Diet, Oxidative Stress, and Blood Serum Nutrients in Various Types of Glaucoma: A Systematic Review
Abstract
:1. Introduction
2. Nutrition, Blood Nutrient Levels of Oxidative Stress, and Glaucoma
2.1. Primary Open-Angle Glaucoma
2.2. Normal Tension Glaucoma
2.3. Pseudoexfoliation Glaucoma
2.4. Angle-Closure Glaucoma
3. Glaucoma and Vitamins
4. Discussion
5. Conclusions
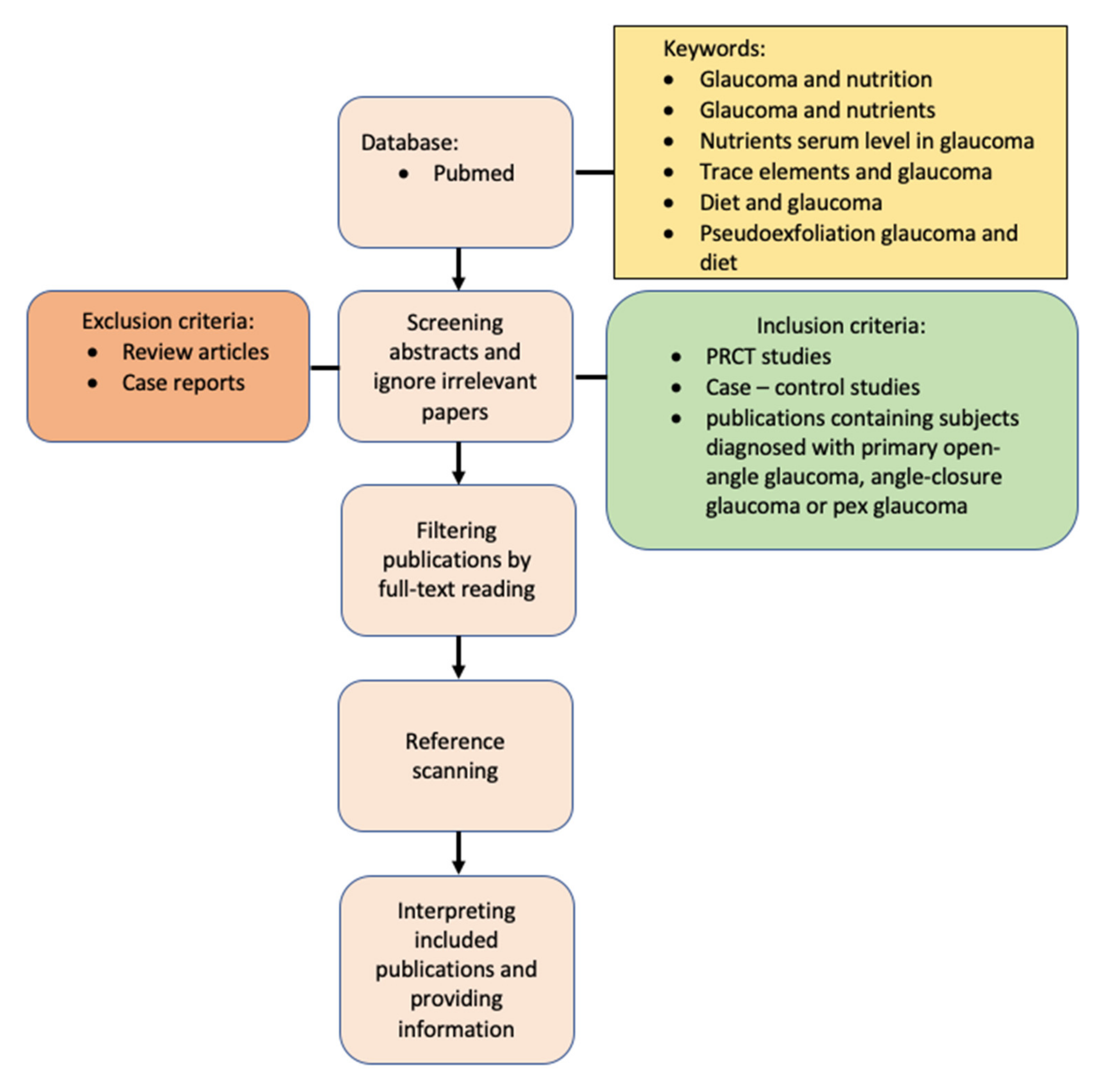
6. Method of Literature Search
6.1. Risk of Bias Assessment
6.2. Data Extraction
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gupta, D.; Chen, P.P. Glaucoma. Am. Fam. Phys. 2016, 93, 668–674. [Google Scholar]
- Schuster, A.K.; Erb, C.; Hoffmann, E.M.; Dietlein, T.; Pfeiffer, N. The diagnosis and treatment of glaucoma. Dtsch. Arztebl. Int. 2020, 117, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Gong, H.; Swain, D.L. The histopathological changes in the trabecular outflow pathway and their possible effects on aqueous outflow in eyes with primary open-angle glaucoma. In Glaucoma Research and Clinical Advances 2016 to 2018; Kugler: McCook, NE, USA, 2016; Chapter 2. [Google Scholar]
- Schlötzer-Schrehardt, U. Genetics and genomics of pseudoexfoliation syndrome/glaucoma. Middle East Afr. J. Ophthalmol. 2011, 18, 30–36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Konopińska, J.; Lewczuk, K.; Jabłońska, J.; Mariak, Z.; Rękas, M. Microinvasive Glaucoma Surgery: A Review of Schlemm’s Canal-Based Procedures. Clin. Ophthalmol. 2021, 15, 1109–1118. [Google Scholar] [CrossRef]
- Dhawan, M.; Hans, T.; Sandhu, P.S.; Midha, N. Evaluation of Vision-Related Quality of Life in Patients with Glaucoma: A Hospital-Based Study. J. Curr. Glaucoma Pract. 2019, 13, 9–15. [Google Scholar]
- Tanito, M.; Kaidzu, S.; Takai, Y.; Ohira, A. Association between systemic oxidative stress and visual field damage in open-angle glaucoma. Sci. Rep. 2016, 6, 25792. [Google Scholar] [CrossRef] [Green Version]
- Moreno-Montañés, J.; Gándara, E.; Gutierrez-Ruiz, I.; Moreno-Galarraga, L.; Ruiz-Canela, M.; Bes-Rastrollo, M.; Martínez-González, M.Á.; Fernandez-Montero, A. Healthy lifestyle score and incidence of glaucoma: The Sun Project. Nutrients 2022, 14, 779. [Google Scholar] [CrossRef]
- Adornetto, A.; Rombolà, L.; Morrone, L.A.; Nucci, C.; Corasaniti, M.T.; Bagetta, G.; Russo, R. Natural products: Evidence for neuroprotection to be exploited in glaucoma. Nutrients 2020, 12, 3158. [Google Scholar] [CrossRef]
- Mozaffarieh, M.; Schoetzau, A.; Sauter, M.; Grieshaber, M.; Orgül, S.; Golubnitschaja, O.; Flammer, J. Comet assay analysis of single-stranded DNA breaks in circulating leukocytes of glaucoma patients. Mol. Vis. 2008, 14, 1584–15488. [Google Scholar]
- Haefliger, I.O.; Dettmann, E.; Liu, R.; Meyer, P.; Prünte, C.; Messerli, J.; Flammer, J. Potential role of nitric oxide and endothelin in the pathogenesis of glaucoma. Surv. Ophthalmol. 1999, 43 (Suppl. 1), S51–S58. [Google Scholar] [CrossRef]
- Ellis, D.Z.; Dismuke, W.M.; Chokshi, B.M. Characterization of soluble guanylate cyclase in NO-induced increases in aqueous humor outflow facility and in the trabecular meshwork. Investig. Ophthalmol. Vis. Sci. 2009, 50, 1808–1813. [Google Scholar] [CrossRef] [PubMed]
- Stamer, W.D.; Lei, Y.; Boussommier-Calleja, A.; Overby, D.R.; Ethier, C.R. eNOS, a pressure-dependent regulator of intraocular pressure. Investig. Ophthalmol. Vis. Sci. 2011, 52, 9438–9444. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Predescu, D.; Predescu, S.; Shimizu, J.; Miyawaki-Shimizu, K.; Malik, A.B. Constitutive eNOS-derived nitric oxide is a determinant of endothelial junctional integrity. Am. J. Physiol. Lung Cell Mol. Physiol. 2005, 289, L371–L381. [Google Scholar] [CrossRef]
- Aliancy, J.; Stamer, W.D.; Wirostko, B. A Review of Nitric Oxide for the Treatment of Glaucomatous Disease. Ophthalmol. Ther. 2017, 6, 221–232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neufeld, A.H.; Sawada, A.; Becker, B. Inhibition of nitric-oxide synthase 2 by aminoguanidine provides neuroprotection of retinal ganglion cells in a rat model of chronic glaucoma. Proc. Natl. Acad. Sci. USA 1999, 96, 9944–9948. [Google Scholar] [CrossRef] [Green Version]
- Barthelmes, J.; Nägele, M.P.; Ludovici, V.; Ruschitzka, F.; Sudano, I.; Flammer, A.J. Endothelial dysfunction in cardiovascular disease and Flammer syndrome-similarities and differences. EPMA J. 2017, 8, 99–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramdas, W.D.; Wolfs, R.C.; Kiefte-de Jong, J.C.; Hofman, A.; de Jong, P.T.; Vingerling, J.R.; Jansonius, N.M. Nutrient intake and risk of open-angle glaucoma: The Rotterdam Study. Eur. J. Epidemiol. 2012, 27, 385–393. [Google Scholar] [CrossRef] [Green Version]
- Braakhuis, A.; Raman, R.; Vaghefi, E. The association between dietary intake of antioxidants and ocular disease. Diseases 2017, 5, 3. [Google Scholar] [CrossRef] [Green Version]
- Yoserizal, M.; Hirooka, K.; Yoneda, M.; Ohno, H.; Kobuke, K.; Kawano, R.; Kiuchi, Y. Associations of nutrient intakes with glaucoma among Japanese Americans. Medicine 2019, 98, e18314. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Shao, M.; Li, D.; Tang, B.; Cao, W.; Sun, X. Association of serum uric acid levels with primary open-angle glaucoma: A 5-year case-control study. Acta Ophthalmol. 2019, 97, e356–e363. [Google Scholar] [CrossRef]
- Kang, J.H.; Willett, W.C.; Rosner, B.A.; Buys, E.; Wiggs, J.L.; Pasquale, L.R. Association of dietary nitrate intake with primary open-angle glaucoma: A prospective analysis from the Nurses’ Health Study and Health Professionals Follow-up Study. JAMA Ophthalmol. 2016, 134, 294–303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, S.Y.; Singh, K.; Lin, S.C. The association between glaucoma prevalence and supplementation with the oxidants calcium and iron. Investig. Ophthalmol. Vis. Sci. 2012, 53, 725–731. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, S.C.; Wang, S.Y.; Yoo, C.; Singh, K.; Lin, S.C. Association between serum ferritin and glaucoma in the South Korean population. JAMA Ophthalmol. 2014, 132, 1414–1420. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, S.S. Coenzyme Q and its role in glaucoma. Saudi J. Ophthalmol. 2020, 34, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Tian, K.; Shibata-Germanos, S.; Pahlitzsch, M.; Cordeiro, M.F. Current perspective of neuroprotection and glaucoma. Clin. Ophthalmol. 2015, 9, 2109–2118. [Google Scholar]
- Edwards, G.; Lee, Y.; Kim, M.; Bhanvadia, S.; Kim, K.Y.; Ju, W.K. Effect of ubiquinol on glaucomatous neurodegeneration and oxidative stress: Studies for retinal ganglion cell survival and/or visual function. Antioxidants 2020, 9, 952. [Google Scholar] [CrossRef]
- Nakajima, Y.; Inokuchi, Y.; Nishi, M.; Shimazawa, M.; Otsubo, K.; Hara, H. Coenzyme Q10 protects retinal cells against oxidative stress in vitro and in vivo. Brain Res. 2008, 1226, 226–233. [Google Scholar] [CrossRef]
- Lee, D.; Shim, M.S.; Kim, K.Y.; Noh, Y.H.; Kim, H.; Kim, S.Y.; Weinreb, R.N.; Ju, W.K. Coenzyme Q10 inhibits glutamate excitotoxicity and oxidative stress-mediated mitochondrial alteration in a mouse model of glaucoma. Investig. Ophthalmol. Vis. Sci. 2014, 55, 993–1005. [Google Scholar] [CrossRef] [Green Version]
- Sim, R.H.; Sirasanagandla, S.R.; Das, S.; Teoh, S.L. Treatment of Glaucoma with Natural Products and Their Mechanism of Action: An Update. Nutrients 2022, 14, 534. [Google Scholar] [CrossRef]
- Russo, R.; Cavaliere, F.; Rombola, L.; Gliozzi, M.; Cerulli, A.; Nucci, C.; Fazzi, E.; Bagetta, G.; Corasaniti, M.T.; Morrone, L.A. Rational basis for the development of coenzyme Q10 as a neurotherapeutic agent for retinal protection. Prog. Brain Res. 2008, 173, 575–582. [Google Scholar]
- Nucci, C.; Tartaglione, R.; Cerulli, A.; Mancino, R.; Spano, A.; Cavaliere, F.; Rombola, L.; Bagetta, G.; Corasaniti, M.T.; Morrone, L.A. Retinal damage caused by high intraocular pressure-induced transient ischemia is prevented by coenzyme Q10 in rat. Int. Rev. Neurobiol. 2007, 82, 397–406. [Google Scholar] [PubMed]
- Parisi, V.; Centofanti, M.; Gandolfi, S.; Marangoni, D.; Rossetti, L.; Tanga, L.; Tardini, M.; Traina, S.; Ungaro, N.; Vetrugno, M.; et al. Effects of coenzyme Q10 in conjunction with vitamin E on retinal-evoked and cortical-evoked responses in patients with open-angle glaucoma. J. Glaucoma 2014, 23, 391–404. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.I.; Kim, Y.C.; Park, C.K. Dietary niacin and open-angle glaucoma: The Korean National Health and Nutrition Examination Survey. Nutrients 2018, 10, 387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.Y.; Kim, J.M.; Lee, K.Y.; Kim, B.; Lee, M.Y.; Park, K.H. Relationships between obesity, nutrient supply and primary open angle glaucoma in Koreans. Nutrients 2020, 12, 878. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pang, R.; Feng, S.; Cao, K.; Sun, Y.; Guo, Y.; Ma, D.; Pang, C.P.; Liu, X.; Qian, J.; Xie, Y.; et al. Association of Serum Retinol Concentration with Normal-Tension Glaucoma. Eye 2021. ahead of print. [Google Scholar] [CrossRef]
- Yoshida, K.; Ohguro, I.; Ohguro, H. Black currant anthocyanins normalized abnormal levels of serum concentrations of endothelin-1 in patients with glaucoma. J. Ocul. Pharmacol. Ther. 2013, 29, 480–487. [Google Scholar] [CrossRef]
- Chung, H.S.; Harris, A.; Kristinsson, J.K.; Ciulla, T.A.; Kagemann, C.; Ritch, R. Ginkgo biloba extract increases ocular blood flow velocity. J. Ocul. Pharmacol. Ther. 1999, 15, 233–240. [Google Scholar] [CrossRef]
- Flammer, J.; Mozaffarieh, M. What is the present pathogenetic concept of glaucomatous optic neuropathy? Surv. Ophthalmol. 2007, 52 (Suppl. 2), S162–S173. [Google Scholar] [CrossRef]
- Zhang, J.; Fu, S.; Liu, S.; Mao, T.; Xiu, R. The therapeutic effect of Ginkgo biloba extract in SHR rats and its possible mechanisms based on cerebral microvascular flow and vasomotion. Clin. Hemorheol. Microcirc. 2000, 23, 133–138. [Google Scholar]
- Szabo, M.E.; Droy-Lefaix, M.T.; Doly, M. Direct measurement of free radicals in ischemic/reperfused diabetic rat retina. Clin. Neurosci. 1997, 4, 240–245. [Google Scholar]
- Uhler, T.A.; Piltz-Seymour, J. Optic disc hemorrhages in glaucoma and ocular hypertension: Implications and recommendations. Curr. Opin. Ophthalmol. 2008, 19, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Cybulska-Heinrich, A.K.; Mozaffarieh, M.; Flammer, J. Ginkgo biloba: An adjuvant therapy for progressive normal and high tension glaucoma. Mol. Vis. 2012, 18, 390–402. [Google Scholar] [PubMed]
- Ou, H.C.; Lee, W.J.; Lee, I.T.; Chiu, T.H.; Tsai, K.L.; Lin, C.Y.; Sheu, W.H. Ginkgo biloba extract attenuates oxLDL-induced oxidative functional damages in endothelial cells. J. Appl. Physiol. 2009, 106, 1674–1685. [Google Scholar] [CrossRef] [Green Version]
- Eckert, A.; Keil, U.; Scherping, I.; Hauptmann, S.; Müller, W.E. Stabilization of mitochondrial membrane potential and improvement of neuronal energy metabolism by Ginkgo biloba extract EGb 761. Ann. N. Y. Acad. Sci. 2005, 1056, 474–485. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Sohn, S.W.; Kee, C. Effect of Ginkgo biloba extract on visual field progression in normal tension glaucoma. J. Glaucoma 2013, 22, 780–784. [Google Scholar] [CrossRef] [PubMed]
- Shim, S.H.; Kim, J.M.; Choi, C.Y.; Kim, C.Y.; Park, K.H. Ginkgo biloba extract and bilberry anthocyanins improve visual function in patients with normal tension glaucoma. J. Med. Food 2012, 15, 818–823. [Google Scholar] [CrossRef] [Green Version]
- Guo, X.; Kong, X.; Huang, R.; Jin, L.; Ding, X.; He, M.; Liu, X.; Patel, M.C.; Congdon, N.G. Effect of Ginkgo biloba on visual field and contrast sensitivity in Chinese patients with normal tension glaucoma: A randomized, crossover clinical trial. Investig. Ophthalmol. Vis. Sci. 2014, 55, 110–116. [Google Scholar] [CrossRef] [Green Version]
- Ohguro, H.; Ohguro, I.; Yagi, S. Effects of black currant anthocyanins on intraocular pressure in healthy volunteers and patients with glaucoma. J. Ocul. Pharmacol. Ther. 2013, 29, 61–67. [Google Scholar] [CrossRef]
- Downie, L.E.; Vingrys, A.J. Oral Omega-3 Supplementation Lowers Intraocular Pressure in Normotensive Adults. Transl. Vis. Sci. Technol. 2018, 7, 3. [Google Scholar] [CrossRef] [Green Version]
- Bazan, N.G.; Scott, B.L. Dietary omega-3 fatty acids and accumulation of docosahexaenoic acid in rod photoreceptor cells of the retina and at synapses. Upsala J. Med. Sci. Suppl. 1990, 48, 97–107. [Google Scholar]
- Saccà, S.C.; Corazza, P.; Gandolfi, S.; Ferrari, D.; Sukkar, S.; Iorio, E.L.; Traverso, C.E. Substances of interest that support glaucoma therapy. Nutrients 2019, 11, 239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.E.; Tseng, V.L.; Yu, F.; Caprioli, J.; Coleman, A.L. Association of dietary fatty acid intake with glaucoma in the United States. JAMA Ophthalmol. 2018, 136, 141–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Villadoniga, R.S.; Rodrıguez Garcıa, E.; Sagastagoia Epelde, O.; Dıaz, M.D.A.; Pedrol, J.C.D. Effects of oral supplementation with docosahexaenoic acid (DHA) plus antioxidants in pseudoexfoliative glaucoma: A 6-month open-label randomized trial. J. Ophthalmol. 2018, 2018, 8259371. [Google Scholar]
- Acar, N.; Creuzot-Garcher, C.P.; Bron, A.M.; Bretillon, L. Dietary polyunsaturated fatty acids, intraocular pressure, and glaucoma. In Handbook of Nutrition, Diet, and the Eye; Academic Press: Cambridge, MA, USA, 2014; Chapter 12. [Google Scholar]
- Pekmezci, M.; Vo, B.; Lim, A.K.; Hirabayashi, D.R.; Tanaka, G.H.; Weinreb, R.N.; Lin, S.C. The characterisation of glaucoma in Japanese Americans. Arch. Ophthalmol. 2009, 127, 167–671. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aydin, B.; Onol, M.; Hondur, A.; Kaya, M.G.; Ozdemir, H.; Cengel, A.; Hasanreisoglu, B. The effect of oral magnesium therapy on visual field and ocular blood flow in normotensive glaucoma. Eur. J. Ophthalmol. 2010, 20, 131–135. [Google Scholar] [CrossRef]
- Flammer, J.; Konieczka, K. The discovery of the Flammer syndrome: A historical and personal perspective. EPMA J. 2017, 8, 75–97. [Google Scholar] [CrossRef]
- Konieczka, K.; Choi, H.J.; Koch, S.; Fankhauser, F.; Schoetzau, A.; Kim, D.M. Relationship between normal tension glaucoma and Flammer syndrome. EPMA J. 2017, 8, 111–117. [Google Scholar] [CrossRef]
- Koc, H.; Kaya, F. Relationship between homocysteine levels, anterior chamber depth, and pseudoexfoliation glaucoma in patients with pseudoexfoliation. Int. Ophthalmol. 2020, 40, 1731–1737. [Google Scholar] [CrossRef]
- Scaglione, F.; Panzavolta, G. Folate, folic acid and 5-methyltetrahydrofolate are not the same thing. Xenobiotica 2014, 44, 480–488. [Google Scholar] [CrossRef]
- Kang, J.H.; Loomis, S.J.; Wiggs, J.L.; Willett, W.C.; Pasquale, L.R. A prospective study of folate, vitamin b6, and vitamin b12 intake in relation to exfoliation glaucoma or suspected exfoliation glaucoma. JAMA Ophthalmol. 2014, 132, 549–559. [Google Scholar] [CrossRef] [Green Version]
- Abu-Amero, K.K.; Kondkar, A.A.; Mousa, A.; Osman, E.A.; Al-Obeidan, S.A. Decreased total antioxidants status in the plasma of patients with pseudoexfoliation glaucoma. Mol. Vis. 2011, 17, 2769–2775. [Google Scholar] [PubMed]
- Ceylan, O.M.; Can Demirdöğen, B.; Mumcuoğlu, T.; Aykut, O. Evaluation of essential and toxic trace elements in pseudoexfoliation syndrome and pseudoexfoliation glaucoma. Biol. Trace Elem. Res. 2013, 153, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Shao, M.; Li, Y.; Li, X.; Wan, Y.; Sun, X.; Cao, W. Relationship between oxidative stress biomarkers and visual field progression in patients with primary angle closure glaucoma. Oxid. Med. Cell. Longev. 2020, 2020, 2701539. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Shao, M.; Tang, B.; Zhang, A.; Cao, W.; Sun, X. The association between serum uric acid and glaucoma severity in primary angle closure glaucoma: A retrospective case-control study. Oncotarget 2017, 8, 2816–2824. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.Y.; Singh, K.; Lin, S.C. Glaucoma and vitamins A, C, and E supplement intake and serum levels in a population-based sample of the United States. Eye 2013, 27, 487–494. [Google Scholar] [CrossRef]
- Zanon-Moreno, V.; Asensio-Marquez, E.M.; Ciancotti-Oliver, L.; Garcia-Medina, J.J.; Sanz, P.; Ortega-Azorin, C.; Pinazo-Duran, M.D.; Ordovás, J.M.; Corella, D. Effects of polymorphisms in vitamin E-, vitamin C-, and glutathione peroxidase-related genes on serum biomarkers and associations with glaucoma. Mol. Vis. 2013, 19, 231–242. [Google Scholar]
- Krefting, E.A.; Jorde, R.; Christoffersen, T.; Grimnes, G. Vitamin D and intraocular pressure—results from a case-control and an intervention study. Acta Ophthalmol. 2014, 92, 345–349. [Google Scholar] [CrossRef]
- Kim, H.T.; Kim, J.M.; Kim, J.H.; Lee, M.Y.; Won, Y.S.; Lee, J.Y.; Park, K.H. The relationship between vitamin D and glaucoma: A Kangbuk Samsung Health Study. Korean J. Ophthalmol. 2016, 30, 426–433. [Google Scholar] [CrossRef] [Green Version]
- Lv, Y.; Yao, Q.; Ma, W.; Liu, H.; Ji, J.; Li, X. Associations of vitamin D deficiency and vitamin D receptor (Cdx-2, Fok I, Bsm I and Taq I) polymorphisms with the risk of primary open-angle glaucoma. BMC Ophthalmol. 2016, 16, 116. [Google Scholar] [CrossRef] [Green Version]
- Turgut, B.; Kaya, M.; Arslan, S.; Demir, T.; Güler, M.; Kaya, M.K. Levels of circulating homocysteine, vitamin B6, vitamin B12, and folate in different types of open-angle glaucoma. Clin. Interv. Aging. 2010, 5, 133–139. [Google Scholar] [CrossRef] [Green Version]
- Mousa, A.; Kondkar, A.A.; Al-Obeidan, S.A.; Azad, T.A.; Sultan, T.; Osman, E.; Abu-Amero, K.K. Association of total antioxidants level with glaucoma type and severity. Saudi Med. J. 2015, 36, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Abu-Amero, K.K.; Kondkar, A.A.; Mousa, A.; Osman, E.A.; Al-Obeidan, S.A. Decreased total antioxidants in patients with primary open angle glaucoma. Curr. Eye Res. 2013, 38, 959–964. [Google Scholar] [CrossRef] [PubMed]
- Aydın Yaz, Y.; Yıldırım, N.; Yaz, Y.; Tekin, N.; İnal, M.; Şahin, F.M. Role of oxidative stress in pseudoexfoliation syndrome and pseudoexfoliation glaucoma. Turk. J. Ophthalmol. 2019, 49, 61–67. [Google Scholar] [CrossRef]
- Renard, J.P.; Rouland, J.F.; Bron, A.; Sellem, E.; Nordmann, J.P.; Baudouin, C.; Denis, P.; Villain, M.; Chaine, G.; Colin, J.; et al. Nutritional, lifestyle and environmental factors in ocular hypertension and primary open-angle glaucoma: An exploratory case-control study. Acta Ophthalmol. 2013, 91, 505–513. [Google Scholar] [CrossRef]
- Garcia-Medina, J.J.; Garcia-Medina, M.; Garrido-Fernandez, P.; Galvan-Espinosa, J.; Garcia-Maturana, C.; Zanon-Moreno, V.; Pinazo-Duran, M.D. A two-year follow-up of oral antioxidant supplementation in primary open-angle glaucoma: An open-label, randomized, controlled trial. Acta Ophthalmol. 2015, 93, 546–554. [Google Scholar] [CrossRef] [PubMed]
- Giaconi, J.A.; Yu, F.; Stone, K.L.; Pedula, K.L.; Ensrud, K.E.; Cauley, J.A.; Hochberg, M.C.; Coleman, A.L.; Study of Osteoporotic Fractures Research Group. The association of consumption of fruits/vegetables with decreased risk of glaucoma among older African-American women in the study of osteoporotic fractures. Am. J. Ophthalmol. 2012, 154, 635–644. [Google Scholar] [CrossRef] [Green Version]
| References | Examined Factor | Risk of Glaucoma Occurrence |
|---|---|---|
| [18] | Low antioxidant intake | ↑ |
| [19] | High antioxidants intake | ↓ |
| [20] | Low vitamin A and vegetable oil intake | ↑ |
| [20,23] | High iron intake | ↑ |
| [34] | Low vitamin B3 intake | ↑ |
| [35] | Low BMI in woman | ↑ |
| [22] | High green leaves (source of NO) intake | ↓ |
| [62] | High vitamin B11 (folic acid) intake | ↓ |
| [76] | Low omega-3 fats intake | ↑ |
| [47] | Anthocyanins and Ginkgo biloba extract administration | Not mentioned, but improvement of visual function |
| References | Examined Factor | Observation | Risk of Glaucoma Occurrence |
|---|---|---|---|
| [7] | Low BAP | Defects in the visual field | ↑ |
| [60] | High homocysteine level | PEX material deposition | ↑ |
| [63] | Low TAS | PEX material deposition | ↑ |
| [65] | Low TAS | Angle closure glaucoma | ↑ |
| [24] | High serum ferritin level | Increased iron resources | ↑ |
| [21] | Low serum uric acid level | Defects in the visual field | ↑ |
| [66] | Low serum uric acid level | Angle closure glaucoma | ↑ |
| [67] | Serum vitamins A, E, C level | No observation | No impact |
| [69] | Serum vitamin D 25 (OH) level | No observation | No impact |
| [64] | Higher serum level of molybdenum, manganese, mercury | PEX material deposition | ↑ |
| [75] | Higher level of MDA | Higher oxidant level | ↑ |
| [36] | Lower level of retinol | Normal tension glaucoma | ↑ |
| [68] | Polymorphism in vitamin C and E | Lower level of vitamin C | ↑ |
| [70] | Lower vitamin D 25 (OH) level | Glaucomatous changes | ↑ |
| [71] | Polymorphism in vitamin D3 | Lower level of vitamin D | ↑ |
| [72] | NTG and POAG occurrence | Higher level of vitamin B6 | ↑ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Młynarczyk, M.; Falkowska, M.; Micun, Z.; Obuchowska, I.; Kochanowicz, J.; Socha, K.; Konopińska, J. Diet, Oxidative Stress, and Blood Serum Nutrients in Various Types of Glaucoma: A Systematic Review. Nutrients 2022, 14, 1421. https://doi.org/10.3390/nu14071421
Młynarczyk M, Falkowska M, Micun Z, Obuchowska I, Kochanowicz J, Socha K, Konopińska J. Diet, Oxidative Stress, and Blood Serum Nutrients in Various Types of Glaucoma: A Systematic Review. Nutrients. 2022; 14(7):1421. https://doi.org/10.3390/nu14071421
Chicago/Turabian StyleMłynarczyk, Maryla, Martyna Falkowska, Zuzanna Micun, Iwona Obuchowska, Jan Kochanowicz, Katarzyna Socha, and Joanna Konopińska. 2022. "Diet, Oxidative Stress, and Blood Serum Nutrients in Various Types of Glaucoma: A Systematic Review" Nutrients 14, no. 7: 1421. https://doi.org/10.3390/nu14071421
APA StyleMłynarczyk, M., Falkowska, M., Micun, Z., Obuchowska, I., Kochanowicz, J., Socha, K., & Konopińska, J. (2022). Diet, Oxidative Stress, and Blood Serum Nutrients in Various Types of Glaucoma: A Systematic Review. Nutrients, 14(7), 1421. https://doi.org/10.3390/nu14071421








