Adherence to Healthy Dietary Patterns and Glioma: A Matched Case-Control Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Dietary Intake Assessment
2.3. Assessment of Dietary Patterns Based on Priori Methods
2.4. Assessment of Dietary Patterns Based on the Posterior Method
2.5. Covariates
2.6. Statistical Analysis
3. Results
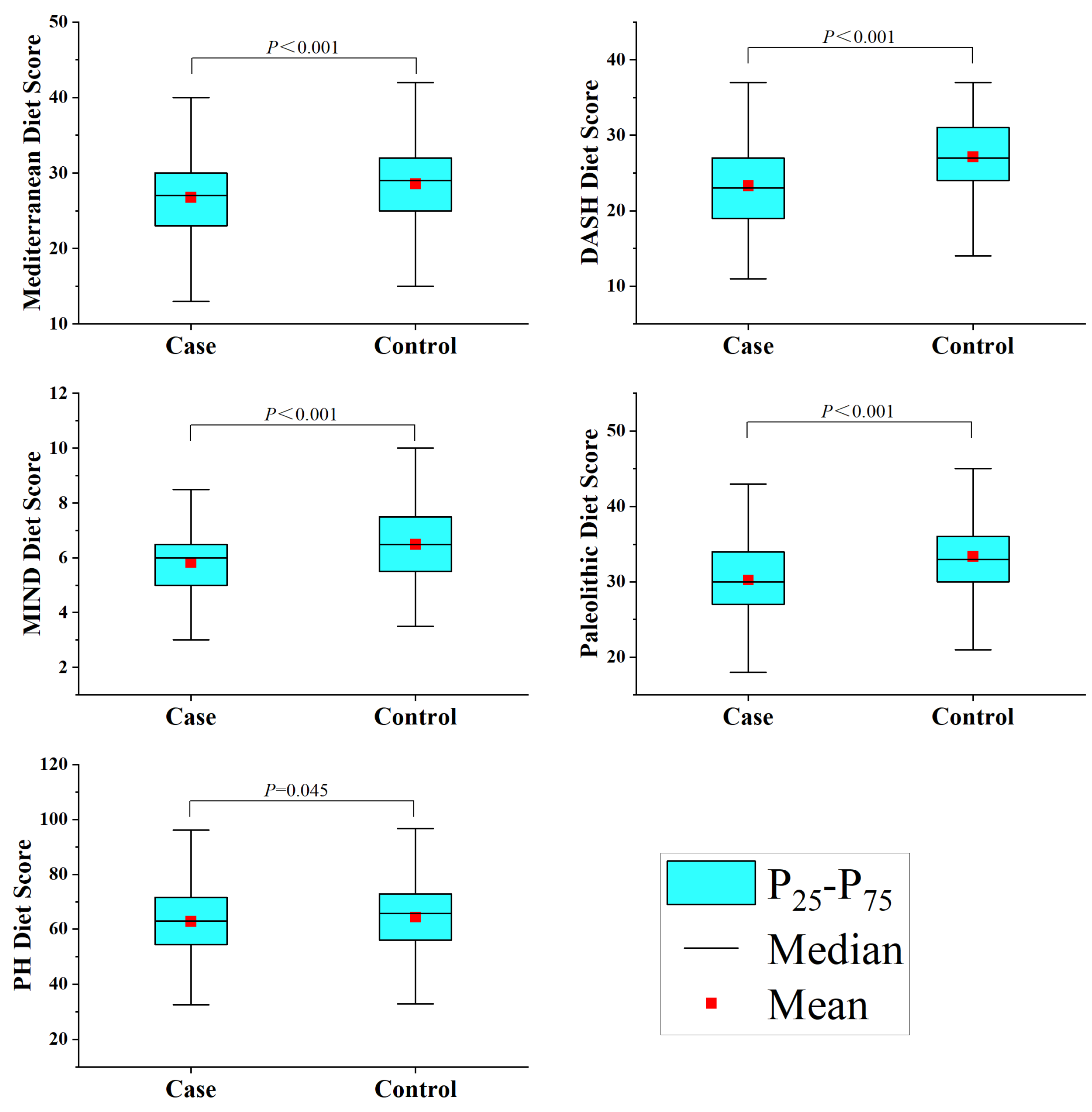
3.1. Characteristics of the Study Population and Dietary Patterns
3.2. Association between Dietary Pattern Score and Glioma
3.3. Dietary Pattern Score and Pathological Classification and Grade of Glioma
3.4. Posterior Method and Risk of Glioma
3.5. Subgroup Analysis
3.6. Dose–Response Relationship
3.7. Mediating Effect Based on BMI
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ostrom, Q.T.; Price, M.; Ryan, K.; Edelson, J.; Neff, C.; Cioffi, G.; Waite, K.A.; Kruchko, C.; Barnholtz-Sloan, J.S. CBTRUS Statistical Report: Pediatric Brain Tumor Foundation Childhood and Adolescent Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2014–2018. Neuro-Oncology 2022, 24, i1–i38. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.D.; Ostrom, Q.T.; Kruchko, C.; Patil, N.; Tihan, T.; Cioffi, G.; Fuchs, H.E.; Waite, K.A.; Jemal, A.; Siegel, R.L.; et al. Brain and other central nervous system tumor statistics, 2021. CA-Cancer J. Clin. 2021, 71, 381–406. [Google Scholar] [CrossRef] [PubMed]
- Bellail, A.C.; Hunter, S.B.; Brat, D.J.; Tan, C.; Van Meir, E.G. Microregional extracellular matrix heterogeneity in brain modulates glioma cell invasion. Int. J. Biochem. Cell Biol. 2004, 36, 1046–1069. [Google Scholar] [CrossRef] [PubMed]
- Walsh, K.M.; Claus, E.B. Diet and risk of glioma: Targets for prevention remain elusive. Neuro-Oncology 2019, 21, 832–833. [Google Scholar] [CrossRef]
- Clinton, S.K.; Giovannucci, E.L.; Hursting, S.D. The World Cancer Research Fund/American Institute for Cancer Research Third Expert Report on Diet, Nutrition, Physical Activity, and Cancer: Impact and Future Directions. J. Nutr. 2020, 150, 663–671. [Google Scholar] [CrossRef]
- Mayne, S.T.; Playdon, M.C.; Rock, C.L. Diet, nutrition, and cancer: Past, present and future. Nat. Rev. Clin. Oncol. 2016, 13, 504–515. [Google Scholar] [CrossRef]
- Aune, D.; Giovannucci, E.; Boffetta, P.; Fadnes, L.T.; Keum, N.; Norat, T.; Greenwood, D.C.; Riboli, E.; Vatten, L.J.; Tonstad, S. Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality—A systematic review and dose-response meta-analysis of prospective studies. Int. J. Epidemiol. 2017, 46, 1029–1056. [Google Scholar] [CrossRef]
- Nagao, M.; Tsugane, S. Cancer in Japan: Prevalence, prevention and the role of heterocyclic amines in human carcinogenesis. Genes Environ. 2016, 38, 16. [Google Scholar] [CrossRef]
- Bielecka, J.; Markiewicz-Zukowska, R. The Influence of Nutritional and Lifestyle Factors on Glioma Incidence. Nutrients 2020, 12, 1812. [Google Scholar] [CrossRef]
- Terry, M.B.; Howe, G.; Pogoda, J.M.; Zhang, F.F.; Ahlbom, A.; Choi, W.; Giles, G.G.; Little, J.; Lubin, F.; Menegoz, F.; et al. An international case-control study of adult diet and brain tumor risk: A histology-specific analysis by food group. Ann. Epidemiol. 2009, 19, 161–171. [Google Scholar] [CrossRef]
- Lee, M.; Wrensch, M.; Miike, R. Dietary and tobacco risk factors for adult onset glioma in the San Francisco Bay Area (California, USA). Cancer Cause Control 1997, 8, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Shahrestani, M.A.; Saneei, P.; Shayanfar, M.; Mohammad-Shirazi, M.; Sharifi, G.; Sadeghi, O.; Esmaillzadeh, A. The relationship between rice consumption and glioma: A case-control study in adults. Sci. Rep. 2021, 11, 6073. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Wang, Z.; Jin, Y.; Guo, J. Association between tea and coffee consumption and brain cancer risk: An updated meta-analysis. World J. Surg. Oncol. 2019, 17, 51. [Google Scholar] [CrossRef]
- Zhang, W.; Jiang, J.; Li, X.; He, Y.; Chen, F.; Li, W. Dietary Factors and Risk of Glioma in Adults: A Systematic Review and Dose-Response Meta-Analysis of Observational Studies. Front. Nutr. 2022, 9, 834258. [Google Scholar] [CrossRef] [PubMed]
- Dianatinasab, M.; Wesselius, A.; Salehi-Abargouei, A.; Yu, E.; Brinkman, M.; Fararouei, M.; van den Brandt, P.; White, E.; Weiderpass, E.; Le Calvez-Kelm, F.; et al. Adherence to a Western dietary pattern and risk of bladder cancer: A pooled analysis of 13 cohort studies of the Bladder Cancer Epidemiology and Nutritional Determinants international study. Int. J. Cancer 2020, 147, 3394–3403. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.B. Dietary pattern analysis: A new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Mahamat-Saleh, Y.; Cervenka, I.; Al, R.M.; Savoye, I.; Mancini, F.R.; Trichopoulou, A.; Boutron-Ruault, M.C.; Kvaskoff, M. Mediterranean dietary pattern and skin cancer risk: A prospective cohort study in French women. Am. J. Clin. Nutr. 2019, 110, 993–1002. [Google Scholar] [CrossRef]
- Gioxari, A.; Tzanos, D.; Kostara, C.; Papandreou, P.; Mountzios, G.; Skouroliakou, M. Mediterranean Diet Implementation to Protect against Advanced Lung Cancer Index (ALI) Rise: Study Design and Preliminary Results of a Randomised Controlled Trial. Int. J. Env. Res. Pub. Health 2021, 18, 3700. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, J.; Choi, I.J.; Kim, Y.I.; Kim, J. Dietary patterns and gastric cancer risk in a Korean population: A case-control study. Eur. J. Nutr. 2021, 60, 389–397. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Shayanfar, M.; Rigi, S.; Mohammad-Shirazi, M.; Sharifi, G.; Esmaillzadeh, A. Adherence to the Mediterranean dietary pattern in relation to glioma: A case-control study. Clin. Nutr. 2021, 40, 313–319. [Google Scholar] [CrossRef]
- Kuan, A.S.; Green, J.; Kitahara, C.M.; Berrington, D.G.A.; Key, T.; Reeves, G.K.; Floud, S.; Balkwill, A.; Bradbury, K.; Liao, L.M.; et al. Diet and risk of glioma: Combined analysis of 3 large prospective studies in the UK and USA. Neuro-Oncology 2019, 21, 944–952. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; He, Y.; Kang, X.; Wang, C.; Chen, F.; Kang, Z.; Yang, S.; Zhang, R.; Peng, Y.; Li, W. Association between dietary minerals and glioma: A case-control study based on Chinese population. Front. Nutr. 2023, 10, 1118997. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Perry, A.; Wesseling, P.; Brat, D.J.; Cree, I.A.; Figarella-Branger, D.; Hawkins, C.; Ng, H.K.; Pfister, S.M.; Reifenberger, G.; et al. The 2021 WHO Classification of Tumors of the Central Nervous System: A summary. Neuro-Oncology 2021, 23, 1231–1251. [Google Scholar] [CrossRef]
- Zhao, W.-H.; Huang, Z.P.; Zhang, X.; He, L.; Willett, W.; Wang, J.-L.; Hasegawa, K.; Chen, J.-S. Reproducibility and Validity of a Chinese Food Frequency Questionnaire. Biomed. Environ. Sci. 2010, 23, 1–38. [Google Scholar] [CrossRef]
- Yang, Y.X. China Food Composition Tables; Standard Edition; Peking University Medical Press: Beijing, China, 2018. [Google Scholar]
- Morgan, L.L.; Miller, A.B.; Sasco, A.; Davis, D.L. Mobile phone radiation causes brain tumors and should be classified as a probable human carcinogen (2A) (review). Int. J. Oncol. 2015, 46, 1865–1871. [Google Scholar] [CrossRef]
- Craig, C.L.; Marshall, A.L.; Sjostrom, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exer. 2003, 35, 1381–1395. [Google Scholar] [CrossRef]
- Zhang, W.; Du, J.; Li, H.; Yang, Y.; Cai, C.; Gao, Q.; Xing, Y.; Shao, B.; Li, G. Multiple-element exposure and metabolic syndrome in Chinese adults: A case-control study based on the Beijing population health cohort. Environ. Int. 2020, 143, 105959. [Google Scholar] [CrossRef]
- Moon, K.A.; Guallar, E.; Umans, J.G.; Devereux, R.B.; Best, L.G.; Francesconi, K.A.; Goessler, W.; Pollak, J.; Silbergeld, E.K.; Howard, B.V.; et al. Association between exposure to low to moderate arsenic levels and incident cardiovascular disease. A prospective cohort study. Ann. Intern. Med. 2013, 159, 649–659. [Google Scholar] [CrossRef]
- Imai, K.; Keele, L.; Tingley, D. A general approach to causal mediation analysis. Psychol. Methods 2010, 15, 309–334. [Google Scholar] [CrossRef]
- Valente, M.J.; Rijnhart, J.; Smyth, H.L.; Muniz, F.B.; MacKinnon, D.P. Causal Mediation Programs in R, Mplus, SAS, SPSS, and Stata. Struct. Equ. Model. 2020, 27, 975–984. [Google Scholar] [CrossRef]
- Tosti, V.; Bertozzi, B.; Fontana, L. Health Benefits of the Mediterranean Diet: Metabolic and Molecular Mechanisms. J. Gerontol. A-Biol. 2018, 73, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Schulpen, M.; van den Brandt, P.A. Adherence to the Mediterranean Diet and Overall Cancer Incidence: The Netherlands Cohort Study. J. Acad. Nutr. Diet. 2021, 121, 242–252. [Google Scholar] [CrossRef] [PubMed]
- Krusinska, B.; Hawrysz, I.; Wadolowska, L.; Slowinska, M.A.; Biernacki, M.; Czerwinska, A.; Golota, J.J. Associations of Mediterranean Diet and a Posteriori Derived Dietary Patterns with Breast and Lung Cancer Risk: A Case-Control Study. Nutrients 2018, 10, 470. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Schwedhelm, C.; Galbete, C.; Hoffmann, G. Adherence to Mediterranean Diet and Risk of Cancer: An Updated Systematic Review and Meta-Analysis. Nutrients 2017, 9, 1063. [Google Scholar] [CrossRef] [PubMed]
- Naska, A.; Trichopoulou, A. Back to the future: The Mediterranean diet paradigm. Nutr. Metab. Cardiovas. 2014, 24, 216–219. [Google Scholar] [CrossRef] [PubMed]
- Emma, M.R.; Augello, G.; Di Stefano, V.; Azzolina, A.; Giannitrapani, L.; Montalto, G.; Cervello, M.; Cusimano, A. Potential Uses of Olive Oil Secoiridoids for the Prevention and Treatment of Cancer: A Narrative Review of Preclinical Studies. Int. J. Mol. Sci. 2021, 22, 1234. [Google Scholar] [CrossRef] [PubMed]
- Mentella, M.C.; Scaldaferri, F.; Ricci, C.; Gasbarrini, A.; Miggiano, G. Cancer and Mediterranean Diet: A Review. Nutrients 2019, 11, 2059. [Google Scholar] [CrossRef]
- Sanchez-Melgar, A.; Munoz-Lopez, S.; Albasanz, J.L.; Martin, M. Antitumoral Action of Resveratrol through Adenosinergic Signaling in C6 Glioma Cells. Front. Neurosci. 2021, 15, 702817. [Google Scholar] [CrossRef]
- Bona, N.P.; Soares, M.; Pedra, N.S.; Spohr, L.; Da, S.D.S.F.; de Farias, A.S.; Alvez, F.L.; de Moraes, M.B.; Luduvico, K.P.; Spanevello, R.M.; et al. Tannic Acid Attenuates Peripheral and Brain Changes in a Preclinical Rat Model of Glioblastoma by Modulating Oxidative Stress and Purinergic Signaling. Neurochem. Res. 2022, 47, 1541–1552. [Google Scholar] [CrossRef]
- Maimaitili, A.; Shu, Z.; Cheng, X.; Kaheerman, K.; Sikandeer, A.; Li, W. Arctigenin, a natural lignan compound, induces G0/G1 cell cycle arrest and apoptosis in human glioma cells. Oncol. Lett. 2017, 13, 1007–1013. [Google Scholar] [CrossRef]
- Tamtaji, O.R.; Razavi, Z.S.; Razzaghi, N.; Aschner, M.; Barati, E.; Mirzaei, H. Quercetin and Glioma: Which Signaling Pathways are Involved? Curr. Mol. Pharmacol. 2022, 15, 962–968. [Google Scholar] [CrossRef] [PubMed]
- Amor, S.; Chalons, P.; Aires, V.; Delmas, D. Polyphenol Extracts from Red Wine and Grapevine: Potential Effects on Cancers. Diseases 2018, 6, 106. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Critselis, E. Mediterranean diet and longevity. Eur. J. Cancer Prev. 2004, 13, 453–456. [Google Scholar] [CrossRef] [PubMed]
- Soltani, S.; Arablou, T.; Jayedi, A.; Salehi-Abargouei, A. Adherence to the dietary approaches to stop hypertension (DASH) diet in relation to all-cause and cause-specific mortality: A systematic review and dose-response meta-analysis of prospective cohort studies. Nutr. J. 2020, 19, 37. [Google Scholar] [CrossRef] [PubMed]
- Toorang, F.; Sasanfar, B.; Hadji, M.; Esmaillzadeh, A.; Zendehdel, K. Adherence to “dietary approaches to stop hypertension” eating plan in relation to gastric cancer. Nutr. J. 2020, 19, 40. [Google Scholar] [CrossRef]
- Benisi-Kohansal, S.; Shayanfar, M.; Mohammad-Shirazi, M.; Tabibi, H.; Sharifi, G.; Saneei, P.; Esmaillzadeh, A. Adherence to the Dietary Approaches to Stop Hypertension-style diet in relation to glioma: A case-control study. Brit. J. Nutr. 2016, 115, 1108–1116. [Google Scholar] [CrossRef]
- McCullough, M.L.; Giovannucci, E.L. Diet and cancer prevention. Oncogene 2004, 23, 6349–6364. [Google Scholar] [CrossRef]
- Hung, C.F.; Lu, K.H. Vitamin C inhibited DNA adduct formation and arylamine N-acetyltransferase activity and gene expression in rat glial tumor cells. Neurochem. Res. 2001, 26, 1107–1112. [Google Scholar] [CrossRef]
- Hervouet, E.; Debien, E.; Campion, L.; Charbord, J.; Menanteau, J.; Vallette, F.M.; Cartron, P.F. Folate supplementation limits the aggressiveness of glioma via the remethylation of DNA repeats element and genes governing apoptosis and proliferation. Clin. Cancer Res. 2009, 15, 3519–3529. [Google Scholar] [CrossRef]
- Lijinsky, W. N-Nitroso compounds in the diet. Mutat. Res.-Genet. Toxicol. Environ. Mutagen. 1999, 443, 129–138. [Google Scholar] [CrossRef]
- Mirvish, S.S. Role of N-nitroso compounds (NOC) and N-nitrosation in etiology of gastric, esophageal, nasopharyngeal and bladder cancer and contribution to cancer of known exposures to NOC. Cancer Lett. 1995, 93, 17–48. [Google Scholar] [CrossRef] [PubMed]
- Goth, R.; Rajewsky, M.F. Persistence of O6-ethylguanine in rat-brain DNA: Correlation with nervous system-specific carcinogenesis by ethylnitrosourea. Proc. Natl. Acad. Sci. USA 1974, 71, 639–643. [Google Scholar] [CrossRef] [PubMed]
- Galvao, R.P.; Zong, H. Inflammation and Gliomagenesis: Bi-Directional Communication at Early and Late Stages of Tumor Progression. Curr. Pathobiol. Rep. 2013, 1, 19–28. [Google Scholar] [CrossRef]
- Blaylock, R.L. Immunoexcitatory mechanisms in glioma proliferation, invasion and occasional metastasis. Surg. Neurol. Int. 2013, 4, 15. [Google Scholar] [CrossRef] [PubMed]
- Conti, A.; Guli, C.; La Torre, D.; Tomasello, C.; Angileri, F.F.; Aguennouz, M. Role of inflammation and oxidative stress mediators in gliomas. Cancers 2010, 2, 693–712. [Google Scholar] [CrossRef] [PubMed]
- Hodson, L.; Harnden, K.E.; Roberts, R.; Dennis, A.L.; Frayn, K.N. Does the DASH diet lower blood pressure by altering peripheral vascular function? J. Hum. Hypertens. 2010, 24, 312–319. [Google Scholar] [CrossRef]
- Azadbakht, L.; Surkan, P.J.; Esmaillzadeh, A.; Willett, W.C. The Dietary Approaches to Stop Hypertension eating plan affects C-reactive protein, coagulation abnormalities, and hepatic function tests among type 2 diabetic patients. J. Nutr. 2011, 141, 1083–1088. [Google Scholar] [CrossRef]
- Marcason, W. What Are the Components to the MIND Diet? J. Acad. Nutr. Diet. 2015, 115, 1744. [Google Scholar] [CrossRef]
- Morris, M.C.; Tangney, C.C.; Wang, Y.; Sacks, F.M.; Barnes, L.L.; Bennett, D.A.; Aggarwal, N.T. MIND diet slows cognitive decline with aging. Alzheimers Dement. 2015, 11, 1015–1022. [Google Scholar] [CrossRef]
- Aghamohammadi, V.; Salari-Moghaddam, A.; Benisi-Kohansal, S.; Taghavi, M.; Azadbakht, L.; Esmaillzadeh, A. Adherence to the MIND Diet and Risk of Breast Cancer: A Case-control Study. Clin. Breast Cancer 2021, 21, e158–e164. [Google Scholar] [CrossRef]
- Sheikhhossein, F.; Imani, H.; Amini, M.R.; Hosseini, F.; Shab-Bidar, S. The association between adherence to MIND diet and risk of breast cancer: A case-control study. Int. J. Clin. Pract. 2021, 75, e14780. [Google Scholar] [CrossRef] [PubMed]
- Soltani, S.; Shayanfar, M.; Benisi-Kohansal, S.; Mohammad-Shirazi, M.; Sharifi, G.; Djazayeri, A.; Esmaillzadeh, A. Adherence to the MIND diet in relation to glioma: A case-control study. Nutr. Neurosci. 2022, 25, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Lehrer, S. Glioma and Alzheimer’s Disease. J. Alzheimers Dis. Rep. 2018, 2, 213–218. [Google Scholar] [CrossRef] [PubMed]
- de la O, V.; Zazpe, I.; Martinez, J.A.; Santiago, S.; Carlos, S.; Zulet, M.A.; Ruiz-Canela, M. Scoping review of Paleolithic dietary patterns: A definition proposal. Nutr. Res. Rev. 2021, 34, 78–106. [Google Scholar] [CrossRef]
- de la O, V.; Zazpe, I.; Goni, L.; Santiago, S.; Martin-Calvo, N.; Bes-Rastrollo, M.; Martinez, J.A.; Martinez-Gonzalez, M.A.; Ruiz-Canela, M. A score appraising Paleolithic diet and the risk of cardiovascular disease in a Mediterranean prospective cohort. Eur. J. Nutr. 2022, 61, 957–971. [Google Scholar] [CrossRef]
- Whalen, K.A.; Judd, S.; McCullough, M.L.; Flanders, W.D.; Hartman, T.J.; Bostick, R.M. Paleolithic and Mediterranean Diet Pattern Scores Are Inversely Associated with All-Cause and Cause-Specific Mortality in Adults. J. Nutr. 2017, 147, 612–620. [Google Scholar] [CrossRef]
- Klement, R.J.; Koebrunner, P.S.; Krage, K.; Weigel, M.M.; Sweeney, R.A. Short-term effects of a Paleolithic lifestyle intervention in breast cancer patients undergoing radiotherapy: A pilot and feasibility study. Med. Oncol. 2020, 38, 1. [Google Scholar] [CrossRef]
- Fenton, T.R.; Fenton, C.J. Paleo diet still lacks evidence. Am. J. Clin. Nutr. 2016, 104, 844. [Google Scholar] [CrossRef]
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radic. Bio. Med. 2010, 49, 1603–1616. [Google Scholar] [CrossRef]
- Nowsheen, S.; Aziz, K.; Kryston, T.B.; Ferguson, N.F.; Georgakilas, A. The interplay between inflammation and oxidative stress in carcinogenesis. Curr. Mol. Med. 2012, 12, 672–680. [Google Scholar] [CrossRef]
- Willett, W.; Rockstrom, J.; Loken, B.; Springmann, M.; Lang, T.; Vermeulen, S.; Garnett, T.; Tilman, D.; DeClerck, F.; Wood, A.; et al. Food in the Anthropocene: The EAT-Lancet Commission on healthy diets from sustainable food systems. Lancet 2019, 393, 447–492. [Google Scholar] [CrossRef] [PubMed]
- Montejano, V.R.; Schulz, C.A.; van de Locht, K.; Oluwagbemigun, K.; Alexy, U.; Nothlings, U. Associations of Adherence to a Dietary Index Based on the EAT-Lancet Reference Diet with Nutritional, Anthropometric, and Ecological Sustainability Parameters: Results from the German DONALD Cohort Study. J. Nutr. 2022, 152, 1763–1772. [Google Scholar] [CrossRef]
- Cacau, L.T.; De Carli, E.; de Carvalho, A.M.; Lotufo, P.A.; Moreno, L.A.; Bensenor, I.M.; Marchioni, D.M. Development and Validation of an Index Based on EAT-Lancet Recommendations: The Planetary Health Diet Index. Nutrients 2021, 13, 1698. [Google Scholar] [CrossRef]
- Marchioni, D.M.; Cacau, L.T.; De Carli, E.; Carvalho, A.M.; Rulli, M.C. Low Adherence to the EAT-Lancet Sustainable Reference Diet in the Brazilian Population: Findings from the National Dietary Survey 2017–2018. Nutrients 2022, 14, 1187. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Chai, L. How Far Are We from the Planetary Health Diet? A Threshold Regression Analysis of Global Diets. Foods 2022, 11, 986. [Google Scholar] [CrossRef]
- Malmir, H.; Shayanfar, M.; Mohammad-Shirazi, M.; Tabibi, H.; Sharifi, G.; Esmaillzadeh, A. Patterns of nutrients intakes in relation to glioma: A case-control study. Clin. Nutr. 2019, 38, 1406–1413. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Chen, J.; Wang, J.; Gong, S.; Jin, H.; Sheng, P.; Qi, X.; Lv, L.; Dong, Y.; Hou, L. Body mass index and risk of brain tumors: A systematic review and dose-response meta-analysis. Eur. J. Clin. Nutr. 2016, 70, 757–765. [Google Scholar] [CrossRef]
- Rieger, J.; Bahr, O.; Maurer, G.D.; Hattingen, E.; Franz, K.; Brucker, D.; Walenta, S.; Kammerer, U.; Coy, J.F.; Weller, M.; et al. ERGO: A pilot study of ketogenic diet in recurrent glioblastoma. Int. J. Oncol. 2014, 44, 1843–1852. [Google Scholar] [CrossRef]
- Porper, K.; Shpatz, Y.; Plotkin, L.; Pechthold, R.G.; Talianski, A.; Champ, C.E.; Furman, O.; Shimoni-Sebag, A.; Symon, Z.; Amit, U.; et al. A Phase I clinical trial of dose-escalated metabolic therapy combined with concomitant radiation therapy in high-grade glioma. J. Neuro-Oncol. 2021, 153, 487–496. [Google Scholar] [CrossRef]
- Zhao, J.; Li, Z.; Gao, Q.; Zhao, H.; Chen, S.; Huang, L.; Wang, W.; Wang, T. A review of statistical methods for dietary pattern analysis. Nutr. J. 2021, 20, 37. [Google Scholar] [CrossRef]
- Panagiotakos, D.B.; Pitsavos, C.; Stefanadis, C. Dietary patterns: A Mediterranean diet score and its relation to clinical and biological markers of cardiovascular disease risk. Nutr. Metab. Cardiovas. 2006, 16, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Vogt, T.M.; Appel, L.J.; Obarzanek, E.; Moore, T.J.; Vollmer, W.M.; Svetkey, L.P.; Sacks, F.M.; Bray, G.A.; Cutler, J.A.; Windhauser, M.M.; et al. Dietary Approaches to Stop Hypertension: Rationale, design, and methods. DASH Collaborative Research Group. J. Am. Diet. Assoc. 1999, 99, S12–S18. [Google Scholar] [CrossRef] [PubMed]
- Bertoia, M.L.; Triche, E.W.; Michaud, D.S.; Baylin, A.; Hogan, J.W.; Neuhouser, M.L.; Tinker, L.F.; Van Horn, L.; Waring, M.E.; Li, W.; et al. Mediterranean and Dietary Approaches to Stop Hypertension dietary patterns and risk of sudden cardiac death in postmenopausal women. Am. J. Clin. Nutr. 2014, 99, 344–351. [Google Scholar] [CrossRef] [PubMed]

| Age ≤ 40 (n = 500) | Age > 40 (n = 512) | |||||
|---|---|---|---|---|---|---|
| Case (n = 239) | Control (n = 261) | p a | Case (n = 267) | Control (n = 245) | p a | |
| Age (years) | 31.10 ± 6.14 | 30.70 ± 5.10 | 0.427 | 52.93 ± 8.11 | 52.28 ± 8.53 | 0.379 |
| Sex, n (%) | 0.880 | 0.885 | ||||
| Male | 55.6 | 56.3 | 56.6 | 55.9 | ||
| Female | 44.4 | 43.7 | 43.4 | 44.1 | ||
| BMI (kg/m2) | 24.01 ± 3.63 | 22.35 ± 3.40 | <0.001 | 24.04 ± 2.88 | 23.80 ± 2.96 | 0.347 |
| High-risk residential area, (%) | 0.051 | 0.295 | ||||
| Yes | 23.4 | 16.5 | 19.5 | 23.3 | ||
| No | 76.6 | 83.5 | 80.5 | 76.7 | ||
| Occupation, (%) | 0.058 | 0.327 | ||||
| Manual workers | 20.1 | 12.3 | 32.2 | 29.0 | ||
| Mental workers | 71.5 | 78.1 | 35.2 | 41.6 | ||
| Others | 8.4 | 9.6 | 32.6 | 29.4 | ||
| Education level, (%) | <0.001 | <0.001 | ||||
| Primary school and below | 1.3 | 0.8 | 12.0 | 4.5 | ||
| Middle school | 31.0 | 11.1 | 50.9 | 40.0 | ||
| University and above | 67.7 | 88.1 | 37.1 | 55.5 | ||
| Household income, (%) | <0.001 | <0.001 | ||||
| <3000 ¥/month | 5.4 | 14.6 | 13.5 | 22.0 | ||
| 3000–10,000 ¥/month | 78.7 | 52.1 | 73.4 | 46.2 | ||
| >10,000 ¥/month | 15.9 | 33.3 | 13.1 | 31.8 | ||
| Smoking status, (%) | 0.017 | 0.285 | ||||
| Never | 72.0 | 81.6 | 68.2 | 68.5 | ||
| Former smoker | 7.5 | 3.1 | 17.6 | 13.5 | ||
| Current smoker | 20.5 | 15.3 | 14.2 | 18.0 | ||
| Alcohol consumption, (%) | <0.001 | <0.001 | ||||
| Never | 68.2 | 56.3 | 60.3 | 56.3 | ||
| Occasional drinker | 13.8 | 32.6 | 12.0 | 27.8 | ||
| Frequent drinker | 18.0 | 11.1 | 27.7 | 15.9 | ||
| History of allergies, (%) | 0.005 | 0.037 | ||||
| Yes | 7.9 | 16.1 | 7.5 | 13.1 | ||
| No | 92.1 | 83.9 | 92.5 | 86.9 | ||
| History of head trauma, (%) | 0.923 | 0.269 | ||||
| Yes | 10.5 | 10.7 | 12.0 | 9.0 | ||
| No | 89.5 | 89.3 | 88.0 | 91.0 | ||
| Family history of cancer, (%) | 0.814 | <0.001 | ||||
| Yes | 24.3 | 23.4 | 35.2 | 18.8 | ||
| No | 75.7 | 76.6 | 64.8 | 81.2 | ||
| Physical activity, (%) | <0.001 | <0.001 | ||||
| Low | 15.9 | 48.7 | 11.6 | 42.8 | ||
| Moderate | 45.2 | 33.7 | 37.8 | 39.2 | ||
| Extreme | 38.9 | 17.6 | 50.6 | 18.0 | ||
| T1 | T2 | T3 | Continuous c | p-continuous | |
|---|---|---|---|---|---|
| Mediterranean Diet | ≤26 | 27–30 | >30 | ||
| Case/Control | 245/166 | 144/151 | 117/189 | ||
| Model 1 a | 1 | 0.61 (0.45–0.84) | 0.43 (0.31–0.58) | 0.94 (0.92–0.96) | <0.001 |
| Model 2 b | 1 | 0.41 (0.23–0.74) | 0.29 (0.17–0.52) | 0.92 (0.88–0.96) | <0.001 |
| DASH Diet | ≤23 | 24–28 | >28 | ||
| Case/Control | 268/117 | 160/183 | 78/206 | ||
| Model 1 a | 1 | 0.35 (0.25–0.50) | 0.15 (0.10–0.23) | 0.84 (0.82–0.87) | <0.001 |
| Model 2 b | 1 | 0.30 (0.16–0.56) | 0.09 (0.04–0.18) | 0.80 (0.74–0.85) | <0.001 |
| MIND | ≤5.5 | 6–6.5 | >6.5 | ||
| Case/Control | 249/142 | 146/150 | 111/214 | ||
| Model 1 a | 1 | 0.57 (0.41–0.78) | 0.28 (0.20–0.40) | 0.64 (0.57–0.71) | <0.001 |
| Model 2 b | 1 | 0.48 (0.28–0.85) | 0.25 (0.14–0.44) | 0.55 (0.44–0.68) | <0.001 |
| Paleolithic Diet | ≤30 | 31–34 | >34 | ||
| Case/Control | 271/131 | 141/163 | 94/212 | ||
| Model 1 a | 1 | 0.35 (0.25–0.50) | 0.20 (0.14–0.28) | 0.85 (0.83–0.88) | <0.001 |
| Model 2 b | 1 | 0.31 (0.16–0.58) | 0.13 (0.06–0.25) | 0.82 (0.77–0.87) | <0.001 |
| PH Diet | ≤58.61 | 58.61–69.64 | >69.64 | ||
| Case/Control | 182/156 | 171/166 | 153/184 | ||
| Model 1 a | 1 | 0.87 (0.64–1.18) | 0.70 (0.51–0.95) | 0.99 (0.98–1.00) | 0.035 |
| Model 2 b | 1 | 0.94 (0.55–1.60) | 0.61 (0.35–1.08) | 0.99 (0.97–1.01) | 0.198 |
| Pathological Classification a | Model 1 b | p | Model 2 c | p |
|---|---|---|---|---|
| Astrocytoma | ||||
| Mediterranean Diet | 0.91 (0.86–0.97) | 0.005 | 0.84 (0.72–0.99) | 0.031 |
| DASH Diet | 0.79 (0.71–0.87) | <0.001 | 0.62 (0.45–0.85) | 0.003 |
| MIND Diet | 0.64 (0.50–0.83) | 0.001 | 0.48 (0.27–0.86) | 0.013 |
| Paleolithic Diet | 0.84 (0.78–0.91) | <0.001 | 0.65 (0.48–0.88) | 0.006 |
| PH Diet | 0.99 (0.97–1.01) | 0.466 | 1.01 (0.97–1.05) | 0.794 |
| Glioblastoma | ||||
| Mediterranean Diet | 0.94 (0.91–0.98) | 0.001 | 0.91 (0.84–0.99) | 0.028 |
| DASH Diet | 0.84 (0.80–0.88) | <0.001 | 0.73 (0.62–0.85) | <0.001 |
| MIND Diet | 0.65 (0.55–0.77) | <0.001 | 0.44 (0.27–0.72) | 0.001 |
| Paleolithic Diet | 0.86 (0.82–0.90) | <0.001 | 0.77 (0.67–0.88) | <0.001 |
| PH Diet | 0.99 (0.98–1.01) | 0.265 | 1.00 (0.97–1.04) | 0.990 |
| T1 | T2 | T3 | p-trend | |
|---|---|---|---|---|
| Factor 1 | ≤−0.46 | −0.46–0.94 | >0.94 | |
| Case/Control | 328/169 | 123/169 | 55/168 | |
| Model 1 a | 1 | 0.34 (0.24–0.48) | 0.17 (0.11–0.25) | <0.001 |
| Model 2 b | 1 | 0.16 (0.08–0.30) | 0.03 (0.01–0.08) | <0.001 |
| Factor 2 | ≤−0.94 | −0.94–−0.26 | >−0.26 | |
| Case/Control | 85/171 | 81/167 | 340/168 | |
| Model 1 a | 1 | 0.98 (0.65–1.46) | 3.97 (2.79–5.64) | <0.001 |
| Model 2 b | 1 | 1.27 (0.66–2.46) | 4.99 (2.56–9.71) | <0.001 |
| Factor 3 | ≤−0.12 | −0.12–0.33 | >0.33 | |
| Case/Control | 201/171 | 181/169 | 124/166 | |
| Model 1 a | 1 | 0.87 (0.64–1.18) | 0.60 (0.43–0.84) | 0.005 |
| Model 2 b | 1 | 0.57 (0.34–0.95) | 0.44 (0.26–0.77) | 0.003 |
| Factor 4 | ≤−0.40 | −0.40–0.01 | >0.01 | |
| Case/Control | 246/178 | 114/161 | 146/167 | |
| Model 1 a | 1 | 0.50 (0.36–0.69) | 0.60 (0.44–0.82) | 0.014 |
| Model 2 b | 1 | 0.37 (0.21–0.63) | 0.41 (0.23–0.74) | 0.018 |
| Factor 5 | ≤−0.35 | −0.35–0.25 | >0.25 | |
| Case/Control | 136/171 | 192/167 | 178/168 | |
| Model 1 a | 1 | 1.48 (1.08–2.02) | 1.37 (0.99–1.89) | 0.062 |
| Model 2 b | 1 | 1.33 (0.79–2.25) | 0.93 (0.55–1.56) | 0.709 |
| Factor 6 | ≤−0.38 | −0.38–−0.08 | >−0.08 | |
| Case/Control | 64/171 | 161/167 | 281/168 | |
| Model 1 a | 1 | 2.92 (1.96–4.37) | 5.42 (3.61–8.13) | <0.001 |
| Model 2 b | 1 | 2.99 (1.63–5.47) | 3.75 (1.89–7.44) | 0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, W.; He, Y.; Wang, C.; Chen, F.; Jiang, B.; Li, W. Adherence to Healthy Dietary Patterns and Glioma: A Matched Case-Control Study. Nutrients 2023, 15, 4886. https://doi.org/10.3390/nu15234886
Zhang W, He Y, Wang C, Chen F, Jiang B, Li W. Adherence to Healthy Dietary Patterns and Glioma: A Matched Case-Control Study. Nutrients. 2023; 15(23):4886. https://doi.org/10.3390/nu15234886
Chicago/Turabian StyleZhang, Weichunbai, Yongqi He, Ce Wang, Feng Chen, Bo Jiang, and Wenbin Li. 2023. "Adherence to Healthy Dietary Patterns and Glioma: A Matched Case-Control Study" Nutrients 15, no. 23: 4886. https://doi.org/10.3390/nu15234886
APA StyleZhang, W., He, Y., Wang, C., Chen, F., Jiang, B., & Li, W. (2023). Adherence to Healthy Dietary Patterns and Glioma: A Matched Case-Control Study. Nutrients, 15(23), 4886. https://doi.org/10.3390/nu15234886






