Differences in the Microbiological Profile of Raw and Pasteurized Breastmilk from Hospital and Community-Based Donors at the First Human Milk Bank in Vietnam
Abstract
1. Introduction
2. Methods
2.1. Study Data and Donation Selection
2.2. Donor Human Milk Collection and Processing
2.3. Microbiology Testing
2.3.1. Sample Collection and Transportation
2.3.2. Inoculation and Incubation
2.3.3. Counting of Colony-Forming Unit
2.3.4. Identifying Microorganisms
2.3.5. Reporting the Results of Microorganisms
2.4. Decisions Based on Microbiological Tests
2.5. Data Analysis
3. Results
3.1. General Characteristics
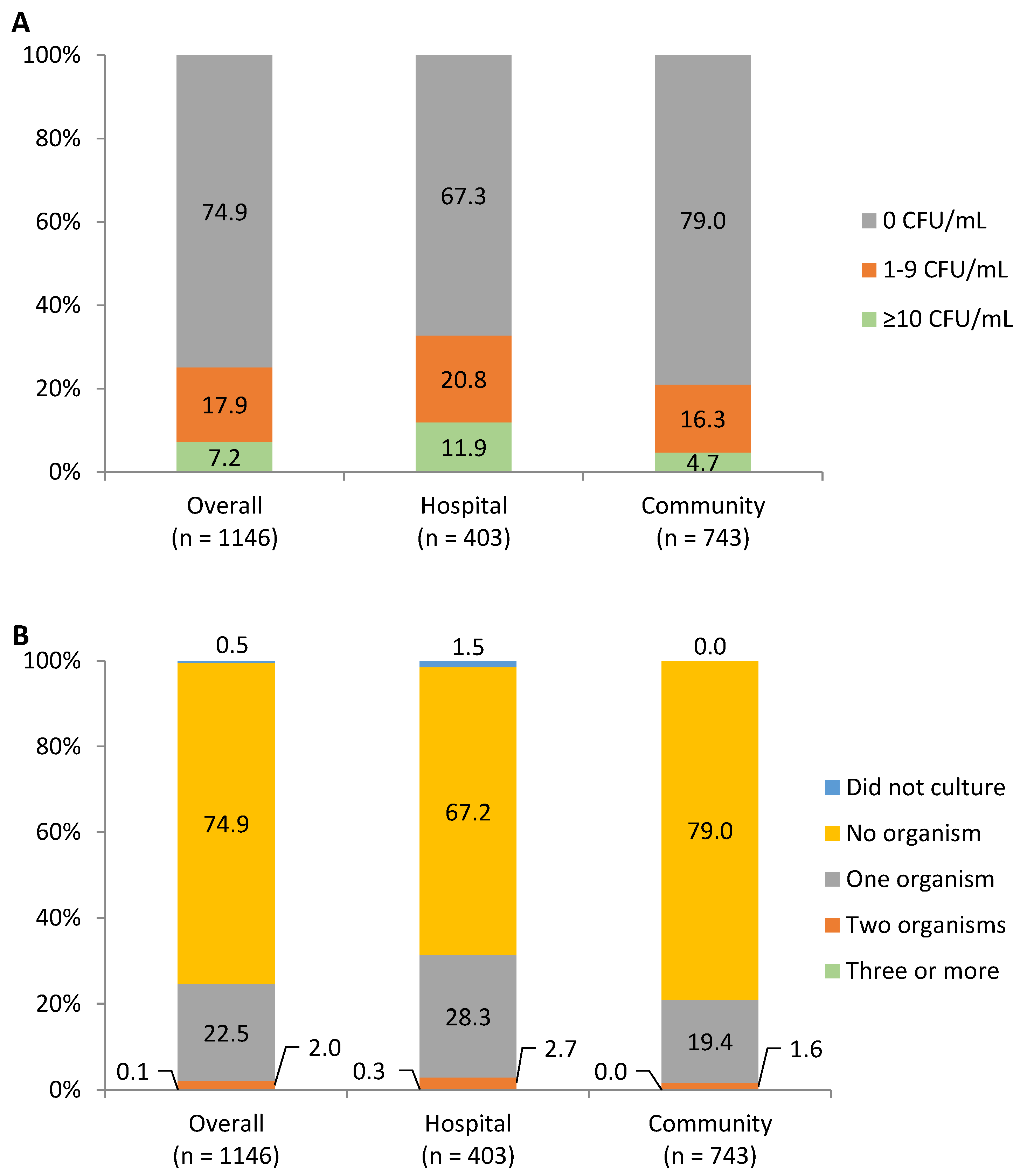
3.2. The Count of Microorganisms
3.3. Types of Microorganisms
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Tran, H.T.; Nguyen, T.T.; Mathisen, R. The use of human donor milk. BMJ 2020, 371, m4243. [Google Scholar] [CrossRef]
- Mansen, K.; Nguyen, T.T.; Nguyen, N.Q.; Do, C.T.; Tran, H.T.; Nguyen, N.T.; Mathisen, R.; Nguyen, V.D.; Ngo, Y.T.K.; Israel-Ballard, K. Strengthening Newborn Nutrition Through Establishment of the First Human Milk Bank in Vietnam. J. Hum. Lact. 2020, 890334420948448. [Google Scholar] [CrossRef] [PubMed]
- Tran, H.T.; Nguyen, T.T.; Barnett, D.; Weaver, G.; Nguyen, O.T.X.; Van Ngo, Q.; Le, H.T.T.; Huynh, L.T.; Do, C.T.; Mathisen, R. Trends and Dynamics in the First Four Years of Operation of the First Human Milk Bank in Vietnam. Nutrients 2021, 13, 1107. [Google Scholar] [CrossRef] [PubMed]
- Human Milk Bank Global Map. Available online: https://public.tableau.com/app/profile/human.milk.bank.global.map/viz/HumanMilkBankGlobalMap_0/HumanMilkBankGlobalMap (accessed on 24 November 2022).
- PATH. Strengthening Human Milk Banking: A Resource Toolkit for Establishing and Integrating Human Milk Bank Programs—A Global Implementation Framework; PATH: Seattle, DC, USA, 2019. [Google Scholar]
- Almutawif, Y.; Hartmann, B.; Lloyd, M.; Erber, W.; Geddes, D. A retrospective audit of bacterial culture results of donated human milk in Perth, Western Australia. Early Hum. Dev. 2017, 105, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Landers, S.; Updegrove, K. Bacteriological screening of donor human milk before and after Holder pasteurization. Breastfeed. Med. 2010, 5, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Chang, F.Y.; Cheng, S.W.; Wu, T.Z.; Fang, L.J. Characteristics of the first human milk bank in Taiwan. Pediatr. Neonatol. 2013, 54, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Xiaoshan, H.; Xue, C.; Jun, Z.; Feng, L.; Xiaohui, C.; Zhangbin, Y.; Shuping, H. Eight-year operation status and data analysis of the first human milk bank in East China. Int. Breastfeed. J. 2022, 17, 65. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.H.; Han, S.P.; Wei, Q.F.; Zheng, F.Y.; Zhang, T.; Chen, H.M.; Mao, M. The data and characteristics of the human milk banks in mainland China. World J. Pediatr. 2019, 15, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Gidrewicz, D.A.; Fenton, T.R. A systematic review and meta-analysis of the nutrient content of preterm and term breast milk. BMC Pediatr. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Pimenteira Thomaz, C.A.; Maia Loureiro, L.V.; da Silva Oliveira, T.; de Mendonça Furtado Montenegro, N.C.; Dantas Almeida Júnior, E.; Fernando Rodrigues Soriano, C.; Calado Cavalcante, J. The human milk donation experience: Motives, influencing factors, and regular donation. J. Hum. Lact. 2008, 24, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Jang, H.L.; Cho, J.Y.; Kim, M.J.; Kim, E.J.; Park, E.Y.; Park, S.A.; Kim, I.Y.; Choi, Y.S.; Bae, C.W.; Chung, S.H. The Experience of Human Milk Banking for 8 Years: Korean Perspective. J. Korean Med. Sci. 2016, 31, 1775–1783. [Google Scholar] [CrossRef] [PubMed]
- Clifford, V.; Klein, L.D.; Sulfaro, C.; Karalis, T.; Hoad, V.; Gosbell, I.; Pink, J. What are Optimal Bacteriological Screening Test Cut-Offs for Pasteurized Donor Human Milk Intended for Feeding Preterm Infants? J. Hum. Lact. 2020, 890334420981013. [Google Scholar] [CrossRef] [PubMed]
- Padín Fontán, M.; Martín-Forero Maestre, M.; Rodríguez Otero, I.; Durán Fernández-Feijoo, C.; Suárez Albo, M.; Concheiro Guisán, A. Influence of donor profile on pre and post-pasteurization bacteriology of donated human milk. Nutr. Hosp. 2022, 39, 1064–1067. [Google Scholar] [CrossRef]
- Strom, K.; Jarzynka, S.; Minkiewicz-Zochniak, A.; Barbarska, O.; Olędzka, G.; Wesolowska, A. Microbiological Quality of Milk Donated to the Regional Human Milk Bank in Warsaw in the First Four Years of Activity. Healthcare 2022, 10, 444. [Google Scholar] [CrossRef]
- Asbury, M.R.; Butcher, J.; Copeland, J.K.; Unger, S.; Bando, N.; Comelli, E.M.; Forte, V.; Kiss, A.; LeMay-Nedjelski, L.; Sherman, P.M.; et al. Mothers of Preterm Infants Have Individualized Breast Milk Microbiota that Changes Temporally Based on Maternal Characteristics. Cell Host Microbe 2020, 28, 669–682.e4. [Google Scholar] [CrossRef] [PubMed]
- Beghetti, I.; Barone, M.; De Fazio, L.; Laderchi, E.; Biagi, E.; Turroni, S.; Brigidi, P.; Pession, A.; Corvaglia, L.; Aceti, A. A Pilot Study on Donor Human Milk Microbiota: A Comparison with Preterm Human Milk Microbiota and the Effect of Pasteurization. Nutrients 2022, 14, 2483. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Mushajiang, S.; Luo, B.; Tian, F.; Ni, Y.; Yan, W. The Composition and Concordance of Lactobacillus Populations of Infant Gut and the Corresponding Breast-Milk and Maternal Gut. Front. Microbiol. 2020, 11, 597911. [Google Scholar] [CrossRef] [PubMed]
- Adjidé, C.C.; Léké, A.; Mullié, C. Bacillus cereus contamination of pasteurized human milk donations: Frequency, origin, seasonal distribution, molecular typing of strains and proposed corrective/preventive actions. J. Matern. Fetal Neonatal Med. 2022, 35, 1554–1561. [Google Scholar] [CrossRef] [PubMed]
- Mallardi, D.; Piemontese, P.; Liotto, N.; Colombo, R.M.; Dodaro, A.; Schiavello, A.; Tabasso, C.; Plevani, L.; Bezze, E.; Menis, C.; et al. New Operating Approach to Limit Bacillus Cereus Contamination of Donor Human Milk. J. Hum. Lact. 2022, 38, 102–107. [Google Scholar] [CrossRef] [PubMed]
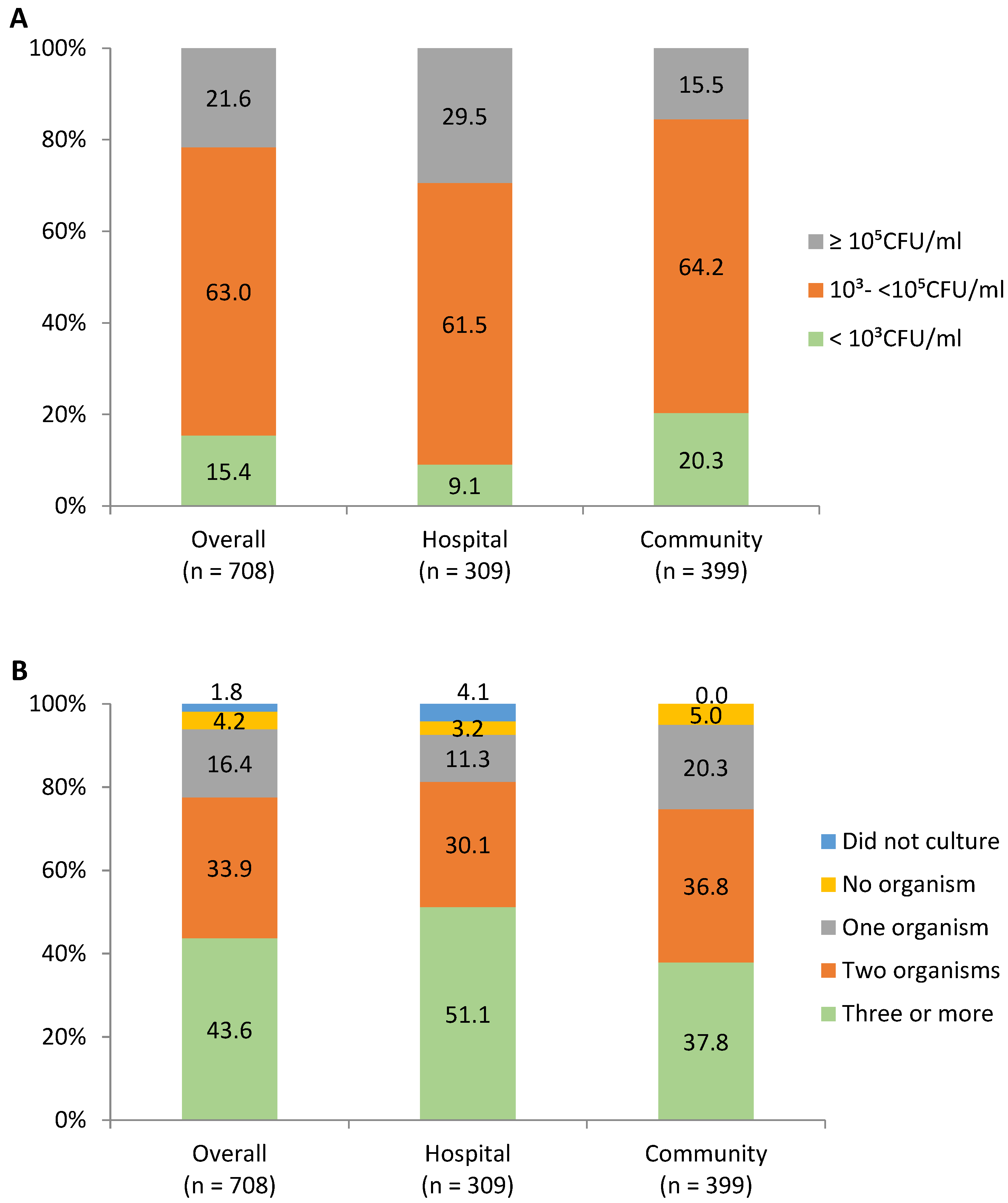
| Characteristics | Total (n = 491) | Hospital (n = 239) | Community (n = 252) |
|---|---|---|---|
| Donor characteristics | |||
| Donor age, Mean ± SD | 28.5 ± 4.1 | 28.2 ± 4.6 | 28.8 ± 3.7 |
| One child, n (%) | 299 (60.9) | 136 (56.9) | 163 (64.7) |
| Preterm births, n (%) | 202 (41.1) | 182 (76.2) | 20 (7.9) |
| Cesarean sections, n (%) | 243 (49.5) | 98 (41.0) | 145 (57.5) |
| Donors from Da Nang, n (%) | 348 (70.9) | 110 (46.0) | 238 (94.4) |
| Education level | |||
| College, university, or higher, n (%) | 276 (56.2) | 89 (37.2) | 187 (74.2) |
| Diploma (2–3 years after high school), n (%) | 90 (18.3) | 55 (23.0) | 35 (13.9) |
| Up to high school, n (%) | 125 (25.5) | 95 (39.8) | 30 (11.9) |
| Occupation | |||
| Office staff, n (%) | 194 (39.5) | 70 (29.3) | 124 (49.2) |
| Teachers, n (%) | 47 (9.6) | 24 (10.0) | 23 (9.1) |
| Health workers, n (%) | 59 (12.0) | 16 (6.7) | 43 (17.1) |
| Factory workers, n (%) | 32 (6.5) | 22 (9.2) | 10 (4.0) |
| Sale or small traders, n (%) | 45 (9.2) | 29 (12.1) | 16 (6.3) |
| Housewife, n (%) | 89 (18.1) | 59 (24.7) | 30 (11.9) |
| Other (farmer, self-employed service), n (%) | 25 (4.9) | 19 (8.0) | 6 (1.2) |
| Human donor milk and pasteurization | |||
| Amount of donor milk (L) | 9504.9 | 3372.1 | 6132.8 |
| Amount of donor milk passed pre- and post- pasteurization test, L (%) | 8069.7 (84.9) | 2682.3 (79.5) | 5387.5 (87.9) |
| Number of pasteurizations | 1244 | 478 (38.4) | 766 (61.6) |
| Number of pre-pasteurization samples tested | 708 | 317 | 391 |
| Number of post-pasteurization samples tested | 1144 | 421 | 723 |
| Passed pre-pasteurization test, n (%) | 559 (79.0) | 226 (71.3) | 333 (85.2) |
| Passed post-pasteurization test, n (%) | 1058 (92.5) | 373 (88.6) | 685 (94.7) |
| Average days of donation, median (IQR) | 45 (24–96) | 30 (20–61) | 70 (29–116) |
| Donors passed all microbiology tests, n (%) | 321 (65.4) | 139 (58.2) | 182 (72.2) |
| Amount of milk per donor, median (IQR) L | 14.2 (7.4–25.2) | 8.4 (5.0–7.0) | 17.5 (8.5–26.3) |
| Number of pasteurizations per donor, mean (SD) | 2.53 ± 2.49 | 2 ± 2.02 | 3.04 ± 2.78 |
| Number passed per donor, mean (SD) | 2.12 ± 2.49 | 1.51 ± 1.98 | 2.70 ± 2.79 |
| Pre-Pasteurization | Post-Pasteurization | |||||
|---|---|---|---|---|---|---|
| Overall (n = 708) | Hospital (n = 309) | Comm. (n = 399) | Overall (n = 1146) | Hospital (n = 403) | Comm. (n = 743) | |
| Staphylococcus epidermidis | 525 (74.2) | 224 (72.5) | 301 (75.4) | 45 (3.9) | 29 (7.2) | 16 (2.2) |
| Acinetobacter sp. | 369 (52.1) | 189 (61.2) | 180 (45.1) | 10 (0.9) | 9 (2.2) | 1 (0.1) |
| Gram-positive bacilli | 366 (51.7) | 156 (50.5) | 210 (52.6) | 241 (21.0) | 95 (23.6) | 146 (19.7) |
| Staphylococcus coagulase negative | 112 (15.8) | 55 (17.8) | 57 (14.3) | 1 (0.1) | 0 | 1 (0.1) |
| Staphylococcus aureus | 74 (10.5) | 41 (13.3) | 33 (8.3) | 1 (0.1) | 1 (0.3) | 0 |
| Pseudomonas sp. | 64 (9.0) | 35 (11.3) | 29 (7.3) | 1 (0.1) | 1 (0.3) | 0 |
| Pseudomonas aeruginosa | 40 (5.7) | 28 (12.5) | 12 (3.0) | 1 (0.1) | 1 (0.3) | 0 |
| Enterococcus sp. | 16 (2.3) | 8 (2.6) | 8 (2.0) | 0 | 0 | 0 |
| Streptococcus sp. | 18 (2.5) | 7 (2.3) | 11 (2.8) | 0 | 0 | 0 |
| Klebsiella sp. | 15 (2.1) | 7 (2.3) | 8 (2.0) | 0 | 0 | 0 |
| Ecoli | 13 (1.8) | 7 (2.3) | 6 (1.5) | 0 | 0 | 0 |
| Serratia sp. | 12 (1.7) | 5 (1.6) | 7 (1.8) | 0 | 0 | 0 |
| Candida albicans | 9 (1.3) | 3 (1.0) | 6 (1.5) | 2 (0.2) | 1 (0.3) | 1 (0.1) |
| Pseudomonas putida | 2 (0.3) | 2 (0.7) | 0 | 0 | 0 | 0 |
| Stenomatophia | 2 (0.3) | 1 (0.3) | 1 (0.3) | 0 | 0 | 0 |
| Candida spp. | 3 (0.4) | 1 (0.3) | 2 (0.5) | 1 (0.1) | 1 (0.3) | 0 |
| Enterobacter sp. | 2 (0.3) | 0 | 2 (0.5) | 0 | 0 | 0 |
| Pantoea sp. | 1 (0.1) | 0 | 1 (0.3) | 0 | 0 | 0 |
| Klebsiella pneumonia | 1 (0.1) | 0 | 1 (0.3) | 0 | 0 | 0 |
| Mold | 1 (0.1) | 0 | 1 (0.3) | 0 | 0 | 0 |
| Stenomatophia | 2 (0.3) | 1 (0.3) | 1 (0.3) | 0 | 0 | 0 |
| Stenotrophoonas maltophilia | 1 (0.1) | 0 | 1 (0.3) | 0 | 0 | 0 |
| Yeast | 1 (0.1) | 0 | 1 (0.3) | 0 | 0 | 0 |
| Proteus spp. | 1 (0.1) | 0 | 1 (0.3) | 0 | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tran, H.T.; Nguyen, T.T.; Nguyen, O.T.X.; Huynh, L.T.; Nguyen, L.T.; Nguyen, T.T.; Le, H.T.T.; Barnett, D.; Weaver, G.; Mathisen, R. Differences in the Microbiological Profile of Raw and Pasteurized Breastmilk from Hospital and Community-Based Donors at the First Human Milk Bank in Vietnam. Nutrients 2023, 15, 412. https://doi.org/10.3390/nu15020412
Tran HT, Nguyen TT, Nguyen OTX, Huynh LT, Nguyen LT, Nguyen TT, Le HTT, Barnett D, Weaver G, Mathisen R. Differences in the Microbiological Profile of Raw and Pasteurized Breastmilk from Hospital and Community-Based Donors at the First Human Milk Bank in Vietnam. Nutrients. 2023; 15(2):412. https://doi.org/10.3390/nu15020412
Chicago/Turabian StyleTran, Hoang Thi, Tuan Thanh Nguyen, Oanh Thi Xuan Nguyen, Le Thi Huynh, Le Thi Nguyen, Thao Thi Nguyen, Huong Thi Thanh Le, Debbie Barnett, Gillian Weaver, and Roger Mathisen. 2023. "Differences in the Microbiological Profile of Raw and Pasteurized Breastmilk from Hospital and Community-Based Donors at the First Human Milk Bank in Vietnam" Nutrients 15, no. 2: 412. https://doi.org/10.3390/nu15020412
APA StyleTran, H. T., Nguyen, T. T., Nguyen, O. T. X., Huynh, L. T., Nguyen, L. T., Nguyen, T. T., Le, H. T. T., Barnett, D., Weaver, G., & Mathisen, R. (2023). Differences in the Microbiological Profile of Raw and Pasteurized Breastmilk from Hospital and Community-Based Donors at the First Human Milk Bank in Vietnam. Nutrients, 15(2), 412. https://doi.org/10.3390/nu15020412









