Lifestyle Interventions to Prevent Type 2 Diabetes in Women with a History of Gestational Diabetes: A Systematic Review and Meta-Analysis through the Lens of Health Equity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy and Selection Criteria
2.2. Quality Assessment
2.3. Data Extraction
2.4. Data Synthesis and Analysis
3. Results
3.1. Risk of Bias and Quality Assessment
3.2. Study Characteristics of PROGRESS Framework
3.2.1. Place of Residence (P)
3.2.2. Race/Ethnicity/Culture/Language (R)
3.2.3. Occupation (O)
3.2.4. Gender (G)
3.2.5. Religion (R)
3.2.6. Education (E)
3.2.7. Socioeconomic Status (S): Income
3.2.8. Social Capital (S)
| Study | Sample | Country Classification | Intervention Type (Diet or Physical Activity) | Country | Residence (Urban vs. Rural) | Ethnicity | Occupation | Educational Status | Income Level c |
|---|---|---|---|---|---|---|---|---|---|
| Brazeau 2014 [57] | 36 | HIC | Combined | Canada | NR | NR; not specified | NR | NR | NR |
| Combined | |||||||||
| Brokaw 2018 [42] | 283 | HIC | Combined | USA | Urban and rural | NR; not specified | NR | NR | NR |
| Cheung 2011 [58] | 43 | HIC | Physical activity | Australia | NR | NR; not specified | NR | NR | NR |
| Cheung 2019 [32]/2022 [59] | 60 | HIC | Combined | Australia | NR | Mixed: South Asian, Southeast Asian, Australian, others | NR | NR | NR |
| Ferrara 2011 [48] | 197 | HIC | Combined | USA | NR | Mixed: Non-Hispanic white, Black/African American, Asian or Pacific Islander, Hispanic origin, others | ≥50% employed | ≥50% tertiary | NR |
| Ferrara 2016 [60] | 2280 | HIC | Combined | USA | NR | Mixed: Asian, Non-Hispanic white, Hispanic, African American, multiracial, Pacific Islander, others | NR | NR | NR |
| Geng 2014 [49] | 100 | MIC | Combined | China | NR | East Asian (Chinese) b | NR | NR | NR |
| Guo 2021 [43]; Chen 2022 [45]; Zhong 2023 [46] | 320 | MIC | Combined | China | Rural | East Asian (Chinese)—Han and others | ≥50% employed | <50% tertiary | High |
| Holmes 2018 [50] | 60 | HIC | Combined | UK (Ireland) | NR | White | ≥50% employed | ≥50% tertiary | NR |
| Hu 2012 [35]; Liu 2008 [61] | 1180 | MIC | Combined | China | Urban | East Asian (Chinese) b | NR | ≥50% tertiary | Low |
| Kapoor 2019 [62] | 56 | MIC | Combined | India | NR | South Asian: Indian | <50% employed | ≥50% tertiary | NR |
| Kim 2012 [63] | 49 | HIC | Physical activity | USA | NR | Mixed: Non-Hispanic white, Asian (South and East), African American, others | NR | ≥50% tertiary | High |
| Kim 2021 [64] | 119 | HIC | Combined | South Korea | NR | East Asian (Korean) b | NR | NR | NR |
| Lee 2022 [65] | 298 | MIC | Combined | Malaysia | Urban and semi-urban | Mixed (mixed Asians): Malays, Chinese, Indians, others | NR | <50% tertiary | NR |
| Li 2021 [40] | 404 | MIC | Combined | China | Rural | East Asian (Chinese): Han and others | <50% employed | <50% tertiary | High |
| Lim 2017 [39] | 33 | HIC | Combined | Australia | Urban | Mixed: Australia-born and born outside Australia | NR | ≥50% tertiary | High |
| Lim 2021 [41] | 200 | HIC | Combined | Singapore | Urban | Mixed (Mixed Asians): Malays, Chinese, Indians, others | ≥50% employed | ≥50% tertiary | NR |
| Man 2021 [47]; Aroda 2015 [66]; Ratner 2008 [67] | 350 | HIC | Combined | USA | NR | Mixed: White, African American, Hispanic, others | NR | NR | NR |
| McCurley 2017 [30] | 24 | HIC | Combined | USA | NR | Mexican Americans | NR | <50% tertiary | Low |
| McIntyre 2012 [68] | 28 | HIC | Combined | Australia | NR | NR; not specified | NR | ≥50% tertiary | NR |
| McManus 2018 [51]; Barton 2019 [69] | 178 | HIC | Combined | Canada | NR | Mixed: White and others | NR | ≥50% tertiary | High |
| Nicholson 2016 [70] | 23 | HIC | Combined | USA | NR | Mixed: White, African American, Asian, Hispanic, others | ≥50% employed | ≥50% tertiary | NR |
| Nicklas 2014 [52] | 75 | HIC | Combined | USA | NR | Mixed: White, African American, Asian, Hispanic or Latina | NR | ≥50% tertiary | High |
| O’Dea 2015 [54] | 50 | HIC | Combined | Ireland | NR | White b | NR | NR | NR |
| O’Reilly 2016/2019 [33,71] | 573 | HIC | Combined | Australia | NR | Mixed: Asian, Australian and New Zealander, Aboriginal, and Torres Strait Islander | <50% employed | ≥50% tertiary | High |
| Peacock 2015 [53] | 31 | HIC | Combined | Australia | NR | White | NR | NR | NR |
| Perez-Ferre 2015 [72] | 237 | HIC | Combined | Spain | NR | Mixed: White and Hispanic | NR | NR | NR |
| Philis-Tsimikas 2014 [31] | 84 | HIC | Combined | USA | NR | Mexican Americans | <50% employed | <50% tertiary | Low |
| Potzel 2022 [56] | 66 | HIC | Combined | Germany | NR | White b | NR | ≥50% tertiary | NR |
| Rautio 2014 [73] | 115 | HIC | Combined | Finland | NR | White b | NR | NR | NR |
| Reinhardt 2012 [44] | 38 | HIC | Combined | Australia | Rural | NR; not specified | NR | NR | NR |
| Rollo 2020 [29] | 29 | HIC | Combined | Australia | NR | White | NR | ≥50% tertiary | High |
| Shek 2014 [74] | 450 | MIC | Combined | China | NR | East Asian (Chinese) | NR | NR | NR |
| Sheng Yu 2012 [75] | 130 | MIC | Combined | China | NR | East Asian (Chinese) b | NR | ≥50% tertiary | Low |
| Shyam 2013/15 [76,77] | 77 | MIC | Combined | Malaysia | NR | Mixed (Mixed Asians): Malays, Chinese, Indians, others | NR | ≥50% tertiary | Low |
| Smith 2014 [55] | 59 | HIC | Combined | Australia | NR | Mixed: Australian, Asian | NR | NR | NR |
| Tandon 2022 [38] | 1612 | MIC | Combined | Bangladesh India Sri Lanka | Urban | South Asian: Bengali Indian, Singhalese | <50% employed | ≥50% tertiary | NR |
| Wein 1999 [78] | 200 | HIC | Diet only | Australia | NR | Mixed: Australian and New Zealander, Mediterranean and Middle Eastern, Northern European, Southeast Asian, Indian subcontinental | NR | NR | NR |
| Yu Xiao 2012 [79] | 126 | MIC | Combined | China | NR | East Asian (Chinese) b | NR | NR | NR |
| Zilberman-Kravits 2018 [80] | 180 | HIC | Combined | Israel | NR | Middle Eastern (Jewish and Bedouins) | NR | <50% tertiary | NR |
| PROGRESS Characteristics | Number Studies | Number of Participants |
|---|---|---|
| Place of residence country based on economy (World Bank Classification) | ||
| High-income country | 29 | 5700 |
| Upper/middle-income country | 9 | 3085 |
| Lower/middle-income country | 2 | 1668 |
| Low-income country | 0 | 0 |
| Continent | ||
| Asia | 14 | 5252 |
| Australia | 10 | 1094 |
| North America | 11 | 3579 |
| Europe | 5 | 528 |
| South America | 0 | 0 |
| Africa | 0 | 0 |
| Asia (n = 14) | ||
| China | 7 | 2710 |
| Malaysia | 2 | 375 |
| Singapore | 1 | 200 |
| India, Sri Lanka, and Bangladesh | 1 | 1612 |
| South Korea | 1 | 119 |
| India | 1 | 56 |
| Israel (Near East) | 1 | 180 |
| North America (n = 11) | ||
| USA | 9 | 3365 |
| Canada | 2 | 214 |
| Europe (n = 4) | ||
| Ireland (UK) | 2 | 110 |
| Spain | 1 | 237 |
| Finland | 1 | 115 |
| Germany | 1 | 66 |
| Urban vs. Rural | ||
| Urban | 4 | 3025 |
| Urban and semi-urban | 1 | 298 |
| Urban and Rural | 1 | 283 |
| Rural | 3 | 762 |
| NR | 31 | 6085 |
| Ethnicity a | ||
| Mixed b | 16 | 4889 |
| East Asian | 8 | 2829 |
| White | 6 | 351 |
| South Asian | 2 | 1668 |
| Mexican Americans | 2 | 108 |
| Middle Eastern | 1 | 180 |
| Not specified c | 5 | 428 |
| Occupation/employment | ||
| Reported | 10 | 3529 |
| Not reported | 30 | 6924 |
| Occupation reported (n = 10) | ||
| Mostly unemployed | 5 | 2729 |
| Mostly employed | 5 | 800 |
| Gender: women | 40 | 10,457 |
| Religion: not reported | 40 | 10,457 |
| Educational status | Number studies | Participants |
| Reported | 23 | 58,876 |
| Not reported | 17 | 4577 |
| Educational status reported (n = 23) | Number studies | Participants |
| Mostly with tertiary education | 17 | 4566 |
| Mostly without tertiary education | 6 | 1310 |
| Socioeconomic status/income (n= 39) | Number studies | Participants |
| Reported | 13 | 3156 |
| Not reported | 27 | 7297 |
| Income reported (n = 13) | Number studies | Participants |
| Above average | 8 | 1661 |
| Below average | 5 | 1495 |
| Social capital not reported | 40 | 10,457 |
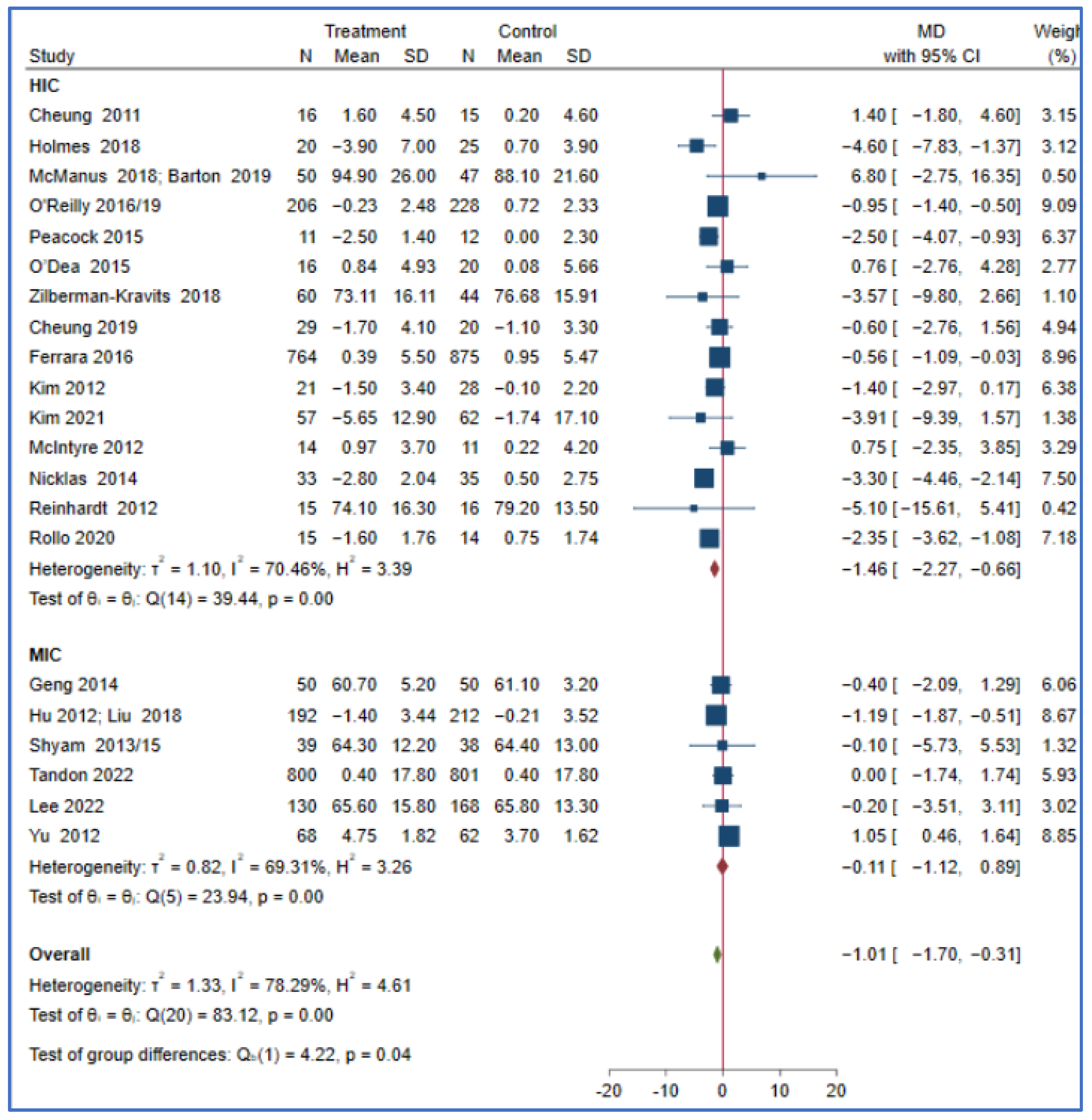
3.3. Meta-Analysis
Intervention Effect of PROGRESS Characteristics
4. Discussion
5. Strengths and Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hill-Briggs, F.; Adler, N.E.; Berkowitz, S.A.; Chin, M.H.; Gary-Webb, T.L.; Navas-Acien, A.; Thornton, P.L.; Haire-Joshu, D. Social determinants of health and diabetes: A scientific review. Diabetes Care 2021, 44, 258–279. [Google Scholar] [CrossRef]
- IDF. Diabetes Atlas, 10th ed.; IDF: Springfield, MO, USA, 2021. [Google Scholar]
- Sommer, I.; Griebler, U.; Mahlknecht, P.; Thaler, K.; Bouskill, K.; Gartlehner, G.; Mendis, S. Socioeconomic inequalities in non-communicable diseases and their risk factors: An overview of systematic reviews. BMC Public Health 2015, 15, 914. [Google Scholar] [CrossRef]
- Agardh, E.; Allebeck, P.; Hallqvist, J.; Moradi, T.; Sidorchuk, A. Type 2 diabetes incidence and socio-economic position: A systematic review and meta-analysis. Int. J. Epidemiol. 2011, 40, 804–818. [Google Scholar] [CrossRef] [PubMed]
- Peykari, N.; Djalalinia, S.; Qorbani, M.; Sobhani, S.; Farzadfar, F.; Larijani, B. Socioeconomic inequalities and diabetes: A systematic review from Iran. J. Diabetes Metab. Disord. 2015, 14, 8. [Google Scholar] [CrossRef] [PubMed]
- Maty, S.C.; Everson-Rose, S.A.; Haan, M.N.; Raghunathan, T.E.; Kaplan, G.A. Education, income, occupation, and the 34-year incidence (1965–99) of type 2 diabetes in the Alameda County Study. Int. J. Epidemiol. 2005, 34, 1274–1281. [Google Scholar] [CrossRef] [PubMed]
- Tung, E.L.; Baig, A.A.; Huang, E.S.; Laiteerapong, N.; Chua, K.-P. Racial and ethnic disparities in diabetes screening between Asian Americans and other adults: BRFSS 2012–2014. J. Gen. Intern. Med. 2017, 32, 423–429. [Google Scholar] [CrossRef]
- Wang, H.; Li, N.; Chivese, T.; Werfalli, M.; Sun, H.; Yuen, L.; Hoegfeldt, C.A.; Powe, C.C.; Immanuel, J.; Karuranga, S.; et al. IDF diabetes atlas: Estimation of global and regional gestational diabetes mellitus prevalence for 2021 by International Association of Diabetes in Pregnancy Study Group’s Criteria. Diabetes Res. Clin. Pract. 2022, 183, 109050. [Google Scholar] [CrossRef]
- Yuen, L.; Wong, V.W. Gestational diabetes mellitus: Challenges for different ethnic groups. World J. Diabetes 2015, 6, 1024. [Google Scholar] [CrossRef]
- Shah, N.S.; Wang, M.C.; Freaney, P.M.; Perak, A.M.; Carnethon, M.R.; Kandula, N.R.; Gunderson, E.P.; Bullard, K.M.; Grobman, W.A.; O’Brien, M.J.; et al. Trends in gestational diabetes at first live birth by race and ethnicity in the US, 2011–2019. JAMA 2021, 326, 660–669. [Google Scholar] [CrossRef]
- Vounzoulaki, E.; Khunti, K.; Abner, S.C.; Tan, B.K.; Davies, M.J.; Gillies, C.L. Progression to type 2 diabetes in women with a known history of gestational diabetes: Systematic review and meta-analysis. BMJ 2020, 369, m1361. [Google Scholar] [CrossRef]
- CDC. NCHHSTP. social determinants of health. Retrieved March 2014, 8, 2016. [Google Scholar]
- Currie, C.; Zanotti, C.; Morgan, A.; Currie, D.; De Looze, M.; Roberts, C.; Samdal, O.; Smith, O.R.F.; Barnekow, V. Social determinants of health and well-being among young people. Health Behav. Sch.-Aged Child. (HBSC) Study Int. Rep. 2009, 2010, 271. [Google Scholar]
- Braveman, P.; Gottlieb, L. The social determinants of health: It’s time to consider the causes of the causes. Public Health Rep. 2014, 129 (Suppl. 2), 19–31. [Google Scholar] [CrossRef] [PubMed]
- Girgis, C.M.; Gunton, J.E.; Cheung, N.W. The influence of ethnicity on the development of type 2 diabetes mellitus in women with gestational diabetes: A prospective study and review of the literature. Int. Sch. Res. Not. 2012, 2012, 341638. [Google Scholar] [CrossRef]
- Marmot, M.; Friel, S.; Bell, R.; Houweling, T.A.; Taylor, S.; on behalf of the Commission on Social Determinants of Health. Closing the gap in a generation: Health equity through action on the social determinants of health. Lancet 2008, 372, 1661–1669. [Google Scholar] [CrossRef] [PubMed]
- Lorenc, T.; Petticrew, M.; Welch, V.; Tugwell, P. What types of interventions generate inequalities? Evidence from systematic reviews. J. Epidemiol. Community Health 2013, 67, 190–193. [Google Scholar] [CrossRef] [PubMed]
- Chapman, A.R.; Carbonetti, B. Human rights protections for vulnerable and disadvantaged groups: The contributions of the UN Committee on Economic, Social and Cultural Rights. Hum. Rights Q. 2011, 33, 682. [Google Scholar] [CrossRef]
- Hart, J.T. The inverse care law. Lancet 1971, 297, 405–412. [Google Scholar] [CrossRef]
- Gordon, D.; Shaw, M. Inequalities in Health: The Evidence Presented to the Independent Inquiry into Inequalities in Health, Chaired by Sir Donald Acheson; Policy Press: Bristol, UK, 1999. [Google Scholar]
- Chatzi, G.; Whittaker, W.; Chandola, T.; Mason, T.; Soiland-Reyes, C.; Sutton, M.; Bower, P. Diabetes Prevention Programme and socioeconomic inequalities in Type 2 Diabetes in England: Georgia Chatzi. Eur. J. Public Health 2022, 32 (Suppl. 3), ckac129–ckac159. [Google Scholar] [CrossRef]
- Welch, V.A.; Petkovic, J.; Jull, J.; Hartling, L.; Klassen, T.; Kristjansson, E.; Pardo, J.P.; Petticrew, M.; Stott, D.J.; Thomson, D.; et al. Equity and specific populations. In Cochrane Handbook for Systematic Reviews of Interventions; John Wiley and Sons: Hoboken, NJ, USA, 2019; pp. 433–449. [Google Scholar]
- O’Neill, J.; Tabish, H.; Welch, V.; Petticrew, M.; Pottie, K.; Clarke, M.; Evans, T.; Pardo, J.P.; Waters, E.; White, H.; et al. Applying an equity lens to interventions: Using PROGRESS ensures consideration of socially stratifying factors to illuminate inequities in health. J. Clin. Epidemiol. 2014, 67, 56–64. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Syst. Rev. 2021, 10, 89. [Google Scholar] [CrossRef]
- Sterne, J.A.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef] [PubMed]
- Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ott. Ott. Hosp. Res. Inst. 2011, 2, 1–12. [Google Scholar]
- Fantom, N.J.; Serajuddin, U. The World Bank’s Classification of Countries by Income; World Bank Policy Research Working Paper; World Bank Group: Washington, DC, USA, 2016; p. 7528. [Google Scholar]
- Rollo, M.E.; Baldwin, J.N.; Hutchesson, M.; Aguiar, E.J.; Wynne, K.; Young, A.; Callister, R.; Haslam, R.; Clare, E.; Collins, C.E. The feasibility and preliminary efficacy of an eHealth lifestyle program in women with recent gestational diabetes mellitus: A pilot study. Int. J. Environ. Res. Public Health 2020, 17, 7115. [Google Scholar] [CrossRef] [PubMed]
- McCurley, J.L.; Fortmann, A.L.; Gutierrez, A.P.; Gonzalez, P.; Euyoque, J.; Clark, T.; Preciado, J.; Ahmad, A.; Philis-Tsimikas, A.; Gallo, L.C. Pilot test of a culturally appropriate diabetes prevention intervention for at-risk Latina women. Diabetes Educ. 2017, 43, 631–640. [Google Scholar] [CrossRef]
- Philis-Tsimikas, A.; Fortmann, A.L.; Dharkar-Surber, S.; Euyoque, J.A.; Ruiz, M.; Schultz, J.; Gallo, L.C. Dulce Mothers: An intervention to reduce diabetes and cardiovascular risk in Latinas after gestational diabetes. Transl. Behav. Med. 2014, 4, 18–25. [Google Scholar] [CrossRef]
- Cheung, N.W.; Blumenthal, C.; Smith, B.J.; Hogan, R.; Thiagalingam, A.; Redfern, J.; Barry, T.; Cinnadaio, N.; Chow, C.K. A pilot randomised controlled trial of a text messaging intervention with customisation using linked data from wireless wearable activity monitors to improve risk factors following gestational diabetes. Nutrients 2019, 11, 590. [Google Scholar] [CrossRef]
- O’Reilly, S.L.; Dunbar, J.A.; Versace, V.; Janus, E.; Best, J.D.; Carter, R.; Oats, J.J.N.; Skinner, T.; Ackland, M.; Phillips, P.A.; et al. Mothers after Gestational Diabetes in Australia (MAGDA): A randomised controlled trial of a postnatal diabetes prevention program. PLoS Med. 2016, 13, e1002092. [Google Scholar] [CrossRef]
- Chen, M.; Ukke, G.G.; Moran, L.J.; Sood, S.; Bennett, C.J.; Bahri Khomami, M.; Absetz, P.; Teede, H.; Harrison, C.L.; Lim, S. The effect of lifestyle intervention on diabetes prevention by ethnicity: A systematic review of intervention characteristics using the tidier framework. Nutrients 2021, 13, 4118. [Google Scholar] [CrossRef]
- Hu, G.; Tian, H.; Zhang, F.; Liu, H.; Zhang, C.; Zhang, S.; Wang, L.; Liu, G.; Yu, Z.; Yang, X.; et al. Tianjin Gestational Diabetes Mellitus Prevention Program: Study design, methods, and 1-year interim report on the feasibility of lifestyle intervention program. Diabetes Res. Clin. Pract. 2012, 98, 508–517. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- StataCorp LLC. Stata Statistical Software: Release Vol. 16; StataCorp LLC: College Station, TX, USA, 2019. [Google Scholar]
- Tandon, N.; Gupta, Y.; Kapoor, D.; Lakshmi, J.K.; Praveen, D.; Bhattacharya, A.; Billot, L.; Naheed, A.; de Silva, A.; Gupta, I.; et al. Effects of a Lifestyle Intervention to Prevent Deterioration in Glycemic Status Among South Asian Women With Recent Gestational Diabetes: A Randomized Clinical Trial. JAMA Netw. Open 2022, 5, e220773. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.; Dunbar, J.A.; Versace, V.L.; Janus, E.; Wildey, C.; Skinner, T.; O’Reilly, S. Comparing a telephone-and a group-delivered diabetes prevention program: Characteristics of engaged and non-engaged postpartum mothers with a history of gestational diabetes. Diabetes Res. Clin. Pract. 2017, 126, 254–262. [Google Scholar] [CrossRef]
- Li, M.; Lin, Q.; Shi, J.; Xi, Y.; Xiang, C.; Yong, C.; Guo, J. The Impact of Lifestyle Intervention on Dietary Quality among Rural Women with Previous Gestational Diabetes Mellitus—A Randomized Controlled Study. Nutrients 2021, 13, 2642. [Google Scholar] [CrossRef]
- Lim, K.; Chan, S.-Y.; Lim, S.L.; Tai, B.C.; Tsai, C.; Wong, S.R.; Ang, S.M.; Yew, T.W.; Tai, E.S.; Yong, E.L. A Smartphone App to Restore Optimal Weight (SPAROW) in Women With Recent Gestational Diabetes Mellitus: Randomized Controlled Trial. JMIR Mhealth Uhealth 2021, 9, e22147. [Google Scholar] [CrossRef]
- Brokaw, S.M.; Carpenedo, D.; Campbell, P.; Butcher, M.K.; Helgerson, S.D.; Harwell, T.S. Does a History of Gestational Diabetes Mellitus Affect Key Outcomes in Women Participating in a Diabetes Prevention Program? Matern. Child Health J. 2018, 22, 529–537. [Google Scholar] [CrossRef]
- Guo, J.; Long, Q.; Yang, J.; Lin, Q.; Wiley, J.; Chen, J.-L. The Efficacy of an Intensive Lifestyle Modification Program on Psychosocial Outcomes among Rural Women with Prior Gestational Diabetes Mellitus: Six Months Follow-Up of a Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2021, 18, 1519. [Google Scholar] [CrossRef]
- Reinhardt, J.A.; Van Der Ploeg, H.P.; Grzegrzulka, R.; Timperley, J.G. Implementing lifestyle change through phone-based motivational interviewing in rural-based women with previous gestational diabetes mellitus. Health Promot. J. Aust. 2012, 23, 5–9. [Google Scholar] [CrossRef]
- Chen, Y.; Zhong, Q.; Luo, J.; Tang, Y.; Li, M.; Lin, Q.; Willey, J.A.; Chen, J.L.; Whittemore, R.; Guo, J. The 6-month efficacy of an intensive lifestyle modification program on type 2 diabetes risk among rural women with prior gestational diabetes mellitus: A cluster randomized controlled trial. Prev. Sci. 2022, 23, 1156–1168. [Google Scholar] [CrossRef]
- Zhong, Q.; Chen, Y.; Luo, M.; Lin, Q.; Tan, J.; Xiao, S.; Willey, J.A.; Chen, J.L.; Whittemore, R.; Guo, J. The 18-month efficacy of an Intensive LifeStyle Modification Program (ILSM) to reduce type 2 diabetes risk among rural women: A cluster randomized controlled trial. Glob. Health 2023, 19, 6. [Google Scholar] [CrossRef] [PubMed]
- Man, B.; Schwartz, A.; Pugach, O.; Xia, Y.; Gerber, B. A clinical diabetes risk prediction model for prediabetic women with prior gestational diabetes. PLoS ONE 2021, 16, e0252501. [Google Scholar] [CrossRef]
- Ferrara, A.; Hedderson, M.M.; Albright, C.L.; Ehrlich, S.F.; Quesenberry, C.P., Jr.; Peng, T.; Feng, J.; Ching, J.; Crites, Y. A pregnancy and postpartum lifestyle intervention in women with gestational diabetes mellitus reduces diabetes risk factors: A feasibility randomized control trial. Diabetes Care 2011, 34, 1519–1525. [Google Scholar] [CrossRef] [PubMed]
- Ling, G.; Shufeng, W.; Xiaoming, Z.; Suhua, Y. Discussion on postpartum lifestyle intervention plan for women with gestational diabetes mellitus. Henan J. Surg. 2014, 20, 76–78. [Google Scholar]
- Holmes, V.A.; Draffin, C.R.; Patterson, C.C.; Francis, L.; Irwin, J.; McConnell, M.; Farrell, B.; Brennan, S.F.; McSorley, O.; Wotherspoon, A.C.; et al. Postnatal lifestyle intervention for overweight women with previous gestational diabetes: A randomized controlled trial. J. Clin. Endocrinol. Metab. 2018, 103, 2478–2487. [Google Scholar] [CrossRef] [PubMed]
- McManus, R.; Miller, D.; Mottola, M.; Giroux, I.; Donovan, L. Translating healthy living messages to postpartum women and their partners after gestational diabetes (GDM): Body habitus, A1c, lifestyle habits, and program engagement results from the families Defeating diabetes (FDD) randomized trial. Am. J. Health Promot. 2018, 32, 1438–1446. [Google Scholar] [CrossRef]
- Nicklas, J.M.; Zera, C.A.; England, L.J.; Rosner, B.A.; Horton, E.; Levkoff, S.E.; Seely, E.W. A web-based lifestyle intervention for women with recent gestational diabetes mellitus: A randomized controlled trial. Obstet. Gynecol. 2014, 124, 563. [Google Scholar] [CrossRef]
- Peacock, A.; Bogossian, F.E.; Wilkinson, S.; Gibbons, K.; Kim, C.; McIntyre, H. A randomised controlled trial to delay or prevent type 2 diabetes after gestational diabetes: Walking for exercise and nutrition to prevent diabetes for you. Int. J. Endocrinol. 2015, 2015, 423717. [Google Scholar] [CrossRef]
- O’Dea, A.; Tierney, M.; McGuire, B.E.; Newell, J.; Glynn, L.G.; Gibson, I.; Noctor, E.; Danyliv, A.; Connolly, S.B.; Dunne, F.P. Can the onset of type 2 diabetes be delayed by a group-based lifestyle intervention in women with prediabetes following gestational diabetes mellitus (GDM)? Findings from a randomized control mixed methods trial. J. Diabetes Res. 2015, 2015, 798460. [Google Scholar] [CrossRef]
- Smith, B.J.; Cinnadaio, N.; Cheung, N.W.; Bauman, A.; Tapsell, L.C.; van der Ploeg, H.P. Investigation of a lifestyle change strategy for high-risk women with a history of gestational diabetes. Diabetes Res. Clin. Pract. 2014, 106, e60–e63. [Google Scholar] [CrossRef]
- Potzel, A.L.; Gar, C.; Banning, F.; Sacco, V.; Fritsche, A.; Fritsche, L.; Müssig, K.; Dauben, L.; Seissler, J.; Lechner, A. A novel smartphone app to change risk behaviors of women after gestational diabetes: A randomized controlled trial. PLoS ONE 2022, 17, e0267258. [Google Scholar] [CrossRef]
- Brazeau, A.-S.; Leong, A.; Meltzer, S.J.; Cruz, R.; DaCosta, D.; Hendrickson-Nelson, M.; Joseph, L.; Dasgupta, K.; MoMM study group. Group-based activities with on-site childcare and online support improve glucose tolerance in women within 5 years of gestational diabetes pregnancy. Cardiovasc. Diabetol. 2014, 13, 104. [Google Scholar] [CrossRef] [PubMed]
- Cheung, N.W.; Smith, B.; Van Der Ploeg, H.; Cinnadaio, N.; Bauman, A. A pilot structured behavioural intervention trial to increase physical activity among women with recent gestational diabetes. Diabetes Res. Clin. Pract. 2011, 92, e27–e29. [Google Scholar] [CrossRef] [PubMed]
- Cheung, N.W. Post-pregnancy intervention for the prevention of T2 diabetes. Obstet. Med. 2022, 15 (Suppl. 1), 15–16. [Google Scholar]
- Ferrara, A.; Hedderson, M.M.; Brown, S.D.; Albright, C.L.; Ehrlich, S.F.; Tsai, A.-L.; Caan, B.J.; Sternfeld, B.; Gordon, N.P.; Schmittdiel, J.A.; et al. The comparative effectiveness of diabetes prevention strategies to reduce postpartum weight retention in women with gestational diabetes mellitus: The Gestational Diabetes’ Effects on Moms (GEM) cluster randomized controlled trial. Diabetes Care 2016, 39, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, L.; Zhang, S.; Leng, J.; Li, N.; Li, W.; Wang, J.; Tian, H.; Qi, L.; Yang, X.; et al. One-year weight losses in the Tianjin Gestational Diabetes Mellitus Prevention Programme: A randomized clinical trial. Diabetes Obes. Metab. 2018, 20, 1246–1255. [Google Scholar] [CrossRef]
- Kapoor, D.; Gupta, Y.; Desai, A.; Praveen, D.; Joshi, R.; Rozati, R.; Bhatla, N.; Prabhakaran, D.; Reddy, P.; Patel, A.; et al. Lifestyle intervention programme for Indian women with history of gestational diabetes mellitus. Glob. Health Epidemiol. Genom. 2019, 4, e1. [Google Scholar] [CrossRef]
- Kim, C.; Draska, M.; Hess, M.; Wilson, E.; Richardson, C. A web-based pedometer programme in women with a recent history of gestational diabetes. Diabet. Med. 2012, 29, 278–283. [Google Scholar] [CrossRef]
- Kim, S.-H.; Kim, H.J.; Shin, G. Self-management mobile virtual reality program for women with gestational diabetes. Int. J. Environ. Res. Public Health 2021, 18, 1539. [Google Scholar] [CrossRef]
- Lee, K.W.; Tan, S.F.; Omar, A.; Nasir, N.H.; Ching, S.M.; Mohd Noor, M.K.; Masdor, N.A.; Lee, Y.S.; Noordin, S.; Mohamed, H. Effectiveness of system-based intervention in reducing incidence of type 2 diabetes and to improve the postnatal metabolic profiles in women with gestational diabetes mellitus: A randomized controlled study. Gynecol. Endocrinol. 2022, 38, 55–62. [Google Scholar] [CrossRef]
- Aroda, V.; Christophi, C.; Edelstein, S.; Zhang, P.; Herman, W.; Barrett-Connor, E.; Delahanty, L.M.; Montez, M.G.; Ackermann, R.T.; Zhuo, X.; et al. The effect of lifestyle intervention and metformin on preventing or delaying diabetes among women with and without gestational diabetes: The Diabetes Prevention Program outcomes study 10-year follow-up. J. Clin. Endocrinol. Metab. 2015, 100, 1646–1653. [Google Scholar] [CrossRef] [PubMed]
- Ratner, R.E.; Christophi, C.A.; Metzger, B.E.; Dabelea, D.; Bennett, P.H.; Pi-Sunyer, X.; Fowler, S.; Kahn, S.E.; Diabetes Prevention Program Research Group. Prevention of diabetes in women with a history of gestational diabetes: Effects of metformin and lifestyle interventions. J. Clin. Endocrinol. Metab. 2008, 93, 4774–4779. [Google Scholar] [CrossRef]
- McIntyre, H.D.; Peacock, A.; Miller, Y.D.; Koh, D.; Marshall, A.L. Pilot study of an individualised early postpartum intervention to increase physical activity in women with previous gestational diabetes. Int. J. Endocrinol. 2012, 2012, 892019. [Google Scholar] [CrossRef] [PubMed]
- Barton, K.I.; Donovan, L.; Giroux, I.; Miller, D.; Mottola, M.; McManus, R. Glycated hemoglobin measurements at three, 12 and 24 months postpartum after gestational diabetes. Clin. Investig. Med. 2019, 42, E37. [Google Scholar] [CrossRef]
- Nicholson, W.K.; Beckham, A.J.; Hatley, K.; Diamond, M.; Johnson, L.-S.; Green, S.L.; Tate, D. The Gestational Diabetes Management System (GooDMomS): Development, feasibility and lessons learned from a patient-informed, web-based pregnancy and postpartum lifestyle intervention. BMC Pregnancy Childbirth 2016, 16, 277. [Google Scholar] [CrossRef] [PubMed]
- O’Reilly, S.; Versace, V.; Mohebbi, M.; Lim, S.; Janus, E.; Dunbar, J. The effect of a diabetes prevention program on dietary quality in women with previous gestational diabetes. BMC Women’s Health 2019, 19, 88. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Ferre, N.; Del Valle, L.; Torrejón, M.J.; Barca, I.; Calvo, M.I.; Matía, P.; Rubio, M.A.; Calle-Pascual, A.L. Diabetes mellitus and abnormal glucose tolerance development after gestational diabetes: A three-year, prospective, randomized, clinical-based, Mediterranean lifestyle interventional study with parallel groups. Clin. Nutr. 2015, 34, 579–585. [Google Scholar] [CrossRef]
- Rautio, N.; Jokelainen, J.; Korpi-Hyövälti, E.; Oksa, H.; Saaristo, T.; Peltonen, M.; Moilanen, L.; Vanhala, M.; Uusitupa, M.; Tuomilehto, J.; et al. Lifestyle intervention in prevention of type 2 diabetes in women with a history of gestational diabetes mellitus: One-year results of the FIN-D2D project. J. Women’s Health 2014, 23, 506–512. [Google Scholar] [CrossRef]
- Shek, N.W.M.; Ngai, C.S.W.; Lee, C.P.; Chan, J.Y.C.; Lao, T.T.H. Lifestyle modifications in the development of diabetes mellitus and metabolic syndrome in Chinese women who had gestational diabetes mellitus: A randomized interventional trial. Arch. Gynecol. Obstet. 2014, 289, 319–327. [Google Scholar] [CrossRef]
- Yu, S.; Wen, J.W.; Miao, L.H.; Hua, H.G. Effects of lifestyle intervention on the weight of postpartum women with gestational diabetes mellitus and impaired glucose tolerance. J. Nurs. Adm. 2012, 12, 772. [Google Scholar]
- Shyam, S.; Arshad, F.; Abdul Ghani, R.; Wahab, N.A.; Safii, N.S.; Nisak, M.Y.B.; Chinna, K.; Kamaruddin, N.A. Low glycaemic index diets improve glucose tolerance and body weight in women with previous history of gestational diabetes: A six months randomized trial. Nutr. J. 2013, 12, 68. [Google Scholar] [CrossRef] [PubMed]
- Shyam, S.; Fatimah, A.; Rohana, A.; Norasyikin, A.; Nik Shanita, S.; Chinna, K.; Mohod-Yusof, B.N.; Nor, A.K. Effect of including glycaemic index (GI) nutrition education, within the conventional healthy dietary recommendation framework, on body weight and composition of women with prior gestational diabetes mellitus: Results from a one-year randomised controlled trial. J. Nutr. 2015, 21, 269–283. [Google Scholar]
- Wein, P.; Beischer, N.; Harris, C.; Permezel, M. A trial of simple versus intensified dietary modification for prevention of progression to diabetes mellitus in women with impaired glucose tolerance. Aust. N. Z. J. Obstet. Gynaecol. 1999, 39, 162–166. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Wu, X.; Zhang, Y.; Mao, Y. Effects of lifestyle intervention on insulin resistance and islet B-cell function in postpartum gestational diabetes patients with impaired glucose regulation. Chin. J. Prev. Control. Chronic 2012, 20, 560–562. [Google Scholar]
- Zilberman-Kravits, D.; Meyerstein, N.; Abu-Rabia, Y.; Wiznitzer, A.; Harman-Boehm, I. The impact of a cultural lifestyle intervention on metabolic parameters after gestational diabetes mellitus a randomized controlled trial. Matern. Child Health J. 2018, 22, 803–811. [Google Scholar] [CrossRef]
- Yuen, L.; Saeedi, P.; Riaz, M.; Karuranga, S.; Divakar, H.; Levitt, N.; Yang, X.; Simmons, D. Projections of the prevalence of hyperglycaemia in pregnancy in 2019 and beyond: Results from the International Diabetes Federation Diabetes Atlas. Diabetes Res. Clin. Pract. 2019, 157, 107841. [Google Scholar] [CrossRef]
- Hamman, R.F.; Wing, R.R.; Edelstein, S.L.; Lachin, J.M.; Bray, G.A.; Delahanty, L.; Hoskin, M.; Kriska, A.M.; Mayer-Davis, E.J.; Pi-Sunyer, X.; et al. Effect of weight loss with lifestyle intervention on risk of diabetes. Diabetes Care 2006, 29, 2102–2107. [Google Scholar] [CrossRef]
- Carter-Edwards, L.; Østbye, T.; Bastian, L.A.; Yarnall, K.S.; Krause, K.M.; Simmons, T.-J. Barriers to adopting a healthy lifestyle: Insight from postpartum women. BMC Res. Notes 2009, 2, 161. [Google Scholar] [CrossRef]
- Saligheh, M.; McNamara, B.; Rooney, R. Perceived barriers and enablers of physical activity in postpartum women: A qualitative approach. BMC Pregnancy Childbirth 2016, 16, 131. [Google Scholar] [CrossRef]
- Al-Dhahir, I.; Reijnders, T.; Faber, J.S.; van den Berg-Emons, R.J.; Janssen, V.R.; Kraaijenhagen, R.A.; Valentijn, T.; Visch, V.T.; Chavannes, N.H.; Evers, A.W. The barriers and facilitators of eHealth-based lifestyle intervention programs for people with a low socioeconomic status: Scoping review. J. Med. Internet Res. 2022, 24, e34229. [Google Scholar] [CrossRef]
- Fontil, V.; McDermott, K.; Tieu, L.; Rios, C.; Gibson, E.; Sweet, C.C.; Payne, M.; Lyles, C.R. Adaptation and feasibility study of a digital health program to prevent diabetes among low-income patients: Results from a partnership between a digital health company and an academic research team. J. Diabetes Res. 2016, 2016, 8472391. [Google Scholar] [CrossRef]
- Whitehead, L.; Talevski, J.; Fatehi, F.; Beauchamp, A. Barriers to and Facilitators of Digital Health Among Culturally and Linguistically Diverse Populations: Qualitative Systematic Review. J. Med. Internet Res. 2023, 25, e42719. [Google Scholar] [CrossRef] [PubMed]
- Joiner, K.L.; Nam, S.; Whittemore, R. Lifestyle interventions based on the diabetes prevention program delivered via eHealth: A systematic review and meta-analysis. Prev. Med. 2017, 100, 194–207. [Google Scholar] [CrossRef]
- Dugani, S.B.; Mielke, M.M.; Vella, A. Burden and management of type 2 diabetes in rural United States. Diabetes/Metab. Res. Rev. 2021, 37, e3410. [Google Scholar] [CrossRef]
- AIHW. Diabetes: Australian Facts. Available online: https://www.aihw.gov.au/reports/diabetes/diabetes/contents/how-common-is-diabetes/all-diabetes (accessed on 9 February 2023).
- Harding, C.; Seal, A.; Mills, N. Evaluation of a Lifestyle and Weight Management Program Within Rural General Practice. J. Prim. Care Community Health 2022, 13, 21501319221084166. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Moon, J.H.; Kim, H.J.; Kong, M.H.; Oh, Y.H. Sedentary lifestyle: Overview of updated evidence of potential health risks. Korean J. Fam. Med. 2020, 41, 365. [Google Scholar] [CrossRef] [PubMed]
- Mbuagbaw, L.; Aves, T.; Shea, B.; Jull, J.; Welch, V.; Taljaard, M.; Yoganathan, M.; Greer-Smith, R.; Wells, G.; Tugwell, P. Considerations and guidance in designing equity-relevant clinical trials. Int. J. Equity Health 2017, 16, 93. [Google Scholar] [CrossRef] [PubMed]
- Kauh, T.J.; Minnis, T.A.; Anand, M.; Berry, M.; Gold, R. Building an Equitable Future Through Data Disaggregation. Health Equity 2023, 7, 251–260. [Google Scholar] [CrossRef]
- Yang, J.; Xia, Y.; Sun, Y.; Guo, Y.; Shi, Z.; do Vale Moreira, N.C.; Zuo, H.; Hussain, A. Effect of lifestyle intervention on HbA1c levels in overweight and obese adults with type 2 diabetes across ethnicities: A systematic review and meta-analysis of randomized controlled trials. Diabetes Res. Clin. Pract. 2023, 199, 110662. [Google Scholar] [CrossRef]
- Lynch, E.B.; Mack, L.; Avery, E.; Wang, Y.; Dawar, R.; Richardson, D.; Keim, K.; Ventrelle, J.; Appelhans, B.M.; Tahsin, B.; et al. Randomized trial of a lifestyle intervention for urban low-income African Americans with type 2 diabetes. J. Gen. Intern. Med. 2019, 34, 1174–1183. [Google Scholar] [CrossRef]
- Khare, M.M.; Cursio, J.F.; Locklin, C.A.; Bates, N.J.; Loo, R.K. Lifestyle intervention and cardiovascular disease risk reduction in low-income Hispanic immigrant women participating in the Illinois WISEWOMAN program. J. Community Health 2014, 39, 737–746. [Google Scholar] [CrossRef]
- Burgess, E.; Hassmén, P.; Pumpa, K.L. Determinants of adherence to lifestyle intervention in adults with obesity: A systematic review. Clin. Obes. 2017, 7, 123–135. [Google Scholar] [CrossRef]
- Coupe, N.; Cotterill, S.; Peters, S. Tailoring lifestyle interventions to low socio-economic populations: A qualitative study. BMC Public Health 2018, 18, 967. [Google Scholar]
- Fokas, E. Religion and Human Rights in Greece. In Global Eastern Orthodoxy; Springer: Berlin/Heidelberg, Germany, 2020; pp. 101–124. [Google Scholar]
- Lim, F.K.; Sng, B.B. Social media, religion and shifting boundaries in globalizing China. Glob. Media China 2020, 5, 261–274. [Google Scholar] [CrossRef]
- Bosch-Capblanch, X.; Zuske, M.-K.; Auer, C. Research on subgroups is not research on equity attributes: Evidence from an overview of systematic reviews on vaccination. Int. J. Equity Health 2017, 16, 95. [Google Scholar] [CrossRef] [PubMed]
- Koenig, H.G. Religion, spirituality, and health: The research and clinical implications. Int. Sch. Res. Not. 2012, 2012, 278730. [Google Scholar] [CrossRef]
- Swihart, D.L.; Yarrarapu, S.N.S.; Martin, R.L. Cultural Religious Competence in Clinical Practice; StatPearls Publishing: Treasure Island, FL, USA, 2018. [Google Scholar]
- Rivenbark, J.G.; Ichou, M. Discrimination in healthcare as a barrier to care: Experiences of socially disadvantaged populations in France from a nationally representative survey. BMC Public Health 2020, 20, 31. [Google Scholar] [CrossRef]
- Tan, S.T.; Low, P.T.A.; Howard, N.; Yi, H. Social capital in the prevention and management of non-communicable diseases among migrants and refugees: A systematic review and meta-ethnography. BMJ Glob. Health 2021, 6, e006828. [Google Scholar] [CrossRef]
- Sanjari, M.; Aalaa, M.; Amini, M.R.; Aghaei Meybodi, H.R.; Qorbani, M.; Adibi, H.; Mehrdad, N. Social-capital determinants of the women with diabetes: A population-based study. J. Diabetes Metab. Disord. 2021, 20, 511–521. [Google Scholar] [CrossRef]
- Jiang, H.; Xingling, C.; Yuxiu, H.; Feng, T. Progress and Inspiration of Social Capital in the Prevention and Control of Diabetes. Iran. J. Public Health 2020, 49, 196–197. [Google Scholar] [CrossRef]
- Keyvanara, M.; Afshari, M.; Dezfoulian, E. The relationship between social capital and quality of life among patients referring to diabetes centers in Isfahan, Iran. J. Diabetes Res. 2018, 2018, 9353858. [Google Scholar] [CrossRef]
- Grootaert, C. Measuring Social Capital: An Integrated Questionnaire; World Bank Publications: Washington, DC, USA, 2004. [Google Scholar]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ukke, G.G.; Boyle, J.A.; Reja, A.; Lee, W.K.; Chen, M.; Ko, M.S.M.; Alycia, C.; Kwon, J.; Lim, S. Lifestyle Interventions to Prevent Type 2 Diabetes in Women with a History of Gestational Diabetes: A Systematic Review and Meta-Analysis through the Lens of Health Equity. Nutrients 2023, 15, 4666. https://doi.org/10.3390/nu15214666
Ukke GG, Boyle JA, Reja A, Lee WK, Chen M, Ko MSM, Alycia C, Kwon J, Lim S. Lifestyle Interventions to Prevent Type 2 Diabetes in Women with a History of Gestational Diabetes: A Systematic Review and Meta-Analysis through the Lens of Health Equity. Nutrients. 2023; 15(21):4666. https://doi.org/10.3390/nu15214666
Chicago/Turabian StyleUkke, Gebresilasea Gendisha, Jacqueline A. Boyle, Ahmed Reja, Wai Kit Lee, Mingling Chen, Michelle Shi Min Ko, Chelsea Alycia, Jane Kwon, and Siew Lim. 2023. "Lifestyle Interventions to Prevent Type 2 Diabetes in Women with a History of Gestational Diabetes: A Systematic Review and Meta-Analysis through the Lens of Health Equity" Nutrients 15, no. 21: 4666. https://doi.org/10.3390/nu15214666
APA StyleUkke, G. G., Boyle, J. A., Reja, A., Lee, W. K., Chen, M., Ko, M. S. M., Alycia, C., Kwon, J., & Lim, S. (2023). Lifestyle Interventions to Prevent Type 2 Diabetes in Women with a History of Gestational Diabetes: A Systematic Review and Meta-Analysis through the Lens of Health Equity. Nutrients, 15(21), 4666. https://doi.org/10.3390/nu15214666