Adherence to Data-Driven Dietary Patterns and Lung Cancer Risk: A Systematic Review and Dose–Response Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Systematic Search and Criteria for Selection
2.2. Data Extraction and Quality Assessment
2.3. Statistical Analysis
3. Results
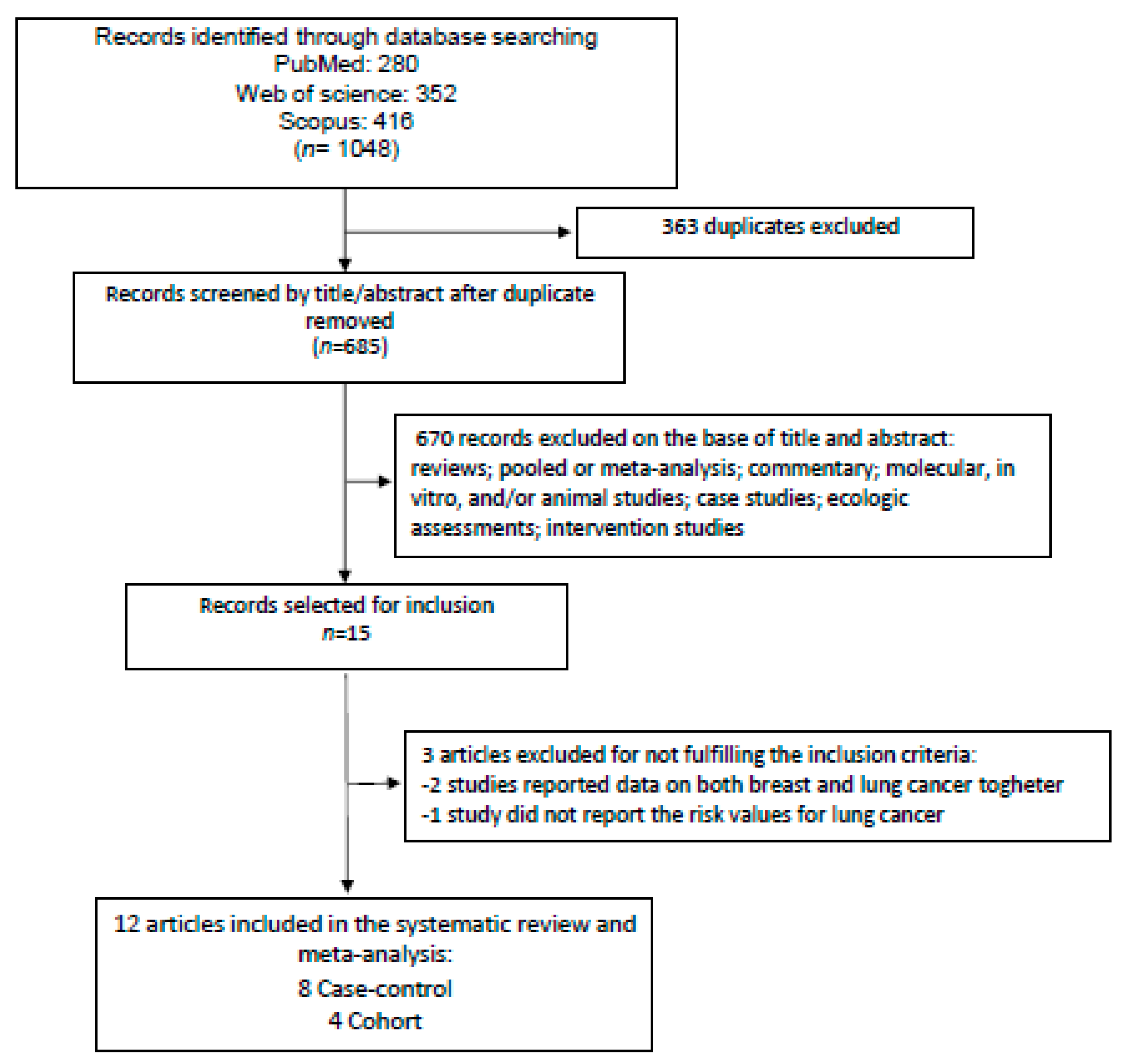
3.1. Study Selection, Characteristics, and Quality Assessment
3.2. Meta-Analysis
3.3. Dose–Response Analysis
3.4. Publication Bias and Sensitivity Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Bade, B.C.; Dela Cruz, C.S. Lung cancer 2020: Epidemiology, etiology, and prevention. Clin. Chest. Med. 2020, 41, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Pyo, J.S.; Kim, N.Y.; Kang, D.W. Impacts of outdoor particulate matter exposure on the incidence of lung cancer and mortality. Medicina 2022, 58, 1159. [Google Scholar] [CrossRef] [PubMed]
- Hamra, G.B.; Laden, F.; Cohen, A.J.; Raaschou-Nielsen, O.; Brauer, M.; Loomis, D. Lung cancer and exposure to nitrogen dioxide and traffic: A systematic review and meta-analysis. Environ. Health Perspect. 2015, 123, 1107–1112. [Google Scholar] [CrossRef] [PubMed]
- Brey, C.; Gouveia, F.T.; Silva, B.S.; Sarquis, L.M.M.; Miranda, F.M.D.; Consonni, D. Lung cancer related to occupational exposure: An integrative review. Rev. Gaucha Enferm. 2020, 41, e20190378. [Google Scholar] [CrossRef]
- Martin-Gisbert, L.; Ruano-Ravina, A.; Varela-Lema, L.; Penabad, M.; Giraldo-Osorio, A.; Candal-Pedreira, C.; Rey-Brandariz, J.; Mourino, N.; Pérez-Ríos, M. Lung cancer mortality attributable to residential radon: A systematic scoping review. J. Expo. Sci. Environ. Epidemiol. 2023, 33, 368–376. [Google Scholar] [CrossRef]
- Fakhri, G.; Al Assaad, M.; Tfayli, A. Association of various dietary habits and risk of lung cancer: An updated comprehensive literature review. Tumori 2020, 106, 445–456. [Google Scholar] [CrossRef]
- Yang, T.; Wang, C.; Li, S.; Guo, X.F.; Li, D. Dietary intakes of fruits and vegetables and lung cancer risk in participants with different smoking status: A meta-analysis of prospective cohort studies. Asia Pac. J. Clin. Nutr. 2019, 28, 770–782. [Google Scholar] [CrossRef]
- Cao, C.; Gan, X.; He, Y.; Nong, S.; Su, Y.; Liu, Z.; Zhang, Y.; Hu, X.; Peng, X. Association between nut consumption and cancer risk: A meta-analysis. Nutr. Cancer 2023, 75, 82–94. [Google Scholar] [CrossRef]
- Wang, J.; Gao, J.; Xu, H.L.; Qian, Y.; Xie, L.; Yu, H.; Qian, B.Y. Citrus fruit intake and lung cancer risk: A meta-analysis of observational studies. Pharmacol. Res. 2021, 166, 105430. [Google Scholar] [CrossRef]
- Lv, D.; Wang, R.; Chen, M.; Li, Y.; Cao, C. Fish intake, dietary polyunsaturated fatty acids, and lung cancer: Systematic review and dose-response meta-analysis of 1.7 million men and women. Nutr. Cancer 2022, 74, 1976–1985. [Google Scholar] [CrossRef] [PubMed]
- Farvid, M.S.; Sidahmed, E.; Spence, N.D.; Mante Angua, K.; Rosner, B.A.; Barnett, J.B. Consumption of red meat and processed meat and cancer incidence: A systematic review and meta-analysis of prospective studies. Eur. J. Epidemiol. 2021, 36, 937–951. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wang, X.; Yao, Q.; Qin, L.; Xu, C. Dairy product, calcium intake and lung cancer risk: A systematic review with meta-analysis. Sci. Rep. 2016, 6, 20624. [Google Scholar] [CrossRef] [PubMed]
- Schulz, C.A.; Oluwagbemigun, K.; Nöthlings, U. Advances in dietary pattern analysis in nutritional epidemiology. Eur. J. Nutr. 2021, 60, 4115–4130. [Google Scholar] [CrossRef]
- Du, H.; Zhang, T.; Lu, X.; Chen, M.; Li, X.; Li, Z. Glycemic index, glycemic load, and lung cancer risk: A meta-analysis of cohort and case-control studies. PLoS ONE 2022, 17, e0273943. [Google Scholar] [CrossRef]
- Bahrami, A.; Khalesi, S.; Makiabadi, E.; Alibeyk, S.; Hajigholam-Saryazdi, M.; Hejazi, E. Adherence to the Mediterranean diet and the risk of lung cancer: A systematic review and dose-response meta-analysis of observational studies. Nutr. Rev. 2022, 80, 1118–1128. [Google Scholar] [CrossRef]
- Sun, Y.; Li, Z.; Li, J.; Li, Z.; Han, J. A Healthy Dietary Pattern Reduces Lung Cancer Risk: A Systematic Review and Meta-Analysis. Nutrients. 2016, 8, 134. [Google Scholar] [CrossRef]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Non-Randomised Studies in Meta-Analyses. 2015. Available online: http://www.ohri.ca/programs/clinical-epidemiology/oxford.asp (accessed on 1 March 2023).
- Willemsen, R.F.; McNeil, J.; Heer, E.; Johnson, S.T.; Friedenreich, C.M.; Brenner, D.R. Dietary patterns with combined and site-specific cancer incidence in Alberta’s Tomorrow Project cohort. Eur. J. Clin. Nutr. 2021, 76, 360–372. [Google Scholar] [CrossRef]
- Wei, X.; Zhu, C.; Ji, M.; Fan, J.; Xie, J.; Huang, Y.; Jiang, X.; Xu, J.; Yin, R.; Du, L.; et al. Diet and risk of incident lung cancer: A large prospective cohort study in UK biobank. Am. J. Clin. Nutr. 2021, 114, 2043–2051. [Google Scholar] [CrossRef]
- Hawrysz, I.; Wadolowska, L.; Slowinska, M.A.; Czerwinska, A.; Golota, J.J. Adherence to Prudent and Mediterranean Dietary Patterns Is Inversely Associated with Lung Cancer in Moderate but Not Heavy Male Polish Smokers: A Case-Control Study. Nutrients 2020, 12, 3788. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Xiao, R.D.; Xiong, W.M.; Xu, Q.P.; Li, X.; Liu, Z.Q.; He, B.C.; Hu, Z.J.; Cai, L. Dietary patterns, BCMO1 polymorphisms, and primary lung cancer risk in a Han Chinese population: A case-control study in Southeast China. BMC Cancer 2018, 18, 445. [Google Scholar] [CrossRef] [PubMed]
- Tu, H.; Heymach, J.V.; Wen, C.P.; Ye, Y.; Pierzynski, J.A.; Roth, J.A.; Wu, X. Different dietary patterns and reduction of lung cancer risk: A large case-control study in the U.S. Sci. Rep. 2016, 6, 26760. [Google Scholar] [CrossRef] [PubMed]
- Gnagnarella, P.; Maisonneuve, P.; Bellomi, M.; Rampinelli, C.; Bertolotti, R.; Spaggiari, L.; Palli, D.; Veronesi, G. Nutrient intake and nutrient patterns and risk of lung cancer among heavy smokers: Results from the COSMOS screening study with annual low-dose CT. Eur. J. Epidemiol. 2013, 28, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Gorlova, O.Y.; Weng, S.F.; Hernandez, L.; Spitz, M.R.; Forman, M.R. Dietary patterns affect lung cancer risk in never smokers. Nutr. Cancer 2011, 63, 842–849. [Google Scholar] [CrossRef] [PubMed]
- De Stefani, E.; Ronco, A.L.; Deneo-Pellegrini, H.; Correa, P.; Boffetta, P.; Acosta, G.; Mendilaharsu, M. Dietary patterns and risk of adenocarcinoma of the lung in males: A factor analysis in Uruguay. Nutr. Cancer 2011, 63, 699–706. [Google Scholar] [CrossRef] [PubMed]
- De Stefani, E.; Deneo-Pellegrini, H.; Boffetta, P.; Ronco, A.L.; Aune, D.; Acosta, G.; Mendilaharsu, M.; Brennan, P.; Ferro, G. Dietary patterns and risk of cancer: A factor analysis in Uruguay. Int. J. Cancer 2009, 124, 1391–1397. [Google Scholar] [CrossRef]
- De Stefani, E.; Boffetta, P.; Ronco, A.L.; Deneo-Pellegrini, H.; Acosta, G.; Gutiérrez, L.P.; Mendilaharsu, M. Nutrient patterns and risk of lung cancer: A factor analysis in Uruguayan men. Lung Cancer 2008, 61, 283–291. [Google Scholar] [CrossRef]
- Balder, H.F.; Goldbohm, R.A.; van den Brandt, P.A. Dietary patterns associated with male lung cancer risk in the The Netherlands Cohort Study. Cancer Epidemiol. Biomarkers Prev. 2005, 14, 483–490. [Google Scholar] [CrossRef]
- Tsai, Y.Y.; McGlynn, K.A.; Hu, Y.; Cassidy, A.B.; Arnold, J.; Engstrom, P.F.; Buetow, K.H. Genetic susceptibility and dietary patterns in lung cancer. Lung Cancer 2003, 41, 269–281. [Google Scholar] [CrossRef]
- Shim, S.R.; Lee, J. Dose-response meta-analysis: Application and practice using the R software. Epidemiol. Health 2019, 41, e2019006. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2014; Available online: http://www.R-project.org/ (accessed on 1 March 2023).
- Higgins, J.P.; Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Begg, C.B.; Mazumdar, M. Operating characteristics of a rank correlation test for publication bias. Biometrics 1994, 50, 1088–1101. [Google Scholar] [CrossRef]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Krusinska, B.; Hawrysz, I.; Wadolowska, L.; Slowinska, M.A.; Biernacki, M.; Czerwinska, A.; Golota, J.J. Associations of Mediterranean Diet and a Posteriori Derived Dietary Patterns with Breast and Lung Cancer Risk: A Case-Control Study. Nutrients 2018, 10, 470. [Google Scholar] [CrossRef]
- Krusińska, B.; Hawrysz, I.; Słowińska, M.A.; Wądołowska, L.; Biernacki, M.; Czerwińska, A.; Gołota, J.J. Dietary patterns and breast or lung cancer risk: A pooled analysis of 2 case-control studies in north-eastern Poland. Adv. Clin. Exp. Med. 2017, 26, 1367–1375. [Google Scholar] [CrossRef]
- De Stefani, E.; Boffetta, P.; Correa, P.; Deneo-Pellegrini, H.; Ronco, A.L.; Acosta, G.; Mendilaharsu, M. Dietary patterns and risk of cancers of the upper aerodigestive tract: A factor analysis in Uruguay. Nutr. Cancer 2013, 65, 384–389. [Google Scholar] [CrossRef]
- Jayedi, A.; Soltani, S.; Abdolshahi, A.; Shab-Bidar, S. Healthy and unhealthy dietary patterns and the risk of chronic disease: An umbrella review of meta-analyses of prospective cohort studies. Br. J. Nutr. 2020, 124, 1133–1144. [Google Scholar] [CrossRef]
- Dianatinasab, M.; Forozani, E.; Akbari, A.; Azmi, N.; Bastam, D.; Fararouei, M.; Wesselius, A.; Zeegres, M.P. Dietary patterns and risk of bladder cancer: A systematic review and meta-analysis. BMC Public Health 2022, 22, 73. [Google Scholar] [CrossRef]
- Garcia-Larsen, V.; Morton, V.; Norat, T.; Moreira, A.; Potts, J.F.; Reeves, T.; Bakolis, I. Dietary patterns derived from principal component analysis (PCA) and risk of colorectal cancer: A systematic review and meta-analysis. Eur. J. Clin. Nutr. 2019, 73, 366–386. [Google Scholar] [CrossRef] [PubMed]
- Bertuccio, P.; Rosato, V.; Andreano, A.; Ferraroni, M.; Decarli, A.; Edefonti, V.; La Vecchia, C. Dietary patterns and gastric cancer risk: A systematic review and meta-analysis. Ann. Oncol. 2013, 24, 1450–1458. [Google Scholar] [CrossRef] [PubMed]
- Fabiani, R.; Minelli, L.; Bertarelli, G.; Bacci, S. A Western Dietary Pattern Increases Prostate Cancer Risk: A Systematic Review and Meta-Analysis. Nutrients 2016, 8, 626. [Google Scholar] [CrossRef] [PubMed]
- Dianatinasab, M.; Rezaian, M.; Haghighat-Nezad, E.; Bagheri-Hosseinabadi, Z.; Amanat, S.; Rezaeian, S.; Masoudi, A.; Ghiasvand, R. Dietary Patterns and Risk of Invasive Ductal and Lobular Breast Carcinomas: A Systematic Review and Meta-analysis. Clin. Breast Cancer 2020, 20, e516–e528. [Google Scholar] [CrossRef]
- Shin, S.; Fu, J.; Shin, W.K.; Huang, D.; Min, S.; Kang, D. Association of food groups and dietary pattern with breast cancer risk: A systematic review and meta-analysis. Clin. Nutr. 2023, 42, 282–297. [Google Scholar] [CrossRef]
- Zheng, J.; Guinter, M.A.; Merchant, A.T.; Wirth, M.D.; Zhang, J.; Stolzenberg-Solomon, R.Z.; Steck, S.E. Dietary patterns and risk of pancreatic cancer: A systematic review. Nutr. Rev. 2017, 75, 883–908. [Google Scholar] [CrossRef]
- Bouvard, V.; Loomis, D.; Guyton, K.Z.; Grosse, Y.; Ghissassi, F.E.; Benbrahim-Tallaa, L.; Guha, N.; Mattock, H.; Straif, K. International Agency for Research on Cancer Monograph Working Group. Carcinogenicity of consumption of red and processed meat. Lancet Oncol. 2015, 16, 1599–1600. [Google Scholar] [CrossRef]
- Gnagnarella, P.; Caini, S.; Maisonneuve, P.; Gandini, S. Carcinogenicity of High Consumption of Meat and Lung Cancer Risk Among Non-Smokers: A Comprehensive Meta-Analysis. Nutr. Cancer 2018, 70, 1–13. [Google Scholar] [CrossRef]
- Tappel, A. Heme of consumed red meat can act as a catalyst of oxidative damage and could initiate colon, breast and prostate cancers, heart disease and other diseases. Med. Hypotheses 2007, 68, 562–564. [Google Scholar] [CrossRef]
- Tao, J.; Jatoi, A.; Crawford, J.; Lam, W.W.T.; Ho, J.C.; Wang, X.; Pang, H. Role of dietary carbohydrates on risk of lung cancer. Lung Cancer 2021, 155, 87–93. [Google Scholar] [CrossRef]
- Kubczak, M.; Szustka, A.; Rogalińska, M. Molecular Targets of Natural Compounds with Anti-Cancer Properties. Int. J. Mol. Sci. 2021, 22, 13659. [Google Scholar] [CrossRef] [PubMed]
- Amararathna, M.; Johnston, M.R.; Rupasinghe, H.P. Plant Polyphenols as Chemopreventive Agents for Lung Cancer. Int. J. Mol. Sci. 2016, 17, 1352. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Kase, B.; Zheng, J.; Steck, S.E. Dietary Patterns and Risk of Lung Cancer: A Systematic Review and Meta-Analyses of Observational Studies. Curr. Nutr. Rep. 2023, 12, 338–357. [Google Scholar] [CrossRef] [PubMed]
- Alsharairi, N.A. Dietary Antioxidants and Lung Cancer Risk in Smokers and Non-Smokers. Healthcare 2022, 10, 2501. [Google Scholar] [CrossRef] [PubMed]
- Kuśnierczyk, P. Genetic differences between smokers and never-smokers with lung cancer. Front. Immunol. 2023, 14, 1063716. [Google Scholar] [CrossRef]
- Hu, F.B. Dietary pattern analysis: A new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef]



| Author, Year Location | Study Design, Name, and Population Case/Control Follow-Up Incident Cases Age | Dietary Pattern Assessment and Identification Method | Dietary Pattern Type and Characteristics | Pattern Score | OR/RR (95% CI) | p for Trend | Matched or Adjusted Variables | NOS |
|---|---|---|---|---|---|---|---|---|
| Willemsen et al., 2021 [20] Canada | Cohort Alberta’s Tomorrow Project (ATP) 26,462 subjects Follow-up: 13.3 ± 3.3 years Incident cases: 252 Age: 35–69 years | 124-item FFQ 1 30 food groups PCA 2 Varimax rotation, EIG 3 > 0.35 Loading ≥ 0.35 3 factors, VE 4 42.4% RRR 5 4 factors, VE 88.3% | PCA 1. Western: grain, non-whole grains, vegetables, white potatoes, cheese, lamb, pork, beef, luncheon meats (red and processed meats), discretionary fats, added sugar | Quartile 1 Quartile 2 Quartile 3 Quartile 4 | 1.00 (Ref.) 1.06 (0.74–1.52) 1.10 (0.76–1.59) 1.10 (0.70–1.73) | 0.64 | Age, sex, BMI 6, energy intake, smoking status, physical activity | 9 |
| 2. Prudent: vegetables, fruits, lean meat from fish and other sea food | Quartile 1 Quartile 2 Quartile 3 Quartile 4 | 1.00 (Ref.) 0.77 (0.55–1.07) 0.71 (0.50–1.01) 0.72 (0.50–1.04) | 0.50 | |||||
| 3. Sugar, fruits, and dairy: grain servings, especially whole grains, fruits, dairy, and teaspoons of added sugar | Quartile 1 Quartile 4 | 1.00 (Ref.) 0.67 (0.46–0.98) | 0.007 | |||||
| RRR 1. Dietary fiber: grain servings, vegetables, and fruits 2. vitamin D: dairy, fish and other seafood 3. Fructose: fruits and teaspoons of added sugar 4. Discretionary fat: solid fats present within the “Milk” and “Meat and Beans | Quartile 1 Quartile 4 Quartile 1 Quartile 4 Quartile 1 Quartile 4 Quartile 1 Quartile 4 | 1.00 (Ref.) 0.66 (0.41–1.06) 1.00 (Ref.) 0.79 (0.55–1.13) 1.00 (Ref.) 1.54 (1.09–2.18) 1.00 (Ref.) 0.66 (0.44–0.98) | <0.0001 <0.0001 <0.0001 0.058 | |||||
| Wei et al., 2021 [21] UK | Cohort UK Biobank 416,588 subjects Follow-up: 7.13 years Incident cases: 1782 Age: 40–69 years | FFQ/24 h dietary intake 16 food groups PCA Varimax rotation, EIG > 1 Loading ≥ 0.3 3 factors, VE 32% | 1. Western: beef, lamb, mutton, pork and processed meat | Quartile 1 Quartile 2 Quartile 3 Quartile 4 | 1.00 (Ref.) 1.00 (0.87–1.16) 1.05 (0.91–1.21) 1.27 (1.11–1.46) | Age, sex, geographical region, smoking status, ethnicity | 9 | |
| 2. Prudent: salad, raw vegetables, cooked vegetables, fresh fruit, dried fruit, oily fish, non-oily fish and water | Quartile 1 Quartile 2 Quartile 3 Quartile 4 | 1.00 (Ref.) 0.96 (0.84–1.09) 0.88 (0.77–1.00) 0.84 (0.73–0.96) | ||||||
| 3. Open sandwich: processed meat, bread, tea and cheese | Quartile 1 Quartile 4 | 1.00 (Ref.) 1.08 (0.94–1.24) | ||||||
| Hawrysz et al., 2020 [22] Poland | HB 7 case-control Cases: 187 Control: 252 Men Age: 45–80 years, mean 62.6 ± 7.2 years | 62-item FFQ 23 food groups PCA Varimax rotation, EIG > 1.0 Loading > 0.3 3 factors, VE 31% | 1. Westernized Traditional: red and processed meats, white meat, potatoes, other fats, vegetables, refined grain, sweetened beverages, energy drinks, sugar, honey, sweets | Tertile 1 Tertile 2 Tertile 3 | 1.0 (Ref.) 0.79 (0.45–1.37) 0.81 (0.60–1.08) | Age, BMI, smoking, socioeconomic status, physical, occurrence of lung cancer in relatives, occupational exposure in the workplace | 8 | |
| 2. Prudent: whole grain, fruits, nuts, seeds, vegetables, fish, legumes, fruit, vegetable-fruit juices | Tertile 1 Tertile 2 Tertile 3 | 1.0 (Ref.) 0.63 (0.37–1.08) 0.72 (0.53–0.96) | ||||||
| 3. Sweet Dairy: animal fats, milk, fermented and sweetened milk drinks and cheese, eggs, cheese, sugar, honey, sweets, breakfast cereals, refined grain products, vegetable oils, dried fruit and preserves | Tertile 1 Tertile 3 | 1.0 (Ref.) 0.99 (0.75–1.30) | ||||||
| He et al., 2018 [23] Southeast China | PB 8 case-control Cases: 1166 Control: 1179 Age: mean 58.93 ± 15.44 years | 20-item FFQ 11 food groups PCA Varimax rotation Loading > 0.4 4 factors, VE 49.53% | 1. Cereals/wheat and meat: high quality protein, such as seafood, kelp and seaweed, egg and beans | Quartile 1 Quartile 4 | 1.0 (Ref.) 0.831 (0.645–1.070) | 0.230 | BMI, incomes, occupation, education, family history of lung cancer, history of lung diseases, environmental tobacco smoke, smoking status | 6 |
| 2. Fruits and vegetables: milk, fruits and vegetables | Quartile 1 Quartile 2 Quartile 3 Quartile 4 | 1.0 (Ref.) 0.447 (0.354–0.566) 0.285 (0.221–0.368) 0.216 (0.164–0.284) | <0.001 | |||||
| 3. Frugal pattern: cereals/wheat and meat: pork, beef, lamb, poultry | Quartile 1 Quartile 2 Quartile 3 Quartile 4 | 1.0 (Ref.) 0.873 (0.675–1.129) 0.897 (0.695–1.159) 1.235 (0.966–1.581) | 0.073 | |||||
| 4. High quality protein: sweet potato and salty vegetables | Quartile 1 Quartile 4 | 1.0 (Ref.) 1.283 (0.999–1.643) | 0.063 | |||||
| Tu et al., 2016 [24] USA | HB case-control Cases: 2139 Age: mean 61.8 ± 10.4 years Control: 2163 Age: mean 61.9 ± 9.7 years | 117-item FFQ 30 food groups PCA Varimax rotation, EIG > 1.0 Loading ≥ 0.38 3 factors, VE 26% | 1. American/Western: hamburgers, cheeseburgers, French fries, fried potatoes, fried chicken, biscuits, rolls, chicken fried steak, gravies, pork chops, pork roasts, dinner, ham, bacons, sausage, chorizo, cheese dishes | Quintile 1 Quintile 2 Quintile 3 Quintile 4 Quintile 5 | 1.0 (Ref.) 1.02 (0.83–1.26) 1.10 (0.89–1.35) 1.33 (1.09–1.64) 1.45 (1.18–1.78) | <0.001 | Age, sex, education, smoking status, pack-years, family history of lung cancer among 1° relatives, body mass index, physical activity, and total energy intake | 9 |
| 2. Fruits and Vegetables: deep yellow vegetables, cruciferous vegetables, dark leafy green vegetable, apples, pears, melons, tomatoes, grapes, strawberries, bananas, peaches | Quintile 1 Quintile 2 Quintile 3 Quintile 4 Quintile 5 | 1.0 (Ref.) 0.94 (0.77–1.14) 0.85 (0.69–1.03) 0.84 (0.68–1.03) 0.68 (0.55–0.85) | 0.001 | |||||
| 3. Tex-Mex: salsa, enchiladas, Spanish rice, refried beans, pinto beans, green chilis, jalapenos, serrano, peppers, avocado, guacamole, flour tortillas, soft tacos, flautas, crispy tacos, corn tortillas | Quintile 1 Quintile 5 | 1.0 (Ref.) 0.45 (0.37–0.56) | <0.001 | |||||
| Gnagnarella et al., 2013 [25] Italy | Cohort COSMOS 4336 subjects Incident cases: 178 Follow-up: 5.7 years Heavy smokers | 188-item FFQ 27 food groups PCA Varimax rotation, EIG > 1.0 Loading ≥ 0.63 4 factors, VE 81.38% | 1. Animal product: animal protein, SFA, linoleic acid, Cholesterol, phosphorus, zinc, vitamin B2 | Quartile 1 Quartile 2 Quartile 3 Quartile 4 | 1.00 (Ref.) 1.00 (0.64–1.56) 1.34 (0.88–2.04) 1.23 (0.80–1.89) | 0.18 | Baseline risk probability (age, sex, smoking history and exposure to asbestos) other nutrient patterns | 6 |
| 2. Vitamins and fiber: dietary fiber, potassium, vitamin C, total folate, b-Carotene, Vitamin E | Quartile 1 Quartile 2 Quartile 3 Quartile 4 | 1.00 (Ref.) 0.96 (0.66–1.41) 0.82 (0.55–1.22) 0.57 (0.36–0.90) | 0.01 | |||||
| 3. Starch-rich: vegetable protein, starch, sodium | Quartile 1 Quartile 4 | 1.00 (Ref.) 1.00 (0.66–1.51) | 0.94 | |||||
| 4. Other PUFA: other PUFA, vitamin D | Quartile 1 Quartile 4 | 1.00 (Ref.) 0.88 (0.58–1.34) | 0.59 | |||||
| Gorlova et al., 2011 [26] USA | HB case-control Cases: 299 Age: mean 61.52 ± 13.1 years Control: 317 Age: mean 61.53 ± 12.62 years Never smokers | 201-item FFQ PCA Loading > 0.3 2 factors, VE 6.76% | 1. Mixed dishes: onions raw/cooked, refried/pinto beans, spaghetti, lasagna, summer squash, cheese dishes without tomato souce, lettuce salad, green peas, avocado, guacamole, salsa, soft tacos, corn, including on the cob, Spanish rice, mayonnaise, grapes, dishes made with mole, raw tomatoes, boiled, baked, mashed potatoes, doughnuts, pastries, ketchup | Tertile 1 Tertile 3 | 1.00 (Ref.) 0.71 (0.41–1.19) | Age, gender, caloric intake, education Never Smokers | 8 | |
| 2. Healthy eating: Low fat salad dressing, carrots, celery, broccoli, apples, applesauce, low fat yogurt, raw spinach, raw tomatoes, nonfat milk in cereal | Tertile 1 Tertile 2 Tertile 3 | 1.00 (Ref.) 0.95 (0.64–1.42) 0.65 (0.42–0.98) | ||||||
| De Stefani et al., 2011 [27] Uruguay | HB case-control Cases: 200 Control: 800 Men Age: 30–79 years | 64-item FFQ PCA Varimax rotation Loading > 0.39 4 factors, VE 37.4% | 1. Western: red meat, processed meat, wine | Quartile 1 Quartile 2 Quartile 3 Quartile 4 | 1.0 (Ref.) 1.30 (0.73–1.32 1.73 (0.98–3.06 1.94 (1.08–3.45) | 0.01 | Age, residence, interviewer, hospital, education, family history of lung cancer, BMI, smoking, total energy intake | 8 |
| 2. Prudent: white meat, cheese, leafy vegetables, total fruits | Quartile 1 Quartile 2 Quartile 3 Quartile 4 | 1.0 (Ref.) 0.77 (0.49–1.22 0.65 (0.40–1.05 0.54 (0.32–0.92) | 0.01 | |||||
| 3. Starchy vegetables: vegetables potato, sweet potato, winter squash | Quartile 1 Quartile 4 | 1.0 (Ref.) 0.49 (0.28–0.86) | 0.007 | |||||
| 4. Milk/coffee: whole milk, coffee | Quartile 1 Quartile 4 | 1.0 (Ref.) 2.30 (1.35–3.90) | 0.0002 | |||||
| De Stefani et al., 2009 [28] Uruguay | HB case-control Cases: 920 Control: 2532 Age: mean 58/66 years | 64-item FFQ PCA Varimax rotation Loading > 0.39 4 factors, VE 37.8% | 1. Western: fried red meat, barbecue and eggs | Tertile 1 Tertile 2 Tertile 3 | 1.0 (Ref.) 1.23 (0.98–1.54) 1.69 (1.35–2.11) | Age, residence, urban/rural status, education, BMI, smoking, total energy intake, all the dietary patterns | 8 | |
| 2. Prudent: poultry, fish, fresh vegetables, and total fruits. | Tertile 1 Tertile 2 Tertile 3 | 1.0 (Ref.) 1.00 (0.56–1.77) 1.00 (0.58–1.74) | ||||||
| 3. Traditional: total grains, all tubers, desserts, and dairy foods | Tertile 1 Tertile 3 | 1.0 (Ref.) 1.08 (0.82–1.42) | ||||||
| 4. Drinker: beer, wine and hard liquor | Tertile 1 Tertile 3 | 1.0 (Ref.) 1.28 (1.03–1.59) | ||||||
| De Stefani et al., 2008 [29] Uruguay | HB case-control Cases: 846 Control: 846 Men Age: 30–89 years | 64-item FFQ PCA Varimax rotation Loading > 0.49 3 factors, VE 0,93% | 1. High-meat protein: saturated fat, monounsaturated fat, linoleic acid, linolenic acid, cholesterol | Tertile 1 Tertile 2 Tertile 3 | 1.0 (Ref.) 1.61 (1.16–2.35) 2.90 (1.91–4.40) | <0.0001 | Age, residence, urban/rural status, education, family history of lung cancer BMI, smoking, alcohol, mate consumption, total energy intake | 8 |
| 2. Antioxidants: glucose, fructose, carotenoids, vitamin C, Vitamin E, folate | Tertile 1 Tertile 2 Tertile 3 | 1.0 (Ref.) 0.66 (0.50–0.89) 0.69 (0.51–0.96) | 0.02 | |||||
| 3. Carbohydrates: Starch, dietary fiber, thiamine, riboflavine, sodium, iron | Tertile 1 Tertile 3 | 1.0 (Ref.) 1.04 (0.72–1.52) | 0.86 | |||||
| Balder et al., 2005 [30] Netherlands | Cohort Netherlands Cohort Study 58,279 subjects Men Incident cases: 1426 Age: 62.6 years Follow-up: 9.3 years | 150-item FFQ 51 food groups PCA Varimax rotation, EIG > 1.0 Loading > 0.35 5 factors, VE 23% | 1. Salad vegetables: Leaf vegetables, allium vegetables, tomatoes, mushrooms, rice, pasta, oil, wine | Quintile 1 Quintile 2 Quintile 3 Quintile 4 Quintile 5 | 1.0 (Ref.) 1.07 (0.81–1.40) 1.02 (0.77–1.35) 0.75 (0.56–1.01) 0.75 (0.55–1.01) | 0.008 | Age, total energy intake, smoking, higher vocational or university education, family history of lung cancer, physical activity | 7 |
| 2. Cooked vegetables: Legumes, cabbages, leaf vegetables, cooked leaf vegetables | Quintile 1 Quintile 5 | 1.0 (Ref.) 0.86 (0.63–1.16) | 0.18 | |||||
| 3. Pork, processed meat and potatoes: Potatoes and potato products, bread, crackers, pork, processed meat, low-fat margarine, coffee | Quintile 1 Quintile 2 Quintile 3 Quintile 4 Quintile 5 | 1.0 (Ref.) 1.18 (0.87–1.61) 1.32 (0.96–1.80) 1.24 (0.90–1.71) 1.44 (0.99–2.09) | 0.08 | |||||
| 4. Sweet foods: Strawberries, savory snacks, cakes, sweet breads, cookies, and biscuits, added sugar | Quintile 1 Quintile 5 | 1.0 (Ref.) 0.62 (0.43–0.89) | 0.002 | |||||
| 5. Brown/white bread substitution: Apples, pears, bread, crackers, brown/whole meal types | Quintile 1 Quintile 5 | 1.0 (Ref.) 0.89 (0.65–1.20) | 0.18 | |||||
| Tsai et al., 2003 [31] USA | HB Case-control Cases: 254 Control: 184 Age: mean 63.13 ± 9.26 years | 61-item FFQ Cluster analysis 2 factors | 1. Unhealthy hight-fat low-fiber: alcohol, animal protein, saturated fat and cholesterol | Sex, age, smoking | 7 | |||
| 2. Healthy high-fiber-low-fat: carbohydrates, dietary fiber (folate, carotene, vitamin A, calcium, magnesium, potassium, copper) | 1.0 (Ref.) 0.93 (0.59–1.44) |
| Combined Risk Estimate | Test of Heterogeneity | Publication Bias | |||||
|---|---|---|---|---|---|---|---|
| Value (95% CI) | p | Q | I2 % | p | p (Egger test) | p (Begg test) | |
| “Western/meat” dietary pattern | |||||||
| Study type | |||||||
| Case-control (n = 6) 3 | 1.50 (1.12–2.00) | 0.006 | 29.98 | 83.32 | <0.0001 | 0.583 | 0.851 |
| Cohort (n = 4) | 1.27 (1.13–1.43) | 0.0001 | 0.84 | 0.00 | 0.839 | 0.869 | 0.174 |
| Pooled 4 (n = 10) | 1.39 (1.17–1.65) | 0.0002 | 32.66 | 72.45 | 0.0001 | 0.580 | 0.655 |
| Smoking status | |||||||
| Current smokers (n = 7) | 1.35 (1.06–1.71) | 0.015 | 16.70 | 64.06 | 0.01 | 0.587 | 0.881 |
| Former smokers (n = 4) | 1.93 (1.11–3.36) | 0.019 | 26.41 | 88.64 | <0.0001 | 0.380 | 0.174 |
| Never smokers (n = 3) | 1.25 (0.80–1.93) | 0.325 | 7.77 | 74.27 | 0.021 | 0.398 | 0.602 |
| “Healthy/Prudent” dietary pattern | |||||||
| Study type | |||||||
| Case-control (n = 8) | 0.62 (0.43–0.89) | 0.010 | 63.34 | 88.95 | <0.0001 | 0.528 | 0.805 |
| Cohort (n = 4) | 0.79 (0.70–0.89) | 0.0001 | 3.07 | 2.26 | 0.381 | 0.051 | 0.042 |
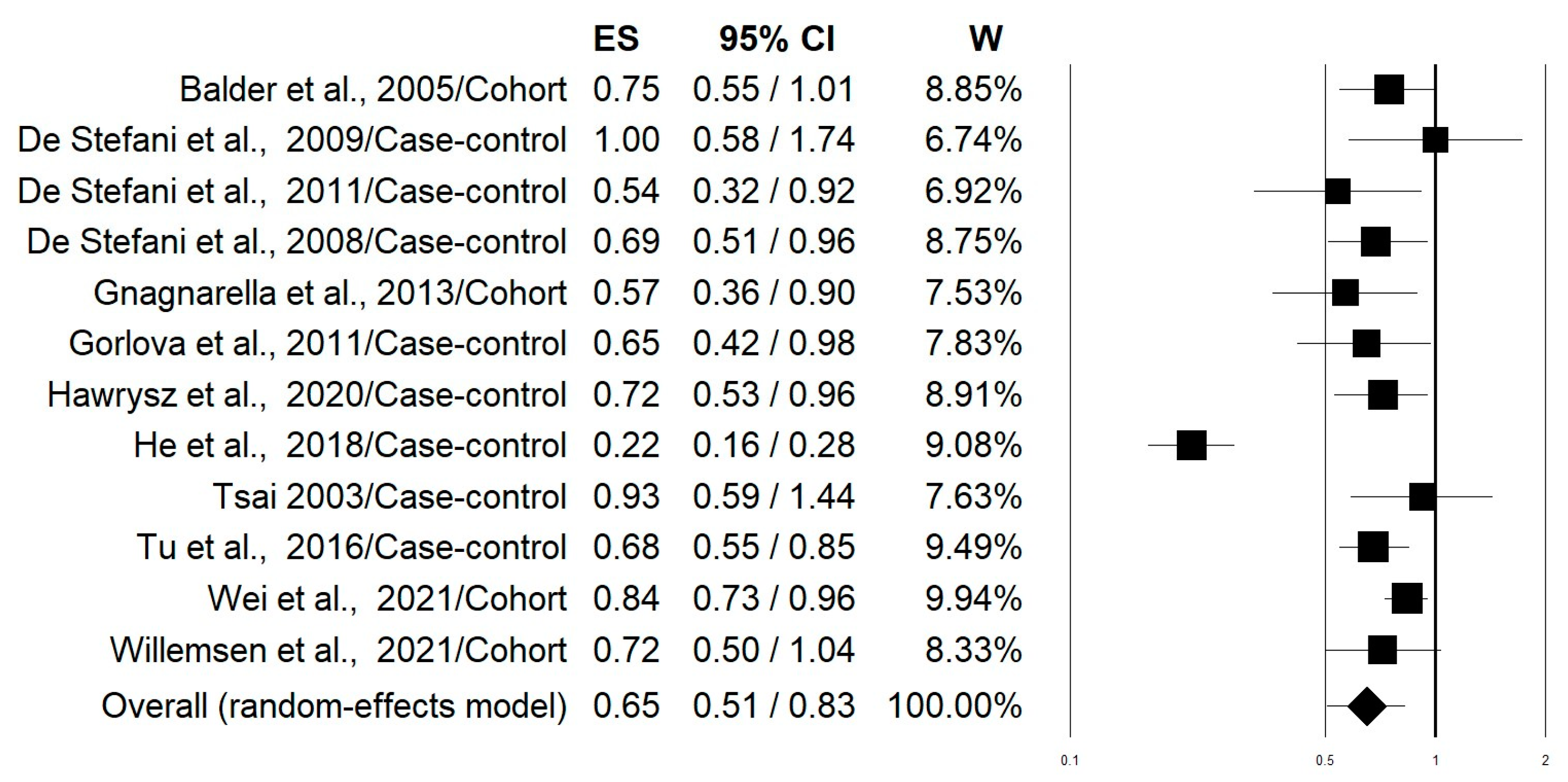
| Pooled 4 (n = 12) | 0.65 (0.51–0.83) | 0.001 | 81.93 | 86.57 | <0.0001 | 0.555 | 0.583 |
| Smoking status | |||||||
| Current smokers (n = 8) | 0.64 (0.46–0.88) | 0.007 | 43.33 | 83.85 | <0.0001 | 0.156 | 0.805 |
| Former smokers (n = 4) | 0.61 (0.44–0.85) | 0.003 | 8.39 | 64.25 | 0.039 | 0.241 | 0.497 |
| Never smokers (n = 4) | 0.60 (0.24–1.49) | 0.266 | 71.17 | 95.78 | <0.0001 | 0.464 | 0.999 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fabiani, R.; La Porta, G.; Li Cavoli, L.; Rosignoli, P.; Chiavarini, M. Adherence to Data-Driven Dietary Patterns and Lung Cancer Risk: A Systematic Review and Dose–Response Meta-Analysis. Nutrients 2023, 15, 4406. https://doi.org/10.3390/nu15204406
Fabiani R, La Porta G, Li Cavoli L, Rosignoli P, Chiavarini M. Adherence to Data-Driven Dietary Patterns and Lung Cancer Risk: A Systematic Review and Dose–Response Meta-Analysis. Nutrients. 2023; 15(20):4406. https://doi.org/10.3390/nu15204406
Chicago/Turabian StyleFabiani, Roberto, Gianandrea La Porta, Laura Li Cavoli, Patrizia Rosignoli, and Manuela Chiavarini. 2023. "Adherence to Data-Driven Dietary Patterns and Lung Cancer Risk: A Systematic Review and Dose–Response Meta-Analysis" Nutrients 15, no. 20: 4406. https://doi.org/10.3390/nu15204406
APA StyleFabiani, R., La Porta, G., Li Cavoli, L., Rosignoli, P., & Chiavarini, M. (2023). Adherence to Data-Driven Dietary Patterns and Lung Cancer Risk: A Systematic Review and Dose–Response Meta-Analysis. Nutrients, 15(20), 4406. https://doi.org/10.3390/nu15204406









