Molecular Mechanisms and Health Benefits of Ghrelin: A Narrative Review
Abstract
1. Introduction
2. Ghrelin and the GI Tract
3. Ghrelin and Orexis
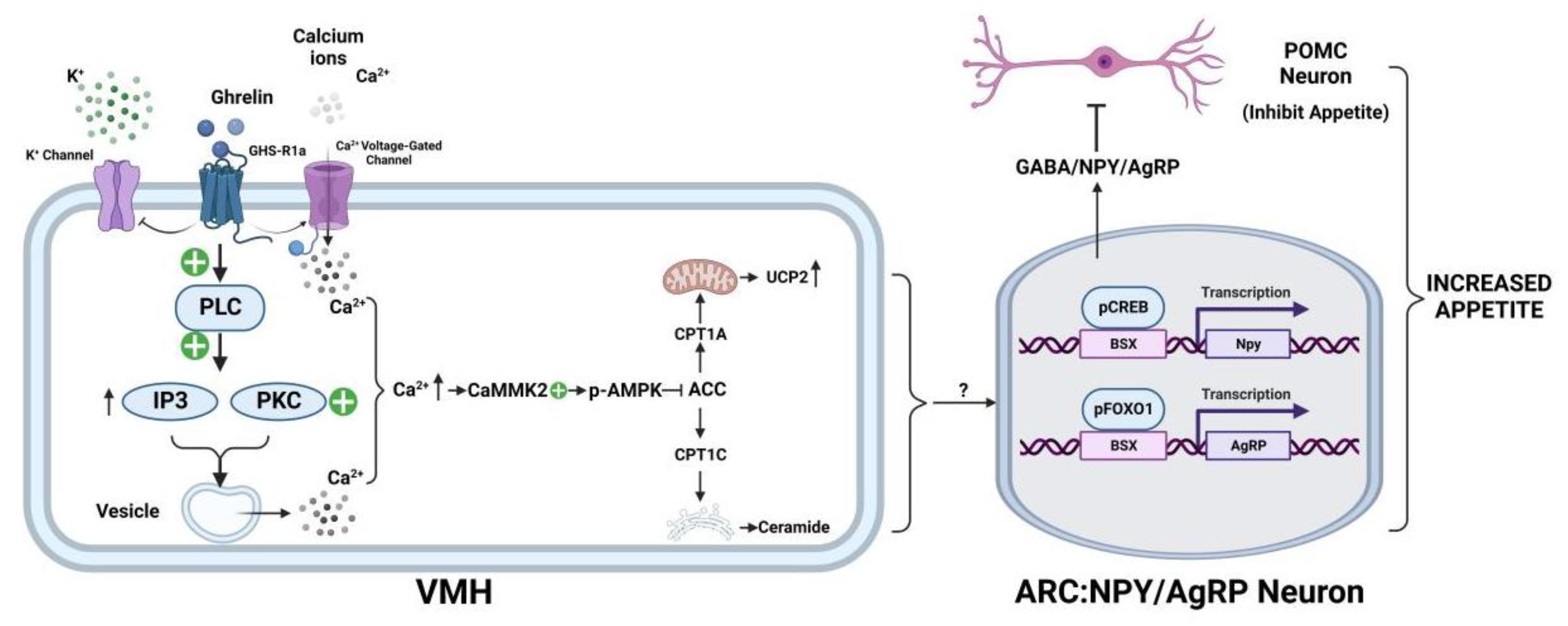
3.1. Ghrelin’s Involvement in the Neuroregulation of Appetite
3.2. Ghrelin in Obesity
3.3. Ghrelin and Anorexia Nervosa
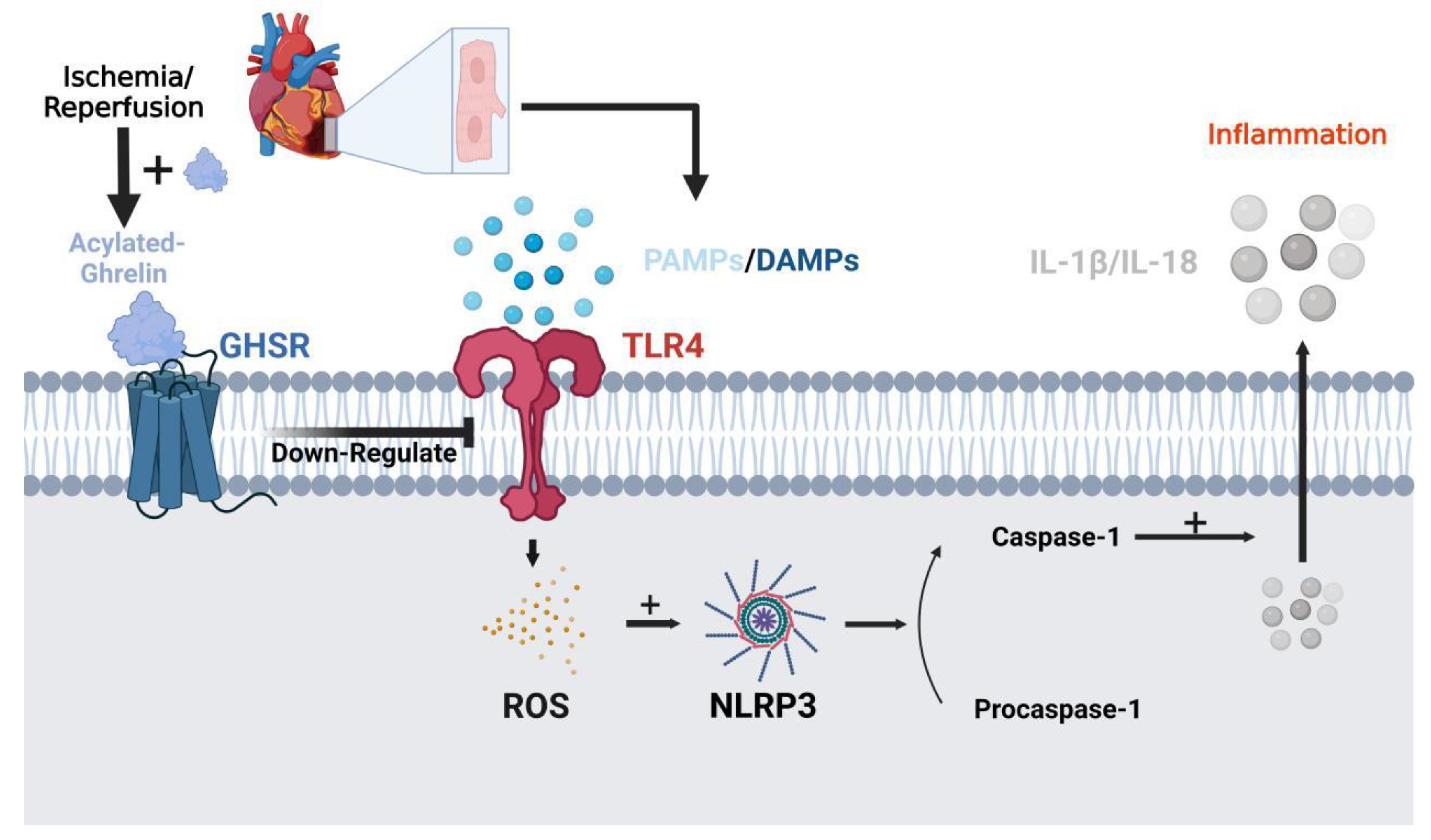
4. Ghrelin in the Cardiovascular System
5. Ghrelin in Stress-Induced Anxiety
6. Ghrelin in Cachexia
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kojima, M.; Hosoda, H.; Date, Y.; Nakazato, M.; Matsuo, H.; Kangawa, K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 1999, 402, 656–660. [Google Scholar] [CrossRef] [PubMed]
- Date, Y.; Kojima, M.; Hosoda, H.; Sawaguchi, A.; Mondal, M.S.; Suganuma, T.; Matsukura, S.; Kangawa, K.; Nakazato, M. Ghrelin, a novel growth hormone-releasing acylated peptide, is synthesized in a distinct endocrine cell type in the gastrointestinal tracts of rats and humans. Endocrinology 2000, 141, 4255–4261. [Google Scholar] [CrossRef]
- Kojima, M.; Kangawa, K. Ghrelin: Structure and function. Physiol. Rev. 2005, 85, 495–522. [Google Scholar] [CrossRef]
- Zhang, J.V.; Ren, P.G.; Avsian-Kretchmer, O.; Luo, C.W.; Rauch, R.; Klein, C.; Hsueh, A.J. Obestatin, a peptide encoded by the ghrelin gene, opposes ghrelin’s effects on food intake. Science 2005, 310, 996–999. [Google Scholar] [CrossRef] [PubMed]
- Moechars, D.; Depoortere, I.; Moreaux, B.; de Smet, B.; Goris, I.; Hoskens, L.; Daneels, G.; Kass, S.; Ver, D.L.; Peeters, T.; et al. Altered gastrointestinal and metabolic function in the GPR39-obestatin receptor-knockout mouse. Gastroenterology 2006, 131, 1131–1141. [Google Scholar] [CrossRef] [PubMed]
- Lauwers, E.; Landuyt, B.; Arckens, L.; Schoofs, L.; Luyten, W. Obestatin does not activate orphan G protein-coupled receptor GPR39. Biochem. Biophys. Res. Commun. 2006, 351, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Holst, B.; Egerod, K.L.; Schild, E.; Vickers, S.P.; Cheetham, S.; Gerlach, L.O.; Storjohann, L.; Stidsen, C.E.; Jones, R.; Beck-Sickinger, A.G.; et al. GPR39 signaling is stimulated by zinc ions but not by obestatin. Endocrinology 2007, 148, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Popovics, P.; Stewart, A.J. GPR39: A Zn(2+)-activated G protein-coupled receptor that regulates pancreatic, gastrointestinal and neuronal functions. Cell. Mol. Life Sci. 2011, 68, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Villarreal, D.; Pradhan, G.; Zhou, Y.; Xue, B.; Sun, Y. Diverse and Complementary Effects of Ghrelin and Obestatin. Biomolecules 2022, 12, 517. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Brown, M.S.; Liang, G.; Grishin, N.V.; Goldstein, J.L. Identification of the acyltransferase that octanoylates ghrelin, an appetite-stimulating peptide hormone. Cell 2008, 132, 387–396. [Google Scholar] [CrossRef]
- Gutierrez, J.A.; Solenberg, P.J.; Perkins, D.R.; Willency, J.A.; Knierman, M.D.; Jin, Z.; Witcher, D.R.; Luo, S.; Onyia, J.E.; Hale, J.E. Ghrelin octanoylation mediated by an orphan lipid transferase. Proc. Natl. Acad. Sci. USA 2008, 105, 6320–6325. [Google Scholar] [CrossRef]
- Hattori, N.; Saito, T.; Yagyu, T.; Jiang, B.H.; Kitagawa, K.; Inagaki, C. GH, GH receptor, GH secretagogue receptor, and ghrelin expression in human T cells, B cells, and neutrophils. J. Clin. Endocrinol. Metab. 2001, 86, 4284–4291. [Google Scholar] [CrossRef] [PubMed]
- Howard, A.D.; Feighner, S.D.; Cully, D.F.; Arena, J.P.; Liberator, P.A.; Rosenblum, C.I.; Hamelin, M.; Hreniuk, D.L.; Palyha, O.C.; Anderson, J.; et al. A receptor in pituitary and hypothalamus that functions in growth hormone release. Science 1996, 273, 974–977. [Google Scholar] [CrossRef] [PubMed]
- Guan, X.M.; Yu, H.; Palyha, O.C.; McKee, K.K.; Feighner, S.D.; Sirinathsinghji, D.J.; Smith, R.G.; Van der Ploeg, L.H.; Howard, A.D. Distribution of mRNA encoding the growth hormone secretagogue receptor in brain and peripheral tissues. Brain Res. Mol. Brain Res. 1997, 48, 23–29. [Google Scholar] [CrossRef]
- Gnanapavan, S.; Kola, B.; Bustin, S.A.; Morris, D.G.; McGee, P.; Fairclough, P.; Bhattacharya, S.; Carpenter, R.; Grossman, A.B.; Korbonits, M. The tissue distribution of the mRNA of ghrelin and subtypes of its receptor, GHS-R, in humans. J. Clin. Endocrinol. Metab. 2002, 87, 2988. [Google Scholar] [CrossRef] [PubMed]
- Garcia, A.; Alvarez, C.V.; Smith, R.G.; Dieguez, C. Regulation of Pit-1 expression by ghrelin and GHRP-6 through the GH secretagogue receptor. Mol. Endocrinol. 2001, 15, 1484–1495. [Google Scholar] [CrossRef][Green Version]
- Chowen, J.A.; Argente, J. Ghrelin: A Link Between Energy Homeostasis and the Immune System. Endocrinology 2017, 158, 2077–2081. [Google Scholar] [CrossRef] [PubMed]
- Steinert, R.E.; Feinle-Bisset, C.; Asarian, L.; Horowitz, M.; Beglinger, C.; Geary, N. Ghrelin, CCK, GLP-1, and PYY(3–36): Secretory Controls and Physiological Roles in Eating and Glycemia in Health, Obesity, and After RYGB. Physiol. Rev. 2017, 97, 411–463. [Google Scholar] [CrossRef] [PubMed]
- Callaghan, B.; Hunne, B.; Hirayama, H.; Sartor, D.M.; Nguyen, T.V.; Abogadie, F.C.; Ferens, D.; McIntyre, P.; Ban, K.; Baell, J.; et al. Sites of action of ghrelin receptor ligands in cardiovascular control. Am. J. Physiol. Heart Circ. Physiol. 2012, 303, H1011–H1021. [Google Scholar] [CrossRef]
- Lutter, M.; Sakata, I.; Osborne-Lawrence, S.; Rovinsky, S.A.; Anderson, J.G.; Jung, S.; Birnbaum, S.; Yanagisawa, M.; Elmquist, J.K.; Nestler, E.J.; et al. The orexigenic hormone ghrelin defends against depressive symptoms of chronic stress. Nat. Neurosci. 2008, 11, 752–753. [Google Scholar] [CrossRef] [PubMed]
- Levin, F.; Edholm, T.; Schmidt, P.T.; Gryback, P.; Jacobsson, H.; Degerblad, M.; Hoybye, C.; Holst, J.J.; Rehfeld, J.F.; Hellstrom, P.M.; et al. Ghrelin stimulates gastric emptying and hunger in normal-weight humans. J. Clin. Endocrinol. Metab. 2006, 91, 3296–3302. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.; Camilleri, M.; Eckert, D.; Burton, D.; Joyner, M.; Acosta, A. Physical activity is associated with accelerated gastric emptying and increased ghrelin in obesity. Neurogastroenterol. Motil. 2020, 32, e13879. [Google Scholar] [CrossRef]
- James, J.; Mair, S.; Doll, W.; Sandefer, E.; Wurtman, D.; Maurer, A.; Deane, A.M.; Harris, M.S. The effects of ulimorelin, a ghrelin agonist, on liquid gastric emptying and colonic transit in humans. Neurogastroenterol. Motil. 2020, 32, e13784. [Google Scholar] [CrossRef] [PubMed]
- Dembinski, A.; Warzecha, Z.; Ceranowicz, P.; Bielanski, W.; Cieszkowski, J.; Dembinski, M.; Pawlik, W.W.; Kuwahara, A.; Kato, I.; Konturek, P.C. Variable effect of ghrelin administration on pancreatic development in young rats. Role of insulin-like growth factor-1. J. Physiol. Pharmacol. 2005, 56, 555–570. [Google Scholar]
- Warzecha, Z.; Dembinski, A.; Ceranowicz, P.; Dembinski, M.; Cieszkowski, J.; Konturek, S.J.; Polus, A.; Pawlik, W.W.; Kuwahara, A.; Kato, I.; et al. Influence of ghrelin on gastric and duodenal growth and expression of digestive enzymes in young mature rats. J. Physiol. Pharmacol. 2006, 57, 425–437. [Google Scholar]
- Warzecha, Z.; Dembinski, A.; Ceranowicz, P.; Dembinski, M.; Cieszkowski, J.; Bielanski, W.; Pawlik, W.W.; Kuwahara, A.; Kato, I. Dual age-dependent effect of ghrelin administration on serum level of insulin-like growth factor-1 and gastric growth in young rats. Eur. J. Pharmacol. 2006, 529, 145–150. [Google Scholar] [CrossRef]
- Frascarelli, S.; Ghelardoni, S.; Ronca-Testoni, S.; Zucchi, R. Effect of ghrelin and synthetic growth hormone secretagogues in normal and ischemic rat heart. Basic Res. Cardiol. 2003, 98, 401–405. [Google Scholar] [CrossRef]
- Takeda, R.; Nishimatsu, H.; Suzuki, E.; Satonaka, H.; Nagata, D.; Oba, S.; Sata, M.; Takahashi, M.; Yamamoto, Y.; Terauchi, Y.; et al. Ghrelin improves renal function in mice with ischemic acute renal failure. J. Am. Soc. Nephrol. 2006, 17, 113–121. [Google Scholar] [CrossRef]
- Zhang, Q.; Huang, C.; Meng, B.; Tang, T.; Shi, Q.; Yang, H. Acute effect of Ghrelin on ischemia/reperfusion injury in the rat spinal cord. Int. J. Mol. Sci. 2012, 13, 9864–9876. [Google Scholar] [CrossRef] [PubMed]
- Dembinski, A.; Warzecha, Z.; Ceranowicz, P.; Cieszkowski, J.; Pawlik, W.W.; Tomaszewska, R.; Kusnierz-Cabala, B.; Naskalski, J.W.; Kuwahara, A.; Kato, I. Role of growth hormone and insulin-like growth factor-1 in the protective effect of ghrelin in ischemia/reperfusion-induced acute pancreatitis. Growth Horm. IGF Res. 2006, 16, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Warzecha, Z.; Ceranowicz, P.; Dembinski, A.; Cieszkowski, J.; Kusnierz-Cabala, B.; Tomaszewska, R.; Kuwahara, A.; Kato, I. Therapeutic effect of ghrelin in the course of cerulein-induced acute pancreatitis in rats. J. Physiol. Pharmacol. 2010, 61, 419–427. [Google Scholar]
- Ceranowicz, D.; Warzecha, Z.; Dembinski, A.; Ceranowicz, P.; Cieszkowski, J.; Kusnierz-Cabala, B.; Tomaszewska, R.; Kuwahara, A.; Kato, I. Role of hormonal axis, growth hormone—IGF-1, in the therapeutic effect of ghrelin in the course of cerulein-induced acute pancreatitis. J. Physiol. Pharmacol. 2010, 61, 599–606. [Google Scholar] [PubMed]
- Bukowczan, J.; Warzecha, Z.; Ceranowicz, P.; Kusnierz-Cabala, B.; Tomaszewska, R.; Dembinski, A. Therapeutic effect of ghrelin in the course of ischemia/reperfusion-induced acute pancreatitis. Curr. Pharm. Des. 2015, 21, 2284–2290. [Google Scholar] [CrossRef]
- Ceranowicz, P.; Warzecha, Z.; Dembinski, A.; Sendur, R.; Cieszkowski, J.; Ceranowicz, D.; Pawlik, W.W.; Kuwahara, A.; Kato, I.; Konturek, P.C. Treatment with ghrelin accelerates the healing of acetic acid-induced gastric and duodenal ulcers in rats. J. Physiol. Pharmacol. 2009, 60, 87–98. [Google Scholar] [PubMed]
- Warzecha, Z.; Ceranowicz, D.; Dembinski, A.; Ceranowicz, P.; Cieszkowski, J.; Kuwahara, A.; Kato, I.; Dembinski, M.; Konturek, P.C. Ghrelin accelerates the healing of cysteamine-induced duodenal ulcers in rats. Med. Sci. Monit. 2012, 18, R181–R187. [Google Scholar] [CrossRef]
- Maduzia, D.; Matuszyk, A.; Ceranowicz, D.; Warzecha, Z.; Ceranowicz, P.; Fyderek, K.; Galazka, K.; Dembinski, A. The influence of pretreatment with ghrelin on the development of acetic-acid-induced colitis in rats. J. Physiol. Pharmacol. 2015, 66, 875–885. [Google Scholar] [PubMed]
- Matuszyk, A.; Ceranowicz, D.; Warzecha, Z.; Ceranowicz, P.; Fyderek, K.; Galazka, K.; Cieszkowski, J.; Bonior, J.; Jaworek, J.; Pihut, M.; et al. The Influence of Ghrelin on the Development of Dextran Sodium Sulfate-Induced Colitis in Rats. Biomed Res. Int. 2015, 2015, 718314. [Google Scholar] [CrossRef]
- Matuszyk, A.; Ceranowicz, P.; Warzecha, Z.; Cieszkowski, J.; Ceranowicz, D.; Galazka, K.; Bonior, J.; Jaworek, J.; Bartus, K.; Gil, K.; et al. Exogenous Ghrelin Accelerates the Healing of Acetic Acid-Induced Colitis in Rats. Int. J. Mol. Sci. 2016, 17, 1455. [Google Scholar] [CrossRef] [PubMed]
- Warzecha, Z.; Kownacki, P.; Ceranowicz, P.; Dembinski, M.; Cieszkowski, J.; Dembinski, A. Ghrelin accelerates the healing of oral ulcers in non-sialoadenectomized and sialoadenectomized rats. J. Physiol. Pharmacol. 2013, 64, 657–668. [Google Scholar]
- Cieszkowski, J.; Warzecha, Z.; Ceranowicz, P.; Ceranowicz, D.; Kusnierz-Cabala, B.; Pedziwiatr, M.; Dembinski, M.; Ambrozy, T.; Kaczmarzyk, T.; Pihut, M.; et al. Therapeutic effect of exogenous ghrelin in the healing of gingival ulcers is mediated by the release of endogenous growth hormone and insulin-like growth factor-1. J. Physiol. Pharmacol. 2017, 68, 609–617. [Google Scholar]
- Jang, E.J.; Park, S.W.; Park, J.S.; Park, S.J.; Hahm, K.B.; Paik, S.Y.; Sin, M.K.; Lee, E.S.; Oh, S.W.; Park, C.Y.; et al. The influence of the eradication of Helicobacter pylori on gastric ghrelin, appetite, and body mass index in patients with peptic ulcer disease. J. Gastroenterol. Hepatol. 2008, 23 (Suppl. S2), S278–S285. [Google Scholar] [CrossRef] [PubMed]
- Tatsuguchi, A.; Miyake, K.; Gudis, K.; Futagami, S.; Tsukui, T.; Wada, K.; Kishida, T.; Fukuda, Y.; Sugisaki, Y.; Sakamoto, C. Effect of Helicobacter pylori infection on ghrelin expression in human gastric mucosa. Am. J. Gastroenterol. 2004, 99, 2121–2127. [Google Scholar] [CrossRef]
- Warzecha, Z.; Ceranowicz, P.; Dembinski, M.; Cieszkowski, J.; Ginter, G.; Ptak-Belowska, A.; Dembinski, A. Involvement of cyclooxygenase-1 and cyclooxygenase-2 activity in the therapeutic effect of ghrelin in the course of ethanol-induced gastric ulcers in rats. J. Physiol. Pharmacol. 2014, 65, 95–106. [Google Scholar]
- Sibilia, V.; Rindi, G.; Pagani, F.; Rapetti, D.; Locatelli, V.; Torsello, A.; Campanini, N.; Deghenghi, R.; Netti, C. Ghrelin protects against ethanol-induced gastric ulcers in rats: Studies on the mechanisms of action. Endocrinology 2003, 144, 353–359. [Google Scholar] [CrossRef]
- Allam, M.M.; El-Gohary, O.A. Gastroprotective effect of ghrelin against indomethacin-induced gastric injury in rats: Possible role of heme oxygenase-1 pathway. Gen. Physiol. Biophys. 2017, 36, 321–330. [Google Scholar] [CrossRef]
- Iwasaki, E.; Suzuki, H.; Masaoka, T.; Nishizawa, T.; Hosoda, H.; Kangawa, K.; Hibi, T. Enhanced gastric ghrelin production and secretion in rats with gastric outlet obstruction. Dig. Dis. Sci. 2012, 57, 858–864. [Google Scholar] [CrossRef]
- Bonior, J.; Warzecha, Z.; Ceranowicz, P.; Gajdosz, R.; Pierzchalski, P.; Kot, M.; Leja-Szpak, A.; Nawrot-Porabka, K.; Link-Lenczowski, P.; Pedziwiatr, M.; et al. Capsaicin-Sensitive Sensory Nerves Are Necessary for the Protective Effect of Ghrelin in Cerulein-Induced Acute Pancreatitis in Rats. Int. J. Mol. Sci. 2017, 18, 1402. [Google Scholar] [CrossRef] [PubMed]
- Bonior, J.; Ceranowicz, P.; Gajdosz, R.; Kusnierz-Cabala, B.; Pierzchalski, P.; Warzecha, Z.; Dembinski, A.; Pedziwiatr, M.; Kot, M.; Leja-Szpak, A.; et al. Molecular Ghrelin System in the Pancreatic Acinar Cells: The Role of the Polypeptide, Caerulein and Sensory Nerves. Int. J. Mol. Sci. 2017, 18, 929. [Google Scholar] [CrossRef] [PubMed]
- Davenport, A.P.; Bonner, T.I.; Foord, S.M.; Harmar, A.J.; Neubig, R.R.; Pin, J.P.; Spedding, M.; Kojima, M.; Kangawa, K. International Union of Pharmacology. LVI. Ghrelin receptor nomenclature, distribution, and function. Pharmacol. Rev. 2005, 57, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Kojima, M.; Hosoda, H.; Kangawa, K. Purification and distribution of ghrelin: The natural endogenous ligand for the growth hormone secretagogue receptor. Horm Res. 2001, 56 (Suppl. S1), 93–97. [Google Scholar] [CrossRef]
- Chen, C.; Wu, D.; Clarke, I.J. Signal transduction systems employed by synthetic GH-releasing peptides in somatotrophs. J. Endocrinol. 1996, 148, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Kohno, D.; Gao, H.Z.; Muroya, S.; Kikuyama, S.; Yada, T. Ghrelin directly interacts with neuropeptide-Y-containing neurons in the rat arcuate nucleus: Ca2+ signaling via protein kinase A and N-type channel-dependent mechanisms and cross-talk with leptin and orexin. Diabetes 2003, 52, 948–956. [Google Scholar] [CrossRef]
- Anderson, K.A.; Ribar, T.J.; Lin, F.; Noeldner, P.K.; Green, M.F.; Muehlbauer, M.J.; Witters, L.A.; Kemp, B.E.; Means, A.R. Hypothalamic CaMKK2 contributes to the regulation of energy balance. Cell Metab. 2008, 7, 377–388. [Google Scholar] [CrossRef]
- Dietrich, M.O.; Antunes, C.; Geliang, G.; Liu, Z.W.; Borok, E.; Nie, Y.; Xu, A.W.; Souza, D.O.; Gao, Q.; Diano, S.; et al. Agrp neurons mediate Sirt1’s action on the melanocortin system and energy balance: Roles for Sirt1 in neuronal firing and synaptic plasticity. J. Neurosci. 2010, 30, 11815–11825. [Google Scholar] [CrossRef] [PubMed]
- Lam, T.K.; Schwartz, G.J.; Rossetti, L. Hypothalamic sensing of fatty acids. Nat. Neurosci. 2005, 8, 579–584. [Google Scholar] [CrossRef] [PubMed]
- Sierra, A.Y.; Gratacos, E.; Carrasco, P.; Clotet, J.; Urena, J.; Serra, D.; Asins, G.; Hegardt, F.G.; Casals, N. CPT1c is localized in endoplasmic reticulum of neurons and has carnitine palmitoyltransferase activity. J. Biol. Chem. 2008, 283, 6878–6885. [Google Scholar] [CrossRef] [PubMed]
- Hao, F.; Tian, M.; Zhang, X.; Jin, X.; Jiang, Y.; Sun, X.; Wang, Y.; Peng, P.; Liu, J.; Xia, C.; et al. Butyrate enhances CPT1A activity to promote fatty acid oxidation and iTreg differentiation. Proc. Natl. Acad. Sci. USA 2021, 118, e2014681118. [Google Scholar] [CrossRef]
- Andrews, Z.B.; Liu, Z.W.; Walllingford, N.; Erion, D.M.; Borok, E.; Friedman, J.M.; Tschop, M.H.; Shanabrough, M.; Cline, G.; Shulman, G.I.; et al. UCP2 mediates ghrelin’s action on NPY/AgRP neurons by lowering free radicals. Nature 2008, 454, 846–851. [Google Scholar] [CrossRef]
- Lage, R.; Vazquez, M.J.; Varela, L.; Saha, A.K.; Vidal-Puig, A.; Nogueiras, R.; Dieguez, C.; Lopez, M. Ghrelin effects on neuropeptides in the rat hypothalamus depend on fatty acid metabolism actions on BSX but not on gender. FASEB J. 2010, 24, 2670–2679. [Google Scholar] [CrossRef]
- Diano, S. New aspects of melanocortin signaling: A role for PRCP in alpha-MSH degradation. Front. Neuroendocrinol. 2011, 32, 70–83. [Google Scholar] [CrossRef]
- Morton, G.J.; Cummings, D.E.; Baskin, D.G.; Barsh, G.S.; Schwartz, M.W. Central nervous system control of food intake and body weight. Nature 2006, 443, 289–295. [Google Scholar] [CrossRef]
- Horvath, T.L.; Naftolin, F.; Kalra, S.P.; Leranth, C. Neuropeptide-Y innervation of beta-endorphin-containing cells in the rat mediobasal hypothalamus: A light and electron microscopic double immunostaining analysis. Endocrinology 1992, 131, 2461–2467. [Google Scholar] [CrossRef]
- Kamegai, J.; Tamura, H.; Shimizu, T.; Ishii, S.; Sugihara, H.; Wakabayashi, I. Chronic central infusion of ghrelin increases hypothalamic neuropeptide Y and Agouti-related protein mRNA levels and body weight in rats. Diabetes 2001, 50, 2438–2443. [Google Scholar] [CrossRef]
- Mani, B.K.; Uchida, A.; Lee, Y.; Osborne-Lawrence, S.; Charron, M.J.; Unger, R.H.; Berglund, E.D.; Zigman, J.M. Hypoglycemic Effect of Combined Ghrelin and Glucagon Receptor Blockade. Diabetes 2017, 66, 1847–1857. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.K.; Dall, R.; Hosoda, H.; Kojima, M.; Kangawa, K.; Christiansen, J.S.; Jorgensen, J.O. Weight loss increases circulating levels of ghrelin in human obesity. Clin. Endocrinol. 2002, 56, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Resnick, J.L.; Nicholls, R.D.; Wevrick, R. Recommendations for the investigation of animal models of Prader-Willi syndrome. Mamm. Genome 2013, 24, 165–178. [Google Scholar] [CrossRef] [PubMed]
- Cummings, D.E.; Clement, K.; Purnell, J.Q.; Vaisse, C.; Foster, K.E.; Frayo, R.S.; Schwartz, M.W.; Basdevant, A.; Weigle, D.S. Elevated plasma ghrelin levels in Prader Willi syndrome. Nat. Med. 2002, 8, 643–644. [Google Scholar] [CrossRef] [PubMed]
- DelParigi, A.; Tschop, M.; Heiman, M.L.; Salbe, A.D.; Vozarova, B.; Sell, S.M.; Bunt, J.C.; Tataranni, P.A. High circulating ghrelin: A potential cause for hyperphagia and obesity in prader-willi syndrome. J. Clin. Endocrinol. Metab. 2002, 87, 5461–5464. [Google Scholar] [CrossRef]
- Holsen, L.M.; Savage, C.R.; Martin, L.E.; Bruce, A.S.; Lepping, R.J.; Ko, E.; Brooks, W.M.; Butler, M.G.; Zarcone, J.R.; Goldstein, J.M. Importance of reward and prefrontal circuitry in hunger and satiety: Prader-Willi syndrome vs simple obesity. Int. J. Obes. 2012, 36, 638–647. [Google Scholar] [CrossRef]
- Miller, J.L.; Lacroix, A.; Bird, L.M.; Shoemaker, A.H.; Haqq, A.; Deal, C.L.; Clark, K.A.; Ames, M.H.; Suico, J.G.; de la Pena, A.; et al. The Efficacy, Safety, and Pharmacology of a Ghrelin O-Acyltransferase Inhibitor for the Treatment of Prader-Willi Syndrome. J. Clin. Endocrinol. Metab. 2022, 107, e2373–e2380. [Google Scholar] [CrossRef] [PubMed]
- Yanagi, S.; Sato, T.; Kangawa, K.; Nakazato, M. The Homeostatic Force of Ghrelin. Cell Metab. 2018, 27, 786–804. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, N.; Delanoue, R.; Mauri, A.; Basco, D.; Pasco, M.; Thorens, B.; Leopold, P. The Drosophila TNF Eiger Is an Adipokine that Acts on Insulin-Producing Cells to Mediate Nutrient Response. Cell Metab. 2016, 23, 675–684. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, G.; Maccio, A.; Lai, P.; Massa, E.; Ghiani, M.; Santona, M.C. Cytokine activity in cancer-related anorexia/cachexia: Role of megestrol acetate and medroxyprogesterone acetate. Semin. Oncol. 1998, 25, 45–52. [Google Scholar] [PubMed]
- Noguchi, Y.; Yoshikawa, T.; Matsumoto, A.; Svaninger, G.; Gelin, J. Are cytokines possible mediators of cancer cachexia? Surg. Today 1996, 26, 467–475. [Google Scholar] [CrossRef]
- Krause, A.; Sillard, R.; Kleemeier, B.; Kluver, E.; Maronde, E.; Conejo-Garcia, J.R.; Forssmann, W.G.; Schulz-Knappe, P.; Nehls, M.C.; Wattler, F.; et al. Isolation and biochemical characterization of LEAP-2, a novel blood peptide expressed in the liver. Protein Sci. 2003, 12, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.; Yang, H.; Bednarek, M.A.; Galon-Tilleman, H.; Chen, P.; Chen, M.; Lichtman, J.S.; Wang, Y.; Dalmas, O.; Yin, Y.; et al. LEAP2 Is an Endogenous Antagonist of the Ghrelin Receptor. Cell Metab. 2018, 27, 461–469. [Google Scholar] [CrossRef]
- Mani, B.K.; Puzziferri, N.; He, Z.; Rodriguez, J.A.; Osborne-Lawrence, S.; Metzger, N.P.; Chhina, N.; Gaylinn, B.; Thorner, M.O.; Thomas, E.L.; et al. LEAP2 changes with body mass and food intake in humans and mice. J. Clin. Investig. 2019, 129, 3909–3923. [Google Scholar] [CrossRef]
- Cummings, D.E.; Weigle, D.S.; Frayo, R.S.; Breen, P.A.; Ma, M.K.; Dellinger, E.P.; Purnell, J.Q. Plasma ghrelin levels after diet-induced weight loss or gastric bypass surgery. N. Engl. J. Med. 2002, 346, 1623–1630. [Google Scholar] [CrossRef]
- Tschop, M.; Weyer, C.; Tataranni, P.A.; Devanarayan, V.; Ravussin, E.; Heiman, M.L. Circulating ghrelin levels are decreased in human obesity. Diabetes 2001, 50, 707–709. [Google Scholar] [CrossRef]
- Shiiya, T.; Nakazato, M.; Mizuta, M.; Date, Y.; Mondal, M.S.; Tanaka, M.; Nozoe, S.; Hosoda, H.; Kangawa, K.; Matsukura, S. Plasma ghrelin levels in lean and obese humans and the effect of glucose on ghrelin secretion. J. Clin. Endocrinol. Metab. 2002, 87, 240–244. [Google Scholar] [CrossRef]
- Fittipaldi, A.S.; Hernandez, J.; Castrogiovanni, D.; Lufrano, D.; De Francesco, P.N.; Garrido, V.; Vitaux, P.; Fasano, M.V.; Fehrentz, J.A.; Fernandez, A.; et al. Plasma levels of ghrelin, des-acyl ghrelin and LEAP2 in children with obesity: Correlation with age and insulin resistance. Eur. J. Endocrinol. 2020, 182, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Barnett, B.P.; Hwang, Y.; Taylor, M.S.; Kirchner, H.; Pfluger, P.T.; Bernard, V.; Lin, Y.Y.; Bowers, E.M.; Mukherjee, C.; Song, W.J.; et al. Glucose and weight control in mice with a designed ghrelin O-acyltransferase inhibitor. Science 2010, 330, 1689–1692. [Google Scholar] [CrossRef] [PubMed]
- Teubner, B.J.; Garretson, J.T.; Hwang, Y.; Cole, P.A.; Bartness, T.J. Inhibition of ghrelin O-acyltransferase attenuates food deprivation-induced increases in ingestive behavior. Horm. Behav. 2013, 63, 667–673. [Google Scholar] [CrossRef] [PubMed]
- Morris, J.; Twaddle, S. Anorexia nervosa. BMJ 2007, 334, 894–898. [Google Scholar] [CrossRef] [PubMed]
- Zipfel, S.; Giel, K.E.; Bulik, C.M.; Hay, P.; Schmidt, U. Anorexia nervosa: Aetiology, assessment, and treatment. Lancet Psychiatry 2015, 2, 1099–1111. [Google Scholar] [CrossRef]
- Otto, B.; Cuntz, U.; Fruehauf, E.; Wawarta, R.; Folwaczny, C.; Riepl, R.L.; Heiman, M.L.; Lehnert, P.; Fichter, M.; Tschop, M. Weight gain decreases elevated plasma ghrelin concentrations of patients with anorexia nervosa. Eur. J. Endocrinol. 2001, 145, 669–673. [Google Scholar] [CrossRef] [PubMed]
- Legrand, R.; Lucas, N.; Breton, J.; Azhar, S.; Do, R.J.; Dechelotte, P.; Coeffier, M.; Fetissov, S.O. Ghrelin treatment prevents development of activity based anorexia in mice. Eur. Neuropsychopharmacol. 2016, 26, 948–958. [Google Scholar] [CrossRef] [PubMed]
- Miljic, D.; Pekic, S.; Djurovic, M.; Doknic, M.; Milic, N.; Casanueva, F.F.; Ghatei, M.; Popovic, V. Ghrelin has partial or no effect on appetite, growth hormone, prolactin, and cortisol release in patients with anorexia nervosa. J. Clin. Endocrinol. Metab. 2006, 91, 1491–1495. [Google Scholar] [CrossRef]
- Fazeli, P.K.; Lawson, E.A.; Faje, A.T.; Eddy, K.T.; Lee, H.; Fiedorek, F.T.; Breggia, A.; Gaal, I.M.; DeSanti, R.; Klibanski, A. Treatment With a Ghrelin Agonist in Outpatient Women With Anorexia Nervosa: A Randomized Clinical Trial. J. Clin. Psychiatry 2018, 79, 7823. [Google Scholar] [CrossRef]
- Broglio, F.; Gianotti, L.; Destefanis, S.; Fassino, S.; Abbate, D.G.; Mondelli, V.; Lanfranco, F.; Gottero, C.; Gauna, C.; Hofland, L.; et al. The endocrine response to acute ghrelin administration is blunted in patients with anorexia nervosa, a ghrelin hypersecretory state. Clin. Endocrinol. 2004, 60, 592–599. [Google Scholar] [CrossRef]
- Lambert, E.; Lambert, G.; Ika-Sari, C.; Dawood, T.; Lee, K.; Chopra, R.; Straznicky, N.; Eikelis, N.; Drew, S.; Tilbrook, A.; et al. Ghrelin modulates sympathetic nervous system activity and stress response in lean and overweight men. Hypertension 2011, 58, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Nagaya, N.; Miyatake, K.; Uematsu, M.; Oya, H.; Shimizu, W.; Hosoda, H.; Kojima, M.; Nakanishi, N.; Mori, H.; Kangawa, K. Hemodynamic, renal, and hormonal effects of ghrelin infusion in patients with chronic heart failure. J. Clin. Endocrinol. Metab. 2001, 86, 5854–5859. [Google Scholar] [CrossRef] [PubMed]
- Krapalis, A.F.; Reiter, J.; Machleidt, F.; Iwen, K.A.; Dodt, C.; Lehnert, H.; Sayk, F. Ghrelin modulates baroreflex-regulation of sympathetic vasomotor tone in healthy humans. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2012, 302, R1305–R1312. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nagaya, N.; Uematsu, M.; Kojima, M.; Date, Y.; Nakazato, M.; Okumura, H.; Hosoda, H.; Shimizu, W.; Yamagishi, M.; Oya, H.; et al. Elevated circulating level of ghrelin in cachexia associated with chronic heart failure: Relationships between ghrelin and anabolic/catabolic factors. Circulation 2001, 104, 2034–2038. [Google Scholar] [CrossRef] [PubMed]
- Nagaya, N.; Uematsu, M.; Kojima, M.; Ikeda, Y.; Yoshihara, F.; Shimizu, W.; Hosoda, H.; Hirota, Y.; Ishida, H.; Mori, H.; et al. Chronic administration of ghrelin improves left ventricular dysfunction and attenuates development of cardiac cachexia in rats with heart failure. Circulation 2001, 104, 1430–1435. [Google Scholar] [CrossRef]
- Sun, N.; Mei, Y.; Hu, Z.; Xing, W.; Lv, K.; Hu, N.; Zhang, T.; Wang, D. Ghrelin attenuates depressive-like behavior, heart failure, and neuroinflammation in postmyocardial infarction rat model. Eur. J. Pharmacol. 2021, 901, 174096. [Google Scholar] [CrossRef]
- Torres-Fuentes, C.; Pastor-Cavada, E.; Cano, R.; Kandil, D.; Shanahan, R.; Juan, R.; Shaban, H.; McGlacken, G.P.; Schellekens, H. Quinolones Modulate Ghrelin Receptor Signaling: Potential for a Novel Small Molecule Scaffold in the Treatment of Cachexia. Int. J. Mol. Sci. 2018, 19, 1605. [Google Scholar] [CrossRef]
- Chang, L.; Ren, Y.; Liu, X.; Li, W.G.; Yang, J.; Geng, B.; Weintraub, N.L.; Tang, C. Protective effects of ghrelin on ischemia/reperfusion injury in the isolated rat heart. J. Cardiovasc. Pharmacol. 2004, 43, 165–170. [Google Scholar] [CrossRef]
- Locatelli, V.; Rossoni, G.; Schweiger, F.; Torsello, A.; De Gennaro, C.V.; Bernareggi, M.; Deghenghi, R.; Muller, E.E.; Berti, F. Growth hormone-independent cardioprotective effects of hexarelin in the rat. Endocrinology 1999, 140, 4024–4031. [Google Scholar] [CrossRef]
- Pei, X.M.; Yung, B.Y.; Yip, S.P.; Ying, M.; Benzie, I.F.; Siu, P.M. Desacyl ghrelin prevents doxorubicin-induced myocardial fibrosis and apoptosis via the GHSR-independent pathway. Am. J. Physiol. Endocrinol. Metab. 2014, 306, E311–E323. [Google Scholar] [CrossRef]
- Baldanzi, G.; Filigheddu, N.; Cutrupi, S.; Catapano, F.; Bonissoni, S.; Fubini, A.; Malan, D.; Baj, G.; Granata, R.; Broglio, F.; et al. Ghrelin and des-acyl ghrelin inhibit cell death in cardiomyocytes and endothelial cells through ERK1/2 and PI 3-kinase/AKT. J. Cell Biol. 2002, 159, 1029–1037. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Lin, P.; Li, P.; Feng, L.; Ren, Q.; Xie, X.; Xu, J. Ghrelin protects the heart against ischemia/reperfusion injury via inhibition of TLR4/NLRP3 inflammasome pathway. Life Sci. 2017, 186, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Huynh, D.N.; Elimam, H.; Bessi, V.L.; Menard, L.; Burelle, Y.; Granata, R.; Carpentier, A.C.; Ong, H.; Marleau, S. A Linear Fragment of Unacylated Ghrelin (UAG6–13) Protects Against Myocardial Ischemia/Reperfusion Injury in Mice in a Growth Hormone Secretagogue Receptor-Independent Manner. Front. Endocrinol. 2018, 9, 798. [Google Scholar] [CrossRef]
- Harisseh, R.; Pillot, B.; Gharib, A.; Augeul, L.; Gallo-Bona, N.; Ferrera, R.; Loufouat, J.; Delale, T.; Allas, S.; Abribat, T.; et al. Unacylated ghrelin analog prevents myocardial reperfusion injury independently of permeability transition pore. Basic Res. Cardiol. 2017, 112, 4. [Google Scholar] [CrossRef]
- Hausenloy, D.J.; Yellon, D.M. New directions for protecting the heart against ischaemia-reperfusion injury: Targeting the Reperfusion Injury Salvage Kinase (RISK)-pathway. Cardiovasc. Res. 2004, 61, 448–460. [Google Scholar] [CrossRef]
- Hung, C.M.; Garcia-Haro, L.; Sparks, C.A.; Guertin, D.A. mTOR-dependent cell survival mechanisms. Cold Spring Harb. Perspect. Biol. 2012, 4, a008771. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Liu, Y.; Gui, Q.; Zhu, X.; Zeng, L.; Meng, J.; Qing, J.; Gao, L.; Jackson, A.O.; Feng, J.; et al. Ghrelin attenuates myocardial fibrosis after acute myocardial infarction via inhibiting endothelial-to mesenchymal transition in rat model. Peptides 2019, 111, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Eid, R.A.; Alkhateeb, M.A.; Al-Shraim, M.; Eleawa, S.M.; Shatoor, A.S.; El-Kott, A.F.; Zaki, M.; Shatoor, K.A.; Bin-Jaliah, I.; Al-Hashem, F.H. Ghrelin prevents cardiac cell apoptosis during cardiac remodelling post experimentally induced myocardial infarction in rats via activation of Raf-MEK1/2-ERK1/2 signalling. Arch. Physiol. Biochem. 2019, 125, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Kerendi, F.; Kin, H.; Halkos, M.E.; Jiang, R.; Zatta, A.J.; Zhao, Z.Q.; Guyton, R.A.; Vinten-Johansen, J. Remote postconditioning. Brief renal ischemia and reperfusion applied before coronary artery reperfusion reduces myocardial infarct size via endogenous activation of adenosine receptors. Basic Res. Cardiol. 2005, 100, 404–412. [Google Scholar] [CrossRef]
- Kharbanda, R.K.; Mortensen, U.M.; White, P.A.; Kristiansen, S.B.; Schmidt, M.R.; Hoschtitzky, J.A.; Vogel, M.; Sorensen, K.; Redington, A.N.; MacAllister, R. Transient limb ischemia induces remote ischemic preconditioning in vivo. Circulation 2002, 106, 2881–2883. [Google Scholar] [CrossRef]
- Eid, R.A.; Alkhateeb, M.A.; Eleawa, S.; Al-Hashem, F.H.; Al-Shraim, M.; El-Kott, A.F.; Zaki, M.; Dallak, M.A.; Aldera, H. Cardioprotective effect of ghrelin against myocardial infarction-induced left ventricular injury via inhibition of SOCS3 and activation of JAK2/STAT3 signaling. Basic Res. Cardiol. 2018, 113, 13. [Google Scholar] [CrossRef] [PubMed]
- Hausenloy, D.J.; Iliodromitis, E.K.; Andreadou, I.; Papalois, A.; Gritsopoulos, G.; Anastasiou-Nana, M.; Kremastinos, D.T.; Yellon, D.M. Investigating the signal transduction pathways underlying remote ischemic conditioning in the porcine heart. Cardiovasc. Drugs Ther. 2012, 26, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Sawashita, Y.; Hirata, N.; Yoshikawa, Y.; Terada, H.; Tokinaga, Y.; Yamakage, M. Remote ischemic preconditioning reduces myocardial ischemia-reperfusion injury through unacylated ghrelin-induced activation of the JAK/STAT pathway. Basic Res. Cardiol. 2020, 115, 50. [Google Scholar] [CrossRef] [PubMed]
- Hill, M.F.; Singal, P.K. Right and left myocardial antioxidant responses during heart failure subsequent to myocardial infarction. Circulation 1997, 96, 2414–2420. [Google Scholar] [CrossRef]
- Liu, X.; Gai, Y.; Liu, F.; Gao, W.; Zhang, Y.; Xu, M.; Li, Z. Trimetazidine inhibits pressure overload-induced cardiac fibrosis through NADPH oxidase-ROS-CTGF pathway. Cardiovasc. Res. 2010, 88, 150–158. [Google Scholar] [CrossRef]
- Wang, Q.; Liu, A.D.; Li, T.S.; Tang, Q.; Wang, X.C.; Chen, X.B. Ghrelin ameliorates cardiac fibrosis after myocardial infarction by regulating the Nrf2/NADPH/ROS pathway. Peptides 2021, 144, 170613. [Google Scholar] [CrossRef]
- Hu, Z.; Zhang, T.; Mei, Y.; Sun, N.; Lv, K.; Wang, D. Impact of Ghrelin on Ventricular Arrhythmia and Related Mechanism After Myocardial Infarction. Pharmacology 2022, 107, 102–110. [Google Scholar] [CrossRef]
- Banks, W.A.; Tschop, M.; Robinson, S.M.; Heiman, M.L. Extent and direction of ghrelin transport across the blood-brain barrier is determined by its unique primary structure. J. Pharmacol. Exp. Ther. 2002, 302, 822–827. [Google Scholar] [CrossRef]
- Huang, H.J.; Zhu, X.C.; Han, Q.Q.; Wang, Y.L.; Yue, N.; Wang, J.; Yu, R.; Li, B.; Wu, G.C.; Liu, Q.; et al. Ghrelin alleviates anxiety- and depression-like behaviors induced by chronic unpredictable mild stress in rodents. Behav. Brain Res. 2017, 326, 33–43. [Google Scholar] [CrossRef]
- Bouillon-Minois, J.B.; Trousselard, M.; Thivel, D.; Gordon, B.A.; Schmidt, J.; Moustafa, F.; Oris, C.; Dutheil, F. Ghrelin as a Biomarker of Stress: A Systematic Review and Meta-Analysis. Nutrients 2021, 13, 784. [Google Scholar] [CrossRef]
- Harmatz, E.S.; Stone, L.; Lim, S.H.; Lee, G.; McGrath, A.; Gisabella, B.; Peng, X.; Kosoy, E.; Yao, J.; Liu, E.; et al. Central Ghrelin Resistance Permits the Overconsolidation of Fear Memory. Biol. Psychiatry 2017, 81, 1003–1013. [Google Scholar] [CrossRef]
- Lu, Y.; Niu, M.; Qiu, X.; Cao, H.; Xing, B.; Sun, Y.; Zhou, Z.; Zhou, Y. Acute But Not Chronic Calorie Restriction Defends against Stress-Related Anxiety and Despair in a GHS-R1a-Dependent Manner. Neuroscience 2019, 412, 94–104. [Google Scholar] [CrossRef]
- Zhang, F.; Xu, F.; Mi, X.; Dong, L.; Xiao, Y.; Jiang, S.; Li, G.D.; Zhou, Y. Ghrelin/GHS-R1a signaling plays different roles in anxiety-related behaviors after acute and chronic caloric restriction. Biochem. Biophys. Res. Commun. 2020, 529, 1131–1136. [Google Scholar] [CrossRef]
- Yousufzai, M.; Harmatz, E.S.; Shah, M.; Malik, M.O.; Goosens, K.A. Ghrelin is a persistent biomarker for chronic stress exposure in adolescent rats and humans. Transl. Psychiatry 2018, 8, 74. [Google Scholar] [CrossRef]
- Meyer, R.M.; Burgos-Robles, A.; Liu, E.; Correia, S.S.; Goosens, K.A. A ghrelin-growth hormone axis drives stress-induced vulnerability to enhanced fear. Mol. Psychiatry 2014, 19, 1284–1294. [Google Scholar] [CrossRef] [PubMed]
- Mitra, R.; Sapolsky, R.M. Acute corticosterone treatment is sufficient to induce anxiety and amygdaloid dendritic hypertrophy. Proc. Natl. Acad. Sci. USA 2008, 105, 5573–5578. [Google Scholar] [CrossRef]
- Zigman, J.M.; Jones, J.E.; Lee, C.E.; Saper, C.B.; Elmquist, J.K. Expression of ghrelin receptor mRNA in the rat and the mouse brain. J. Comp. Neurol. 2006, 494, 528–548. [Google Scholar] [CrossRef] [PubMed]
- Duan, K.; Gu, Q.; Petralia, R.S.; Wang, Y.X.; Panja, D.; Liu, X.; Lehmann, M.L.; Zhu, H.; Zhu, J.; Li, Z. Mitophagy in the basolateral amygdala mediates increased anxiety induced by aversive social experience. Neuron 2021, 109, 3793–3809. [Google Scholar] [CrossRef] [PubMed]
- Anacker, C.; Luna, V.M.; Stevens, G.S.; Millette, A.; Shores, R.; Jimenez, J.C.; Chen, B.; Hen, R. Hippocampal neurogenesis confers stress resilience by inhibiting the ventral dentate gyrus. Nature 2018, 559, 98–102. [Google Scholar] [CrossRef]
- Huang, H.J.; Chen, X.R.; Han, Q.Q.; Wang, J.; Pilot, A.; Yu, R.; Liu, Q.; Li, B.; Wu, G.C.; Wang, Y.Q.; et al. The protective effects of Ghrelin/GHSR on hippocampal neurogenesis in CUMS mice. Neuropharmacology 2019, 155, 31–43. [Google Scholar] [CrossRef] [PubMed]
- Esteban-Cornejo, I.; Stillman, C.M.; Rodriguez-Ayllon, M.; Kramer, A.F.; Hillman, C.H.; Catena, A.; Erickson, K.I.; Ortega, F.B. Physical fitness, hippocampal functional connectivity and academic performance in children with overweight/obesity: The ActiveBrains project. Brain Behav. Immun. 2021, 91, 284–295. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.Q.; Wang, Y.; Hu, K.; Lin, S.; Lin, X.H. Hippocampal Glycerol-3-Phosphate Acyltransferases 4 and BDNF in the Progress of Obesity-Induced Depression. Front. Endocrinol. 2021, 12, 667773. [Google Scholar] [CrossRef] [PubMed]
- Pierre, A.; Regin, Y.; Van Schuerbeek, A.; Fritz, E.M.; Muylle, K.; Beckers, T.; Smolders, I.J.; Singewald, N.; De Bundel, D. Effects of disrupted ghrelin receptor function on fear processing, anxiety and saccharin preference in mice. Psychoneuroendocrino 2019, 110, 104430. [Google Scholar] [CrossRef]
- Suzuki, H.; Asakawa, A.; Amitani, H.; Nakamura, N.; Inui, A. Cancer cachexia--pathophysiology and management. J. Gastroenterol. 2013, 48, 574–594. [Google Scholar] [CrossRef]
- Dixit, V.D.; Schaffer, E.M.; Pyle, R.S.; Collins, G.D.; Sakthivel, S.K.; Palaniappan, R.; Lillard, J.J.; Taub, D.D. Ghrelin inhibits leptin- and activation-induced proinflammatory cytokine expression by human monocytes and T cells. J. Clin. Investig. 2004, 114, 57–66. [Google Scholar] [CrossRef]
- Pritchett, N.R.; Maziarz, M.; Shu, X.O.; Kamangar, F.; Dawsey, S.M.; Fan, J.H.; Ji, B.T.; Gao, Y.T.; Xiang, Y.B.; Qiao, Y.L.; et al. Serum ghrelin and esophageal and gastric cancer in two cohorts in China. Int. J. Cancer 2020, 146, 2728–2735. [Google Scholar] [CrossRef] [PubMed]
- Blum, D.; de Wolf-Linder, S.; Oberholzer, R.; Brandle, M.; Hundsberger, T.; Strasser, F. Natural ghrelin in advanced cancer patients with cachexia, a case series. J. Cachexia Sarcopenia Muscle 2021, 12, 506–516. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, N.; Miki, K.; Tsubouchi, H.; Sakamoto, A.; Arimura, Y.; Yanagi, S.; Iiboshi, H.; Yoshida, M.; Souma, R.; Ishimoto, H.; et al. Ghrelin administration for chronic respiratory failure: A randomized dose-comparison trial. Lung 2015, 193, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Strasser, F.; Lutz, T.A.; Maeder, M.T.; Thuerlimann, B.; Bueche, D.; Tschop, M.; Kaufmann, K.; Holst, B.; Brandle, M.; von Moos, R.; et al. Safety, tolerability and pharmacokinetics of intravenous ghrelin for cancer-related anorexia/cachexia: A randomised, placebo-controlled, double-blind, double-crossover study. Br. J. Cancer 2008, 98, 300–308. [Google Scholar] [CrossRef]
- Grundmann, O.; Yoon, S.L.; Williams, J.J.; Gordan, L.; George, T.J. Augmentation of Cancer Cachexia Components With Targeted Acupuncture in Patients With Gastrointestinal Cancers: A Randomized Controlled Pilot Study. Integr. Cancer Ther. 2019, 18, 1870041701. [Google Scholar] [CrossRef]
- Morozumi, N.; Hanada, T.; Habara, H.; Yamaki, A.; Furuya, M.; Nakatsuka, T.; Inomata, N.; Minamitake, Y.; Ohsuye, K.; Kangawa, K. The role of C-terminal part of ghrelin in pharmacokinetic profile and biological activity in rats. Peptides 2011, 32, 1001–1007. [Google Scholar] [CrossRef] [PubMed]
- Hamauchi, S.; Furuse, J.; Takano, T.; Munemoto, Y.; Furuya, K.; Baba, H.; Takeuchi, M.; Choda, Y.; Higashiguchi, T.; Naito, T.; et al. A multicenter, open-label, single-arm study of anamorelin (ONO-7643) in advanced gastrointestinal cancer patients with cancer cachexia. Cancer-Am. Cancer Soc. 2019, 125, 4294–4302. [Google Scholar] [CrossRef] [PubMed]
- Katakami, N.; Uchino, J.; Yokoyama, T.; Naito, T.; Kondo, M.; Yamada, K.; Kitajima, H.; Yoshimori, K.; Sato, K.; Saito, H.; et al. Anamorelin (ONO-7643) for the treatment of patients with non-small cell lung cancer and cachexia: Results from a randomized, double-blind, placebo-controlled, multicenter study of Japanese patients (ONO-7643–04). Cancer-Am. Cancer Soc. 2018, 124, 606–616. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, H.; Arai, H.; Inui, A. The regulatory approval of anamorelin for treatment of cachexia in patients with non-small cell lung cancer, gastric cancer, pancreatic cancer, and colorectal cancer in Japan: Facts and numbers. J. Cachexia Sarcopenia Muscle 2021, 12, 14–16. [Google Scholar] [CrossRef] [PubMed]
- Miyake, M.; Hori, S.; Itami, Y.; Oda, Y.; Owari, T.; Fujii, T.; Ohnishi, S.; Morizawa, Y.; Gotoh, D.; Nakai, Y.; et al. Supplementary Oral Anamorelin Mitigates Anorexia and Skeletal Muscle Atrophy Induced by Gemcitabine Plus Cisplatin Systemic Chemotherapy in a Mouse Model. Cancers 2020, 12, 1942. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiao, Z.-T.; Luo, Q. Molecular Mechanisms and Health Benefits of Ghrelin: A Narrative Review. Nutrients 2022, 14, 4191. https://doi.org/10.3390/nu14194191
Jiao Z-T, Luo Q. Molecular Mechanisms and Health Benefits of Ghrelin: A Narrative Review. Nutrients. 2022; 14(19):4191. https://doi.org/10.3390/nu14194191
Chicago/Turabian StyleJiao, Zheng-Tong, and Qi Luo. 2022. "Molecular Mechanisms and Health Benefits of Ghrelin: A Narrative Review" Nutrients 14, no. 19: 4191. https://doi.org/10.3390/nu14194191
APA StyleJiao, Z.-T., & Luo, Q. (2022). Molecular Mechanisms and Health Benefits of Ghrelin: A Narrative Review. Nutrients, 14(19), 4191. https://doi.org/10.3390/nu14194191





