Serine Metabolism in Health and Disease and as a Conditionally Essential Amino Acid
Abstract
1. Introduction
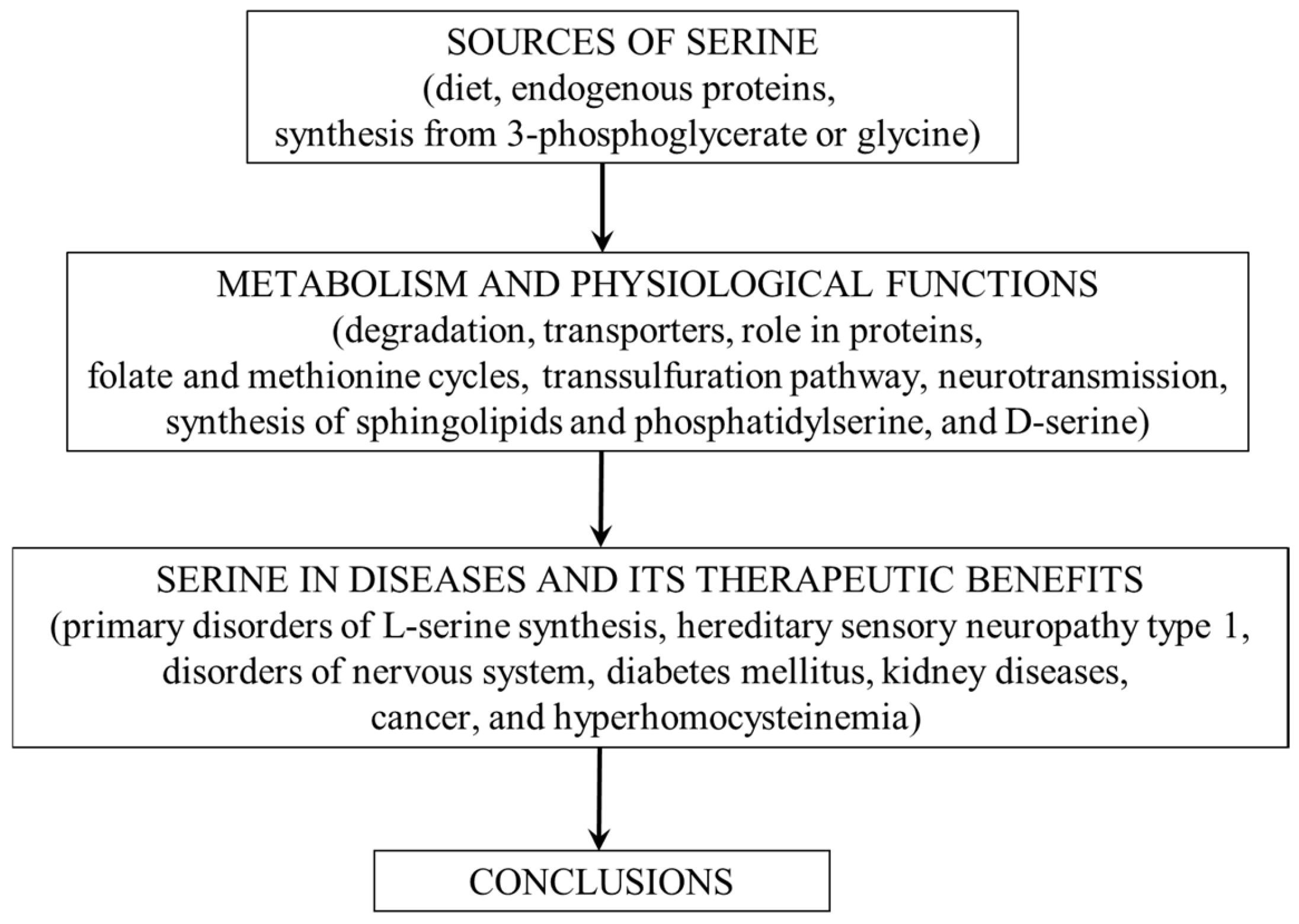
2. Sources of Serine
2.1. Dietary Sources
2.2. Degradation of Endogenous Proteins
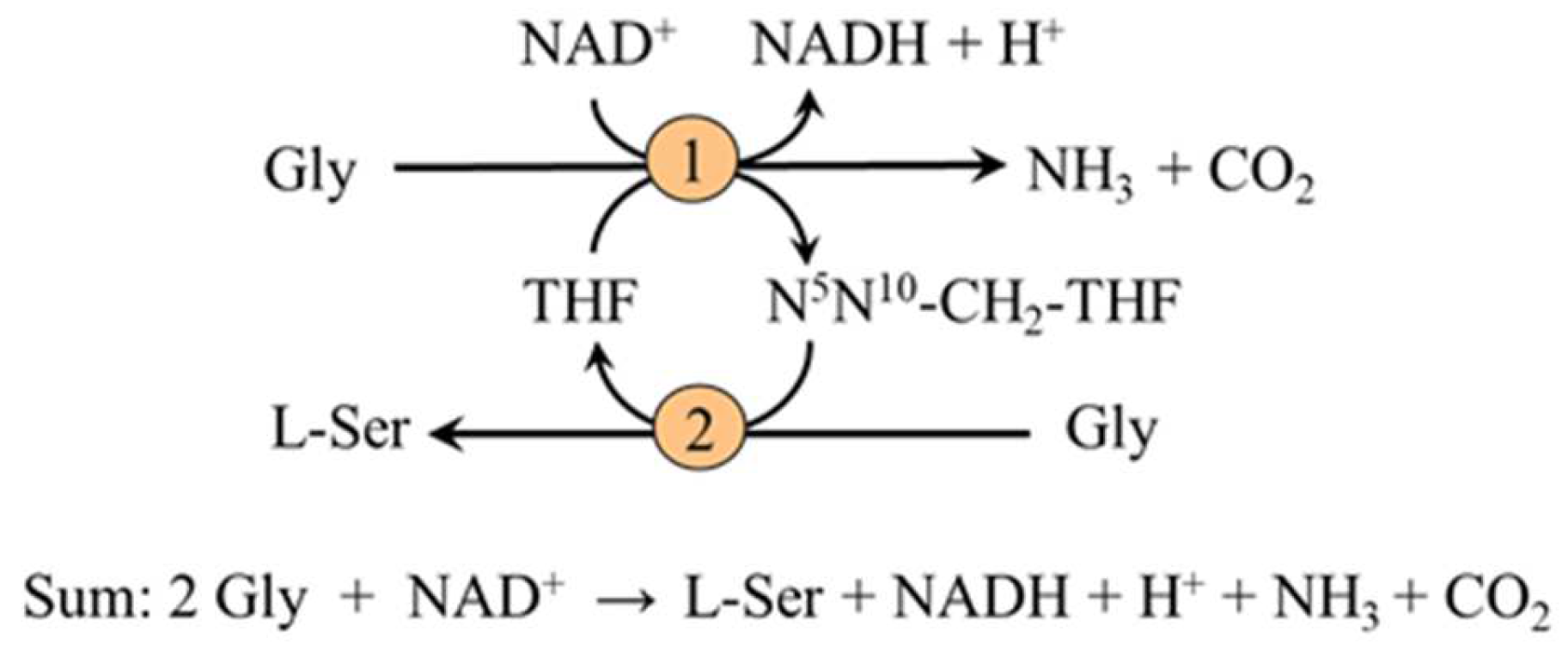
2.3. Synthesis from 3-PG and Glycine
2.3.1. Synthesis from 3-PG
2.3.2. Synthesis from Glycine
2.3.3. Role of the Kidneys in Serine Synthesis
3. Serine Degradation
4. Transporters
- System A (alanine preferring)—a sodium-dependent transporter for small neutral amino acids. It is found in most cell types of the body and includes transporters SNAT1 (SLC38A1), SNAT2 (SLC38A2), and SNAT4 (SLC38A4). Transporter recruitment from vesicles under the plasma membrane to the cell surface is sensitive to amino acid levels and hormones (notably to insulin and glucagon). The system plays a role in tissue amino acid uptake after food intake and in hepatic amino acid uptake to be used for gluconeogenesis during starvation [22,23,24].
- System ASC (alanine, serine, and cysteine preferring)— a sodium-dependent transporter, which includes transporters ASCT1 (SLC1A4) and ASCT2 (SLC1A5). It is supposed that ASCT1 is the main transporter for serine in the brain, where it plays a key role in transporting serine from astrocytes and serine uptake by neurons [25]. ASCT1 mutations cause a disorder of intellectual disability, progressive microcephaly, spasticity and thin corpus callosum [26].
- System asc—a heterodimeric (SLC7A10/SLC3A2) sodium-independent amino acid exchanger. The system includes transporter Asc-1, which is expressed in the brain and has a high affinity for both L- and D-serine [27].
5. Physiological Functions
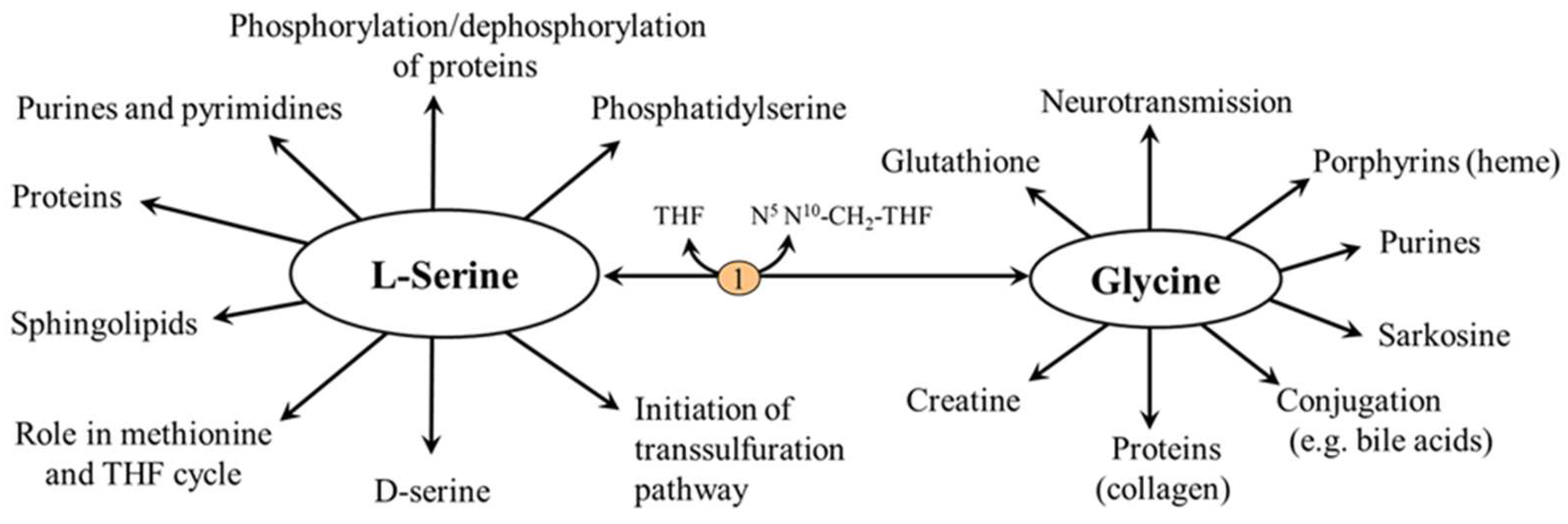
5.1. L-Serine and Proteins
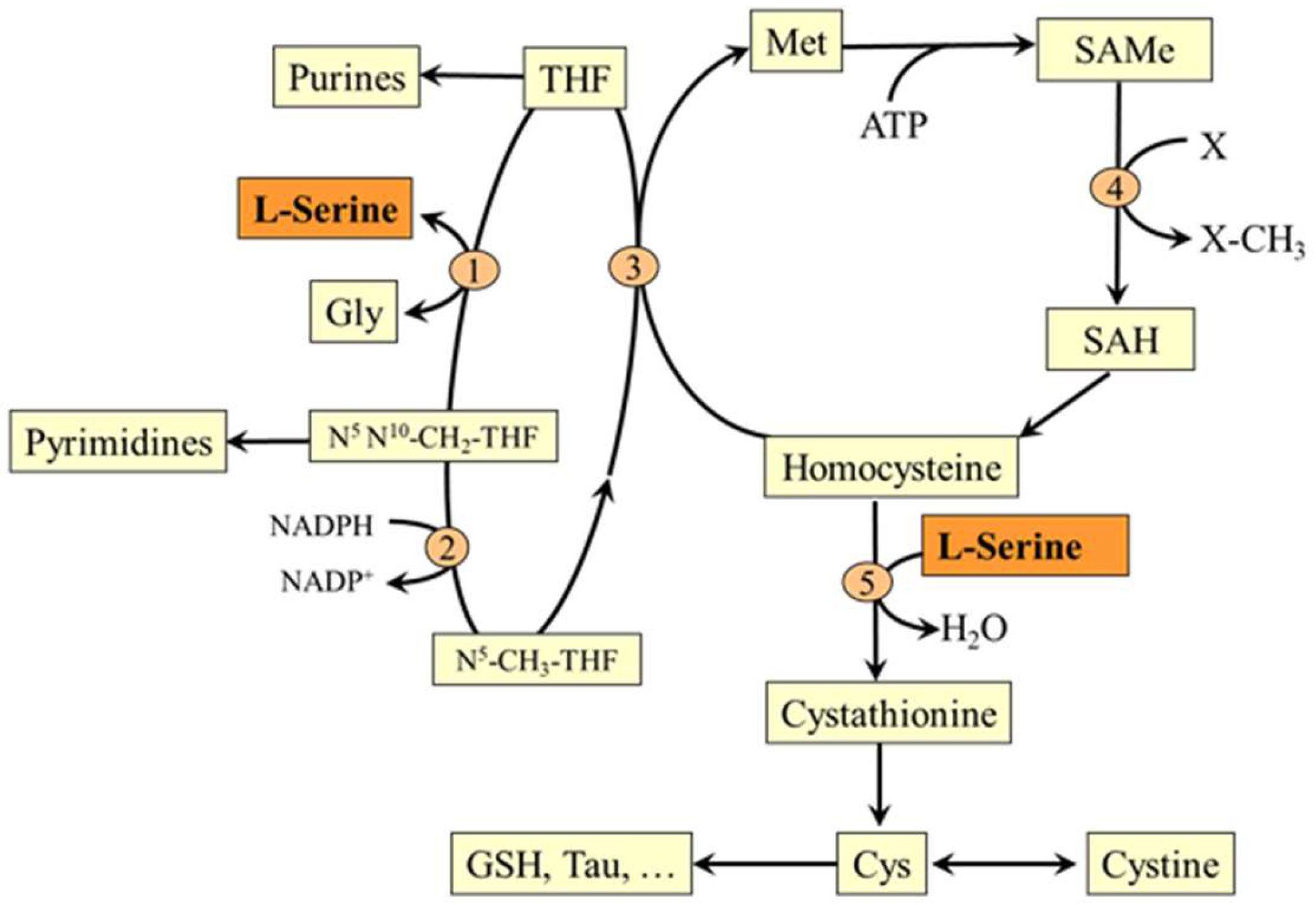
5.2. L-Serine and Folate and Methionine Cycles
5.3. L-Serine and Transsulfuration Pathway
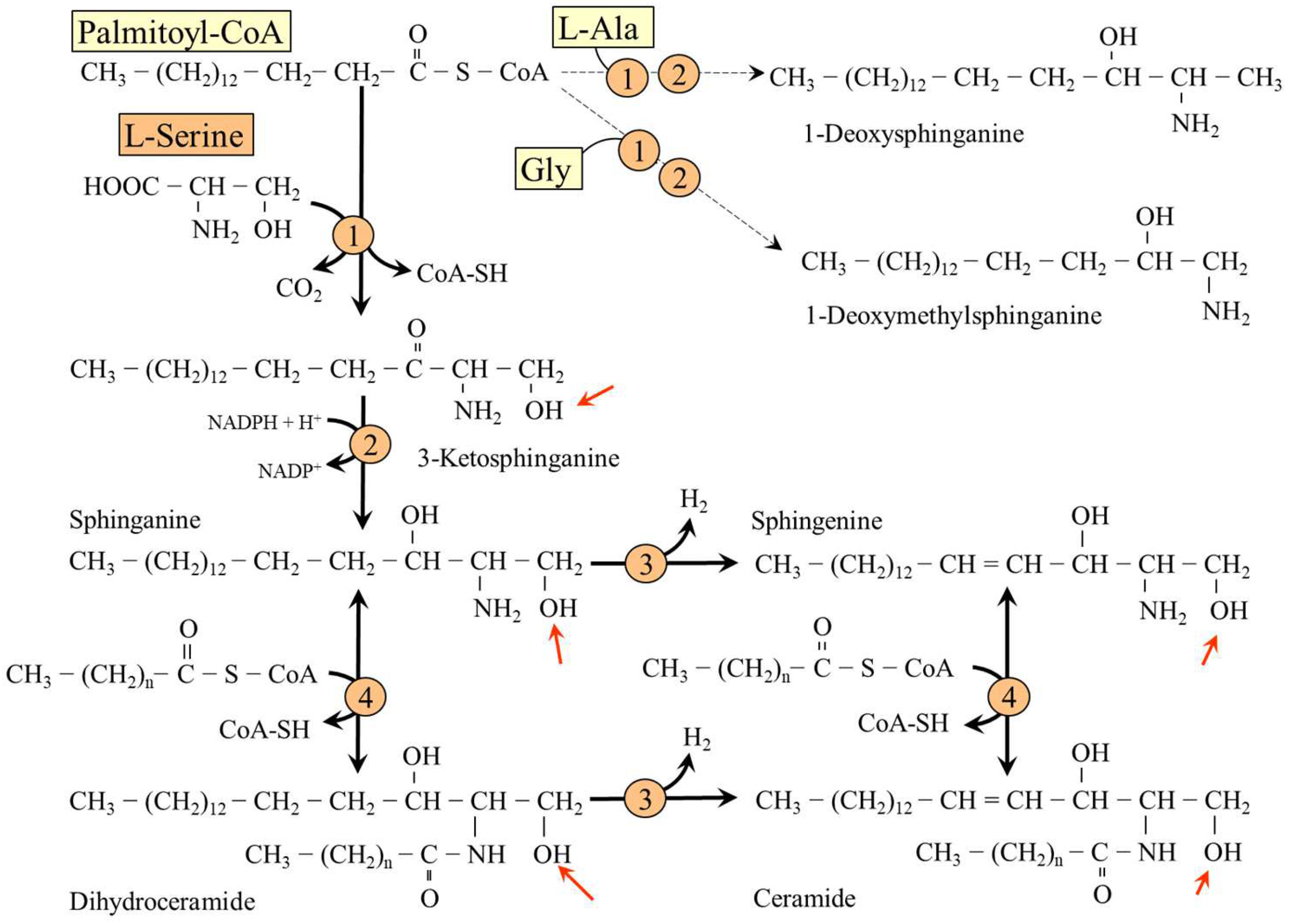
5.4. L-Serine and Sphingolipids (SL)
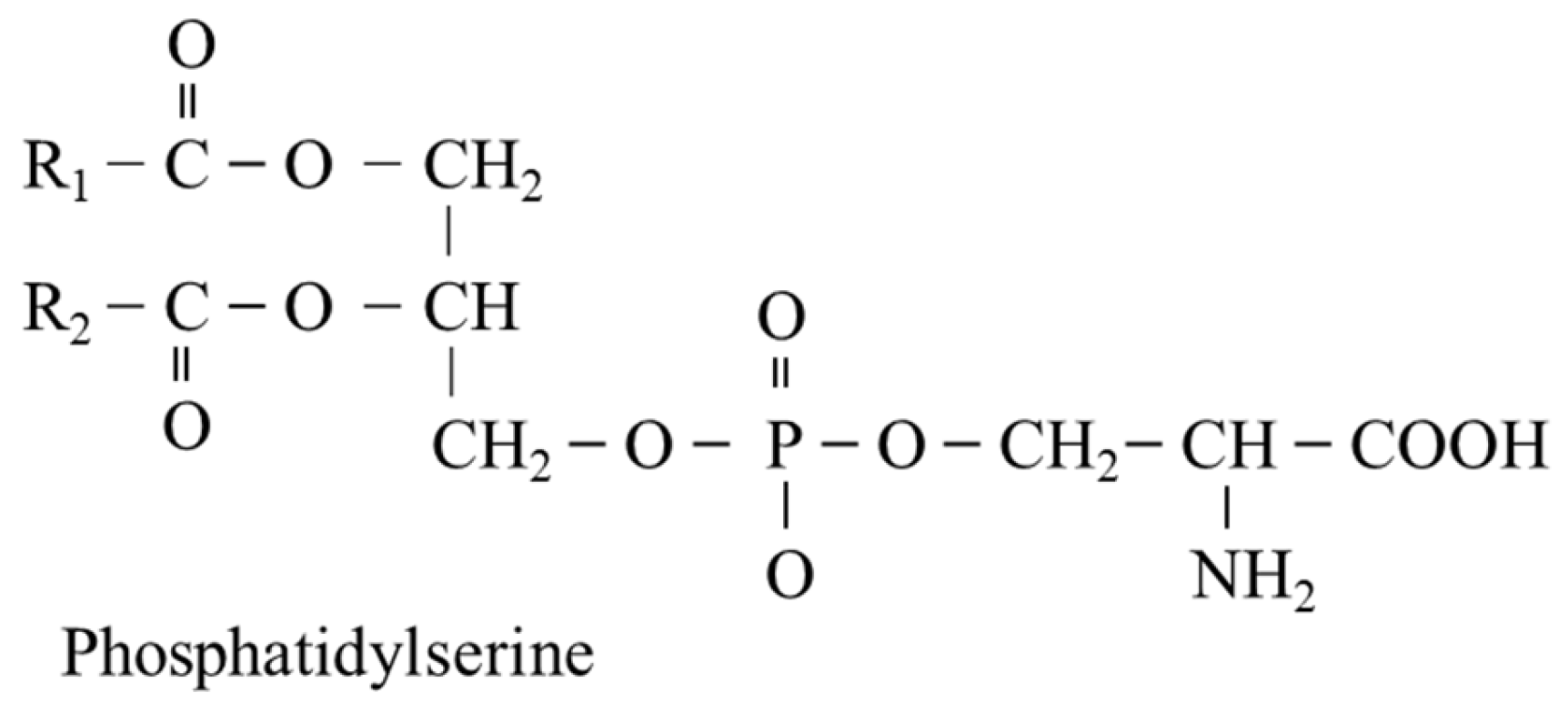
5.5. L-Serine and Phosphatidylserine (PS)
5.6. Serine and Neurotransmission (D-Serine)
5.6.1. L-Serine
5.6.2. Glycine
5.6.3. D-Serine
6. Serine and Disease
6.1. Primary Disorders of L-Serine Synthesis
6.2. Hereditary Sensory Neuropathy Type 1
6.3. Disorders of the Nervous System
- L-serine supplementation has been investigated in the therapy of central nervous system injury, such as that due to cerebral ischemia, stroke, and trauma [40].
- L-serine has shown therapeutic potential in amyotrophic lateral sclerosis [8].
- Clinical trials indicate that PS may prevent and treat depression and age-related cognitive disorders like Alzheimer’s disease [39].
6.4. Serine and Diabetes Mellitus
6.5. Serine and Kidney Diseases
D-Serine
6.6. Serine and Cancer
6.7. Serine and Hyperhomocysteinemia
7. L-Serine as a Conditionally Essential Amino Acid
8. Conclusions
- There are primary disorders of L-serine synthesis resulting in L-serine deficiency.
- Humans cannot synthesize L-serine in sufficient quantities in diabetes and chronic kidney diseases.
- L-serine deficiency is associated with severe neurological abnormalities.
- It was proved that L-serine supplementation is effective in therapy of primary disorders of serine metabolism and diabetic neuropathy.
Funding
Conflicts of Interest
References
- Calianese, D.C.; Birge, R.B. Biology of phosphatidylserine (PS): Basic physiology and implications in immunology, infectious disease, and cancer. Cell Commun. Signal. 2020, 18, 41. [Google Scholar] [CrossRef] [PubMed]
- Penno, A.; Reilly, M.M.; Houlden, H.; Laurá, M.; Rentsch, K.; Niederkofler, V.; Stoeckli, E.T.; Nicholson, G.; Eichler, F.; Brown, R.H., Jr.; et al. Hereditary sensory neuropathy type 1 is caused by the accumulation of two neurotoxic sphingolipids. J. Biol. Chem. 2010, 285, 11178–11187. [Google Scholar] [CrossRef] [PubMed]
- Othman, A.; Rütti, M.F.; Ernst, D.; Saely, C.H.; Rein, P.; Drexel, H.; Porretta-Serapiglia, C.; Lauria, G.; Bianchi, R.; Von Eckardstein, A.; et al. Plasma deoxysphingolipids: A novel class of biomarkers for the metabolic syndrome? Diabetologia 2012, 55, 421–431. [Google Scholar] [CrossRef] [PubMed]
- Othman, A.; Saely, C.H.; Muendlein, A.; Vonbank, A.; Drexel, H.; Von Eckardstein, A.; Hornemann, T. Plasma 1-deoxysphingolipids are predictive biomarkers for type 2 diabetes mellitus. BMJ Open Diabetes Res. Care 2015, 3, e000073. [Google Scholar] [CrossRef] [PubMed]
- Zuellig, R.A.; Hornemann, T.; Othman, A.; Hehl, A.B.; Bode, H.; Güntert, T.; Ogunshola, O.O.; Saponara, E.; Grabliauskaite, K.; Jang, J.H.; et al. Deoxysphingolipids, novel biomarkers for type 2 diabetes, are cytotoxic for insulin-producing cells. Diabetes 2014, 63, 1326–1339. [Google Scholar] [CrossRef]
- Brassier, A.; Valayannopoulos, V.; Bahi-Buisson, N.; Wiame, E.; Hubert, L.; Boddaert, N.; Kaminska, A.; Habarou, F.; Desguerre, I.; Van Schaftingen, E.; et al. Two new cases of serine deficiency disorders treated with l-serine. Eur. J. Paediatr. Neurol. 2016, 20, 53–60. [Google Scholar] [CrossRef]
- De Koning, T.J.; Duran, M.; Dorland, L.; Gooskens, R.; Van Schaftingen, E.; Jaeken, J.; Blau, N.; Berger, R.; Poll-The, B.T. Beneficial effects of L-serine and glycine in the management of seizures in 3-phosphoglycerate dehydrogenase deficiency. Ann. Neurol. 1998, 44, 261–265. [Google Scholar] [CrossRef]
- Levine, T.D.; Miller, R.G.; Bradley, W.G.; Moore, D.H.; Saperstein, D.S.; Flynn, L.E.; Katz, J.S.; Forshew, D.A.; Metcalf, J.S.; Banack, S.A.; et al. Phase I clinical trial of safety of L-serine for ALS patients. Amyotroph. Lateral Scler. Front. Degener. 2017, 18, 107–111. [Google Scholar] [CrossRef]
- Holm, L.J.; Haupt-Jorgensen, M.; Larsen, J.; Giacobini, J.D.; Bilgin, M.; Buschard, K. L-serine supplementation lowers diabetes incidence and improves blood glucose homeostasis in NOD mice. PLoS ONE 2018, 13, e0194414. [Google Scholar] [CrossRef]
- Gorissen, S.H.M.; Crombag, J.J.R.; Senden, J.M.G.; Waterval, W.A.H.; Bierau, J.; Verdijk, L.B.; Van Loon, L.J.C. Protein content and amino acid composition of commercially available plant-based protein isolates. Amino Acids 2018, 50, 1685–1695. [Google Scholar] [CrossRef]
- Cohn, J.S.; Kamili, A.; Wat, E.; Chung, R.W.; Tandy, S. Dietary phospholipids and intestinal cholesterol absorption. Nutrients 2010, 2, 116–127. [Google Scholar] [CrossRef] [PubMed]
- Kalhan, S.C.; Hanson, R.W. Resurgence of serine: An often neglected but indispensable amino acid. J. Biol. Chem. 2012, 287, 19786–19791. [Google Scholar] [CrossRef] [PubMed]
- Knox, W.E.; Herzfeld, A.; Hudson, J. Phosphoserine phosphatase distribution in normal and neoplastic rat tissues. Arch. Biochem. Biophys. 1969, 132, 397–403. [Google Scholar] [CrossRef]
- Achouri, Y.; Robbi, M.; Van Schaftingen, E. Role of cysteine in the dietary control of the expression of 3-phosphoglycerate dehydrogenase in rat liver. Biochem. J. 1999, 344, 15–21. [Google Scholar] [CrossRef]
- Antflick, J.E.; Baker, G.B.; Hampson, D.R. The effects of a low protein diet on amino acids and enzymes in the serine synthesis pathway in mice. Amino Acids 2010, 39, 145–153. [Google Scholar] [CrossRef]
- Lamers, Y.; Williamson, J.; Gilbert, L.R.; Stacpoole, P.W.; Gregory, J.F., 3rd. Glycine turnover and decarboxylation rate quantified in healthy men and women using primed, constant infusions of [1,2-(13)C2]glycine and [(2)H3]leucine. J. Nutr. 2007, 137, 2647–2652. [Google Scholar] [CrossRef]
- Van de Poll, M.C.; Soeters, P.B.; Deutz, N.E.; Fearon, K.C.; Dejong, C.H. Renal metabolism of amino acids: Its role in interorgan amino acid exchange. Am. J. Clin. Nutr. 2004, 79, 185–197. [Google Scholar] [CrossRef]
- Lowry, M.; Hall, D.E.; Brosnan, J.T. Serine synthesis in rat kidney: Studies with perfused kidney and cortical tubules. Am. J. Physiol. 1986, 250, F649–F658. [Google Scholar] [CrossRef]
- Lowry, M.; Hall, D.E.; Hall, M.S.; Brosnan, J.T. Renal metabolism of amino acids in vivo: Studies on serine and glycine fluxes. Am. J. Physiol. 1987, 252, F304–F309. [Google Scholar] [CrossRef]
- Jois, M.; Hall, D.E.; Brosnan, J.T. Serine synthesis by the rat kidney. Contrib. Nephrol. 1988, 63, 136–140. [Google Scholar]
- Mauron, J.; Mottu, F.; Spohr, G. Reciprocal induction and repression of serine dehydratase and phosphoglycerate dehydrogenase by proteins and dietary-essential amino acids in rat liver. Eur. J. Biochem. 1973, 32, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Ling, R.; Bridges, C.C.; Sugawara, M.; Fujita, T.; Leibach, F.H.; Prasad, P.D.; Ganapathy, V. Involvement of transporter recruitment as well as gene expression in the substrate-induced adaptive regulation of amino acid transport system A. Biochim. Biophys. Acta. 2001, 1512, 15–21. [Google Scholar] [CrossRef][Green Version]
- Liao, K.; Lane, M.D. Expression of a novel insulin-activated amino acid transporter gene during differentiation of 3T3-L1 preadipocytes into adipocytes. Biochem. Biophys. Res. Commun. 1995, 208, 1008–1015. [Google Scholar] [CrossRef] [PubMed]
- Bröer, S. Amino acid transporters as modulators of glucose homeostasis. Trends Endocrinol. Metab. 2022, 33, 120–135. [Google Scholar] [CrossRef]
- El-Hattab, A.W. Serine biosynthesis and transport defects. Mol. Genet. Metab. 2016, 118, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Heimer, G.; Marek-Yagel, D.; Eyal, E.; Barel, O.; Oz Levi, D.; Hoffmann, C.; Ruzzo, E.K.; Ganelin-Cohen, E.; Lancet, D.; Pras, E.; et al. SLC1A4 mutations cause a novel disorder of intellectual disability, progressive microcephaly, spasticity and thin corpus callosum. Clin. Genet. 2015, 88, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, H.; Kanai, Y.; Tokunaga, M.; Nakata, T.; Chairoungdua, A.; Ishimine, H.; Tsukada, S.; Ooigawa, H.; Nawashiro, H.; Kobayashi, Y.; et al. High affinity D- and L-serine transporter Asc-1: Cloning and dendritic localization in the rat cerebral and cerebellar cortices. Neurosci. Lett. 2004, 358, 123–126. [Google Scholar] [CrossRef] [PubMed]
- Davis, S.R.; Stacpoole, P.W.; Williamson, J.; Kick, L.S.; Quinlivan, E.P.; Coats, B.S.; Shane, B.; Bailey, L.B.; Gregory, J.F., 3rd. Tracer-derived total and folate-dependent homocysteine remethylation and synthesis rates in humans indicate that serine is the main one-carbon donor. Am. J. Physiol. Endocrinol. Metab. 2004, 286, E272–E279. [Google Scholar] [CrossRef]
- Holeček, M.; Vodeničarovová, M. Effects of histidine supplementation on amino acid metabolism in rats. Physiol. Res. 2020, 69, 99–111. [Google Scholar] [CrossRef]
- Holeček, M.; Vodeničarovová, M. Effects of histidine load on ammonia, amino acid, and adenine nucleotide concentrations in rats. Amino Acids 2019, 51, 1667–1680. [Google Scholar] [CrossRef]
- Hirabayashi, Y.; Furuya, S. Roles of l-serine and sphingolipid synthesis in brain development and neuronal survival. Prog. Lipid Res. 2008, 47, 188–203. [Google Scholar] [CrossRef] [PubMed]
- Esaki, K.; Sayano, T.; Sonoda, C.; Akagi, T.; Suzuki, T.; Ogawa, T.; Okamoto, M.; Yoshikawa, T.; Hirabayashi, Y.; Furuya, S. L-Serine deficiency elicits intracellular accumulation of cytotoxic deoxysphingolipids and lipid body formation. J. Biol. Chem. 2015, 290, 14595–14609. [Google Scholar] [CrossRef] [PubMed]
- Gui, T.; Li, Y.; Zhang, S.; Alecu, I.; Chen, Q.; Zhao, Y.; Hornemann, T.; Kullak-Ublick, G.A.; Gai, Z. Oxidative stress increases 1-deoxysphingolipid levels in chronic kidney disease. Free Radic. Biol. Med. 2021, 164, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Bertea, M.; Rütti, M.F.; Othman, A.; Marti-Jaun, J.; Hersberger, M.; Von Eckardstein, A.; Hornemann, T. Deoxysphingoid bases as plasma markers in diabetes mellitus. Lipids Health Dis. 2010, 9, 84. [Google Scholar] [CrossRef] [PubMed]
- Fridman, V.; Zarini, S.; Sillau, S.; Harrison, K.; Bergman, B.C.; Feldman, E.L.; Reusch, J.E.B.; Callaghan, B.C. Altered plasma serine and 1-deoxydihydroceramide profiles are associated with diabetic neuropathy in type 2 diabetes and obesity. J. Diabetes Complicat. 2021, 35, 107852. [Google Scholar] [CrossRef]
- Birge, R.B.; Boeltz, S.; Kumar, S.; Carlson, J.; Wanderley, J.; Calianese, D.; Barcinski, M.; Brekken, R.A.; Huang, X.; Hutchins, J.T.; et al. Phosphatidylserine is a global immunosuppressive signal in efferocytosis, infectious disease, and cancer. Cell Death Differ. 2016, 23, 962–978. [Google Scholar] [CrossRef]
- Schuiki, I.; Daum, G. Phosphatidylserine decarboxylases, key enzymes of lipid metabolism. IUBMB Life 2009, 61, 151–162. [Google Scholar] [CrossRef]
- Vance, J.E.; Tasseva, G. Formation and function of phosphatidylserine and phosphatidylethanolamine in mammalian cells. Biochim. Biophys. Acta 2013, 1831, 543–554. [Google Scholar] [CrossRef]
- Pepping, J. Phosphatidylserine. Am. J. Health Syst. Pharm. 1999, 56, 2043–2044. [Google Scholar] [CrossRef]
- Ye, L.; Sun, Y.; Jiang, Z.; Wang, G. L-serine, an endogenous amino acid, is a potential neuroprotective agent for neurological disease and injury. Front. Mol. Neurosci. 2021, 14, 726665. [Google Scholar] [CrossRef]
- Maugard, M.; Vigneron, P.A.; Bolaños, J.P.; Bonvento, G. l-Serine links metabolism with neurotransmission. Prog. Neurobiol. 2021, 197, 101896. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Qiang, R.; Yang, Y.; Jiang, Z.L.; Wang, G.H.; Zhao, G.W.; Ren, T.J.; Jiang, R.; Xu, L.H. L-serine treatment may improve neurorestoration of rats after permanent focal cerebral ischemia potentially through improvement of neurorepair. PLoS ONE 2014, 9, e93405. [Google Scholar] [CrossRef]
- Hardingham, G.E.; Bading, H. Synaptic versus extrasynaptic NMDA receptor signalling: Implications for neurodegenerative disorders. Nat. Rev. Neurosci. 2010, 11, 682–696. [Google Scholar] [CrossRef] [PubMed]
- Papouin, T.; Ladépêche, L.; Ruel, J.; Sacchi, S.; Labasque, M.; Hanini, M.; Groc, L.; Pollegioni, L.; Mothet, J.P.; Oliet, S.H.R. Synaptic and extrasynaptic NMDA receptors are gated by different endogenous coagonists. Cell 2012, 150, 633–646. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.F. D-Serine as a glial modulator of nerve cells. Glia 2004, 47, 275–283. [Google Scholar] [CrossRef]
- Mothet, J.P.; Parent, A.T.; Wolosker, H.; Brady, R.O., Jr.; Linden, D.J.; Ferris, C.D.; Rogawski, M.A.; Snyder, S.H. D-serine is an endogenous ligand for the glycine site of the N-methyl-D-aspartate receptor. Proc. Natl. Acad. Sci. USA 2000, 97, 4926–4931. [Google Scholar] [CrossRef]
- Yang, Y.; Ge, W.; Chen, Y.; Zhang, Z.; Shen, W.; Wu, C.; Poo, M.; Duan, S. Contribution of astrocytes to hippocampal long-term potentiation through release of D-serine. Proc. Natl. Acad. Sci. USA 2003, 100, 15194–15199. [Google Scholar] [CrossRef]
- Montesinos Guevara, C.; Mani, A.R. The role of D-serine in peripheral tissues. Eur. J. Pharmacol. 2016, 780, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Heresco-Levy, U.; Javitt, D.C.; Ebstein, R.; Vass, A.; Lichtenberg, P.; Bar, G.; Catinari, S.; Ermilov, M. D-serine efficacy as add-on pharmacotherapy to risperidone and olanzapine for treatment-refractory schizophrenia. Biol. Psychiatry 2005, 57, 577–585. [Google Scholar] [CrossRef]
- Goh, K.K.; Wu, T.H.; Chen, C.H.; Lu, M.L. Efficacy of N-methyl-D-aspartate receptor modulator augmentation in schizophrenia: A meta-analysis of randomised, placebo-controlled trials. J. Psychopharmacol. 2021, 35, 236–252. [Google Scholar] [CrossRef]
- Tabatabaie, L.; Klomp, L.W.; Berger, R.; De Koning, T.J. L-serine synthesis in the central nervous system: A review on serine deficiency disorders. Mol. Genet. Metab. 2010, 99, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Glade, M.J.; Smith, K. Phosphatidylserine and the human brain. Nutrition 2015, 31, 781–786. [Google Scholar] [CrossRef] [PubMed]
- Acuna-Hidalgo, R.; Schanze, D.; Kariminejad, A.; Nordgren, A.; Kariminejad, M.H.; Conner, P.; Grigelioniene, G.; Nilsson, D.; Nordenskjöld, M.; Wedell, A.; et al. Neu-Laxova syndrome is a heterogeneous metabolic disorder caused by defects in enzymes of the L-serine biosynthesis pathway. Am. J. Hum. Genet. 2014, 95, 285–293. [Google Scholar] [CrossRef] [PubMed]
- De Koning, T.J.; Klomp, L.W.; Van Oppen, A.C.; Beemer, F.A.; Dorland, L.; Van den Berg, I.; Berger, R. Prenatal and early postnatal treatment in 3-phosphoglycerate-dehydrogenase deficiency. Lancet 2004, 364, 2221–2222. [Google Scholar] [CrossRef]
- Hart, C.E.; Race, V.; Achouri, Y.; Wiame, E.; Sharrard, M.; Olpin, S.E.; Watkinson, J.; Bonham, J.R.; Jaeken, J.; Matthijs, G.; et al. Phosphoserine aminotransferase deficiency: A novel disorder of the serine biosynthesis pathway. Am. J. Hum. Genet. 2007, 80, 931–937. [Google Scholar] [CrossRef]
- Garofalo, K.; Penno, A.; Schmidt, B.P.; Lee, H.J.; Frosch, M.P.; Von Eckardstein, A.; Brown, R.H.; Hornemann, T.; Eichler, F.S. Oral L-serine supplementation reduces production of neurotoxic deoxysphingolipids in mice and humans with hereditary sensory autonomic neuropathy type 1. J. Clin. Investig. 2011, 121, 4735–4745. [Google Scholar] [CrossRef]
- Le Douce, J.; Maugard, M.; Veran, J.; Matos, M.; Jégo, P.; Vigneron, P.A.; Faivre, E.; Toussay, X.; Vandenberghe, M.; Balbastre, Y.; et al. Impairment of glycolysis-derived l-serine production in astrocytes contributes to cognitive deficits in Alzheimer’s disease. Cell Metab. 2020, 31, 503–517. [Google Scholar] [CrossRef]
- Holeček, M.; Vodeničarovová, M.; Fingrová, R. Dual effects of beta-hydroxy-beta-methylbutyrate (HMB) on amino acid, energy, and protein metabolism in the liver and muscles of rats with streptozotocin-induced type 1 diabetes. Biomolecules 2020, 10, 1475. [Google Scholar] [CrossRef]
- Scharff, R.; Wool, I.G. Effect of diabetes on the concentration of amino acids in plasma and heart muscle of rats. Biochem. J. 1966, 99, 173–178. [Google Scholar] [CrossRef]
- Bervoets, L.; Massa, G.; Guedens, W.; Louis, E.; Noben, J.P.; Adriaensens, P. Metabolic profiling of type 1 diabetes mellitus in children and adolescents: A case-control study. Diabetol. Metab. Syndr. 2017, 9, 48. [Google Scholar] [CrossRef]
- Drábková, P.; Šanderová, J.; Kovařík, J.; Kanďár, R. An assay of selected serum amino acids in patients with type 2 diabetes mellitus. Adv. Clin. Exp. Med. 2015, 24, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Kamaura, M.; Nishijima, K.; Takahashi, M.; Ando, T.; Mizushima, S.; Tochikubo, O. Lifestyle modification in metabolic syndrome and associated changes in plasma amino acid profiles. Circ. J. 2010, 74, 2434–2440. [Google Scholar] [CrossRef] [PubMed]
- Mook-Kanamori, D.O.; De Mutsert, R.; Rensen, P.C.; Prehn, C.; Adamski, J.; Den Heijer, M.; Le Cessie, S.; Suhre, K.; Rosendaal, F.R.; Van Dijk, K.W. Type 2 diabetes is associated with postprandial amino acid measures. Arch. Biochem. Biophys. 2016, 589, 138–144. [Google Scholar] [CrossRef]
- Enquobahrie, D.A.; Denis, M.; Tadesse, M.G.; Gelaye, B.; Ressom, H.W.; Williams, M.A. Maternal early pregnancy serum metabolites and risk of gestational diabetes mellitus. J. Clin. Endocrinol. Metab. 2015, 100, 4348–4356. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Hornemann, T.; Štefanić, S.; Schraner, E.M.; Zuellig, R.; Reding, T.; Malagola, E.; Henstridge, D.C.; Hills, A.P.; Graf, R.; et al. Serine administration as a novel prophylactic approach to reduce the severity of acute pancreatitis during diabetes in mice. Diabetologia 2020, 63, 1885–1899. [Google Scholar] [CrossRef] [PubMed]
- Holecek, M.; Sprongl, L.; Tilser, I.; Tichý, M. Leucine and protein metabolism in rats with chronic renal insufficiency. Exp. Toxicol. Pathol. 2001, 53, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Suvanapha, R.; Tungsanga, K.; Laorpatanaskul, S.; Sitprija, V.; Suwan, S. Plasma amino acid patterns in normal Thais and in patients with chronic renal failure. J. Med. Assoc. Thail. 1991, 74, 211–217. [Google Scholar]
- Canepa, A.; Divino Filho, J.C.; Forsberg, A.M.; Perfumo, F.; Carrea, A.; Gusmano, R.; Bergström, J. Nutritional status and muscle amino acids in children with end-stage renal failure. Kidney Int. 1992, 41, 1016–1022. [Google Scholar] [CrossRef][Green Version]
- Ceballos, I.; Chauveau, P.; Guerin, V.; Bardet, J.; Parvy, P.; Kamoun, P.; Jungers, P. Early alterations of plasma free amino acids in chronic renal failure. Clin. Chim. Acta 1990, 188, 101–108. [Google Scholar] [CrossRef]
- McDonald, S.P.; Whiting, M.J.; Tallis, G.A.; Barbara, J.A. Relationships between homocysteine and related amino acids in chronic hemodialysis patients. Clin. Nephrol. 2001, 55, 465–470. [Google Scholar]
- Van Guldener, C.; Stehouwer, C.D. Homocysteine and methionine metabolism in renal failure. Semin. Vasc. Med. 2005, 5, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Hamed, S.A. Neurologic conditions and disorders of uremic syndrome of chronic kidney disease: Presentations, causes, and treatment strategies. Expert Rev. Clin. Pharmacol. 2019, 12, 61–90. [Google Scholar] [CrossRef] [PubMed]
- Kimura, T.; Hesaka, A.; Isaka, Y. D-Amino acids and kidney diseases. Clin. Exp. Nephrol. 2020, 24, 404–410. [Google Scholar] [CrossRef] [PubMed]
- Ganote, C.E.; Peterson, D.R.; Carone, F.A. The nature of D-serine-induced nephrotoxicity. Am. J. Pathol. 1974, 77, 269–282. [Google Scholar]
- Maekawa, M.; Okamura, T.; Kasai, N.; Hori, Y.; Summer, K.H.; Konno, R. D-amino-acid oxidase is involved in D-serine-induced nephrotoxicity. Chem. Res. Toxicol. 2005, 18, 1678–1682. [Google Scholar] [CrossRef]
- Lin, C.S.; Hung, S.F.; Huang, H.S.; Ma, M.C. Blockade of the N-methyl-D-aspartate glutamate receptor ameliorates lipopolysaccharide-induced renal insufficiency. PLoS ONE 2015, 10, e0132204. [Google Scholar] [CrossRef]
- Snell, K.; Natsumeda, Y.; Eble, J.N.; Glover, J.L.; Weber, G. Enzymic imbalance in serine metabolism in human colon carcinoma and rat sarcoma. Br. J. Cancer 1988, 57, 87–90. [Google Scholar] [CrossRef]
- Pan, S.; Fan, M.; Liu, Z.; Li, X.; Wang, H. Serine, glycine and one-carbon metabolism in cancer (Review). Int. J. Oncol. 2021, 58, 158–170. [Google Scholar] [CrossRef]
- Possemato, R.; Marks, K.M.; Shaul, Y.D.; Pacold, M.E.; Kim, D.; Birsoy, K.; Sethumadhavan, S.; Woo, H.K.; Jang, H.G.; Jha, A.K.; et al. Functional genomics reveal that the serine synthesis pathway is essential in breast cancer. Nature 2011, 476, 346–350. [Google Scholar] [CrossRef]
- Xie, M.; Pei, D.S. Serine hydroxymethyltransferase 2: A novel target for human cancer therapy. Investig. New Drugs 2021, 39, 1671–1681. [Google Scholar] [CrossRef]
- Maddocks, O.D.K.; Athineos, D.; Cheung, E.C.; Lee, P.; Zhang, T.; Van den Broek, N.J.F.; Mackay, G.M.; Labuschagne, C.F.; Gay, D.; Kruiswijk, F.; et al. Modulating the therapeutic response of tumours to dietary serine and glycine starvation. Nature 2017, 544, 372–376. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Li, B.; He, W.; Huang, C. Dietary serine supplementation: Friend or foe? Curr. Opin. Pharmacol. 2021, 61, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Stead, L.M.; Brosnan, M.E.; Brosnan, J.T. Characterization of homocysteine metabolism in the rat liver. Biochem. J. 2000, 350, 685–692. [Google Scholar] [CrossRef] [PubMed]
- Benevenga, N.J.; Harper, A.E. Effect of glycine and serine on methionine metabolism in rats fed diets high in methionine. J. Nutr. 1970, 100, 1205–1214. [Google Scholar] [CrossRef] [PubMed]
- Fukada, S.; Shimada, Y.; Morita, T.; Sugiyama, K. Suppression of methionine-induced hyperhomocysteinemia by glycine and serine in rats. Biosci. Biotechnol. Biochem. 2006, 70, 2403–2409. [Google Scholar] [CrossRef]
- Verhoef, P.; Steenge, G.R.; Boelsma, E.; Van Vliet, T.; Olthof, M.R.; Katan, M.B. Dietary serine and cystine attenuate the homocysteine-raising effect of dietary methionine: A randomized crossover trial in humans. Am. J. Clin. Nutr. 2004, 80, 674–679. [Google Scholar] [CrossRef]
- Sim, W.C.; Yin, H.Q.; Choi, H.S.; Choi, Y.J.; Kwak, H.C.; Kim, S.K.; Lee, B.H. L-serine supplementation attenuates alcoholic fatty liver by enhancing homocysteine metabolism in mice and rats. J. Nutr. 2015, 145, 260–267. [Google Scholar] [CrossRef]
- Cox, P.A.; Davis, D.A.; Mash, D.C.; Metcalf, J.S.; Banack, S.A. Dietary exposure to an environmental toxin triggers neurofibrillary tangles and amyloid deposits in the brain. Proc. Biol. Sci. 2016, 283, 20152397. [Google Scholar] [CrossRef]
- Soto, D.; Olivella, M.; Grau, C.; Armstrong, J.; Alcon, C.; Gasull, X.; Santos-Gómez, A.; Locubiche, S.; Gómez de Salazar, M.; García-Díaz, R.; et al. L-Serine dietary supplementation is associated with clinical improvement of loss-of-function GRIN2B-related pediatric encephalopathy. Sci. Signal. 2019, 12, eaaw0936. [Google Scholar] [CrossRef]
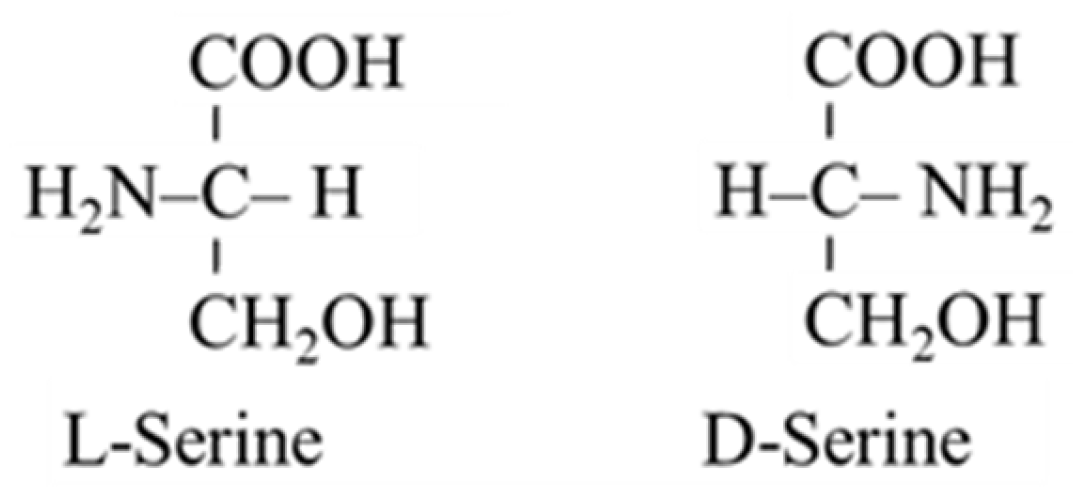

| Disease | Reference(s) |
|---|---|
| Primary disorders of serine synthesis | [6,7,54,55] |
| Hereditary sensory neuropathy type 1 | [2,56] |
| Amyotrophic lateral sclerosis | [8,88] |
| Encephalopathy due to mutations of NMDA receptor | [89] |
| Diabetes | [4,9] |
| Hyperhomocysteinemia | [86] |
| Chronic renal injury | None |
| CNS injury | None |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Holeček, M. Serine Metabolism in Health and Disease and as a Conditionally Essential Amino Acid. Nutrients 2022, 14, 1987. https://doi.org/10.3390/nu14091987
Holeček M. Serine Metabolism in Health and Disease and as a Conditionally Essential Amino Acid. Nutrients. 2022; 14(9):1987. https://doi.org/10.3390/nu14091987
Chicago/Turabian StyleHoleček, Milan. 2022. "Serine Metabolism in Health and Disease and as a Conditionally Essential Amino Acid" Nutrients 14, no. 9: 1987. https://doi.org/10.3390/nu14091987
APA StyleHoleček, M. (2022). Serine Metabolism in Health and Disease and as a Conditionally Essential Amino Acid. Nutrients, 14(9), 1987. https://doi.org/10.3390/nu14091987






