Effectiveness of Nutritional Ingredients on Upper Gastrointestinal Conditions and Symptoms: A Narrative Review
Abstract
1. Introduction
1.1. Background on Upper GI Issues
1.2. Background on Nutritional Ingredients and Use for Upper GI Support
1.3. Primary Aim of This Review
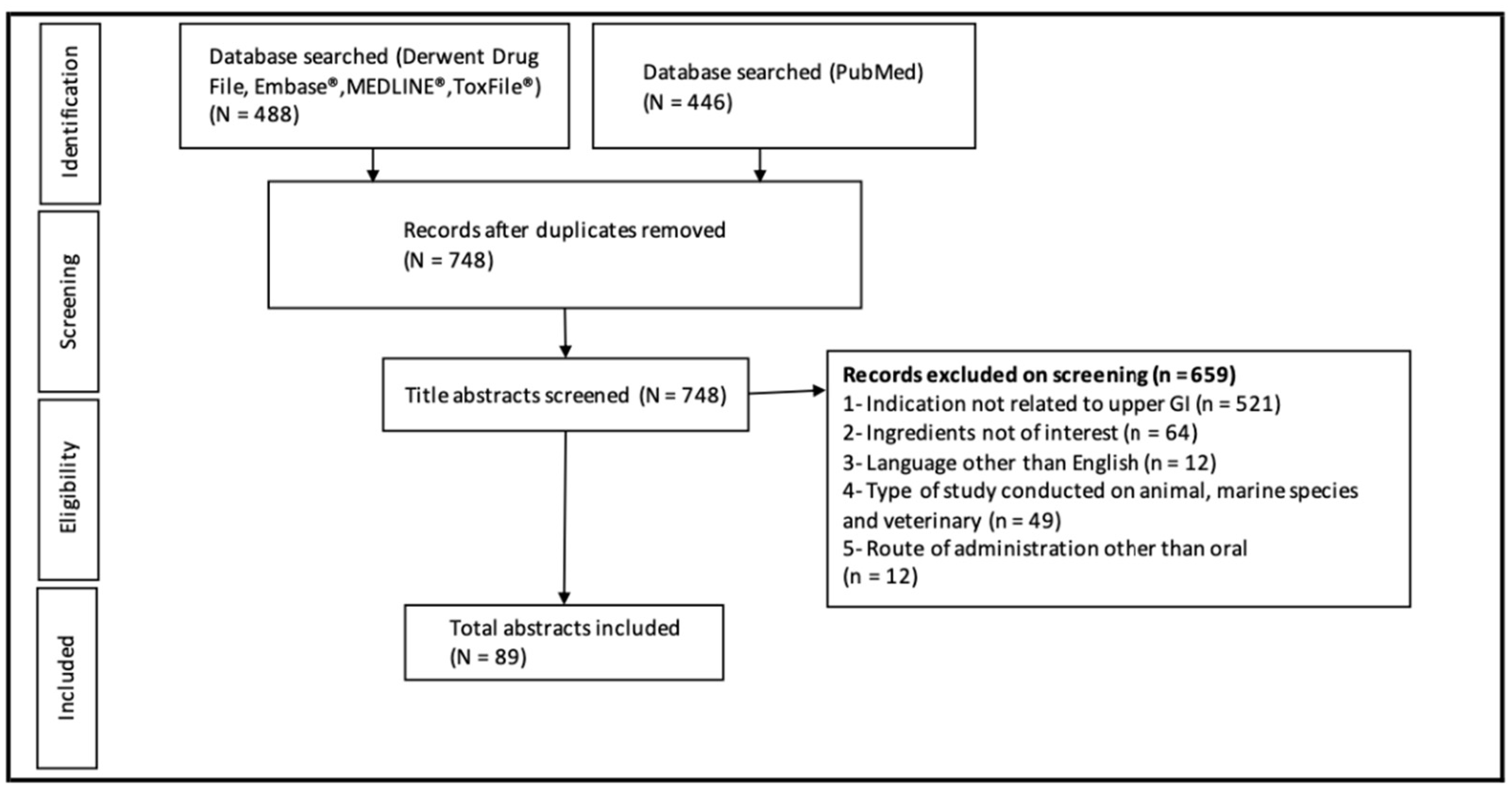
2. Materials and Methods
3. Results and Discussion
3.1. Identified Trials
3.2. Botanical Ingredients Addressing Heartburn, GERD, and Gastric Conditions: Fiber, Other Botanicals, and Combinations
3.2.1. Fiber
Fenugreek and Galactomannan
Marine Alginate
3.2.2. “Other” Botanicals
Aloe Vera
Apple Cider Vinegar
Cardamom
D-limonene
Fennel
Ginger
Licorice
Papaya
3.2.3. Combination Products
Peppermint Oil and Caraway Oil
3.3. Non-Botanical Ingredients Addressing Heartburn, GERD, and Gastric Conditions
3.3.1. Activated Charcoal
3.3.2. Melatonin
3.3.3. Zinc-l-carnosine
3.4. Areas for Improvement of Future Clinical Trials
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Andreasson, A.; Talley, N.J.; Walker, M.M.; Jones, M.P.; Platts, L.G.; Wallner, B.; Kjellstrom, L.; Hellstrom, P.M.; Forsberg, A.; Agrocus, L. An Increasing Incidence of Upper Gastrointestinal Disorders Over 23 Years: A Prospective Population-Based Study in Sweden. Am. J. Gastroenterol. 2021, 116, 210–213. [Google Scholar] [CrossRef] [PubMed]
- Sperber, A.D.; Bangdiwala, S.I.; Drossman, D.A.; Ghoshal, U.C.; Simren, M.; Tack, J.; Whitehead, W.E.; Dumitrascu, D.L.; Fang, X.; Fukudo, S.; et al. Worldwide Prevalence and Burden of Functional Gastrointestinal Disorders, Results of Rome Foundation Global Study. Gastroenterology 2021, 160, 99–114.e3. [Google Scholar] [CrossRef] [PubMed]
- Black, C.J.; Drossman, D.A.; Talley, N.J.; Ruddy, J.; Ford, A.C. Functional gastrointestinal disorders: Advances in understanding and management. Lancet 2020, 396, 1664–1674. [Google Scholar] [CrossRef]
- Delshad, S.D.; Almario, C.V.; Chey, W.D.; Spiegel, B.M.R. Prevalence of Gastroesophageal Reflux Disease and Proton Pump Inhibitor-Refractory Symptoms. Gastroenterology 2020, 158, 1250–1261.e2. [Google Scholar] [CrossRef] [PubMed]
- Fass, R.; Achem, S.R. Noncardiac chest pain: Epidemiology, natural course and pathogenesis. J. Neurogastroenterol. Motil. 2011, 17, 110–123. [Google Scholar] [CrossRef] [PubMed]
- De Brito, B.B.; da Silva, F.A.F.; Soares, A.S.; Pereira, V.A.; Santos, M.L.C.; Sampaio, M.M.; Neves, P.H.M.; de Melo, F.F. Pathogenesis and clinical management of Helicobacter pylori gastric infection. World J. Gastroenterol. 2019, 25, 5578–5589. [Google Scholar] [CrossRef]
- Drossman, D.A. Functional Gastrointestinal Disorders: History, Pathophysiology, Clinical Features, and Rome IV. Gastroenterology 2016, 150, 1262–1279.e2. [Google Scholar] [CrossRef]
- Peery, A.F.; Crockett, S.D.; Murphy, C.C.; Lund, J.L.; Dellon, E.S.; Williams, J.L.; Jensen, E.T.; Shaheen, N.J.; Barritt, A.S.; Lieber, S.R.; et al. Burden and Cost of Gastrointestinal, Liver, and Pancreatic Diseases in the United States: Update 2018. Gastroenterology 2019, 156, 254–272.e11. [Google Scholar] [CrossRef]
- Fifi, A.C.; Axelrod, C.H.; Chakraborty, P.; Saps, M. Herbs and Spices in the Treatment of Functional Gastrointestinal Disorders: A Review of Clinical Trials. Nutrients 2018, 10, 1715. [Google Scholar] [CrossRef]
- Smith, T.; May, G.; Eckl, V.; Morton Reynolds, C. US Sales of Herbal Supplements Increase by 8.6% in 2019. HerbalGram 2020, 127, 54–69. [Google Scholar]
- Lahner, E.; Bellentani, S.; Bastiani, R.D.; Tosetti, C.; Cicala, M.; Esposito, G.; Arullani, P.; Annibale, B. A survey of pharmacological and nonpharmacological treatment of functional gastrointestinal disorders. United Eur. Gastroenterol. J. 2013, 1, 385–393. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef]
- Schardt, C.; Adams, M.B.; Owens, T.; Keitz, S.; Fontelo, P. Utilization of the PICO framework to improve searching PubMed for clinical questions. BMC Med. Inf. Decis. Mak. 2007, 7, 16. [Google Scholar] [CrossRef]
- DiSilvestro, R.A.; Verbruggen, M.A.; Offutt, E.J. Anti-heartburn effects of a fenugreek fiber product. Phytother. Res. 2011, 25, 88–91. [Google Scholar] [CrossRef] [PubMed]
- Abenavoli, L.; Luigiano, C.; Pendlimari, R.; Fagoonee, S.; Pellicano, R. Efficacy and tolerability of a novel galactomannan-based formulation for symptomatic treatment of gastroesophageal reflux disease: A randomized, double-blind, placebo-controlled study. Eur. Rev. Med. Pharm. Sci. 2021, 25, 4128–4138. [Google Scholar]
- Yao, D.; Zhang, B.; Zhu, J.; Zhang, Q.; Hu, Y.; Wang, S.; Wang, Y.; Cao, H.; Xiao, J. Advances on application of fenugreek seeds as functional foods: Pharmacology, clinical application, products, patents and market. Crit. Rev. Food Sci. Nutr. 2020, 60, 2342–2352. [Google Scholar] [CrossRef] [PubMed]
- Kovalik, A.C.; Bisetto, P.; Pochapski, M.T.; Campagnoli, E.B.; Pilatti, G.L.; Santos, F.A. Effects of an orabase formulation with ethanolic extract of Malva sylvestris L. in oral wound healing in rats. J. Med. Food. 2014, 17, 618–624. [Google Scholar] [CrossRef] [PubMed]
- Maton, P.N.; Burton, M.E. Antacids revisited: A review of their clinical pharmacology and recommended therapeutic use. Drugs 1999, 57, 855–870. [Google Scholar] [CrossRef] [PubMed]
- Gasparetto, J.C.; Martins, C.A.F.; Hayashi, S.S.; Otuky, M.F.; Pontarolo, R. Ethnobotanical and scientific aspects of Malva sylvestris L.: A millennial herbal medicine. J. Pharm. Pharmacol. 2012, 64, 172–189. [Google Scholar] [CrossRef]
- Ialenti, A.; Di Rosa, M. Hyaluronic acid modulates acute and chronic inflammation. Agents Actions 1994, 43, 44–47. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Liu, J.; Li, L.; Liu, W.; Sun, M. A brief review of nutraceutical ingredients in gastrointestinal disorders: Evidence and suggestions. Int. J. Mol. Sci. 2020, 21, 1822. [Google Scholar] [CrossRef] [PubMed]
- Kapadia, C.J.; Mane, V.B. Raft-Forming Agents: Antireflux Formulations. Drug Dev. Ind. Pharm. 2007, 33, 1350–1361. [Google Scholar] [CrossRef]
- Suja Pandian, R.; Anuradha, C.V.; Viswanathan, P. Gastroprotective effect of fenugreek seeds (Trigonella foenum graecum) on experimental gastric ulcer in rats. J. Ethnopharmacol. 2002, 81, 393–397. [Google Scholar] [CrossRef]
- Phillips, G.O.; Williams, P.A. (Eds.) Hydrocolloid Thickeners and Their Applications. In Gums and Stabilisers for the Food Industry 12; Royal Society of Chemistry: Cambridge, UK, 2004; Volume 12. [Google Scholar]
- Morozov, S.; Isakov, V.; Konovalova, M. Fiber-enriched diet helps to control symptoms and improves esophageal motility in patients with non-erosive gastroesophageal reflux disease. World J. Gastroenterol. 2018, 24, 2291–2299. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.X.; Wang, J.W.; Gong, M. Efficacy and safety of alginate formulations in patients with gastroesophageal reflux disease: A systematic review and meta-analysis of randomized controlled trials. Eur. Rev. Med. Pharm. Sci. 2020, 24, 11845–11857. [Google Scholar]
- Panahi, Y.; Aslani, J.; Hajihashemi, A.; Kalkhorani, M.; Ghanei, M.; Sahebkar, A. Effect of aloe vera and pantoprazole on Gastroesophageal reflux symptoms in mustard gas victims: A randomized controlled trial. Pharm. Sci. 2016, 22, 190–194. [Google Scholar] [CrossRef][Green Version]
- Panahi, Y.; Khedmat, H.; Valizadegan, G.; Mohtashami, R.; Sahebkar, A. Efficacy and safety of Aloe vera syrup for the treatment of gastro-esophageal reflux disease: A pilot randomized positive-controlled trial. J. Tradit. Chin. Med. 2015, 35, 632–636. [Google Scholar] [CrossRef]
- Attari, V.E.; Somi, M.H.; Jafarabadi, M.A.; Ostadrahimi, A.; Moaddab, S.-Y.; Lotfi, N. The gastro-protective effect of ginger (Zingiber officinale roscoe) in Helicobacter pylori positive functional dyspepsia. Adv. Pharm. Bull. 2019, 9, 321–324. [Google Scholar] [CrossRef]
- Panda, S.K.; Nirvanashetty, S.; Parachur, V.A.; Krishnamoorthy, C.; Dey, S. A Randomized, Double-Blind, Placebo Controlled, Parallel-Group, Comparative Clinical Study to Evaluate the Efficacy and Safety of OLNP-06 versus Placebo in Subjects with Functional Dyspepsia. J. Diet. Suppl. 2020, 1–13. [Google Scholar] [CrossRef]
- Bhargava, R.; Chasen, M.; Elten, M.; MacDonald, N. The effect of ginger (Zingiber officinale Roscoe) in patients with advanced cancer. Support Care Cancer 2020, 28, 3279–3286. [Google Scholar] [CrossRef]
- Prajapati, S.M.; Patel, B.R. A comparative clinical study of Jethimala (Taverniera nummularia Baker.) and Yashtimadhu (Glycyrrhiza glabra Linn.) in the management of Amlapitta. Ayu 2015, 36, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Raveendra, K.R.; Jayachandra; Srinivasa, V.; Sushma, K.R.; Allan, J.J.; Goudar, K.S.; Shivaprasad, H.N.; Venkateshwarlu, K.; Geetharani, P.; Sushma, G.; et al. An Extract of Glycyrrhiza glabra (GutGard) Alleviates Symptoms of Functional Dyspepsia: A Randomized, Double-Blind, Placebo-Controlled Study. Evid.-Based Complement. Altern. Med. 2012, 2012, 216970. [Google Scholar] [CrossRef] [PubMed]
- Muss, C.; Mosgoeller, W.; Endler, T. Papaya preparation (CaricolB®) in digestive disorders. Neuro-Endocrinol. Lett. 2013, 34, 38–46. [Google Scholar] [PubMed]
- Weiser, F.-A.; Fangl, M.; Mosgoeller, W. Supplementation of CaricolB®-Gastro reduces chronic gastritis disease associated pain. Neuro-Endocrinol. Lett. 2018, 39, 19–25. [Google Scholar] [PubMed]
- Park, C.H.; Son, H.U.; Yoo, C.Y.; Lee, S.H. Low molecular-weight gel fraction of Aloe vera exhibits gastroprotection by inducing matrix metalloproteinase-9 inhibitory activity in alcohol-induced acute gastric lesion tissues. Pharm. Biol. 2017, 55, 2110–2115. [Google Scholar] [CrossRef] [PubMed]
- Pazyar, N.; Yaghoobi, R.; Rafiee, E.; Mehrabian, A.; Feily, A. Skin wound healing and phytomedicine: A review. Ski. Pharm. Physiol. 2014, 27, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, N. Inflammation and oxidative stress in gastroesophageal reflux disease. J. Clin. Biochem. Nutr. 2007, 40, 13–23. [Google Scholar] [CrossRef]
- Nahar, T.; Uddin, B.; Hossain, S.; Sikder, A.M.; Ahmed, S. Aloe vera gel protects liver from oxidative stress-induced damage in experimental rat model. J. Complement. Integr. Med. 2013, 10, 1–7. [Google Scholar] [CrossRef]
- Duansak, D.; Somboonwong, J.; Patumraj, S. Effects of Aloe vera on leukocyte adhesion and TNF-O;± and IL-6 levels in burn wounded rats. Clin. Hemorheol. Microcirc. 2003, 29, 239–246. [Google Scholar]
- Johnston, C.S.; Kim, C.M.; Buller, A.J. Vinegar Improves Insulin Sensitivity to a High-Carbohydrate Meal in Subjects with Insulin Resistance or Type 2 Diabetes. Diabetes Care 2004, 27, 281–282. [Google Scholar] [CrossRef]
- Leeman, M.; Ostman, E.; Björck, I. Vinegar dressing and cold storage of potatoes lowers postprandial glycaemic and insulinaemic responses in healthy subjects. Eur. J. Clin. Nutr. 2005, 59, 1266–1271. [Google Scholar] [CrossRef]
- Ostman, E.; Granfeldt, Y.; Persson, L.; Björck, I. Vinegar supplementation lowers glucose and insulin responses and increases satiety after a bread meal in healthy subjects. Eur. J. Clin. Nutr. 2005, 59, 983–988. [Google Scholar] [CrossRef]
- Brown, R.; Sam, C.H.Y.; Green, T.; Wood, S. Effect of GutsyGumm, A Novel Gum, on Subjective Ratings of Gastro Esophageal Reflux Following a Refluxogenic Meal. J. Diet. Suppl. 2015, 12, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Ozgoli, G.; Saei Ghare Naz, M. Effects of Complementary Medicine on Nausea and Vomiting in Pregnancy: A Systematic Review. Int. J. Prev. Med. 2018, 9, 75. [Google Scholar] [PubMed]
- Ozgoli, G.; Gharayagh Zandi, M.; Nazem Ekbatani, N.; Allavi, H.; Moattar, F. Cardamom powder effect on nausea and vomiting during pregnancy. Complement. Med. J. 2015, 14, 1056–1076. [Google Scholar]
- Jafri, M.A.; Farah; Javed, K.; Singh, S. Evaluation of the gastric antiulcerogenic effect of large cardamom (fruits of Amomum subulatum Roxb). J. Ethnopharmacol. 2001, 75, 89–94. [Google Scholar] [CrossRef]
- Jamal, A.; Javed, K.; Aslam, M.; Jafri, M.A. Gastroprotective effect of cardamom, Elettaria cardamomum Maton. fruits in rats. J. Ethnopharmacol. 2006, 103, 149–153. [Google Scholar] [CrossRef]
- Anandakumar, P.; Kamaraj, S.; Vanitha, M.K. Dlimonene: A multifunctional compound with potent therapeutic effects. J. Food Biochem. 2021, 45, e13566. [Google Scholar] [CrossRef]
- Wilkins, J.S., Jr. Method for Treating Gastrointestinal Disorder. U.S. Patent US6420435B1, 16 July 2002. [Google Scholar]
- Asano, T.; Takenaga, M. Restoration of delayed gastric emptying and impaired gastric compliance by anethole in an animal model of functional dyspepsia. FASEB J. 2018, 32, 840.7. [Google Scholar] [CrossRef]
- Savino, F.; Cresi, F.; Castagno, E.; Silvestro, L.; Oggero, R. A randomized double-blind placebo-controlled trial of a standardized extract of Matricariae recutita, Foeniculum vulgare and Melissa officinalis (ColiMil) in the treatment of breastfed colicky infants. Phytother. Res. 2005, 19, 335–340. [Google Scholar] [CrossRef]
- Vakil, N.; Laine, L.; Talley, N.J.; Zakko, S.F.; Tack, J.; Chey, W.D.; Kralstein, J.; Earnest, D.L.; Ligozio, G.; Cohard-Radice, M. Tegaserod treatment for dysmotility-like functional dyspepsia: Results of two randomized, controlled trials. Am. J. Gastroenterol. 2008, 103, 1906–1919. [Google Scholar] [CrossRef] [PubMed]
- Palatty, P.L.; Haniadka, R.; Valder, B.; Arora, R.; Baliga, M.S. Ginger in the Prevention of Nausea and Vomiting: A Review. Crit. Rev. Food Sci. Nutr. 2013, 53, 659–669. [Google Scholar] [CrossRef]
- Mohd Yusof, Y.A. Gingerol and Its Role in Chronic Diseases. In Drug Discovery from Mother Nature; Gupta, S.C., Prasad, S., Aggarwal, B.B., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 177–207. [Google Scholar]
- Nikkhah Bodagh, M.; Maleki, I.; Hekmatdoost, A. Ginger in gastrointestinal disorders: A systematic review of clinical trials. Food Sci. Nutr. 2019, 7, 96–108. [Google Scholar] [CrossRef]
- Anh, N.H.; Kim, S.J.; Long, N.P.; Min, J.E.; Yoon, Y.C.; Lee, E.G.; Kim, M.; Kim, T.J.; Yang, Y.Y.; Son, E.Y.; et al. Ginger on Human Health: A Comprehensive Systematic Review of 109 Randomized Controlled Trials. Nutrients 2020, 12, 157. [Google Scholar] [CrossRef]
- Phillips, S.; Hutchinson, S.; Ruggier, R. Zingiber officinale does not affect gastric emptying rate: A randomised, placebo-controlled, crossover trial. Anaesthesia 1993, 48, 393–395. [Google Scholar] [CrossRef]
- Lien, H.C.; Sun, W.M.; Chen, Y.H.; Kim, H.; Hasler, W.; Owyang, C. Effects of ginger on motion sickness and gastric slow-wave dysrhythmias induced by circular vection. Am. J. Physiol. Gastrointest. Liver Physiol. 2003, 284, G481–G489. [Google Scholar] [CrossRef] [PubMed]
- Gonlachanvit, S.; Chen, Y.H.; Hasler, W.L.; Sun, W.M.; Owyang, C. Ginger reduces hyperglycemia-evoked gastric dysrhythmias in healthy humans: Possible role of endogenous prostaglandins. J. Pharm. Exp. Ther. 2003, 307, 1098–1103. [Google Scholar] [CrossRef]
- Mentis, A.; Lehours, P.; Mégraud, F. Epidemiology and Diagnosis of Helicobacter pylori infection. Helicobacter. 2015, 20, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Selgrad, M.; Kandulski, A.; Malfertheiner, P. Dyspepsia and Helicobacter pylori. Dig. Dis. 2008, 26, 210–214. [Google Scholar] [CrossRef] [PubMed]
- Ali, B.H.; Blunden, G.; Tanira, M.O.; Nemmar, A. Some phytochemical, pharmacological and toxicological properties of ginger (Zingiber officinale Roscoe): A review of recent research. Food Chem. Toxicol. 2008, 46, 409–420. [Google Scholar] [CrossRef]
- Jalilzadeh-Amin, G.; Najarnezhad, V.; Anassori, E.; Mostafavi, M.; Keshipour, H. Antiulcer properties of Glycyrrhiza glabra L. extract on experimental models of gastric ulcer in mice. Iran. J. Pharm. Res. 2015, 14, 1163–1170. [Google Scholar] [PubMed]
- Bennett, A.; Clark-Wibberley, T.; Stamford, I.F.; Wright, J.E. Aspirin-induced gastric mucosal damage in rats: Cimetidine and degly-cyhrrhizinated liquorice together give greater protection than low doses of either drug alone. J. Pharm. Pharmacol. 1980, 32, 151. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wang, S.; Bao, Y.-R.; Li, T.-j.; Yang, G.-L.; Chang, X.; Meng, X.-S. Anti-ulcer effect and potential mechanism of licoflavone by regulating inflammation mediators and amino acid metabolism. J. Ethnopharmacol. 2017, 199, 175–182. [Google Scholar] [CrossRef] [PubMed]
- HMPC. Assessment Report on Glycyrrhiza glabr L. and/or Glycyrrhiza inflata Bat. and/or Glycyrrhiza uralensis Fisch. radix; European Medicine Agency: London, UK, 2013; pp. 1–40.
- Asha, M.Z.; Khalil, S.F.H. Efficacy and Safety of Probiotics, Prebiotics and Synbiotics in the Treatment of Irritable Bowel Syndrome: A systematic review and meta-analysis. Sultan Qaboos Univ. Med. J. 2020, 20, e13–e24. [Google Scholar] [CrossRef]
- Osato, J.A.; Santiago, L.A.; Remo, G.M.; Cuadra, M.S.; Mori, A. Antimicrobial and antioxidant activities of unripe papaya. Life Sci. 1993, 53, 1383–1389. [Google Scholar] [CrossRef]
- Chen, C.F.; Chen, S.M.; Chow, S.Y.; Han, P.W. Protective effects of Carica papaya Linn on the exogenous gastric ulcer in rats. Am. J. Chin. Med. 1981, 9, 205–212. [Google Scholar] [CrossRef]
- Cho, C.H.; Han, P.W. Papain reduces gastric acid secretion induced by histamine and other secretagogues in anesthetized rats. Proc. Natl. Sci. Counc. Repub. China B 1984, 8, 177–181. [Google Scholar]
- Annahazi, A.; Schroeder, A.; Schemann, M. Region-specific effects of the cysteine protease papain on gastric motility. Neurogastroenterol. Motil. 2021, 33, e14105. [Google Scholar] [CrossRef]
- Ried, K.; Travica, N.; Dorairaj, R.; Sali, A. Herbal formula improves upper and lower gastrointestinal symptoms and gut health in Australian adults with digestive disorders. Nutr. Res. 2020, 76, 37–51. [Google Scholar] [CrossRef]
- Ahuja, A.; Ahuja, N.K. Popular Remedies for Esophageal Symptoms: A Critical Appraisal. Curr. Gastroenterol. Rep. 2019, 21, 39. [Google Scholar] [CrossRef]
- Moazzez, R.; Bartlett, D.; Anggiansah, A. The Effect of Chewing Sugar-free Gum on Gastro-esophageal Reflux. J. Dent. Res. 2005, 84, 1062–1065. [Google Scholar] [CrossRef] [PubMed]
- Chumpitazi, B.P.; Kearns, G.L.; Shulman, R.J. Review article: The physiological effects and safety of peppermint oil and its efficacy in irritable bowel syndrome and other functional disorders. Aliment. Pharm. Ther. 2018, 47, 738–752. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Lv, L.; Zhang, J.; Xu, L.; Zeng, E.; Zhang, Z.; Wang, F.; Tang, X. A Combination of Peppermint Oil and Caraway Oil for the Treatment of Functional Dyspepsia: A Systematic Review and Meta-Analysis. Evid.-Based Complement. Altern. Med. 2019, 2019, 7654947. [Google Scholar] [CrossRef] [PubMed]
- Botschuijver, S.; Welting, O.; Levin, E.; Maria-Ferreira, D.; Koch, E.; Montijn, R.C.; Seppen, J.; Hakvoort, T.B.M.; Schuren, F.H.J.; de Jonge, W.J.; et al. Reversal of visceral hypersensitivity in rat by Menthacarin, a proprietary combination of essential oils from peppermint and caraway, coincides with mycobiome modulation. Neurogastroenterol. Motil. 2018, 30, e13299. [Google Scholar] [CrossRef] [PubMed]
- Micklefield, G.; Jung, O.; Greving; May, B. Effects of intraduodenal application of peppermint oil (WS (R) 1340) and caraway oil (WS (R) 1520) on gastroduodenal motility in healthy volunteers. Phytother. Res. 2003, 17, 135–140. [Google Scholar] [CrossRef]
- Lecuyer, M.; Cousin, T.; Monnot, M.N.; Coffin, B. Efficacy of an activated charcoal-simethicone combination in dyspeptic syndrome: Results of a placebo-controlled prospective study in general practice. Gastroentérol. Clin. Biol. 2009, 33, 478–484. [Google Scholar] [CrossRef]
- Coffin, B.; Bortolloti, C.; Bourgeois, O.; Denicourt, L. Efficacy of a simethicone, activated charcoal and magnesium oxide combination (CarbosymagB®) in functional dyspepsia: Results of a general practice-based randomized trial. Clin. Res. Hepatol. Gastroenterol. 2011, 35, 494–499. [Google Scholar] [CrossRef]
- Tan, B.; Luo, H.Q.; Xu, H.; Lv, N.H.; Shi, R.H.; Luo, H.S.; Li, J.S.; Ren, J.L.; Zou, Y.Y.; Li, Y.Q.; et al. Polaprezinc combined with clarithromycin-based triple therapy for Helicobacter pylori-associated gastritis: A prospective, multicenter, randomized clinical trial. PLoS ONE 2017, 12, e0175625. [Google Scholar] [CrossRef]
- Potter, T.; Ellis, C.; Levitt, M. Activated charcoal: In vivo and in vitro studies of effect on gas formation. Gastroenterology 1985, 88, 620–624. [Google Scholar] [CrossRef]
- Hall, R.G., Jr.; Thompson, H.; Strother, A. Effects of orally administered activated charcoal on intestinal gas. Am. J. Gastroenterol. 1981, 75, 192–196. [Google Scholar]
- Jain, N.K.; Patel, V.P.; Pitchumoni, S. Activated charcoal, simethicone, and intestinal gas: A double-blind study. Ann. Intern. Med. 1986, 105, 61–62. [Google Scholar] [CrossRef]
- Jain, N.K.; Patel, V.P.; Pitchumoni, C.S. Efficacy of activated charcoal in reducing intestinal gas: A double-blind clinical trial. Am. J. Gastroenterol. 1986, 81, 532–535. [Google Scholar] [PubMed]
- Holtmann, G.; Gschossmann, J.; Karaus, M.; Fischer, T.; Becker, B.; Mayr, P.; Gerken, G. Randomised double-blind comparison of simethicone with cisapride in functional dyspepsia. Aliment. Pharmacol. Ther. 1999, 13, 1459–1465. [Google Scholar] [CrossRef] [PubMed]
- Holtmann, G.; Gschossmann, J.; Mayr, P.; Talley, N.J. A randomized placebo-controlled trial of simethicone and cisapride for the treatment of patients with functional dyspepsia. Aliment. Pharmacol. Ther. 2002, 16, 1641–1648. [Google Scholar] [CrossRef] [PubMed]
- Klupińska, G.; Wiśniewska-Jarosińska, M.; Harasiuk, A.; Chojnacki, C.; Stec-Michalska, K.; Błasiak, J.; Reiter, R.J.; Chojnacki, J. Nocturnal secretion of melatonin in patients with upper digestive tract disorders. J. Physiol. Pharm. 2006, 57 (Suppl. 5), 41–50. [Google Scholar]
- Kandil, T.S.; Mousa, A.A.; El-Gendy, A.A.; Abbas, A.M. The potential therapeutic effect of melatonin in Gastro-Esophageal Reflux Disease. BMC Gastroenterol. 2010, 10, 7. [Google Scholar] [CrossRef]
- Bang, C.S.; Yang, Y.J.; Baik, G.H. Melatonin for the treatment of gastroesophageal reflux disease; protocol for a systematic review and meta-analysis. Medicine 2019, 98, e14241. [Google Scholar] [CrossRef]
- Kato, K.; Murai, I.; Asai, S.; Takahashi, Y.; Matsuno, Y.; Komuro, S.; Kurosaka, H.; Iwasaki, A.; Ishikawa, K.; Arakawa, Y. Central nervous system action of melatonin on gastric acid and pepsin secretion in pylorus-ligated rats. Neuroreport 1998, 9, 3989–3992. [Google Scholar] [CrossRef]
- Konturek, S.J.; Konturek, P.C.; Brzozowska, I.; Pawlik, M.; Sliwowski, Z.; Cześnikiewicz-Guzik, M.; Kwiecień, S.; Brzozowski, T.; Bubenik, G.A.; Pawlik, W.W. Localization and biological activities of melatonin in intact and diseased gastrointestinal tract (GIT). J. Physiol. Pharm. 2007, 58, 381–405. [Google Scholar]
- Miyoshi, A.; Matsuo, H.; Miwa, T.; Namiki, M.; Taniuchi, A.; Masamune, K.; Asaki, S.; Mori, H.; Nakajima, M. Clinical Evaluation of Z-103 on Gastric Ulcer: A multi-center double blind comparative study with Cetraxate Hydrochloride. Jpn. Pharm. Ther. 1992, 20, 199–223. [Google Scholar]
- Hiraishi, H.; Sasai, T.; Oinuma, T.; Shimada, T.; Sugaya, H.; Terano, A. Polaprezinc protects gastric mucosal cells from noxious agents through antioxidant properties in vitro. Aliment Pharm. Ther. 1999, 13, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Matsukura, T.; Tanaka, H. Applicability of zinc complex of L-carnosine for medical use. Biochemistry 2000, 65, 817–823. [Google Scholar] [PubMed]
- Hudson, T. A combination of zinc and L-carnosine improves gastric ulcers. Nat. Med. J. 2013, 5, 1–8. [Google Scholar]
- Ko, J.K.; Leung, C.C. Ginger extract and polaprezinc exert gastroprotective actions by anti-oxidant and growth factor modulating effects in rats. J. Gastroenterol. Hepatol. 2010, 25, 1861–1869. [Google Scholar] [CrossRef]
- Ito, M.; Tanaka, T.; Suzuki, Y. Effect of N-(3-Aminopropionyl)-L-Histidinato Zinc (Z-103) on Healing and Hydrocortisone-Induced Relapse of Acetic Acid Ulcers in Rats with Limited Food-Intake-Time. Jpn. J. Pharmacol. 1990, 52, 513–521. [Google Scholar] [CrossRef]
- Choi, H.S.; Lim, J.Y.; Chun, H.J.; Lee, M.; Kim, E.S.; Keum, B.; Seo, Y.S.; Jeen, Y.T.; Um, S.H.; Lee, H.S.; et al. The effect of polaprezinc on gastric mucosal protection in rats with ethanol-induced gastric mucosal damage: Comparison study with rebamipide. Life Sci. 2013, 93, 69–77. [Google Scholar] [CrossRef]
- Naito, Y.; Yoshikawa, T.; Yagi, N.; Matsuyama, K.; Yoshida, N.; Seto, K.; Yoneta, T. Effects of polaprezinc on lipid peroxidation, neutrophil accumulation, and TNF-alpha expression in rats with aspirin-induced gastric mucosal injury. Dig. Dis. Sci. 2001, 46, 845–851. [Google Scholar] [CrossRef]
| Ingredient | Alternative Name |
|---|---|
| Activated Charcoal | Activated Carbon |
| Aloe Vera | Aloe barbadensis Miller; Aloe aborescens |
| Apple Cider Vinegar | ACV |
| Ashwagandha | Withania somnifera |
| Burdock Root | Arctium lappa |
| Cardamom | Zingiberaceae elettaria; Zingiberaceae amomum |
| Chamomile | Matricaria recutita |
| Chicory | Cichorium intybus |
| Clove Oil | Syzygium aromaticum |
| Dandelion | Taraxacum officinale |
| D-Limonene | Limonene |
| Fennel | Foeniculum vulgare |
| Fenugreek | Trigonella foenum-graecum |
| Galactomannan | - |
| Ginger | Zingiber officinale Roscoe |
| Gum Arabic | Acacia gum |
| Lemon Balm | Melissa officinalis |
| Licorice | Glycyrrhiza glabra |
| Marine Alginate | Alginate |
| Melatonin | N-acetyl-5-methoxytryptamine |
| Papaya | Carica papaya, papain, papaw |
| Partially Hydrolyzed Guar Gum | PHGG |
| Peppermint | Mentha pipereta |
| Slippery Elm | Ulmus rubra |
| Zinc-L-Carnosine | Z-103, Polaprezinc, L-CAZ, N(3 aminopropionyl)-L-histidine |
| Parameter | Criteria | Exclusion |
|---|---|---|
| Population | Healthy and occasionally or chronically ill adults (≥18 years) | Individuals <18 years, animals, in vitro |
| Intervention | Galactomannan, Gum Arabic, Partially Hydrolyzed Guar Gum, Fenugreek, Zinc Carnosine, Chicory, Burdock Root Arctium, Slippery Elm, Activated Charcoal, Clove Oil, Papaya, Cardamom, Ginger, Fennel, Aloe, Chamomile, Lemon Balm, Dandelion, Ashwagandha, Peppermint, Marine Alginate, Melatonin, Apple Cider Vinegar, Licorice, and D-Limonene | N/A |
| Comparator | Placebo, control, none, or standard care | N/A |
| Outcome | Improvement of upper GI symptoms | N/A |
| Study Design | RCTs, clinical studies, and review articles | Case reports, observational studies, editorials, comments, notes, and letters |
| S. No | Searched for | Databases |
|---|---|---|
| 1 | Key words used for intervention: ti,ab(“Withania somnifera” OR Ashwagandha OR Taraxacum OR “yellow flowers” OR Dandelion OR “Melissa officinalis” OR “Lemon balm” OR Camomile OR Chamomile OR “Matricaria chamomilla” OR Aloe OR “Foeniculum vulgare” OR fennel OR “Zingiber officinale” OR Ginger OR cardamon OR cardamum OR Elettaria OR “Carica papaya” OR papaw OR papain OR “papaya” OR eugenol OR “clove oil” OR “Activated charcoal” OR “Ulmus rubra” OR “Slippery elm” OR “Burdock root “ OR Arctium OR “Cichorium intybus” OR “Zinc carnosine” OR “Trigonella foenum” OR “gum sudani” OR “acacia gum” OR “Arabic gum” OR “gum acacia” OR “Senegal gum” OR “Gum Arabic” OR “galactomannan” OR “Partially hydrolyzed guar gum” OR “Mentha piperita” OR “peppermint oil” OR “marine alginate” OR “alginate” OR “Melatonin” OR “N-acetyl-5-methoxytryptamine” OR “D-limonene” OR “Limonene” OR “Apple cider vinegar” OR “ACV” OR “Licorice” OR “Glycyrrhiza glabra” OR “Liquorice” OR “DGL” OR “deglycerized licorice” OR “deglycyrrhizinated licorice” OR “deglycyrrhizinated liquorice” OR “Liquiritiae radix”) | Embase, Medline, Derwent drug file, PubMed and ToXfile |
| 2 | Indications ti,ab(“gastric gas” OR belching OR Eructation OR Dyspepsia OR indigestion OR “gastroesophageal reflux” OR pyrosis OR Heartburn OR satiety OR overfeed OR Overfill OR “difficulty in swallowing” OR “abdominal bloating” OR “appetite loss” OR distention OR Nausea OR vomit* OR burp* OR dysphagia OR “stomach spasm” OR “stomach pain” OR “Stomach fullness” OR flatulence OR “stomach gas” OR “upper gastrointestinal” OR bloating OR “postprandial fullness” OR “loss of appetite” OR “food regurgitation” OR acidity OR retching OR gagging OR “motion sickness” OR GERD) | Embase, Medline, Derwent drug file, PubMed and ToXfile |
| Nutritional Ingredient | Reference | Population (n) | Characteristics | Study Design | Duration | Intervention | Control/Comparator | Outcome Measured | Results |
|---|---|---|---|---|---|---|---|---|---|
| Fenugreek | DiSilvestro et al., 2011 [14] | n = 45 | Age 43 ± 8; 24 females and 21 males; subjects experiencing heartburn after 3–8 meals per week for at least a month | RCT; placebo CG and IG were blinded, ranitidine positive CG group was unblinded; rescue medication tablets of chewable calcium carbonate were allowed | 2 weeks | IG: 2000 mg 2x/day fenugreek capsules (4 g daily dose) | Placebo CG: starch capsules, four capsules taken twice/day; Positive CG: Ranitidine (Zantac 75), 75 mg, 2x/day, (150 mg daily dose) | Heartburn | The severity and incidence of heartburn significantly decreased with both the IG and the positive CG for both the first and second intervention week. The placebo also yielded significant effects for the second but not the first intervention week. All three interventions yielded statistically significant reductions in rescue medication use. |
| Galactomannan | Abenevoli et al., 2021 [15] | n = 60 | Age ≥18; 47 males and 13 females; adults with GERD symptoms not taking PPIs | Single-center, RCT | 2 weeks | IG: 10 mL, 3x/day liquid blend (calcium carbonate, sodium bicarbonate, Malva sylvestris, hyaluronic acid) (30 mL total) | CG: Placebo, one sachet containing 10 mL liquid, three times per day (30 mL total) | GERD | 100% of patients reported at least 30% reduction in symptoms from baseline to week 3 of the trial (p < 0.001) as compared to placebo group. Symptom frequency and intensity were progressively significantly reduced from baseline to visit 4 (p < 0.001) in the IG as compared to placebo. Heartburn significantly decreased from baseline to day 14 for the IG (p < 0.001) versus an increase in score in CG (p > 0.05). There was a significant decrease in GERD symptoms from visit 2 to 3 to 4 (p < 0.001, p < 0.001, and p = 0.001 respectively) as compared to the CG. |
| Nutritional Ingredient | Reference | Population (n) | Characteristics | Study Design | Duration | Intervention | Control/Comparator | Outcome Measured | Results |
|---|---|---|---|---|---|---|---|---|---|
| Aloe Vera | Panahi et al., 2015 [28] | n = 79 | Age: 18–65; 45 females and 34 males; GERD patients | RCT | 4 weeks | IG: 10 mL 1x/day A. vera syrup (standardized to 5.0 mg polysaccharide per mL of syrup) (10 mL total) | CG1: omeprazole capsule (20 g once a day) CG2: ranitidine tablet (150 mg in a fasted state in the morning and 150 mg 30 min before sleep at night) (300 mg total) | GERD | A. vera was effective in significantly reducing the frequencies of all GERD symptoms except vomiting at weeks 2 and 4 for within-group comparison to baseline (p < 0.05). Omeprazole and ranitidine were both more effective at reducing heartburn and flatulence than A. vera. The frequency of other symptoms did not differ between the groups. |
| Aloe Vera | Panahi et al., 2016 [27] | n = 85 | Age >40 years; male GERD veterans with sulphur mustard gas exposure | RCT | 6 weeks | IG: pantoprazole (40 mg before breakfast) plus 5 mL, 2x/day A. vera syrup (10 mL total) | CG: pantoprazole (40 mg before breakfast) | GERD | Both IG and CG resulted in a progressive decrease in RSI score from baseline to weeks 3 and 6 ((p < 0.001) with the IG having a significantly greater reduction (p < 0.001) compared to CG. No adverse events were reported. |
| Ginger | Panda et al., 2020 [30] | n = 48 | Age: 18–55; subjects with FD per Rome III criteria | RCT, parallel group | 4 weeks | IG: 200 mg, 2x/day high concentration gingerol powder extract (400 mg total) | CG: placebo, 200 mg twice daily | FD symptoms | The IG had significantly more subjects who were “extremely” or “markedly” improved as compared to CG. (79% vs. 21%; p < 0.05). Elimination rate of symptoms both individually and collectively was greater in the IG than CG (64% vs. 13% of subjects) (p < 0.05). |
| Ginger | Attari et al., 2019 [29] | n = 15 | Age: 18–65; 5 males and 10 females; patients with H. pylori positive FD | Pilot study | 4 weeks | IG: 3 g, 1x/day ginger powder tablets (3 g total) | CG: none | H. pylori positive FD and FD | Ginger supplementation resulted in significant improvement of all dyspepsia symptoms including fullness, early satiety, nausea, belching, gastric pain, and gastric burn, but not vomiting (p = 0.180). |
| Ginger | Bhargava et al., 2020 [31] | n = 15 | Age: 35–79; 8 males and 7 females; patients with anorexia-cachexia syndrome (ACS) in addition to a variety of advanced cancer diagnoses | Single-arm intervention trial | 2 weeks | IG: 1650 mg 1×/day of ginger powder capsule (1650 mg total) | CG: none | ACS GI symptoms such as nausea, vomiting, dysmotility-, ulcer-, and reflux-like symptoms | Over half of the patients reported significant improvements in GI symptoms including nausea (p < 0.02), dysmotility-like (p < 0.01), reflux-like (p < 0.01), and ulcer-like symptoms (p = 0.05). |
| Licorice | Prajapati and Patel, 2015 [32] | n = 40 | Age: 21–60; Amlapitta (acid gastritis) patients including symptoms of indigestion, exhaustion, eructation with bitter or sour taste, burning sensation in the chest and throat, and anorexia | RCT | 2 weeks | IG1: 2 g, 3×/day of Licorice root powder (6 total) IG2: 2 g, 3×/day of Jethimala (Taverniera nummularia Baker) (commercial licorice substitute) | CG: none | Gastritis; heartburn; anorexia; reflux | Licorice root and Jethimala were both effective in significantly reducing all symptoms in both groups, with licorice treatment showing overall better effects. There was no significant difference between the two treatments in treatment efficacy (p > 0.05) other than in the symptom of anorexia (p-0.001). |
| Licorice | Raveendra et al., 2012 [33] | n = 50 | Age: 18–65; 31 males and 19 females; patients with FD as diagnosed by Rome III criteria | RCT | 30 days | IG: 75 mg, 2×/day of flavonoid-rich extract of licorice (150 mg total) | CG: placebo | Functional dyspepsia | As compared to CG, IG showed a significant decrease in total symptom scores (p ≤ 0.05), and a significant improvement in quality of life (p ≤ 0.05) as well as overall treatment efficacy. |
| Papaya | Muss et al., 2013 [34] | n = 84 | Age: 18–75; subjects with dysfunctions of the GI tract such as constipation, heartburn, and irritable bowel syndrome (IBS) | RCT; participants were labeled as “early” (2 days) or “late” (3–16 days) returnees based on when they returned to the trial center to complete the endpoint questionnaire | 40 days | IG: 20 mL, 1×/day papaya formulation (standardized to higher papain activity) | CG: 20 mL, 1×/day of placebo | Heartburn, constipation, and bloating | In the “early returnees,” the IG showed significant improvements in symptoms of constipation (p < 0.031) and flatulence (p = 0.017) as compared to placebo. Regarding heartburn, 85% of evaluable participants (n = 13) reported improvement (p = 0.114). These effects vanished in the “late returnees,” but those in the IG showed more beneficial effects than the CG. |
| Papaya | Weiser et al., 2018 [35] | n = 60 | Age: 18–75; 22 males and 38 females; patients with endoscopically confirmed chronic gastritis | RCT | 30 days | IG: 20 g, 2×/day of papaya blend before meal (papaya pulp, organic whole meal oat flour, apple juice concentrate, natural aroma, and water) | CG: 20 g, 2×/day placebo before a main meal | Chronic Gastritis | There was a reduction in all symptoms in both the IG and CG with greater reduction in scores for the IG for acute stomach ache pain, pain severity, impact on daily routine, nausea, bloating, and pain in the upper abdomen, but no significant difference between the groups. The only symptom which was significantly reduced in the IG compared to CG was pain load (p = 0.048). |
| Nutritional Ingredient | Reference | Population (n) | Characteristics | Study Design | Duration | Intervention | Control/Comparator | Outcome Measured | Results |
|---|---|---|---|---|---|---|---|---|---|
| Curcumin, aloe vera, slippery elm, guar gum, pectin, peppermint oil, and glutamine | Ried et al., 2020 [73] | n = 43 | Mean age: 50; 76% of participants were female; adults with moderate upper and/or lower GI disturbances | Single-arm pre-post study | 16 weeks (4-week run-in period and 12-week intervention period) | IG: 5 g, 1×/day formula mixed with water for 4 weeks, followed by 10 g/d for the second month and finally the patient’s preferred dose (0/5/10 g/d) for the third month (Curcumin, Aloe vera, slippery elm, guar gum, pectin, peppermint oil, and glutamine) | CG: 4-week run-in period | Upper and lower GI symptoms | There was a significant improvement of upper GI symptoms including indigestion, heartburn, regurgitation (acid reflux), and nausea (p < 0.001), overall decreased upper GI pain (p < 0.001) as well as improved quality of life (QoL) (p < 0.001) after 12 weeks. |
| ACV, Licorice, papain | Brown, et al., 2015 [44] | n = 24 | Age ≥18; mean age: 34 ± 14; 17 females and 7 males; GERD patients | Double-blind, placebo controlled, crossover trial with 1-week washout between treatments; reflux causing meal: big hamburger, French fries, hot sauce, soda | Single day intervention | IG: 30 min chewing intervention gum following reflux causing meal (gum active ingredients: calcium carbonate (500 mg), licorice extract, papain, and apple cider vinegar) | CG: 30 min chewing placebo gum following reflux causing meal | GERD and heartburn | Adjusted mean heartburn score and mean acid reflux score were significantly decreased in IG as compared to CG (p = 0.034 and p = 0.013 respectively). There were no significant differences between groups for pain, nausea, and belching, although they trended towards greater improvement in the IG group. |
| Nutritional Ingredient | Reference | Population (n) | Characteristics | Study Design | Duration | Intervention | Control/Comparator | Outcome Measured | Results |
|---|---|---|---|---|---|---|---|---|---|
| Activated Charcoal (AC) | Coffin et al., 2011 [80] | n = 276 | Age: 18–49; mean age 39 ± 10 years; 70% female; functional dyspeptic patients per ROME III criteria | RCT, phase III trial | 30 days | IG: 2 capsules 3x per day of AC formula (gastro-soluble capsule containing 140mg of AC, 45mg of simethicone INN, and 180mg of magnesium oxide, plus enteric coated capsule containing 140mg of AC and 45mg of simethicone INN) | CG: 2 placebo capsules, 3x per day | Functional dyspepsia | IG saw significantly greater absolute and relative symptom reductions compared to placebo. The IG observed a significant reduction in post-prandial fullness (p = 0.034), epigastric pain (p = 0.045), epigastric burning (p = 0.03), and bloating (p = 0.03), in comparison to placebo, and early satiety approached significance (p = 0.051). |
| Activated Charcoal | Lecuyer et al., 2009 [81] | n = 132 | Age: 18–49; mean age: 39.0±8.8 years; functional dyspepsia patients | RCT | 3 months and 2-month follow-up period | IG: 2 capsules, 3x per day AC formula during meals (gastro-soluble and enteric capsules containing 140 mg AC and 45 mg simethicone) | CG: 2 capsules placebo, 3x per day during meals | Functional dyspepsia | Greater percentage of patients with a reduction of at least two points on the symptoms intensity scale in IG compared to CG (p = 0.043) although there was a nonsignificant difference between the two groups regarding overall patient complaints (p = 0.115). IG also saw greater reduction in intensity of abdominal fullness, bloating, and the sensation of slow digestion versus placebo (p < 0.05). |
| Zinc-l-Carnosine (polaprezinc) (ZnC) | Tan et al., 2017 [82] | n = 303 | Age: 18–70; 168 females and 164 males; patients with H. pylori-associated gastritis | RCT | 2 weeks | IG: Arm A: triple therapy (omeprazole 20 mg, amoxicillin 1 g, and clarithromycin 500 mg, each twice daily) plus polaprezinc 75 mg, twice daily (150 mg total); Arm B: triple therapy plus polaprezinc 150 mg, twice daily (300 mg total) | CG: Arm C triple therapy alone | H. pylori associated gastritis | All three arms saw significant gastrointestinal symptom improvement, including abdominal pain, acid reflux, belching, heartburn, bloating, nausea, and vomiting at days 7, 14, and 28 when compared to baseline (p < 0.0001) with no significant difference between groups. Both the intention to treat (ITT) and per-protocol (PP) analyses showed that Arms A and B had a significantly higher rate of H. pylori eradication than Arm C while there was no significant difference between the rate of eradication in Arms A and B. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schulz, R.M.; Ahuja, N.K.; Slavin, J.L. Effectiveness of Nutritional Ingredients on Upper Gastrointestinal Conditions and Symptoms: A Narrative Review. Nutrients 2022, 14, 672. https://doi.org/10.3390/nu14030672
Schulz RM, Ahuja NK, Slavin JL. Effectiveness of Nutritional Ingredients on Upper Gastrointestinal Conditions and Symptoms: A Narrative Review. Nutrients. 2022; 14(3):672. https://doi.org/10.3390/nu14030672
Chicago/Turabian StyleSchulz, Rebekah M., Nitin K. Ahuja, and Joanne L. Slavin. 2022. "Effectiveness of Nutritional Ingredients on Upper Gastrointestinal Conditions and Symptoms: A Narrative Review" Nutrients 14, no. 3: 672. https://doi.org/10.3390/nu14030672
APA StyleSchulz, R. M., Ahuja, N. K., & Slavin, J. L. (2022). Effectiveness of Nutritional Ingredients on Upper Gastrointestinal Conditions and Symptoms: A Narrative Review. Nutrients, 14(3), 672. https://doi.org/10.3390/nu14030672






