The Nature of Available Choices Affects the Intake and Meal Patterns of Rats Offered a Palatable Cafeteria-Style Diet
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
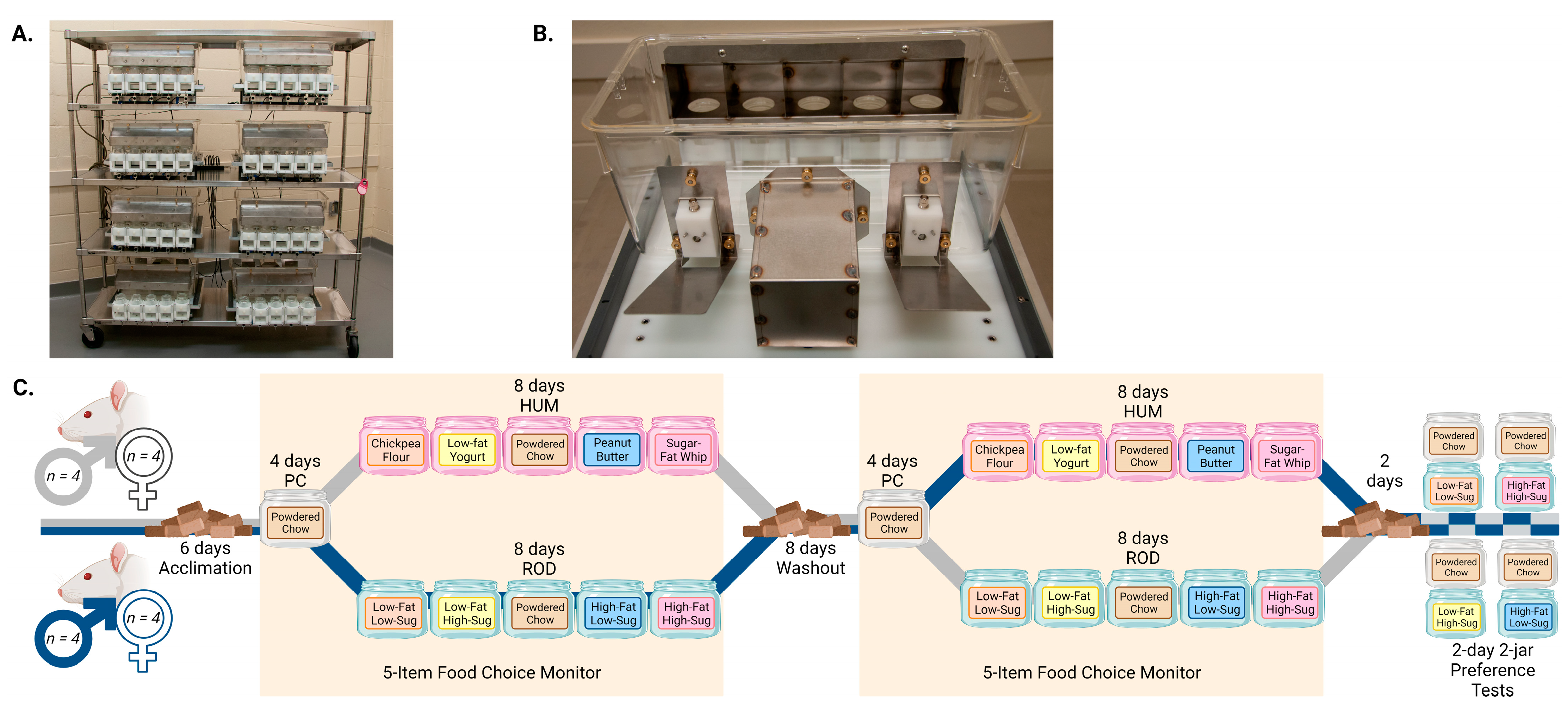
2.2. Apparatus
2.3. Foods
2.4. Experimental Design
2.5. Two-Jar Tests
2.6. FCM Output and Statistical Analysis
3. Results
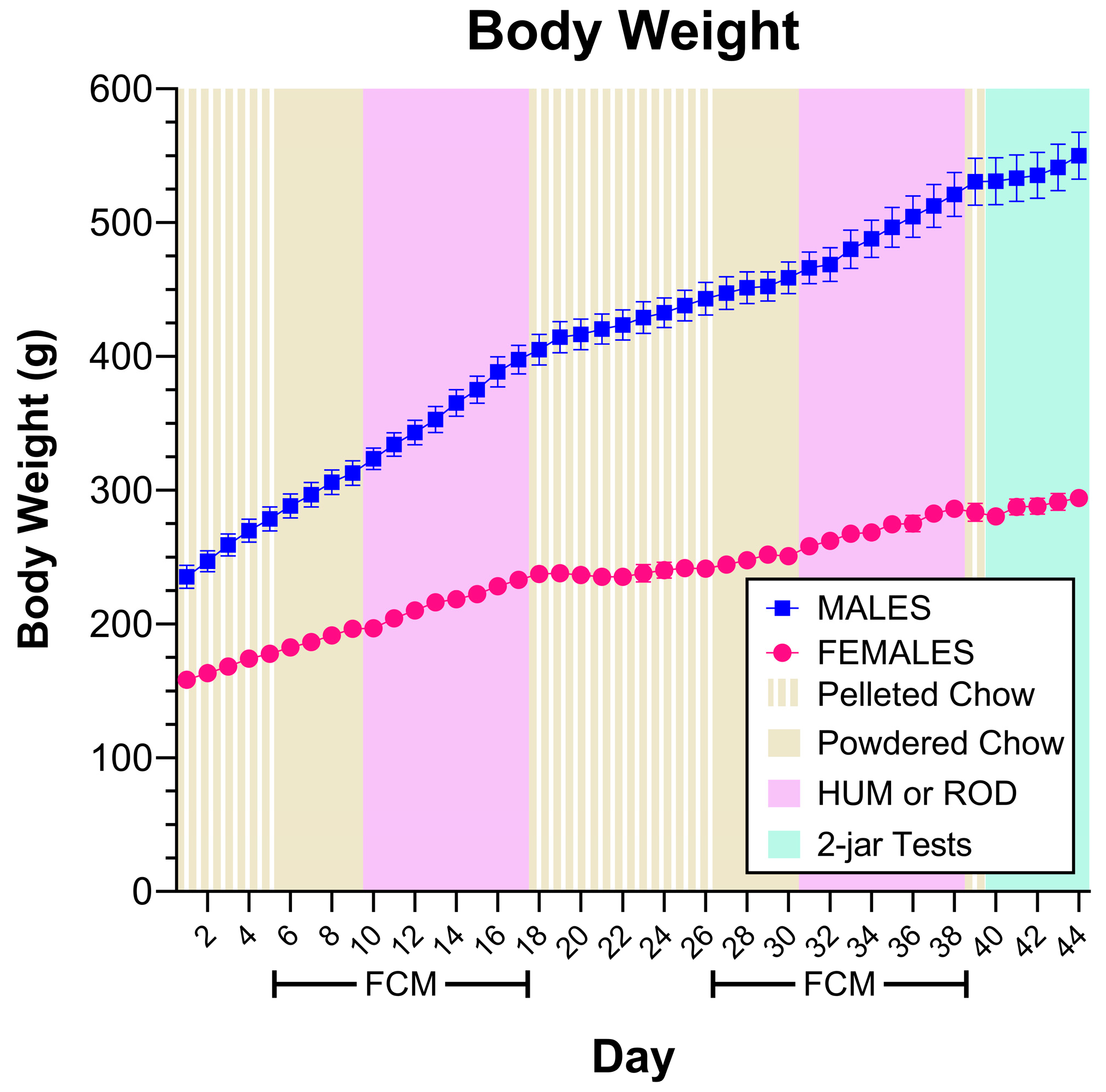
3.1. Body Weight
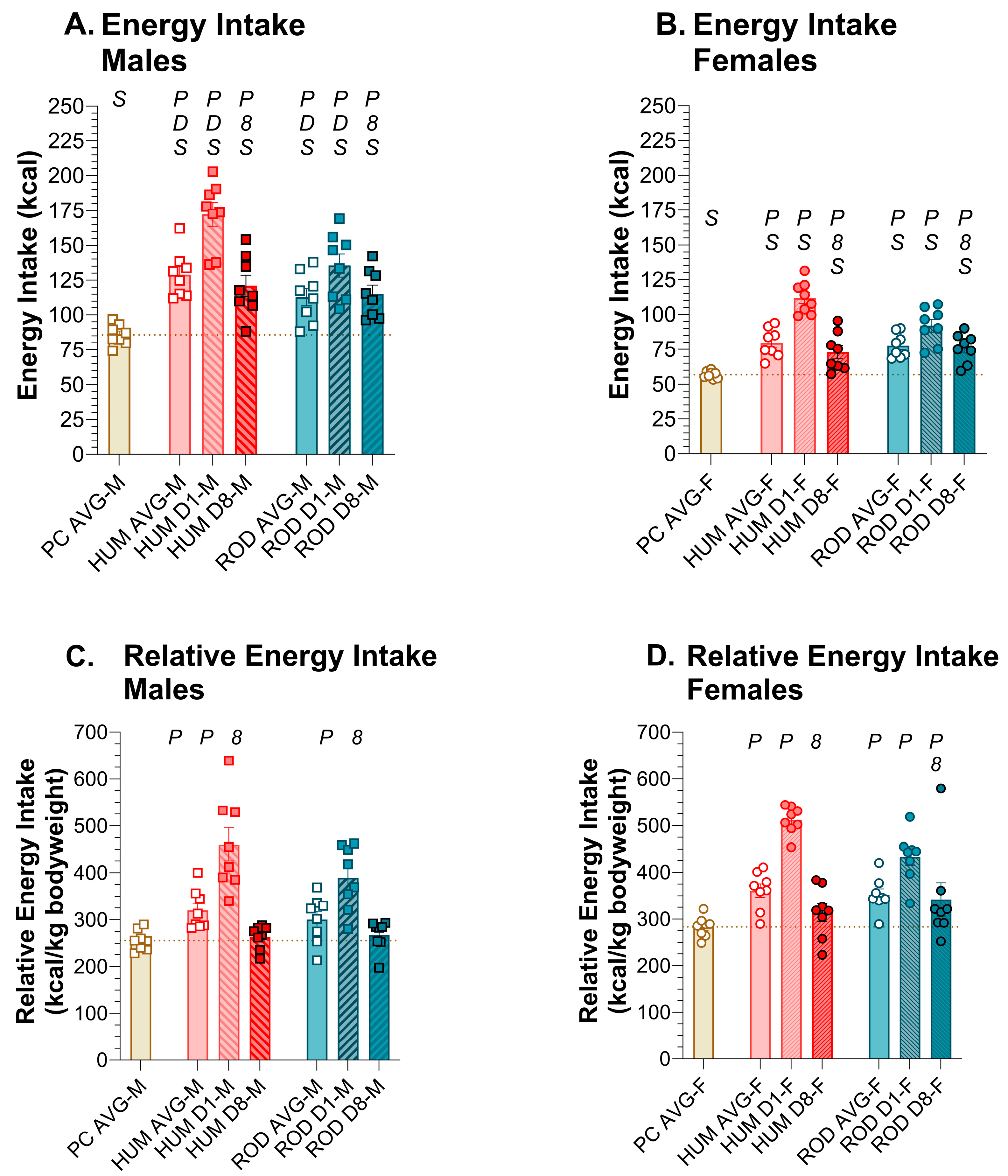
3.2. Energy Intake
3.3. Water Intake
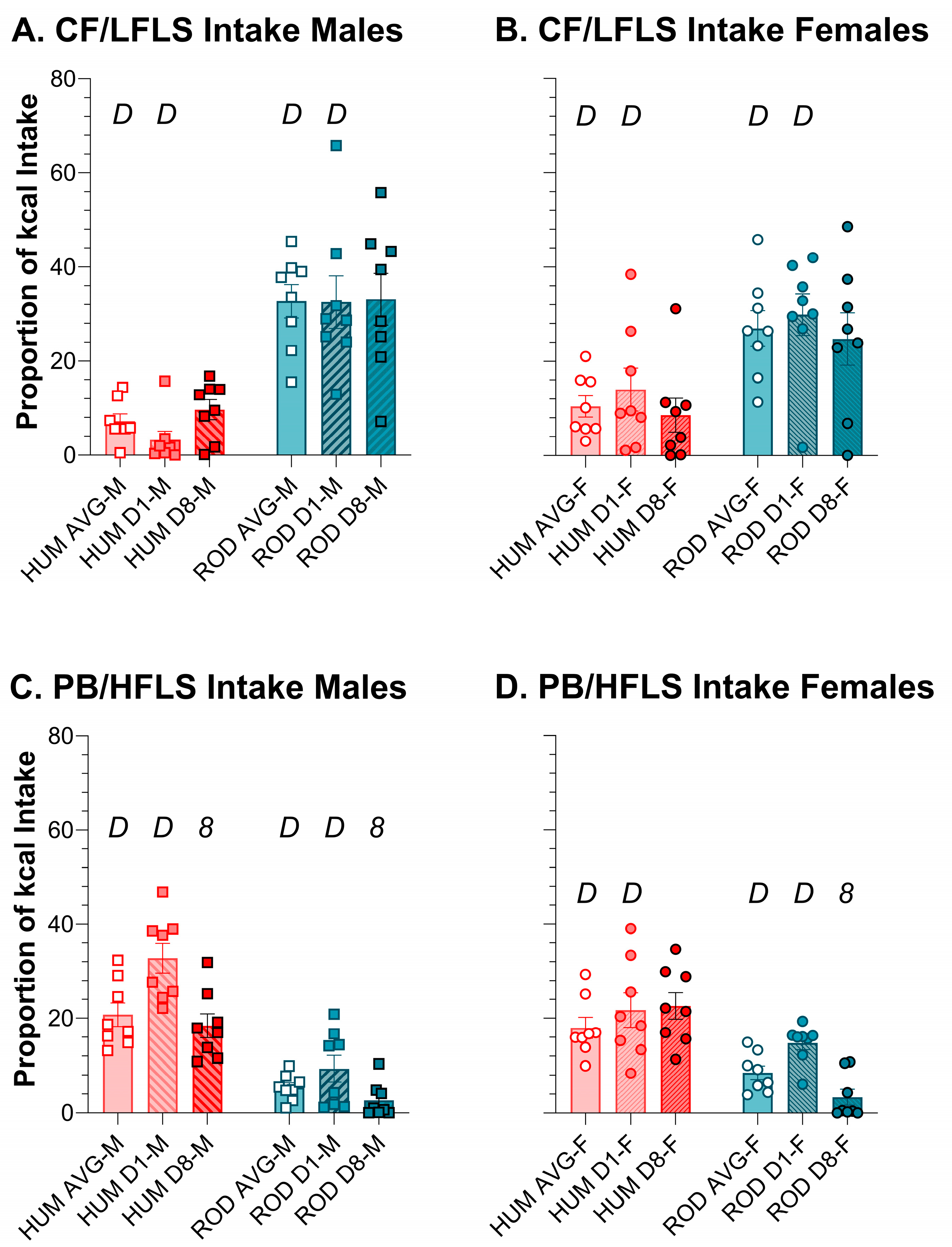
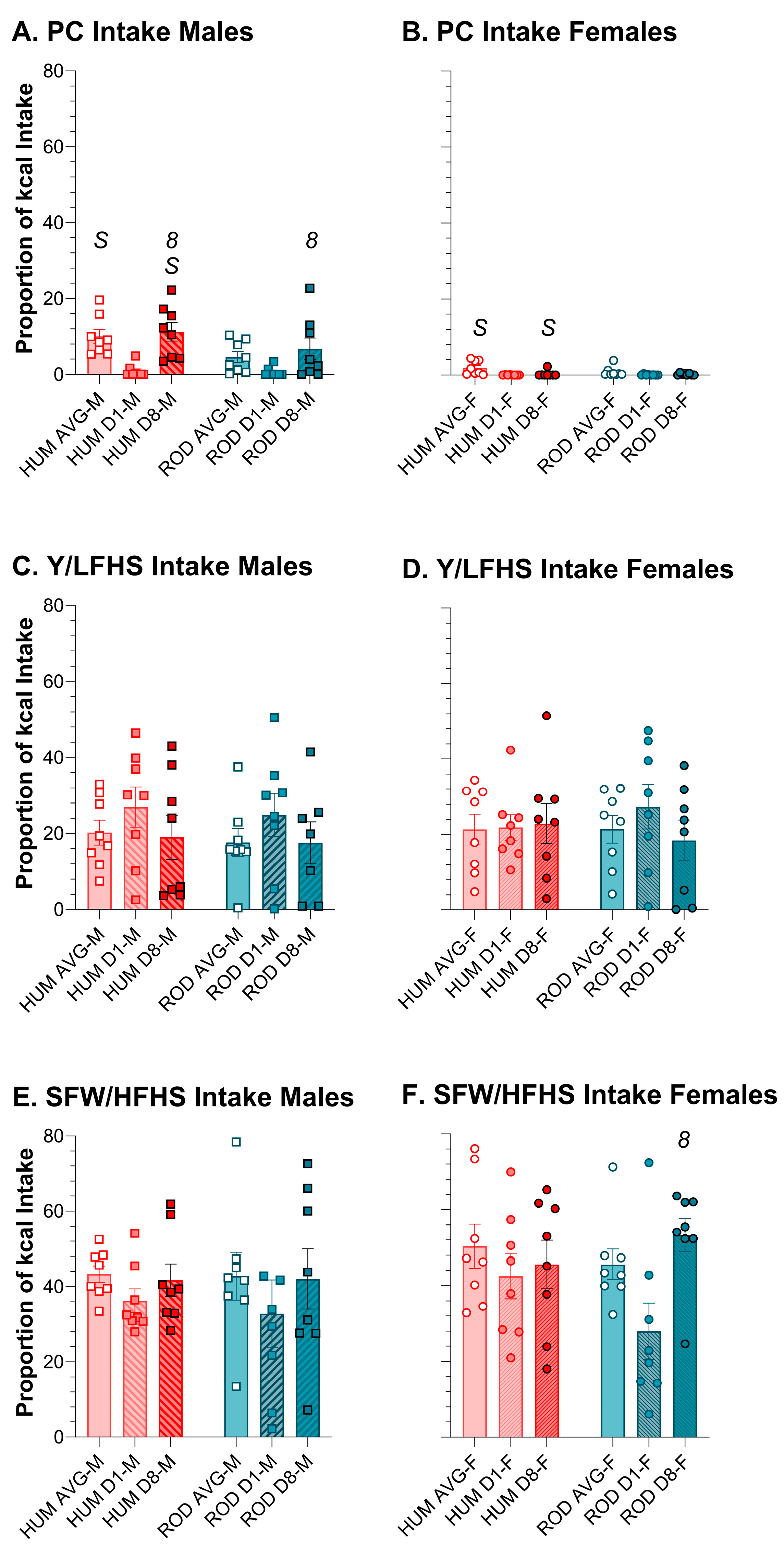
3.4. Intake of Comparable Foods: ROD and HUM
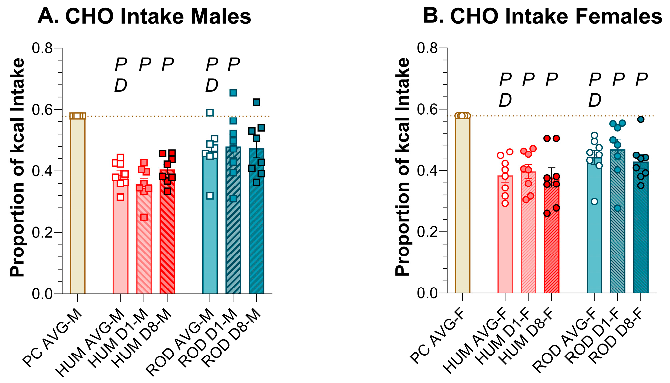
3.5. Macronutrients and Sugar Intake: ROD, HUM, and PC
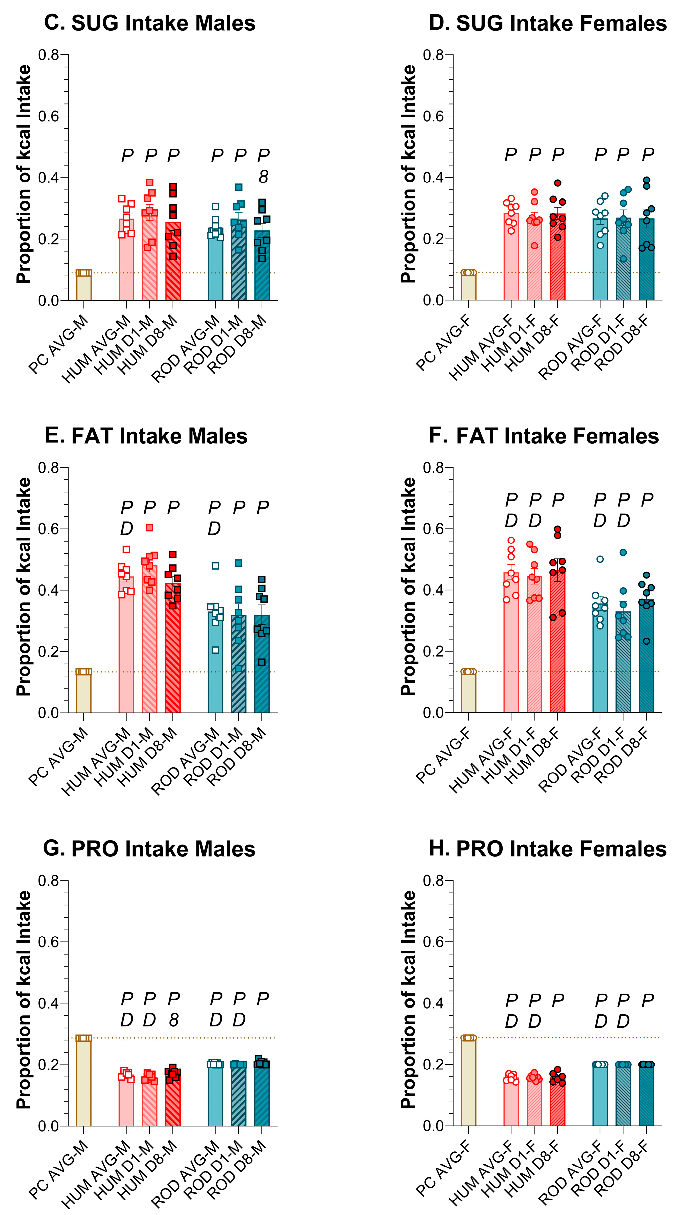
3.6. Meal Characteristics
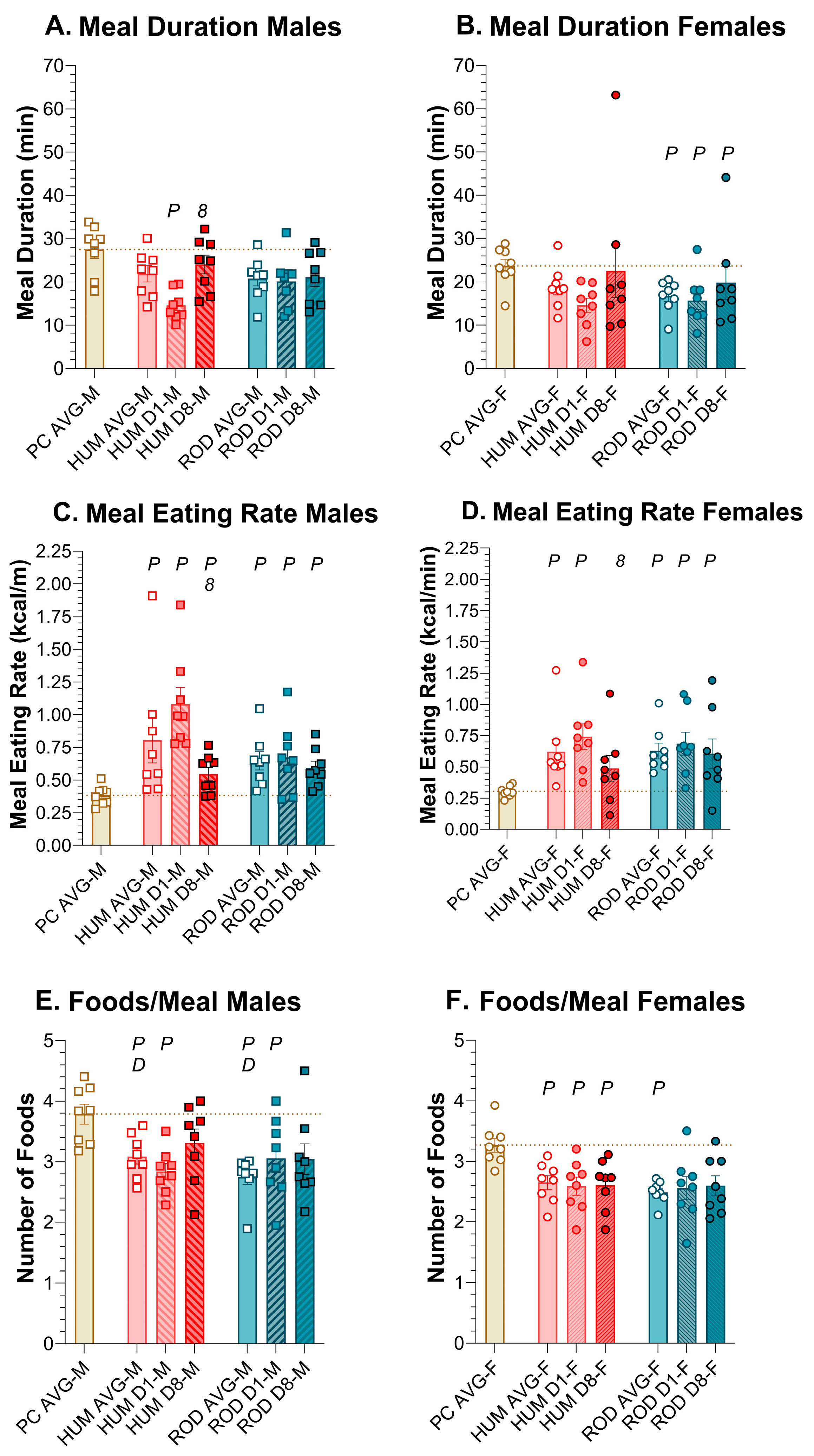
3.6.1. Number of Meals
3.6.2. Meal Size
3.6.3. Meal Duration
3.6.4. Meal Rate
3.6.5. Number of Foods Per Meal
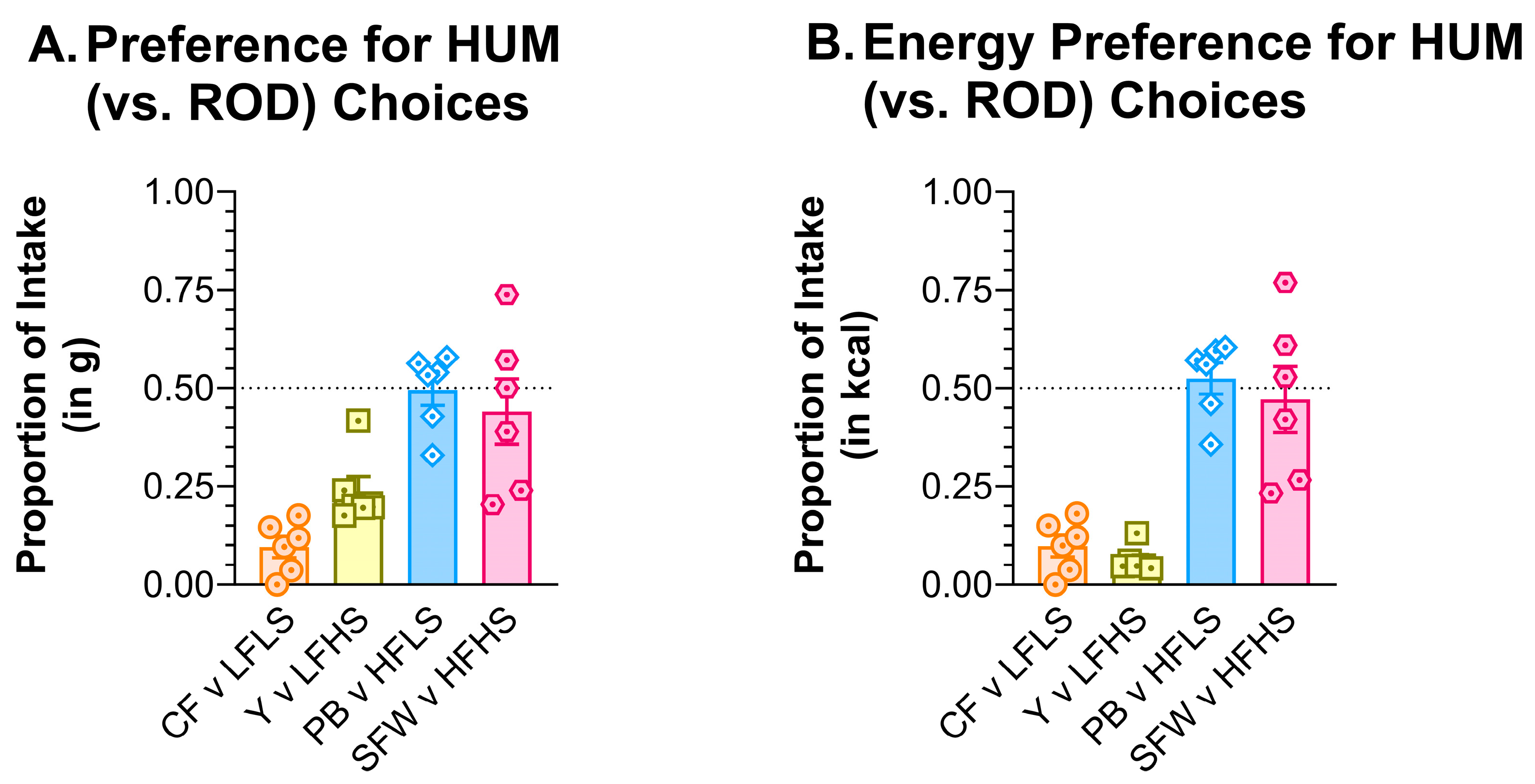
3.7. Two-Jar Tests
3.7.1. ROD v PC
3.7.2. ROD v HUM
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rolls, B.J.; Rowe, E.A.; Rolls, E.T.; Kingston, B.; Megson, A.; Gunary, R. Variety in a meal enhances food intake in man. Physiol. Behav. 1981, 26, 215–221. [Google Scholar] [CrossRef]
- Rolls, B.J.; Van Duijvenvoorde, P.; Rowe, E.A. Variety in the diet enhances intake in a meal and contributes to the development of obesity in the rat. Physiol. Behav. 1983, 31, 21–27. [Google Scholar] [CrossRef]
- Blonde, G.D.; Fletcher, F.H.; Tang, T.; Newsome, R.; Spector, A.C. A new apparatus to analyze meal-related ingestive behaviors in rats fed a complex multi-food diet. Physiol. Behav. 2022, 252, 113824. [Google Scholar] [CrossRef]
- Rogers, P.J.; Blundell, J.E. Meal patterns and food selection during the development of obesity in rats fed a cafeteria diet. Neurosci. Biobehav. Rev. 1985, 8, 441–453. [Google Scholar] [CrossRef]
- Gac, L.; Kanaly, V.; Ramirez, V.; Teske, J.; Pinto, M.; Perez-Leighton, C. Behavioral characterization of a model of differential susceptibility to obesity induced by standard and personalized cafeteria diet feeding. Physiol. Behav. 2015, 152, 315–322. [Google Scholar] [CrossRef]
- Kendig, M.D.; Westbrook, R.F.; Morris, M.J. Pattern of access to cafeteria-style diet determines fat mass and degree of spatial memory impairments in rats. Sci. Rep. 2019, 9, 13516. [Google Scholar] [CrossRef]
- Leigh, S.-J.; Kaakoush, N.O.; Bertoldo, M.J.; Westbrook, R.F.; Morris, M.J. Intermittent cafeteria diet identifies fecal microbiome changes as a predictor of spatial recognition memory impairment in female rats. Transl. Psychiatry 2020, 10, 36. [Google Scholar] [CrossRef]
- Maniam, J.; Morris, M.J. Palatable cafeteria diet ameliorates anxiety and depression-like symptoms following an adverse early environment. Psychoneuroendocrinology 2010, 35, 717–728. [Google Scholar] [CrossRef]
- Zeeni, N.; Daher, C.; Fromentin, G.; Tome, D.; Darcel, N.; Chaumontet, C. A cafeteria diet modifies the response to chronic variable stress in rats. Stress 2012, 16, 211–219. [Google Scholar] [CrossRef]
- Martire, S.I.; Westbrook, R.F.; Morris, M.J. Effects of long-term cycling between palatable cafeteria diet and regular chow on intake, eating patterns, and response to saccharin and sucrose. Physiol. Behav. 2015, 139, 80–88. [Google Scholar] [CrossRef]
- Fam, J.; Clemens, K.J.; Westbrook, R.F.; Morris, M.J.; Kendig, M.D. Chronic exposure to cafeteria-style diet in rats alters sweet taste preference and reduces motivation for, but not ‘liking’ of sucrose. Appetite 2021, 168, 105742. [Google Scholar] [CrossRef]
- Palframan, K.M.; Myers, K.P. Modern ‘junk food’ and minimally-processed ‘natural food’ cafeteria diets alter the response to sweet taste but do not impair flavor-nutrient learning in rats. Physiol. Behav. 2016, 157, 146–157. [Google Scholar] [CrossRef]
- Osiak, J.; Mikut, K.; Wijata, A.; Kędziora-Kornatowska, K. Obesity treatment—Overview of pharmacological and surgical methods. J. Educ. Health Sport 2022, 12, 40–48. [Google Scholar] [CrossRef]
- Müller, T.D.; Blüher, M.; Tschöp, M.H.; DiMarchi, R.D. Anti-obesity drug discovery: Advances and challenges. Nat. Rev. Drug Discov. 2021, 21, 201–223. [Google Scholar] [CrossRef]
- Perdomo, C.M.; Cohen, R.V.; Sumithran, P.; Clément, K.; Frühbeck, G. Contemporary medical, device, and surgical therapies for obesity in adults. Lancet 2023, 401, 1116–1130. [Google Scholar] [CrossRef]
- Blonde, G.D.; Price, R.K.; le Roux, C.W.; Spector, A.C. Meal Patterns and Food Choices of Female Rats Fed a Cafeteria-Style Diet Are Altered by Gastric Bypass Surgery. Nutrients 2021, 13, 3856. [Google Scholar] [CrossRef]
- Mathes, C.M.; Letourneau, C.; Blonde, G.D.; le Roux, C.W.; Spector, A.C. Roux-en-Y gastric bypass in rats progressively decreases the proportion of fat calories selected from a palatable cafeteria diet. Am. J. Physiol. Integr. Comp. Physiol. 2016, 310, R952–R959. [Google Scholar] [CrossRef]
- Lalanza, J.F.; Snoeren, E.M. The cafeteria diet: A standardized protocol and its effects on behavior. Neurosci. Biobehav. Rev. 2020, 122, 92–119. [Google Scholar] [CrossRef]
- Ackroff, K.; Bonacchi, K.; Magee, M.; Yiin, Y.-M.; Graves, J.V.; Sclafani, A. Obesity by choice revisited: Effects of food availability, flavor variety and nutrient composition on energy intake. Physiol. Behav. 2007, 92, 468–478. [Google Scholar] [CrossRef][Green Version]
- Rolls, B.J.; A Rowe, E.; Turner, R.C. Persistent obesity in rats following a period of consumption of a mixed, high energy diet. J. Physiol. 1980, 298, 415–427. [Google Scholar] [CrossRef]
- Goebel-Stengel, M.; Stengel, A.; Wang, L.; Ohning, G.; Taché, Y.; Reeve, J.R. CCK-8 and CCK-58 differ in their effects on nocturnal solid meal pattern in undisturbed rats. Am. J. Physiol. Integr. Comp. Physiol. 2012, 303, R850–R860. [Google Scholar] [CrossRef] [PubMed]
- Farley, C.; Cook, J.A.; Spar, B.D.; Austin, T.M.; Kowalski, T.J. Meal Pattern Analysis of Diet-Induced Obesity in Susceptible and Resistant Rats. Obes. Res. 2003, 11, 845–851. [Google Scholar] [CrossRef]
- Wang, L.; Jacobs, J.P.; Lagishetty, V.; Yuan, P.-Q.; Wu, S.V.; Million, M.; Reeve, J.R.; Pisegna, J.R.; Taché, Y.; Klingbeil, E.; et al. High-protein diet improves sensitivity to cholecystokinin and shifts the cecal microbiome without altering brain inflammation in diet-induced obesity in rats. Am. J. Physiol. Integr. Comp. Physiol. 2017, 313, R473–R486. [Google Scholar] [CrossRef]
- Molle, R.D.; Laureano, D.P.; Alves, M.B.; Reis, T.M.; Desai, M.; Ross, M.G.; Silveira, P.P. Intrauterine growth restriction increases the preference for palatable foods and affects sensitivity to food rewards in male and female adult rats. Brain Res. 2015, 1618, 41–49. [Google Scholar] [CrossRef]
- Eckel, L.A.; Langhans, W.; Kahler, A.; Campfield, L.A.; Smith, F.J.; Geary, N. Chronic administration of OB protein decreases food intake by selectively reducing meal size in female rats. Am. J. Physiol. Integr. Comp. Physiol. 1998, 275, R186–R193. [Google Scholar] [CrossRef]
- Sclafani, A.; Vigorito, M.; Pfeiffer, C.L. Starch-induced overeating and overweight in rats: Influence of starch type and form. Physiol. Behav. 1988, 42, 409–415. [Google Scholar] [CrossRef]
- Sclafani, A.; Nissenbaum, J.W.; Vigorito, M. Starch Preference in Rats. Neurosci. Biobehav. Rev. 1987, 11, 253–262. [Google Scholar] [CrossRef]
- Glass, M.J.; Cleary, J.P.; Billington, C.J.; Levine, A.S. Role of carbohydrate type on diet selection in neuropeptide Y-stimulated rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 1997, 273, R2040–R2045. [Google Scholar] [CrossRef]
- Yadav, S.S.; Redden, R.J.; Chen, W.; Sharma, B. Chickpea Breeding and Management; CAB International: Wallingford, UK, 2007. [Google Scholar]
- Zhang, H.; Yin, L.; Zheng, Y.; Shen, J. Rheological, textural, and enzymatic hydrolysis properties of chickpea starch from a Chinese cultivar. Food Hydrocoll. 2016, 54, 23–29. [Google Scholar] [CrossRef]
- Epstein, L.H.; Robinson, J.L.; Temple, J.L.; Roemmich, J.N.; Marusewski, A.; Nadbrzuch, R. Sensitization and habituation of motivated behavior in overweight and non-overweight children. Learn. Motiv. 2008, 39, 243–255. [Google Scholar] [CrossRef]
- Arcego, D.M.; Krolow, R.; Lampert, C.; Toniazzo, A.P.; Garcia, E.d.S.; Lazzaretti, C.; Costa, G.; Scorza, C.; Dalmaz, C. Chronic high-fat diet affects food-motivated behavior and hedonic systems in the nucleus accumbens of male rats. Appetite 2020, 153, 104739. [Google Scholar] [CrossRef] [PubMed]
- Cawthon, C.R.; de La Serre, C.B. The critical role of CCK in the regulation of food intake and diet-induced obesity. Peptides 2021, 138, 170492. [Google Scholar] [CrossRef] [PubMed]
- Ferrario, C.R.; Labouèbe, G.; Liu, S.; Nieh, E.H.; Routh, V.H.; Xu, S.; O’Connor, E.C. Homeostasis Meets Motivation in the Battle to Control Food Intake. J. Neurosci. 2016, 36, 11469–11481. [Google Scholar] [CrossRef] [PubMed]


| HUM Choices | %CHO | %SUG | %PRO | %FAT | kcal/g | ROD Choices | Research Diets No. | %CHO | %SUG | %PRO | %FAT | kcal/g |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PC | 57.9 | 9.0 | 28.7 | 13.4 | 3.36 | PC | n/a | 57.9 | 9.0 | 28.7 | 13.4 | 3.36 |
| CF | 71.5 | 3.4 | 17.0 | 11.5 | 3.92 | LFLS | D21102802 | 67.0 | 4.0 | 20.0 | 13.0 | 3.79 |
| Y | 64.6 | 61.5 | 21.5 | 13.8 | 0.76 | LFHS | D21102803 | 67.0 | 66.5 | 20.0 | 13.0 | 3.79 |
| PB | 15.7 | 5.9 | 13.7 | 70.6 | 6.18 | HFLS | D21102804 | 10.0 | 4.0 | 20.0 | 70.0 | 5.41 |
| SFW | 28.6 | 27.5 | 13.6 | 57.8 | 5.76 | HFHS | D21102801 | 25.4 | 24.8 | 20.0 | 54.6 | 4.85 |
| Males | Females | ||||
|---|---|---|---|---|---|
| Friedman Statistic | Bonferroni-Corrected p-Value | Friedman Statistic | Bonferroni-Corrected p-Value | ||
| Energy Intake | PC avg v HUM avg | 10.6066 | <0.0001 | 5.3135 | 0.0003 |
| PC avg v ROD avg | 6.3640 | <0.0001 | 6.2796 | <0.0001 | |
| HUM avg v ROD avg | 4.2426 | 0.0024 | 0.9661 | 1.0512 | |
| PC avg v HUM1 | 11.0258 | <0.0001 | 9.7900 | <0.0001 | |
| PC avg v HUM8 | 5.2832 | <0.0001 | 3.5411 | 0.0030 | |
| HUM1 v HUM8 | 5.7426 | <0.0001 | 6.2490 | <0.0001 | |
| PC avg v ROD1 | 7.8099 | <0.0001 | 7.4988 | <0.0001 | |
| PC avg v ROD8 | 4.5941 | <0.0001 | 4.3743 | 0.0003 | |
| ROD1 v ROD8 | 3.2159 | 0.0075 | 3.1245 | 0.0096 | |
| HUM1 v ROD1 | 3.2159 | 0.0075 | 3.0000 | 0.0597 | |
| Relative Energy Intake | PC avg v HUM avg | 3.1180 | 0.0228 | 5.6125 | 0.0003 |
| PC avg v ROD avg | 2.4944 | 0.0771 | 5.6125 | 0.0003 | |
| HUM avg v ROD avg | 0.6236 | 1.6287 | 0.0000 | 3.0000 | |
| PC avg v HUM1 | 6.2796 | <0.0001 | 7.5609 | <0.0001 | |
| PC avg v HUM8 | 0.9661 | 1.0512 | 2.1602 | 0.1458 | |
| HUM1 v HUM8 | 5.3135 | 0.0003 | 5.4006 | 0.0003 | |
| PC avg v ROD1 | 5.6125 | 0.0003 | 6.7626 | <0.0001 | |
| PC avg v ROD8 | 0.0000 | 3.0000 | 3.3813 | 0.0135 | |
| ROD1 v ROD8 | 5.6125 | 0.0003 | 3.3813 | 0.0135 | |
| HUM1 v ROD1 | 0.0000 | 3.0000 | 2.1413 | 0.1143 | |
| Water Intake | PC avg v HUM avg | 13.5607 | <0.0001 | 13.2400 | <0.0001 |
| PC avg v ROD avg | 6.4037 | <0.0001 | 5.0097 | <0.0001 | |
| HUM avg v ROD avg | 7.1570 | <0.0001 | 8.2303 | <0.0001 | |
| PC avg v HUM1 | 10.6066 | <0.0001 | 10.6066 | <0.0001 | |
| PC avg v HUM8 | 6.3640 | <0.0001 | 6.3640 | <0.0001 | |
| HUM1 v HUM8 | 4.2426 | 0.0024 | 4.2426 | 0.0024 | |
| PC avg v ROD1 | 10.1705 | <0.0001 | 10.6066 | <0.0001 | |
| PC avg v ROD8 | 3.0135 | 0.0132 | 6.3640 | <0.0001 | |
| ROD1 v ROD8 | 7.1570 | <0.0001 | 4.2426 | <0.0001 | |
| HUM1 v ROD1 | 7.5337 | <0.0001 | 8.9460 | <0.0001 | |
| U | p-Value | ||
|---|---|---|---|
| Energy Intake | PC avg | 0.0000 | 0.0056 |
| HUM avg | 0.0000 | 0.0056 | |
| HUM Day 1 | 0.0000 | 0.0056 | |
| HUM Day 8 | 1.0000 | 0.0077 | |
| ROD avg | 2.0000 | 0.0112 | |
| ROD Day 1 | 2.0000 | 0.0112 | |
| ROD Day 8 | 0.0000 | 0.0056 | |
| Relative Energy Intake | PC avg | 52.0000 | 0.2499 |
| HUM avg | 52.0000 | 0.2499 | |
| HUM Day 1 | 44.0000 | 1.0000 | |
| HUM Day 8 | 51.0000 | 0.3220 | |
| ROD avg | 54.0000 | 0.1463 | |
| ROD Day 1 | 40.0000 | 1.0000 | |
| ROD Day 8 | 56.0000 | 0.0819 | |
| Water Intake | PC avg | 0.0000 | 0.0056 |
| HUM avg | 12.0000 | 0.2499 | |
| HUM Day 1 | 32.0000 | 1.0000 | |
| HUM Day 8 | 16.0000 | 0.6503 | |
| ROD avg | 0.0000 | 0.0056 | |
| ROD Day 1 | 11.5000 | 0.2184 | |
| ROD Day 8 | 0.0000 | 0.0056 | |
| Males | Females | ||||
|---|---|---|---|---|---|
| Friedman Statistic | Bonferroni-Corrected p-Value | Friedman Statistic | Bonferroni-Corrected p-Value | ||
| PC | HUM avg v ROD avg | 1.5954 | 0.3588 | 1.1741 | 0.7449 |
| HUM1 v HUM8 | 4.9858 | <0.0001 | 0.3914 | 2.0937 | |
| ROD1 v ROD8 | 3.3903 | 0.0051 | 1.1741 | 0.7449 | |
| HUM1 v ROD1 | 0.3989 | 2.0772 | 0.9784 | 1.0038 | |
| CF/ LFLS | HUM avg v ROD avg | 5.2647 | <0.0001 | 3.5355 | 0.0036 |
| HUM1 v HUM8 | 1.1445 | 0.7806 | 1.7678 | 0.2574 | |
| ROD1 v ROD8 | 0.4578 | 1.9497 | 1.0607 | 0.8883 | |
| HUM1 v ROD1 | 5.4936 | <0.0001 | 2.6517 | 0.0360 | |
| Y/ LFHS | HUM avg v ROD avg | 0.6709 | 1.5201 | 0.2667 | 2.3739 |
| HUM1 v HUM8 | 0.9393 | 1.0620 | 0.2667 | 2.3739 | |
| ROD1 v ROD8 | 1.6102 | 0.3489 | 2.1337 | 0.1197 | |
| HUM1 v ROD1 | 0.2684 | 2.3700 | 1.2002 | 0.7143 | |
| PB/ HFLS | HUM avg v ROD avg | 6.4550 | <0.0001 | 2.9580 | 0.0165 |
| HUM1 v HUM8 | 4.8412 | <0.0001 | 0.0000 | 3.0000 | |
| ROD1 v ROD8 | 3.5502 | 0.0033 | 3.8032 | 0.0015 | |
| HUM1 v ROD1 | 8.3915 | <0.0001 | 2.5355 | 0.0477 | |
| SFW/ HFHS | HUM avg v ROD avg | 0.2643 | 2.3793 | 0.9917 | 0.9846 |
| HUM1 v HUM8 | 0.2643 | 2.3793 | 0.1417 | 2.6646 | |
| ROD1 v ROD8 | 0.2643 | 2.3793 | 2.5500 | 0.0459 | |
| HUM1 v ROD1 | 0.2643 | 2.3793 | 1.2750 | 0.6321 | |
| U | p-Value | ||
|---|---|---|---|
| PC Intake | HUM avg | 0.0000 | 0.0024 |
| HUM Day 1 | 16.0000 | 0.2181 | |
| HUM Day 8 | 0.0000 | 0.0018 | |
| ROD avg | 12.0000 | 0.1071 | |
| ROD Day 1 | 33.0000 | 1.0000 | |
| ROD Day 8 | 12.0000 | 0.0930 | |
| CF/LFLS Intake | HUM avg | 42.0000 | 0.8808 |
| HUM Day 1 | 52.0000 | 0.1071 | |
| HUM Day 8 | 24.0000 | 1.0000 | |
| ROD avg | 22.0000 | 0.8808 | |
| ROD Day 1 | 37.0000 | 1.0000 | |
| ROD Day 8 | 22.000 | 0.8808 | |
| Y/LFHS Intake | HUM avg | 35.0000 | 1.0000 |
| HUM Day 1 | 24.0000 | 1.0000 | |
| HUM Day 8 | 37.0000 | 1.0000 | |
| ROD avg | 40.0000 | 1.0000 | |
| ROD Day 1 | 36.0000 | 1.0000 | |
| ROD Day 8 | 34.0000 | 1.0000 | |
| PB/HFLS Intake | HUM avg | 24.0000 | 1.0000 |
| HUM Day 1 | 13.0000 | 0.1380 | |
| HUM Day 8 | 42.0000 | 0.8808 | |
| ROD avg | 45.0000 | 0.5166 | |
| ROD Day 1 | 45.0000 | 0.5166 | |
| ROD Day 8 | 33.0000 | 1.0000 | |
| SFW/HFHS Intake | HUM avg | 38.0000 | 1.0000 |
| HUM Day 1 | 37.0000 | 1.0000 | |
| HUM Day 8 | 37.0000 | 1.0000 | |
| ROD avg | 38.0000 | 1.0000 | |
| ROD Day 1 | 27.0000 | 1.0000 | |
| ROD Day 8 | 39.0000 | 1.0000 | |
| Males | Females | ||||
|---|---|---|---|---|---|
| Friedman Statistic | Bonferroni-Corrected p-Value | Friedman Statistic | Bonferroni-Corrected p-Value | ||
| CHO Intake | PC avg v HUM avg | 6.7626 | <0.0001 | 10.6066 | <0.0001 |
| PC avg v ROD avg | 3.3813 | 0.0135 | 6.3640 | <0.0001 | |
| HUM avg v ROD avg | 3.3813 | 0.0135 | 4.2426 | 0.0024 | |
| PC avg v HUM1 | 7.5609 | <0.0001 | 5.6125 | 0.0003 | |
| PC avg v HUM8 | 5.4006 | 0.0003 | 5.6125 | 0.0003 | |
| HUM1 v HUM8 | 2.1602 | 0.1458 | 0.0000 | 3.0000 | |
| PC avg v ROD1 | 3.1180 | 0.0228 | 5.4006 | 0.0003 | |
| PC avg v ROD8 | 2.4944 | 0.0771 | 7.5609 | <0.0001 | |
| ROD1 v ROD8 | 0.6236 | 1.6287 | 2.1602 | 0.1458 | |
| HUM1 v ROD1 | 1.5275 | 0.5115 | 1.5275 | 0.5115 | |
| SUGAR Intake | PC avg v HUM avg | 7.5609 | <0.0001 | 5.6125 | 0.0003 |
| PC avg v ROD avg | 5.4006 | 0.0003 | 5.6125 | 0.0003 | |
| HUM avg v ROD avg | 2.1602 | 0.1458 | 0.0000 | 3.0000 | |
| PC avg v HUM1 | 5.6125 | 0.0003 | 5.6125 | 0.0003 | |
| PC avg v HUM8 | 5.6125 | 0.0003 | 5.6125 | 0.0003 | |
| HUM1 v HUM8 | 0.0000 | 3.0000 | 0.0000 | 3.0000 | |
| PC avg v ROD1 | 10.6066 | <0.0001 | 6.2796 | <0.0001 | |
| PC avg v ROD8 | 6.3640 | <0.0001 | 6.3135 | 0.0003 | |
| ROD1 v ROD8 | 4.2426 | 0.0024 | 0.9661 | 1.0512 | |
| HUM1 v ROD1 | 0.6831 | 1.5495 | 0.0000 | 3.0000 | |
| FAT Intake | PC avg v HUM avg | 10.6066 | <0.0001 | 7.3196 | <0.0001 |
| PC avg v ROD avg | 6.3640 | <0.0001 | 3.0819 | 0.0108 | |
| HUM avg v ROD avg | 4.2426 | 0.0024 | 4.2376 | 0.0003 | |
| PC avg v HUM1 | 7.5609 | <0.0001 | 5.6125 | 0.0003 | |
| PC avg v HUM8 | 5.4006 | 0.0003 | 5.6125 | 0.0003 | |
| HUM1 v HUM8 | 2.1602 | 0.1458 | 0.0000 | 3.0000 | |
| PC avg v ROD1 | 6.2796 | <0.0001 | 5.4006 | 0.0003 | |
| PC avg v ROD8 | 5.3135 | 0.0003 | 7.5609 | <0.0001 | |
| ROD1 v ROD8 | 0.9661 | 1.0512 | 2.1602 | 0.1458 | |
| HUM1 v ROD1 | 3.0000 | 0.0597 | 4.0450 | 0.0006 | |
| PRO Intake | PC avg v HUM avg | 14.1421 | <0.0001 | 11.3837 | <0.0001 |
| PC avg v ROD avg | 4.5962 | <0.0001 | 3.8944 | 0.0009 | |
| HUM avg v ROD avg | 9.5459 | <0.0001 | 7.4893 | <0.0001 | |
| PC avg v HUM1 | 10.6066 | <0.0001 | 5.6125 | 0.0003 | |
| PC avg v HUM8 | 6.3640 | <0.0001 | 5.6125 | 0.0003 | |
| HUM1 v HUM8 | 4.2426 | 0.0024 | 0.0000 | 3.0000 | |
| PC avg v ROD1 | 6.2796 | <0.0001 | 5.3135 | 0.0003 | |
| PC avg v ROD8 | 5.3135 | 0.0003 | 6.2796 | <0.0001 | |
| ROD1 v ROD8 | 0.9661 | 1.0512 | 0.9661 | 1.0512 | |
| HUM1 v ROD1 | 8.4853 | <0.0001 | 6.5906 | <0.0001 | |
| U | p-Value | ||
|---|---|---|---|
| CHO Intake | HUM avg | 32.0000 | 1.0000 |
| HUM Day 1 | 42.0000 | 1.0000 | |
| HUM Day 8 | 23.0000 | 1.0000 | |
| ROD avg | 26.0000 | 1.0000 | |
| ROD Day 1 | 32.0000 | 1.0000 | |
| ROD Day 8 | 25.0000 | 1.0000 | |
| SUG Intake | HUM avg | 41.0000 | 1.0000 |
| HUM Day 1 | 23.0000 | 1.0000 | |
| HUM Day 8 | 38.0000 | 1.0000 | |
| ROD avg | 43.0000 | 1.0000 | |
| ROD Day 1 | 35.0000 | 1.0000 | |
| ROD Day 8 | 40.0000 | 1.0000 | |
| FAT Intake | HUM avg | 34.0000 | 1.0000 |
| HUM Day 1 | 24.0000 | 1.0000 | |
| HUM Day 8 | 42.0000 | 1.0000 | |
| ROD avg | 41.0000 | 1.0000 | |
| ROD Day 1 | 32.0000 | 1.0000 | |
| ROD Day 8 | 41.0000 | 1.0000 | |
| PRO Intake | HUM avg | 14.0000 | 0.3522 |
| HUM Day 1 | 28.0000 | 1.0000 | |
| HUM Day 8 | 16.0000 | 0.5574 | |
| ROD avg | 10.0000 | 0.1254 | |
| ROD Day 1 | 35.0000 | 1.0000 | |
| ROD Day 8 | 12.0000 | 0.2112 | |
| Males | Females | ||||
|---|---|---|---|---|---|
| Friedman Statistic | Bonferroni-Corrected p-Value | Friedman Statistic | Bonferroni-Corrected p-Value | ||
| Number of Meals | PC avg v HUM avg | 0.2357 | 2.4513 | 0.7568 | 1.3851 |
| PC avg v ROD avg | 0.2357 | 2.4513 | 0.0000 | 3.0000 | |
| HUM avg v ROD avg | 0.4714 | 1.9338 | 0.7568 | 1.3851 | |
| PC avg v HUM1 | 3.2051 | 0.0192 | 3.1180 | 0.0228 | |
| PC avg v HUM8 | 0.3374 | 2.2224 | 0.6236 | 1.6287 | |
| HUM1 v HUM8 | 2.8677 | 0.0372 | 2.4944 | 0.0771 | |
| PC avg v ROD1 | 2.2215 | 0.1299 | 0.1231 | 2.7114 | |
| PC avg v ROD8 | 0.1587 | 2.6286 | 0.1231 | 2.7114 | |
| ROD1 v ROD8 | 2.0629 | 0.1746 | 0.2462 | 2.4273 | |
| HUM1 v ROD1 | 2.3760 | 0.1476 | 3.0000 | 0.0597 | |
| Meal Size (kcal) | PC avg v HUM avg | 5.3072 | 0.0003 | 5.3135 | 0.0003 |
| PC avg v ROD avg | 2.0412 | 0.1815 | 6.2796 | <0.0001 | |
| HUM avg v ROD avg | 3.2660 | 0.0168 | 0.9661 | 1.0512 | |
| PC avg v HUM1 | 3.5824 | 0.0090 | 3.5824 | 0.0090 | |
| PC avg v HUM8 | 2.2797 | 0.1164 | 1.3027 | 0.6411 | |
| HUM1 v HUM8 | 1.3027 | 0.6411 | 2.2797 | 0.1164 | |
| PC avg v ROD1 | 2.5677 | 0.0669 | 4.4900 | 0.0015 | |
| PC avg v ROD8 | 1.7118 | 0.3270 | 3.3675 | 0.0138 | |
| ROD1 v ROD8 | 0.8559 | 1.2195 | 1.1225 | 0.8415 | |
| HUM1 v ROD1 | 3.0000 | 0.0597 | 0.0000 | 3.0000 | |
| U | p-Value | ||
|---|---|---|---|
| Number of Meals | PC avg | 31.0000 | 1.0000 |
| HUM avg | 31.5000 | 1.0000 | |
| HUM Day 1 | 33.0000 | 1.0000 | |
| HUM Day 8 | 32.0000 | 1.0000 | |
| ROD avg | 27.5000 | 1.0000 | |
| ROD Day 1 | 20.0000 | 1.0000 | |
| ROD Day 8 | 22.5000 | 1.0000 | |
| Meal Size (kcal) | PC avg | 4.0000 | 0.0231 |
| HUM avg | 5.0000 | 0.0322 | |
| HUM Day 1 | 9.0000 | 0.1099 | |
| HUM Day 8 | 9.0000 | 0.0889 | |
| ROD avg | 10.0000 | 0.1463 | |
| ROD Day 1 | 15.0000 | 0.5194 | |
| ROD Day 8 | 18.0000 | 0.9905 | |
| Meal Duration (min) | PC avg | 17.0000 | 0.8064 |
| HUM avg | 23.0000 | 1.0000 | |
| HUM Day 1 | 35.0000 | 1.0000 | |
| HUM Day 8 | 18.0000 | 0.9905 | |
| ROD avg | 14.0000 | 0.4109 | |
| ROD Day 1 | 18.0000 | 0.9905 | |
| ROD Day 8 | 25.0000 | 1.0000 | |
| Meal Eating Rate (kcal/min) | PC avg | 10.0000 | 0.1463 |
| HUM avg | 24.0000 | 1.0000 | |
| HUM Day 1 | 13.0000 | 0.3220 | |
| HUM Day 8 | 24.0000 | 1.0000 | |
| ROD avg | 27.0000 | 1.0000 | |
| ROD Day 1 | 31.0000 | 1.0000 | |
| ROD Day 8 | 30.0000 | 1.0000 | |
| Foods/Meal | PC avg | 13.0000 | 0.3220 |
| HUM avg | 11.0000 | 0.1918 | |
| HUM Day 1 | 22.0000 | 1.0000 | |
| HUM Day 8 | 13.0000 | 0.3220 | |
| ROD avg | 8.0000 | 0.0819 | |
| ROD Day 1 | 17.0000 | 0.8064 | |
| ROD Day 8 | 18.0000 | 0.9821 | |
| Males | Females | ||||
|---|---|---|---|---|---|
| Friedman Statistic | Bonferroni-Corrected p-Value | Friedman Statistic | Bonferroni-Corrected p-Value | ||
| Meal Duration (min) | PC avg v HUM avg | 1.9522 | 0.2136 | 2.4944 | 0.0771 |
| PC avg v ROD avg | 2.2311 | 0.1275 | 3.1180 | 0.0228 | |
| HUM avg v ROD avg | 0.2789 | 2.3532 | 0.6236 | 1.6287 | |
| PC avg v HUM1 | 5.3072 | 0.0003 | 2.5677 | 0.0669 | |
| PC avg v HUM8 | 2.0412 | 0.1815 | 1.7118 | 0.3270 | |
| HUM1 v HUM8 | 3.2660 | 0.0168 | 0.8559 | 1.2195 | |
| PC avg v ROD1 | 2.2311 | 0.1275 | 2.7681 | 0.0453 | |
| PC avg v ROD8 | 1.9522 | 0.2136 | 2.7681 | 0.0453 | |
| ROD1 v ROD8 | 0.2789 | 2.3532 | 0.0000 | 3.0000 | |
| HUM1 v ROD1 | 3.0000 | 0.0597 | 0.6831 | 1.5495 | |
| Meal Eating Rate (kcal/min) | PC avg v HUM avg | 5.6125 | 0.0003 | 3.3675 | 0.0138 |
| PC avg v ROD avg | 5.6125 | 0.0003 | 4.4900 | 0.0015 | |
| HUM avg v ROD avg | 0.0000 | 3.0000 | 1.1225 | 0.8415 | |
| PC avg v HUM1 | 7.8619 | <0.0001 | 5.3072 | 0.0003 | |
| PC avg v HUM8 | 3.1448 | 0.0090 | 2.0412 | 0.1815 | |
| HUM1 v HUM8 | 4.7171 | <0.0001 | 3.2660 | 0.0168 | |
| PC avg v ROD1 | 5.6125 | 0.0003 | 2.7681 | 0.0453 | |
| PC avg v ROD8 | 5.6125 | 0.0003 | 2.7681 | 0.0453 | |
| ROD1 v ROD8 | 0.0000 | 3.0000 | 0.0000 | 3.0000 | |
| HUM1 v ROD1 | 3.0000 | 0.0597 | 1.5275 | 0.5115 | |
| Foods/Meal | PC avg v HUM avg | 3.3813 | 0.0135 | 5.4006 | 0.0003 |
| PC avg v ROD avg | 6.7626 | <0.0001 | 7.5609 | <0.0001 | |
| HUM avg v ROD avg | 3.3813 | 0.0135 | 2.1602 | 0.1458 | |
| PC avg v HUM1 | 3.5824 | 0.0090 | 3.9605 | 0.0042 | |
| PC avg v HUM8 | 1.3027 | 0.6411 | 3.6004 | 0.0087 | |
| HUM1 v HUM8 | 2.2797 | 0.1164 | 0.3600 | 2.1726 | |
| PC avg v ROD1 | 3.1180 | 0.0228 | 2.5677 | 0.0669 | |
| PC avg v ROD8 | 2.4944 | 0.0771 | 1.7118 | 0.3270 | |
| ROD1 v ROD8 | 0.6236 | 1.6287 | 0.8559 | 1.2195 | |
| HUM1 v ROD1 | 0.6831 | 1.5495 | 0.6831 | 1.5495 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cawthon, C.R.; Spector, A.C. The Nature of Available Choices Affects the Intake and Meal Patterns of Rats Offered a Palatable Cafeteria-Style Diet. Nutrients 2023, 15, 5093. https://doi.org/10.3390/nu15245093
Cawthon CR, Spector AC. The Nature of Available Choices Affects the Intake and Meal Patterns of Rats Offered a Palatable Cafeteria-Style Diet. Nutrients. 2023; 15(24):5093. https://doi.org/10.3390/nu15245093
Chicago/Turabian StyleCawthon, Carolina R., and Alan C. Spector. 2023. "The Nature of Available Choices Affects the Intake and Meal Patterns of Rats Offered a Palatable Cafeteria-Style Diet" Nutrients 15, no. 24: 5093. https://doi.org/10.3390/nu15245093
APA StyleCawthon, C. R., & Spector, A. C. (2023). The Nature of Available Choices Affects the Intake and Meal Patterns of Rats Offered a Palatable Cafeteria-Style Diet. Nutrients, 15(24), 5093. https://doi.org/10.3390/nu15245093






