Structure, Health Benefits, Mechanisms, and Gut Microbiota of Dendrobium officinale Polysaccharides: A Review
Abstract
:1. Introduction
2. Extraction and Structure of Polysaccharides from Dendrobium officinale
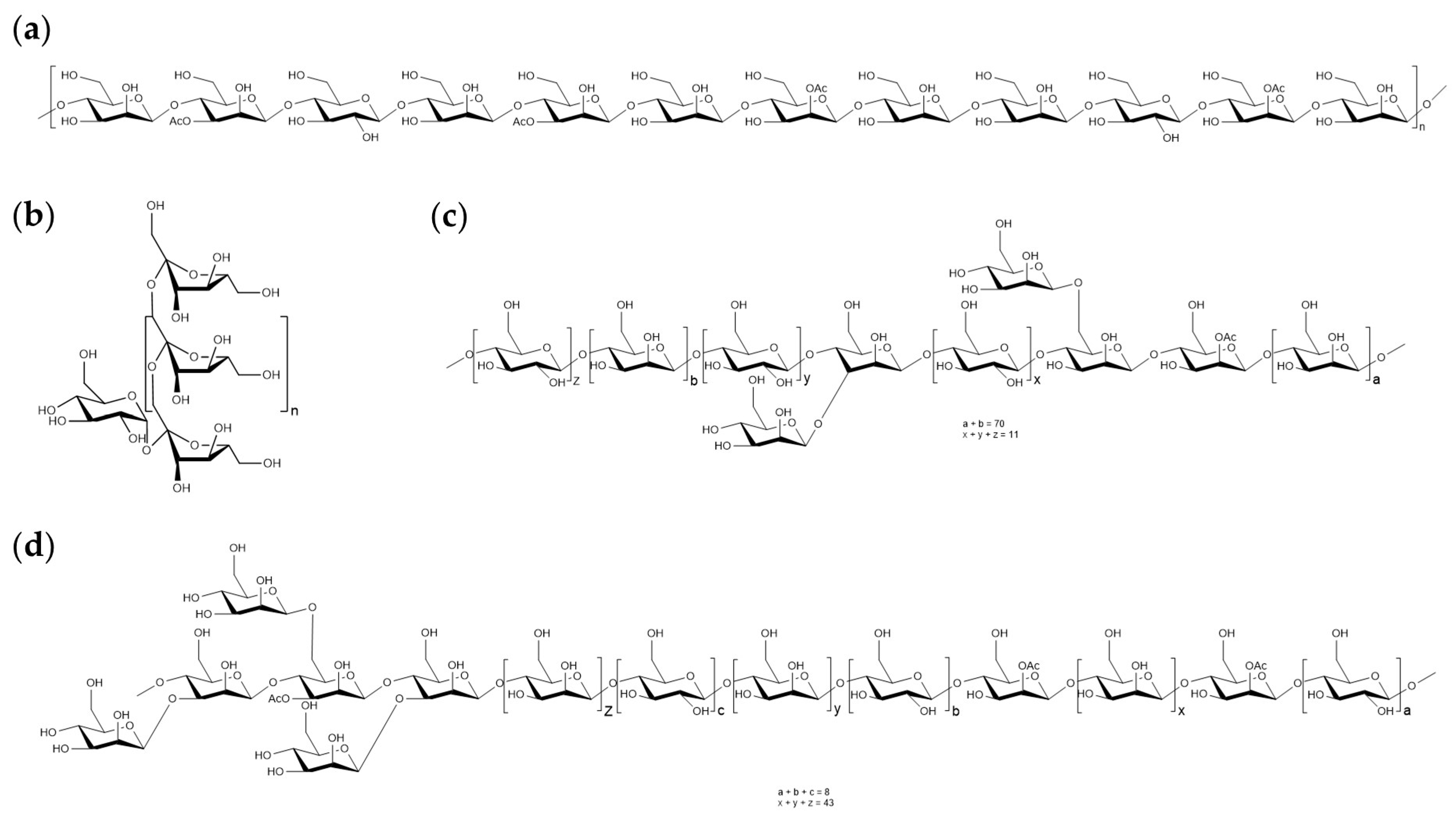
| Compound | Extraction Methods | Yield/% | Monosaccharide Compositions | Structures | Mw/Da | Ref. |
|---|---|---|---|---|---|---|
| HDOP | 70 °C deionized water extraction, 75% ethanol precipitation | 19.12% | Manp/Glup = 6.9:1.0 | Backbone chain: →4)-2,6-di-O-acetyl-β-d-Manp-(1→, →4)-3,6-di-O-acetyl-β-d-Manp-(1→, and →4)-6-O-acetyl-β-d-Manp-(1→ | 3.12 × 105 | [37] |
| DOP | 70~75 °C hot-water extraction and 85% ethanol precipitation | Unknown | Manp/Glup/Arap/Galp = 6.2: 2.3:2.1:0.1. | Backbone consisting of 2-O-acetyl-Manp and (1→4)-linked-β-d-Manp and (1→4)-linked-β-d-Glcp residues | 8.5 × 103 | [38] |
| DOPs | 80 °C distilled water extraction, 75% soluble component | 2.76% | Manp/Glup = 5.78:1 | Backbone consisting of α-(1→3)-Glcp, branch consisting of α-(1→4)-Glcp and β-(1→4)-Manp | 4.56 × 103 | [30] |
| DOPA-1 | 90 °C deionized water extraction, 85% ethanol precipitation | Unknown | Manp/Glup/Galp = 1:0.42:0.27 | Backbone consisting of (1→3), (1→2), and (1→6) linkages. | 2.29 × 105 | [39] |
| DOP-1-A1 | 100 °C distilled water and 1% polyvinylpyrrolidone extraction, 60% alcohol precipitation | 3.16% | Manp/Glup/Arap = 40.2:8.4:1.0 | Backbone consisting of (1→4)-linked β-d-Manp and β-d-Glcp (in 6:1 ratio) | 1.30 × 105 | [29] |
| DOPa | 100 °C deionized water extraction, 71.25% ethanol precipitation | Unknown | Manp/Glup = 5.6: 1.0 | Backbone consisting of β-(1→4)-D-Manp, β-(1→4)-D-Glcp residues | 8.1 × 105 | [40] |
| DOPS-1 | Boiling water extraction, 80% ethanol precipitation | Unknown | Manp/Glup/Galp = 3.2: 1.3: 1.0 | Backbone consisting of (1→4)-β-D-Glcp, (1→4)-β-D-Manp and 2-O-acetyl-(1→4)-β-D-Manp | 1.53 × 106 | [41] |
| DOP-W3-b | 100 °C deionized water extraction, 80% ethanol precipitation | 1.59% | Manp/Glup = 4.5:1.0 | Backbone consisting of β-(1→4)-D-Manp, β-(1→4)-D-Glcp, and β-(1→3,6)-D-Manp residues; branch consisting of β-(1→4)-D-Manp, β-(1→4)-D-Glcp, and terminal β-D-Glcp, and O-acetyl groups attached to O-2 of β-(1→4)-D-Manp | 1.543 × 104 | [42] |
| DOP1-DES | Deep eutectic solvents extraction | Unknown | Manp/Glup = 2.2:1.0 | Backbone consisting of (1→4)-β-D-Manp and (1→4)-β-D-Glcp | 2.98 × 105 | [22] |
| DO | Mechanochemical-assisted extraction | Unknown | Unknown | Unknown | 0.66~6.19 × 105 | [26] |
| FP | Flash Extraction | 26.3% | Manp/Glup = 3.75~25.60:1 | Unknown | Unknown | [27] |
| DOPCP | Cold pressing | 13.79% | Manp/Glup = 3.71:1 | Unknown | 3.78 × 105 | [4] |
| DOPFTCP | Freeze–thawing cold pressing | 20.33% | Manp/Glup = 3.63:1 | Unknown | 3.69 × 105 | [4] |
| DOPUHWE | Ultrasonic-assisted hot water extraction | 20.55% | Manp/Glup = 2.29:1 | Unknown | 1.97 × 105 | [4] |
| DOPMHWE | Microwave-assisted hot water extraction | 17.74% | Manp/Glup = 3.57:1 | Unknown | 3.24 × 105 | [4] |
| DOPEHWE | Enzyme-assisted hot water extraction | 18.50% | Manp/Glup = 2.01:1 | Unknown | 2.98 × 105 | [4] |
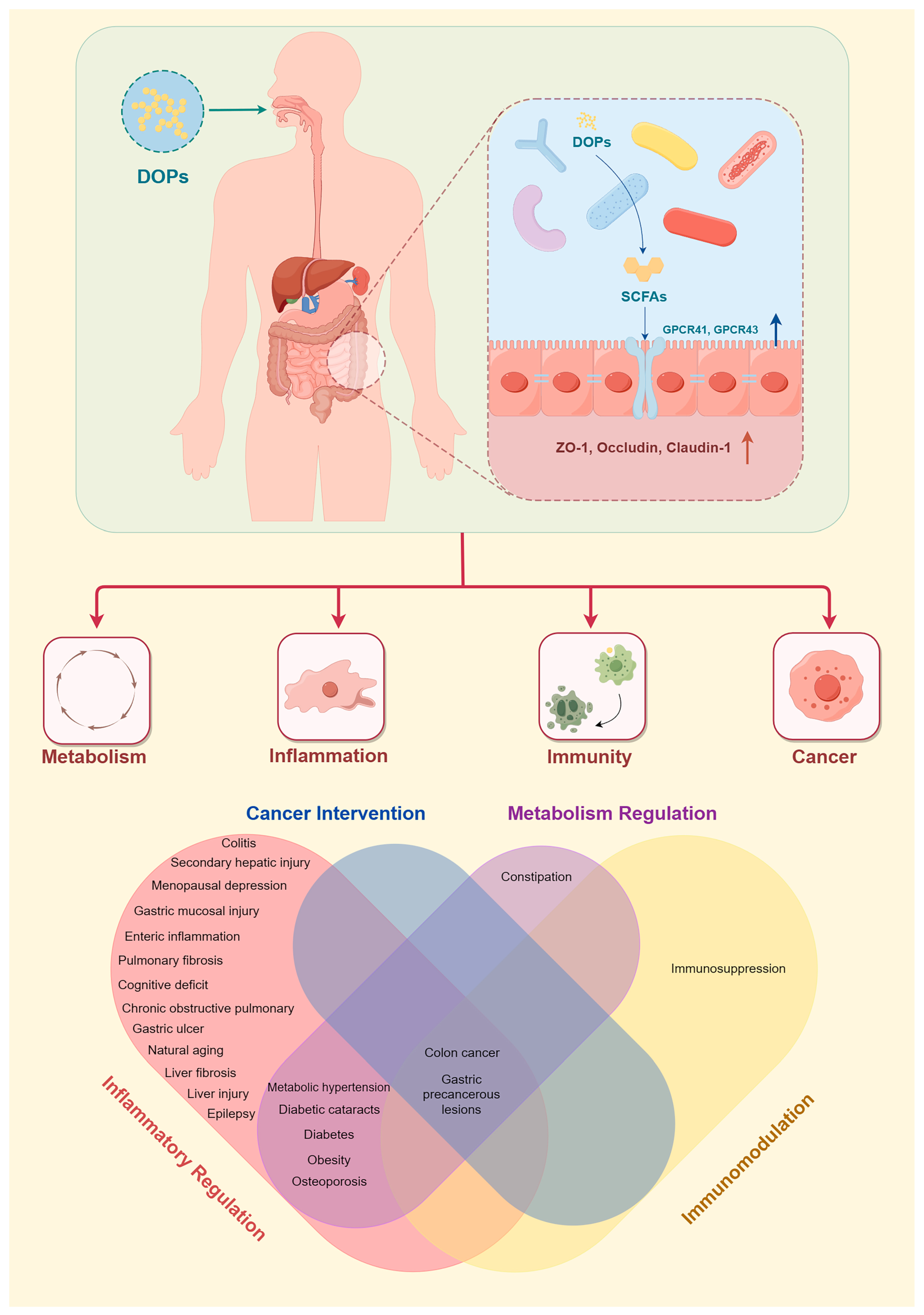
3. Dendrobium officinale Polysaccharides–Host Interactions: Health Benefits, Cellular Mechanisms, and the Structure–Function Relationship
3.1. Metabolism Regulations
| Substance | Object | Dosage and Duration | Health Outcome | Potential Mechanism | Ref. |
|---|---|---|---|---|---|
| Unknown | HFD-induced obese male KM mice | 350 mg/kg/d intervention for 8 weeks | Improve intestinal mucosal barrier function. | Regulate gut microbial composition, carbohydrate metabolism, and SCFAs. | [53] |
| Manp/Glup/Galp = 59.23:35.82:1.61 Mw = unknown | HFD-induced obese male C57BL/6 mice | 1.0 g/kg/d intervention for 11 weeks | Alleviate obesity and hepatic steatosis; improve insulin pathway, hepatic glycogen synthesis, and glucose homeostasis; upregulate energy metabolism; increase acetic acid and taurine. | Increase the abundance of beneficial bacteria and SCFAs; upregulate UCP1, PGC-1α, Gck, Gys2, ZO-1, and occludin; downregulate Pck1 and LPS. | [12] |
| Manp/Glup/Arap = 5.5:1:0.12 Mw = 393.8 kDa | HFD-induced obese male C57BL/6 and ob/ob mice | 150 mg/kg/d intervention for 3 months | Improve lipid metabolism, insulin resistance, and purine metabolism; increase glucose tolerance. | Unknown. | [13] |
| Manp/Glup = 6.9:1 Mw = 312 kDa | STZ-induced type 2 diabetic male Wistar rats | 20, 40, 80, 160 mg/kg/d intervention for 8 weeks | Improve lipid metabolism, insulin resistance, and the metabolism of fatty acid, glycerolipid, sphingolipid, phospholipid, bile acid, and amino acid; decrease oxidative stress. | Upregulate GLP-1. | [50] |
| Manp/Glup =1.38:1.00 Mw = 395 kDa | STZ-induced type 2 diabetic male Balb/c mice | 100, 200, 400 mg/kg/d intervention for 4 weeks | Improve hyperglycemia, lipid metabolism, and insulin resistance. | Activate the PI3K/Akt signaling pathways; upregulate IRS1, PI3K, Akt, GS, GLUT4, IR-β, and IR-β; downregulate PPAR-γ, JNK, GSK-3β, and PTP 1B. | [14] |
| Manp/Glup = 6.9:1 Mw = 312 kDa | STZ-induced type 2 diabetic male Wistar rats | 160 mg/kg/d intervention for 4 weeks | Improve hyperglycemia, lipid metabolism, and insulin resistance; normalize the glomerular structure and function in diabetic nephropathy; decrease serum uric acid, urea, and Crea-J. | Improve the metabolic of purine, aldehyde/acetate, purine, tyrosine, bile acid biosynthesis, pyrimidine, glycine/serine, gluconeogenesis, amino sugars, citric acid cycle, and aspartate. | [15] |
| DOPS-1 | HFD-induced type 2 diabetic male Wistar rats | Unknown | Improve hyperglycemia, insulin resistance, and the metabolic disorders of branched-chain amino acids, saccharides, cholic acids, nucleotides, carnitine, indoles, and lipids. | Improve branched-chain amino acid metabolism by decreasing the microbial abundance of valine, leucine, and isoleucine. | [10] |
| Manp/Glup = 4.41:1.00 Mw = 190 kDa | HFD/STZ-induced type 2 diabetes in male C57BL/6J mice | 100, 200, 400 mg/kg/d for 4 weeks | Improve hyperglycemia, insulin resistance, hepatic glycogen synthesis, the stability of hepatic glycogen structure, and liver glucose metabolism; inhibit hepatic glycogenolysis and gluconeogenesis. | Upregulate GS and Akt/Fox01; downregulate G6Pase, PEPCK, cAMP-PKA, and GP. | [49] |
| Manp/Glup = 6.9:1 Mw = 312 kDa | STZ-induced type 2 diabetes in male Wistar rats | 20, 40, 80, 160 mg/kg/d for 8 weeks | Improve the metabolism disorders of methionine, fatty acid, triglyceride, glycerophospholipid, sphingolipid, bile acid, and carbohydrate; normalize levels of dipeptides, hemolytic phospholipids, salicylates, and others. | Unknown. | [48] |
| Manp/Glup/Arap/Galp = 1.0:1.5:3.5:1.2 Mw = unknown | Alloxan-induced diabetes in male ICR mice | 40 mg/kg/d for 3 weeks | Improve hyperglycemia; increase the serum insulin. | Unknown. | [54] |
| The content of Glup, Manp, Galp, and GalA is 56.24%, 18.68%, 3.67%, 1.29% Mw = 312 kDa Uronic acids = 3.02% | HFD-induced type 2 diabetes mellitus in male C57BL/6J mice | 200 mg/kg/d for 8 weeks | Improve hyperglycemia, lipid metabolism, and insulin resistance; improve mitochondrial function in the brain cortex, inhibit apoptosis of brain neurons, and enhance the tricarboxylic acid cycle in the brain cortex; decrease oxidative stress. | Upregulate Bcl-2, Bcl-2/Bax, 5hmC, 5fC, TET2, and p-AMPK/AMPK; downregulate cleaved caspase 3/caspase 3. | [52] |
| Manp/Glup = 5.18:1 and 4.78:1 Mw = 6.8 kDa and 14.3 kDa | STZ/HFD-induced diabetes in male SD mice | 25 and 100 mg/kg/d for 28 days | Improve hyperglycemia; increase the serum insulin. | Upregulate GLP-1; regulate the Ca2+/CaM/CaMKII and MAPK signaling pathways. | [35] |
| Manp/Glup = 1.9:1.0 Mw = unknown | Excessive alcohol consumption and HFD-induced metabolic hypertension in male SD rats | 200 mg/kg/d for 7 weeks | Decrease the blood pressure; improve lipid metabolism, intestinal barrier, and endothelial function. | Regulate intestinal microbial composition and SCFAs; activate the SCFA-GPCR43/41 pathways; upregulate occludin, claudin, ZO-1, GPCR41, GPCR41/43, aortic eNOS, and serum NO. | [55] |
| The content of Manp and Glup is 59.19 and 830.98 mg/g Mw = 8.404 kDa | Difenoxin or deprivation of water-induced constipation in ICR mice | 29, 57, 114 mg/kg once a time | Improve constipation; increase intestinal transit rate; facilitate stool evacuation characteristics. | Upregulate motilin, gastrin, acetyl cholinesterase, and substance P; downregulate somatostatin. | [56] |
| Manp/Glup = 69.70:30.30 Mw = 731 kDa | HFD/STZ-induced type 2 diabetes mellitus in male C57BL/6J mice | 120 mg/kg/d for 8 weeks | Improve hyperglycemia, lipid metabolism, insulin resistance, and tissue damage; decrease oxidative stress. | Regulate gut microbial composition. | [57] |
3.2. Inflammation Modulation
- (1)
- DOPs exert anti-inflammatory effects by modulating the composition of the gut microbiota and promoting the secretion of SCFAs. Transplantation of the fecal microbiota has shown that the anti-inflammatory, antioxidant and stress-relieving effects of DOPs are transferable to obese mice induced by HFD [12].
- (2)
- DOPs restore intestinal barrier function and protect the intestinal mucosal barrier. In animal models, DOPs enhance the expression of intestinal epithelial tight junction proteins such as occludin and ZO-1, thereby maintaining a stable mucosal barrier, strengthening intestinal barrier function, reducing lipopolysaccharide (LPS) translocation, and lowering serum LPS levels [62,63]. In addition, DOPs reduce the expression of the LPS binding site toll-like receptor 4 (TLR4), inhibit the NF-κB signaling pathway, and reduce the secretion of inflammatory cytokines [58,63].
- (3)
- DOPs alleviate intestinal inflammation by promoting the packaging of miR-433-3p into extracellular vesicles (EVs). Upregulation of miR-433-3p was observed in a DSS-induced colitis mouse model, and DOPs were found to regulate hnRNPA2B1 to facilitate the engulfment of miR-433-3p into gut-derived small EVs (sEVs) in Caco2 cells. Subsequently, these sEVs are transported to LPS-induced macrophages in the lamina propria of the intestine, where miR-433-3p targets the MAPK8 gene, inhibits MAPK signaling pathways, and decreases the secretion of TNF-α and IL-6 by macrophages [64].
| Substance | Object | Dosage and Duration | Health Outcome | Potential Mechanism | Ref. |
|---|---|---|---|---|---|
| Manp/Glup/Arap = 5.5:1:0.12 Mw = 393.8 kDa | HFD-induced obesity in male C57BL/6 mice and ob/ob mice | 150 mg/kg/d for 3 months | Improve insulin resistance and visceral adipose tissue inflammation. | Unknown. | [13] |
| Manp/Glup = 6.9:1 Mw = 312 kDa | STZ-induced type 2 diabetes in male Wistar rats | 20, 40, 80, 160 mg/kg/d for 8 weeks | Improve proliferation of adipocytes; decrease inflammatory infiltration, glycoprotein deposition on capillary basement membrane, and oxidative stress in adipose tissue cells. | Unknown. | [50] |
| Manp/Glup/Arap Mw = 393.8 kDa | Osteoporosis in elderly mice | 150 mg/kg/d for 3 months | Increase bone formation rate and mineral absorption; decrease bone marrow adipose tissue accumulation and oxidative stress. | Activate the Nrf-2 signaling pathways; upregulate Nrf-2 and HO-1. | [65] |
| Manp/Glup = 6.9:1 Mw = 312 kDa | STZ-induced type 2 diabetes in male Wistar rats | 20, 40, 80, 160 mg/kg/d for 8 weeks. | Improve hepatic oxidative stress and inflammation. | Unknown. | [48] |
| Manp/Glup = 6.9:1 Mw = 312 kDa | LPS-induced enteric inflammation in male C57BL/6 mice | 160 mg/kg/d for 2 weeks | Inhibit Th1 cell responses in serum and spleen, exerting systemic anti-inflammatory effects; induce Th17 cell differentiation in spleen and mesenteric lymph nodes. | Upregulate Rorc; down-regulate Foxp3, Tbx21, and HIF-1. | [6] |
| Unknown | STZ-induced diabetic cataracts in male Wistar rats | 0.1 g/kg two times a day for 12 weeks | Decrease the severity of diabetic cataracts. | Inhibit The MAPK signaling pathways; downregulate ERK1, Raf, Ras, and MiRNA-125b. | [69] |
| Manp/Glup = 1.9:1.0 Mw = unknown | Excessive alcohol consumption and HFD-induced metabolic hypertension in male SD rats | 200 mg/kg/d for 7 weeks | Improve lipid metabolism, intestinal barrier, and endothelial function; alleviate hepatic inflammation lesions and ameliorate fatty inflammation. | Regulate gut microbial composition and SCFAs; activate the SCFA-GPCR43/41 signaling pathways; upregulate GPCR41, GPCR41/43, occludin, claudin, ZO-1, aortic eNOS, and serum NO. | [55] |
| The content of Manp and Glup is 57.3 mg/g and 670.2 mg/g | 40 cases of moderate chronic obstructive pulmonary disease patients in China | 1.2 g three times a day for 12 weeks | Improve serum and pulmonary inflammation; increase patient lung capacity to improve pulmonary function. | Upregulate aquaporin-5; downregulate mucin-5AC. | [16] |
| The content of Manp and Glup = 57.3 mg/g and 670.2 mg/g | Passive smoking models in male SD rats | 50, 200 mg/kg/d for 28 days | Alleviate infiltration of inflammatory cells in lung tissue; decrease lymphocyte and monocyte counts in serum and oxidative stress in lung. | Inhibit the ERK, p38 MAPK, and NF-κB signaling pathways; downregulate MCP-1 and CINC-1. | [17] |
| Manp and Glup in a molar percent of 71.2% and 98.1% | Bleomycin-induced pulmonary fibrosis in male SD rats | 200 mg/kg/d for 28 days | Improve pulmonary fibrosis and inflammation; reduce collagen deposition; decrease the transformation of rat alveolar epithelial type 2 cells into myofibroblasts. | Inhibit the TGFβ1-Smad2/3 signaling pathways; downregulate serum TGFβ1, Smad2/3, pSmad2/3, collagen I, and fibronectin protein expression. | [70] |
| Manp and Glup = 3.8:1.0 Mw = 132 kDa | Ethanol-induced gastric mucosal injury in male Sprague Dawley rats | 124, 248 mg/kg/d for 7 days | Improve gastric mucosal injury; inhibit ethanol-induced mucosal protein loss and cell apoptosis. | Upregulate Bcl-2; downregulate Bax. | [71] |
| Manp and Glup = 3.8:1.0 Mw = 132 kDa | H2O2-induced HFE145 cells | 50 μg/mL and 500 μg/ml | Protect cells and inhibit apoptosis. | Activate PPAR signaling pathways; downregulate p-NF-κBp65/NF-κBp65, Bax, and cleaved caspase 3; upregulate Bcl-2. | [71] |
| Manp/Glup/Arap = 5.55:1:0.12 Mw = 393 kDa | DSS-induced colitis in male Balb/c mice | 50, 100, 200 mg/kg/d for 7 days | Improve colitis-induced lung injury and pulmonary edema; decrease inflammatory cell infiltration, inflammatory response, and oxidative stress. | Activate the Nrf-2 signaling pathways; upregulate HO-1, NQO-1, and ZO-1; downregulate Ly6G and TLR4. | [58] |
| Manp/Glup/Arap = 5.55:1:0.12 Mw = 393 kDa | DSS-induced ulcerative colitis in male Balb/c mice | 50, 100, 200 mg/kg/d for 7 days | Improve inflammation and histopathological changes; inhibit neutrophil infiltration, splenomegaly, and thymic atrophy to restore the immune system damage. | Inhibit the NLRP3 inflammasome signaling pathways; downregulate Ly6G, NLRP3, ASC, and caspase 1. | [72] |
| Manp/Glup/Arap = 5.55:1:0.12 Mw = 393 kDa | DSS-induced acute colitis and secondary hepatic injury in male Balb/c mice | 50, 100, 200 mg/kg/d for 14 days | Improve dyslipidemia; inhibit infiltration of inflammatory cells into hepatic macrophages; decrease oxidative stress and liver damage. | Activate the Nrf-2 signaling pathways; upregulate Nrf-2, HO-1, and NQO-1. | [67] |
| Manp/Glup = 4.76:1.00 Mw = 2.921 kDa And Manp/Glup = 4.44:1.00 Mw = 141.2 kDa | DSS-induced colitis in male Balb/c mice | 200 mg/kg/d for 7 days | Improve the clinical symptoms of colitis, mucosal damage, and inflammatory cell infiltration; inhibit splenomegaly caused by colitis and inflammation. | Activate the GPR41/43 signaling pathways; upregulate GPR41, GPR43, the abundance of gut microbiota, acetic acid, i-butyric acid, and total SCFAs. | [61] |
| Manp/Glup = 4.17:1 Mw = 618.029 kDa | DSS-induced colitis in female Balb/c mice | 200 mg/kg/d for 20 days | Improve intestinal microenvironment homeostasis; inhibit inflammatory cell infiltration in the intestinal lamina propria and intestinal inflammation. | Upregulate MiR-433-3p. | [64] |
| Manp/Glup = 6.9:1 Mw = 312 kDa | DSS-induced colitis in female BALB/c mice | 200 mg/kg/d for 18 days | Improve the clinical symptoms of colitis; increase thymus index, colon length, crypt depth, intestinal wall thickness, and intestinal mucosal integrity; repair colonic mucosal damage; decrease inflammatory reactions. | Downregulate TLR-2, TLR-4, TLR-6, and TLR-9. | [60] |
| Manp/Glup = 6.9:1 Mw = 312 kDa | Healthy female Balb/c mice | 40, 80, 160 mg/kg /d for 10, 20, 30 days | Maintain colon health; increase in colon length and in fecal water content; decrease in defecation time. | Improve the fermentation and regulation of the colon microenvironment; upregulate acetic acid, n-butyric acid, propionic acid, isovaleric acid, i-butyric acid, n-valeric acid, and total SCFAs. | [73] |
| Manp/Glup/Gal = 59.23:35.82:1.61 Mw = unknown | HFD-induced obesity in male C57BL/6 mice | 1.0 g/kg/d for 11 weeks | Improve intestinal barrier function and systemic anti-inflammatory activity; decrease inflammation and oxidative stress. | Upregulate the abundance of beneficial bacteria, ZO-1, and occludin; downregulate NOX2 and NOX4. | [12] |
| Unknown | Anhydrous ethanol-induced gastric ulcer in male SD rats | 0.12, 0.23, 0.46 g/kg/d for 1 week | Improve gastric epithelial defects caused by gastric mucosal injury; inhibit inflammatory cell infiltration and inflammation. | Inhibit the MAPK signaling pathways; downregulate MEK1, MEK, ERK1, and Raf-2; upregulate EGFR and TFF1. | [74] |
| Manp/Glup/Arap = 5.55:1:0.12 Mw = 393 kDa | Ovariectomy- and galactose-induced learning and memory deficits in female Kunming mice | 140 mg/kg/d for 3 months | Alleviate neuroinflammation and oxidative stress. Improve learning and memory deficits and hippocampal neuronal cells; inhibit activation of astrocytes and microglia. | Activate the Nrf-2/HO-1 signaling pathways; upregulate Nrf-2 and HO-1; downregulate GFAP and Iba-1. | [68] |
| Unknown | Senescence-accelerated mouse susceptible male 8 mice and control senescence-accelerated male mouse resistant 1 cognitive impairment | 40 mg/kg/d for 3 months | Improve cognitive and inflammation; modulate microglia activation; increase transition from M1 to M2 phenotype and Aβ degradation to ameliorate aberrant phosphorylation of Tau and Aβ accumulation. | Upregulate NEP, IDE, BDNF, NGF, and PSD95. | [59] |
| Manp/Glup/Arap = 5.55:1:0.12 Mw = 393.8 kDa | Female Kunming naturally aging mice | 70 mg/kg/d for 10 weeks | Improve mitochondrial activity; decrease inflammatory reactions, oxidative stress, and pathological damage to the ovary; increase the number of follicular cells in different stages of the ovary. | Upregulate Bcl-2 and estradiol; downregulate IL-12p70, p53, and p-p65. | [75] |
| Manp/Glup = 2.55:1.00 Mw = 746.52 kDa | Cognitive impairment in male C57BL/6J mice due to circadian rhythm disruption | 200 mg/kg/d for 4 weeks | Improve gut microbial disorders and mucosal integrity; inhibit hippocampal neuronal damage and inflammatory cell infiltration. | Regulate gut microbial composition; upregulate ZO-1, occludin, eletriptan, moclobemide, and paliperidone; downregulate Aβ. | [62] |
| Mw = 8.551 kDa | Acetaminophen-induced liver injury in male ICR mice | 50, 100, 200 mg/kg/d for 30 days | Improve liver damage; decrease inflammatory infiltration and oxidative stress. | Activate the Nrf-2 signaling pathways; upregulate GCLC, GCLM, HO-1, and NQO1; downregulate Keap1. | [66] |
| Manp/Glup = 4.41:1.00 Mw = 195 kDa | CCl4-induced liver fibrosis in male SD rats | 200, 400, 800 mg/kg/d for 8 weeks | Maintain intestinal homeostasis; improve intestinal mucosal barrier; decrease intestinal cell permeability and apoptosis; decrease inflammation and prevent hepatic fibrosis. | Upregulate ZO-1, Bcl-2, occludin, and claudin-1; inhibit the LPS-TLR4-NF-κB pathways; downregulate LPS, TLR4, NF-κB, and p-IκBα; downregulate Bax, caspase 3, TGF-β, α-SMA, and collagen I. | [63] |
| Unknown | PTZ-induced epilepsy in male SD rats | 1.5 g/kg/d for 4 weeks | Decrease brain inflammation and seizures; protect brain neurons. | Downregulate the MAPK pathways, p-ERK1/2, p-JNK, and p-p38; upregulate p-MKP-1. | [76] |
| Unknown | Ovariectomy-induced menopausal depression in female Kunming mice | 150, 300, 600 mg/kg/d for 4 weeks | Improve the clinical symptoms of depression; decrease inflammation. | Inhibition of microglia activation; upregulate estradiol; downregulate CRH, ACTH, and corticosterone. | [77] |
3.3. Immunity Moderation
| Substance | Object | Dosage and Duration | Health Outcome | Potential Mechanism | Ref. |
|---|---|---|---|---|---|
| Manp/Glup = 5.59:1 Mw = unknown | Cyclophosphamides-induced immunosuppression in female Kunming mice | 40, 80, 160 mg/kg/d for 30 days | Improve cellular immunity, humoral immunity, and phagocytosis of monocytes; increase the quality of immune organs; decrease oxidative stress. | Upregulate CD3, CD4, CD8, CD4/CD8, IL-4, IL-6, IL-10, TNF-α, IgM, IgG, and hemolysin. | [18] |
| Manp/Glup = 5.16:1.3 Mw = 262.4 ku | Cyclophosphamides-induced immunosuppression in male BALB/c mice | 160 mg/kg/d for 7 days | Improve intestinal health, immunomodulation, and the balance between Th1 and Th2 types; increase mass of immune organs. | Upregulate acetic acid, propionic acid, butyric acid, valeric acid, total SCFAs, IFN-γ, TNF-α, and IL-6; downregulate IL-4 and IgE. | [81] |
| Manp/Glup = 6.9:1 Mw = 312 kDa | Cyclophosphamides-induced immunosuppression in female Balb/c mice | 40, 80, 160 mg/kg/d for 7 days | Improve immunomodulation; stimulate plasma cell differentiation, the secretion of immunoglobulin, and cytokines; increase the expression of Th1-type T cells. | Upregulate CD4, CD8 T lymphocytes, T-bet/GATA-3, Pax5, XBP-1, Blimp-1, TNF-α, IFN-γ, IL-4, IgA, IgM, and IgG. | [78] |
| Manp/Glup = 4.5:1 Mw = 15.43 kDa | Healthy female ICR mice | 0.5, 2 g/kg/d for 7 days. | Improve intestinal mucosal structure, intestinal mucosal immunoreactivity, and the balance between Th1 and Th2 types. | Upregulate IFN-γ, IL-4, and IgA. | [42] |
| Manp/Glup = 6.9:1 Mw = 312 kDa | Cyclophosphamides-induced immunosuppression in male Balb/c mice | 40, 80, 160 mg/kg/d for 4 weeks | Improve immunomodulation and proliferation of splenocytes; accelerate phagocytosis by peritoneal macrophages. | Upregulate CD3, CD4/CD8, TNF-α, IL-6, IgM, IgG, and serum hemolysin. | [82] |
| Glup/GluA = 19.4:1.2 Mw = 39.4 kDa | Healthy female C57BL/6 mice | 0.25% DOP aqueous solution instead of drinking water, free water intervention for 25 days | Improve the production of more butyrate by gut microorganisms to participate in immune mediation. | Regulate gut microbial composition; upregulate butyrate, acetic acids, Muc-2, IL-10, TNF-α, IL-1β, and IgM; downregulate fecal lipocalin-2. | [80] |
3.4. Cancer Intervention
| Substance | Object | Dosage and Duration | Health Outcome | Potential Mechanism | Ref. |
|---|---|---|---|---|---|
| Manp/Glup/Arap = 5.5:1:0.12 Mw = 393.8 kDa | AOM/DSS-induced colon cancer in male Balb/c mice | 50, 100, 200 mg/kg/d for 6 weeks | Improve intestinal epithelial barrier function, intestinal anti-tumor immune response, and inflammation; inhibit the formation and growth of colon tumors. | Upregulate ZO-1, occludin, CD8 CTLs, ATP, and glucose of tumor-infiltrating lymphocytes; downregulate TNF-α, IL-1β, and PD-1. | [86] |
| Mw = 3.5 and 1000 kDa | MNNG-induced gastric precancerous lesions in male Wistar rats | 2.4, 4.8, 9.6 g/kg/d for 7 months | Prevent gastric precancerous lesions; protect gastric mucosa and subsequent liver and kidney damage; improve the weight loss and reduce intestinal epithelial chemotaxis, simultaneously. | Activate the Nrf-2 pathways; upregulate Nrf-2, HO-1, and NQO-1; downregulate 8-OHdG. | [19] |
| Mw = 3.5 and 1000 kDa | MNNG-induced gastric precancerous lesions in male Wistar rats | 2.4, 4.8, 9.6 g/kg/d for 7 months | Modulate endogenous metabolites; ameliorate oxidative stress; inhibit induced gastric precancerous lesions. | Inhibit the Wnt/β-catenin pathways; downregulate Wnt2β, Gsk3β, PCNA, CyclinD1, and β-catenin; upregulate serum endogenous metabolites such as betaine. | [88] |
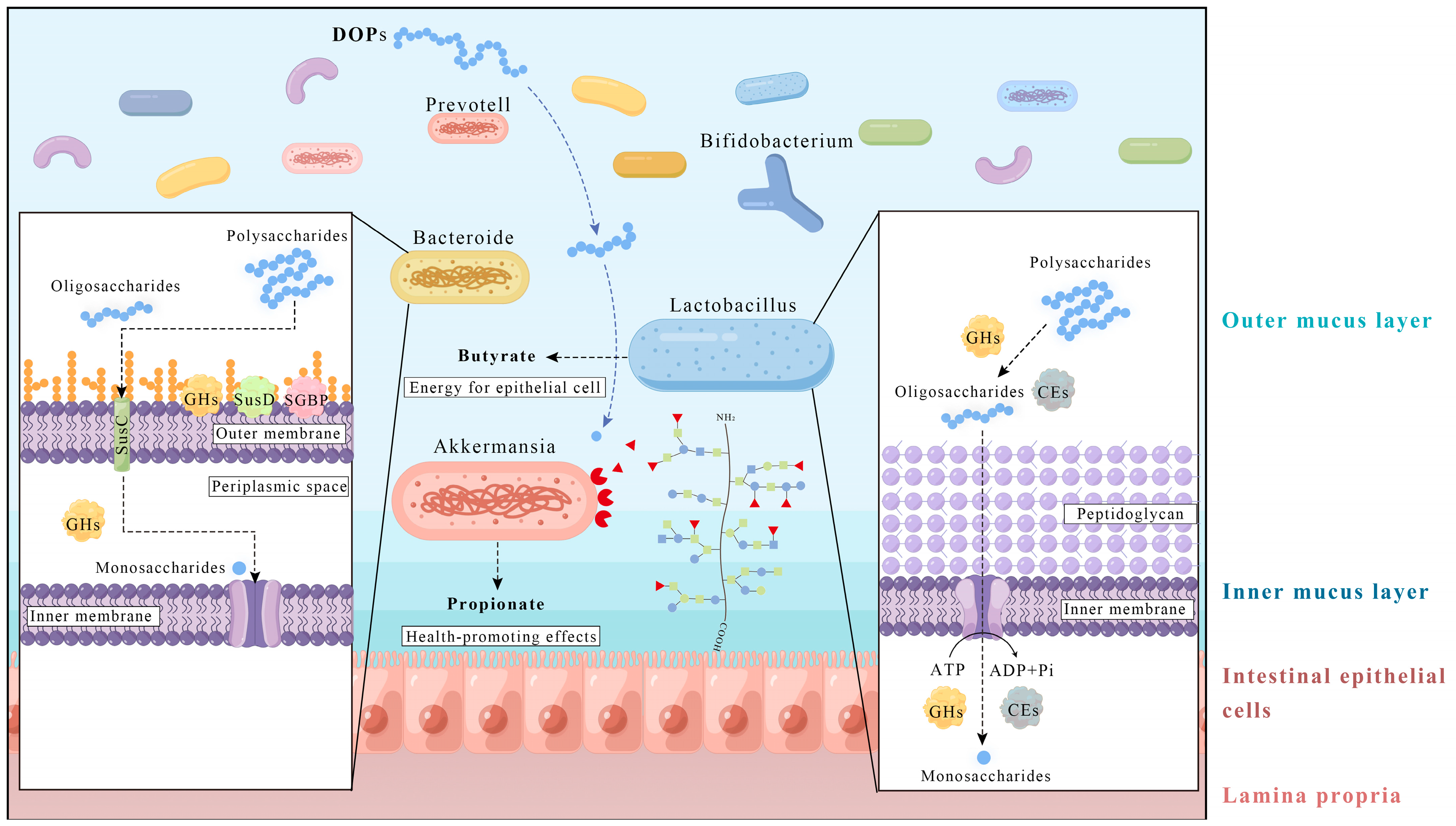
4. Relationship between Dendrobium officinale Polysaccharides and Gut Microbiota
| Structure | Object | Dosage and Duration | The Genus Level of Gut Microbiota | Ref. | |
|---|---|---|---|---|---|
| Increase | Decrease | ||||
| The content of Manp, Glup, and Galp is 49.03%, 16.54%, 14.83% | In vitro fermentation of healthy human feces | 200 mg/15 mL culture fluid for 48 h | Bacteroides, Prevotella, and Faecalibacterium | Citrobacter | [5] |
| The content of Manp and Glup is 61.10% and 36.92% Mw = 277 KDa and 1318 Da | In vitro fermentation of healthy human feces | 10 mg/5 mL culture fluid for 24 h | Bifidobacterium, Bacteroides, Lactobacillus, Enterococcus, Streptococcus (in women), and Prevotella_9 (in men) | Lachnospiraceae UCG-004, Lachnoclostridium Escherichia-Shigella, and Paraclostridium | [31] |
| Manp/Glup/Galp/Rhap = 59.31:33.31:1.00:0.51 Mw = 291 kDa | In vitro fermentation of healthy human feces | 20, 40, 80 mg/10 mL culture fluid for 24 h | Bifidobacterium, Prevotella_9, Lactobacillus, Faeca-libacterium, Pseudobutyrivibrio, Pediococcus, and Lachnoclostridium | Bacteroides, Escherichia-Shigella, Enterobacter, Dialister, and ParaBacteroides | [11] |
| Manp/Glup/Galp/Rhap = 59.31:33.31:1.00:0.51 Mw = 291 kDa | Healthy male ICR mice | 100, 200 mg/kg/d for 21 days | Lactobacillus, Desulfovibrionaceae_unclassified, Klebsiella, and Lactococcus | Ruminococcaceae_unclassified, Desulfomicrobium, Papillibacter, Desulfovibrio, Pseudomonas, Sandaracinobacter, and Corynebacterium | [11] |
| Manp/Glup = 4.0:1.0 Mw = 277.3 kDa | Healthy male mice | 200 mg/kg/for 4 weeks | Lactobacillus, Bifidobacterium, and g-Roseburia | Proteobacteria | [92] |
| Manp/Glup =1.9:1.0 Mw = unknown | Excessive alcohol consumption and HFD-induced metabolic hypertension in male SD rats. | 200 mg/kg/d for 7 weeks | Lachnospiraceae_NK4A136_group, Lactobacillus, and NK4A214_group | Blautia | [55] |
| Unknown | HFD-induced obese male KM mice | 350 mg/kg/d for 8 weeks | Lachnospiraceae_NK4A136_group, Lactobacillus, and Candidatus_Arthromitus | Corynebacterium_1 and Staphylococcus | [53] |
| Manp/Glup = 4.76:1.00 Mw = 2.921 kDa And Manp/Glup = 4.44:1.00 Mw = 141.2 kDa | DSS-induced colitis in male Balb/c mice | 200 mg/kg/d for 7 days | Ruminococcaceae_UCG—014, Bacteroides, and Lactobacillus | Akkermansia | [61] |
| Glup/GluA = 19.4:1.2 Mw = 39.4 kDa | Heathy female C57BL/6 mice | 0.25% DOP aqueous solution instead of drinking water, free water intervention for 25 days | Akkermansia muciniphila, Ruminococcus, Eubacterium, Clostridium, Bifidobacterium, and Parabacteroides | Proteobacteria and Prevotella | [80] |
| Manp/Glup = 2.55:1.00 Mw = 746.52 kDa | Cognitive impairment in male C57BL/6J mice due to circadian rhythm disruption | 200 mg/kg/d for 4 weeks | Akkermansia, Alistipes, and Dubosiella | Desulfovibrio, Candidatus_Saccharimon, and Clostridia | [62] |
| Manp/Glup/Galp = 59.23:35.82:1.61 Mw = unknown | HFD-induced obesity in male C57BL/6 mice | 1.0 g/kg/d for 11 weeks | Muribaculum, Akkermansia, and Bifidobacterium | Blautia, Lachnoclostridium, Bilophila, and Mucispirillum | [12] |
| Manp/Glup = 6.9:1 Mw = 312 kDa | STZ-induced type 2 diabetes in male Wistar rats | 160 mg/kg/d for 4 weeks | Clostridium, Bacteroides, Prevotella, Klebsiella, Escherichia, Streptococcus, and Staphylococcus aureus | [10] | |
| Manp/Glup = 69.70:30.30 Mw = 731 kDa | HFD/STZ-induced T2DM in male C57BL/6J mice | 120 mg/kg/d for 8 weeks | Dubosiella, Lysinibacillus, Ileibacterium valens, Faecalibaculum rodentium, and Akkermansia muciniphila | Lactobacillus reuteri, Lactobacillus johnsonii, Enterococcus Casseliflavus, and Eubacterium Plexicaudatum | [57] |
| Manp/Glup = 7.32:1 Mw = 279 kDa | Cyclophosphamides-induced immunosuppression in male BALB/c mice | 100 mg/kg/d for 9 days | Lactobacillus, AlloPrevotella, Akkermansia, Ruminococcaceae_UCG-014, and Alistipes | Helicobacter | [83] |
5. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tang, H.; Zhao, T.; Sheng, Y.; Zheng, T.; Fu, L.; Zhang, Y. Dendrobium officinale Kimura et Migo: A Review on Its Ethnopharmacology, Phytochemistry, Pharmacology, and Industrialization. Evid.-Based Complement. Altern. Med. 2017, 2017, 7436259. [Google Scholar] [CrossRef] [PubMed]
- National Health Commission of the People’s Republic of China. Announcement on 9 New Substances Including Codonopsis pilosula That Are Both Food and Traditional Chinese Medicine. Available online: http://www.nhc.gov.cn/sps/s7892/202311/f0d6ef3033b54333a882e3d009ff49bf.shtml (accessed on 17 November 2023).
- Yuan, Y.; Tang, X.; Jia, Z.; Li, C.; Ma, J.; Zhang, J. The Effects of Ecological Factors on the Main Medicinal Components of Dendrobium officinale under Different Cultivation Modes. Forests 2020, 11, 94. [Google Scholar] [CrossRef]
- He, L.; Yan, X.; Liang, J.; Li, S.; He, H.; Xiong, Q.; Lai, X.; Hou, S.; Huang, S. Comparison of Different Extraction Methods for Polysaccharides from Dendrobium officinale Stem. Carbohydr. Polym. 2018, 198, 101–108. [Google Scholar] [CrossRef]
- Fu, Y.; Zhang, J.; Chen, K.; Xiao, C.; Fan, L.; Zhang, B.; Ren, J.; Fang, B. An In Vitro Fermentation Study on the Effects of Dendrobium officinale Polysaccharides on Human Intestinal Microbiota from Fecal Microbiota Transplantation Donors. J. Funct. Foods 2019, 53, 44–53. [Google Scholar] [CrossRef]
- Fan, S.; Zhang, Z.; Zhong, Y.; Li, C.; Huang, X.; Geng, F.; Nie, S. Microbiota-Related Effects of Prebiotic Fibres in Lipopolysaccharide-Induced Endotoxemic Mice: Short Chain Fatty Acid Production and Gut Commensal Translocation. Food Funct. 2021, 12, 7343–7357. [Google Scholar] [CrossRef]
- Li, L.; Yao, H.; Li, X.; Zhang, Q.; Wu, X.; Wong, T.; Zheng, H.; Fung, H.; Yang, B.; Ma, D.; et al. Destiny of Dendrobium officinale Polysaccharide after Oral Administration: Indigestible and Nonabsorbing, Ends in Modulating Gut Microbiota. J. Agric. Food Chem. 2019, 67, 5968–5977. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Zhang, Y.; Nie, G.; Liu, J.; Mei, H.; He, Z.; Dou, P.; Wang, K. Tracking the Gastrointestinal Digestive and Metabolic Behaviour of Dendrobium officinale Polysaccharides by Fluorescent Labelling. Food Funct. 2022, 13, 7274–7286. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Gong, F.; Wei, F.; Lai, F.; Zhang, X.; Wu, H. Artificial Simulated Gastrointestinal Digestion of Four Carbohydrates Containing Beta-d-1 → 4 Linkages and New GC-TQ/MS-MS Method for Characterising Released Monosaccharides. Int. J. Food Sci. Technol. 2018, 53, 1992–2005. [Google Scholar] [CrossRef]
- Chen, H.; Nie, Q.; Hu, J.; Huang, X.; Yin, J.; Nie, S. Multiomics Approach to Explore the Amelioration Mechanisms of Glucomannans on the Metabolic Disorder of Type 2 Diabetic Rats. J. Agric. Food Chem. 2021, 69, 2632–2645. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Tao, W.; Wang, M.; Liu, W.; Xing, J.; Yang, Y. Dendrobium officinale Xianhu 2 Polysaccharide Helps Forming a Healthy Gut Microbiota and Improving Host Immune System: An In Vitro and In Vivo Study. Food Chem. 2023, 401, 134211. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Ji, H.; Fan, K.; Xu, H.; Huang, Y.; Zheng, Y.; Xu, Q.; Li, C.; Zhao, L.; Li, Y.; et al. Targeting Gut Microbiota and Host Metabolism with Dendrobium officinale Dietary Fiber to Prevent Obesity and Improve Glucose Homeostasis in Diet-Induced Obese Mice. Mol. Nutr. Food Res. 2022, 66, e2100772. [Google Scholar] [CrossRef] [PubMed]
- Qu, J.; Tan, S.Y.; Xie, X.Y.; Wu, W.Q.; Zhu, H.H.; Li, H.; Liao, X.B.; Wang, J.J.; Zhou, Z.A.; Huang, S.; et al. Dendrobium officinale Polysaccharide Attenuates Insulin Resistance and Abnormal Lipid Metabolism in Obese Mice. Front. Pharmacol. 2021, 12, 659626. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.P.; Wang, H.X.; Liu, Y.G.; Shui, W.Z.; Wang, J.F.; Cao, P.; Wang, H.J.; You, R.X.; Zhang, Y. Dendrobiurn officinale Polysaccharide Attenuates Type 2 Diabetes Mellitus via the Regulation of PI3K/Akt-Mediated Glycogen Synthesis and Glucose Metabolism. J. Funct. Foods 2018, 40, 261–271. [Google Scholar] [CrossRef]
- Chen, H.; Nie, Q.; Hu, J.; Huang, X.; Zhang, K.; Nie, S. Glucomannans Alleviated the Progression of Diabetic Kidney Disease by Improving Kidney Metabolic Disturbance. Mol. Nutr. Food Res. 2019, 63, 1801008. [Google Scholar] [CrossRef] [PubMed]
- Song, T.H.; Chen, X.X.; Tang, S.C.W.; Ho, J.C.M.; Lao, L.X.; Ng, T.B.; Lee, K.F.; Sze, S.C.W.; Zhang, K.Y. Dendrobium officinale Polysaccharides Ameliorated Pulmonary Function While Inhibiting Mucin-5AC and Stimulating Aquaporin-Expression. J. Funct. Foods 2016, 21, 359–371. [Google Scholar] [CrossRef]
- Liang, Y.; Du, R.; Chen, R.; Chu, P.H.; Ip, M.S.M.; Zhang, K.Y.B.; Mak, J.C.W. Therapeutic Potential and Mechanism of Dendrobium officinale Polysaccharides on Cigarette Smoke-Induced Airway Inflammation in Rat. Biomed. Pharmacother. 2021, 143, 112101. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Xu, L.; Guo, X.; Cui, X.; Yang, Y. Optimization of the Extraction Process of Polysaccharides from Dendrobium officinale and Evaluation of the In Vivo Immunmodulatory Activity. J. Food Process Preserv. 2018, 42, e13598. [Google Scholar] [CrossRef]
- Zhao, Y.; Sun, Y.; Wang, G.; Ge, S.; Liu, H. Dendrobium officinale Polysaccharides Protect against MNNG-Induced PLGC in Rats via Activating the NRF2 and Antioxidant Enzymes HO-1 and NQO-1. Oxid. Med. Cell Longev. 2019, 2019, 9310245. [Google Scholar] [CrossRef]
- Chen, W.; Wu, J.; Li, X.; Lu, J.; Wu, W.; Sun, Y.; Zhu, B.; Qin, L. Isolation, Structural Properties, Bioactivities of Polysaccharides from Dendrobium officinale Kimura et. Migo: A Review. Int. J. Biol. Macromol. 2021, 184, 1000–1013. [Google Scholar] [CrossRef]
- He, Y.; Li, L.; Chang, H.; Cai, B.; Gao, H.; Chen, G.; Hou, W.; Jappar, Z.; Yan, Y. Research Progress on Extraction, Purification, Structure and Biological Activity of Dendrobium officinale Polysaccharides. Front. Nutr. 2022, 9, 965073. [Google Scholar] [CrossRef]
- Liang, J.; Zeng, Y.; Wang, H.; Lou, W. Extraction, Purification and Antioxidant Activity of Novel Polysaccharides from Dendrobium officinale by Deep Eutectic Solvents. Nat. Prod. Res. 2019, 33, 3248–3253. [Google Scholar] [CrossRef]
- Pan, L.-H.; Wang, J.; Ye, X.-Q.; Zha, X.-Q.; Luo, J.-P. Enzyme-Assisted Extraction of Polysaccharides from Dendrobium chrysotoxum and Its Functional Properties and Immunomodulatory Activity. LWT—Food Sci. Technol. 2015, 60, 1149–1154. [Google Scholar] [CrossRef]
- Cui, R.; Zhu, F. Ultrasound Modified Polysaccharides: A Review of Structure, Physicochemical Properties, Biological Activities and Food Applications. Trends Food Sci. Technol. 2021, 107, 491–508. [Google Scholar] [CrossRef]
- Chen, X.; Yang, J.; Shen, M.; Chen, Y.; Yu, Q.; Xie, J. Structure, Function and Advance Application of Microwave-Treated Polysaccharide: A Review. Trends Food Sci. Technol. 2022, 123, 198–209. [Google Scholar] [CrossRef]
- Ma, H.; Zhang, K.; Jiang, Q.; Dai, D.; Li, H.; Bi, W.; Chen, D.D.Y. Characterization of Plant Polysaccharides from Dendrobium officinale by Multiple Chromatographic and Mass Spectrometric Techniques. J. Chromatogr. A 2018, 1547, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Chen, Y.; Zhu, M.; Lu, Y. Homogenate Extraction and Quality Evaluation of Polysaccharides from Dendrobium officinale. J. Pharm. Res. 2018, 37, 270–274. [Google Scholar]
- YUE, H.; ZENG, H.; DING, K. A Review of Isolation Methods, Structure Features and Bioactivities of Polysaccharides from Dendrobium Species. Chin. J. Nat. Med. 2020, 18, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Hua, Y.; Zhang, M.; Fu, C.; Chen, Z.; Chan, G.Y.S. Structural Characterization of a 2-O-Acetylglucomannan from Dendrobium officinale Stem. Carbohydr. Res. 2004, 339, 2219–2224. [Google Scholar] [CrossRef]
- Sun, S.J.; Deng, P.; Peng, C.E.; Ji, H.Y.; Mao, L.F.; Peng, L.Z. Extraction, Structure and Immunoregulatory Activity of Low Molecular Weight Polysaccharide from Dendrobium officinale. Polymers 2022, 14, 2899. [Google Scholar] [CrossRef] [PubMed]
- Tao, W.; Liu, W.; Wang, M.; Zhou, W.; Xing, J.; Xu, J.; Pi, X.; Wang, X.; Lu, S.; Yang, Y. Dendrobium officinale Polysaccharides Better Regulate the Microbiota of Women Than Men. Foods 2022, 11, 1641. [Google Scholar] [CrossRef] [PubMed]
- Borreani, J.; Llorca, E.; Larrea, V.; Hernando, I. Adding Neutral or Anionic Hydrocolloids to Dairy Proteins under In Vitro Gastric Digestion Conditions. Food Hydrocoll. 2016, 57, 169–177. [Google Scholar] [CrossRef]
- Biedrzycka, E.; Bielecka, M. Prebiotic Effectiveness of Fructans of Different Degrees of Polymerization. Trends Food Sci. Technol. 2004, 15, 170–175. [Google Scholar] [CrossRef]
- Ronkart, S.N.; Blecker, C.S.; Fourmanoir, H.; Fougnies, C.; Deroanne, C.; Van Herck, J.C.; Paquot, M. Isolation and Identification of Inulooligosaccharides Resulting from Inulin Hydrolysis. Anal. Chim. Acta 2007, 604, 81–87. [Google Scholar] [CrossRef]
- Kuang, M.T.; Li, J.Y.; Yang, X.B.; Yang, L.; Xu, J.Y.; Yan, S.; Lv, Y.F.; Ren, F.C.; Hu, J.M.; Zhou, J. Structural Characterization and Hypoglycemic Effect via Stimulating Glucagon-like Peptide-1 Secretion of Two Polysaccharides from Dendrobium officinale. Carbohydr. Polym. 2020, 241, 116326. [Google Scholar] [CrossRef]
- Tao, S.; Lei, Z.; Huang, K.; Li, Y.; Ren, Z.; Zhang, X.; Wei, G.; Chen, H. Structural Characterization and Immunomodulatory Activity of Two Novel Polysaccharides Derived from the Stem of Dendrobium officinale Kimura et Migo. J. Funct. Foods 2019, 57, 121–134. [Google Scholar] [CrossRef]
- Xing, X.; Cui, S.W.; Nie, S.; Phillips, G.O.; Goff, H.D.; Wang, Q. Study on Dendrobium officinale O-Acetyl-Glucomannan (Dendronan®): Part II. Fine Structures of O-Acetylated Residues. Carbohydr. Polym. 2015, 117, 422–433. [Google Scholar] [CrossRef] [PubMed]
- Luo, Q.; Tang, Z.; Zhang, X.; Zhong, Y.; Yao, S.; Wang, L.; Lin, C.; Luo, X. Chemical Properties and Antioxidant Activity of a Water-Soluble Polysaccharide from Dendrobium officinale. Int. J. Biol. Macromol. 2016, 89, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Wang, L.; Wang, D.; Wang, D.; Wen, C.; Han, B.; Ouyang, Z. Characterization and Anti-Tumor Activity of a Polysaccharide Isolated from Dendrobium officinale Grown in the Huoshan County. Chin. Med. 2018, 13, 47. [Google Scholar] [CrossRef]
- Wei, W.; Feng, L.; Bao, W.R.; Ma, D.L.; Leung, C.H.; Nie, S.P.; Han, Q. Bin Structure Characterization and Immunomodulating Effects of Polysaccharides Isolated from Dendrobium officinale. J. Agric. Food Chem. 2016, 64, 881–889. [Google Scholar] [CrossRef]
- Wang, L.-X.; Li, C.-Y.; Hu, C.; Gong, P.-S.; Zhao, S.-H. Purification and Structural Characterization of Dendrobium officinale Polysaccharides and Its Activities. Chem. Biodivers. 2021, 18, e2001023. [Google Scholar] [CrossRef]
- Xie, S.Z.; Liu, B.; Zhang, D.D.; Zha, X.Q.; Pan, L.H.; Luo, J.P. Intestinal Immunomodulating Activity and Structural Characterization of a New Polysaccharide from Stems of Dendrobium officinale. Food Funct. 2016, 7, 2789–2799. [Google Scholar] [CrossRef]
- Tilg, H.; Adolph, T.E.; Dudek, M.; Knolle, P. Non-Alcoholic Fatty Liver Disease: The Interplay between Metabolism, Microbes and Immunity. Nat. Metab. 2021, 3, 1596–1607. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.M.; Guo, Q.; Yang, W.G.; Wang, Y.L.; Sun, Z.J.; Wu, H.Y. Mapping Knowledge Landscapes and Emerging Trends of the Links between Bone Metabolism and Diabetes Mellitus: A Bibliometric Analysis from 2000 to 2021. Front. Public Health 2022, 10, 918483. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.W.; Cao, M.M.; Li, Y.J.; Dai, G.C.; Lu, P.P.; Zhang, M.; Bai, L.Y.; Chen, X.X.; Zhang, C.; Shi, L.; et al. The Regulative Effect and Repercussion of Probiotics and Prebiotics on Osteoporosis: Involvement of Brain-Gut-Bone Axis. Crit. Rev. Food Sci. Nutr. 2023, 63, 7510–7528. [Google Scholar] [CrossRef] [PubMed]
- Xue, M.; Xu, W.; Ou, Y.N.; Cao, X.P.; Tan, M.S.; Tan, L.; Yu, J.T. Diabetes Mellitus and Risks of Cognitive Impairment and Dementia: A Systematic Review and Meta-Analysis of 144 Prospective Studies. Ageing Res. Rev. 2019, 55, 100944. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Zhang, W.H.; Li, S.; Yang, H. The Role of Cholesterol Metabolism in Cancer. Am. J. Cancer Res. 2019, 9, 219–227. [Google Scholar] [PubMed]
- Yang, J.R.; Chen, H.H.; Nie, Q.X.; Huang, X.J.; Nie, S.P. Dendrobium officinale Polysaccharide Ameliorates the Liver Metabolism Disorders of Type II Diabetic Rats. Int. J. Biol. Macromol. 2020, 164, 1939–1948. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yang, L.; Zhang, Y.; Liu, X.; Wu, Z.; Gilbert, R.G.; Deng, B.; Wang, K. Dendrobium officinale Polysaccharide Ameliorates Diabetic Hepatic Glucose Metabolism via Glucagon-Mediated Signaling Pathways and Modifying Liver-Glycogen Structure. J. Ethnopharmacol. 2020, 248, 112308. [Google Scholar] [CrossRef]
- Chen, H.H.; Nie, Q.X.; Hu, J.L.; Huang, X.J.; Huang, W.Q.; Nie, S.P. Metabolism Amelioration of Dendrobium officinale Polysaccharide on Type II Diabetic Rats. Food Hydrocoll. 2020, 102, 105582. [Google Scholar] [CrossRef]
- Rovira-Llopis, S.; Bañuls, C.; Diaz-Morales, N.; Hernandez-Mijares, A.; Rocha, M.; Victor, V.M. Mitochondrial Dynamics in Type 2 Diabetes: Pathophysiological Implications. Redox Biol. 2017, 11, 637–645. [Google Scholar] [CrossRef]
- Chen, L.; He, X.; Wang, H.; Fang, J.; Zhang, Z.; Zhu, X.; Qi, Z.; Guo, Y.; Liu, R.; Li, D. Dendrobium officinale Polysaccharide Prevents Neuronal Apoptosis via TET2-Dependent DNA Demethylation in High-Fat Diet-Induced Diabetic Mice. Int. J. Biol. Macromol. 2023, 233, 123288. [Google Scholar] [CrossRef] [PubMed]
- Xie, G.; Tang, Y.; Qiu, X.; Tan, Z. Effects of Dendrobium officinale Polysaccharides on the Intestinal Mucosal Structure and Microbiota in Mice Fed a High-Fat Diet. Biotechnol. Bull. 2022, 38, 150–157. [Google Scholar]
- Tang, Z.; Zhou, X.; Feng, J.; Liu, H.; Li, J.; Wu, Y.; Ye, X.; Ni, S. The Hypoglycemic Activity of Homogeneous Polysaccharides from Dendrobium officinale. J. Nanjing Univ. Tradit. Chin. Med. 2016, 32, 566–570. [Google Scholar]
- Li, B.; Wang, H.-Y.; Huang, J.-H.; Xu, W.-F.; Feng, X.-J.; Xiong, Z.-P.; Dong, Y.-J.; Li, L.-Z.; He, X.; Wu, H.-S.; et al. Polysaccharide, the Active Component of Dendrobium officinale, Ameliorates Metabolic Hypertension in Rats via Regulating Intestinal Flora-SCFAs-Vascular Axis. Front. Pharmacol. 2022, 13, 935714. [Google Scholar] [CrossRef] [PubMed]
- Luo, D.; Qu, C.; Lin, G.; Zhang, Z.; Xie, J.; Chen, H.; Liang, J.; Li, C.; Wang, H.; Su, Z. Character and Laxative Activity of Polysaccharides Isolated from Dendrobium officinale. J. Funct. Foods 2017, 34, 106–117. [Google Scholar] [CrossRef]
- Peng, D.; Tian, W.; An, M.; Chen, Y.; Zeng, W.; Zhu, S.; Li, P.; Du, B. Characterization of Antidiabetic Effects of Dendrobium officinale Derivatives in a Mouse Model of Type 2 Diabetes Mellitus. Food Chem. 2023, 399, 133974. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Xiao, H.; Liu, Y.; Yang, Y.; Wang, Y.; Xu, S.; Huang, S.; Hou, S.; Liang, J. Polysaccharides from Dendrobium officinale Ameliorate Colitis-Induced Lung Injury via Inhibiting Inflammation and Oxidative Stress. Chem. Biol. Interact. 2021, 347, 109615. [Google Scholar] [CrossRef] [PubMed]
- Feng, C.Z.; Cao, L.; Luo, D.; Ju, L.S.; Yang, J.J.; Xu, X.Y.; Yu, Y.P. Dendrobium Polysaccharides Attenuate Cognitive Impairment in Senescence-Accelerated Mouse Prone 8 Mice via Modulation of Microglial Activation. Brain Res. 2019, 1704, 1–10. [Google Scholar] [CrossRef]
- Zhang, L.J.; Huang, X.J.; Shi, X.D.; Chen, H.H.; Cui, S.W.; Nie, S.P. Protective Effect of Three Glucomannans from Different Plants against DSS Induced Colitis in Female BALB/c Mice. Food Funct. 2019, 10, 1928–1939. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, Z.; Liu, J.; Zheng, Z.; Li, Q.; Wang, H.J.; Chen, Z.; Wang, K. Identification of the Core Active Structure of a Dendrobium officinale Polysaccharide and Its Protective Effect against Dextran Sulfate Sodium-Induced Colitis via Alleviating Gut Microbiota Dysbiosis. Food Res. Int. 2020, 137, 109641. [Google Scholar] [CrossRef]
- Sun, Y.; Zeng, X.; Liu, Y.; Zhan, S.; Wu, Z.; Zheng, X.; Zhang, X. Dendrobium officinale Polysaccharide Attenuates Cognitive Impairment in Circadian Rhythm Disruption Mice Model by Modulating Gut Microbiota. Int. J. Biol. Macromol. 2022, 217, 677–688. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Yang, X.; Wu, Z.; Wang, H.; Li, Q.; Mei, H.; You, R.; Zhang, Y. Dendrobium officinale Polysaccharide Protected CCl4-Induced Liver Fibrosis through Intestinal Homeostasis and the LPS-TLR4-NF-ΚB Signaling Pathway. Front. Pharmacol. 2020, 11, 240. [Google Scholar] [CrossRef]
- Liu, H.F.; Liang, J.X.; Zhong, Y.M.; Xiao, G.S.; Efferth, T.; Georgiev, M.I.; Vargas-De-la-Cruz, C.; Bajpai, V.K.; Caprioli, G.; Liu, J.L.; et al. Dendrobium officinale Polysaccharide Alleviates Intestinal Inflammation by Promoting Small Extracellular Vesicle Packaging of MiR-433-3p. J. Agric. Food Chem. 2021, 69, 13510–13523. [Google Scholar] [CrossRef]
- Peng, H.; Yang, M.; Guo, Q.; Su, T.; Xiao, Y.; Xia, Z.Y. Dendrobium officinale Polysaccharides Regulate Age-Related Lineage Commitment between Osteogenic and Adipogenic Differentiation. Cell Prolif. 2019, 52, e12624. [Google Scholar] [CrossRef] [PubMed]
- Lin, G.; Luo, D.; Liu, J.; Wu, X.; Chen, J.; Huang, Q.; Su, L.; Zeng, L.; Wang, H.; Su, Z. Hepatoprotective Effect of Polysaccharides Isolated from Dendrobium officinale against Acetaminophen-Induced Liver Injury in Mice via Regulation of the Nrf2-Keap1 Signaling Pathway. Oxid. Med. Cell Longev. 2018, 2018, 6962439. [Google Scholar] [CrossRef]
- Liang, J.; Chen, S.; Hu, Y.; Yang, Y.; Yuan, J.; Wu, Y.; Li, S.; Lin, J.; He, L.; Hou, S.; et al. Protective Roles and Mechanisms of Dendrobium officinal Polysaccharides on Secondary Liver Injury in Acute Colitis. Int. J. Biol. Macromol. 2018, 107, 2201–2210. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Wu, Y.F.; Yuan, H.; Yang, Y.Q.; Xiong, Q.P.; Liang, C.Y.; Li, Z.M.; Li, C.T.; Zhang, G.F.; Lai, X.P.; et al. Dendrobium officinale Polysaccharides Attenuate Learning and Memory Disabilities via Anti-Oxidant and Anti-Inflammatory Actions. Int. J. Biol. Macromol. 2019, 126, 414–426. [Google Scholar] [CrossRef]
- Chen, Z.; Lan, Q.; Chen, S.; Hu, Y.; Ke, F.; Hu, J.; Feng, C.; Qi, M.; Huang, X. Effects of Dendrobium candidum Polysaccharides on MicroRNA-125b and Mitogen-Activated Protein Kinase Signaling Pathways in Diabetic Cataract Rats. Tradit. Med. Res. 2021, 6, 45. [Google Scholar] [CrossRef]
- Chen, J.; Lu, J.; Wang, B.; Zhang, X.; Huang, Q.; Yuan, J.; Hao, H.; Chen, X.; Zhi, J.; Zhao, L.; et al. Polysaccharides from Dendrobium officinale Inhibit Bleomycin-Induced Pulmonary Fibrosis via the TGFβ1-Smad2/3 Axis. Int. J. Biol. Macromol. 2018, 118, 2163–2175. [Google Scholar] [CrossRef]
- Zeng, Q.; Ko, C.H.; Siu, W.S.; Li, L.F.; Han, X.Q.; Yang, L.; Lau, C.B.S.; Hu, J.M.; Leung, P.C. Polysaccharides of Dendrobium officinale Kimura & Migo Protect Gastric Mucosal Cell against Oxidative Damage-Induced Apoptosis In Vitro and In Vivo. J. Ethnopharmacol. 2017, 208, 214–224. [Google Scholar] [CrossRef]
- Liang, J.; Chen, S.X.; Chen, J.H.; Lin, J.Z.; Xiong, Q.P.; Yang, Y.Q.; Yuan, J.; Zhou, L.; He, L.; Hou, S.Z.; et al. Therapeutic Roles of Polysaccharides from Dendrobium officinale on Colitis and Its Underlying Mechanisms. Carbohydr. Polym. 2018, 185, 159–168. [Google Scholar] [CrossRef]
- Zhang, G.Y.; Nie, S.P.; Huang, X.J.; Hu, J.L.; Cui, S.W.; Xie, M.Y.; Phillips, G.O. Study on Dendrobium officinale O-Acetyl-Glucomannan (Dendronan). 7. Improving Effects on Colonic Health of Mice. J. Agric. Food Chem. 2016, 64, 2485–2491. [Google Scholar] [CrossRef]
- Ma, S.; Wu, Q.; Zhao, Z.; Xiong, J.; Niu, J.; Liu, C.; Liu, T.; Chai, Y.; Qu, X.; Ma, Z.; et al. Mechanisms of Dendrobium officinale Polysaccharides in Repairing Gastric Mucosal Injuries Based on Mitogen-Activated Protein Kinases (MAPK) Signaling Pathway. Bioengineered 2022, 13, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Liang, C.; Liu, T.; Liang, Y.; Li, S.; Lu, Y.; Liang, J.; Yuan, X.; Li, C.; Hou, S.; et al. Protective Roles and Mechanisms of Polysaccharides from Dendrobium officinal on Natural Aging-Induced Premature Ovarian Failure. Biomed. Pharmacother. 2018, 101, 953–960. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Peng, H.; Xu, J.; Xu, Y.; Yin, Y.; He, B.; Zhuang, J. Effects of Dendrobium officinale Polysaccharides on Brain Inflammation of Epileptic Rats. Int. J. Polym. Sci. 2019, 2019, 9058161. [Google Scholar] [CrossRef]
- Zhang, Q.P.; Cheng, J.; Liu, Q.; Xu, G.H.; Li, C.F.; Yi, L.T. Dendrobium officinale Polysaccharides Alleviate Depression-like Symptoms via Regulating Gut Microbiota-Neuroinflammation in Perimenopausal Mice. J. Funct. Foods 2022, 88, 104912. [Google Scholar] [CrossRef]
- Li, M.Z.; Huang, X.J.; Hu, J.L.; Cui, S.W.; Xie, M.Y.; Nie, S.P. The Protective Effects against Cyclophosphamide (CTX)-Induced Immunosuppression of Three Glucomannans. Food Hydrocoll. 2020, 100, 105445. [Google Scholar] [CrossRef]
- Lei, S.; Xiao, R.; Zhang, Y.; Huang, J.; Yan, J.; Lin, L.; Xia, B.; Wu, P. Seperation and Purification of Neutral Polysaccharides from Dendrobium officinale and Study of Its Immunoregulatory Effects. Tradit. Chin. Drug Res. Clin. Pharmacol. 2018, 29, 748–753. [Google Scholar] [CrossRef]
- Li, M.X.; Yue, H.; Wang, Y.Q.; Guo, C.L.; Du, Z.Y.; Jin, C.; Ding, K. Intestinal Microbes Derived Butyrate Is Related to the Immunomodulatory Activities of Dendrobium officinale Polysaccharide. Int. J. Biol. Macromol. 2020, 149, 717–723. [Google Scholar] [CrossRef]
- Zhang, S.; Tong, W.; Hu, J.; Huang, X.; Yin, J.; Nie, S. Immunomodulation of Polysaccharide Fractions from Dendrobium officinale and Their Effects on Colon Health of Mice. J. Chin. Inst. Food Sci. Technol. 2019, 19, 14–21. [Google Scholar] [CrossRef]
- Huang, X.; Nie, S.; Cai, H.; Zhang, G.; Cui, S.W.; Xie, M.; Phillips, G.O. Study on Dendrobium officinale O-Acetyl-Glucomannan (Dendronan): Part IV. Immunomodulatory Activity In Vivo. J. Funct. Foods 2015, 15, 525–532. [Google Scholar] [CrossRef]
- Shan, Z.; Wang, Y.; Jin, Z.; Liu, J.; Wang, N.; Guo, X.; Cui, S.W.; Guo, Q. Insight into the Structural and Immunomodulatory Relationships of Polysaccharides from Dendrobium officinale—An In Vivo Study. Food Hydrocoll. 2023, 139, 108560. [Google Scholar] [CrossRef]
- Furusawa, Y.; Obata, Y.; Fukuda, S.; Endo, T.A.; Nakato, G.; Takahashi, D.; Nakanishi, Y.; Uetake, C.; Kato, K.; Kato, T.; et al. Commensal Microbe-Derived Butyrate Induces the Differentiation of Colonic Regulatory T Cells. Nature 2013, 504, 446–450. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; He, T.; Cuan, X.; Wang, X.; Hu, J.; Sheng, J. 1,4-β-D-Glucomannan from Dendrobium officinale Activates Nf-Κb via Tlr4 to Regulate the Immune Response. Molecules 2018, 23, 2658. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Li, H.L.; Chen, J.Q.; He, L.; Du, X.H.; Zhou, L.; Xiong, Q.P.; Lai, X.P.; Yang, Y.Q.; Huang, S.; et al. Dendrobium officinale Polysaccharides Alleviate Colon Tumorigenesis via Restoring Intestinal Barrier Function and Enhancing Anti-Tumor Immune Response. Pharmacol. Res. 2019, 148, 104417. [Google Scholar] [CrossRef]
- Scharping, N.E.; Menk, A.V.; Moreci, R.S.; Whetstone, R.D.; Dadey, R.E.; Watkins, S.C.; Ferris, R.L.; Delgoffe, G.M. The Tumor Microenvironment Represses T Cell Mitochondrial Biogenesis to Drive Intratumoral T Cell Metabolic Insufficiency and Dysfunction. Immunity 2016, 45, 374–388. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, B.; Wang, G.; Ge, S.; Lan, X.; Xu, G.; Liu, H. Dendrobium officinale Polysaccharides Inhibit 1-Methyl-2-Nitro-1-Nitrosoguanidine Induced Precancerous Lesions of Gastric Cancer in Rats through Regulating Wnt/β-Catenin Pathway and Altering Serum Endogenous Metabolites. Molecules 2019, 24, 2660. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Zhang, N.; Xu, G.; Jiang, P.; Huang, S.; Zhao, Q.; He, Y. Anti-Tumor and Immunomodulation Activity of Polysaccharides from Dendrobium officinale in S180 Tumor-Bearing Mice. J. Funct. Foods 2022, 94, 105105. [Google Scholar] [CrossRef]
- Zhang, L.Y.; Wang, F.B.; Ren, X.B. Inhibitory Effect of Dendrobium officinale Polysaccharide on Human Gastric Cancer Cell Xenografts in Nude Mice. Food Sci. Technol. 2018, 38, 78–83. [Google Scholar] [CrossRef]
- Li, N.Y.; Wang, C.F.; Georgiev, M.I.; Bajpai, V.K.; Tundis, R.; Simal-Gandara, J.; Lu, X.M.; Xiao, J.B.; Tang, X.Z.; Qiao, X.G. Advances in Dietary Polysaccharides as Anticancer Agents: Structure-Activity Relationship. Trends Food Sci. Technol. 2021, 111, 360–377. [Google Scholar] [CrossRef]
- Cai, M.; Zhu, H.; Xu, L.; Wang, J.; Xu, J.; Li, Z.; Yang, K.; Wu, J.; Sun, P. Structure, Anti-Fatigue Activity and Regulation on Gut Microflora in Vivo of Ethanol-Fractional Polysaccharides from Dendrobium officinale. Int. J. Biol. Macromol. 2023, 234, 123572. [Google Scholar] [CrossRef] [PubMed]
- Zeng, C.; Tan, H.M. Gut Microbiota and Heart, Vascular Injury. In Gut Microbiota and Pathogenesis of Organ Injury; Chen, P., Ed.; Springer: Singapore, 2020; Volume 1238, pp. 107–141. ISBN 978-981-15-2384-7. [Google Scholar]
- Canfora, E.E.; Jocken, J.W.; Blaak, E.E. Short-Chain Fatty Acids in Control of Body Weight and Insulin Sensitivity. Nat. Rev. Endocrinol. 2015, 11, 577–591. [Google Scholar] [CrossRef] [PubMed]
- Hughes, R.L.; Kable, M.E.; Marco, M.; Keim, N.L. The Role of the Gut Microbiome in Predicting Response to Diet and the Development of Precision Nutrition Models. Part II: Results. Adv. Nutr. 2019, 10, 979–998. [Google Scholar] [CrossRef]
- Vrieze, A.; Van Nood, E.; Holleman, F.; Salojärvi, J.; Kootte, R.S.; Bartelsman, J.F.W.M.; Dallinga-Thie, G.M.; Ackermans, M.T.; Serlie, M.J.; Oozeer, R.; et al. Transfer of Intestinal Microbiota from Lean Donors Increases Insulin Sensitivity in Individuals with Metabolic Syndrome. Gastroenterology 2012, 143, 913–916.e7. [Google Scholar] [CrossRef] [PubMed]
- Mocanu, V.; Zhang, Z.; Deehan, E.C.; Kao, D.H.; Hotte, N.; Karmali, S.; Birch, D.W.; Samarasinghe, K.K.; Walter, J.; Madsen, K.L. Fecal Microbial Transplantation and Fiber Supplementation in Patients with Severe Obesity and Metabolic Syndrome: A Randomized Double-Blind, Placebo-Controlled Phase 2 Trial. Nat. Med. 2021, 27, 1272–1279. [Google Scholar] [CrossRef] [PubMed]
- Ndeh, D.; Gilbert, H.J. Biochemistry of Complex Glycan Depolymerisation by the Human Gut Microbiota. FEMS Microbiol. Rev. 2018, 42, 146–164. [Google Scholar] [CrossRef]
- Tan, H.; Zhai, Q.; Chen, W. Investigations of Bacteroides spp. towards next-Generation Probiotics. Food Res. Int. 2019, 116, 637–644. [Google Scholar] [CrossRef]
- Bolam, D.N.; Koropatkin, N.M. Glycan Recognition by the Bacteroidetes Sus-like Systems. Curr. Opin. Struct. Biol. 2012, 22, 563–569. [Google Scholar] [CrossRef]
- Martens, E.C.; Lowe, E.C.; Chiang, H.; Pudlo, N.A.; Wu, M.; McNulty, N.P.; Abbott, D.W.; Henrissat, B.; Gilbert, H.J.; Bolam, D.N.; et al. Recognition and Degradation of Plant Cell Wall Polysaccharides by Two Human Gut Symbionts. PLoS Biol. 2011, 9, e1001221. [Google Scholar] [CrossRef]
- Gao, G.; Cao, J.; Mi, L.; Feng, D.; Deng, Q.; Sun, X.; Zhang, H.; Wang, Q.; Wang, J. BdPUL12 Depolymerizes β-Mannan-like Glycans into Mannooligosaccharides and Mannose, Which Serve as Carbon Sources for Bacteroides dorei and Gut Probiotics. Int. J. Biol. Macromol. 2021, 187, 664–674. [Google Scholar] [CrossRef]
- Ulsemer, P.; Toutounian, K.; Kressel, G.; Goletz, C.; Schmidt, J.; Karsten, U.; Hahn, A.; Goletz, S. Impact of Oral Consumption of Heat-Treated Bacteroides xylanisolvens DSM 23964 on the Level of Natural TFa-Specific Antibodies in Human Adults. Benef. Microbes 2016, 7, 485–500. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Zhao, J.; Zhang, H.; Zhai, Q.; Chen, W. Novel Strains of Bacteroides fragilis and Bacteroides ovatus Alleviate the LPS-Induced Inflammation in Mice. Appl. Microbiol. Biotechnol. 2019, 103, 2353–2365. [Google Scholar] [CrossRef]
- Wu, M.; McNulty, N.P.; Rodionov, D.A.; Khoroshkin, M.S.; Griffin, N.W.; Cheng, J.; Latreille, P.; Kerstetter, R.A.; Terrapon, N.; Henrissat, B.; et al. Genetic Determinants of In Vivo Fitness and Diet Responsiveness in Multiple Human Gut Bacteroides. Science 2015, 350, aac5992. [Google Scholar] [CrossRef]
- Huang, J.; Wang, Q.; Xu, Q.; Zhang, Y.; Lin, B.; Guan, X.; Qian, L.; Zheng, Y. In Vitro Fermentation of O-acetyl-arabinoxylan from Bamboo Shavings by Human Colonic Microbiota. Int. J. Biol. Macromol. 2019, 125, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Accetto, T.; Avguštin, G. Polysaccharide Utilization Locus and CAZYme Genome Repertoires Reveal Diverse Ecological Adaptation of Prevotella Species. Syst. Appl. Microbiol. 2015, 38, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Emerson, E.L.; Weimer, P.J. Fermentation of Model Hemicelluloses by Prevotella strains and Butyrivibrio fibrisolvens in Pure Culture and in Ruminal Enrichment Cultures. Appl. Microbiol. Biotechnol. 2017, 101, 4269–4278. [Google Scholar] [CrossRef]
- Shahi, S.K.; Freedman, S.N.; Murra, A.C.; Zarei, K.; Sompallae, R.; Gibson-Corley, K.N.; Karandikar, N.J.; Murray, J.A.; Mangalam, A.K. Prevotella histicola, a Human Gut Commensal, Is as Potent as COPAXONE® in an Animal Model of Multiple Sclerosis. Front. Immunol. 2019, 10, 462. [Google Scholar] [CrossRef] [PubMed]
- Khalili, L.; Alipour, B.; Jafar-Abadi, M.A.; Faraji, I.; Hassanalilou, T.; Abbasi, M.M.; Vaghef-Mehrabany, E.; Sani, M.A. The Effects of Lactobacillus casei on Glycemic Response, Serum Sirtuin1 and Fetuin-A Levels in Patients with Type 2 Diabetes Mellitus: A Randomized Controlled Trial. Iran. Biomed. J. 2019, 23, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Akbari, E.; Asemi, Z.; Kakhaki, R.D.; Bahmani, F.; Kouchaki, E.; Tamtaji, O.R.; Hamidi, G.A.; Salami, M. Effect of Probiotic Supplementation on Cognitive Function and Metabolic Status in Alzheimer’s Disease: A Randomized, Double-Blind and Controlled Trial. Front. Aging Neurosci. 2016, 8, 256. [Google Scholar] [CrossRef] [PubMed]
- Kobyliak, N.; Falalyeyeva, T.; Mykhalchyshyn, G.; Kyriienko, D.; Komissarenko, I. Effect of Alive Probiotic on Insulin Resistance in Type 2 Diabetes Patients: Randomized Clinical Trial. Diabetes Metab. Syndr. Clin. Res. Rev. 2018, 12, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.W.; Gu, Y.L.; Mao, X.Q.; Zhang, L.; Pei, Y.F. Effects of Probiotics on Type II Diabetes Mellitus: A Meta-Analysis. J. Transl. Med. 2020, 18, 30. [Google Scholar] [CrossRef]
- Vakadaris, G.; Stefanis, C.; Giorgi, E.; Brouvalis, M.; Voidarou, C.; Kourkoutas, Y.; Tsigalou, C.; Bezirtzoglou, E. The Role of Probiotics in Inducing and Maintaining Remission in Crohn’s Disease and Ulcerative Colitis: A Systematic Review of the Literature. Biomedicines 2023, 11, 494. [Google Scholar] [CrossRef] [PubMed]
- Sheridan, P.O.; Martin, J.C.; Lawley, T.D.; Browne, H.P.; Harris, H.M.B.; Bernalier-Donadille, A.; Duncan, S.H.; O’Toole, P.W.; Scott, K.P.; Flint, H.J. Polysaccharide Utilization Loci and Nutritional Specialization in a Dominant Group of Butyrate-Producing Human Colonic Firmicutes. Microb. Genom. 2016, 2, e000043. [Google Scholar] [CrossRef]
- Usta-Gorgun, B.; Yilmaz-Ersan, L. Short-Chain Fatty Acids Production by Bifidobacterium Species in the Presence of Salep. Electron. J. Biotechnol. 2020, 47, 29–35. [Google Scholar] [CrossRef]
- Al-Ghazzewi, F.H.; Khanna, S.; Tester, R.F.; Piggott, J. The Potential Use of Hydrolysed Konjac Glucomannan as a Prebiotic. J. Sci. Food Agric. 2007, 87, 1758–1766. [Google Scholar] [CrossRef]
- Wang, C.H.; Lai, P.; Chen, M.E.; Chen, H.L. Antioxidative Capacity Produced by Bifidobacterium- and Lactobacillus Acidophilus-Mediated Fermentations of Konjac Glucomannan and Glucomannan Oligosaccharides. J. Sci. Food Agric. 2008, 88, 1294–1300. [Google Scholar] [CrossRef]
- Cozzolino, A.; Vergalito, F.; Tremonte, P.; Iorizzo, M.; Lombardi, S.J.; Sorrentino, E.; Luongo, D.; Coppola, R.; Di Marco, R.; Succi, M. Preliminary Evaluation of the Safety and Probiotic Potential of Akkermansia muciniphila DSM 22959 in Comparison with Lactobacillus rhamnosus GG. Microorganisms 2020, 8, 189. [Google Scholar] [CrossRef] [PubMed]
- Dao, M.C.; Everard, A.; Aron-Wisnewsky, J.; Sokolovska, N.; Prifti, E.; Verger, E.O.; Kayser, B.D.; Levenez, F.; Chilloux, J.; Hoyles, L.; et al. Akkermansia muciniphila and Improved Metabolic Health during a Dietary Intervention in Obesity: Relationship with Gut Microbiome Richness and Ecology. Gut 2016, 65, 426–436. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Hong, J.; Xu, X.; Feng, Q.; Zhang, D.; Gu, Y.; Shi, J.; Zhao, S.; Liu, W.; Wang, X.; et al. Gut Microbiome and Serum Metabolome Alterations in Obesity and after Weight-Loss Intervention. Nat. Med. 2017, 23, 859–868. [Google Scholar] [CrossRef]
- Li, J.; Zhao, F.; Wang, Y.; Chen, J.; Tao, J.; Tian, G.; Wu, S.; Liu, W.; Cui, Q.; Geng, B.; et al. Gut Microbiota Dysbiosis Contributes to the Development of Hypertension. Microbiome 2017, 5, 14. [Google Scholar] [CrossRef]
- Yassour, M.; Lim, M.Y.; Yun, H.S.; Tickle, T.L.; Sung, J.; Song, Y.M.; Lee, K.; Franzosa, E.A.; Morgan, X.C.; Gevers, D.; et al. Sub-Clinical Detection of Gut Microbial Biomarkers of Obesity and Type 2 Diabetes. Genome Med. 2016, 8, 17. [Google Scholar] [CrossRef] [PubMed]
- Brahe, L.K.; Le Chatelier, E.; Prifti, E.; Pons, N.; Kennedy, S.; Hansen, T.; Pedersen, O.; Astrup, A.; Ehrlich, S.D.; Larsen, L.H. Specific Gut Microbiota Features and Metabolic Markers in Postmenopausal Women with Obesity. Nutr. Diabetes 2015, 5, e159. [Google Scholar] [CrossRef] [PubMed]
- Depommier, C.; Everard, A.; Druart, C.; Plovier, H.; Van Hul, M.; Vieira-Silva, S.; Falony, G.; Raes, J.; Maiter, D.; Delzenne, N.M.; et al. Supplementation with Akkermansia muciniphila in Overweight and Obese Human Volunteers: A Proof-of-Concept Exploratory Study. Nat. Med. 2019, 25, 1096–1103. [Google Scholar] [CrossRef] [PubMed]
- van Passel, M.W.J.; Kant, R.; Zoetendal, E.G.; Plugge, C.M.; Derrien, M.; Malfatti, S.A.; Chain, P.S.G.; Woyke, T.; Palva, A.; de Vos, W.M.; et al. The Genome of Akkermansia muciniphila, a Dedicated Intestinal Mucin Degrader, and Its Use in Exploring Intestinal Metagenomes. PLoS ONE 2011, 6, e16876. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Li, Q.; Cheng, L.; Buch, H.; Zhang, F. Akkermansia muciniphila Is a Promising Probiotic. Microb. Biotechnol. 2019, 12, 1109–1125. [Google Scholar] [CrossRef]
- Shuoker, B.; Pichler, M.J.; Jin, C.; Sakanaka, H.; Wu, H.; Gascueña, A.M.; Liu, J.; Nielsen, T.S.; Holgersson, J.; Nordberg Karlsson, E.; et al. Sialidases and Fucosidases of Akkermansia muciniphila Are Crucial for Growth on Mucin and Nutrient Sharing with Mucus-Associated Gut Bacteria. Nat. Commun. 2023, 14, 1833. [Google Scholar] [CrossRef]
- Kirmiz, N.; Galindo, K.; Cross, K.L.; Luna, E.; Rhoades, N.; Podar, M.; Flores, G.E. Comparative Genomics Guides Elucidation of Vitamin B12 Biosynthesis in Novel Human-Associated Akkermansia Strains. Appl. Environ. Microbiol. 2020, 86, e02117-19. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Kawahara, T.; Inoue, S.; Kohda, N. Akkermansia biwaensis sp. Nov., an Anaerobic Mucin-Degrading Bacterium Isolated from Human Faeces. Int. J. Syst. Evol. Microbiol. 2023, 73, 005697. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, W.; Zhao, Z.; Zhao, Z.; Zhang, D.; Zhang, Q.; Zhang, J.; Fang, Z.; Bai, Y.; Guo, X. Structure, Health Benefits, Mechanisms, and Gut Microbiota of Dendrobium officinale Polysaccharides: A Review. Nutrients 2023, 15, 4901. https://doi.org/10.3390/nu15234901
Wu W, Zhao Z, Zhao Z, Zhang D, Zhang Q, Zhang J, Fang Z, Bai Y, Guo X. Structure, Health Benefits, Mechanisms, and Gut Microbiota of Dendrobium officinale Polysaccharides: A Review. Nutrients. 2023; 15(23):4901. https://doi.org/10.3390/nu15234901
Chicago/Turabian StyleWu, Weijie, Ziqi Zhao, Zhaoer Zhao, Dandan Zhang, Qianyi Zhang, Jiayu Zhang, Zhengyi Fang, Yiling Bai, and Xiaohui Guo. 2023. "Structure, Health Benefits, Mechanisms, and Gut Microbiota of Dendrobium officinale Polysaccharides: A Review" Nutrients 15, no. 23: 4901. https://doi.org/10.3390/nu15234901
APA StyleWu, W., Zhao, Z., Zhao, Z., Zhang, D., Zhang, Q., Zhang, J., Fang, Z., Bai, Y., & Guo, X. (2023). Structure, Health Benefits, Mechanisms, and Gut Microbiota of Dendrobium officinale Polysaccharides: A Review. Nutrients, 15(23), 4901. https://doi.org/10.3390/nu15234901






