Disgust and Self-Disgust in Eating Disorders: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria for Systematic Review
2.2. Eligibility Criteria for Meta-Analysis
2.3. Information Sources
2.4. Literature Search
2.5. Study Selection and Data Extraction
3. Synthesis of Results
3.1. Meta-Analyses
3.2. Sensitivity Analyses
4. Quality Assessment
5. Results
5.1. Study Selection
5.2. Systematic Review
5.2.1. Methods to Investigate Disgust and Self-Disgust
Questionnaires
Stimuli to Trigger Disgust
Cognitive Experimental Tasks
Brain imaging and Neurophysiology
Qualitative Studies
5.2.2. Findings of Qualitative Studies
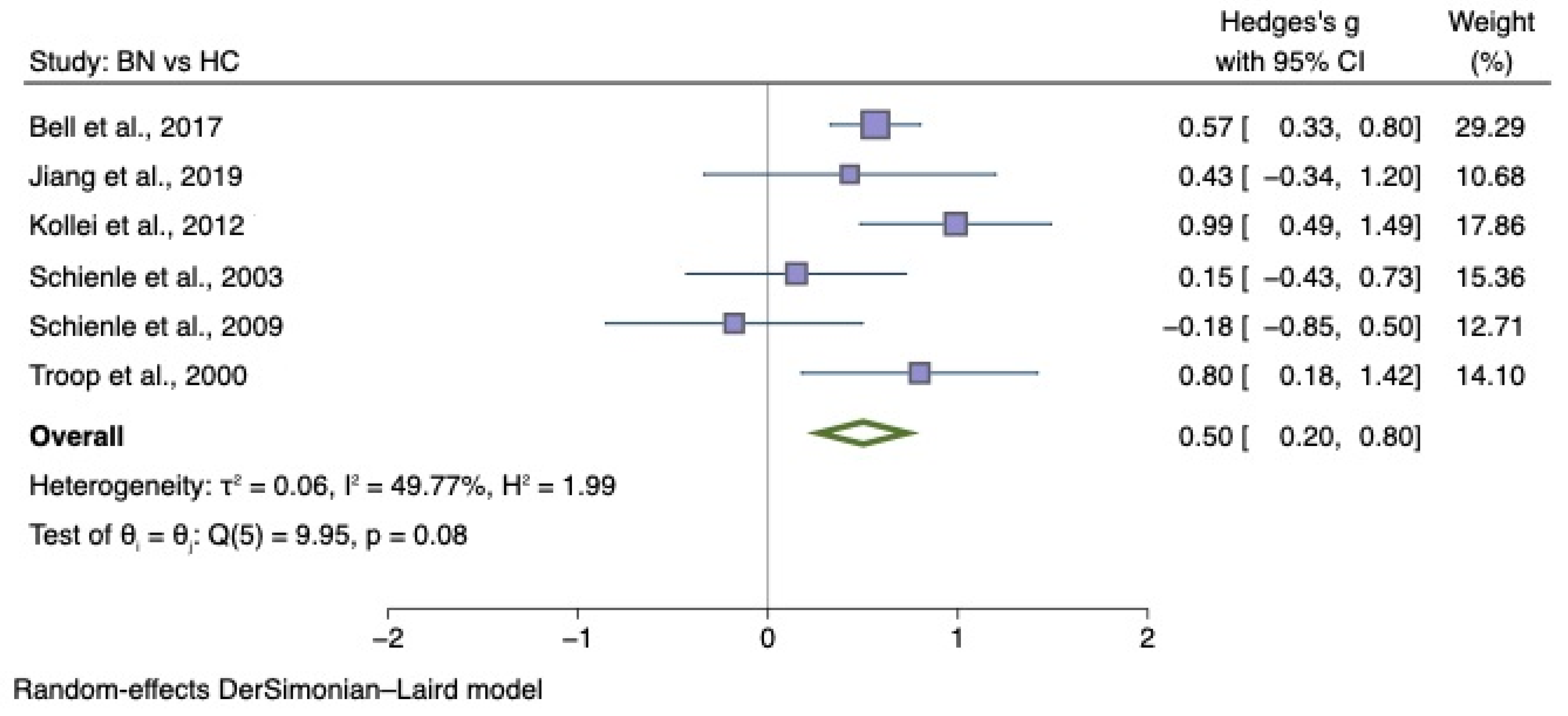
5.3. Meta-Analysis
5.3.1. Disgust Elicited by Food Images
5.3.2. Generic Disgust Sensitivity
| Author (Year) Country | Gender (n) | Sample Size | Age M (SD) | Study Design | Method | Main Findings | Effect Size (Cohen’s d) of Main Findings | ||
|---|---|---|---|---|---|---|---|---|---|
| Clinical | Control | Clinical | Control | ||||||
| Disgust | |||||||||
| Marzola et al. (2020) [37] * Italy | F adults | AN (33): | 39 | 26.2 (10.3) | 23.92 (2.7) | Case-control | DS | -The minimum difference between AN and HC on the baseline disgust sensitivity level. | d = −0.16 |
| -The minimum correlation between disgust sensitivity and eating psychopathology in AN. | d = −0.09 | ||||||||
| Kot et al. (2021) [38] * Poland | F adults | AN-R (29) AN-BP (34) | 57 | 25.73 (5.99) | 25.21 (5.60) | Case-control | DS-R | -The level of disgust sensitivity of AN patients was greater than HC. | d = 0.41 |
| -The minimum correlation between self-disgust and overall disgust sensitivity in AN. | d = −0.14 | ||||||||
| Aharoni & Hertz (2012) [35] * Denmark | F adults | AN-R (37) AN-BP (25) | 62 | General: 27.77 (6.74) | Case-control | DSQ | -The scores of overall disgust sensitivity (DS) and specific sub-scales (i.e., food, magical thinking, body products) were higher among AN than HC. | d for Overall DS = 0.76 Magical thinking = 0.84 Food = 0.94 Animal = 0.34 Body products = 0.59 Sex = 0.30 Body envelope violations = 0.11 Death = 0.13 Hygiene = 0.43 | |
| Troop et al. (2000) [34] * UK | F (82) M (7) adults | AN-R (16) AN-BP (12) BN (33) EDNOS (7) Binge eater (6) | 15 | AN-R: 21.9 (5.1) AN-BP: 29.2 (8.9) BN: 29.7 (7.5) EDNOS: 21.9 (4.2) Binge eater: 30.5 (7.8) | 28.7 (7.3) | Case-control | DSQ | -The minimum difference among groups on overall disgust sensitivity. | d = 0.01 |
| -Drive for thinness was positively correlated with disgust sensitivity to food and magical contagion, but it was marginally associated with overall disgust sensitivity level. | d for Food = 0.77 Magical contagion = 0.52 Overall DS = 0.41 | ||||||||
| -Bulimia symptoms were positively associated with disgust sensitivity to animals, death, body envelope violations and magical contagion, but it is marginally correlated with overall disgust sensitivity level. | d for Animal = 0.52 Death = 0.56 Body envelope violations = 0.56 Magical contagion = 0.61 Overall DS = 0.41 | ||||||||
| Troop et al. (2002) [40] UK | F adults | Remission Group (57): AN-R (12) AN-BP (31) BN (10) EDNOS (4) | 215 | General: 31.6 (10.01) | NI | Case-control | DQ | -Further analysis of a previous study by Troop et al. (2000) [34] showed that the ED group had a higher overall disgust sensitivity and disgust sensitivity to food, animal, and body products than HC. | d for Overall DS = 0.84 Food = 0.81 Animal = 0.85 Body products = 0.93 |
| Clinical Group (148): AN-R (36) AN-BP (30) BN (38) EDNOS (44) | -Both ED groups (remitted and clinical) reported a higher level of disgust towards the human body and body products and foodstuffs of animal origin than the other three domains (invertebrate animals, gastro-enteric, sexual practices). | N/A | |||||||
| Davey et al. (1998) [39] UK | F adolescents | AN (10) | Student girls (27) | 16.1 | 15.2 | Case-control | DQ | -The scores of patients with AN were higher in three sub-scales (foodstuffs of animal origin, human body and body products, gastro-enteric products) than HC. | d for Foodstuffs of animal origin = 1.20 Human body and body products = 0.83 Invertebrate animals = 0.02 Gastro-enteric products = 1.16 Sexual practices= 0.50 |
| Jiang et al. (2019) [32] * France | F adults | AN-R (14) BN (13) | 12 | AN-R: 24.94 (4.67) BN: 22.50 (2.88) | 24.14 (3.06) | Case-control | DPSS | -AN-R patients reported higher levels of overall disgust sensitivity than HC participants. | d = 1.20 |
| Bell et al. (2017) [33] * UK | F adults | AN (270) BN (104) | 217 | General: 25.36 (9.67) | Case-control | DPSS-R | -The minimum correlation between self-disgust and overall disgust sensitivity for people with EDs. | d for Overall DS = 0.19 | |
| Schienle (2003) [30] * Germany | F (214) M (136) adults | BN (13) | 150 | BN: 26.0 (8.4) | 40.2 (11.0) | Case-control | QADS | -The minimum difference between female BN and female HC on the disgust sensitivity level. | d = −0.15 |
| BN group was entirely female | Other psychiatric disorders (187) | -The minimum difference between female BN and female other psychiatric disorders on disgust sensitivity level. | N/A | ||||||
| Schienle et al. (2017) [31] Germany | F adults | Patients with binge-eating symptoms (36) | 38 | NI | Experimental | QADP | -The minimum difference between ED and HC on baseline disgust proneness levels in bitter and neutral conditions. | d for bitter condition ED vs. HC = −0.27 neutral condition ED vs. HC = −0.19 | |
| -The minimum difference between bitter and neutral conditions for people with ED and HC. | ED bitter vs. neutral = 0.10 HC bitter vs. neutral = 0.16 | ||||||||
| Fox and Froom (2009) [54] UK | F adults | Individuals were recruited from the BEAT database (52) | N/A | 31.74 (10.06) | N/A | Cross-sectional | BES ** | -The positive and large correlation between ED symptomatology and state disgust. | d = 1.50 |
| -This association stayed large with depression and anxiety scores partialled out of the analysis. | d (depression) = 0.97 d (anxiety) = 1.05 | ||||||||
| -After accounting for state sadness and anger within the regression model, this association disappeared. | N/A | ||||||||
| Fox et al. (2013) [36] * UK | F adults | AN (22) | 19 | 23.70 (4.20) | 23.38 (3.03) | First stage: cross-sectional Second stage: case-control | First stage: BES ** Second stage: DS-R ** | -State disgust was positively correlated with negative self-belief. | d = 1.61 |
| -State disgust was negatively correlated with positive self-belief. | d = −1.06 | ||||||||
| The positive and large correlation between state disgust and body size/shape estimation. | d = 1.02 | ||||||||
| -Following anger induction, the AN group reported more elevated disgust than HC. | d = 0.93 | ||||||||
| Kollei et al. (2012) [27] * Germany | F (105) M (25) adults | AN (32) BN (34) BDD (31) | 33 | AN: 26.94 (9.15) BN: 25.94 (8.25) BDD: 28.77 (8.91) | 26.91 (8.48) | Case-control | DES; DES-Body | -AN and BN patients reported a higher level of overall disgust and disgust towards the body than HC. | d for AN vs. HC = 1.05 BN vs. HC = 1.0 |
| -The minimum difference between BDD and EDs patients (AN and BN) on emotional experiences (overall disgust and disgust towards body). | d for BDD vs. AN = 0.14 BDD vs. BN = 0.11 | ||||||||
| Zeeck et al. (2011) [26] Germany | F adults | BED and Obesity (20) Obesity without BED (23) | NW (20) | BED and Obesity: 39.3 (12.7) Obesity without BED: 45.4 (11.3) | 39.7 (11.6) | Case-control | DAS | -The feeling of disgust was one of the strongest emotional experiences aggravating the association between a desire to eat and binge eating. | d = 1.74 |
| Bornholt et al. (2005) [28] Australia | F adolescents | AN (28) | Schoolgirls (113) -Low BMI -Low–medium BMI -Medium–high BMI -High BMI | 14.9 (1.8) | 13.5 (1.5) | Case-control | 5-point Likert Scale *** -Item: yuk, sick, disgust | -Adolescents with AN reported more disgust feelings about their bodies than schoolgirls with low BMI groups. | d = 0.80 |
| -The minimum correlation between self-concepts and disgust feelings about the body among individuals with AN. | d = 0.26 | ||||||||
| Cooper et al. (1988) [41] US | F adults | Patients with bulimia (binge–purge cycle): -with depression -without depression | N/A | 25.6 | N/A | Cross-sectional | The Diagnostic Survey for EDs contained a brief adjective checklist to determine emotions experienced by participants during a binge (phase 1), after a binge (phase 2) and after purging (phase 3), retrospectively. | -Factor 1, indicating feeling of guilty, disgusted, and angry, was at the highest level compared to the other three factors in the period between binge and purge. Following the purge, this level decreased by reaching the same level reported in phase 1. | d Between Phase 1 and Phase 2 for depressed people: 0.69 for non-depressed people: 0.86 Between Phase 2 and Phase 3: for depressed people: −0.86 for non-depressed people: −0.60 Between Phase 1 and Phase 3: for depressed people: −0.15 for non-depressed people: 0.23 |
| -Disgust was not measured separately: Factor 1: feeling of guilty, disgusted, angry Factor 2: energized, excited Factor 3: secure, relieved Factor 4: panicked, helpless, not calm | -The minimum difference among bulimic patients with and without depression in Factor 1 level during the binge-purge cycle. | d for difference between depressed and non-depressed people -at phase 1: 0.23 -at phase 2: −0.10 -at phase 3: 0.15 | |||||||
| Richson et al. (2020) [42] US | F (177) M (36) Transgender (2) | AN-BP (13) BN (103) BED (14) OSFED sub-BN (73) OSFED sub-BED (16) | N/A | General: 24.73 (9.12) | Cross-sectional | EPSI-CRV used to measure Criterion B symptoms for BED | - Feeling disgusted/depressed/guilty was not a predictor of binge-eating severity. | d = 0.27 | |
| -Item: feeling disgusted/depressed/guilty | |||||||||
| Buvat-Herbaut et al. (1983) [29] France | Young F | AN emaciation state (54) AN weight restoration (27) | Schoolgirls (288) | Range: 14–27 | Range: 12–26 | Case-control | Administration of the Questionnaire Disgust-relevant Items: -I am disgusted at being pregnant -Sexuality disgusts me | -Young females with AN reported more disgust feelings about pregnancy than HC. | N/A |
| -The proportion of AN patients (at the phase of weight restoration, 28% and at the phase of emaciation, 33.3%) who were disgusted by the idea of an enlarged stomach during pregnancy was higher than that of HC (20.6%). | N/A | ||||||||
| -The proportion of AN patients (at the phase of weight restoration, 37.5%) who were disgusted by sexuality was higher than the proportions of AN patients in the acute phase (at the phase of emaciation, 18.4%) and HC (14%). | N/A | ||||||||
| Kockler et al. (2017) [55] Germany | F adults | BN (20) PTSD (28) BPD (43) | 28 | BN: 23.70 (5.97) PTSD: 35.25 (7.53) BPD: 26.72 (7.07) | 28.82 (7.47) | Case-control | DialogPad E-Diary Software measures emotion sequences in 4 categories: activation, persistence, switch, down-regulation | -BN patients experienced a most frequent change from anger to disgust than BPD, PTSD, and HC. | d for BN vs. BPD = 0.42 BN vs. PTSD = 0.47 BN vs. HC = 0.52 |
| -The most frequent switch was from disgust to an unspecific emotion for patients with BN relative to those with BPD and HC. | d for BN vs. BPD = 0.46 BN vs. HC = 0.56 | ||||||||
| Self-Disgust | |||||||||
| Bell et al. (2017) [33] * UK | F adults | AN (270) BN (104) | 217 | General: 25.36 (9.67) | Case-control | SDS | -ED group reported a higher level of self-disgust than HC. | d = 1.19 d for Anxiety = 0.36 Low registration = 0.35 Sensation seeking = −0.23 | |
| -Self-disgust was positively associated with anxiety symptoms, low registration and negatively correlated with sensation seeking among the AN group. | |||||||||
| -Self-disgust was positively associated with anxiety symptoms, sensation avoidance, and sensation seeking among the BN group. | d for Anxiety = 0.30 Low registration = 0.37 Sensation seeking = −0.26 | ||||||||
| Ille et al. (2014) [44] * Austria | F (93) M (19) adults ED group is entirely female | Clinical sample consisted of AN (16) and BN (24) | 112 | No ED-specific age details | 31.10 (13.0) | Case-control | QASD | -Individuals diagnosed with EDs reported higher personal and behavioural disgust than HC. | d for personal disgust = 1.68 behavioural disgust = 1.59 |
| -For EDs patients, whereas interpersonal sensitivity, depression, and obsession were predictors for personal disgust (corrected R2 = 0.70), the best predictor of behavioural disgust was anxiety (corrected R2 = 0.26). | N/A | ||||||||
| Kot et al. (2021) [38] * Poland | F adults | AN-R (29) AN-BP (34) | 57 | 25.73 (5.99) | 25.21 (5.60) | Case-control | SDS Female patients with AN aged between 18 and 45 years | -The level of self-disgust among patients with AN was greater than in HC. | d for overall self-disgust = 0.41 |
| -The minimum correlation between self-disgust and overall disgust sensitivity in AN and HC. | d for AN = −0.14 HC = 0.26 | ||||||||
| -Self-disgust predicted the severity of EDs characteristics. | N/A | ||||||||
| -Self-disgust mediated the associations between ED characteristics and depressive symptoms and trait anxiety in AN and HC. | N/A | ||||||||
| Marques et al. (2021) [43] * Portugal | F adults | 62 | 119 | 32.16 (13.19) | 22.45 (3.50) | Case-control | MSDS | -In comparison with the community sample, ED patients reported higher levels of self-disgust. | d = 1.71 |
| -Self-disgust was positively correlated with a drive for thinness and external shame and negatively correlated with self-compassion level. | d for Drive for thinness = 0.77 External shame = 1.25 Self-compassion = −1.25 | ||||||||
| -Self-compassion played a moderator role (b = − 0.24; p = 0.033) in the relationship between self-disgust and ED symptomatology. | N/A | ||||||||
| Author (Year) Country | Gender (n) | Sample Size | Age M (SD) | Study Design | Method | Main Findings | Effect Size (Cohen’s d) of Main Findings | ||
|---|---|---|---|---|---|---|---|---|---|
| Clinical | Control | Clinical | Control | ||||||
| Uher et al. (2005) [53] * UK | F adults | BN (9) AN (13) | 18 | BN: 29.6 (9.3) AN: 25.4 (10.2) | 26.6 (8.6) | Case-control | Numeric Analogue Scale of 1–7 for disgust and fear to visual stimuli: underweight, normal, and overweight female bodies in swimming costumes | -Each body shape category was more aversive to ED patients than it was to HC. This effect was more marked in AN than in BN. | d for Underweight = 0.99 Normal weight = 1.30 Overweight = 2.13 |
| -The most aversive body shape category was underweight for HC, while it was overweight for ED group. | N/A | ||||||||
| -AN patients reported more aversion to normal-weight bodies compared to BN and HC. | N/A | ||||||||
| Schienle et al. (2017) [31] Germany | F adults | Patients with binge-eating symptoms (36) | 38 | NI | Experimental | 9-point Likert Scale | -Aftertaste ratings showed wormwood was perceived as more disgusting than water by each participant. | d = −6.32 | |
| visual stimuli (food pictures) and gustatory fluid stimuli, including water (neutral tastant) and wormwood (bitter/aversive tastant) | |||||||||
| Marzola et al. (2020) [37] ** Italy | F adults | AN-R (21) AN-BP (12) | 39 | 26.2 (10.3) | 23.92 (2.7) | Case-control | VAS (disgust towards gustatory stimuli: supplement) | -The supplement induced more food-related disgust than the juice in patients with AN in comparison with HC. | d = 0.51 |
| Joos et al. (2012) [51] Germany | F adults | AN (23) BN (29) Depression (35) | 25 | AN: 24.0 (4.7) BN: 26.2 (6.3) Depression: 27.6 (5.7) | 27.4 (5.5) | Case-control | VAS (disgust towards visual stimuli: clear or blended emotional facial expressions) | -Medium difference among groups (EDs vs. HC; EDs vs. Depression) on disgust responses to angry facial stimuli. | d for EDs vs. HC = 0.40 ED vs. Depression = 0.38 |
| Joos et al. (2009) [48] ** Germany | F adults | AN-R (15) BN (19) | 25 | AN-R: 25.0 (4.5) BN: 25.4 (6.4) | 27.4 (5.5) | Case-control | VAS (disgust towards visual stimuli: facial expressions of different clear or blended emotions) | -The small difference between AN-R and BN on disgust response towards angry facial expressions. | d = 0.49 |
| -The moderate difference between AN-R and HC on disgust response towards angry facial expressions. | d = 0.68 | ||||||||
| -Following depression score covariation, AN-R patients reported elevated disgust levels towards angry facial expressions compared to BN and HC (df =1, t =22.58, p = 0.013). | N/A | ||||||||
| Hay and Katsikitis (2014) [46] ** Australia | F adults | 26 | Psychiatric control (PC: 20) HC (61) | 26.1 (8.3) | PC: 30.9 (10.9) HC: 26.1 (7.7) | Case-control | VAS (disgust towards visual stimuli: food and non-food pictures shown) | -The disgust responses to food images in the ED group were higher than ones in either control group. | d for ED vs. PC = 1.205 ED vs. HC = 1.337 |
| Uher et al. (2004) [52] ** UK | F adults | AN (16) BN (10) | 19 | AN: 26.93 (12.14) BN: 29.80 (8.80) | 26.6 (8.34) | Case-control | VAS (disgust towards visual stimuli: food vs. non-food images; aversive vs. neutral) | -ED patients reported a higher level of disgust towards food stimuli than HC. | d = 1.66 |
| Joos et al. (2011a) [49] ** Germany | F adults | AN-R (11) | 11 | 25.0 (5.0) | 26.0 (5.2) | Case-control | VAS (disgust towards visual stimuli: food or non-food pictures) | -AN-R patients reported a higher level of disgust towards food photographs than HC. | d = 1.975 |
| - AN-R patients’ disgust levels increased when viewing high-calorie food photographs. | N/A | ||||||||
| Joos et al. (2011b) [50] ** Germany | F adults | Medication-free BN (13) | 13 | 25.2 (5.1) | 27.0 (6.0) | Case-control | VAS (disgust towards visual stimuli: food or non-food pictures) | - BN patients reported a higher level of disgust towards food photographs than HC. | d = 0.67 |
| Horndasch et al. (2012) [47] Germany | F adolescents | AN (13) | Typically developing girls (18) | 15.7 (1.8) | 16.6 (1.8) | Case-control | VAS (disgust towards visual stimuli: underweight, normal, and overweight female body pictures) | -Both groups reported higher levels of disgust towards underweight and overweight body pictures compared to normal ones. | d for Normal vs. under-weight in AN = 1.25 Normal vs. over-weight in AN = 1.55 Normal vs. under-weight in HC = 1.51 Normal vs. over-weight in AN = 1.69 |
| Schienle et al. (2004) [57] Germany | F adults | BN (11) | 12 | 25.4 (9.0) | 26.3 (6.4) | Case-control | VAS (disgust ratings for visual stimuli: disgust- vs. fear-inducing vs. neutral pictures) | -BN patients found disgust-inducing pictures as highly repulsive as fear-inducing ones. | d = 1.35 |
| Gagnon et al. (2018) [56] Canada | F adults | AN-R (5) AN-BP (5) BN (13) | 23 | General: 30.35 (11.31) | 25.91 (5.86) | Case-control | Temporal Bisection Task as a time perception task during visual stimuli: disgusting vs. joyful vs. neutral food pictures | -AN patients tended to perceive the duration of disgusting food pictures longer than those with BN. | d = 0.99 |
| -AN patients tended to perceive the duration of disgusting food pictures longer than neutral ones. | d = 0.53 | ||||||||
| Foroughi et al. (2020) [45] ** Australia | F adults | AN (61) AAN (26) BN (39) BED (13) | 41 | AN: 16.47 (2.08) AAN: M = 18.92 (7.53) BN: 25.20 (10.26) BED: 31.38 (15.13) | 21.02 (8.02) | Case-control | VAS (disgust towards visual stimuli: disgust-eliciting food and non-food images shown by PowerPoint slides) | -Disgust ratings towards food images were higher in individuals with ED than HC but did not differ between ED groups. | d for HC vs. EDs = 1.52 AN vs. AAN vs. BN vs. BED = 0.53 |
| AN and AAN (180) -pre-treatment -post weight-gain | N/A | N/A | Longitudinal | -Following weight gain, the disgust of AN and AAN patients towards food images declined, but it remained higher than HC. | d = −0.78 | ||||
| Author (Year) Country | Gender (n) | Sample Size | Age M (SD) | Study Design | Method | Main Findings | Effect Size (Cohen’s d) of Main Findings | ||
|---|---|---|---|---|---|---|---|---|---|
| Clinical | Control | Clinical | Control | ||||||
| Pollatos et al. (2008) [65] Germany | F adults | AN-R (12) | 12 | 23.86 (4.25) | 22.39 (4.78) | Case-control | Emotional Face Recognition Task (six emotional faces from KDEF by Lundqvist et al. 1998) [64] | -AN patients had lower recognition for disgusted faces compared to HC. | N/A * |
| Lule et al. (2014) [66] Switzerland | F adolescents | AN (15) | 15 | 16.2 (1.26) | 16.5 (1.09) | Case-control | FEEL Test (facial emotion recognition from JACFEE by Matsumoto and Ekman, 1988) [61] | -AN group tended to recognize disgust with less accuracy than HC (F = 3.39 p = 0.08), but this moderate difference disappeared with the correction of the depression score. | d = 0.70 |
| -Negative correlation between disgust recognition ability and the psychological characteristics “perfectionism” and “trait anxiety”. | d for Perfectionism = −1.09 Trait anxiety = −0.72 | ||||||||
| Wyssen et al. (2019) [67] Switzerland and Germany | F adults | AN (61) BN (58) A mixed group in which patients with depression (36) Anxiety (23) were found | 130 | AN: 22.87 (4.57) BN: 23.16 (3.96) Mixed: 25.92 (4.79) | 21.53 (2.18) | Case-control | A Computerized Emotion Recognition Task (facial expressions of disgust from the series of Ekman and Friesen 1971) [59] | -BN group had more difficulty in recognizing expressions of disgust than HC. | d for BN vs. HC = 0.35 BN vs. AN = 0.40 |
| -Mixed group needed more information to accurately recognise disgust than HC and AN. | d for Mixed group vs. HC = 0.49 Mixed group vs. AN = 0.59 | ||||||||
| -Each group had confusion between expressions of disgust and anger (17–22%). | N/A | ||||||||
| Duriez et al. (2021) [68] France | F adults | AN (33) | 33 | 25.03 (7.04) | 26.27 (6.28) | Case-control | Facial Emotion Recognition Task-Multi-Morph Technique (disgust was one of the proto-typical emotions taken from the series of Ekman and Friesen, 1976) [60] | -Patients with AN accurately identified disgust more often than HC. | d = 0.60 |
| -Accuracy in recognizing disgust was predicted in the AN group (vs. control group) after controlling depression scores. | d = 0.58 | ||||||||
| -Higher depressive scores were related to faster and more accurate disgust recognition among the AN group, which was not observed in HC. | d for accuracy = −0.85 speed = 0.72 | ||||||||
| -No difference between disgust recognition performance and physical activity level. | d = 0.02 | ||||||||
| Dapelo et al. (2016) [69] UK | F adults | AN (35) Medicated patients with AN (21) Unmedicated patients with AN (14) | 42 | 27.54 (8.36) | 26.98 (7.55) | Case-control | Facial Emotion Recognition Task (disgust expression (Young et al., 2002) [62] depicted at different ambiguous proportions: 50%, 70%, and 90%) | -AN group manifested less accurate recognition of disgust depicted at the proportion of 90%. | Disgust recognition at 90% d = −0.85 70% d = −0.47 50% d = −0.15 |
| -No difference between AN and HC on emotion recognition accuracy at the proportion of 70% and 50%. | N/A | ||||||||
| -The minimum difference between AN and HC on response bias towards emotions shown at the proportions of 90%, 70% and 50%. | Disgust response bias at 90% d = 0.21 70% d = 0.16 50% d = 0.01 | ||||||||
| -The minimum correlation between disgust recognition accuracy and medication situation among participants with AN. | d = 0.10 | ||||||||
| Dapelo et al. (2017) [70] UK | F adults | BN (26) AN (35) | 42 | 27.54 (8.36) | 26.98 (7.55) | Case-control | Facial Emotion Recognition Task (disgust expression (Young et al., 2002) [62] depicted at different ambiguous proportions: 50%, 70%, and 90%) | at the proportion of 90% -ED group showed lower disgust recognition accuracy than HC. | d for AN vs. HC at 90% = −0.93 BN vs. HC at 90% = −0.54 |
| at the proportion of 90% -The moderate difference among AN and BN on disgust recognition accuracy. | AN vs. BN at 90% = −0.51 | ||||||||
| at the proportion of 70% -No difference among groups on disgust recognition accuracy. | AN vs. HC at 70% = −0.47 BN vs. HC at 70% = −0.12 AN vs. BN at 70% = −0.36 | ||||||||
| at the proportion of 50% -No difference among groups on disgust recognition accuracy. | AN vs. HC at 50% = −0.19 BN vs. HC at 50% = −0.10 AN vs. BN at 50% = −0.10 | ||||||||
| -BN participants misinterpreted disgust depicted at a proportion of 90% as anger compared to HC. | N/A | ||||||||
| Jänsch et al. (2009) [71] UK | F adults | AN (28) Medicated vs. Unmedicated | 28 | 27.11 (7.51) | 28.21 (7.03) | Case-control | Facial Expression Recognition Task (facial expression of disgust taken from the series of Ekman and Friesen, 1976) [60] -reaction time -accuracy -misclassification | -Increased level of ED symptomatology was associated with fewer misclassification of faces as disgusted among people with and without medication. | d (with medication) = −1.58 d (without medication) = −0.63 |
| -Those in the medicated group recognized disgust more quickly. | N/A | ||||||||
| - In the unmedicated group, only accuracy for disgust decreased with a higher level of ED symptoms. | d = −1.58 | ||||||||
| Fujiwara et al. (2017) [75] Canada | F adults | AN (19) BN (5) | HC with -low alexithymia (HC-LA: 25) -high alexithymia (HC-HA: 25) | 23.33 (7.12) | HC-LA: 19.92 (3.80) HC-HA: 18.60 (2.04) | Case-control | Facial Emotion Discrimination Task (clear vs. blended/ambiguous disgust–anger facial expressions), eye tracking) | -ED group judged ambiguous disgust–anger expressions with less accuracy than HC-LA and HC-HA. -ED group spent less time looking at disgust–anger expressions than HC-LA and HC-HA. | d (accuracy) for ED vs. HC-LA = −0.86 ED vs. HC-HA = −0.67 d (time) for ED vs. HC-LA = 0.74 ED vs. HC-HA = 0.64 |
| -Accuracy in judging ambiguous disgust–anger expressions was less than that of clear expressions among all participants (η2 partial= 0.24). | N/A | ||||||||
| -In ED group only, difficulty judging ambiguous disgust–anger faces was predicted by less visual attention to the faces (β = 0.88, t = 3.44, p = 0.004) and a lesser tendency to gaze at the faces’ eye (β = 0.38, t = 1.84, p = 0.09). | N/A | ||||||||
| Hildebrandt et al. (2018) [74] US | F adolescents | AN (16) | N/A | 16.0 (1.4) | N/A | Case-control | Voluntary Facial Expression Task: Simultaneous fMRI-EMG data were collected for yuck/disgust vs. happy facial expressions. | -EMG measuring the patterns of voluntary muscle activation (Levator labii that contributes to facial expression and movement of the mouth and upper lip) can be used to distinguish disgust from happiness. | N/A |
| -Levator labii was more active in response to disgusted faces than Zygomaticus (Mdiff = 0.294, SE = 0.022, 95% CI = 0.251, 0.337, t = 13.39, p < 0.0001) and Corrugator (Mdiff = 0.091, SE = 0.022, 95% CI = 0.134, 0.048). | N/A | ||||||||
| Dapelo et al. (2016) [76] UK | F adults | AN-R (17) AN-BP (19) BN (25) | 42 | AN: 27.50 (8.24) BN: 26.32 (6.64) | 26.98 (7.55) | Case-control | Pose Expression Task and Imitated Expression Task (facial expressions of disgust from the series of Ekman and Friesen, 1976) [60] | Non-disgust-specific findings -Those with AN and BN were less accurate at posing facial expressions of emotions. | N/A * |
| -Those with AN had lower performance than HC at imitating facial expressions, whereas BN participants did not differ from those with AN and HC. | N/A * | ||||||||
| Kim et al. (2014) [72] South Korea | F adults | AN (31) | 33 | 23.10 (9.35) | 22.18 (2.14) | Cross-over RCT | Visual Probe Detection Task (disgust expression photos from KOFEE facial expression photos from the series of Park et al. 2011) [63] | - Attentional bias to the disgust stimuli was observed in AN and HC under the placebo condition. | d = 0.60 |
| -Oxytocin had a small effect on attentional bias in the AN group. | d = 0.42 | ||||||||
| Hildebrandt et al. (2016) [73] US | F adolescents | AN-R (21) AN-BP (11) | 20 | 16.68 (3.14) | 17.91 (2.45) | Case-control | Emotional Go/No-Go Task (disgusted facial expressions taken from the MacBrain face stimulus set) [58] | -Patients with AN committed more commission errors for disgust stimuli than HC. | d = 0.74 |
| -For patients with AN, lower testosterone predicted greater behavioural disinhibition for disgusted faces (β = −0.67, 95% CI [−1.22, −0.12]). | N/A | ||||||||
| Gagnon et al. (2018) [56] Canada | AN-R (5) AN-BP (5) BN (13) | 23 | 30.35 (11.31) | 25.91 (5.86) | Case-control | Temporal Bisection Task as a time perception task during visual stimuli: disgusting vs. joyful vs. neutral food pictures | -AN patients tended to perceive the duration of disgusting food pictures longer than those with BN. | d = 0.99 | |
| -AN patients tended to perceive the duration of disgusting food pictures longer than neutral ones. | d = 0.53 | ||||||||
| Author (Year) Country | Gender (n) | Sample Size | Age M (SD) | Study Design | Method | Main Findings | Effect Size (Cohen’s d) of Main Findings | ||
|---|---|---|---|---|---|---|---|---|---|
| Clinical | Control | Clinical | Control | ||||||
| Uher et al. (2004) [52] UK | F adults | AN (16) BN (10) | 19 | AN: 26.93 (12.14) BN: 29.80 (8.80) | 26.6 8 (8.34) | Case-control | fMRI | Non-disgust-specific findings: | |
| -Greater activation in the left medial orbito-frontal and anterior cingulate cortices and less activation in the lateral prefrontal cortex, inferior parietal lobule, and cerebellum in response to food images among patients with EDs compared to HC. | N/A * | ||||||||
| -BN patients had less activation in the lateral and apical prefrontal cortex in response to food images than HC. | N/A * | ||||||||
| Uher et al. (2005) [53] UK | F adults | BN (9) AN (13) | 18 | BN: 29.6 (9.3) AN: 25.4 (10.2) | 26.6 (8.6) | Case-control | fMRI visual stimuli: underweight, normal, and overweight female bodies in swimming costumes | - Higher aversion scores were reported in response to all body shape categories associated with greater activation in the right medial apical prefrontal cortex among ED patients. | N/A |
| Schienle et al. (2004) [57] Germany | F adults | BN (11) | 12 | 25.4 (9.0) | 26.3 (6.4) | Case-control | fMRI Contrasts: Disgust > Neutral Disgust > Fear Fear > Disgust visual stimuli: disgust- vs. fear-inducing vs. neutral pictures | BN patients had greater activation in the left amygdala and right cuneus when comparing disgust with neutral and fear conditions. | N/A |
| - No significant difference was found between BN and HC for each contrast. | N/A | ||||||||
| Schienle et al. (2009) [77] Germany | F adults | BED (17) BN-purging type (14) | Normal Weight (NW: 19) Overweight (OW: 17) | BED: 26.4 (6.4) BN: 23.1 (3.8) | NW: 22.3 (2.6) OW: 25.0 (4.7) | Case-control | fMRI visual stimuli: disgust-inducing pictures Defined ROIs: amygdala, insula, and lateral OFC Contrast: Disgust > Neutral | -Disgust pictures induced greater activation in the defined ROIs among each group. | N/A |
| -Greater insula activation to disgust-inducing pictures in BN relative to OW | d = 1.52 | ||||||||
| -Greater insula and lateral OFC activations to disgust-inducing pictures in NW relative to BED. | d for insula in BED vs. NW = 1.30 d for lateral OFC in BED vs. NW = 1.34 | ||||||||
| Ashworth et al. (2011) [78] UK | F adults | Medication-free patients with BN (12) | 16 | 24.4 (4.8) | 27.4 (5.4) | Case-control | fMRI visual stimuli: disgusted female and male faces (Matsumoto and Ekman, 1988) [61] | -No significant difference was found in insula and amygdala activation in response to disgusted faces between BN and HC. | N/A |
| -BN patients had reduced activation in the praecuneus/cuneal cortex towards disgusted faces compared to HC. | N/A | ||||||||
| Joos et al. (2011a) [49] Germany | F adults | AN-R (11) | 11 | 25.0 (5.0) | 26.0 (5.2) | Case-control | fMRI visual stimuli: food or non-food pictures | -The disgust level in response to food pictures was negatively associated with the right amygdala signal. | d = −3.10 |
| Hildebrandt et al. (2018) [74] US | F adolescents | AN (17) | N/A | 16.0 (1.4) | N/A | Case-control | Facial Expression Recognition Task (facial expression of disgust) -reaction time -accuracy -misclassification | -Increased levels of ED symptomatology were associated with fewer misclassifications of faces as disgusted among people with and without medication. | d (with medication) = −1.58 d (without medication) = −0.63 |
| -Those in the medicated group recognized disgust more quickly. | N/A | ||||||||
| -In the unmedicated group, only accuracy for disgust decreased with a higher level of ED symptoms. | d = −1.58 | ||||||||
| Hildebrandt et al. (2015) [79] US | F adults | AN-R (14) | 15 | 15.05 (1.87) | 17.64 (2.71) | Case-control | Facial EMG recording during an experimental task (reversal learning paradigm) using theoretically aversive ED stimuli (chocolate candies) | -AN-R group had a distinct spike in levator labii activation (as a disgust marker) to food-cue during the acquisition phase compared to HC. | d = 1.36 |
| -The number of levator labii spikes predicted impaired extinction in reversal for AN-R group only. | d = 0.93 | ||||||||
| Soussignan et al. (2010) [80] France | F adolescents | AN (16) | 25 | 26.68 (7.30) | 24.6 (6.03) | Case-control | EMG during visual stimuli: palatable food pictures just after subliminal exposure to facial expressions (disgust/fear) | -Subliminal disgust expressions did not prime corrugator muscle reactivity to food stimuli in fasting AN patients, but subliminal fear expressions did. | d for disgust = N/A d for fear = 0.54 |
| Horndasch et al. (2012) [47] Germany | F adolescents | AN (13) | 15.7 (1.8) | 15.7 (1.8) | 16.6 (1.8) | Case-control | EEG recording visual stimuli: underweight, normal, and overweight female body pictures | -The highest earlier (16.4 ± 10.2 μV) and late (12.0 ± 6.4 μV) Late Positive Potential (LPP) amplitudes were found for underweight body pictures in the AN group. | N/A |
| Schienle et al. (2017) [31] Germany | F adults | Patients with binge-eating symptoms (36) | 33 | No age-specific information | Experimental | EEG recording during both visual stimuli (food pictures) and gustatory fluid stimuli, including water (neutral tastant) and wormwood (bitter/aversive tastant) | -Atypical/Enhanced Late Positive Potentials (LPP) towards visual food images during tasting wormwood among people with binge-eating symptoms (p = 0.04). | N/A | |
| Pollatos et al. (2008) [65] Germany | F adults | AN-R (12) | 12 | 23.86 (4.25) | 22.39 (4.78) | Case-control | EEG recording visual stimuli: emotional faces | -In the AN group, EEG recording showed increased higher N200 amplitudes to all face categories (η2 = 0.25) and lower P300 amplitudes in response to unpleasant emotional faces (η2 = 0.37), different from HC. | N/A |
| Author (Year) Country | Gender (n) | Sample Size | Age M | Study Design | Method | Main Findings | Effect Size (Cohen’s d) of Main Findings | ||
|---|---|---|---|---|---|---|---|---|---|
| Clinical | Control | Clinical | Control | ||||||
| Brooks et al. (1998) [82] Australia | F (10) M (1) adults | BN (11) | N/A | Range: 19–53 | N/A | Qualitative | Semi-structured interviews (Discourse Analytic Approach) | -Participants with BN reported disgust feelings towards themselves and their diagnosis | N/A |
| Espeset et al. (2012) [83] Australia | F adults | AN (14) | N/A | 29.1 | N/A | Qualitative | Semi-structured interviews (Grounded Theory Methodology) | Possible triggers of experiencing disgust: | N/A |
| -Social situations (i.e., feeling sensitive towards criticism or negative feedback from others). | |||||||||
| -Feelings of being full or satiated and eating food | |||||||||
| -Touch—The association between disgust and body dissatisfaction. -The coping strategy developed for disgust feelings was “avoidance” (of food and body awareness) | |||||||||
| Fox (2009) [54] UK | F adults | AN-R (5) AN-BP (6) | N/A | Range: 19–51 | N/A | Qualitative | Semi-structured interviews (Grounded Theory Methodology) | -Being bullied can trigger feelings of disgust towards the body. | N/A |
| -Disgust and anger were linked | |||||||||
| McNamara et al. (2008) [81] Norway | F adults | BN with (3) and without (2) purge AN-R (1) AN-BP (1) EDNOS (3) | N/A | 29.1 | N/A | Qualitative | Semi-structured interviews with PowerPoint slides involving the images of high- and low-caloric foods (Theoretical Framework Approach) | -The image of “Peking duck” was related to disgust feelings due to thoughts of lack of control over this food. -The image of “block of chocolate” was related to disgust feelings because of negative autobiographical memories. | N/A |
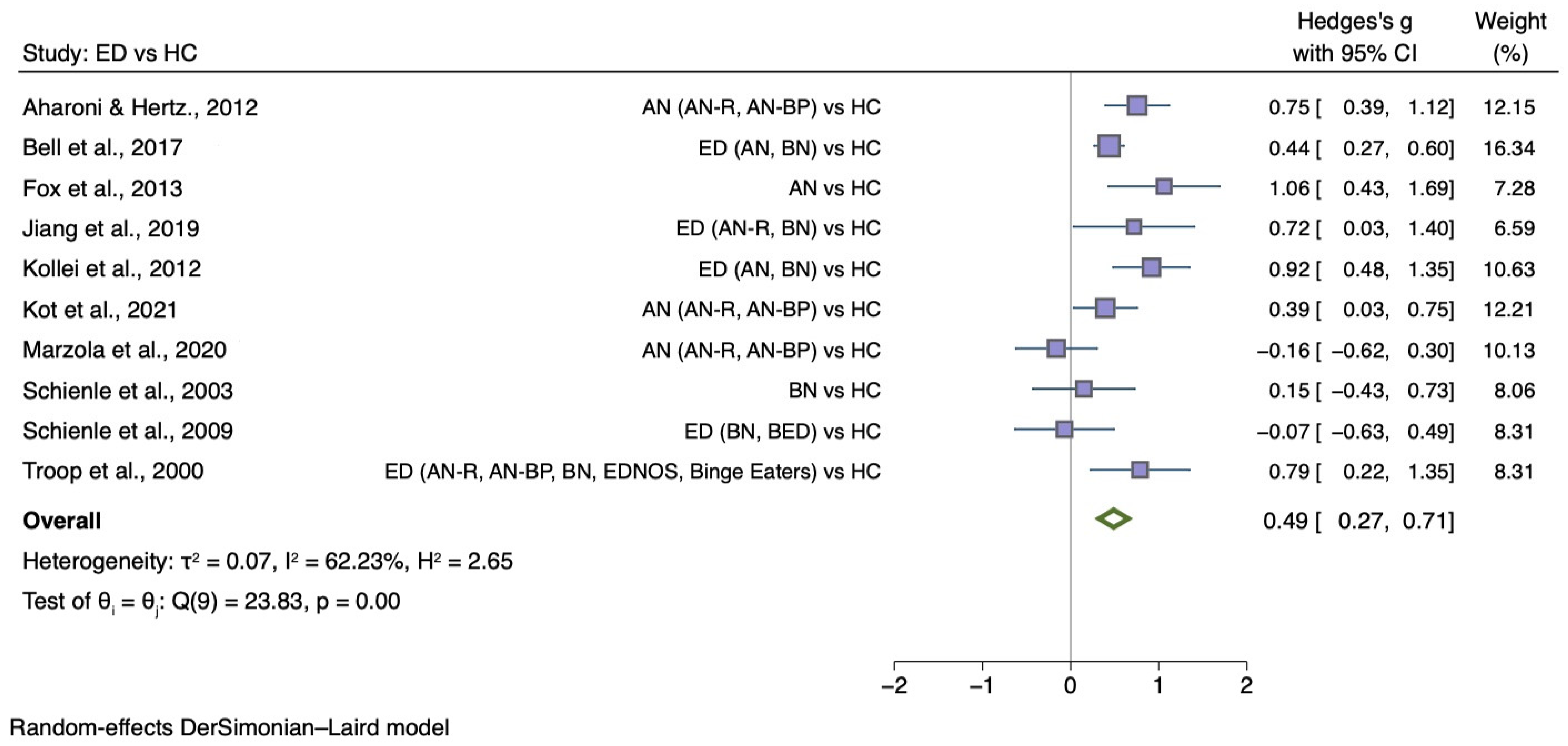
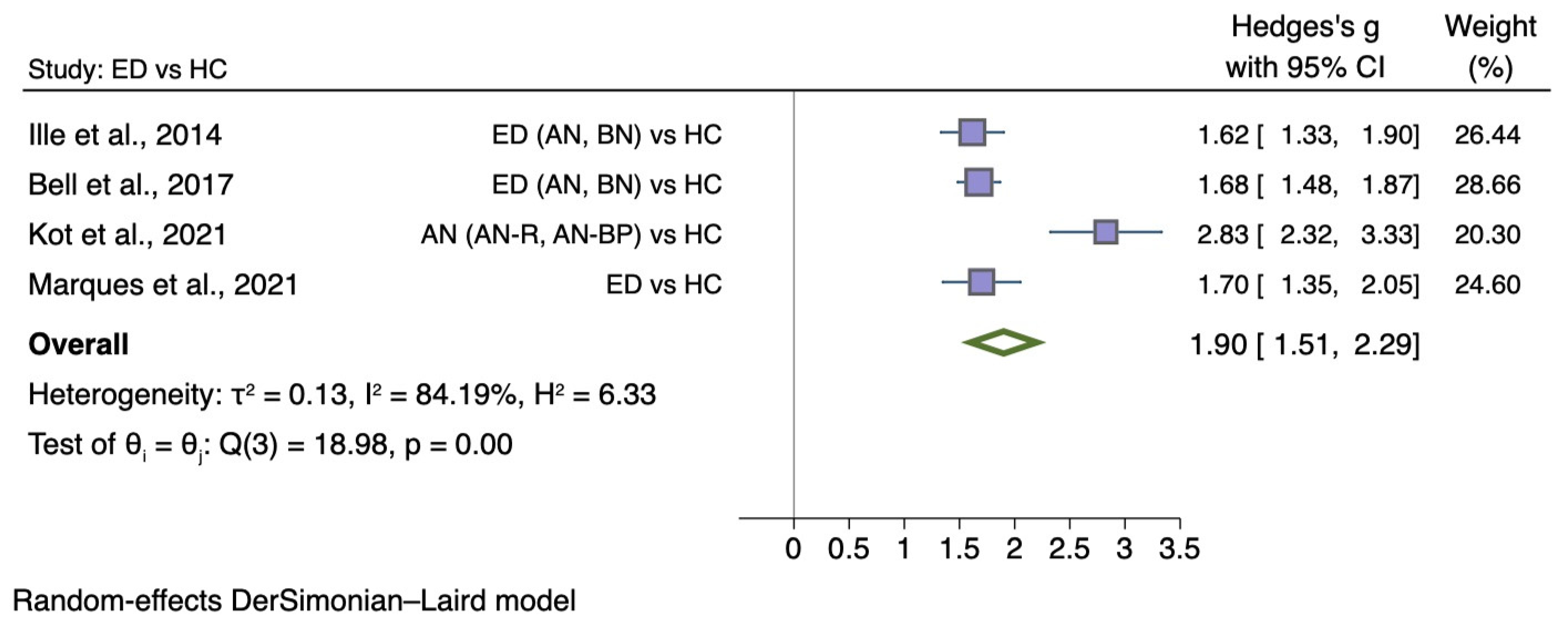
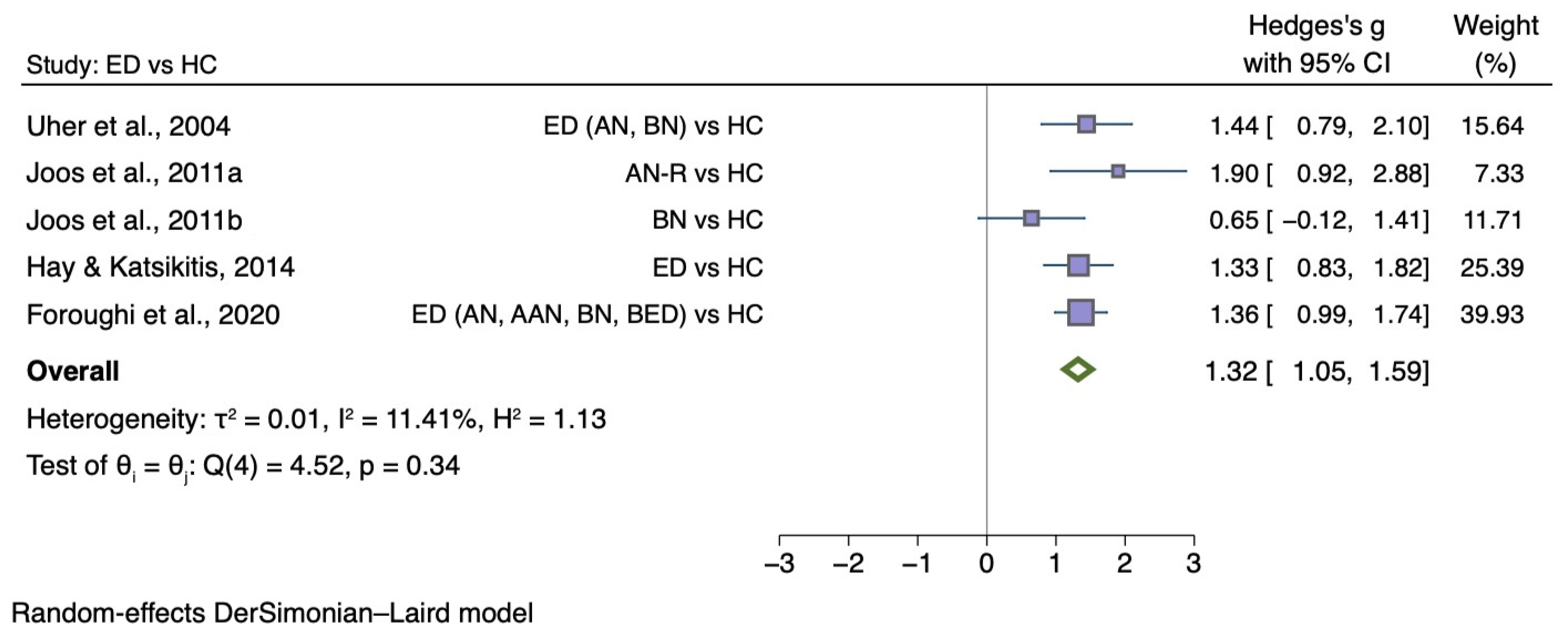
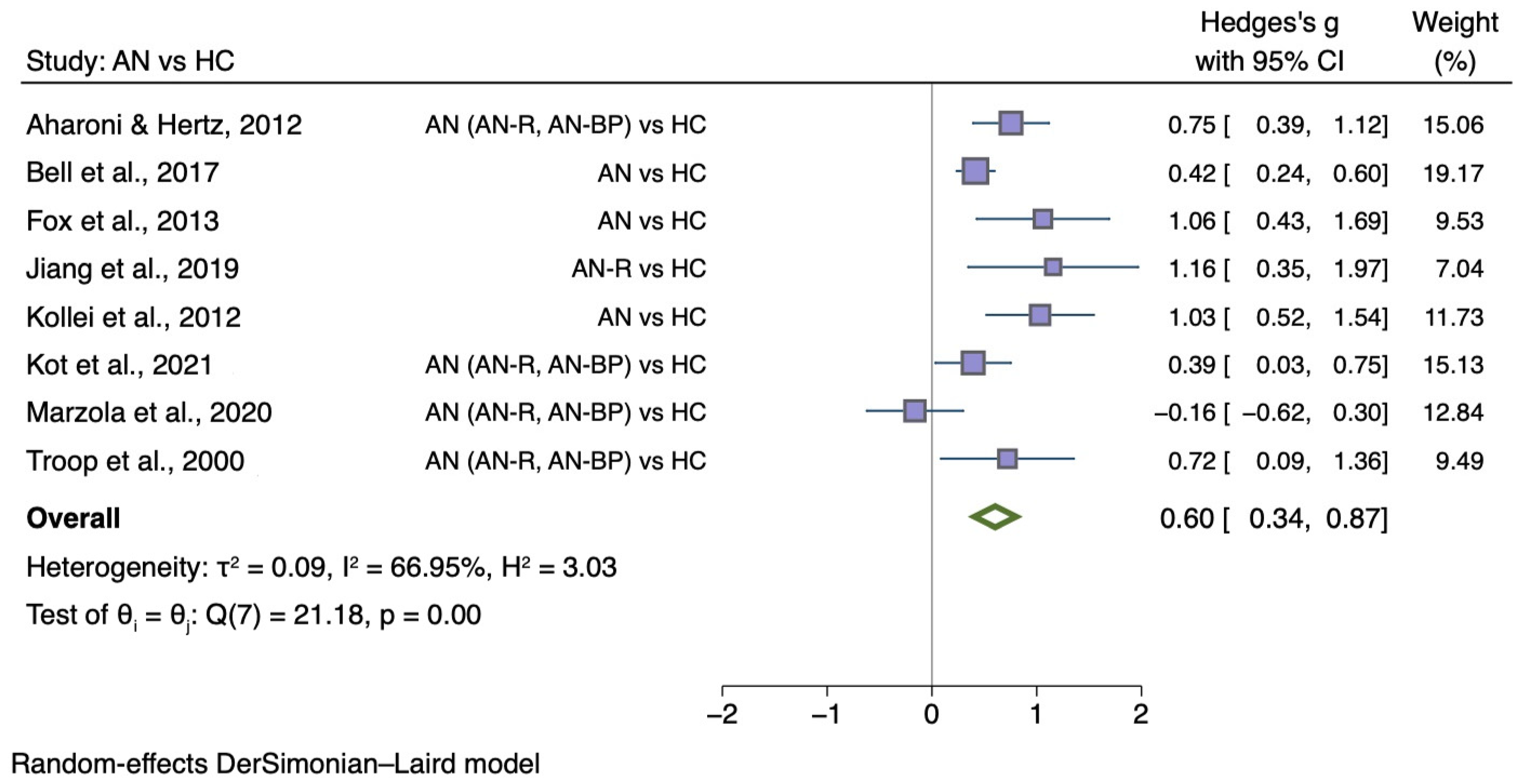
5.3.3. Self-Disgust
5.4. Sensitivity Analyses
6. Discussion
6.1. Summary of Evidence
6.2. Strengths and Limitations
6.3. Clinical Implications
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Moncrieff-Boyd, J.; Byrne, S.; Nunn, K. Disgust and Anorexia Nervosa: Confusion between self and non-self. Adv. Eat. Disord. 2014, 2, 4–18. [Google Scholar] [CrossRef]
- SIGN 164. Eating Disorders: A National Clinical Guideline. 2022. Available online: https://www.sign.ac.uk/#:~:text=SIGN%20164%3A%20Eating%20disorders,of%20type%201%20diabetes%20mellitus (accessed on 1 January 2022).
- Treasure, J.; Duarte, T.A.; Schmidt, U. Eating disorders. Lancet 2020, 395, 899–911. [Google Scholar] [CrossRef]
- Levinson, C.A.; Williams, B.M. Eating disorder fear networks: Identification of central eating disorder fears. Int. J. Eat. Disord. 2020, 53, 1960–1973. [Google Scholar] [CrossRef] [PubMed]
- Lambert, E.; Treasure, J.; Purves, K.L.; McGregor, T.; Bergou, N.; Kan, C.; Breen, G.; Eley, T.C.; Cardi, V. Fear conditioning in women with anorexia nervosa and healthy controls: A preliminary study. J. Abnorm. Psychol. 2021, 130, 490–497. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.M.; Berg, H.; Brown, T.A.; Menzel, J.; Reilly, E.E. The Role of Disgust in Eating Disorders. Curr. Psychiatry Rep. 2021, 23, 4. [Google Scholar] [CrossRef]
- Levinson, C.A.; Vanzhula, I.A.; Christian, C. Development and validation of the eating disorder fear questionnaire and interview: Preliminary investigation of eating disorder fears. Eat. Behav. 2019, 35, 101320. [Google Scholar] [CrossRef]
- Levinson, C.A.; Rapp, J.; Riley, E.N. Addressing the fear of fat: Extending imaginal exposure therapy for anxiety disorders to anorexia nervosa. Eat. Weight Disord. 2014, 19, 521–524. [Google Scholar] [CrossRef]
- Levinson, C.A.; Christian, C.; Ram, S.S.; Vanzhula, I.; Brosof, L.C.; Michelson, L.P.; Williams, B.M. Eating disorder symptoms and core eating disorder fears decrease during online imaginal exposure therapy for eating disorders. J. Affect Disord. 2020, 276, 585–591. [Google Scholar] [CrossRef]
- Porras-Garcia, B.; Serrano-Troncoso, E.; Carulla-Roig, M.; Soto-Usera, P.; Ferrer-Garcia, M.; Figueras-Puigderrajols, N.; Yilmaz, L.; Onur Sen, Y.; Shojaeian, N.; Gutiérrez-Maldonado, J. Virtual Reality Body Exposure Therapy for Anorexia Nervosa. A Case Report with Follow-Up Results. Front. Psychol. 2020, 11, 956. [Google Scholar] [CrossRef]
- Rozin, P.; Fallon, A.E. A perspective on disgust. Psychol. Rev. 1987, 94, 23–41. [Google Scholar] [CrossRef]
- Becker, C.B.; Farrell, N.R.; Waller, G. Exposure Therapy for Eating Disorders; Oxford University Press: Oxford, UK, 2019; ISBN 9780190069742. [Google Scholar]
- Powell, P.A.; Overton, P.G.; Simpson, J.E. The Revolting Self: Perspectives on the Psychological, Social, and Clinical Implications of Self-Directed Disgust; Routledge: Oxfordshire, UK, 2015. [Google Scholar]
- Clarke, A.; Simpson, J.; Varese, F. A systematic review of the clinical utility of the concept of self-disgust. Clin. Psychol. Psychother. 2019, 26, 110–134. [Google Scholar] [CrossRef] [PubMed]
- Glashouwer, K.A.; de Jong, P.J. The revolting body: Self-disgust as a key factor in anorexia nervosa. Curr. Opin. Psychol. 2021, 41, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan—A web and mobile app for systematic reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; Cochrane Book Series; John Wiley & Sons, Ltd.: Chichester, UK, 2008; pp. 187–241. ISBN 978-0-470-51845-8. [Google Scholar]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Routledge: New York, NY, USA, 2013. [Google Scholar]
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials revisited. Contemp. Clin. Trials 2015, 45, 139–145. [Google Scholar] [CrossRef]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Duval, S.; Tweedie, R. A Nonparametric “Trim and Fill” Method of Accounting for Publication Bias in Meta-Analysis. J. Am. Stat. Assoc. 2000, 95, 89–98. [Google Scholar] [CrossRef]
- StataCorp. Stata Statistical Software: Release 17; StataCorp: College Station, TX, USA, 2021. [Google Scholar]
- The Joanna Briggs Institute Critical Appraisal Tools. Available online: https://jbi.global/critical-appraisal-tools (accessed on 1 November 2021).
- Clancy, E. ‘I feel fat when I feel fat’: Affective forces of trauma in anorexia and bulimia. Gend. Place Cult. 2022, 29, 303–322. [Google Scholar] [CrossRef]
- Zeeck, A.; Stelzer, N.; Linster, H.W.; Joos, A.; Hartmann, A. Emotion and eating in binge eating disorder and obesity. Eur. Eat. Disord. Rev. 2011, 19, 426–437. [Google Scholar] [CrossRef]
- Kollei, I.; Brunhoeber, S.; Rauh, E.; de Zwaan, M.; Martin, A. Body image, emotions and thought control strategies in body dysmorphic disorder compared to eating disorders and healthy controls. J. Psychosom. Res. 2012, 72, 321–327. [Google Scholar] [CrossRef]
- Bornholt, L.; Brake, N.; Thomas, S.; Russell, L.; Madden, S.; Anderson, G.; Kohn, M.; Clarke, S. Understanding affective and cognitive self-evaluations about the body for adolescent girls. Br. J. Health Psychol. 2005, 10, 485–503. [Google Scholar] [CrossRef] [PubMed]
- Buvat-Herbaut, M.; Hebbinckuys, P.; Lemaire, A.; Buvat, J. Attitudes toward weight, body image, eating, menstruation, pregnancy, and sexuality in 81 cases of anorexia compared with 288 normal control school girls. Int. J. Eat. Disord. 1983, 2, 45–59. [Google Scholar] [CrossRef]
- Schienle, A.; Schafer, A.; Stark, R.; Walter, B.; Franz, M.; Vaitl, D. Disgust sensitivity in psychiatric disorders: A questionnaire study. J. Nerv. Ment. Dis. 2003, 191, 831–834. [Google Scholar] [CrossRef] [PubMed]
- Schienle, A.; Scharmüller, W.; Schwab, D. Processing of visual food cues during bitter taste perception in female patients with binge-eating symptoms: A cross-modal ERP study. Clin. Neurophysiol. 2017, 128, 2184–2190. [Google Scholar] [CrossRef]
- Jiang, T.; Soussignan, R.; Carrier, E.; Royet, J.P. Dysfunction of the Mesolimbic Circuit to Food Odors in Women with Anorexia and Bulimia Nervosa: A fMRI Study. Front. Hum. Neurosci. 2019, 13, 117. [Google Scholar] [CrossRef]
- Bell, K.; Coulthard, H.; Wildbur, D. Self-Disgust within Eating Disordered Groups: Associations with Anxiety, Disgust Sensitivity and Sensory Processing. Eur. Eat. Disord. Rev. 2017, 25, 373–380. [Google Scholar] [CrossRef]
- Troop, N.A.; Murphy, F.; Bramon, E.; Treasure, J.L. Disgust sensitivity in eating disorders: A preliminary investigation. Int. J. Eat. Disord. 2000, 27, 446–451. [Google Scholar] [CrossRef]
- Aharoni, R.; Hertz, M.M. Disgust sensitivity and anorexia nervosa. Eur. Eat. Disord. Rev. 2012, 20, 106–110. [Google Scholar] [CrossRef]
- Fox, J.R.; Smithson, E.; Baillie, S.; Ferreira, N.; Mayr, I.; Power, M.J. Emotion coupling and regulation in anorexia nervosa. Clin. Psychol. Psychother. 2013, 20, 319–333. [Google Scholar] [CrossRef]
- Marzola, E.; Cavallo, F.; Pradella, P.; Brustolin, A.; Abbate-Daga, G. A tasting experiment comparing food and nutritional supplement in anorexia nervosa. Appetite 2020, 155, 155. [Google Scholar] [CrossRef]
- Kot, E.; Grzegorzewski, P.; Kostecka, B.; Kucharska, K. Self-disgust and disgust sensitivity are increased in anorexia nervosa inpatients, but only self-disgust mediates between comorbid and core psychopathology. Eur. Eat. Disord. Rev. 2021, 29, 879–892. [Google Scholar] [CrossRef] [PubMed]
- Davey, G.C.L.; Buckland, G.; Tantow, B.; Dallos, R. Disgust and eating disorders. Eur. Eat. Disord. Rev. 1998, 6, 201–211. [Google Scholar] [CrossRef]
- Troop, N.A.; Treasure, J.L.; Serpell, L. A further exploration of disgust in eating disorders. Eur. Eat. Disord. Rev. 2002, 10, 218–226. [Google Scholar] [CrossRef]
- Cooper, J.L.; Morrison, T.L.; Bigman, O.L.; Abramowitz, S.I.; Levin, S.; Krener, P. Mood Changes and Affective-Disorder in the Bulimic Binge Purge Cycle. Int. J. Eat. Disord. 1988, 7, 469–474. [Google Scholar] [CrossRef]
- Richson, B.N.; Forbush, K.T.; Schaumberg, K.; Crosby, R.D.; Peterson, C.B.; Crow, S.J.; Mitchell, J.E. Are the Criterion B binge-eating symptoms interchangeable in understanding binge-eating severity? An item response theory analysis. Int. J. Eat. Disord. 2020, 53, 1983–1992. [Google Scholar] [CrossRef]
- Marques, C.; Simão, M.; Guiomar, R.; Castilho, P. Self-disgust and urge to be thin in eating disorders: How can self-compassion help? Eat. Weight Disord. 2021, 26, 2317–2324. [Google Scholar] [CrossRef]
- Ille, R.; Schöggl, H.; Kapfhammer, H.P.; Arendasy, M.; Sommer, M.; Schienle, A. Self-disgust in mental disorders—Symptom-related or disorder-specific? Compr. Psychiatry 2014, 55, 938–943. [Google Scholar] [CrossRef]
- Foroughi, N.; Madden, S.; Clarke, S.; Kohn, M.; Donnelly, B.; Touyz, S.; Hay, P. Do emotional responses to food images differ within different types of eating disorders? Australas. Psychiatry 2020, 28, 128–133. [Google Scholar] [CrossRef]
- Hay, P.; Katsikitis, M. Emotional responses to images of food in adults with an eating disorder: A comparative study with healthy and clinical controls. Eat. Behav. 2014, 15, 371–374. [Google Scholar] [CrossRef]
- Horndasch, S.; Heinrich, H.; Kratz, O.; Moll, G.H. The late positive potential as a marker of motivated attention to underweight bodies in girls with anorexia nervosa. J. Psychosom. Res. 2012, 73, 443–447. [Google Scholar] [CrossRef][Green Version]
- Joos, A.A.; Cabrillac, E.; Hartmann, A.; Wirsching, M.; Zeeck, A. Emotional perception in eating disorders. Int. J. Eat. Disord. 2009, 42, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Joos, A.A.; Saum, B.; van Elst, L.T.; Perlov, E.; Glauche, V.; Hartmann, A.; Freyer, T.; Tuscher, O.; Zeeck, A. Amygdala hyperreactivity in restrictive anorexia nervosa. Psychiatry Res. 2011, 191, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Joos, A.A.B.; Saum, B.; Zeeck, A.; Perlov, E.; Glauche, V.; Hartmann, A.; Freyer, T.; Sandholz, A.; Unterbrink, T.; van Elst, L.T.; et al. Frontocingular Dysfunction in Bulimia Nervosa when Confronted with Disease-specific Stimuli. Eur. Eat. Disord. Rev. 2011, 19, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Joos, A.A.; Gille, M.; Hartmann, A.; Unterbrink, T.; Wetzler-Burmeister, E.; Scheidt, C.; Waller, E.; Bauer, J.; Wirsching, M.; Zeeck, A. Emotional perception in patients with eating disorders in comparison with depressed patients. Eur. Eat. Disord. Rev. 2012, 20, 468–475. [Google Scholar] [CrossRef] [PubMed]
- Uher, R.; Murphy, T.; Brammer, M.J.; Dalgleish, T.; Phillips, M.L.; Ng, V.W.; Andrew, C.M.; Williams, S.C.; Campbell, I.C.; Treasure, J. Medial prefrontal cortex activity associated with symptom provocation in eating disorders. Am. J. Psychiatry 2004, 161, 1238–1246. [Google Scholar] [CrossRef]
- Uher, R.; Murphy, T.; Friederich, H.C.; Dalgleish, T.; Brammer, M.J.; Giampietro, V.; Phillips, M.L.; Andrew, C.M.; Ng, V.W.; Williams, S.C.; et al. Functional neuroanatomy of body shape perception in healthy and eating-disordered women. Biol. Psychiatry 2005, 58, 990–997. [Google Scholar] [CrossRef]
- Fox, J.R. A qualitative exploration of the perception of emotions in anorexia nervosa: A basic emotion and developmental perspective. Clin. Psychol. Psychother. 2009, 16, 276–302. [Google Scholar] [CrossRef]
- Kockler, T.D.; Tschacher, W.; Santangelo, P.S.; Limberger, M.F.; Ebner-Priemer, U.W. Specificity of emotion sequences in borderline personality disorder compared to posttraumatic stress disorder, bulimia nervosa, and healthy controls: An e-diary study. Borderline Personal Disord. Emot. Dysregul. 2017, 4, 26. [Google Scholar] [CrossRef]
- Gagnon, C.; Bégin, C.; Laflamme, V.; Grondin, S. Temporal Processing of Joyful and Disgusting Food Pictures by Women with an Eating Disorder. Front. Hum. Neurosci. 2018, 12, 129. [Google Scholar] [CrossRef]
- Schienle, A.; Stark, R.; Schafer, A.; Walter, B.; Kirsch, P.; Vaitl, D. Disgust and disgust sensitivity in bulimia nervosa: An fMRI study. Eur. Eat. Disord. Rev. 2004, 12, 42–50. [Google Scholar] [CrossRef]
- Tottenham, N. MacBrain Face Stimulus Set; MacArthur Foundation Research Network on Early Experience and Brain Development: Chicago, IL, USA, 1998. [Google Scholar]
- Ekman, P.; Friesen, W.V. Constants across cultures in the face and emotion. J. Pers. Soc. Psychol. 1971, 17, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Ekman, P.; Friesen, W.V. Measuring Facial Movement. Environ. Psychol. Non-Verbal Behav. 1976, 1, 56–75. [Google Scholar] [CrossRef]
- Matsumoto, D.; Ekman, P. Japanese and Caucasian Facial Expressions of Emotion (JACFEE); Intercultural and Emotion Research Laboratory, Department of Psychiatry, University of California: San Francisco, CA, USA, 1988. [Google Scholar]
- Young, A.W.; Perret, D.I.; Calder, A.J.; Sprengelmeyer, R.; Ekman, P. Facial Expressions of Emotion: Stimuli and Tests (FEEST). Available online: file:///C:/Users/k21108448/Downloads/FeestManual.pdf (accessed on 25 March 2022).
- Park, J.Y.; Oh, J.M.; Kim, S.Y.; Lee, M.; Lee, C.; Kim, B.R.; An, S.K. Korean Facial Expressions of Emotion (KOFEE); Section of Affect & Neuroscience, Institute of Behavioral Science in Medicine, Yonsei University College of Medicine: Seoul, Korea, 2011. [Google Scholar]
- Lundqvist, D.; Flykt, A.; Öhman, A. The Karolinska Directed Emotional Faces—KDEF. Available online: https://www.kdef.se/home/aboutKDEF.html (accessed on 25 March 2022).
- Pollatos, O.; Herbert, B.M.; Schandry, R.; Gramann, K. Impaired central processing of emotional faces in anorexia nervosa. Psychosom. Med. 2008, 70, 701–708. [Google Scholar] [CrossRef]
- Lulé, D.; Schulze, U.M.; Bauer, K.; Schöll, F.; Müller, S.; Fladung, A.K.; Uttner, I. Anorexia nervosa and its relation to depression, anxiety, alexithymia and emotional processing deficits. Eat. Weight Disord. 2014, 19, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Wyssen, A.; Lao, J.; Rodger, H.; Humbel, N.; Lennertz, J.; Schuck, K.; Isenschmid, B.; Milos, G.; Trier, S.; Whinyates, K.; et al. Facial Emotion Recognition Abilities in Women Experiencing Eating Disorders. Psychosom. Med. 2019, 81, 155–164. [Google Scholar] [CrossRef]
- Duriez, P.; Guy-Rubin, A.; Kaya Lefèvre, H.; Gorwood, P. Morphing analysis of facial emotion recognition in anorexia nervosa: Association with physical activity. Eat. Weight Disord. 2021, 27, 1053–1061. [Google Scholar] [CrossRef]
- Dapelo, M.M.; Surguladze, S.; Morris, R.; Tchanturia, K. Emotion Recognition in Blended Facial Expressions in Women with Anorexia Nervosa. Eur. Eat. Disord. Rev. 2016, 24, 34–42. [Google Scholar] [CrossRef]
- Dapelo, M.M.; Surguladze, S.; Morris, R.; Tchanturia, K. Emotion Recognition in Face and Body Motion in Bulimia Nervosa. Eur. Eat. Disord. Rev. 2017, 25, 595–600. [Google Scholar] [CrossRef]
- Jansch, C.; Harmer, C.; Cooper, M.J. Emotional processing in women with anorexia nervosa and in healthy volunteers. Eat. Behav. 2009, 10, 184–191. [Google Scholar] [CrossRef]
- Kim, Y.R.; Kim, C.H.; Park, J.H.; Pyo, J.; Treasure, J. The impact of intranasal oxytocin on attention to social emotional stimuli in patients with anorexia nervosa: A double blind within-subject cross-over experiment. PLoS ONE 2014, 9, e90721. [Google Scholar] [CrossRef]
- Hildebrandt, T.; Grotzinger, A.; Schulz, K. Anorexia nervosa, emotional go/no-go, and the distinct effect of testosterone. Int. J. Eat. Disord. 2016, 49, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, T.; Schulz, K.; Fleysher, L.; Griffen, T.; Heywood, A.; Sysko, R. Development of a methodology to combine fMRI and EMG to measure emotional responses in patients with anorexia nervosa. Int. J. Eat. Disord. 2018, 51, 722–729. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, E.; Kube, V.L.; Rochman, D.; Macrae-Korobkov, A.K.; Peynenburg, V.; University of Alberta Hospital Eating Disorder, Program. Visual Attention to Ambiguous Emotional Faces in Eating Disorders: Role of Alexithymia. Eur. Eat. Disord. Rev. 2017, 25, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Dapelo, M.M.; Bodas, S.; Morris, R.; Tchanturia, K. Deliberately generated and imitated facial expressions of emotions in people with eating disorders. J. Affect. Disord. 2016, 191, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Schienle, A.; Schafer, A.; Hermann, A.; Vaitl, D. Binge-eating disorder: Reward sensitivity and brain activation to images of food. Biol. Psychiatry 2009, 65, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Ashworth, F.; Pringle, A.; Norbury, R.; Harmer, C.J.; Cowen, P.J.; Cooper, M.J. Neural response to angry and disgusted facial expressions in bulimia nervosa. Psychol. Med. 2011, 41, 2375–2384. [Google Scholar] [CrossRef]
- Hildebrandt, T.; Grotzinger, A.; Reddan, M.; Greif, R.; Levy, I.; Goodman, W.; Schiller, D. Testing the disgust conditioning theory of food-avoidance in adolescents with recent onset anorexia nervosa. Behav. Res. Ther. 2015, 71, 131–138. [Google Scholar] [CrossRef]
- Soussignan, R.; Jiang, T.; Rigaud, D.; Royet, J.; Schaal, B. Subliminal fear priming potentiates negative facial reactions to food pictures in women with anorexia nervosa. Psychol. Med. 2010, 40, 503–514. [Google Scholar] [CrossRef]
- McNamara, C.; Chur-Hansen, A.; Hay, P. Emotional responses to food in adults with an eating disorder: A qualitative exploration. Eur. Eat. Disord. Rev. 2008, 16, 115–123. [Google Scholar] [CrossRef]
- Brooks, A.; LeCouteur, A.; Hepworth, J. Accounts of experiences of bulimia: A discourse analytic study. Int. J. Eat. Disord. 1998, 24, 193–205. [Google Scholar] [CrossRef]
- Espeset, E.M.S.; Gulliksen, K.S.; Nordb, R.H.S.; Skarderud, F.; Holte, A. The Link Between Negative Emotions and Eating Disorder Behaviour in Patients with Anorexia Nervosa. Eur. Eat. Disord. Rev. 2012, 20, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Olatunji, B.O.; McKay, D. Disgust and Its Disorders: Theory, Assessment, and Treatment Implications; American Psychological Association: Washington, DC, USA, 2009; ISBN 978-1433803970. [Google Scholar]
- Vicario, C.M.; Rafal, R.D.; Martino, D.; Avenanti, A. Core, social and moral disgust are bounded: A review on behavioral and neural bases of repugnance in clinical disorders. Neurosci. Biobehav. Rev. 2017, 80, 185–200. [Google Scholar] [CrossRef] [PubMed]
- Crosby, C.L.; Durkee, C.M.; Meston, D.M. Six dimensions of sexual disgust. Pers. Individ. Differ. 2020, 156, 109714. [Google Scholar] [CrossRef]
- Schmidt, U.; Tiller, J.; Blanchard, M.; Andrews, B.; Treasure, J. Is there a specific trauma precipitating anorexia nervosa? Psychol. Med. 1997, 27, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Hall, N.A.; Everson, A.T.; Billingsley, M.R.; Miller, M.B. Moral injury, mental health and behavioural health outcomes: A systematic review of the literature. Clin. Psychol. Psychother. 2022, 29, 92–110. [Google Scholar] [CrossRef] [PubMed]
- Howkins, S.; Millar, J.F.A.; Salkovskis, P.M. Sensitivity to being betrayed and betraying others in obsessive compulsive disorder and depression. Br. J. Clin. Psychol. 2022, 61, 58–75. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, T.; Peyser, D.; Sysko, R. Lessons learned developing and testing family-based interoceptive exposure for adolescents with low-weight eating disorders. Int. J. Eat. Disord. 2021, 54, 2037–2045. [Google Scholar] [CrossRef]
- Cardi, V.; Leppanen, J.; Mataix-Cols, D.; Campbell, I.C.; Treasure, J. A case series to investigate food-related fear learning and extinction using in vivo food exposure in anorexia nervosa: A clinical application of the inhibitory learning framework. Eur. Eat Disord. Rev. 2019, 27, 173–181. [Google Scholar] [CrossRef]
- Schmidt, U.; Startup, H.; Treasure, J. A Cognitive-Interpersonal Therapy Workbook for Treating Anorexia Nervosa: The Maudsley Model; Routledge: London, UK, 2018; ISBN 9781315728483. [Google Scholar]
- Gilbert, P. The origins and nature of compassion focused therapy. Br. J. Clin. Psychol. 2014, 53, 6–41. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bektas, S.; Keeler, J.L.; Anderson, L.M.; Mutwalli, H.; Himmerich, H.; Treasure, J. Disgust and Self-Disgust in Eating Disorders: A Systematic Review and Meta-Analysis. Nutrients 2022, 14, 1728. https://doi.org/10.3390/nu14091728
Bektas S, Keeler JL, Anderson LM, Mutwalli H, Himmerich H, Treasure J. Disgust and Self-Disgust in Eating Disorders: A Systematic Review and Meta-Analysis. Nutrients. 2022; 14(9):1728. https://doi.org/10.3390/nu14091728
Chicago/Turabian StyleBektas, Sevgi, Johanna Louise Keeler, Lisa M. Anderson, Hiba Mutwalli, Hubertus Himmerich, and Janet Treasure. 2022. "Disgust and Self-Disgust in Eating Disorders: A Systematic Review and Meta-Analysis" Nutrients 14, no. 9: 1728. https://doi.org/10.3390/nu14091728
APA StyleBektas, S., Keeler, J. L., Anderson, L. M., Mutwalli, H., Himmerich, H., & Treasure, J. (2022). Disgust and Self-Disgust in Eating Disorders: A Systematic Review and Meta-Analysis. Nutrients, 14(9), 1728. https://doi.org/10.3390/nu14091728