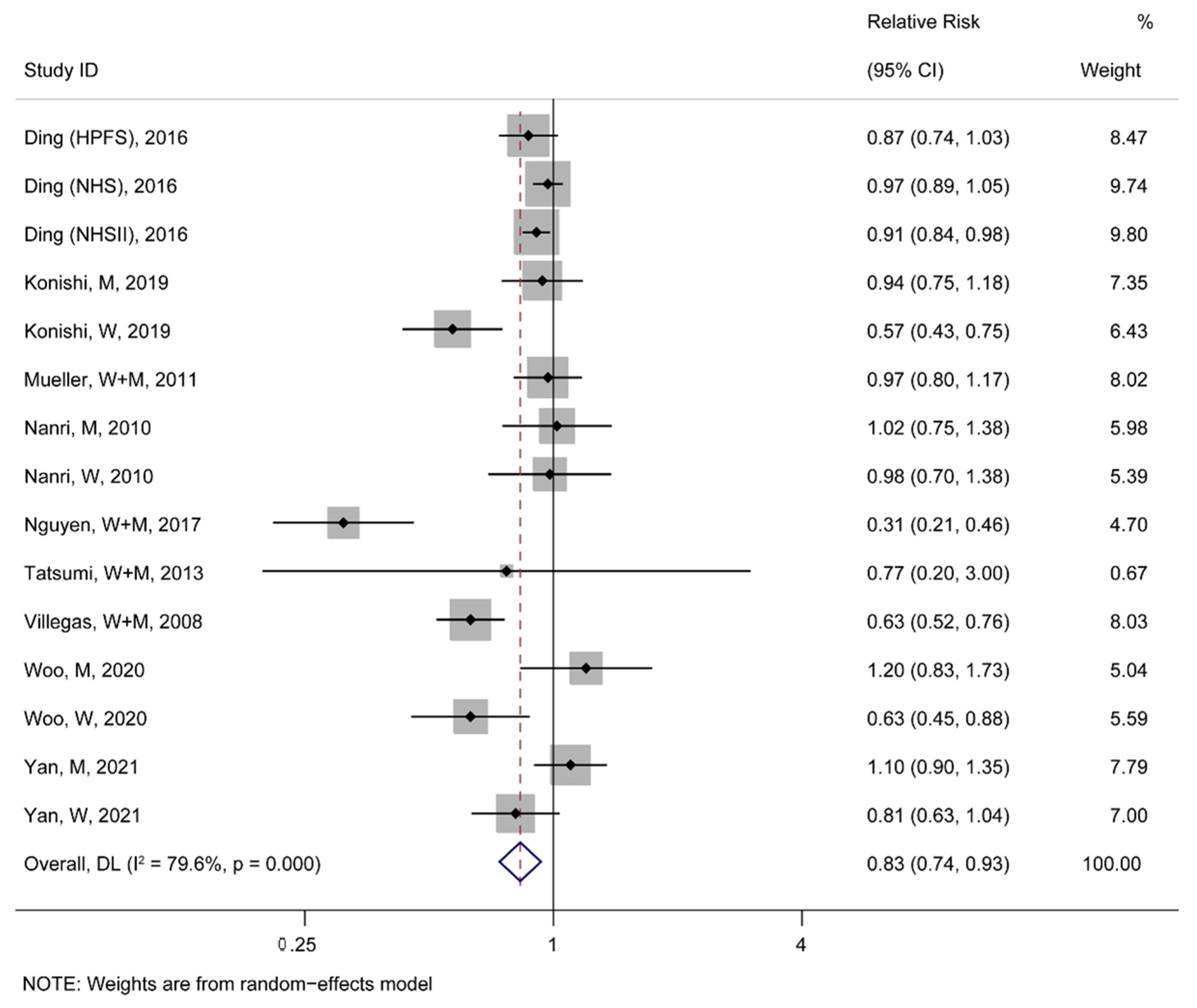
3.3.1. Association between Soy Consumption and the Risk of T2D and CVDs
3.3.2. Subgroup Analysis between Soy Foods Consumption and the Risk of T2D and CVDs Events
3.3.3. Subgroup Analysis between Baseline Characteristics and the Risk of T2D and CVDs Events
3.4. Publication Bias
4. Discussion
4.1. Main Findings and Interpretation
4.2. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chatterjee, C.; Gleddie, S.; Xiao, C.W. Soybean bioactive peptides and their functional properties. Nutrients 2018, 10, 1211. [Google Scholar] [CrossRef] [PubMed]
- Naghshi, S.; Sadeghi, O.; Willett, W.C.; Esmaillzadeh, A. Dietary intake of total, animal, and plant proteins and risk of all cause, cardiovascular, and cancer mortality: Systematic review and dose-response meta-analysis of prospective cohort studies. BMJ 2020, 370, m2412. [Google Scholar] [CrossRef] [PubMed]
- Engelen, M.P.; Rutten, E.P.; Castro, C.L.; Wouters, E.F.; Schols, A.M.; Deutz, N.E. Supplementation of soy protein with branched-chain amino acids alters protein metabolism in healthy elderly and even more in patients with chronic obstructive pulmonary disease. Am. J. Clin. Nutr. 2007, 85, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Rushovich, D.; Weil, R. Sulfur fertility management to enhance methionine and cysteine in soybeans. J. Sci. Food Agric. 2021, 101, 6595–6601. [Google Scholar] [CrossRef] [PubMed]
- Argiles, J.M.; Campos, N.; Lopez-Pedrosa, J.M.; Rueda, R.; Rodriguez-Manas, L. Skeletal muscle regulates metabolism via interorgan crosstalk: Roles in health and disease. J. Am. Med. Dir. Assoc. 2016, 17, 789–796. [Google Scholar] [CrossRef] [PubMed]
- Neinast, M.D.; Jang, C.; Hui, S.; Murashige, D.S.; Chu, Q.; Morscher, R.J.; Li, X.; Zhan, L.; White, E.; Anthony, T.G.; et al. Quantitative analysis of the whole-body metabolic fate of branched-chain amino acids. Cell. Metab. 2019, 29, 417–429.e414. [Google Scholar] [CrossRef]
- Mcgarrah, R.W.; White, P.J. Branched-chain amino acids in cardiovascular disease. Nat. Rev. Cardiol. 2023, 20, 77–89. [Google Scholar] [CrossRef]
- Richardson, N.E.; Konon, E.N.; Schuster, H.S.; Mitchell, A.T.; Boyle, C.; Rodgers, A.C.; Finke, M.; Haider, L.R.; Yu, D.; Flores, V.; et al. Lifelong restriction of dietary branched-chain amino acids has sex-specific benefits for frailty and life span in mice. Nat. Aging 2021, 1, 73–86. [Google Scholar] [CrossRef]
- Ramdath, D.D.; Padhi, E.M.; Sarfaraz, S.; Renwick, S.; Duncan, A.M. Beyond the cholesterol-lowering effect of soy protein: A review of the effects of dietary soy and its constituents on risk factors for cardiovascular disease. Nutrients 2017, 9, 324. [Google Scholar] [CrossRef]
- Bakrim, S.; Benkhaira, N.; Bourais, I.; Benali, T.; Lee, L.H.; El Omari, N.; Sheikh, R.A.; Goh, K.W.; Ming, L.C.; Bouyahya, A.; et al. Health benefits and pharmacological properties of stigmasterol. Antioxidants 2022, 11, 1912. [Google Scholar] [CrossRef]
- Jayachandran, M.; Xu, B. An insight into the health benefits of fermented soy products. Food Chem. 2019, 271, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yu, C.; Lv, J.; Li, L.; Hu, Y.; Liu, K.; Shirai, K.; Iso, H.; Dong, J.Y. Consumption of soy products and cardiovascular mortality in people with and without cardiovascular disease: A prospective cohort study of 0.5 million individuals. Eur. J. Nutr. 2021, 60, 4429–4438. [Google Scholar] [CrossRef]
- Ma, L.; Liu, G.; Ding, M.; Zong, G.; Hu, F.B.; Willett, W.C.; Rimm, E.B.; Manson, J.E.; Sun, Q. Isoflavone intake and the risk of coronary heart disease in us men and women results from 3 prospective cohort studies. Circulation 2020, 141, 1127–1137. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, S.; Joseph, P.; Rangarajan, S.; Islam, S.; Mente, A.; Hystad, P.; Brauer, M.; Kutty, V.R.; Gupta, R.; Wielgosz, A.; et al. Modifiable risk factors, cardiovascular disease, and mortality in 155,722 individuals from 21 high-income, middle-income, and low-income countries (pure): A prospective cohort study. Lancet 2020, 395, 795–808. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.J.L.; Aravkin, A.Y.; Zheng, P.; Abbafati, C.; Abbas, K.M.; Abbasi-Kangevari, M.; Lopez, A.D.; Lim, S.S.; Wang, H.; Lozano, R.; et al. Global burden of 87 risk factors in 204 countries and territories, 1990–2019: A systematic analysis for the global burden of disease study 2019. Lancet 2020, 396, 1223–1249. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2018, 14, 88–98. [Google Scholar] [CrossRef]
- Pan, X.R.; Li, G.W.; Hu, Y.H.; Wang, J.X.; Yang, W.Y.; An, Z.X.; Hu, Z.X.; Lin, J.; Xiao, J.Z.; Cao, H.B.; et al. Effects of diet and exercise in preventing niddm in people with impaired glucose tolerance. Diabetes Care 1997, 20, 537–544. [Google Scholar] [CrossRef]
- Knowler, W.C.; Barrett-Connor, E.; Fowler, S.E.; Hamman, R.F.; Lachin, J.M.; Walker, E.A.; Nathan, D.M.; Diabetes Prevention Program Research Group. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N. Engl. J. Med. 2002, 346, 393–403. [Google Scholar] [CrossRef]
- Brandhorst, S.; Longo, V.D. Dietary restrictions and nutrition in the prevention and treatment of cardiovascular disease. Circ. Res. 2019, 124, 952–965. [Google Scholar] [CrossRef]
- Zhang, Y.B.; Chen, C.; Pan, X.F.; Guo, J.; Li, Y.; Franco, O.H.; Liu, G.; Pan, A. Associations of healthy lifestyle and socioeconomic status with mortality and incident cardiovascular disease: Two prospective cohort studies. BMJ 2021, 373, n604. [Google Scholar] [CrossRef]
- Budhathoki, S.; Sawada, N.; Iwasaki, M.; Yamaji, T.; Goto, A.; Kotemori, A.; Ishihara, J.; Takachi, R.; Charvat, H.; Mizoue, T.; et al. Association of animal and plant protein intake with all-cause and cause-specific mortality in a japanese cohort. JAMA Intern. Med. 2019, 179, 1509–1518. [Google Scholar] [CrossRef]
- Windey, K.; De Preter, V.; Verbeke, K. Relevance of protein fermentation to gut health. Mol. Nutr. Food Res. 2012, 56, 184–196. [Google Scholar] [CrossRef]
- Nachvak, S.M.; Moradi, S.; Anjom-Shoae, J.; Rahmani, J.; Nasiri, M.; Maleki, V.; Sadeghi, O. Soy, soy isoflavones, and protein intake in relation to mortality from all causes, cancers, and cardiovascular diseases: A systematic review and dose-response meta-analysis of prospective cohort studies. J. Acad. Nutr. Diet 2019, 119, 1483–1500.e1417. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Zhang, X.; Li, C.; Jiao, S.; Dong, W. Association between consumption of soy and risk of cardiovascular disease: A meta-analysis of observational studies. Eur. J. Prev. Cardiol. 2017, 24, 735–747. [Google Scholar] [CrossRef]
- Page, M.J.; Mckenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The prisma 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis of observational studies in epidemiology (moose) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.; Shea, B.; O’connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle–Ottawa Scale (Nos) for Assessing the Quality of Non-Randomized Studies in Meta-Analysis. 2014. Available online: https://www.researchgate.net/publication/288802810_The_Newcastle-Ottawa_Scale_NOS_for_Assessing_The_Quality_of_Nonrandomised_Studies_in_Meta-analyses (accessed on 8 February 2023).
- Greenland, S.; Longnecker, M. Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am. J. Epidemiol. 1992, 135, 1301–1309. [Google Scholar] [CrossRef]
- Orsini, N.; Bellocco, R.; Greenland, S. Generalized least squares for trend estimation of summarized dose-response data. Stata J. 2006, 6, 40–57. [Google Scholar] [CrossRef]
- Villegas, R.; Gao, Y.-T.; Yang, G.; Li, H.-L.; Elasy, T.A.; Zheng, W.; Shu, X.O. Legume and soy food intake and the incidence of type 2 diabetes in the shanghai women’s health study. Am. J. Clin. Nutr. 2008, 87, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Eshak, E.S.; Shirai, K.; Dong, J.-Y.; Muraki, I.; Tamakoshi, A.; Iso, H. Soy intake and risk of type 2 diabetes among japanese men and women: Jacc study. Front. Nutr. 2022, 8, 813742. [Google Scholar] [CrossRef]
- Nguyen, C.T.; Pham, N.M.; Do, V.V.; Binns, C.W.; Hoang, V.M.; Dang, D.A.; Lee, A.H. Soyfood and isoflavone intake and risk of type 2 diabetes in vietnamese adults. Eur. J. Clin. Nutr. 2017, 71, 1186–1192. [Google Scholar] [CrossRef]
- Ding, M.; Pan, A.; Manson, J.E.; Willett, W.C.; Malik, V.; Rosner, B.; Giovannucci, E.; Hu, F.B.; Sun, Q. Consumption of soy foods and isoflavones and risk of type 2 diabetes: A pooled analysis of three us cohorts. Eur. J. Clin. Nutr. 2016, 70, 1381–1387. [Google Scholar] [CrossRef] [PubMed]
- Tatsumi, Y.; Morimoto, A.; Deura, K.; Mizuno, S.; Ohno, Y.; Watanabe, S. Effects of soybean product intake on fasting and postload hyperglycemia and type 2 diabetes in japanese men with high body mass index: The saku study. J. Diabetes Investig. 2013, 4, 626–633. [Google Scholar] [CrossRef] [PubMed]
- Woo, H.W.; Kim, M.K.; Lee, Y.H.; Shin, D.H.; Shin, M.H.; Choi, B.Y. Sex-specific associations of habitual intake of soy protein and isoflavones with risk of type 2 diabetes. Clin. Nutr. 2021, 40, 127–136. [Google Scholar] [CrossRef]
- Konishi, K.; Wada, K.; Yamakawa, M.; Goto, Y.; Mizuta, F.; Koda, S.; Uji, T.; Tsuji, M.; Nagata, C. Dietary soy intake is inversely associated with risk of type 2 diabetes in japanese women but not in men. J. Nutr. 2019, 149, 1208–1214. [Google Scholar] [CrossRef]
- Mueller, N.T.; Odegaard, A.O.; Gross, M.D.; Koh, W.-P.; Yu, M.C.; Yuan, J.-M.; Pereira, M. Soy intake and risk of type 2 diabetes mellitus in chinese singaporeans. Eur. J. Nutr. 2012, 51, 1033–1040. [Google Scholar] [CrossRef] [PubMed]
- Nanri, A.; Mizoue, T.; Takahashi, Y.; Kirii, K.; Inoue, M.; Noda, M.; Tsugane, S. Soy product and isoflavone intakes are associated with a lower risk of type 2 diabetes in overweight japanese women. J. Nutr. 2010, 140, 580–586. [Google Scholar] [CrossRef]
- Fang, J.; Foo, S.H.; Fung, C.; Wylie-Rosett, J.; Alderman, M.H. Stroke risk among chinese immigrants in new york city. J. Immigr. Minor. Health 2006, 8, 387–393. [Google Scholar] [CrossRef]
- Guo, J.; Li, W.; Wang, Y.; Chen, T.; Teo, K.; Liu, L.-S.; Yusuf, S.; INTERHEART China Study Investigators. Influence of dietary patterns on the risk of acute myocardial infarction in china population: The interheart china study. Chin. Med. J. 2013, 126, 464–470. [Google Scholar] [CrossRef]
- Ho, S.Y.; Schooling, M.; Hui, L.L.; Mcghee, S.M.; Mak, K.H.; Lam, T.H. Soy consumption and mortality in hong kong: Proxy-reported case-control study of all older adult deaths in 1998. Prev. Med. 2006, 43, 20–26. [Google Scholar] [CrossRef]
- Im, J.; Park, K. Association between soy food and dietary soy isoflavone intake and the risk of cardiovascular disease in women: A prospective cohort study in korea. Nutrients 2021, 13, 1407. [Google Scholar] [CrossRef] [PubMed]
- Katagiri, R.; Sawada, N.; Goto, A.; Yamaji, T.; Iwasaki, M.; Noda, M.; Iso, H.; Tsugane, S.; Japan Public Health Center-Based Prospective Study Group. Association of soy and fermented soy product intake with total and cause specific mortality: Prospective cohort study. BMJ—Br. Med. J. 2020, 368, m34. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Sasazuki, S.; Shimazu, T.; Sawada, N.; Yamaji, T.; Iwasaki, M.; Mizoue, T.; Tsugane, S. Association of dietary diversity with total mortality and major causes of mortality in the japanese population: Jphc study. Eur. J. Clin. Nutr. 2020, 74, 54–66. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, Y.; Iso, H.; Ishihara, J.; Okada, K.; Inoue, M.; Tsugane, S. Association of dietary intake of soy, beans, and isoflavones with risk of cerebral and myocardial infarctions in japanese populations—The japan public health center-based (jphc) study cohort i. Circulation 2007, 116, 2553–2562. [Google Scholar] [CrossRef]
- Liang, W.; Lee, A.H.; Binns, C.W.; Huang, R.; Hu, D.; Shao, H. Soy consumption reduces risk of ischemic stroke: A case-control study in southern china. Neuroepidemiology 2009, 33, 111–116. [Google Scholar] [CrossRef]
- Nagata, C.; Takatsuka, N.; Shimizu, H. Soy and fish oil intake and mortality in a japanese community. Am. J. Epidemiol. 2002, 156, 824–831. [Google Scholar] [CrossRef]
- Nagata, C.; Wada, K.; Tamura, T.; Konishi, K.; Goto, Y.; Koda, S.; Kawachi, T.; Tsuji, M.; Nakamura, K. Dietary soy and natto intake and cardiovascular disease mortality in japanese adults: The takayama study. Am. J. Clin. Nutr. 2017, 105, 426–431. [Google Scholar] [CrossRef]
- Nguyen, H.N.; Miyagawa, N.; Miura, K.; Okuda, B.; Yoshita, K.; Arai, Y.; Nakagawa, H.; Sakata, K.; Ojima, T.; Kadota, A.; et al. Dietary tofu intake and long-term risk of death from stroke in a general population. Clin. Nutr. 2018, 37, 182–188. [Google Scholar] [CrossRef]
- Nouri, F.; Haghighatdoost, F.; Mohammadifard, N.; Mansourian, M.; Sadeghi, M.; Roohafza, H.; Khani, A.; Sarrafzadegan, N. The longitudinal association between soybean and non-soybean legumes intakes and risk of cardiovascular disease: Isfahan cohort study. Br. Food J. 2021, 123, 2864–2879. [Google Scholar] [CrossRef]
- Nozue, M.; Shimazu, T.; Charvat, H.; Mori, N.; Mutoh, M.; Sawada, N.; Iwasaki, M.; Yamaji, T.; Inoue, M.; Kokubo, Y.; et al. Fermented soy products intake and risk of cardiovascular disease and total cancer incidence: The japan public health center-based prospective study. Eur. J. Clin. Nutr. 2021, 75, 954–968. [Google Scholar] [CrossRef]
- Talaei, M.; Koh, W.P.; Van Dam, R.M.; Yuan, J.M.; Pan, A. Dietary soy intake is not associated with risk of cardiovascular disease mortality in singapore chinese adults. J. Nutr. 2014, 144, 921–928. [Google Scholar] [CrossRef]
- Yamasaki, K.; Kayaba, K.; Ishikawa, S. Soy and soy products intake, all-cause mortality, and cause-specific mortality in japan: The jichi medical school cohort study. Asia Pac. J. Public Health 2015, 27, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Shu, X.O.; Li, H.; Yang, G.; Cai, Q.; Xiang, Y.B.; Ji, B.T.; Franke, A.A.; Gao, Y.T.; Zheng, W.; et al. Dietary isoflavones, urinary isoflavonoids, and risk of ischemic stroke in women. Am. J. Clin. Nutr. 2015, 102, 680–686. [Google Scholar] [CrossRef] [PubMed]
- Sasazuki, S.; Group, F.H.S. Case-control study of nonfatal myocardial infarction in relation to selected foods in japanese men and women. Jpn. Circ. J. 2001, 65, 200–206. [Google Scholar] [CrossRef]
- Okamoto, K.; Horisawa, R. Soy products and risk of an aneurysmal rupture subarachnoid hemorrhage in japan. Eur. J. Cardiovasc. Prev. Rehabil. 2006, 13, 284–287. [Google Scholar] [CrossRef]
- Zhang, X.; Shu, X.O.; Gao, Y.-T.; Yang, G.; Li, Q.; Li, H.; Jin, F.; Zheng, W. Soy food consumption is associated with lower risk of coronary heart disease in chinese women. J. Nutr. 2003, 133, 2874–2878. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Ruan, W.; Peng, Y.; Wang, D. Soy and the risk of type 2 diabetes mellitus: A systematic review and meta-analysis of observational studies. Diabetes Res. Clin. Pract. 2018, 137, 190–199. [Google Scholar] [CrossRef]
- Tang, J.; Wan, Y.; Zhao, M.; Zhong, H.; Zheng, J.S.; Feng, F. Legume and soy intake and risk of type 2 diabetes: A systematic review and meta-analysis of prospective cohort studies. Am. J. Clin. Nutr. 2020, 111, 677–688. [Google Scholar] [CrossRef]
- Jiang, H.; Feng, J.; Du, Z.; Zhen, H.; Lin, M.; Jia, S.; Li, T.; Huang, X.; Ostenson, C.G.; Chen, Z. Oral administration of soybean peptide vglycin normalizes fasting glucose and restores impaired pancreatic function in type 2 diabetic wistar rats. J. Nutr. Biochem. 2014, 25, 954–963. [Google Scholar] [CrossRef] [PubMed]
- Tachibana, N.; Yamashita, Y.; Nagata, M.; Wanezaki, S.; Ashida, H.; Horio, F.; Kohno, M. Soy beta-conglycinin improves glucose uptake in skeletal muscle and ameliorates hepatic insulin resistance in goto-kakizaki rats. Nutr. Res. 2014, 34, 160–167. [Google Scholar] [CrossRef]
- Clerici, C.; Nardi, E.; Battezzati, P.M.; Asciutti, S.; Castellani, D.; Corazzi, N.; Giuliano, V.; Gizzi, S.; Perriello, G.; Di Matteo, G.; et al. Novel soy germ pasta improves endothelial function, blood pressure, and oxidative stress in patients with type 2 diabetes. Diabetes Care 2011, 34, 1946–1948. [Google Scholar] [CrossRef] [PubMed]
- Das, D.; Sarkar, S.; Bordoloi, J.; Wann, S.B.; Kalita, J.; Manna, P. Daidzein, its effects on impaired glucose and lipid metabolism and vascular inflammation associated with type 2 diabetes. Biofactors 2018, 44, 407–417. [Google Scholar] [CrossRef]
- Cheong, S.H.; Furuhashi, K.; Ito, K.; Nagaoka, M.; Yonezawa, T.; Miura, Y.; Yagasaki, K. Daidzein promotes glucose uptake through glucose transporter 4 translocation to plasma membrane in l6 myocytes and improves glucose homeostasis in type 2 diabetic model mice. J. Nutr. Biochem. 2014, 25, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Sahebkar, A. Fat lowers fat: Purified phospholipids as emerging therapies for dyslipidemia. Biochim. Biophys. Acta 2013, 1831, 887–893. [Google Scholar] [CrossRef]
- Bonaca, M.P.; Morrow, D.A.; Bergmark, B.A.; Berg, D.D.; Lima, J.A.C.; Hoffmann, U.; Kato, Y.; Lu, M.T.; Kuder, J.; Murphy, S.A.; et al. Randomized, placebo-controlled phase 2b study to evaluate the safety and efficacy of recombinant human lecithin cholesterol acyltransferase in acute st-segment-elevation myocardial infarction: Results of real-timi 63b. Circulation 2022, 146, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Imai, S. Soybean and processed soy foods ingredients, and their role in cardiometabolic risk prevention. Recent Pat. Food Nutr. Agric. 2015, 7, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Zhang, R.; Ji, Y.C.; Hao, J.Y.; Ma, W.W.; Chen, X.D.; Xiao, R.; Yu, H.L. Soy milk powder supplemented with phytosterol esters reduced serum cholesterol level in hypercholesterolemia independently of lipoprotein e genotype: A random clinical placebo-controlled trial. Nutr. Res. 2016, 36, 879–884. [Google Scholar] [CrossRef]
- Butteiger, D.N.; Hibberd, A.A.; Mcgraw, N.J.; Napawan, N.; Hall-Porter, J.M.; Krul, E.S. Soy protein compared with milk protein in a western diet increases gut microbial diversity and reduces serum lipids in golden syrian hamsters. J. Nutr. 2016, 146, 697–705. [Google Scholar] [CrossRef]
- Lukaczer, D.; Liska, D.J.; Lerman, R.H.; Darland, G.; Schiltz, B.; Tripp, M.; Bland, J.S. Effect of a low glycemic index diet with soy protein and phytosterols on cvd risk factors in postmenopausal women. Nutrition 2006, 22, 104–113. [Google Scholar] [CrossRef]
- Li, N.; Wu, X.; Zhuang, W.; Xia, L.; Chen, Y.; Zhao, R.; Yi, M.; Wan, Q.; Du, L.; Zhou, Y. Soy and isoflavone consumption and multiple health outcomes: Umbrella review of systematic reviews and meta-analyses of observational studies and randomized trials in humans. Mol. Nutr. Food Res. 2020, 64, e1900751. [Google Scholar] [CrossRef]
- Messina, M.; Duncan, A.; Messina, V.; Lynch, H.; Kiel, J.; Erdman, J.W., Jr. The health effects of soy: A reference guide for health professionals. Front. Nutr. 2022, 9, 970364. [Google Scholar] [CrossRef]
- Sun, L.; Zong, G.; Li, H.; Lin, X. Fatty acids and cardiometabolic health: A review of studies in chinese populations. Eur. J. Clin. Nutr. 2021, 75, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, S.M.; Jalilpiran, Y.; Karimi, E.; Aune, D.; Larijani, B.; Mozaffarian, D.; Willett, W.C.; Esmaillzadeh, A. Dietary intake of linoleic acid, its concentrations, and the risk of type 2 diabetes: A systematic review and dose-response meta-analysis of prospective cohort studies. Diabetes Care 2021, 44, 2173–2181. [Google Scholar] [CrossRef] [PubMed]
- De Goede, J.; Verschuren, W.M.; Boer, J.M.; Kromhout, D.; Geleijnse, J.M. Gender-specific associations of marine n-3 fatty acids and fish consumption with 10-year incidence of stroke. PLoS ONE 2012, 7, e33866. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Zhang, F.; Ding, X.; Wu, G.; Lam, Y.Y.; Wang, X.; Fu, H.; Xue, X.; Lu, C.; Ma, J.; et al. Gut bacteria selectively promoted by dietary fibers alleviate type 2 diabetes. Science 2018, 359, 1151–1156. [Google Scholar] [CrossRef]
- Tanaka, S.; Yoshimura, Y.; Kamada, C.; Tanaka, S.; Horikawa, C.; Okumura, R.; Ito, H.; Ohashi, Y.; Akanuma, Y.; Yamada, N.; et al. Intakes of dietary fiber, vegetables, and fruits and incidence of cardiovascular disease in japanese patients with type 2 diabetes. Diabetes Care 2013, 36, 3916–3922. [Google Scholar] [CrossRef]
- Threapleton, D.E.; Burley, V.J.; Greenwood, D.C.; Cade, J.E. Dietary fibre intake and risk of ischaemic and haemorrhagic stroke in the uk women’s cohort study. Eur. J. Clin. Nutr. 2015, 69, 467–474. [Google Scholar] [CrossRef]
- Luiking, Y.; Deutz, N.; Jäkel, M.; Soeters, P. Casein and soy protein meals differentially affect whole-body and splanchnic protein metabolism in healthy humans. J. Nutr. 2005, 135, 1080–1087. [Google Scholar] [CrossRef] [PubMed]
- Lynch, C.; Adams, S. Branched-chain amino acids in metabolic signalling and insulin resistance. Nat. Rev. Endocrinol. 2014, 10, 723–736. [Google Scholar] [CrossRef] [PubMed]
- Nagata, C. Soy intake and chronic disease risk: Findings from prospective cohort studies in japan. Eur. J. Clin. Nutr. 2021, 75, 890–901. [Google Scholar] [CrossRef] [PubMed]
- Vedtofte, M.S.; Jakobsen, M.U.; Lauritzen, L.; Heitmann, B.L. Dietary alpha-linolenic acid, linoleic acid, and n-3 long-chain pufa and risk of ischemic heart disease. Am. J. Clin. Nutr. 2011, 94, 1097–1103. [Google Scholar] [CrossRef]
- Soni, K.; Frew, R.; Kebede, B. A review of conventional and rapid analytical techniques coupled with multivariate analysis for origin traceability of soybean. Crit. Rev. Food Sci. Nutr. 2023, 3, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Lukaszuk, J.M.; Luebbers, P.; Gordon, B.A. Preliminary study: Soy milk as effective as skim milk in promoting weight loss. J. Am. Diet. Assoc 2007, 107, 1811–1814. [Google Scholar] [CrossRef]
- Menotti, A.; Kromhout, D.; Blackburn, H.; Fidanza, F.; Buzina, R.; Nissinen, A. Food intake patterns and 25-year mortality from coronary heart disease: Cross-cultural correlations in the seven countries study. Eur. J. Epidemiol. 1999, 15, 507–515. [Google Scholar] [CrossRef]
- Nagura, J.; Iso, H.; Watanabe, Y.; Maruyama, K.; Date, C.; Toyoshima, H.; Yamamoto, A.; Kikuchi, S.; Koizumi, A.; Kondo, T.; et al. Fruit, vegetable and bean intake and mortality from cardiovascular disease among japanese men and women: The jacc study. Br. J. Nutr. 2009, 102, 285–292. [Google Scholar] [CrossRef]
- Nanri, A.; Mizoue, T.; Shimazu, T.; Ishihara, J.; Takachi, R.; Node, M.; So, H.; Sasazuki, S.; Sawada, N.; Tsugane, S.; et al. Dietary patterns and all-cause, cancer, and cardiovascular disease mortality in japanese men and women: The japan public health center-based prospective study. PLoS ONE 2017, 12, e0174848. [Google Scholar] [CrossRef] [PubMed]
- Ozawa, M.; Yoshida, D.; Hata, J.; Ohara, T.; Mukai, N.; Shibata, M.; Uchida, K.; Nagata, M.; Kitazono, T.; Kiyohara, Y.; et al. Dietary protein intake and stroke risk in a general japanese population: The hisayama study. Stroke 2017, 48, 1478–1486. [Google Scholar] [CrossRef] [PubMed]
- Preis, S.R.; Stampfer, M.J.; Spiegelman, D.; Willett, W.C.; Rimm, E.B. Dietary protein and risk of ischemic heart disease in middle-aged men. Am. J. Clin. Nutr. 2010, 92, 1265–1272. [Google Scholar] [CrossRef] [PubMed]
- Tsugane, S.; Sawada, N. The jphc study: Design and some findings on the typical japanese diet. Jpn. J. Clin. Oncol. 2014, 44, 777–782. [Google Scholar] [CrossRef]
- Yu, D.; Zhang, X.; Xiang, Y.B.; Yang, G.; Li, H.; Fazio, S.; Linton, M.; Cai, Q.; Zheng, W.; Gao, Y.T.; et al. Association of soy food intake with risk and biomarkers of coronary heart disease in chinese men. Int. J. Cardiol. 2014, 172, e285–e287. [Google Scholar] [CrossRef]
- Morimoto, Y.; Steinbrecher, A.; Kolonel, L.N.; Maskarinec, G. Soy consumption is not protective against diabetes in hawaii: The multiethnic cohort. Eur. J. Clin. Nutr. 2011, 65, 279–282. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Shu, X.O.; Jin, F.; Elasy, T.; Li, H.L.; Li, Q.; Huang, F.; Zhang, X.L.; Gao, Y.T.; Zheng, W. Soyfood consumption and risk of glycosuria: A cross-sectional study within the shanghai women’s health study. Eur. J. Clin. Nutr. 2004, 58, 615–620. [Google Scholar] [CrossRef]
- Kokubo, Y.; Watanabe, M.; Furukawa, Y.; Miyamoto, Y. Moderate miso soup consumption may reduce the risk of cardiovascular risk factors in japanese prospective cohort study: The suita study. Ann. Nutr. Metab. 2013, 63, 1347. [Google Scholar]
- Tavani, A.; Spertini, L.; Bosetti, C.; Parpinel, M.; Gnagnarella, P.; Bravi, F.; Peterson, J.; Dwyer, J.; Lagiou, P.; Negri, E.; et al. Intake of specific flavonoids and risk of acute myocardial infarction in italy. Public Health Nutr. 2006, 9, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Mink, P.J.; Scrafford, C.G.; Barraj, L.M.; Harnack, L.; Hong, C.P.; Nettleton, J.A.; Jacobs, D.R., Jr. Flavonoid intake and cardiovascular disease mortality: A prospective study in postmenopausal women. Am. J. Clin. Nutr. 2007, 85, 895–909. [Google Scholar] [CrossRef] [PubMed]
- Mccullough, M.L.; Peterson, J.J.; Patel, R.; Jacques, P.F.; Shah, R.; Dwyer, J.T. Flavonoid intake and cardiovascular disease mortality in a prospective cohort of us adults. Am. J. Clin. Nutr. 2012, 95, 454–464. [Google Scholar] [CrossRef]
- Loke, A.Y.; Chan, K.N. Dietary habits of patients with coronary atherosclerosis: Case-control study. J. Adv. Nurs. 2005, 52, 159–169. [Google Scholar] [CrossRef]

| First Author (year) | Cohort Name | Country | Sample Size | Follow-up (Median, y) | Dietary Assessment | Exposure Assessment | Outcome (n) | Comparison | Adjustments | NOS Score |
|---|---|---|---|---|---|---|---|---|---|---|
| Prospective cohort studies | ||||||||||
| Yamasaki (2015) [53] | JMS | Japan | 11,066 | 11.8 | Self-administered, FFQ, 30-item, and validated | Soy | CVDs deaths (198) | Almost daily vs. 1–2 times/week | a and b | 8 |
| Yamasaki (2015) [53] | JMS | Japan | 11,066 | 11.8 | Self-administered, FFQ, 30-item, and validated | Soy products | CVDs deaths (217) | Almost daily vs. 1–4 times per week | a and b | 8 |
| Nagata (2016) [48] | Takayama study | Japan | 29,079 | 16 | Self-administered, FFQ-169, and validated | Natto | CVDs deaths (1678), stroke (677), and CHD (308) | 7.3 vs. 0 g/d | a, b, and c | 9 |
| Zhang (2003) [57] | SWHS | China | 64,915 | 2.5 | Interview, FFQ, and validated | Total soy protein | CHD (62) | Q4 vs. Q1 ≥ 11.19 vs. <4.5 g/d | a, b, and c | 8 |
| Yan (2021) [31] | JACC Study | Japan | 21,925 | 5 | Self-administered, FFQ-40, and validated | Tofu | T2D (593) | Almost daily vs. <3 times/week | a, b, and c | 9 |
| Yan (2021) [31] | JACC Study | Japan | 21,925 | 5 | Self-administered, FFQ-40, and validated | Miso soup | T2D (593) | ≥3 bowls/day vs. <1 bowl/day | a, b, and c | 9 |
| Woo (2020) [35] | MR Cohort | Korea | 8269 | 6.18 | Interview, FFQ-106, and validated | Soy protein | T2D (531) | Q4 vs. Q1 | a, b, and c | 9 |
| Wang-a (2021) [12] | CKB study | China | 487,034 | 8.2 | Interview, FFQ, and validated | Soy | CVDs deaths (12582), CHD (3764), and stroke (5916) | ≥4 days per week vs. never or rarely | a, b, and c | 7 |
| Wang-b (2021) [12] | CKB study | China | 22,923 | 7.8 | Interview, FFQ, and validated | soy | CVDs deaths (2860), CHD (1123), and stroke (1473) | ≥4 days per week vs. never or rarely | a, b, and c | 7 |
| Villegas (2008) [30] | SWHS | China | 64,191 | 4.6 | Interview, FFQ-77, and validated | Soybeans | T2D (1605) | Q5 vs. Q1 32 vs. 2.8 g/d | a, b, and c | 9 |
| Tatsumi (2013) [34] | The Saku Study | Japan | 3039 | 4 | Self-administered, FFQ, and non-validated | soybean products | T2D (204) | ≥4 times/week vs. 0–1 time/week | a, b, and c | 7 |
| Talaei (2014) [52] | SCHS | China | 60,298 | 14.7 | Interview, FFQ-165, and validated | Tofu equivalents | CVDs deaths (4780), CHD (2697), and stroke (1298) | Q4 vs. Q1 197 vs. 42.8 g/d | a, b, and c | 9 |
| Nozue (2020) [51] | JPHC I, II | Japan | 79,648 | 13.3 | Self-administered, FFQ-138, and validated | Total soy products | CVDs events (4427), stroke (3743), and CHD (684) | Q4 vs. Q1 140.9 vs. 33.6 g/d | a, b, and c | 8 |
| Nouri (2021) [50] | ICS | Iran | 5432 | 13 | Interview, FFQ-48, and validated | Soybeans | CVDs events (751) | ≥1 times /week vs. <1 times/week | a and b | 7 |
| Ma (2020) [13] | NHS, NHSII, HPFS | USA | 210,700 | / | Self-administered, FFQ-130, and validated | Tofu | CHD events (8359) | ≥1 serving/week vs. <1 serving/month | a, b, and c | 6 |
| Ding (2016) [33] | NHS, NHSII, HPFS | USA | 163,457 | / | Self-administered, FFQ-116, and validated | Total soy food | T2D (9185) | ≥1 serving/week vs. non-consumer | a, b, and c | 6 |
| Nanri (2010) [38] | JPHC I, II | Japan | 59,791 | 10 | Self-administered, FFQ-147, and validated | Soy products | T2D (1114) | Q5 vs. Q1 ≥ 186 vs. 29 g/d | a, b, and c | 9 |
| Yu (2015) [54] | SWHS | China | 66,832 | 10 | Interview, FFQ-77, and validated | Total soy foods (dry weight) | IS (3110) | Q5 vs. Q1 33.5 vs. 6.6 g/d | a, b, and c | 9 |
| Im (2021) [42] | KGES | Korea | 4713 | 7.4 | Interview, FFQ, and validated | Total soy foods | CVDs events (282) | Q4 vs. Q1 18.03 vs. 5.27 servings/week | a, b, and c | 8 |
| Katagiri (2019) [43] | JPHC | Japan | 92,915 | 14.8 | Self-administered, FFQ-138, and validated | Total soy products | CVDs deaths (3326) and stroke (1326) | Q5 vs. Q1 178 vs. 37 g/d | a, b, and c | 9 |
| Kobayashi (2019) [44] | JPHC | Japan | 79,904 | 14.9 | Self-administered, FFQ-138, and validated | Soy dietary diversity | CVDs deaths (2942) | Q5 vs. Q1 3.5 vs. 1.1 of food items/day | a, b, and c | 9 |
| Kokubo (2007) [45] | JPHC I | Japan | 40,462 | 12.5 | Self-administered, FFQ-44, and NA | Soy | stroke (1230) and MI (308) | Highest vs. lowest ≥ 5 vs. 0–2 days/week | a, b, and c | 8 |
| Konishi (2019) [36] | Takayama study | Japan | 13,521 | 10 | Self-administered, FFQ-169, and validated | Total soy foods | T2D (438) | Q3 vs. Q1 141.2 vs. 50.7 g/d | a, b, and c | 8 |
| Nguyen (2018) [49] | NIP-PON DATA | Japan | 9244 | 24 | Weigh and record all foods and beverages on 3 consecutive days | Tofu | stroke (417) | Q4 vs. Q1 41.5 vs. 0.8 g/1000 kcal | a, b, and c | 8 |
| Mueller (2011) [37] | SCHS | China | 43,176 | 5.7 | Interview, FFQ-165, and validated | Unsweetened soy | T2D (2252) | ≥5/week vs. none | a, b, and c | 8 |
| Case-control studies | Baseline years a | |||||||||
| Guo (2013) [40] | ICS | China | 3547 | 1999.2–2003.3 | Self-reported, FFQ-19, and validated | Tofu | CHD events (1312) | >3 vs. <0.75 times/week | a and b | 6 |
| Liang (2009) [46] | / | China | 838 | 2007–2008 | Interview, FFQ-125, and validated | Total soy foods | IS (374) | ≥300 vs. <50 g | a, b, and c | 7 |
| Okamoto (2006) [56] | / | Japan | 411 | 1992.4–1997.3 | Interview, FFQ, and NA | Soy products | SAH (201) | Q4 vs. Q1 | a and b | 8 |
| Fang (2006) [39] | / | USA | 391 | 2000.9–2003.6 | Interview, FFQ-49, and validated | Soybean | stroke (187) | ≥3 vs. <3 times/week | a | 5 |
| Ho (2006) [41] | / | China | 32,462 | 1997.12–1999.01 | Proxy report, FFQ, and NA | Soy | stroke (2160) and fatal IHD (2016) | 4+/week vs. <1/mon | a, b, and c | 6 |
| Sasazuki (2001) [55] | FHS | Japan | 1846 | 1996.9–1998.9 | Interview, FFQ-23, and NA | Tofu | AMI (632) | 4+/week vs. <2/week | a and b | 8 |
| Sasazuki (2001) [55] | FHS | Japan | 1846 | 1996.9–1998.9 | Interview, FFQ-23, and NA | Miso soup | AMI (632) | 2+/week vs. <1/week | b and c | 8 |
| Nguyen (2017) [32] | / | Vietnam | 1198 | 2013.8–2015.10 | Interview, FFQ-128, and validated | Total soy foods | T2D (599) | >133.9 vs. = <44.1 g/d | a, b, and c | 7 |
| T2D Subgroup | No. | TRR (95% CI) | Ph | I2 (%) | Pd |
|---|---|---|---|---|---|
| Non-fermented soy food | 8 | 0.90 (0.81, 1.00) | <0.001 | 77 | 0.05 |
| Tofu | 7 | 0.91 (0.81,1.02) | 0.09 | 46 | 0.1 |
| Soy milk | 6 | 0.85 (0.68,1.05) | <0.001 | 90 | 0.12 |
| Boiled beans | 2 | 0.97 (0.74,1.26) | 0.28 | 13 | 0.79 |
| Others | 1 | 1.04 (0.86,1.26) | 0.38 | 0 | 0.66 |
| Fermented soy food | 2 | 0.98 (0.71,1.34) | 0.68 | 0 | 0.89 |
| Miso soup | 2 | 0.98 (0.71,1.34) | 0.68 | 0 | 0.89 |
| Other soy food | 5 | 0.75 (0.61,0.93) | <0.001 | 82 | 0.009 |
| Subgroup total | 0.87 (0.80,0.95) | <0.001 | 75 | 0.002 | |
| CVDs Subgroup | |||||
| Non-fermented soy food | 16 | 0.92 (0.84,1.00) | <0.001 | 69 | 0.06 |
| Tofu | 14 | 0.89 (0.80,0.99) | <0.001 | 65 | 0.03 |
| Soy milk | 6 | 0.81 (0.59,1.12) | <0.001 | 77 | 0.2 |
| Fermented soy food | 10 | 0.86 (0.79,0.93) | 0.08 | 41 | <0.001 |
| Natto | 5 | 0.82 (0.75,0.90) | 0.2 | 34 | <0.001 |
| Miso soup | 8 | 0.93 (0.84,1.03) | 0.12 | 39 | 0.17 |
| Subgroup total | 0.88 (0.83,0.93) | <0.001 | 66 | <0.001 | |
| CHD Subgroup | |||||
| Non-fermented soy food | 9 | 0.81 (0.70,0.95) | 0.002 | 68 | 0.009 |
| Tofu | 7 | 0.81 (0.67,0.98) | 0.003 | 70 | 0.03 |
| Soy milk | 3 | 0.80 (0.54,1.20) | 0.10 | 57 | 0.28 |
| Fermented soy food | 7 | 0.85 (0.73,1.00) | 0.65 | 0 | 0.05 |
| Natto | 3 | 0.89 (0.68,1.15) | 0.26 | 26 | 0.36 |
| Miso soup | 6 | 0.96 (0.81,1.14) | 0.78 | 0 | 0.63 |
| Subgroup total | 0.87 (0.80,0.94) | 0.09 | 28 | <0.001 | |
| Stroke Subgroup | |||||
| Non-fermented soy food | 9 | 1.05 (0.97,1.13) | 0.19 | 29 | 0.25 |
| Tofu | 7 | 1.01 (0.90,1.13) | 0.15 | 36 | 0.89 |
| Soy milk | 2 | 0.50 (0.08,3.09) | <0.001 | 91 | 0.45 |
| Fermented soy food | 7 | 0.88 (0.78,1.00) | 0.007 | 66 | 0.04 |
| Natto | 5 | 0.81 (0.72,0.91) | 0.19 | 34 | <0.001 |
| Miso soup | 6 | 0.91 (0.79,1.06) | 0.04 | 57 | 0.23 |
| Subgroup total | 0.90 (0.84,0.97) | <0.001 | 69 | 0.005 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zuo, X.; Zhao, R.; Wu, M.; Wan, Q.; Li, T. Soy Consumption and the Risk of Type 2 Diabetes and Cardiovascular Diseases: A Systematic Review and Meta-Analysis. Nutrients 2023, 15, 1358. https://doi.org/10.3390/nu15061358
Zuo X, Zhao R, Wu M, Wan Q, Li T. Soy Consumption and the Risk of Type 2 Diabetes and Cardiovascular Diseases: A Systematic Review and Meta-Analysis. Nutrients. 2023; 15(6):1358. https://doi.org/10.3390/nu15061358
Chicago/Turabian StyleZuo, Xinrong, Rui Zhao, Minming Wu, Qianyi Wan, and Tao Li. 2023. "Soy Consumption and the Risk of Type 2 Diabetes and Cardiovascular Diseases: A Systematic Review and Meta-Analysis" Nutrients 15, no. 6: 1358. https://doi.org/10.3390/nu15061358
APA StyleZuo, X., Zhao, R., Wu, M., Wan, Q., & Li, T. (2023). Soy Consumption and the Risk of Type 2 Diabetes and Cardiovascular Diseases: A Systematic Review and Meta-Analysis. Nutrients, 15(6), 1358. https://doi.org/10.3390/nu15061358







