Biochemical, Nutritional, and Clinical Parameters of Vitamin B12 Deficiency in Infants: A Systematic Review and Analysis of 292 Cases Published between 1962 and 2022
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Search
2.2. Data Extraction
2.3. Statistical Methods
3. Results
3.1. Descriptive Statistics of Included Studies
3.2. Descriptive Statistics of Included Cases
3.3. Correlation Analyses
3.4. Association between Clinical Symptoms: Chi-Squared Tests
3.5. Subsample Comparisons: Descriptive Statistics, Chi-Squared Tests, and ANOVAs
3.6. Regression Analysis—Predicting Infant Biomarkers
3.7. Regression Analysis—Predicting Clinical Outcomes
3.8. Regression Analysis—Infant Biomarkers and Medical Data as Moderators
4. Discussion
5. Conclusions and Implications for Clinical Practice
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| B12 | Vitamin B12 |
| Cbl | Cobalamin |
| Hcy | Homocysteine |
| tHcy | Total Homocysteine |
| MMA | Methylmalonic acid |
| HoloTC | Holo-Transcobalamin |
| NBS | Newborn Screening |
References
- de Souza, A.; Moloi, M.W. Involuntary movements due to vitamin B12 deficiency. Neurol. Res. 2014, 36, 1121–1128. [Google Scholar] [CrossRef] [PubMed]
- Dror, D.K.; Allen, L.H. Effect of vitamin B12 deficiency on neurodevelopment in infants: Current knowledge and possible mechanisms. Nutr. Rev. 2008, 66, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Huemer, M.; Baumgartner, M.R. The clinical presentation of cobalamin-related disorders: From acquired deficiencies to inborn errors of absorption and intracellular pathways. J. Inherit. Metab. Dis. 2019, 42, 686–705. [Google Scholar] [CrossRef] [PubMed]
- Ströhle, A.; Richter, M.; González-Gross, M.; Neuhäuser-Berthold, M.; Wagner, K.H.; Leschik-Bonnet, E.; Egert, S. German Nutrition Society (DGE). The Revised D-A-CH-Reference Values for the Intake of Vitamin B12: Prevention of Deficiency and Beyond. Mol. Nutr. Food Res. 2019, 63, e1801178. [Google Scholar] [CrossRef]
- Rashid, S.; Meier, V.; Patrick, H. Review of Vitamin B12 deficiency in pregnancy: A diagnosis not to miss as veganism and vegetarianism become more prevalent. Eur. J. Haematol. 2021, 106, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Obeid, R.; Morkbak, A.L.; Munz, W.; Nexo, E.; Herrmann, W. The cobalamin-binding proteins transcobalamin and haptocorrin in maternal and cord blood sera at birth. Clin. Chem. 2006, 52, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Layden, A.J.; O’Brien, K.O.; Pressman, E.K.; Cooper, E.M.; Kent, T.R.; Finkelstein, J.L. Vitamin B12 and placental expression of transcobalamin in pregnant adolescents. Placenta 2016, 45, 1–7. [Google Scholar] [CrossRef]
- Duggan, C.; Srinivasan, K.; Thomas, T.; Samuel, T.; Rajendran, R.; Muthayya, S.; Finkelstein, J.L.; Lukose, A.; Fawzi, W.; Allen, L.H.; et al. Vitamin B-12 supplementation during pregnancy and early lactation increases maternal, breast milk, and infant measures of vitamin B-12 status. J. Nutr. 2014, 144, 758–764. [Google Scholar] [CrossRef]
- Hay, G.; Johnston, C.; Whitelaw, A.; Trygg, K.; Refsum, H. Folate and cobalamin status in relation to breastfeeding and weaning in healthy infants. Am. J. Clin. Nutr. 2008, 88, 105–114. [Google Scholar] [CrossRef]
- Hannibal, L.; Lysne, V.; Bjørke-Monsen, A.L.; Behringer, S.; Grünert, S.C.; Spiekerkoetter, U.; Jacobsen, D.W.; Blom, H.J. Biomarkers and Algorithms for the Diagnosis of Vitamin B12 Deficiency. Front. Mol. Biosci. 2016, 3, 27. [Google Scholar] [CrossRef]
- Page, M.J.; Moher, D.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Bmj 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Moola, S.; Munn, Z.; Tufanaru, C.; Aromataris, E.; Sears, K.; Sfetcu, R.; Currie, M.; Lisy, K.; Qureshi, R.; Mattis, P.; et al. Chapter 7: Systematic reviews of etiology and risk. In JBI Manual for Evidence Synthesis; Aromataris, E., Munn, Z., Eds.; JBI: Adelaide, Australia, 2020; Available online: https://synthesismanual.jbi.global (accessed on 28 February 2023).
- Team R. RStudio: Integrated Development Environment for R RStudio, PBC, Boston, MA. 2020. Available online: http://www.rstudio.com/ (accessed on 28 February 2023).
- Snow, C.F. Laboratory diagnosis of vitamin B12 and folate deficiency: A guide for the primary care physician. Arch. Intern. Med. 1999, 159, 1289–1298. [Google Scholar] [CrossRef]
- Devalia, V.; Hamilton, M.S.; Molloy, A.M. Guidelines for the diagnosis and treatment of cobalamin and folate disorders. Br. J. Haematol. 2014, 166, 496–513. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://bestpractice.bmj.com/topics/en-gb/822?bcgovtm=pique%20newsmagazine (accessed on 21 June 2023).
- Roumeliotis, N.; Dix, D.; Lipson, A. Vitamin B12 deficiency in infants secondary to maternal causes. Can. Med. Assoc. J. 2012, 184, 1593–1598. [Google Scholar] [CrossRef] [PubMed][Green Version]
- von Schenck, U.; Bender-Gotze, C.; Koletzko, B. Persistence of neurological damage induced by dietary vitamin B-12 deficiency in infancy. Arch. Dis. Child. 1997, 77, 137–139. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.; Ahmad, N.; Rossetti, A.L. Vomiting as a Presenting Symptom of Infantile Vitamin B12 Deficiency. Cureus 2022, 14, e25134. [Google Scholar] [CrossRef] [PubMed]
- Feraco, P.; Incandela, F.; Franceschi, R.; Gagliardo, C.; Bellizzi, M. Clinical and Brain Imaging Findings in a Child with Vitamin B12 Deficiency. Pediatr. Rep. 2021, 13, 583–588. [Google Scholar] [CrossRef]
- Kaninde, A.; Katre, M.; Papadopoulou, K.; Ramaswamy, R. 55 Regression of milestones in an infant as presenting feature of Maternal Pernicious anaemia. Arch. Dis. Child. 2021, 106 (Suppl. S2), 23–24. [Google Scholar]
- Subramani, P.; Saranya, C.G.; Chand, G.M.; Narayani, R.S.; James, S.; Vinoth, P.N. Neuroregression in an infant: A rare cause. S. Afr. J. Child Health 2015, 9, 59–60. [Google Scholar]
- Delbet, J.D.; Ulinski, T. Thrombotic microangiopathy and breastfeeding: Where is the link? Questions. Pediatr. Nephrol. 2017, 33, 985–986. [Google Scholar] [CrossRef]
- Sklar, R. Nutritional Vitamin B12 Deficiency in a Breast-Fed Infant of a Vegan-Diet Mother. Clin. Pediatr. 1986, 25, 219–221. [Google Scholar] [CrossRef] [PubMed]
- Sadowitz, P.D.; Livingston, A.; Cavanaugh, R.M. Developmental Regression as an Early Manifestation of Vitamin B12 Deficiency. Clin. Pediatr. 1986, 25, 369–371. [Google Scholar] [CrossRef] [PubMed]
- Chalouhi, C.; Faesch, S.; Anthoine-Milhomme, M.C.; Fulla, Y.; Dulac, O.; Chéron, G. Neurological Consequences of Vitamin B12 Deficiency and Its Treatment. Pediatr. Emerg. Care 2008, 24, 538–541. [Google Scholar] [CrossRef]
- Lampkin, B.C.; Shore, N.A.; Chadwick, D. Megaloblastic Anemia of Infancy Secondary to Maternal Pernicious Anemia. N. Engl. J. Med. 1966, 274, 1168–1171. [Google Scholar] [CrossRef] [PubMed]
- Almadan, M.S.; Al Awamy, B.H.; Al Mulhim, I.A. Nutritional Vitamin B12 deficiency in infancy. Indian J. Pediatr. 1993, 60, 683–685. [Google Scholar] [CrossRef] [PubMed]
- Zetterström, R.; Franzén, S. Megaloblastic Anemia in Infancy: Megaloblastic Anemia Occurring in an Infant of a Mother Suffering from Pernicious Anemia of Pregnancy. Acta Paediatr. 1954, 43, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Schlapbach, L.J.; Schütz, B.; Nuoffer, J.M.; Brekenfeld, C.; Müller, G.; Fluri, S. Floppy baby with macrocytic anemia and vegan mother. Praxis 2007, 96, 1309–1314. [Google Scholar] [CrossRef]
- Akcaboy, M.; Malbora, B.; Zorlu, P.; Altınel, E.; Oguz, M.M.; Senel, S. Vitamin B12 Deficiency in Infants. Indian J. Pediatr. 2015, 82, 619–624. [Google Scholar] [CrossRef]
- Sturm, I.; Hennermann, J.B.; von Arnim-Baas, A.; Driever, P.H.; Massenkeil, G. Thromboembolic events, abortions and a sick infant—Unusual presentation of a vitamin deficiency. Internist 2008, 49, 1507–1511. [Google Scholar] [CrossRef]
- Hasbaoui, B.E.; Mebrouk, N.; Saghir, S.; Yajouri, A.E.; Abilkassem, R.; Agadr, A. Vitamin B12 deficiency: Case report and review of literature. Pan. Afr. Med. J. 2021, 38, 237. [Google Scholar]
- Sharma, N.; Kunwar, S.; Shrestha, A.K. Vitamin B12 Deficiency Resembling Acute Leukemia: A Case Report. JNMA J. Nepal Med. Assoc. 2021, 59, 1182–1184. [Google Scholar] [CrossRef] [PubMed]
- Quentin, C.; Huybrechts, S.; Rozen, L.; De Laet, C.; Demulder, A.; Ferster, A. Vitamin B12 deficiency in a 9-month-old boy. Eur. J. Pediatr. 2012, 171, 193–195. [Google Scholar] [CrossRef] [PubMed]
- Roschitz, B.; Plecko, B.; Huemer, M.; Biebl, A.; Foerster, H.; Sperl, W. Nutritional infantile vitamin B12 deficiency: Pathobiochemical considerations in seven patients. Arch. Dis. Child. Fetal Neonatal Ed. 2005, 90, F281–F282. [Google Scholar] [CrossRef] [PubMed]
- Dubaj, C.; Czyż, K.; Furmaga-Jabłońska, W. Vitamin B12 deficiency as a cause of severe neurological symptoms in breast fed infant—A case report. Ital. J. Pediatr. 2020, 46, 40. [Google Scholar] [CrossRef] [PubMed]
- Guez, S.; Chiarelli, G.; Menni, F.; Salera, S.; Principi, N.; Esposito, S. Severe vitamin B12 deficiency in an exclusively breastfed 5-month-old Italian infant born to a mother receiving multivitamin supplementation during pregnancy. BMC Pediatr. 2012, 12, 85. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.L.; Goh, M.C.; Fu, K.X.; Aw, M.H.; Quak, S.H.; Goh, D.L. Severe vitamin B12 deficiency in a 7-month-old boy. Ann. Acad. Med. Singap. 2015, 44, 109–111. [Google Scholar] [CrossRef]
- Erol, I.; Alehan, F.; Gümüs, A. West syndrome in an infant with vitamin B12 deficiency in the absence of macrocytic anaemia. Dev. Med. Child Neurol. 2007, 49, 774–776. [Google Scholar] [CrossRef]
- McPhee, A.J.; Davidson, G.P.; Leahy, M.; Beare, T. Vitamin B12 deficiency in a breast fed infant. Arch. Dis. Child. 1988, 63, 921–923. [Google Scholar] [CrossRef]
- Kocaoglu, C.; Akin, F.; Caksen, H.; Böke, S.B.; Arslan, S.; Aygün, S. Cerebral atrophy in a vitamin B12-deficient infant of a vegetarian mother. J. Health Popul. Nutr. 2014, 32, 367–371. [Google Scholar]
- Afzal, T.; Ashraf, N.; Munir, S.; Tabassum, R. Megaloblastic anaemia in a 9-weeks old infant: A case report. J. Pak. Med. Assoc. 2020, 70, 923–925. [Google Scholar]
- Ide, E.; Van Biervliet, S.; Thijs, J.; Vande Velde, S.; De Bruyne, R.; Van Winckel, M. Solid food refusal as the presenting sign of vitamin B12 deficiency in a breastfed infant. Eur. J. Pediatr. 2011, 170, 1453–1455. [Google Scholar] [CrossRef] [PubMed]
- Korenke, G.C.; Hunneman, D.H.; Eber, S.; Hanefeld, F. Severe encephalopathy with epilepsy in an infant caused by subclinical maternal pernicious anaemia: Case report and review of the literature. Eur. J. Pediatr. 2004, 163, 196–201. [Google Scholar] [CrossRef] [PubMed]
- Hoey, H.; Linnell, J.C.; Oberholzer, V.G.; Laurance, B.M. Vitamin B12 deficiency in a breastfed infant of a mother with pernicious anaemia. J. R. Soc. Med. 1982, 75, 656–658. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, V.; Dias, A.; Brito, M.J.; Galvão, I.; Ferreira, G.C. Severe megaloblastic anaemia in an infant. BMJ Case Rep. 2011, 2011, bcr0220113835. [Google Scholar] [CrossRef] [PubMed]
- Baatenburg de Jong, R.; Bekhof, J.; Roorda, R.; Zwart, P. Severe nutritional vitamin deficiency in a breast-fed infant of a vegan mother. Eur. J. Pediatr. 2005, 164, 259–260. [Google Scholar] [CrossRef] [PubMed]
- Rachmel, A.; Steinberg, T.; Ashkenazi, S.; Sela, B.A. Cobalamin deficiency in a breast-fed infant of a vegetarian mother. Isr. Med. Assoc. J. 2003, 5, 534–536. [Google Scholar] [PubMed]
- McNeil, K.; Chowdhury, D.; Penney, L.; Rashid, M. Vitamin B12 deficiency with intrinsic factor antibodies in an infant with poor growth and developmental delay. Paediatr. Child Health 2014, 19, 84–86. [Google Scholar] [CrossRef] [PubMed]
- Serin, H.M.; Kara, A.O.; Oğuz, B. West syndrome due to vitamin B12 deficiency. Turk Pediatri Ars. 2015, 50, 251–253. [Google Scholar] [CrossRef]
- Agrawal, S.; Nathani, S. Neuro-regression in vitamin B12 deficiency. BMJ Case Rep. 2009, 2009, bcr0620080235. [Google Scholar] [CrossRef]
- Chong, P.F.; Matsukura, M.; Fukui, K.; Watanabe, Y.; Matsumoto, N.; Kira, R. West Syndrome in an Infant with Vitamin B12 Deficiency Born to Autoantibodies Positive Mother. Front. Pediatr. 2019, 7, 531. [Google Scholar] [CrossRef]
- Tamura, A.; Nino, N.; Yamamoto, N.; Naito, A.; Matsubara, K.; Nakatani, N.; Ichikawa, T.; Nakamura, S.; Saito, A.; Kozaki, A.; et al. Vitamin B12 deficiency anemia in an exclusively breastfed infant born to an ileum-resected mother. Pediatr. Neonatol. 2019, 60, 579–580. [Google Scholar] [CrossRef] [PubMed]
- Lücke, T.; Korenke, G.C.; Poggenburg, I.; Bentele, K.H.; Das, A.M.; Hartmann, H. Maternal vitamin B12 deficiency: Cause for neurological symptoms in infancy. Z. Geburtshilfe Neonatol. 2007, 211, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Glaser, K.; Girschick, H.J.; Schropp, C.; Speer, C.P. Psychomotor development following early treatment of severe infantile vitamin B12 deficiency and West syndrome—Is everything fine? A case report and review of literature. Brain Dev. 2015, 37, 347–351. [Google Scholar] [CrossRef]
- Casella, E.B.; Valente, M.; de Navarro, J.M.; Kok, F. Vitamin B12 deficiency in infancy as a cause of developmental regression. Brain Dev. 2005, 27, 592–594. [Google Scholar] [CrossRef] [PubMed]
- Yaramis, A. A variety of abnormal movements in 13 cases with nutritional cobalamin deficiency in infants. Med. Hypotheses 2020, 142, 109796. [Google Scholar] [CrossRef] [PubMed]
- Taskesen, M.; Yaramis, A.; Pirinccioglu, A.G.; Ekici, F. Cranial magnetic resonance imaging findings of nutritional vitamin B12 deficiency in 15 hypotonic infants. Eur. J. Paediatr. Neurol. 2012, 16, 266–270. [Google Scholar] [CrossRef]
- Pavone, P.; Sullo, F.; Falsaperla, R.; Greco, F.; Crespo, A.; Calvo, A.; Caraballo, R. Vitamin B12 Deficiency and West Syndrome: An Uncommon but Preventable Cause of Neurological Disorder. Report on Three Cases, One of Them with Late Onset during Vitamin B12 Treatment. Neuropediatrics 2021, 52, 333–336. [Google Scholar] [CrossRef]
- Malbora, B.; Yuksel, D.; Aksoy, A.; Ozkan, M. Two infants with infantile spasms associated with vitamin B12 deficiency. Pediatr. Neurol. 2014, 51, 144–146. [Google Scholar] [CrossRef]
- Celiker, M.Y.; Chawla, A. Congenital B12 deficiency following maternal gastric bypass. J. Perinatol. 2009, 29, 640–642. [Google Scholar] [CrossRef]
- Okamura, J.; Miyake, Y.; Kamei, M.; Ito, Y.; Matsubayashi, T. Three infants with megaloblastic anemia caused by maternal vitamin B12 deficiency. Pediatr. Int. 2020, 62, 864–865. [Google Scholar] [CrossRef]
- Reghu, A.; Hosdurga, S.; Sandhu, B.; Spray, C. Vitamin B12 deficiency presenting as oedema in infants of vegetarian mothers. Eur. J. Pediatr. 2005, 164, 257–258. [Google Scholar] [CrossRef] [PubMed]
- Citak, F.E.; Citak, E.C. Severe vitamin B12 deficiency in a breast fed infant with pancytopenia. J. Trop. Pediatr. 2011, 57, 69–70. [Google Scholar] [CrossRef] [PubMed]
- Bicakci, Z. Growth retardation, general hypotonia, and loss of acquired neuromotor skills in the infants of mothers with cobalamin deficiency and the possible role of succinyl-CoA and glycine in the pathogenesis. Medicine 2015, 94, e584. [Google Scholar] [CrossRef] [PubMed]
- Lövblad, K.; Ramelli, G.; Remonda, L.; Nirkko, A.C.; Ozdoba, C.; Schroth, G. Retardation of myelination due to dietary vitamin B12 deficiency: Cranial MRI findings. Pediatr. Radiol. 1997, 27, 155–158. [Google Scholar] [CrossRef] [PubMed]
- Lundgren, J.; Blennow, G. Vitamin B12 deficiency may cause benign familial infantile convulsions: A case report. Acta Paediatr. 1999, 88, 1158–1160. [Google Scholar] [CrossRef] [PubMed]
- Horstmann, M.; Neumaier-Probst, E.; Lukacs, Z.; Steinfeld, R.; Ullrich, K.; Kohlschütter, A. Infantile cobalamin deficiency with cerebral lactate accumulation and sustained choline depletion. Neuropediatrics 2003, 34, 261–264. [Google Scholar] [PubMed]
- Renault, F.; Verstichel, P.; Ploussard, J.P.; Costil, J. Neuropathy in two cobalamin-deficient breast-fed infants of vegetarian mothers. Muscle Nerve 1999, 22, 252–254. [Google Scholar] [CrossRef]
- Dilber, B.; Eyüboğlu, İ. Cranial Magnetic Resonance Imaging Findings in Hypotonic Infants with Cobalamin Deficiency and Combined Methylmalonic Aciduria and Homocystinuria. Klin. Padiatr. 2022, 234, 105–112. [Google Scholar] [CrossRef]
- Grattan-Smith, P.J.; Wilcken, B.; Procopis, P.G.; Wise, G.A. The neurological syndrome of infantile cobalamin deficiency: Developmental regression and involuntary movements. Mov. Disord. 1997, 12, 39–46. [Google Scholar] [CrossRef]
- Muhammad, R.; Fernhoff, P.; Rasmussen, S.; Bowman, B. Neurologic impairment in children associated with maternal dietary deficiency of cobalamin—Georgia, 2001. MMWR Morb. Mortal Wkly. Rep. 2003, 52, 61–64. [Google Scholar]
- Jagadish Kumar, K.; Prudhvi, S.; Balaji, K.; Rahul, R. Persistent diarrhea, hemolytic anemia, and splenohepatomegaly due to Vitamin B12 deficiency in an infant. J. Appl. Hematol. 2018, 9, 148–150. [Google Scholar] [CrossRef]
- Rössler, J.; Breitenstein, S.; Havers, W. Megaloblastäre Anämien durch Vitamin-B12-Mangel im Kindesalter. Monatsschrift Kinderheilkd. 2001, 149, 497–503. [Google Scholar] [CrossRef]
- Belen, B.; Hismi, B.O.; Kocak, U. Severe vitamin B12 deficiency with pancytopenia, hepatosplenomegaly and leukoerythroblastosis in two Syrian refugee infants: A challenge to differentiate from acute leukaemia. BMJ Case Rep. 2014, 2014, bcr2014203742. [Google Scholar] [CrossRef] [PubMed]
- Milankov, O.; Bjelica, M.; Suvajdžić, L.; Maksic, J.; Milankov, V.; Medić, D.; Ilić, N. Vitamin B12-deficient child of a vegan mother. Food Feed. Res. 2019, 46, 219–225. [Google Scholar] [CrossRef]
- Singh, G.; Le, D.; Schnabl, K.; Leaker, M.T.; Steele, M.; Sparkes, R.L. Vitamin B12 Deficiency in Infancy: The Case for Screening. Pediatr. Blood Cancer 2016, 63, 740–742. [Google Scholar] [CrossRef] [PubMed]
- Avci, Z.; Turul, T.; Aysun, S.; Unal, I. Involuntary movements and magnetic resonance imaging findings in infantile cobalamine (vitamin B12) deficiency. Pediatrics 2003, 112, 684–686. [Google Scholar] [CrossRef]
- Emery, E.S.; Homans, A.C.; Colletti, R.B. Vitamin B12 deficiency: A cause of abnormal movements in infants. Pediatrics 1997, 99, 255–256. [Google Scholar] [CrossRef]
- Siddaraju, M.L.; Sathyabama, K.A. Vitamin B12 Deficiency in an Exclusively Breastfed 7-Month-Old Infant Born to a Vegan Mother. Int. J. Sci. Study 2014, 2, 91–93. [Google Scholar]
- Johnson, P.R., Jr.; Roloff, J.S. Vitamin B12 deficiency in an infant strictly breast-fed by a mother with latent pernicious anemia. J. Pediatr. 1982, 100, 917–919. [Google Scholar] [CrossRef]
- Yousif, T.I.; Shukla, P.J.; Gallagher, S. Severe Vitamin B12 Deficiency; an Unusual Cause of Developmental Regression in Infants. Ir. Med. J. 2016, 109, 474. [Google Scholar]
- Serin, H.M.; Arslan, E.A. Neurological symptoms of vitamin B12 deficiency: Analysis of pediatric patients. Acta Clin. Croat. 2019, 58, 295–302. [Google Scholar] [CrossRef]
- Rajasekaran, V.; Sheriff, J.; Moore, H.; McCay, H.; Winstanley, M. Infantile B12 deficiency with severe thrombocytopenia-an under-recognised public health problem? N. Z. Med. J. 2020, 133, 93–96. [Google Scholar] [PubMed]
- Abu-Kishk, I.; Rachmiel, M.; Hoffmann, C.; Lahat, E.; Eshel, G. Infantile encephalopathy due to vitamin deficiency in industrial countries. Childs Nerv. Syst. 2009, 25, 1477–1480. [Google Scholar] [CrossRef] [PubMed]
- Machado, R.; Furtado, F.; Kjöllerström, P.; Cunha, F. Cutaneous hyperpigmentation and cobalamin deficiency. Br. J. Haematol. 2016, 174, 834. [Google Scholar] [CrossRef] [PubMed]
- Van Noolen, L.; Nguyen-Morel, M.A.; Faure, P.; Corne, C. Don’t forget methylmalonic acid quantification in symptomatic exclusively breast-fed infants. Eur. J. Clin. Nutr. 2014, 68, 941–942. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kanra, G.; Cetin, M.; Unal, S.; Haliloglu, G.; Akça, T.; Akalan, N.; Kara, A. Answer to hypotonia: A simple hemogram. J. Child Neurol. 2005, 20, 930–931. [Google Scholar] [CrossRef] [PubMed]
- Lund, A.M. Questions about a vegan diet should be included in differential diagnostics of neurologically abnormal infants with failure to thrive. Acta Paediatr. 2019, 108, 1377–1379. [Google Scholar] [CrossRef]
- Mellin-Sanchez, L.; Sondheimer, N. An Infant Refugee with Anemia and Low Serum Vitamin B12. Clin. Chem. 2018, 64, 1567–1570. [Google Scholar] [CrossRef]
- Ozer, E.A.; Turker, M.; Bakiler, A.R.; Yaprak, I.; Ozturk, C. Involuntary movements in infantile cobalamin deficiency appearing after treatment. Pediatr. Neurol. 2001, 25, 81–83. [Google Scholar] [CrossRef]
- Weber-Ferro, W.; Hertzberg, C.; Röder, H.; Timme, K.; Rossi, R. Intact recovery from early ‘acquired methylmalonic aciduria’ secondary to maternal atrophic gastritis. Acta Paediatr. 2011, 100, e138–e140. [Google Scholar] [CrossRef]
- Patiroglu, T.; Unal, E.; Yildirim, S. Infantile tremor syndrome associated with cobalamin therapy: A case report. Clin. Neurol. Neurosurg. 2013, 115, 1903–1905. [Google Scholar] [CrossRef] [PubMed]
- Dilber, B.; Reis, G.P. Infantile tremor syndrome secondary to peroral vitamin B12 replacement therapy: A report of two cases with myoclonus. Turk. J. Pediatr. 2021, 63, 510–515. [Google Scholar] [CrossRef] [PubMed]
- Danielsson, L.; Enocksson, E.; Hagenfeldt, L.; Rasmussen, E.B.; Tillberg, E. Failure to thrive due to subclinical maternal pernicious anemia. Acta Paediatr. Scand. 1988, 77, 310–311. [Google Scholar] [CrossRef] [PubMed]
- Heaton, D. Another case of megaloblastic anemia of infancy due to maternal pernicious anemia. N. Engl. J. Med. 1979, 300, 202–203. [Google Scholar] [PubMed]
- Choudhry, V.P. Vitamin B12 deficiency in infancy associated with lactose intolerance. Indian J. Pediatr. 1972, 39, 267–269. [Google Scholar] [CrossRef]
- Ozdemir, O.; Baytan, B.; Gunes, A.M.; Okan, M. Involuntary movements during vitamin B12 treatment. J. Child Neurol. 2010, 25, 227–230. [Google Scholar] [CrossRef]
- Baker, S.J.; Mathan, V.I.; Abe, K. Beta-melanocyte stimulating hormone levels in subjects with hyperpigmentation associated with megaloblastic anemia. Blood 1970, 35, 83–85. [Google Scholar] [CrossRef]
- Tosun, A.; Aral, Y.Z.; Çeçen, E.; Aydoğdu, A.; Çetinkaya Çakmak, B. Involuntary movement in infants during vitamin B12 treatment. Turk. J. Haematol. 2011, 28, 317–322. [Google Scholar] [CrossRef]
- Tunçer, G.O.; Köker, A.; Köker, S.A.; Aba, A.; Kara, T.T.; Coban, Y.; Akbas, Y. Infantile Tremor Syndrome after Peroral and Intramuscular Vitamin B12 Therapy: Two Cases. Klin. Padiatr. 2019, 231, 274–277. [Google Scholar] [CrossRef]
- Sharawat, I.K.; Kasinathan, A.; Sankhyan, N. Infantile Tremor Syndrome: Response to B12 Therapy. J. Pediatr. 2018, 196, 323.e1. [Google Scholar] [CrossRef]
- Kamoun, F.; Guirat, R.; Megdich, F.; Ben Ameur, S.; Kallel, C.; Hachicha, M. Frequent Infections, Hypotonia, and Anemia in a Breastfed Infant. J. Pediatr. Hematol. Oncol. 2017, 39, 141–142. [Google Scholar] [CrossRef]
- Banka, S.; Roberts, R.; Plews, D.; Newman, W.G. Early diagnosis and treatment of cobalamin deficiency of infancy owing to occult maternal pernicious anemia. J. Pediatr. Hematol. Oncol. 2010, 32, 319–322. [Google Scholar] [CrossRef]
- Higginbottom, M.C.; Sweetman, L.; Nyhan, W.L. A syndrome of methylmalonic aciduria, homocystinuria, megaloblastic anemia and neurologic abnormalities in a vitamin B12-deficient breast-fed infant of a strict vegetarian. N. Engl. J. Med. 1978, 299, 317–323. [Google Scholar] [CrossRef]
- Codazzi, D.; Sala, F.; Parini, R.; Langer, M. Coma and respiratory failure in a child with severe vitamin B12 deficiency. Pediatr. Crit. Care Med. 2005, 6, 483–485. [Google Scholar] [CrossRef]
- Wighton, M.C.; Manson, J.I.; Speed, I.; Robertson, E.; Chapman, E. Brain damage in infancy and dietary vitamin B12 deficiency. Med. J. Aust. 1979, 2, 1–3. [Google Scholar] [CrossRef]
- Stollhoff, K.; Schulte, F.J. Vitamin B12 and brain development. Eur. J. Pediatr. 1987, 146, 201–205. [Google Scholar] [CrossRef]
- Doyle, J.J.; Langevin, A.M.; Zipursky, A. Nutritional vitamin B12 deficiency in infancy: Three case reports and a review of the literature. Pediatr. Hematol. Oncol. 1989, 6, 161–172. [Google Scholar] [CrossRef]
- Grange, D.K.; Finlay, J.L. Nutritional vitamin B12 deficiency in a breastfed infant following maternal gastric bypass. Pediatr. Hematol. Oncol. 1994, 11, 311–318. [Google Scholar] [CrossRef]
- Yenicesu, I. Pancytopenia due to vitamin B12 deficiency in a breast-fed infant. Pediatr. Hematol. Oncol. 2008, 25, 365–367. [Google Scholar] [CrossRef]
- Kühne, T.; Bubl, R.; Baumgartner, R. Maternal vegan diet causing a serious infantile neurological disorder due to vitamin B12 deficiency. Eur. J. Pediatr. 1991, 150, 205–208. [Google Scholar] [CrossRef] [PubMed]
- Weiss, R.; Fogelman, Y.; Bennett, M. Severe vitamin B12 deficiency in an infant associated with a maternal deficiency and a strict vegetarian diet. J. Pediatr. Hematol. Oncol. 2004, 26, 270–271. [Google Scholar] [CrossRef]
- Kamei, M.; Ito, Y.; Ando, N.; Awaya, T.; Yamada, T.; Nakagawa, M.; Yamaguchi, A.; Ohuchi, M.; Yazaki, M.; Togari, H. Brain atrophy caused by vitamin B12-deficient anemia in an infant. J. Pediatr. Hematol. Oncol. 2011, 33, 556–558. [Google Scholar] [CrossRef]
- Lampkin, B.C.; Saunders, E.F. Nutritional vitamin B12 deficiency in an infant. J. Pediatr. 1969, 75, 1053–1055. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.R., Jr.; Goldenring, J.; Lubin, B.H. Nutritional vitamin B12 deficiency in infants. Am. J. Dis. Child. 1981, 135, 566–567. [Google Scholar] [CrossRef]
- Graham, S.M.; Arvela, O.M.; Wise, G.A. Long-term neurologic consequences of nutritional vitamin B12 deficiency in infants. J. Pediatr. 1992, 121, 710–714. [Google Scholar] [CrossRef]
- Rendle-Short, J.; Tiernan, J.R.; Hawgood, S. Vegan mothers with vitamin B12 deficiency. Med. J. Aust. 1979, 2, 483. [Google Scholar]
- Wardinsky, T.D.; Montes, R.G.; Friederich, R.L.; Broadhurst, R.B.; Sinnhuber, V.; Bartholomew, D. Vitamin B12 deficiency associated with low breast-milk vitamin B12 concentration in an infant following maternal gastric bypass surgery. Arch. Pediatr. Adolesc. Med. 1995, 149, 1281–1284. [Google Scholar] [CrossRef]
- Bobb-Semple, A.A.; Lau, C.S.; Teruya-Feldstein, J.; Wistinghausen, B. A Rare Cause of Pancytopenia in an Exclusively Breastfed Infant. J. Pediatr. Hematol. Oncol. 2019, 41, e322–e324. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.J.; Scott-Jupp, R.; Kohler, J.A. Infantile megaloblastosis secondary to acquired vitamin B12 deficiency. Pediatr. Hematol. Oncol. 1999, 16, 79–81. [Google Scholar] [CrossRef] [PubMed]
- Sobczyńska-Malefora, A.; Ramachandran, R.; Cregeen, D.; Green, E.; Bennett, P.; Harrington, D.J.; Lemonde, H.A. An infant and mother with severe B12 deficiency: Vitamin B12 status assessment should be determined in pregnant women with anaemia. Eur. J. Clin. Nutr. 2017, 71, 1013–1015. [Google Scholar] [CrossRef]
- Bidhuri, N.; Kumar, V.; Singh, R.; Singh, D.P.; Agarwal, S.; Nandan, D. Diaphragmatic palsy in a 10-month-old boy with infantile tremor syndrome causing respiratory failure with full response to vitamin B12 therapy. Paediatr. Int. Child Health 2020, 40, 248–250. [Google Scholar] [CrossRef] [PubMed]
- Gambon, R.C.; Lentze, M.J.; Rossi, E. Megaloblastic anaemia in one of monozygous twins breast fed by their vegetarian mother. Eur. J. Pediatr. 1986, 145, 570–571. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, M.; Webb, J.K.; Vaishnava, S.; Baker, S.J. Vitamin B12 deficiency in Indian infants. A clinical syndrome. Lancet 1962, 2, 903–907. [Google Scholar] [CrossRef]
- Frader, J.; Reibman, B.; Turkewitz, D. Vitamin B12 deficiency in strict vegetarians. N. Engl. J. Med. 1978, 299, 1319–1320. [Google Scholar]
- Honzik, T.; Adamovicova, M.; Smolka, V.; Magner, M.; Hruba, E.; Zeman, J. Clinical presentation and metabolic consequences in 40 breastfed infants with nutritional vitamin B12 deficiency—What have we learned? Eur. J. Paediatr. Neurol. 2010, 14, 488–495. [Google Scholar] [CrossRef]
- Ljungblad, U.W.; Astrup, H.; Mørkrid, L.; Hager, H.B.; Lindberg, M.; Eklund, E.A.; Bjørke-Monsen, A.-L.; Rootwelt, T.; Tangeraas, T. Breastfed Infants with Spells, Tremor, or Irritability: Rule Out Vitamin B12 Deficiency. Pediatr. Neurol. 2022, 131, 4–12. [Google Scholar] [CrossRef] [PubMed]
- Ganesan, S.; Thanawala, N.; Hussain, N. Vitamin B12 deficiency: A treatable cause of developmental delay in infancy. J. Paediatr. Child Health 2013, 49, E348–E349. [Google Scholar] [CrossRef]
- Saritha, K.; Nalini, B.; Anjali, R. B12 Deficiency in a Breastfed Infant Due to Maternal B12 Deficiency: A Case Report. J. Clin. Diagn. Res. 2018, 12, 3–4. [Google Scholar]
- Gotelli, N.; Ellison, A. A Primer of Ecological Statistics; Sinauer Associates: Sunderland, MA, USA, 2004. [Google Scholar]
- Zeuschner, C.L.; Hokin, B.D.; Marsh, K.A.; Saunders, A.V.; Reid, M.A.; Ramsay, M.R. Vitamin B12 and vegetarian diets. Med. J. Aust. 2013, 199, S27–S32. [Google Scholar] [CrossRef]
- Dayaldasani, A.; Ruiz-Escalera, J.; Rodríguez-Espinosa, M.; Rueda, I.; Pérez-Valero, V.; Yahyaoui, R. Serum vitamin B12 levels during the first trimester of pregnancy correlate with newborn screening markers of vitamin B12 deficiency. Int. J. Vitam. Nutr. Res. 2014, 84, 92–97. [Google Scholar] [CrossRef]
- Varsi, K.; Ueland, P.M.; Torsvik, I.K.; Bjørke-Monsen, A.L. Maternal Serum Cobalamin at 18 Weeks of Pregnancy Predicts Infant Cobalamin Status at 6 Months-A Prospective, Observational Study. J. Nutr. 2018, 148, 738–745. [Google Scholar] [CrossRef] [PubMed]
- Ljungblad, U.W.; Paulsen, H.; Mørkrid, L.; Pettersen, R.D.; Hager, H.B.; Lindberg, M.; Astrup, H.; Eklund, E.A.; Bjørke-Monsen, A.L.; Rootwelt, T.; et al. The prevalence and clinical relevance of hyperhomocysteinemia suggesting vitamin B12 deficiency in presumed healthy infants. Eur. J. Paediatr. Neurol. 2021, 35, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Reischl-Hajiabadi, A.T.; Garbade, S.F.; Feyh, P.; Weiss, K.H.; Mütze, U.; Kölker, S.; Hoffmann, G.F.; Gramer, G. Maternal Vitamin B12 Deficiency Detected by Newborn Screening-Evaluation of Causes and Characteristics. Nutrients 2022, 14, 3767. [Google Scholar] [CrossRef] [PubMed]
- Bohn, M.K.; Higgins, V.; Asgari, S.; Leung, F.; Hoffman, B.; Macri, J.; Adeli, K. Paediatric reference intervals for 17 Roche cobas 8000 e602 immunoassays in the CALIPER cohort of healthy children and adolescents. Clin. Chem. Lab. Med. 2019, 57, 1968–1979. [Google Scholar] [CrossRef] [PubMed]
- Abildgaard, A.; Knudsen, C.S.; Hoejskov, C.S.; Greibe, E.; Parkner, T. Reference intervals for plasma vitamin B12 and plasma/serum methylmalonic acid in Danish children, adults and elderly. Clin. Chim. Acta 2022, 525, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Green, R.; Allen, L.H.; Bjørke-Monsen, A.L.; Brito, A.; Guéant, J.L.; Miller, J.W.; Molloy, A.M.; Nexo, E.; Stabler, S.; Toh, B.H.; et al. Vitamin B12 deficiency. Nat. Rev. Dis. Primers 2017, 3, 17040, Erratum in Nat. Rev. Dis. Primers 2017, 3, 17054. [Google Scholar] [CrossRef] [PubMed]
- Huemer, M.; Diodato, D.; Martinelli, D.; Olivieri, G.; Blom, H.; Gleich, F.; Kölker, S.; Kožich, V.; Morris, A.A.; Seifert, B.; et al. EHOD consortium. Phenotype, treatment practice and outcome in the cobalamin-dependent remethylation disorders and MTHFR deficiency: Data from the E-HOD registry. J. Inherit. Metab. Dis. 2019, 42, 333–352. [Google Scholar] [CrossRef]
- Karademir, F.; Suleymanoglu, S.; Ersen, A.; Aydinoz, S.; Gultepe, M.; Meral, C.; Ozkaya, H.; Gocmen, I. Vitamin B12, folate, homocysteine and urinary methylmalonic acid levels in infants. J. Int. Med. Res. 2007, 35, 384–388. [Google Scholar] [CrossRef]
- Victora, C.G.; Bahl, R.; Barros, A.J.; França, G.V.; Horton, S.; Krasevec, J.; Murch, S.; Sankar, M.J.; Walker, N.; Rollins, N.C.; et al. Breastfeeding in the 21st century: Epidemiology, mechanisms, and lifelong effect. Lancet 2016, 387, 475–490. [Google Scholar] [CrossRef]
- Rozmarič, T.; Mitulović, G.; Konstantopoulou, V.; Goeschl, B.; Huemer, M.; Plecko, B.; Spenger, J.; Wortmann, S.B.; Scholl-Bürgi, S.; Karall, D.; et al. Elevated Homocysteine after Elevated Propionylcarnitine or Low Methionine in Newborn Screening Is Highly Predictive for Low Vitamin B12 and Holo-Transcobalamin Levels in Newborns. Diagnostics 2020, 10, 626. [Google Scholar] [CrossRef]
- Gramer, G.; Hoffmann, G.F. Vitamin B12 Deficiency in Newborns and their Mothers-Novel Approaches to Early Detection, Treatment and Prevention of a Global Health Issue. Curr. Med. Sci. 2020, 40, 801–809. [Google Scholar] [CrossRef]
- Tangeraas, T.; Ljungblad, U.W.; Lutvica, E.; Kristensen, E.; Rowe, A.D.; Bjørke-Monsen, A.L.; Rootwelt-Revheim, T.; Sæves, I.; Pettersen, R.D. Vitamin B12 Deficiency (Un-) Detected Using Newborn Screening in Norway. Int. J. Neonatal Screen. 2023, 9, 3. [Google Scholar] [CrossRef] [PubMed]
- Ljungblad, U.W.; Lindberg, M.; Eklund, E.A.; Sæves, I.; Sagredo, C.; Bjørke-Monsen, A.L.; Tangeraas, T. A Retrospective Evaluation of the Predictive Value of Newborn Screening for Vitamin B12 Deficiency in Symptomatic Infants below 1 Year of Age. Int. J. Neonatal Screen. 2022, 8, 66. [Google Scholar] [CrossRef] [PubMed]
- Venkatramanan, S.; Armata, I.E.; Strupp, B.J.; Finkelstein, J.L. Vitamin B-12 and Cognition in Children. Adv. Nutr. 2016, 7, 879–888. [Google Scholar] [CrossRef]
- Lai, J.S.; Mohamad Ayob, M.N.; Cai, S.; Quah, P.L.; Gluckman, P.D.; Shek, L.P.; Yap, F.; Tan, K.H.; Chong, Y.S.; Godfrey, K.M.; et al. Maternal plasma vitamin B12 concentrations during pregnancy and infant cognitive outcomes at 2 years of age. Br. J. Nutr. 2019, 121, 1303–1312. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.; Thomas, T.; Bosch, R.J.; Ramthal, A.; Bellinger, D.C.; Kurpad, A.V.; Duggan, C.P.; Srinivasan, K. Effect of Maternal Vitamin B12 Supplementation on Cognitive Outcomes in South Indian Children: A Randomized Controlled Clinical Trial. Matern. Child Health J. 2019, 23, 155–163. [Google Scholar] [CrossRef]
- Tu, S.J.; Wei, Y.J.; Chen, B.T.; Zhang, X.F.; Luo, C.; Dong, B.Q. Effects of a false-positive result in newborn congenital hypothyroidism screening on parents in Guangxi, China. Front. Pediatr. 2023, 11, 1134923. [Google Scholar] [CrossRef]
- Oliver, E.M.; Grimshaw, K.E.C.; Schoemaker, A.A.; Keil, T.; McBride, D.; Sprikkelman, A.B.; Ragnarsdottir, H.S.; Trendelenburg, V.; Emmanouil, E.; Reche, M.; et al. Dietary habits and supplement use in relation to national pregnancy recommendations: Data from the EuroPrevall birth cohort. Matern. Child Health J. 2014, 18, 2408–2425. [Google Scholar] [CrossRef]




| Variable | Total Sample (N = 292) (% Values Relate to the Number of Reports for Each Variable) |
|---|---|
| Sex female, n (%) | 103 (37) |
| Age at first symptoms (months), M (SD), range | 4 (3), 0–12 |
| Age at diagnosis (months), M (SD), range | 7 (4), 0–30 |
| Diagnostic delay (months), M (SD), range | 3 (3), 0-21 |
| Serum B12 infant log, M (SD), range | 5 (1), 1–7 |
| Serum B12 infant pmol/L, M (SD), range | 107 (87), 2–552 |
| # Probable B12 deficiency, n (%) | 204 (78) |
| # Possible B12 deficiency, n (%) | 38 (15) |
| # Unlikely B12 deficiency, n (%) | 19 (7) |
| Plasma MMA infant log, M (SD), range | 1 (1.35), −2–4 |
| Plasma MMA infant µmol/L, M (SD), range | 5 (9), 0–46 |
| Plasma tHcy infant log, M (SD), range | 3 (1), 0–5 |
| Plasma tHcy infant µmol/L, M (SD), range | 51 (56), 1–239 |
| Serum folate infant log, M (SD), range | 7 (1), −1–5 |
| Serum folate infant nmol/L, M (SD), range | 36 (21), 0–211 |
| Serum B12 mother log, M (SD), range | 5 (1), 3–7 |
| Serum B12 mother pmol/L, M (SD), range | 154 (109), 10–709 |
| Clinical symptoms: Failure to thrive, n (%) | 89 (86) |
| Irritability, n (%) | 38 (43) |
| Anemia, n (%) | 186 (69) |
| Cerebral Atrophy, n (%) | 58 (72) |
| Hypotonia, n (%) | 165 (96) |
| Apathy/lethargy, n (%) | 86 (61) |
| Developmental delay, n (%) | 146 (93) |
| Movement disorder, n (%) | 74 (56) |
| Maternal Diet: Vegan, n (%) | 35 (17) |
| Vegetarian, n (%) | 29 (14) |
| Mostly vegetarian, n (%) | 16 (8) |
| Mixed, n (%) | 126 (61) |
| Maternal B12 malabsorption (with supplementation), n (n) | 52 (4) |
| Maternal B12 supplementation at diagnosis without malabsorption, n (%) | 35 (38) |
| Diet of the infant: Exclusive breastfeeding, n (%) | 224 (86) |
| Partial breastfeeding, n (%) | 12 (7) |
| Partial formula, n (%) | 3 (7) |
| Solid foods vegetarian, n (%) | 2 (1) |
| Solid foods vegan, n (%) | 3 (2) |
| Solid foods mixed, n | 0 |
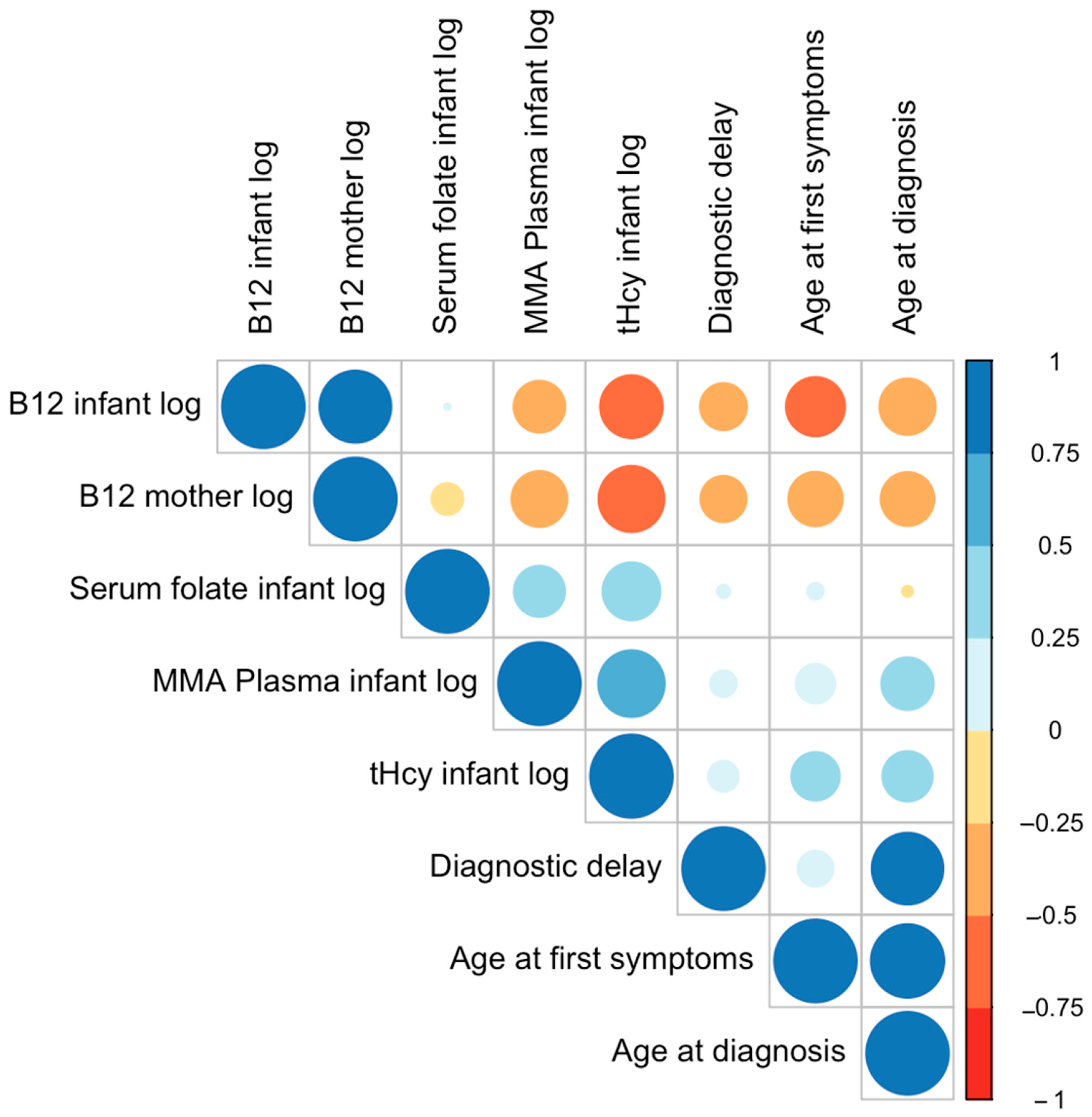
| Variable | M | SD | Age at First Symptoms (Months) | Age at Diagnosis (Months) | Diagnostic Delay (Months) | B12 Infant Log | MMA Plasma Infant Log | tHcy Infant Log | Serum Folate Infant Log |
|---|---|---|---|---|---|---|---|---|---|
| Age at first symptoms | 3.99 | 2.97 | |||||||
| Age at diagnosis | 7.14 | 4.03 | 0.79 ** [0.73, 0.84] | ||||||
| Diagnostic delay | 2.58 | 2.72 | 0.19 ** [0.05, 0.32] | 0.75 ** [0.68, 0.81] | |||||
| B12 infant log | 4.67 | 0.81 | −0.51 ** [−0.61, −0.40] | −0.46 ** [−0.56, −0.36] | −0.32 ** [−0.45, −0.19] | ||||
| MMA Plasma infant log | 0.66 | 1.35 | 0.23 * [0.01, 0.43] | 0.41 ** [0.21, 0.57] | 0.11 [−0.12, 0.32] | −0.39 ** [−0.57, −0.18] | |||
| tHcy infant log | 3.31 | 1.14 | 0.34 ** [0.18, 0.49] | 0.37 ** [0.23, 0.50] | 0.14 [−0.03, 0.31] | −0.58 ** [−0.68, −0.45] | 0.65 ** [0.50, 0.76] | ||
| Serum folate infant log | 2.66 | 0.58 | 0.04 [−0.14, 0.22] | −0.02 [−0.19, 0.15] | 0.03 [−0.16, 0.21] | 0.01 [−0.17, 0.18] | 0.38 ** [0.15, 0.58] | 0.49 ** [0.31, 0.64] | |
| B12 mother log | 5.14 | 0.65 | −0.43 ** [−0.55, −0.30] | −0.42 ** [−0.52, −0.31] | −0.31 ** [−0.44, −0.17] | 0.76 ** [0.70, 0.81] | −0.46 ** [−0.62, −0.025] | −0.64 ** [−0.73, −0.53] | −0.15 [−0.33, 0.04] |
| Count Variable | n | Chi-Squared | p | |
|---|---|---|---|---|
| Anemia | No Anemia | |||
| Irritability | 24 | 12 | 14 | <0.001 |
| Hypotonia | 108 | 40 | 8 | <0.01 |
| Apathy/lethargy | 75 | 8 | 52 | <0.001 |
| Apathy/lethargy | No apathy/lethargy | |||
| Irritability | 16 | 5 | 23 | <0.001 |
| Developmental delay | 69 | 1 | 5 | <0.05 |
| Movement disorders | 29 | 12 | 12 | <0.001 |
| Count Variable | n | Chi-Squared | p | ||
|---|---|---|---|---|---|
| Probable B12 Deficiency | Possible B12 Deficiency | Unlikely B12 Deficiency | |||
| Irritability | 25 | 4 | 4 | 19 | <0.001 |
| Anemia | 149 | 9 | 2 | 69 | <0.001 |
| Hypotonia | 122 | 12 | 11 | 9 | <0.05 |
| Apathy/lethargy | 37 | 1 | 1 | 60 | <0.001 |
| Movement disorder | 46 | 13 | 6 | 7 | <0.05 |
| Exclusive breastfeeding | 158 | 26 | 13 | 13 | <0.05 |
| Vegan diet Mixed diet | 25 | 0 | 0 | 41 | <0.001 |
| Vegetarian diet | 27 | 0 | 0 | ||
| Mixed diet | 64 | 35 | 17 | ||
| Mostly vegetarian diet | 14 | 0 | 0 | ||
| Predictor | M | F | p | ||
|---|---|---|---|---|---|
| Probable Deficiency | Possible Deficiency | Unlikely Deficiency | |||
| A. Criterion: maternal B12 levels | |||||
| (Intercept) | 760 | <0.001 | |||
| B12 infant grouped | 163 | 385 | 421 | 80 | <0.001 |
| B. Criterion: age at diagnosis | |||||
| (Intercept) | 192 | <0.001 | |||
| B12 infant grouped | 8 | 4 | 3 | 39 | <0.001 |
| C. Criterion: age at first symptoms | |||||
| (Intercept) | 106 | <0.001 | |||
| B12 infant grouped | 5 | 2 | 1 | 27 | <0.001 |
| A Predictor | b | b 95% CI [LL, UL] |
| (Intercept) | 0.65 | [−0.90, 2.20] |
| Maternal B12, log | 0.79 ** | [0.55, 1.03] |
| Sex female | 0.11 | [−0.13, 0.36] |
| Age at first symptoms | −0.03 | [−0.09, 0.04] |
| Age at diagnosis | −0.04 | [−0.08, 0.01] |
| Serum folate infant, log | 0.11 | [−0.09, 0.31] |
| (Intercept, reference category vegan) | 4.52 ** | [3.48, 5.56] |
| Maternal diet vegetarian | 0.42 | [−0.14, 0.99] |
| Maternal mixed | 0.72 ** | [0.23, 1.22] |
| Maternal mostly vegetarian | 0.36 | [−0.57, 1.28] |
| Sex female | 0.08 | [−0.18, 0.33] |
| Age at first symptoms | −0.06 | [−0.13, 0.02] |
| Age at diagnosis | −0.06 * | [−0.11, −0.00] |
| Serum folate infant, log | 0.14 | [−0.22, 0.50] |
| (Intercept) | 5.95 ** | [5.27, 6.63] |
| Breastfeeding exclusively | −0.58 ** | [−0.88, −0.27] |
| Sex female | 0.10 | [−0.14, 0.35] |
| Age at first symptoms | −0.05 | [−0.12, 0.02] |
| Age at diagnosis | −0.10 ** | [−0.15, −0.05] |
| Serum folate infant, log | 0.07 | [−0.14, 0.28] |
| (Intercept) | 5.36 ** | [4.74, 5.99] |
| Age at first symptoms | −0.08 * | [−0.15, −0.01] |
| Age at diagnosis | −0.07 ** | [−0.12, −0.02] |
| Sex female | 0.19 | [−0.07, 0.44] |
| Serum folate infant, log | 0.08 | [−0.14, 0.30] |
| (Intercept) | 5.36 ** | [4.74, 5.99] |
| Age at diagnosis | −0.07 ** | [−0.12, −0.02] |
| Age at first symptoms | −0.08 * | [−0.15, −0.01] |
| Sex female | 0.19 | [−0.07, 0.44] |
| Serum folate infant, log | 0.08 | [−0.14, 0.30] |
| (Intercept) | 5.03 ** | [4.30, 5.77] |
| Diagnostic delay | −0.09 ** | [−0.15, −0.04] |
| Sex female | 0.22 | [−0.08, 0.52] |
| Serum folate infant, log | 0.06 | [−0.20, 0.32] |
| B Predictor | b | b 95% CI [LL, UL] |
| (Intercept) | 3.40 * | [0.76, 6.04] |
| Maternal B12, log | −0.51 ** | [−0.86, −0.16] |
| Sex female | 0.11 | [−0.22, 0.44] |
| Age at first symptoms | −0.05 | [−0.15, 0.05] |
| Age at diagnosis | 0.02 | [−0.05, 0.09] |
| Serum folate infant, log | 0.83 ** | [0.35, 1.31] |
| (Intercept, reference category vegan) | 1.76 ** | [0.44, 3.07] |
| Maternal diet vegetarian | −0.55 | [−1.70, 0.60] |
| Maternal mixed | −1.00 * | [−1.81, −0.20] |
| Maternal mostly vegetarian | 0.53 | [−0.85, 1.91] |
| Sex female | 0.18 | [−0.09, 0.44] |
| Age at first symptoms | −0.02 | [−0.10, 0.07] |
| Age at diagnosis | 0.01 | [−0.05, 0.07] |
| Serum folate infant, log | 0.66 ** | [0.24, 1.09] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wirthensohn, M.; Wehrli, S.; Ljungblad, U.W.; Huemer, M. Biochemical, Nutritional, and Clinical Parameters of Vitamin B12 Deficiency in Infants: A Systematic Review and Analysis of 292 Cases Published between 1962 and 2022. Nutrients 2023, 15, 4960. https://doi.org/10.3390/nu15234960
Wirthensohn M, Wehrli S, Ljungblad UW, Huemer M. Biochemical, Nutritional, and Clinical Parameters of Vitamin B12 Deficiency in Infants: A Systematic Review and Analysis of 292 Cases Published between 1962 and 2022. Nutrients. 2023; 15(23):4960. https://doi.org/10.3390/nu15234960
Chicago/Turabian StyleWirthensohn, Miriam, Susanne Wehrli, Ulf Wike Ljungblad, and Martina Huemer. 2023. "Biochemical, Nutritional, and Clinical Parameters of Vitamin B12 Deficiency in Infants: A Systematic Review and Analysis of 292 Cases Published between 1962 and 2022" Nutrients 15, no. 23: 4960. https://doi.org/10.3390/nu15234960
APA StyleWirthensohn, M., Wehrli, S., Ljungblad, U. W., & Huemer, M. (2023). Biochemical, Nutritional, and Clinical Parameters of Vitamin B12 Deficiency in Infants: A Systematic Review and Analysis of 292 Cases Published between 1962 and 2022. Nutrients, 15(23), 4960. https://doi.org/10.3390/nu15234960








