Vitamin D Status and SARS-CoV-2 Infection in a Cohort of Kidney Transplanted Patients
Abstract
:1. Introduction
2. Material and Methods
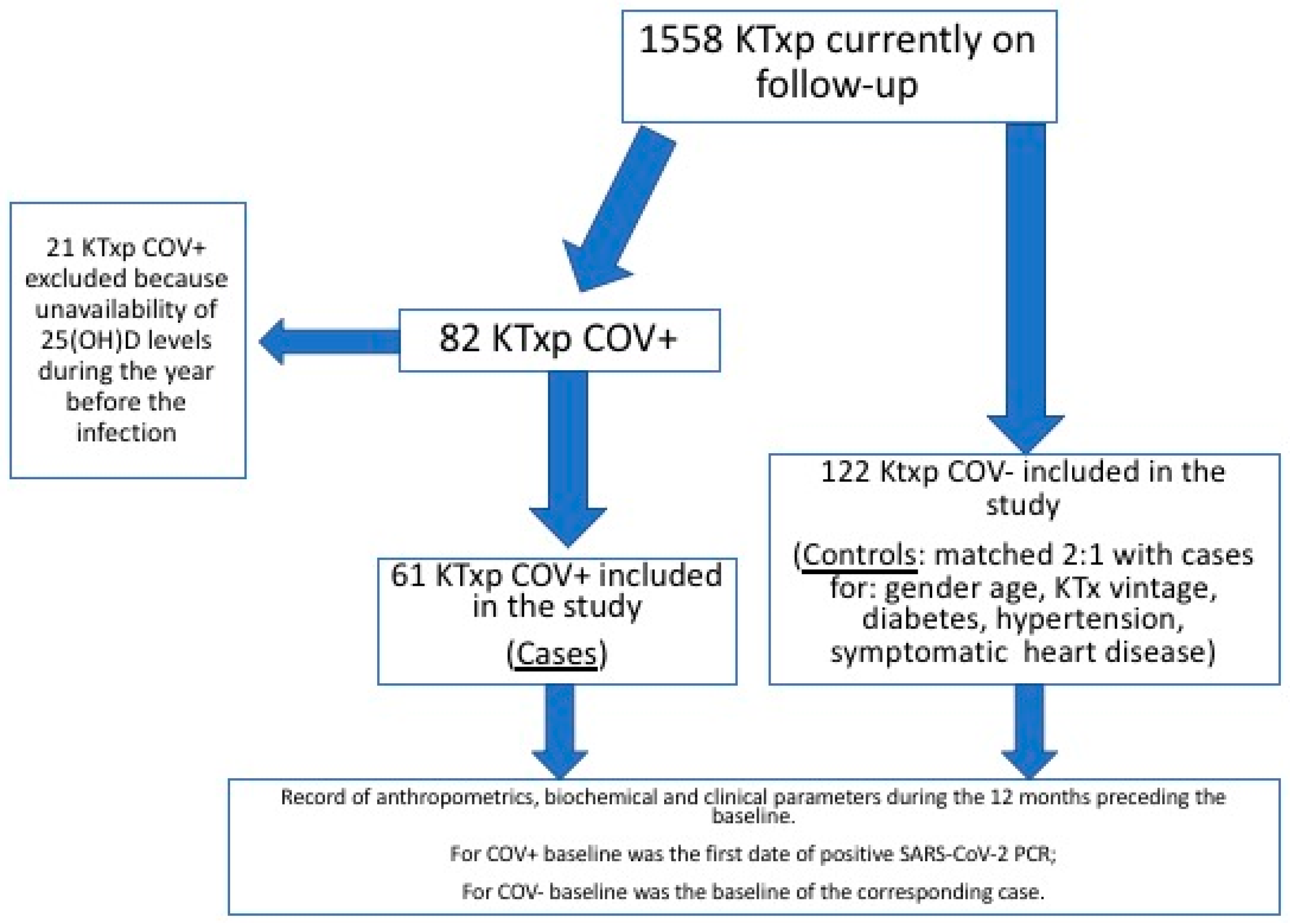
2.1. Studied Cohort and Design
2.2. Laboratory Measures
2.2.1. SARS-CoV-2 Assessment
2.2.2. Vitamin D
2.3. Statistical Analyses
3. Results
3.1. Overall Cohort Characteristics
3.2. Vitamin D Status and Other Biochemical Parameters
3.3. Discriminative Role of Vitamin D Status on COV Positivity
3.4. Therapy
3.5. SARS-CoV-2 Infection Related Outcomes
3.6. Relationship between Vitamin D Status in the 12 Monts before Baseline and SARS-CoV-2 Infection Severity
4. Discussion
4.1. Limitations of the Study
4.2. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Guan, W.-J.; Ni, Z.-Y.; Hu, Y.; Liang, W.-H.; Ou, C.-Q.; He, J.-X.; Liu, L.; Shan, H.; Lei, C.-L.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease in China. N. Engl. J. Med. 2019, 382, 1708–1720. [Google Scholar] [CrossRef]
- Baeke, F.; Takiishi, T.; Korf, H.; Gysemans, C.; Mathieu, C. Vitamin D: Modulator of the immune system. Curr. Opin. Pharmacol. 2010, 10, 482–496. [Google Scholar] [CrossRef] [PubMed]
- Greiller, C.L.; Martineau, A.R. Modulation of the Immune Response to Respiratory Viruses by Vitamin D. Nutrients 2015, 7, 4240–4270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdrabbo, M.; Birch, C.M.; Brandt, M.; Cicigoi, K.A.; Coffey, S.J.; Dolan, C.C.; Dvorak, H.; Gehrke, A.C.; Gerzema, A.E.L.; Hansen, A.; et al. Vitamin D and COVID-19: A review on the role of vitamin D in preventing and reducing the severity of COVID-19 infection. Protein Sci. 2021, 30, 2206–2220. [Google Scholar] [CrossRef]
- Alfieri, C.; Ruzhytska, O.; Vettoretti, S.; Caldiroli, L.; Cozzolino, M.; Messa, P. Native Hypovitaminosis D in CKD Patients: From Experimental Evidence to Clinical Practice. Nutrients 2019, 11, 1918. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Messa, P.; Regalia, A.; Alfieri, C.M. Nutritional Vitamin D in Renal Transplant Patients: Speculations and Reality. Nutrients 2017, 9, 550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Campise, M.; Alfieri, C.; Perego, M.; Tamborini, F.; Cresseri, D.; Gandolfo, M.; Binda, V.; Regalia, A.; Messa, P. COVID-19 Infection in Kidney Transplant Patients: An Italian One Year Single Centre Experience. Pathogens 2021, 10, 964. [Google Scholar] [CrossRef] [PubMed]
- Akalin, E.; Azzi, Y.; Bartash, R.; Seethamraju, H.; Parides, M.; Hemmige, V.; Ross, M.; Forest, S.; Goldstein, Y.D.; Ajaimy, M.; et al. Covid-19 and Kidney Transplantation. N. Engl. J. Med. 2020, 382, 2475–2477. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Interim Guidelines for Collecting, Handling, and Testing Clinical Specimens from Persons for Coronavirus Disease 2019 (COVID-19). 2020. Available online: https://www.cdc.gov/coronavirus/2019-ncov/lab/guidelines-clinical-specimens.html (accessed on 8 December 2021).
- Pecora, F.; Persico, F.; Argentiero, A.; Neglia, C.; Esposito, S. The Role of Micronutrients in Support of the Immune Response against Viral Infections. Nutrients 2020, 12, 3198. [Google Scholar] [CrossRef]
- Mohan, M.; Cherian, J.J.; Sharma, A. Exploring links between vitamin D deficiency and COVID-19. PLoS Pathog. 2020, 16, e1008874. [Google Scholar] [CrossRef]
- Brito, D.T.M.; Ribeiro, L.H.C.; Daltro, C.H.D.C.; Silva, R.B. The possible benefits of vitamin D in COVID-19. Nutrition 2021, 91–92, 111356. [Google Scholar] [CrossRef]
- Brenner, H. Vitamin D Supplementation to Prevent COVID-19 Infections and Deaths-Accumulating Evidence from Epidemiological and Intervention Studies Calls for Immediate Action. Nutrients 2021, 13, 411. [Google Scholar] [CrossRef]
- Boulkrane, M.S.; Ilina, V.; Melchakov, R.; Fedotova, J.; Drago, F.; Gozzo, L.; Das, U.N.; El-Aty, A.M.A.; Baranenko, D. COVID-19 Disease and Vitamin D: A Mini-Review. Front. Pharmacol. 2020, 11, 604579. [Google Scholar] [CrossRef]
- Malaguarnera, L. Vitamin D3 as Potential Treatment Adjuncts for COVID-19. Nutrients 2020, 12, 3512. [Google Scholar] [CrossRef]
- Kumar, R.; Rathi, H.; Haq, A.; Wimalawansa, S.J.; Sharma, A. Putative roles of vitamin D in modulating immune response and immunopathology associated with COVID-19. Virus Res. 2021, 292, 198235. [Google Scholar] [CrossRef] [PubMed]
- D’Avolio, A.; Avataneo, V.; Manca, A.; Cusato, J.; De Nicolò, A.; Lucchini, R.; Keller, F.; Cantù, M. 25-Hydroxyvitamin D Concentrations Are Lower in Patients with Positive PCR for SARS-CoV-2. Nutrients 2020, 12, 1359. [Google Scholar] [CrossRef]
- Kaufman, H.W.; Niles, J.K.; Kroll, M.H.; Bi, C.; Holick, M.F. SARS-CoV-2 positivity rates associated with circulating 25-hydroxyvitamin D levels. PLoS ONE 2020, 15, e0239252. [Google Scholar] [CrossRef] [PubMed]
- Teshome, A.; Adane, A.; Girma, B.; Mekonnen, Z.A. The Impact of Vitamin D Level on COVID-19 Infection: Systematic Review and Meta-Analysis. Front. Public Health 2021, 9, 624559. [Google Scholar] [CrossRef]
- Santaolalla, A.; Beckmann, K.; Kibaru, J.; Josephs, D.; Van Hemelrijck, M.; Irshad, S. Association between Vitamin D and Novel SARS-CoV-2 Respiratory Dysfunction—A Scoping Review of Current Evidence and Its Implication for COVID-19 Pandemic. Front. Physiol. 2020, 11, 564387. [Google Scholar] [CrossRef] [PubMed]
- Bassatne, A.; Basbous, M.; Chakhtoura, M.; El Zein, O.; Rahme, M.; Fuleihan, G.E.-H. The link between COVID-19 and Vitamin D (VIVID): A systematic review and meta-analysis. Metabolism 2021, 119, 154753. [Google Scholar] [CrossRef] [PubMed]
- Raisi-Estabragh, Z.; Martineau, A.R.; Curtis, E.M.; Moon, R.J.; Darling, A.; Lanham-New, S.; Ward, K.A.; Cooper, C.; Munroe, P.B.; Petersen, S.E.; et al. Vitamin D and coronavirus disease 2019 (COVID-19): Rapid evidence review. Aging Clin. Exp. Res. 2021, 33, 2031–2041. [Google Scholar] [CrossRef]
- Crafa, A.; Cannarella, R.; Condorelli, R.A.; Mongioì, L.M.; Barbagallo, F.; Aversa, A.; La Vignera, S.; Calogero, A.E. Influence of 25-hydroxy-cholecalciferol levels on SARS-CoV-2 infection and COVID-19 severity: A systematic review and meta-analysis. EClinicalMedicine 2021, 37, 100967. [Google Scholar] [CrossRef] [PubMed]
- Grove, A.; Osokogu, O.; Al-Khudairy, L.; Mehrabian, A.; Zanganeh, M.; Brown, A.; Court, R.; Taylor-Phillips, S.; Uthman, O.A.; McCarthy, N.; et al. Association between vitamin D supplementation or serum vitamin D level and susceptibility to SARS-CoV-2 infection or COVID-19 including clinical course, morbidity and mortality outcomes? A systematic review. BMJ Open 2021, 11, e043737. [Google Scholar] [CrossRef] [PubMed]
- Mercola, J.; Grant, W.B.; Wagner, C.L. Evidence Regarding Vitamin D and Risk of COVID-19 and Its Severity. Nutrients 2020, 12, 3361. [Google Scholar] [CrossRef] [PubMed]
- Benskin, L.L. A Basic Review of the Preliminary Evidence That COVID-19 Risk and Severity Is Increased in Vitamin D Deficiency. Front. Public Health 2020, 8, 513. [Google Scholar] [CrossRef]
- Campi, I.; Gennari, L.; Merlotti, D.; Mingiano, C.; Frosali, A.; Giovanelli, L.; Torlasco, C.; Pengo, M.F.; Heilbron, F.; Soranna, D.; et al. Vitamin D and COVID-19 severity and related mortality: A prospective study in Italy. BMC Infect. Dis. 2021, 21, 566. [Google Scholar] [CrossRef]
- Radujkovic, A.; Hippchen, T.; Tiwari-Heckler, S.; Dreher, S.; Boxberger, M.; Merle, U. Vitamin D Deficiency and Outcome of COVID-19 Patients. Nutrients 2020, 12, 2757. [Google Scholar] [CrossRef] [PubMed]
- De Smet, D.; De Smet, K.; Herroelen, P.; Gryspeerdt, S.; Martens, G.A. Serum 25(OH)D Level on Hospital Admission Associated with COVID-19 Stage and Mortality. Am. J. Clin. Pathol. 2021, 155, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Castillo, M.E.; Costa, L.M.E.; Barrios, J.M.V.; Díaz, J.F.A.; Miranda, J.L.; Bouillon, R.; Gomez, J.M.Q. Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: A pilot randomized clinical study. J. Steroid Biochem. Mol. Biol. 2020, 203, 105751. [Google Scholar] [CrossRef]
- Stroehlein, J.K.; Wallqvist, J.; Iannizzi, C.; Mikolajewska, A.; Metzendorf, M.-I.; Benstoem, C.; Meybohm, P.; Becker, M.; Skoetz, N.; Stegemann, M.; et al. Vitamin D supplementation for the treatment of COVID-19: A living systematic review. Cochrane Database Syst. Rev. 2021, 2021, CD015043. [Google Scholar]
- Bouillon, R.; Quesada-Gomez, J.M. Vitamin D Endocrine System and COVID-19. JBMR Plus 2021, 5, e10576. [Google Scholar] [CrossRef]
- Jean, G.; Souberbielle, J.C.; Chazot, C. Vitamin D in Chronic Kidney Disease and Dialysis Patients. Nutrients 2017, 9, 328. [Google Scholar] [CrossRef] [PubMed]
- Flythe, J.E.; Assimon, M.M.; Tugman, M.J.; Chang, E.H.; Gupta, S.; Shah, J.; Sosa, M.A.; Renaghan, A.D.; Melamed, M.L.; Wilson, F.P.; et al. Characteristics and Outcomes of Individuals with Pre-existing Kidney Disease and COVID-19 Admitted to Intensive Care Units in the United States. Am. J. Kidney Dis. 2021, 77, 190–203. [Google Scholar] [CrossRef]
- Ozturk, S.; Turgutalp, K.; Arici, M.; Odabas, A.R.; Altiparmak, M.R.; Aydin, Z.; Cebeci, E.; Basturk, T.; Soypacaci, Z.; Sahin, G.; et al. Mortality analysis of COVID-19 infection in chronic kidney disease, haemodialysis and renal transplant patients compared with patients without kidney disease: A nationwide analysis from Turkey. Nephrol. Dial. Transplant. 2020, 35, 2083–2095. [Google Scholar] [CrossRef] [PubMed]
- Oristrell, J.; Oliva, J.C.; Subirana, I.; Casado, E.; Domínguez, D.; Toloba, A.; Aguilera, P.; Esplugues, J.; Fafián, P.; Grau, M. Association of Calcitriol Supplementation with Reduced COVID-19 Mortality in Patients with Chronic Kidney Disease: A Population-Based Study. Biomedicines 2021, 9, 509. [Google Scholar] [CrossRef] [PubMed]
- Arenas Jimenez, M.D.; González-Parra, E.; Riera, M.; Rincón Bello, A.; López-Herradón, A.; Cao, H.; Hurtado, S.; Collado, S.; Ribera, L.; Barbosa, F.; et al. Mortality in Hemodialysis Patients with COVID-19, the Effect of Paricalcitol or Calcimimetics. Nutrients 2021, 13, 2559. [Google Scholar] [CrossRef]
- Drucker, D.J. Diabetes, obesity, metabolism, and SARS-CoV-2 infection: The end of the beginning. Cell Metab. 2021, 33, 479–498. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, H.R.; Adhikari, S.; Pulgarin, C.; Troxel, A.B.; Iturrate, E.; Johnson, S.B.; Hausvater, A.; Newman, J.D.; Berger, J.S.; Berger, S.; et al. Renin-Angiotensin-Aldosterone System Inhibitors and Risk of Covid-19. N. Engl. J. Med. 2020, 382, 2441–2448. [Google Scholar] [CrossRef]
- Alfieri, C.; Regalia, A.; Zanoni, F.; Vettoretti, S.; Cozzolino, M.; Messa, P. The Importance of Adherence in the Treatment of Secondary Hyperparathyroidism. Blood Purif. 2019, 47, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Quesada-Gomez, J.M.; Bouillon, R. Is calcifediol better than cholecalciferol for vitamin D supplementation? Osteoporos Int. 2018, 29, 1697–1711. [Google Scholar] [CrossRef] [PubMed]
- Zelzer, S.; Prüller, F.; Curcic, P.; Sloup, Z.; Holter, M.; Herrmann, M.; Mangge, H. Vitamin D Metabolites and Clinical Outcome in Hospitalized COVID-19 Patients. Nutrients 2021, 13, 2129. [Google Scholar] [CrossRef] [PubMed]
- Hernández, J.L.; Nan, D.; Fernandez-Ayala, M.; García-Unzueta, M.; Hernández-Hernández, M.A.; López-Hoyos, M.; Muñoz-Cacho, P.; Olmos, J.M.; Gutiérrez-Cuadra, M.; Ruiz-Cubillán, J.J.; et al. Vitamin D Status in Hospitalized Patients with SARS-CoV-2 Infection. J. Clin. Endocrinol. Metab. 2021, 106, e1343–e1353. [Google Scholar] [CrossRef] [PubMed]

| Overall Cohort (N 183) | COV+ (N 61) | COV− (N 122) | p | |
|---|---|---|---|---|
| Age, years, median (25–75%ile) | 53 (46–64) | 51 (44–62) | 55 (46–65) | 0.12 |
| Age at KTx, years, median (25–75%ile) | 44 (31–53) | 42 (29–52) | 45 (32–53) | 0.51 |
| KTx vintage, years, median (25–75%ile) | 8 (3–15) | 8 (3–15) | 7 (2–15) | 0.78 |
| Gender, n (F/M, %) | 72/111 (39/61) | 24/37 (39/61) | 48/74 (39/61) | 0.87 |
| Diabetes, n (%) | 33 (18) | 11 (18) | 22 (18) | 0.81 |
| Hypertension, n (%) | 156 (85) | 52 (85) | 104 (85) | 0.82 |
| Symptomatic heart disease, n (%) | 6 (3) | 2 (3) | 4 (3) | 0.66 |
| COV+ (N 61) | COV− (N 122) | p | |
|---|---|---|---|
| BMI, kg/m2 | 24.5 (21.7–26.9) | 24.2 (21.1–26.7) | 0.99 |
| 25(OH)D, ng/mL | 19 (12–26) | 23 (17–31) | 0.01 |
| 1,25(OH)2D, pg/mL * | 42 (34–49) | 48 (34–58) | 0.14 |
| Ca, mg/dL | 9.5 (9.1–9.9) | 9.4 (9.2–9.8) | 0.73 |
| P, mg/dL | 3.1 (2.6–3.5) | 2.9 (2.6–3.4) | 0.44 |
| Mg, mg/dL | 1.8 (1.6–1.8) | 1.7 (1.6–1.8) | 0.09 |
| PTH, pg/ml | 59 (41–85) | 61 (46–91) | 0.37 |
| Serum Creatinine, mg/dL | 1.4 (1.2–1.8) | 1.3 (1.1–1.8) | 0.09 |
| U-Prot, gr/day | 0.15 (0.11–0.33) | 0.18 (0.13–0.31) | 0.53 |
| Serum Albumin, mg/dL | 4.2 (3.9–4.4) | 4.1 (3.9–4.3) | 0.46 |
| CRP, mg/dL | 0.2 (0–1–0.4) | 0.2 (0.1–0.5) | 0.56 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Regalia, A.; Benedetti, M.; Malvica, S.; Alfieri, C.; Campise, M.; Cresseri, D.; Gandolfo, M.T.; Tripodi, F.; Castellano, G.; Messa, P. Vitamin D Status and SARS-CoV-2 Infection in a Cohort of Kidney Transplanted Patients. Nutrients 2022, 14, 317. https://doi.org/10.3390/nu14020317
Regalia A, Benedetti M, Malvica S, Alfieri C, Campise M, Cresseri D, Gandolfo MT, Tripodi F, Castellano G, Messa P. Vitamin D Status and SARS-CoV-2 Infection in a Cohort of Kidney Transplanted Patients. Nutrients. 2022; 14(2):317. https://doi.org/10.3390/nu14020317
Chicago/Turabian StyleRegalia, Anna, Matteo Benedetti, Silvia Malvica, Carlo Alfieri, Mariarosaria Campise, Donata Cresseri, Maria Teresa Gandolfo, Federica Tripodi, Giuseppe Castellano, and Piergiorgio Messa. 2022. "Vitamin D Status and SARS-CoV-2 Infection in a Cohort of Kidney Transplanted Patients" Nutrients 14, no. 2: 317. https://doi.org/10.3390/nu14020317
APA StyleRegalia, A., Benedetti, M., Malvica, S., Alfieri, C., Campise, M., Cresseri, D., Gandolfo, M. T., Tripodi, F., Castellano, G., & Messa, P. (2022). Vitamin D Status and SARS-CoV-2 Infection in a Cohort of Kidney Transplanted Patients. Nutrients, 14(2), 317. https://doi.org/10.3390/nu14020317





