Potential Benefits of Selenium Supplementation in Reducing Insulin Resistance in Patients with Cardiometabolic Diseases: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Search Strategy
2.2. Study Selection
2.3. Data Collection Process
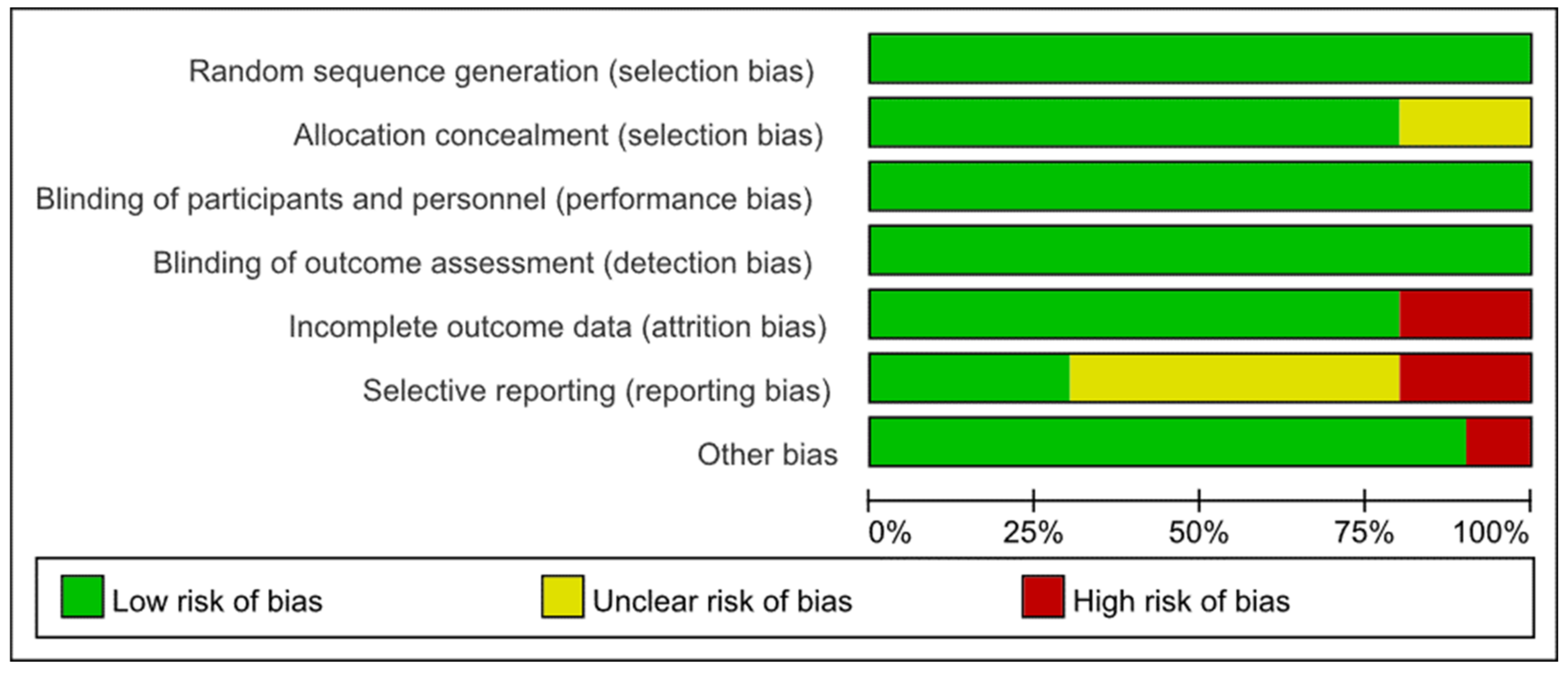
2.4. Risk of Bias Assessment
2.5. Data Synthesis and Statistical Analysis
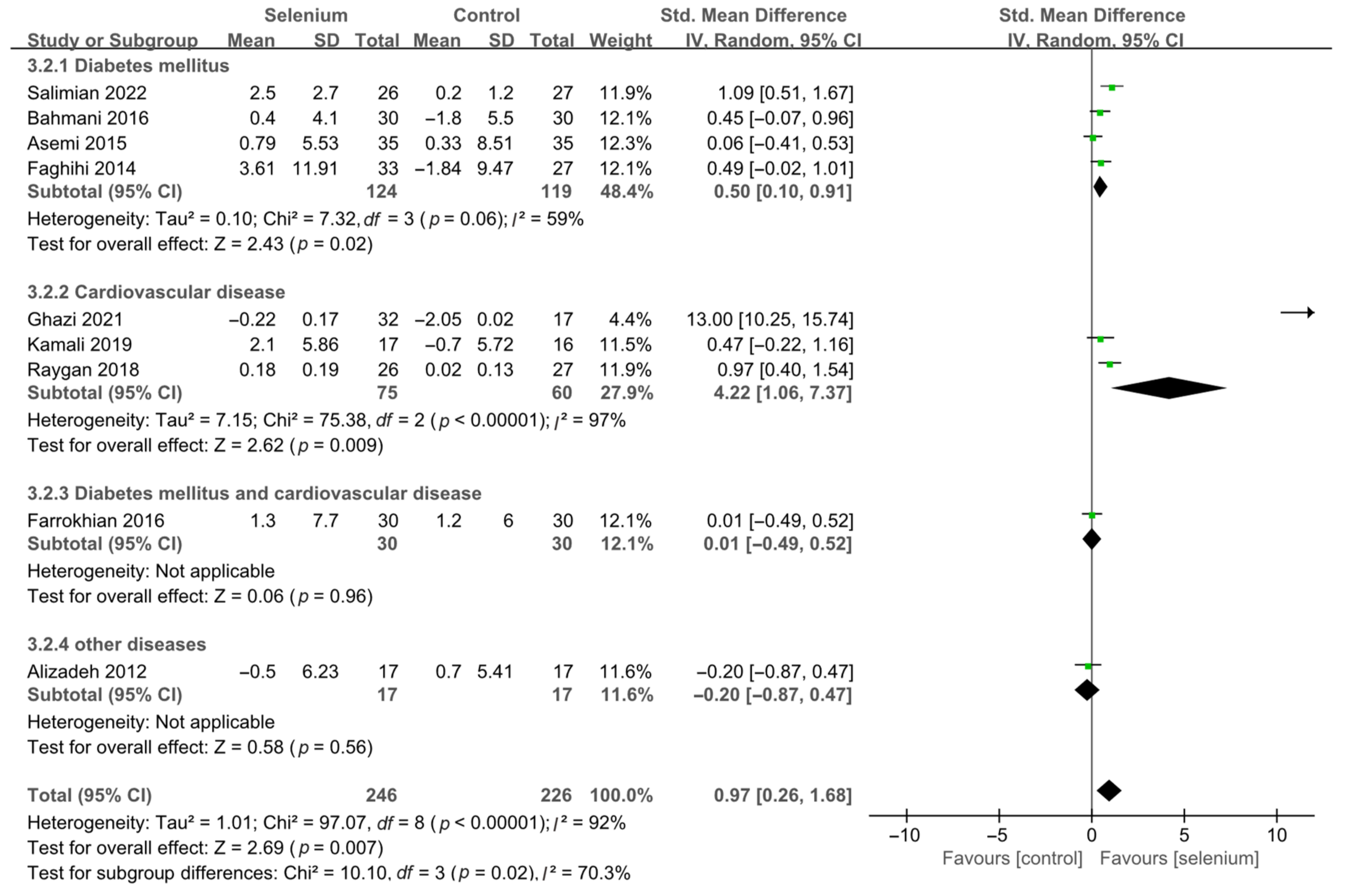
2.6. Analysis of Subgroups or Subsets
3. Results
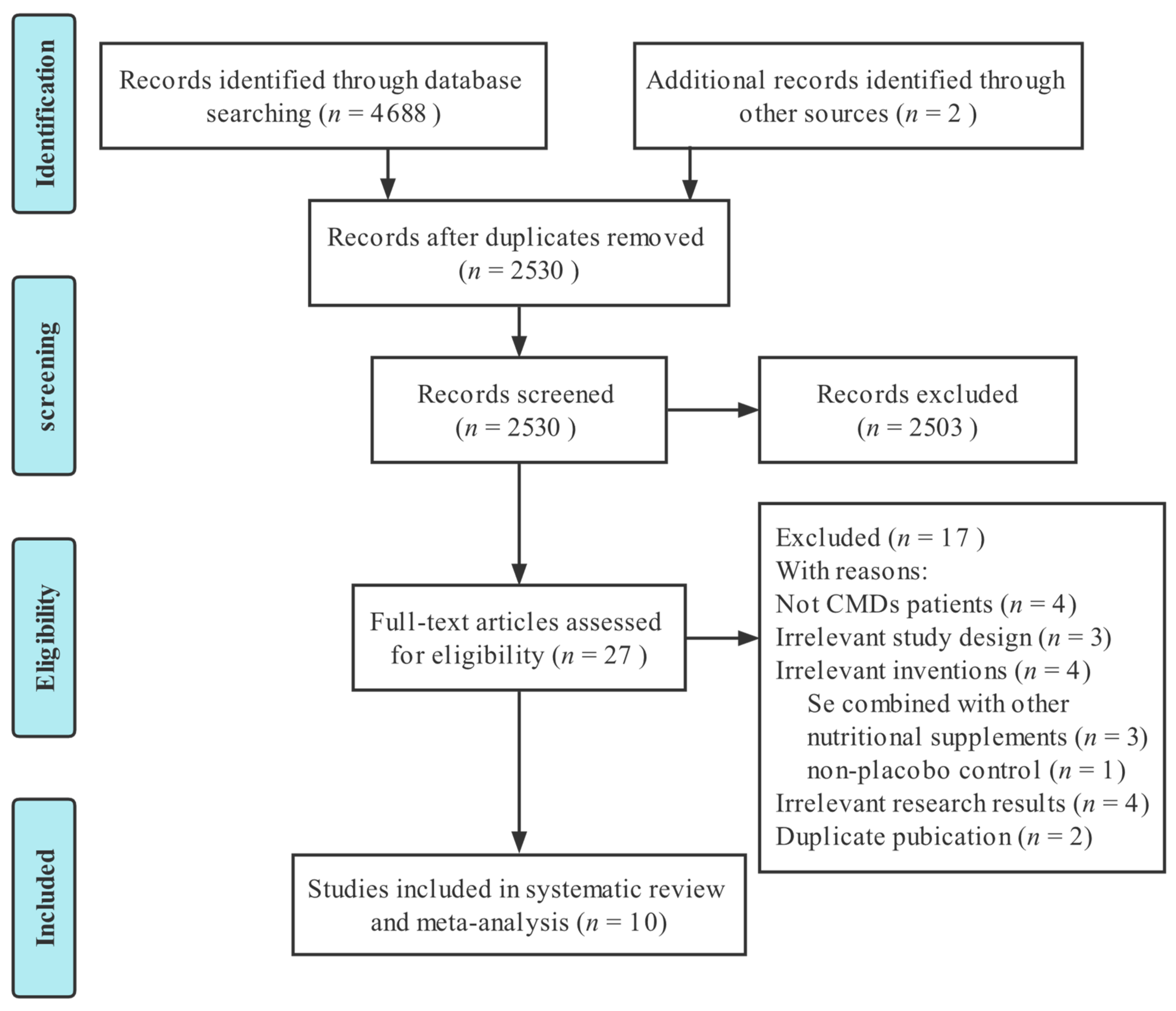
3.1. Literature Selection
3.2. Study Characteristics
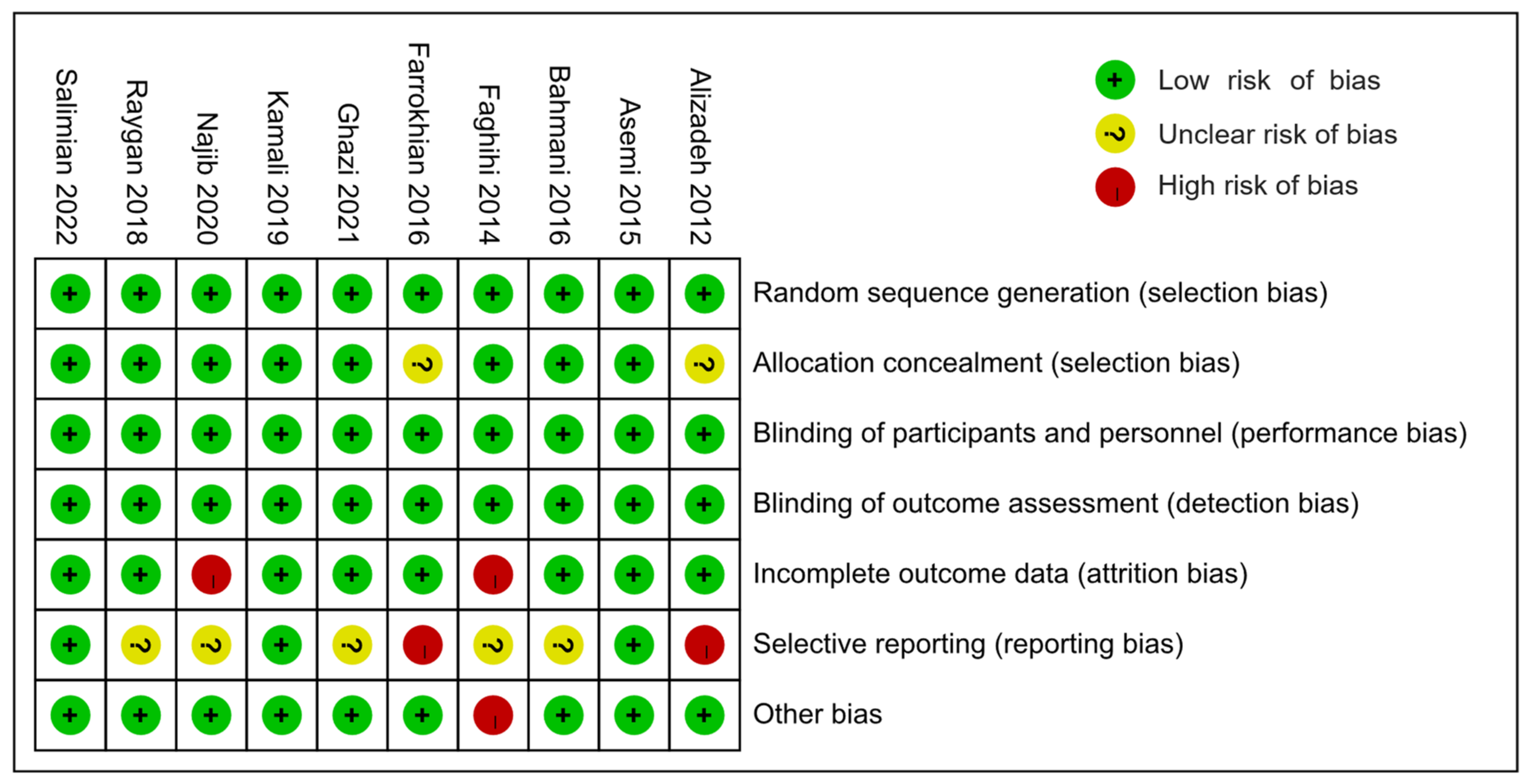
3.3. Risk of Bias Assessments
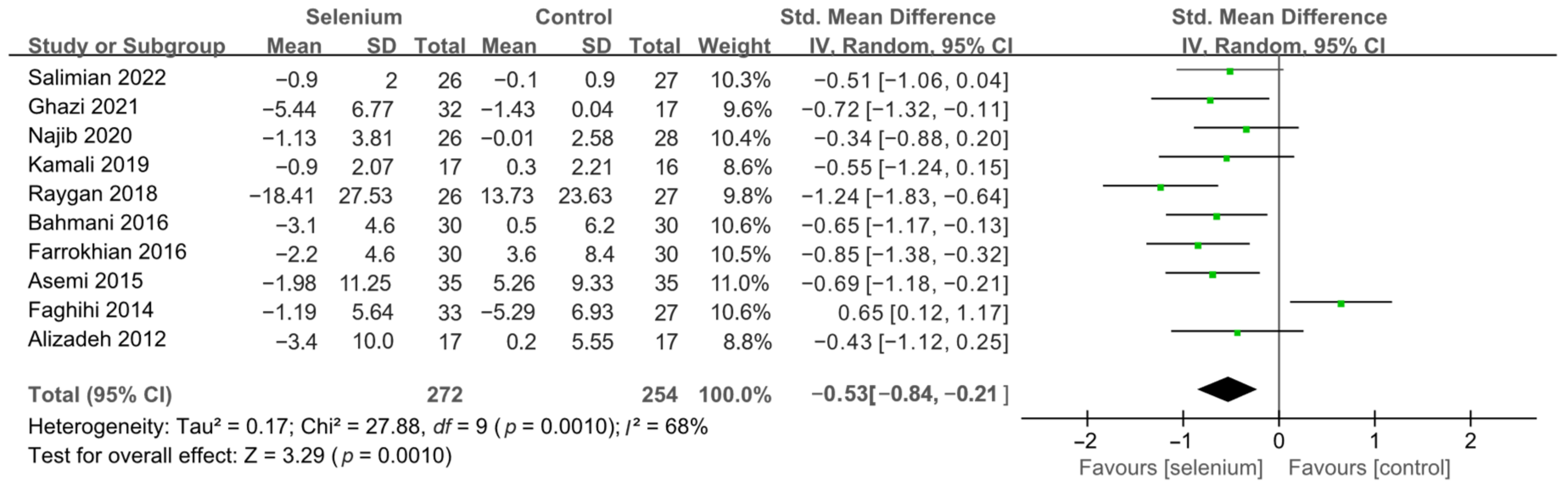
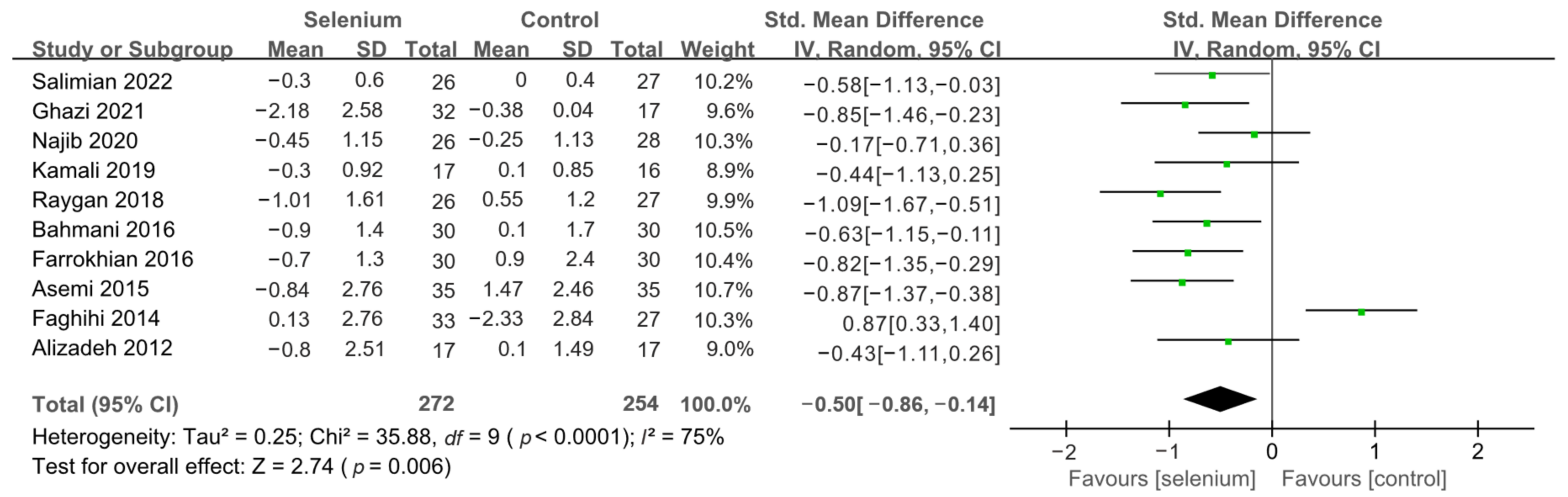
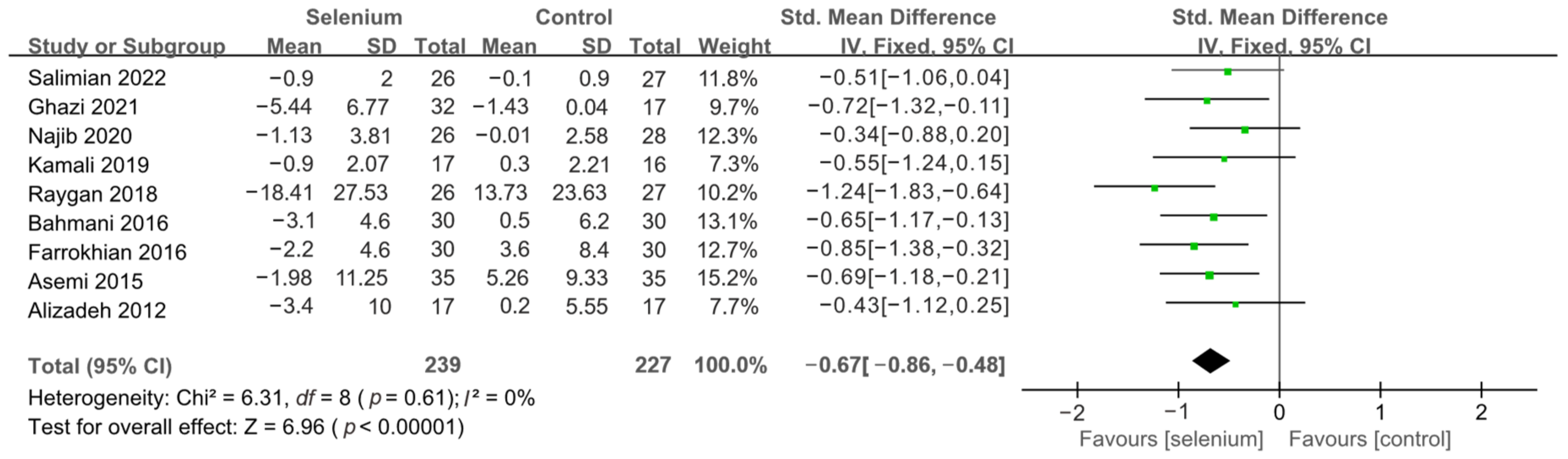
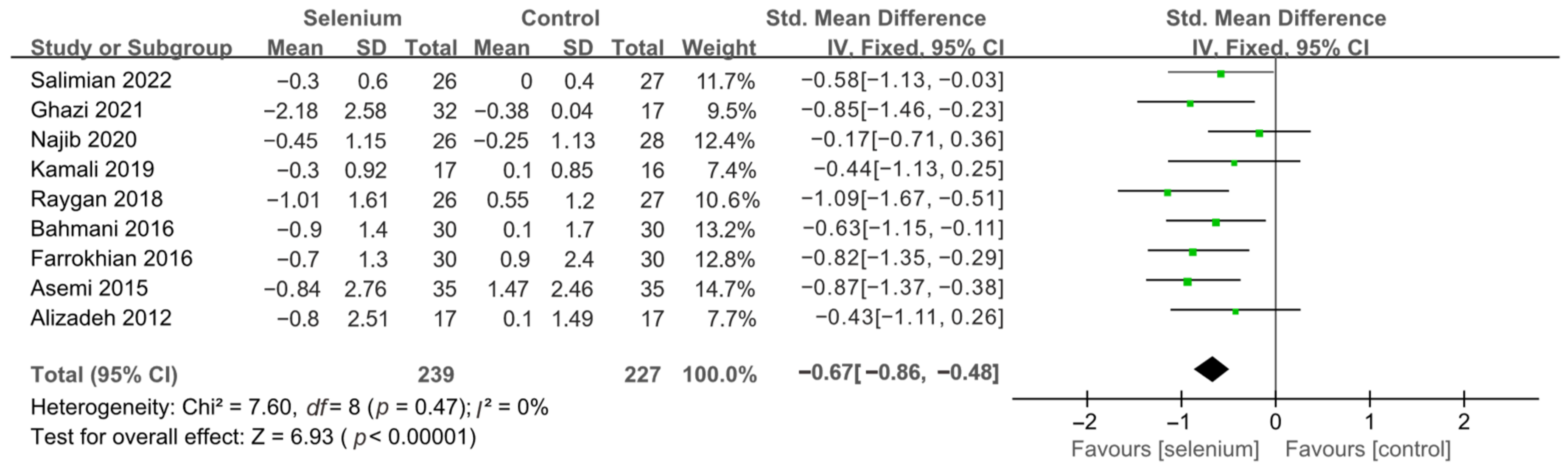
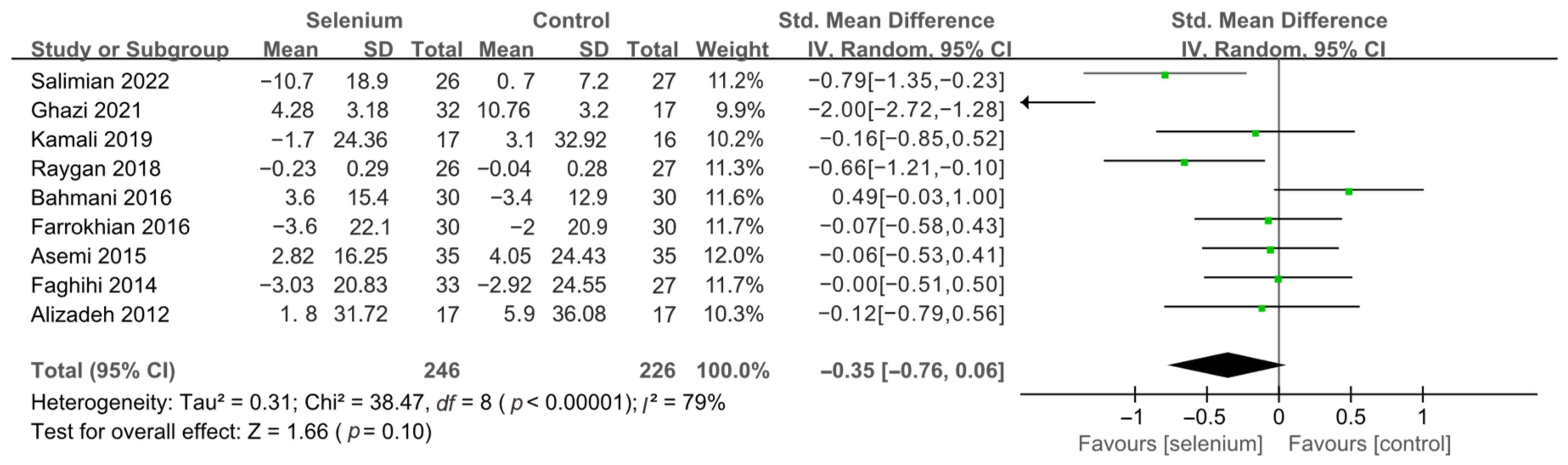
3.4. Meta-Analysis
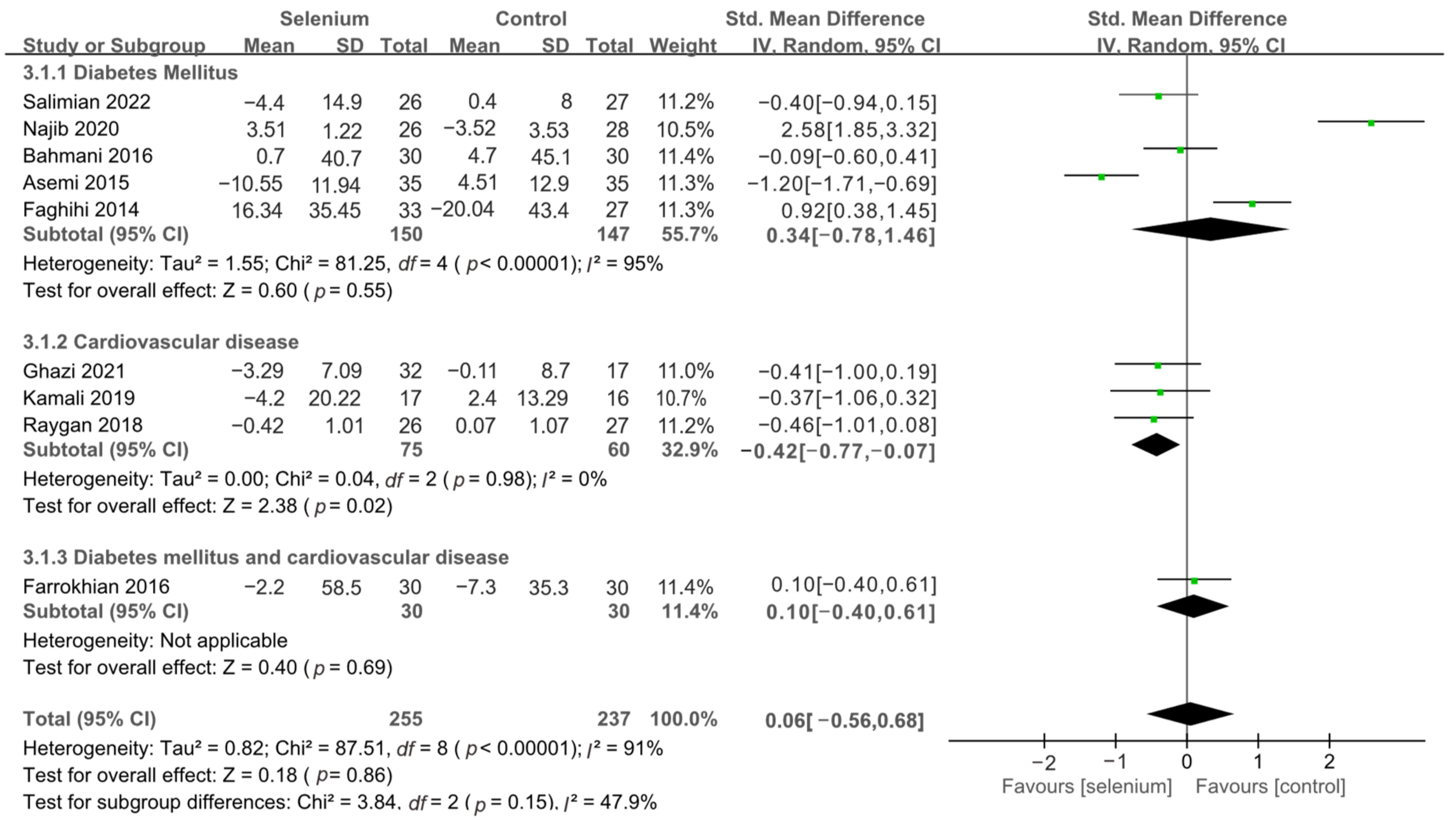
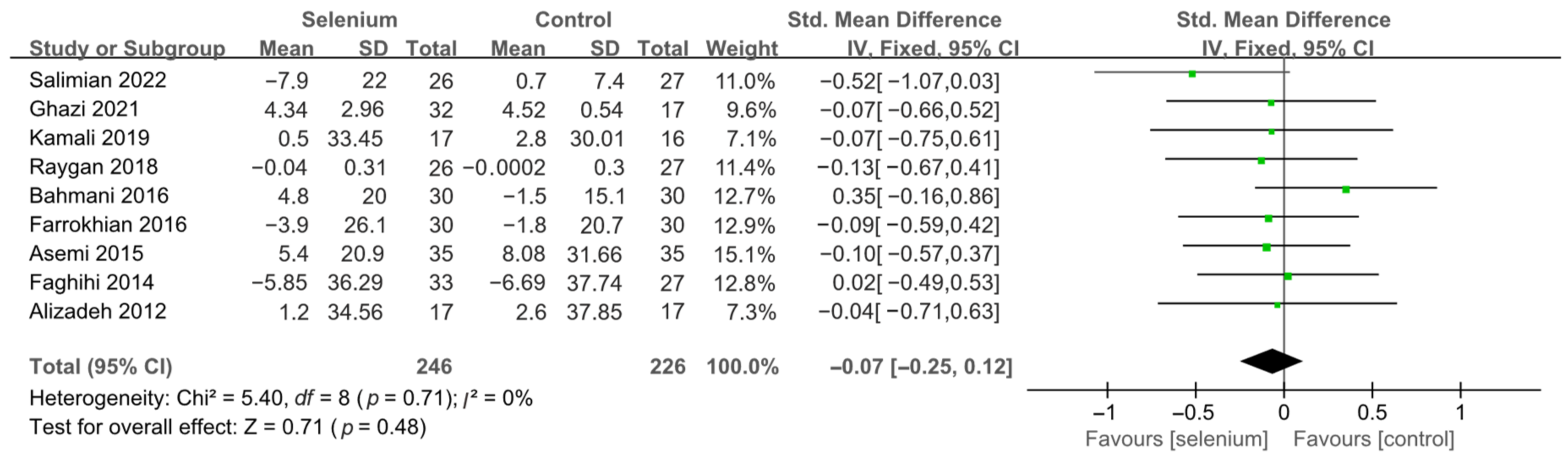
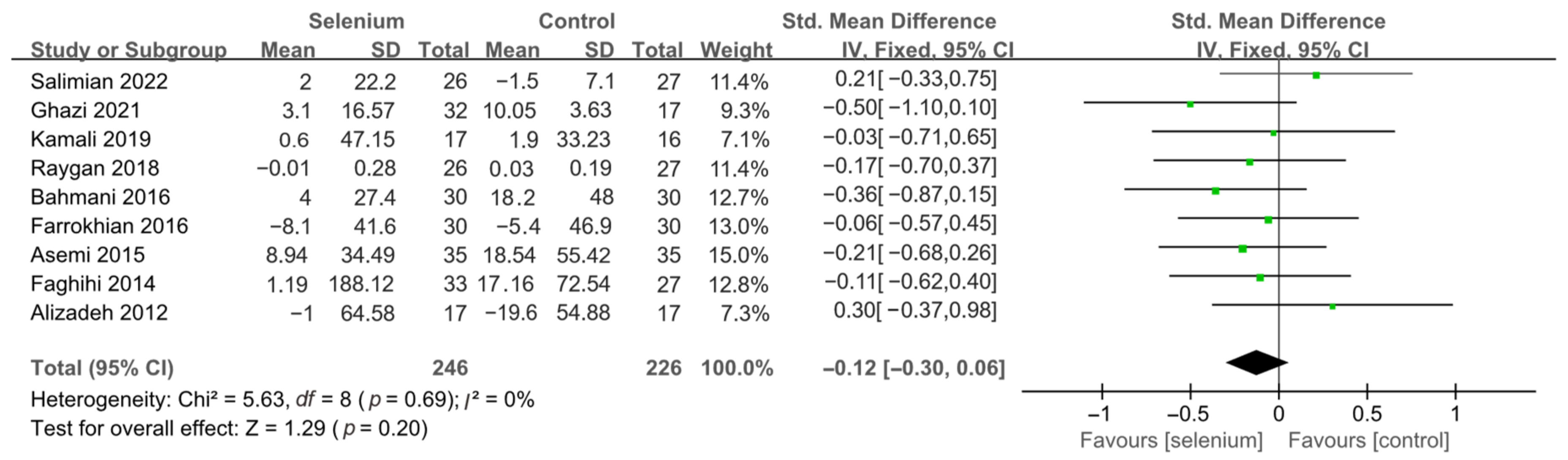
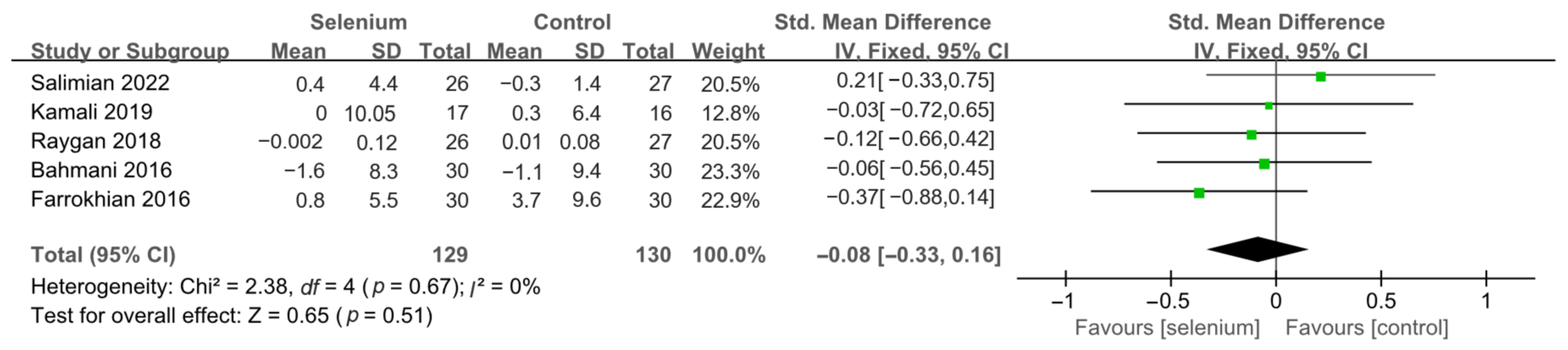
3.4.1. Primary Outcomes
3.4.2. Secondary Outcomes
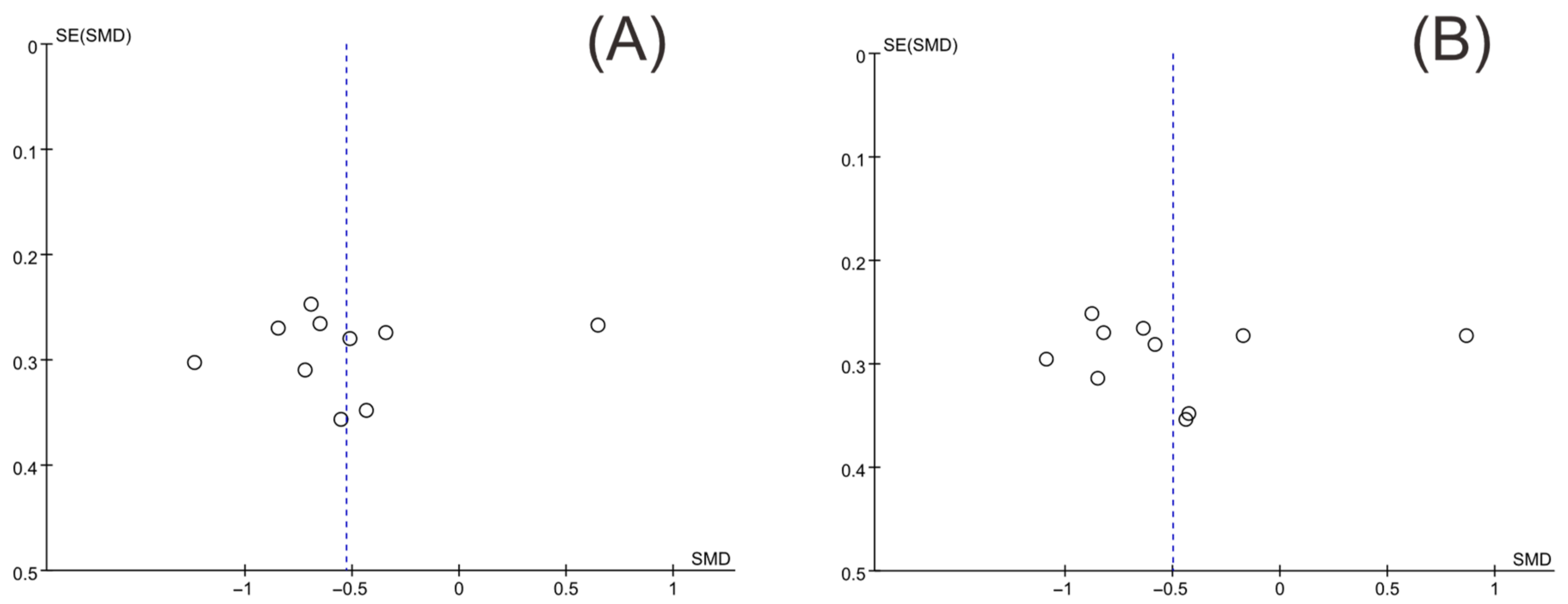
3.5. Publication Bias
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Guo, F.; Moellering, D.R.; Garvey, W.T. The progression of cardiometabolic disease: Validation of a new cardiometabolic disease staging system applicable to obesity. Obesity 2014, 22, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Hodkinson, A.; Kontopantelis, E.; Adeniji, C.; van Marwijk, H.; McMillian, B.; Bower, P.; Panagioti, M. Interventions Using Wearable Physical Activity Trackers Among Adults With Cardiometabolic Conditions: A Systematic Review and Meta-analysis. JAMA Netw. Open 2021, 4, e2116382. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Y.Y.; Wang, L.S.; Jiang, H.R.; Jia, X.F.; Wang, Z.H.; Wang, H.J.; Zhang, B.; Ding, G.G. Epidemiological characteristics and trends of cardiometabolic risk factors in residents aged 18–64 years in 15 provinces of China. Zhonghua Liu Xing Bing Xue Za Zhi 2022, 43, 1254–1261. [Google Scholar] [CrossRef] [PubMed]
- Madlala, S.S.; Hill, J.; Kunneke, E.; Kengne, A.P.; Peer, N.; Faber, M. Dietary Diversity and its Association with Nutritional Status, Cardiometabolic Risk Factors and Food Choices of Adults at Risk for Type 2 Diabetes Mellitus in Cape Town, South Africa. Nutrients 2022, 14, 3191. [Google Scholar] [CrossRef] [PubMed]
- Ghorbani, Z.; Kazemi, A.; Bartolomaeus, T.U.P.; Martami, F.; Noormohammadi, M.; Salari, A.; Löber, U.; Balou, H.A.; Forslund, S.K.; Mahdavi-Roshan, M. The effect of probiotic and synbiotic supplementation on lipid parameters among patients with cardiometabolic risk factors: A systematic review and meta-analysis of clinical trials. Cardiovasc. Res. 2022; in press. [Google Scholar] [CrossRef]
- Jamilian, M.; Modarres, S.Z.; Siavashani, M.A.; Karimi, M.; Mafi, A.; Ostadmohammadi, V.; Asemi, Z. The Influences of Chromium Supplementation on Glycemic Control, Markers of Cardio-Metabolic Risk, and Oxidative Stress in Infertile Polycystic ovary Syndrome Women Candidate for In vitro Fertilization: A Randomized, Double-Blind, Placebo-Controlled Trial. Biol. Trace Elem. Res. 2018, 185, 48–55. [Google Scholar] [CrossRef]
- Gil, Á.; Ortega, R.M. Introduction and Executive Summary of the Supplement, Role of Milk and Dairy Products in Health and Prevention of Noncommunicable Chronic Diseases: A Series of Systematic Reviews. Adv. Nutr. 2019, 10, S67–S73. [Google Scholar] [CrossRef]
- Kim, Y.; Oh, Y.K.; Lee, J.; Kim, E. Could nutrient supplements provide additional glycemic control in diabetes management? A systematic review and meta-analysis of randomized controlled trials of as an add-on nutritional supplementation therapy. Arch. Pharmacal Res. 2022, 45, 185–204. [Google Scholar] [CrossRef]
- Bitterli, C.; Bañuelos, G.S.; Schulin, R. Use of transfer factors to characterize uptake of selenium by plants. J. Geochem. Explor. 2010, 107, 206–216. [Google Scholar] [CrossRef]
- Fordyce, F. Selenium Geochemistry and Health. Ambio 2007, 36, 94–97. [Google Scholar] [CrossRef]
- Hadrup, N.; Ravn-Haren, G. Absorption, distribution, metabolism and excretion (ADME) of oral selenium from organic and inorganic sources: A review. J. Trace Elem. Med. Biol. 2021, 67, 126801. [Google Scholar] [CrossRef]
- Labunskyy, V.M.; Hatfield, D.L.; Gladyshev, V.N. Selenoproteins: Molecular Pathways and Physiological Roles. Physiol. Rev. 2014, 94, 739–777. [Google Scholar] [CrossRef]
- Fairweather-Tait, S.J.; Bao, Y.; Broadley, M.R.; Collings, R.; Ford, D.; Hesketh, J.E.; Hurst, R. Selenium in Human Health and Disease. Antioxid. Redox Signal. 2011, 14, 1337–1383. [Google Scholar] [CrossRef]
- Handy, D.E.; Loscalzo, J. The role of glutathione peroxidase-1 in health and disease. Free Radic. Biol. Med. 2022, 188, 146–161. [Google Scholar] [CrossRef]
- Hariharan, S.; Dharmaraj, S. Selenium and selenoproteins: It’s role in regulation of inflammation. Inflammopharmacology 2020, 28, 667–695. [Google Scholar] [CrossRef]
- Wang, J.; Yang, J.; Wang, C.; Zhao, Z.; Fan, Y. Systematic Review and Meta-Analysis of Oxidative Stress and Antioxidant Markers in Oral Lichen Planus. Oxidative Med. Cell. Longev. 2021, 2021, 9914652. [Google Scholar] [CrossRef]
- Hasani, M.; Djalalinia, S.; Khazdooz, M.; Asayesh, H.; Zarei, M.; Gorabi, A.M.; Ansari, H.; Qorbani, M.; Heshmat, R. Effect of selenium supplementation on antioxidant markers: A systematic review and meta-analysis of randomized controlled trials. Hormones 2019, 18, 451–462. [Google Scholar] [CrossRef]
- Hurrle, S.; Hsu, W.H. The etiology of oxidative stress in insulin resistance. Biomed. J. 2017, 40, 257–262. [Google Scholar] [CrossRef]
- Singh, A.; Kukreti, R.; Saso, L.; Kukreti, S. Mechanistic Insight into Oxidative Stress-Triggered Signaling Pathways and Type 2 Diabetes. Molecules 2022, 27, 950. [Google Scholar] [CrossRef]
- Kattoor, A.J.; Pothineni, N.V.K.; Palagiri, D.; Mehta, J.L. Oxidative Stress in Atherosclerosis. Curr. Atheroscler. Rep. 2017, 19, 42. [Google Scholar] [CrossRef]
- Gunawardena, H.P.; Silva, R.; Sivakanesan, R.; Ranasinghe, P.; Katulanda, P. Poor Glycaemic Control Is Associated with Increased Lipid Peroxidation and Glutathione Peroxidase Activity in Type 2 Diabetes Patients. Curr. Med. Res. Opin. 2019, 2019, 9471697. [Google Scholar] [CrossRef] [PubMed]
- Barchielli, G.; Capperucci, A.; Tanini, D. The Role of Selenium in Pathologies: An Updated Review. Antioxidants 2022, 11, 251. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Zhao, G.; Liu, S.; Zhang, Q.; Wang, P.; Cao, Y.; Wu, L. Supplementation with selenium attenuates autism-like behaviors and improves oxidative stress, inflammation and related gene expression in an autism disease model. J. Nutr. Biochem. 2022, 107, 109034. [Google Scholar] [CrossRef]
- Brodin, O.; Eksborg, S.; Wallenberg, M.; Asker-Hagelberg, C.; Larsen, E.H.; Mohlkert, D.; Lenneby-Helleday, C.; Jacobsson, H.; Linder, S.; Misra, S.; et al. Pharmacokinetics and Toxicity of Sodium Selenite in the Treatment of Patients with Carcinoma in a Phase I Clinical Trial: The SECAR Study. Nutrients 2015, 7, 4978–4994. [Google Scholar] [CrossRef] [PubMed]
- Nandi, A.; Yan, L.-J.; Jana, C.K.; Das, N. Role of Catalase in Oxidative Stress- and Age-Associated Degenerative Diseases. Oxidative Med. Cell. Longev. 2019, 2019, 9613090. [Google Scholar] [CrossRef]
- Li, Z.-Y.; Xu, G.-B.; Xia, T.-A. Prevalence rate of metabolic syndrome and dyslipidemia in a large professional population in Beijing. Atherosclerosis 2006, 184, 188–192. [Google Scholar] [CrossRef]
- Kohler, L.N.; Florea, A.; Kelley, C.P.; Chow, S.; Hsu, P.; Batai, K.; Saboda, K.; Lance, P.; Jacobs, E.T. Higher Plasma Selenium Concentrations Are Associated with Increased Odds of Prevalent Type 2 Diabetes. J. Nutr. 2018, 148, 1333–1340. [Google Scholar] [CrossRef]
- Yin, T.; Zhu, X.; Xu, D.; Lin, H.; Lu, X.; Tang, Y.; Shi, M.; Yao, W.; Zhou, Y.; Zhang, H.; et al. The Association Between Dietary Antioxidant Micronutrients and Cardiovascular Disease in Adults in the United States: A Cross-Sectional Study. Front. Nutr. 2021, 8, 799095. [Google Scholar] [CrossRef]
- Huang, J.; Xie, L.; Song, A.; Zhang, C. Selenium Status and Its Antioxidant Role in Metabolic Diseases. Oxidative Med. Cell. Longev. 2022, 2022, 7009863. [Google Scholar] [CrossRef]
- Kamali, A.; Amirani, E.; Asemi, Z. Effects of Selenium Supplementation on Metabolic Status in Patients Undergoing for Coronary Artery Bypass Grafting (CABG) Surgery: A Randomized, Double-Blind, Placebo-Controlled Trial. Biol. Trace Elem. Res. 2019, 191, 331–337. [Google Scholar] [CrossRef]
- Cardoso, B.R.; Braat, S.; Graham, R.M. Selenium Status Is Associated With Insulin Resistance Markers in Adults: Findings From the 2013 to 2018 National Health and Nutrition Examination Survey (NHANES). Front. Nutr. 2021, 8, 696024. [Google Scholar] [CrossRef]
- Ju, W.; Ji, M.; Li, X.; Li, Z.; Wu, G.; Fu, X.; Yang, X.; Gao, X. Relationship between higher serum selenium level and adverse blood lipid profile. Clin. Nutr. 2018, 37, 1512–1517. [Google Scholar] [CrossRef]
- Ju, W.; Li, X.; Li, Z.; Wu, G.R.; Fu, X.F.; Yang, X.M.; Zhang, X.Q.; Gao, X.B. The effect of selenium supplementation on coronary heart disease: A systematic review and meta-analysis of randomized controlled trials. J. Trace Elem. Med. Biol. 2017, 44, 8–16. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Syst. Rev. 2021, 10, 89. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: Hoboken, NJ, USA, 2019. [Google Scholar]
- Yuan, D.; Sharma, H.; Krishnan, A.; Vangaveti, V.N.; Malabu, U.H. Effect of glucagon-like peptide 1 receptor agonists on albuminuria in adult patients with type 2 diabetes mellitus: A systematic review and meta-analysis. Diabetes Obes. Metab. 2022, 24, 1869–1881. [Google Scholar] [CrossRef]
- Li, T.; Yu, T.; Hawkins, B.S.; Dickersin, K. Design, Analysis, and Reporting of Crossover Trials for Inclusion in a Meta-Analysis. PLoS ONE 2015, 10, e0133023. [Google Scholar] [CrossRef]
- Cumpston, M.; Li, T.; Page, M.J.; Chandler, J.; Welch, V.A.; Higgins, J.P.; Thomas, J. Updated guidance for trusted systematic reviews: A new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst. Rev. 2019, 10, ED000142. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Akbari, M.; Moosazadeh, M.; Ghahramani, S.; Tabrizi, R.; Kolahdooz, F.; Asemi, Z.; Lankarani, K.B. High prevalence of hypertension among Iranian children and adolescents: A systematic review and meta-analysis. J. Hypertens. 2017, 35, 1155–1163. [Google Scholar] [CrossRef]
- Salimian, M.; Soleimani, A.; Bahmani, F.; Tabatabaei, S.M.H.; Asemi, Z.; Talari, H.R. The effects of selenium administration on carotid intima-media thickness and metabolic status in diabetic hemodialysis patients: A randomized, double-blind, placebo-controlled trial. Clin. Nutr. ESPEN 2022, 47, 58–62. [Google Scholar] [CrossRef]
- Ghazi, M.K.K.; Ghaffari, S.; Naemi, M.; Salehi, R.; Sadeghi, M.T.; Barati, M.; Shabestari, A.N.; Kafil, B.; Alamdari, N.M.; Soleimanzadeh, H.; et al. Effects of sodium selenite and selenium-enriched yeast on cardiometabolic indices of patients with atherosclerosis: A double-blind randomized clinical trial study. J. Cardiovasc. Thorac. Res. 2021, 13, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Najib, F.S.; Poordast, T.; Nia, M.R.; Dabbaghmanesh, M.H. Effects of selenium supplementation on glucose homeostasis in women with gestational diabetes mellitus: A randomized, controlled trial. Int. J. Reprod. Biomed. 2020, 18, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Raygan, F.; Behnejad, M.; Ostadmohammadi, V.; Bahmani, F.; Mansournia, M.A.; Karamali, F.; Asemi, Z. Selenium supplementation lowers insulin resistance and markers of cardio-metabolic risk in patients with congestive heart failure: A randomised, double-blind, placebo-controlled trial. Br. J. Nutr. 2018, 120, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Bahmani, F.; Kia, M.; Soleimani, A.; Asemi, Z.; Esmaillzadeh, A. Effect of Selenium Supplementation on Glycemic Control and Lipid Profiles in Patients with Diabetic Nephropathy. Biol. Trace Elem. Res. 2016, 172, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Farrokhian, A.; Bahmani, F.; Taghizadeh, M.; Mirhashemi, S.M.; Aarabi, M.; Raygan, F.; Aghadavod, E.; Asemi, Z. Selenium Supplementation Affects Insulin Resistance and Serum hs-CRP in Patients with Type 2 Diabetes and Coronary Heart Disease. Horm. Metab. Res. 2016, 48, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Asemi, Z.; Jamilian, M.; Mesdaghinia, E.; Esmaillzadeh, A. Effects of selenium supplementation on glucose homeostasis, inflammation, and oxidative stress in gestational diabetes: Randomized, double-blind, placebo-controlled trial. Nutrition 2015, 31, 1235–1242. [Google Scholar] [CrossRef]
- Faghihi, T.; Radfar, M.; Barmal, M.; Amini, P.; Qorbani, M.; Abdollahi, M.; Larijani, B. A Randomized, Placebo-Controlled Trial of Selenium Supplementation in Patients With Type 2 Diabetes: Effects on Glucose Homeostasis, Oxidative Stress, and Lipid Profile. Am. J. Ther. 2014, 21, 491–495. [Google Scholar] [CrossRef]
- Alizadeh, M.; Safaeiyan, A.; Ostadrahimi, A.; Estakhri, R.; Daneghian, S.; Ghaffari, A.; Gargari, B.P. Effect of L-Arginine and Selenium Added to a Hypocaloric Diet Enriched with Legumes on Cardiovascular Disease Risk Factors in Women with Central Obesity: A Randomized, Double-Blind, Placebo-Controlled Trial. Ann. Nutr. Metab. 2012, 60, 157–168. [Google Scholar] [CrossRef]
- Yang, L.; Qi, M.; Du, X.; Xia, Z.; Fu, G.; Chen, X.; Liu, Q.; Sun, N.; Shi, C.; Zhang, R. Selenium concentration is associated with occurrence and diagnosis of three cardiovascular diseases: A systematic review and meta-analysis. J. Trace Elem. Med. Biol. 2022, 70, 126908. [Google Scholar] [CrossRef]
- Xu, W.; Tang, Y.; Ji, Y.; Yu, H.; Li, Y.; Piao, C.; Xie, L. The association between serum selenium level and gestational diabetes mellitus: A systematic review and meta-analysis. Diabetes Metab. Res. Rev. 2022, 38, e3522. [Google Scholar] [CrossRef]
- Fontenelle, L.C.; de Araújo, D.S.C.; da Cunha Soares, T.; Cruz, K.J.C.; Henriques, G.S.; do Nascimento Marreiro, D. Nutritional status of selenium in overweight and obesity: A systematic review and meta-analysis. Clin. Nutr. 2022, 41, 862–884. [Google Scholar] [CrossRef]
- Kuria, A.; Tian, H.; Li, M.; Wang, Y.; Aaseth, J.O.; Zang, J.; Cao, Y. Selenium status in the body and cardiovascular disease: A systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2021, 61, 3616–3625. [Google Scholar] [CrossRef]
- Ding, J.; Liu, Q.; Liu, Z.; Guo, H.; Liang, J.; Zhang, Y. Associations of the Dietary Iron, Copper, and Selenium Level With Metabolic Syndrome: A Meta-Analysis of Observational Studies. Front. Nutr. 2021, 8, 810494. [Google Scholar] [CrossRef]
- Qiu, Z.; Geng, T.; Wan, Z.; Lu, Q.; Guo, J.; Liu, L.; Pan, A.; Liu, G. Serum selenium concentrations and risk of all-cause and heart disease mortality among individuals with type 2 diabetes. Am. J. Clin. Nutr. 2022, 115, 53–60. [Google Scholar] [CrossRef]
- Díaz-Ruiz, M.; Martínez-Triguero, M.L.; López-Ruiz, A.; Fernández-de la Cruz, F.; Bañuls, C.; Hernández-Mijares, A. Metabolic disorders and inflammation are associated with familial combined hyperlipemia. Clin. Chim. Acta 2019, 490, 194–199. [Google Scholar] [CrossRef]
- Püschel, G.P.; Klauder, J.; Henkel, J. Macrophages, Low-Grade Inflammation, Insulin Resistance and Hyperinsulinemia: A Mutual Ambiguous Relationship in the Development of Metabolic Diseases. J. Clin. Med. 2022, 11, 4358. [Google Scholar] [CrossRef]
- DeFronzo, R.A.; Tripathy, D. Skeletal Muscle Insulin Resistance Is the Primary Defect in Type 2 Diabetes. Diabetes Care 2009, 32 (Suppl. S2), S157–S163. [Google Scholar] [CrossRef]
- Norouzi, S.; Adulcikas, J.; Sohal, S.S.; Myers, S. Zinc transporters and insulin resistance: Therapeutic implications for type 2 diabetes and metabolic disease. J. Biomed. Sci. 2017, 24, 87. [Google Scholar] [CrossRef]
- Ogawa-Wong, A.N.; Berry, M.J.; Seale, L.A. Selenium and Metabolic Disorders: An Emphasis on Type 2 Diabetes Risk. Nutrients 2016, 8, 80. [Google Scholar] [CrossRef]
- Ezaki, O. The insulin-like effects of selenate in rat adipocytes. J. Biol. Chem. 1990, 265, 1124–1128. [Google Scholar] [CrossRef]
- Pasquier, A.; Vivot, K.; Erbs, E.; Spiegelhalter, C.; Zhang, Z.; Aubert, V.; Liu, Z.; Senkara, M.; Maillard, E.; Pinget, M.; et al. Lysosomal degradation of newly formed insulin granules contributes to β cell failure in diabetes. Nat. Commun. 2019, 10, 3312. [Google Scholar] [CrossRef] [PubMed]
- Kitabayashi, N.; Nakao, S.; Mita, Y.; Arisawa, K.; Hoshi, T.; Toyama, T.; Ishii, K.-A.; Takamura, T.; Noguchi, N.; Saito, Y. Role of selenoprotein P expression in the function of pancreatic β cells: Prevention of ferroptosis-like cell death and stress-induced nascent granule degradation. Free Radic. Biol. Med. 2022, 183, 89–103. [Google Scholar] [CrossRef] [PubMed]
- Tabrizi, R.; Akbari, M.; Moosazadeh, M.; Lankarani, K.B.; Heydari, S.T.; Kolahdooz, F.; Mohammadi, A.A.; Shabani, A.; Badehnoosh, B.; Jamilian, M.; et al. The Effects of Selenium Supplementation on Glucose Metabolism and Lipid Profiles Among Patients with Metabolic Diseases: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Horm. Metab. Res. 2017, 49, 826–830. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P.; Stranges, S. Epidemiology of selenium and type 2 diabetes: Can we make sense of it? Free Radic. Biol. Med. 2013, 65, 1557–1564. [Google Scholar] [CrossRef] [PubMed]
- Wasserman, D.H. Four grams of glucose. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E11–E21. [Google Scholar] [CrossRef] [PubMed]
- Cherkas, A.; Holota, S.; Mdzinarashvili, T.; Gabbianelli, R.; Zarkovic, N. Glucose as a Major Antioxidant: When, What for and Why It Fails? Antioxidants 2020, 9, 140. [Google Scholar] [CrossRef] [PubMed]
- Greenfield, M.S.; Doberne, L.; Kraemer, F.; Tobey, T.; Reaven, G. Assessment of Insulin Resistance with the Insulin Suppression Test and the Euglycemic Clamp. Diabetes 1981, 30, 387–392. [Google Scholar] [CrossRef]
- Sherwani, S.I.; Khan, H.A.; Ekhzaimy, A.; Masood, A.; Sakharkar, M.K. Significance of HbA1c Test in Diagnosis and Prognosis of Diabetic Patients. Biomark. Insights 2016, 11, 95–104. [Google Scholar] [CrossRef]
- Stróżyk, A.; Osica, Z.; Przybylak, J.D.; Kołodziej, M.; Zalewski, B.M.; Mrozikiewicz-Rakowska, B.; Szajewska, H. Effectiveness and safety of selenium supplementation for type 2 diabetes mellitus in adults: A systematic review of randomised controlled trials. J. Hum. Nutr. Diet. 2019, 32, 635–645. [Google Scholar] [CrossRef]
- Liu, Y.; Zeng, S.; Liu, Y.; Wu, W.; Shen, Y.; Zhang, L.; Li, C.; Chen, H.; Liu, A.; Shen, L.; et al. Synthesis and antidiabetic activity of selenium nanoparticles in the presence of polysaccharides from Catathelasma ventricosum. Int. J. Biol. Macromol. 2018, 114, 632–639. [Google Scholar] [CrossRef]
- Zeng, S.; Ke, Y.; Liu, Y.; Shen, Y.; Zhang, L.; Li, C.; Liu, A.; Shen, L.; Hu, X.; Wu, H.; et al. Synthesis and antidiabetic properties of chitosan-stabilized selenium nanoparticles. Colloids Surf. B Biointerfaces 2018, 170, 115–121. [Google Scholar] [CrossRef]
- Oztürk, Z.; Gurpinar, T.; Vural, K.; Boyacıoglu, S.; Korkmaz, M.; Var, A. Effects of selenium on endothelial dysfunction and metabolic profile in low dose streptozotocin induced diabetic rats fed a high fat diet. Biotech. Histochem. 2015, 90, 506–515. [Google Scholar] [CrossRef]
- Abdulmalek, S.A.; Balbaa, M. Synergistic effect of nano-selenium and metformin on type 2 diabetic rat model: Diabetic complications alleviation through insulin sensitivity, oxidative mediators and inflammatory markers. PLoS ONE 2019, 14, e0220779. [Google Scholar] [CrossRef]
- Kazemi, A.; Shim, S.R.; Jamali, N.; Hassanzadeh-Rostami, Z.; Soltani, S.; Sasani, N.; Mohsenpour, M.A.; Firoozi, D.; Basirat, R.; Hosseini, R.; et al. Comparison of nutritional supplements for glycemic control in type 2 diabetes: A systematic review and network meta-analysis of randomized trials. Diabetes Res. Clin. Pract. 2022, 191, 110037. [Google Scholar] [CrossRef]
- Bhale, A.S.; Venkataraman, K. Leveraging knowledge of HDLs major protein ApoA1: Structure, function, mutations, and potential therapeutics. Biomed. Pharmacother. 2022, 154, 113634. [Google Scholar] [CrossRef]
- Sena-Evangelista, K.C.M.; Pedrosa, L.F.C.; Paiva, M.S.M.O.; Dias, P.C.S.; Ferreira, D.Q.C.; Cozzolino, S.M.F.; Faulin, T.E.S.; Abdalla, D.S.P. The Hypolipidemic and Pleiotropic Effects of Rosuvastatin Are Not Enhanced by Its Association with Zinc and Selenium Supplementation in Coronary Artery Disease Patients: A Double Blind Randomized Controlled Study. PLoS ONE 2015, 10, e0119830. [Google Scholar] [CrossRef]
- Gharipour, M.; Sadeghi, M.; Haghjooy-Javanmard, S.; Hamledari, H.; Khosravi, E.; Dianatkhah, M.; Vaseghi, G. Effects of selenium intake on the expression of prostaglandin-endoperoxide synthase 2 (cyclooxygenase-2) and matrix metallopeptidase-9 genes in the coronary artery disease: Selenegene study, a double-blind randomized controlled trial. ARYA Atheroscler. J. 2021, 17, 2093. [Google Scholar] [CrossRef]
| Study | Study Location | Study Design | Groups | No. of Participants | Mean Age | Gender (M/F) | Intervention | Treatment Period | Type of Disease |
|---|---|---|---|---|---|---|---|---|---|
| Salimian 2022 [41] | Iran | Single-center double-blinded RCT | I | 26 | 57.6 ± 11.5 | NR | selenized yeast 200 μg/day | 24 weeks | Diabetic nephropathy |
| C | 27 | 61.5 ± 9.8 | NR | placebo | |||||
| Ghazi 2021 [42] | Iran | Single-center double-blinded RCT | I1 | 16 | 59.06 ± 8.55 | 12/4 | selenium-enriched yeast 200 μg/day | 8 weeks | Atherosclerosis |
| I2 | 16 | 58.62 ± 9.68 | 16/0 | sodium selenite 200 μg/day | |||||
| C | 17 | 53.58 ± 13.75 | 15/2 | Placebo | |||||
| Najib 2020 [43] | Iran | Multi-center double-blinded RCT | I | 26 | 29.19 ± 6.16 | 0/26 | selenium supplements 100 μg/day | 12 weeks | Gestational diabetes mellitus |
| C | 28 | 31.0 ± 4.43 | 0/28 | Placebo | |||||
| Kamali 2019 [30] | Iran | Single-center double-blinded RCT | I | 17 | 62.6 ± 11.6 | NR | selenium yeast 200 μg/day | 4 weeks | Coronary heart disease |
| C | 16 | 61.2 ± 4.6 | NR | Placebo | |||||
| Raygan 2018 [44] | Iran | Single-center double-blinded RCT | I | 26 | 70.7 ± 10.3 | 8/18 | selenium yeast 200 μg/day | 12 weeks | Congestive heart failure |
| C | 27 | 68.5 ± 7.7 | 8/19 | Placebo | |||||
| Bahmani 2016 [45] | Iran | Single-center double-blinded RCT | I | 30 | 63.1 ± 12.6 | 15/15 | selenium supplements 200 μg/day | 12 weeks | Diabetic nephropathy |
| C | 30 | 61.4 ± 9.3 | 15/15 | Placebo | |||||
| Farrokhian 2016 [46] | Iran | Single-center double-blinded RCT | I | 30 | NR | 10/20 | selenium yeast 200μg/day | 8 weeks | Type 2 diabetes mellitus and coronary heart disease |
| C | 30 | NR | 10/20 | Placebo | |||||
| Asemi 2015 [47] | Iran | Single-center double-blinded RCT | I | 35 | 27.6 ± 5.3 | 0/35 | selenium supplements 200 μg/day | 6 weeks | Gestational diabetes mellitus |
| C | 35 | 29.6 ± 3.6 | 0/35 | Placebo | |||||
| Faghihi 2014 [48] | Iran | Single-center double-blinded RCT | I | 33 | 53.54 ± 7.52 | 16/17 | sodium selenite 200 μg/day | 3 months | Type 2 diabetes mellitus |
| C | 27 | 55.76 ± 7.77 | 18/9 | Placebo | |||||
| Alizadeh 2012 [49] | Iran | Single-center double-blinded RCT | I | 17 | 36.6 ± 8.6 | 0/17 | selenium-enriched yeast 200 μg/day | 6 weeks | Obesity |
| C | 17 | 36.7 ± 8.3 | 0/17 | Placebo |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ouyang, J.; Cai, Y.; Song, Y.; Gao, Z.; Bai, R.; Wang, A. Potential Benefits of Selenium Supplementation in Reducing Insulin Resistance in Patients with Cardiometabolic Diseases: A Systematic Review and Meta-Analysis. Nutrients 2022, 14, 4933. https://doi.org/10.3390/nu14224933
Ouyang J, Cai Y, Song Y, Gao Z, Bai R, Wang A. Potential Benefits of Selenium Supplementation in Reducing Insulin Resistance in Patients with Cardiometabolic Diseases: A Systematic Review and Meta-Analysis. Nutrients. 2022; 14(22):4933. https://doi.org/10.3390/nu14224933
Chicago/Turabian StyleOuyang, Jiahui, Yajie Cai, Yewen Song, Zhuye Gao, Ruina Bai, and Anlu Wang. 2022. "Potential Benefits of Selenium Supplementation in Reducing Insulin Resistance in Patients with Cardiometabolic Diseases: A Systematic Review and Meta-Analysis" Nutrients 14, no. 22: 4933. https://doi.org/10.3390/nu14224933
APA StyleOuyang, J., Cai, Y., Song, Y., Gao, Z., Bai, R., & Wang, A. (2022). Potential Benefits of Selenium Supplementation in Reducing Insulin Resistance in Patients with Cardiometabolic Diseases: A Systematic Review and Meta-Analysis. Nutrients, 14(22), 4933. https://doi.org/10.3390/nu14224933