“Doctor, Can I Drink an Alcohol-Free Beer?” Low-Alcohol and Alcohol-Free Drinks in People with Heavy Drinking or Alcohol Use Disorders: Systematic Review of the Literature
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Selection Criteria
2.3. Data Extraction
3. Results
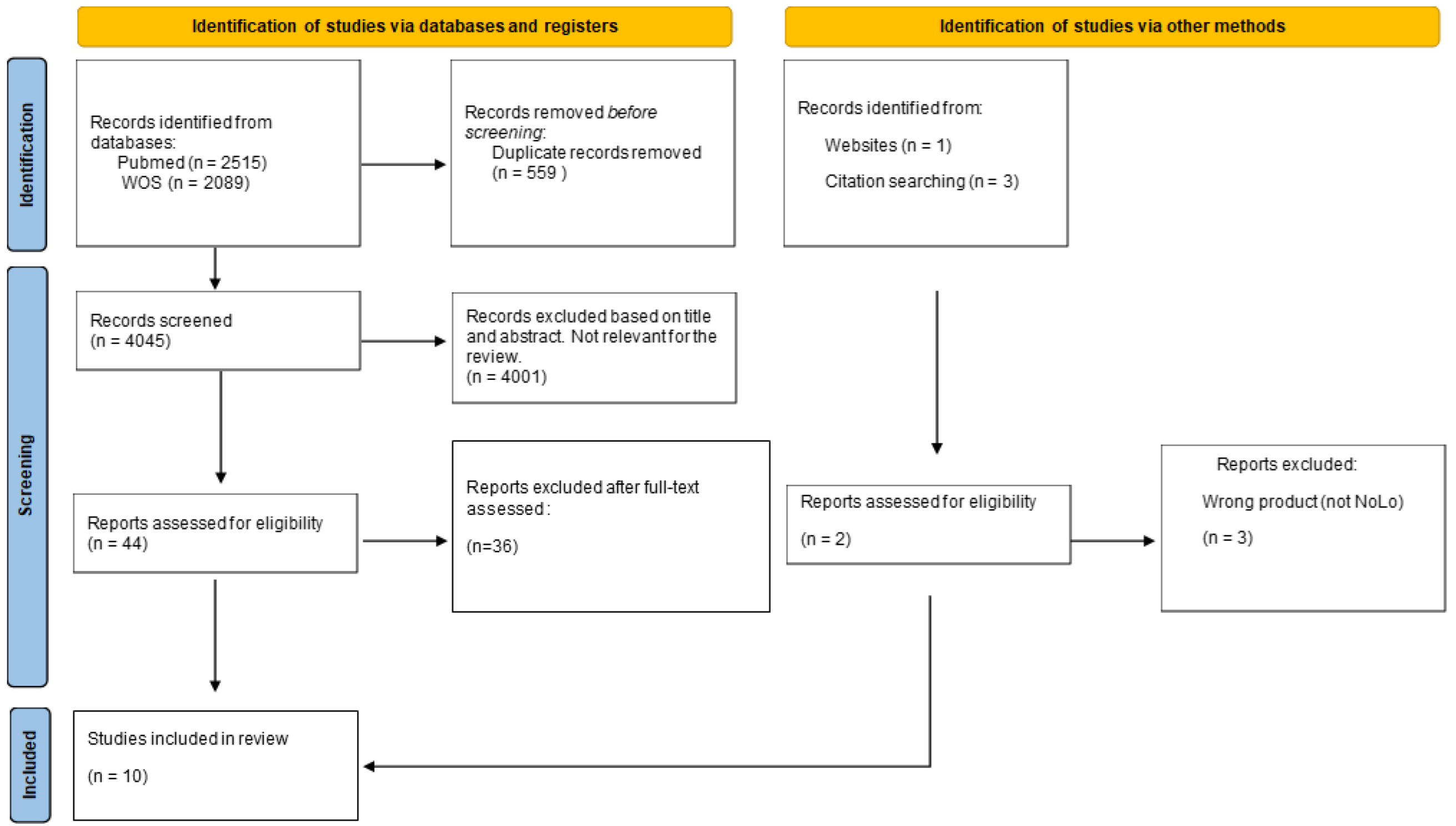
3.1. Clinical and Experimental Studies
3.1.1. Attitudes towards NoLo Drinks and Craving Following Their Use
3.1.2. Autonomic Reactivity to Alcohol-Related Cues
3.2. Epidemiological Studies
3.3. Product Description/Labelling Studies
4. Discussion
4.1. Clinical/Experimental Studies
4.2. Epidemiological Studies
4.3. Product Description/Labelling Studies
4.4. What Advice Can Clinicians Offer Their Patients with AUD, Heavy, or High-Risk Drinking, Regarding NoLo Products, Based on the Current Scientific Evidence?
4.5. Limitations
4.6. Gaps and Future Research
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Murray, C.J.L.; Aravkin, A.Y.; Zheng, P.; Abbafati, C.; Abbas, K.M.; Abbasi-Kangevari, M.; Abd-Allah, F.; Abdelalim, A.; Abdollahi, M.; Abdollahpour, I.; et al. Global burden of 87 risk factors in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1223–1249. [Google Scholar] [CrossRef]
- Roerecke, M.; Gual, A.; Rehm, J. Reduction of alcohol consumption and subsequent mortality in alcohol use disorders: Systematic review and meta-analyses. J. Clin. Psychiatry 2013, 74, e1181–e1189. [Google Scholar] [CrossRef]
- European Medicines Agency Guideline on the Development of Medicinal Products for the Treatment of Alcohol Dependence. 2010. Available online: http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2010/03/WC500074898.pdf. (accessed on 17 August 2022).
- François, C.; Laramée, P.; Rahhali, N.; Chalem, Y.; Aballéa, S.; Millier, A.; Bineau, S.; Toumi, M.; Rehm, J. A Predictive Microsimulation Model to Estimate the Clinical Relevance of Reducing Alcohol Consumption in Alcohol Dependence. Eur. Addict. Res. 2014, 20, 269–284. [Google Scholar] [CrossRef]
- Rehm, J.; Guiraud, J.; Poulnais, R.; Shield, K.D. Alcohol dependence and very high risk level of alcohol consumption: A life-threatening and debilitating disease. Addict. Biol. 2018, 23, 961–968. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization Global Strategy to Reduce the Harmful Use of Alcohol. 2010. Available online: https://www.who.int/publications/i/item/9789241599931 (accessed on 17 August 2022).
- Corfe, S.; Hyde, R.; Shepherd, J. Alcohol-Free and Low-Strength Drinks Understanding Their Role in Reducing Alcohol-Related Harms; Social Market Foundation: London, UK, 2020. [Google Scholar]
- Rehm, J.; Lachenmeier, D.W.; Llopis, E.J.; Imtiaz, S.; Anderson, P. Evidence of reducing ethanol content in beverages to reduce harmful use of alcohol. Lancet Gastroenterol. Hepatol. 2016, 1, 78–83. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group, T.P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Long, C.G.; Cohen, E.M. Low alcohol beers and wines: Attitudes of problem drinkers to their use and their effect on craving. Br. J. Addict. 1989, 84, 777–783. [Google Scholar] [CrossRef]
- Kaplan, R.F.; Meyer, R.E.; Stroebel, C.F. Alcohol Dependence and Responsivity to an Ethanol Stimulus as Predictors of Alcohol Consumption. Br. J. Addict. 1983, 78, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, R.F.; Meyer, R.E.; Virgilio, L.M. Physiological reactivity to alcohol cues and the awareness of an alcohol effect in a double-blind placebo design. Br. J. Addict. 1984, 79, 439–442. [Google Scholar] [CrossRef]
- Dolinsky, Z.S.; Morse, D.E.; Kaplan, R.F.; Meyer, R.E.; Corry, D.; Pomerleau, O.F. Neuroendocrine, psychophysiological and subjective reactivity to an alcohol placebo in male alcoholic patients. Alcohol Clin. Exp. Res. 1987, 11, 296–300. [Google Scholar] [CrossRef]
- Meyer, R.E.; Dolinsky, Z.S. Ethanol beverage anticipation: Effects on plasma testosterone and luteinizing hormone levels—A pilot study. J. Stud. Alcohol. 1990, 51, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Collins, R.L.; Gollnisch, G.; Izzo, C.V. Drinking restraint and alcohol-related outcomes: Exploring the contributions of beverage instructions, beverage content and self-monitoring. J. Stud. Alcohol 1996, 57, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Vasiljevic, M.; Couturier, D.L.; Marteau, T.M. Impact on product appeal of labeling wine and beer with (a) lower strength alcohol verbal descriptors and (b) percent alcohol by volume (%ABV): An experimental study. Psychol. Addict. Behav. 2018, 32, 779–791. [Google Scholar] [CrossRef] [PubMed]
- Vasiljevic, M.; Couturier, D.L.; Frings, D.; Moss, A.C.; Albery, I.P.; Marteau, T.M. Impact of Lower Strength Alcohol Labeling on Consumption: A Randomized Controlled Trial. Health Psychol. 2018, 37, 658. [Google Scholar] [CrossRef] [PubMed]
- Vasiljevic, M.; Frings, D.; Pilling, M.; Marteau, T.M. Do alcohol product labels stating lower strength verbal description, percentage alcohol-by-volume, or their combination affect wine consumption? A bar laboratory adaptive randomised controlled trial. Addiction 2021, 116, 2339–2347. [Google Scholar] [CrossRef]
- Oberlin, B.G.; Dzemidzic, M.; Tran, S.M.; Soeurt, C.M.; Albrecht, D.S.; Yoder, K.K.; Kareken, D.A. Beer flavor provokes striatal dopamine release in male drinkers: Mediation by family history of alcoholism. Neuropsychopharmacology 2013, 38, 1617–1624. [Google Scholar] [CrossRef]
- Oberlin, B.G.; Dzemidzic, M.; Tran, S.M.; Soeurt, C.M.; O’Connor, S.J.; Yoder, K.K.; Kareken, D.A. Beer self-administration provokes lateralized nucleus accumbens dopamine release in male heavy drinkers. Psychopharmacology 2015, 232, 861–870. [Google Scholar] [CrossRef]
- Kareken, D.A.; Claus, E.D.; Sabri, M.; Dzemidzic, M.; Kosobud, A.E.K.; Radnovich, A.J.; Hector, D.; Ramchandani, V.A.; O’Connor, S.J.; Lowe, M.; et al. Alcohol-related olfactory cues activate the nucleus accumbens and ventral tegmental area in high-risk drinkers: Preliminary findings. Alcohol Clin. Exp. Res. 2004, 28, 550–557. [Google Scholar] [CrossRef]
- Anderson, P.; O’donnell, A.; Kokole, D.; Llopis, E.J.; Kaner, E. Is Buying and Drinking Zero and Low Alcohol Beer a Higher Socio-Economic Phenomenon? Analysis of British Survey Data, 2015-2018 and Household Purchase Data 2015–2020. Int. J. Environ. Res. Public Health 2021, 18, 10347. [Google Scholar] [CrossRef]
- Vasiljevic, M.; Couturier, D.L.; Marteau, T.M. What are the perceived target groups and occasions for wines and beers labelled with verbal and numerical descriptors of lower alcohol strength? An experimental study. BMJ Open 2019, 9, e024412. [Google Scholar] [CrossRef]
- Jones, S.C.; Gregory, P. The impact of more visible standard drink labelling on youth alcohol consumption: Helping young people drink (ir)responsibly? Drug Alcohol Rev. 2009, 28, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Maynard, O.M.; Langfield, T.; Attwood, A.S.; Allen, E.; Drew, I.; Votier, A.; Munafò, M.R. No Impact of Calorie or Unit Information on Ad Libitum Alcohol Consumption. Alcohol Alcohol. 2018, 53, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Furtwängler, N.; de Visser, R.O. Motivation to adhere to unit-based guidelines for alcohol consumption and ability to do so is limited among university students. Drugs Educ. Prev. Policy 2017, 24, 418–425. [Google Scholar] [CrossRef]
- Jané Llopis, E.; O’Donnell, A.; Kaner, E.; Anderson, P. Are Lower-Strength Beers Gateways to Higher-Strength Beers? Time Series Analyses of Household Purchases from 64,280 British Households, 2015–2018. Alcohol Alcohol 2022, 57, 520–528. [Google Scholar] [CrossRef] [PubMed]
- Ter Meulen, R.H.J. The ethical basis of the precautionary principle in health care decision making. Toxicol. Appl. Pharmacol. 2005, 207, 663–667. [Google Scholar] [CrossRef] [PubMed]
- Tubiana, M. Le principe de précaution: Ses avantages et ses risques. J. Chir. 2001, 138, 68–80. [Google Scholar]
- Morphett, K.; Hall, W.; Gartner, C. The Misuse of the Precautionary Principle in Justifying Australia’s Ban on the Sale of Nicotine Vaping Products. Nicotine Tob. Res. 2021, 23, 14–20. [Google Scholar] [CrossRef] [PubMed]
| Study | Main Conclusions |
|---|---|
| Clinical and Experimental | |
| Craving [10] |
|
| Autonomic reactivity to alcohol-related cues [11,12] |
|
| Neuroendocrine activity [13,14] |
|
| Drinking restraint (particularly temptation to drink) [15] |
|
| Epidemiological Studies | |
| [7] |
|
| Product Description/Labelling Studies | |
| Product labeling [16,17,18] |
|
| Author and Year | Study Design | Population | Intervention | Comparator | Outcome | Results |
|---|---|---|---|---|---|---|
| Long et al., 1989 [10] | Cross-over | 31 inpatients and 67 former patients who had completed at least 1 week of a 5-week therapy program covering detoxification, education, skill training and relapse prevention strategies. | Educational group session in which the advantages and disadvantages of using low-alcohol drinks were discussed and in which they were offered to taste several drinks. | Soft drinks were offered after an educational session on problem drinking, to control the effects of convivial drinking. | Craving: at baseline (8 assessments during the weeks before and after the low-alcohol drink session), 1 h before, immediately before and immediately after the session Attitudes toward low-alcohol drinks: before and after the session. Use of the low-alcohol drinks to maintain post-treatment goals. | Craving was significantly raised over baseline (r = 3.3, d.f. = 30, p > 0.01) after the low-alcohol drinks session. No differences between subjects who had tested the low-alcohol drinks and those who had not (Chi square = 0.023, d.f. = 1, p >0.05). No increase in craving after the soft drink control session (t = 0.31, d.f= 19; p > 0.05). A significant correlation between severity of dependence and increase craving after sessions was found (Rho = 0.384, N = 20, p < 0.05). Attitudes toward low-alcohol drinks: INPATIENTS: 65% had used low alcohol drinks, and 19% of these have been using them daily, and 24% used them once a week. A total of 42% of the ones that used low drinks before felt that they helped to cope with the urge to drink alcohol. A total of 79% of the sample had a ‘favorable’ attitude toward low-alcohol drinks; 89% disagreed with the idea that low-alcohol drinks contributed to relapse. A total of 58% felt that low-alcohol drinks would be most acceptable alternatives to alcoholic drinks and were useful in helping them to join in at pubs and parties (47%). Of the inpatients, 55% would use the low-alcohol drinks after discharge; 50% would buy them in pubs and 42% at home. FORMER PATIENTS: A total of 47% had used low-alcohol drinks since leaving treatment (26% daily, 40% once/week). In all, 85% had a post-treatment goal of abstinence, 44% felt the use of low-alcohol drinks helped with this. Finally, 39% would recommend the use of these products to problem drinkers. |
| Kaplan et al., 1983 [11] | Double blind, placebo-controlled trial | 16 alcoholic patients (with a history of heavy drinking of at least 5 years) and 16 control subjects (social drinkers) | Participants were randomly assigned on a double-blind basis to either an ethanol or placebo condition. First, they do not drink it, only hold it while measurements are taken. Then, they are asked about the desire to drink, and their belief of whether the drink contained alcohol. Second, An identical second drink was presented and consumed Third, Subjects were instructed that they had an opportunity to work for a third drink or another reward. They must perform an operant task to obtain the reward, after which they must choose the drink or the other reward. The withdrawal symptoms and drinking behavior on the previous month is previously examined. | Placebo beer (non-alcoholic malt beverage) that participants believe contains alcohol. | To investigate the contributions of subclinical withdrawal symptomatology in the previous 30 days to psychophysiological arousal, desire to drink, and operant behavior associated with alcohol within the clinical laboratory. | Alcoholics showed a greater desire to drink than controls. There was also a significant correlation between autonomic arousal and desire to drink among alcoholics but not controls. There was some evidence that arousal was related to alcohol dependence among alcoholics. Placebo responding among alcoholics was also related to alcohol dependence. Desire to drink, withdrawal symptomatology, and heart rate accounted for over 57% of the variance in predicting which alcoholics would choose the drink reward following the operant task. |
| Kaplan et al., 1984 [12] | Double blind, placebo-controlled trial | 16 male alcoholic inpatients (with at least 5 years of alcohol dependence) undergoing alcohol treatment. | Skin conductance level is recorded during the presentation of a beer drink or placebo (randomly assigned) and subjects are asked if they thought they had just consumed an alcoholic drink. | Placebo beer (non-alcoholic malt beverage). | First, to describe the relationship between autonomic reactivity to an alcohol stimulus prior to the consumption, second, to describe the perceptions of an alcohol effect immediately following consumption of either real beer or placebo in alcoholic subjects. | SCL increases to the presentation of beer stimuli prior to consumption were highest among alcoholics who perceived the drink as ‘real beer’ following consumption. Perception of the drink as ‘real beer’ was not related to receiving real beer. |
| Vasiljevic et al., 2018 [17] | RCT | 264 (132 weekly wine drinkers and 132 weekly beer drinkers) | Group 1: label displaying the verbal descriptor Super Low + 4% ABV for wine/1% ABV for beer; Group 2: verbal descriptor Low + 8% ABV for wine/3% ABV for beer | Group 3: no verbal descriptor of strength + the average ABV of 12.9% for wine/4.2% for beer. | Primary: Total volume of drink consumed (in ml). Secondary: product appeal, understanding of alcohol strength, calorie content, guilt related to consumption. Other measures: risky drinking, motivation to reduce consumption, self-licensing. | ml of alcohol consumed increased as the label on the drink denoted successively lower alcohol strength (Lin = 0.71, SE = 0.30, p = 0.015, 95% CI [0.13, 1.30]) Group 1 drank more (M = 213.77, SD = 124.05) vs. Group 3 (M = 176.85, SD = 116.41), BD2 = 1.43, SE = 0.61, p = 0.019, 95% CI [0.24, 2.61]. No differences between Groups 2 and 3 (BD1 = 0.59, SE = 0.63, p = 0.340, 95% CI [−0.66, 1.80]). Risky drinkers drank more than non-risky drinkers, (BD8 = 2.46, SE = 0.72, p = 0.001, 95% CI [1.00, 3.83]). |
| Vasiljevic et al., 2018 [16] | 3 × 6 between-subjects, randomized study | 1697 wine drinkers (41% with a risky drinking pattern); 1693 beer drinkers (55.9% risky drinking pattern) | 18 groups with one of three levels of verbal descriptor (Low; Super Low; No verbal descriptor) and six levels of %ABV (five levels varying for wine and beer, and no level given. | Same as intervention. | Primary: product appeal. Secondary: Understanding of alcohol strength and calorie content. | Appeal decreased significantly as %ABV decreased with lowest appeal for wine with 0%ABV and 4%ABV, and for beer with 1%ABV and 2%ABV (p ≤ 0.001, for the comparison with Regular). Appeal for Low verbal descriptors was lowest when combined with No %ABV, and for Super Low appeal was lowest when combined with 0%ABV. Both Low and Super Low verbal descriptors had a similar detrimental impact on appeal (pswine < 0.001; psbeer < 0.002). Heavy drinking pattern did not affect the results. |
| Vasiljevic et al., 2021 [18] | RCT | 147 weekly wine drinkers | Group 1: verbal descriptor only (Super Low). Group 2: numerical descriptor only (4% ABV); and Group 3: verbal and numerical descriptors combined (Super Low 4%ABV). | Same as intervention. | Primary: Total volume of drink consumed (mL). Secondary: product appeal. | Participants randomized to the numerical descriptor label group (4%ABV: M = 155.12 mL, B = 20.30; 95% CI = 3.92, 36.69; p value = 0.016) and combined verbal and numerical descriptor label group (Super Low 4%ABV: M = 154.59 mL, B = 20.68; 95% CI = 4.32, 37.04; p value = 0.014) drank significantly greater amounts than those randomized to the verbal descriptor label group (Super Low: M = 125.65 mL). Self-reported appeal of the wine did not differ between the three groups (all Ps > 0.082). |
| Dolinsky et al., 1987 [13] | Experimental | EG: 8 male inpatients with alcohol dependence (AD) (DSM-III) hospitalized from 7–14 days of a 21-day alcohol rehabilitation program. 9 control subjects | Smell a real beer and then drink a “placebo” beer they believed contained alcohol. Outcomes were assessed at:
| Heart rate, skin conductance. Plasma insulin, glucagon, and cortisol levels. | Cortisol values were depressed in the EG and remained so throughout the study (group F(9,135) = 7.51, p = 0.05). The EG presented significantly larger and more rapid glucose and insulin responses than the CG following the consumption of the placebo beer, which they believed contained alcohol (group x time: glucose, F(9,135) = 2.28, p = 0.05; Insulin, F(9,135) = 3.5, p < 0.001). Both groups experienced an increase in desire to drink while smelling the real beer (time: F(3,45) = 5.14, p < 0.05). Heart rate was greater in the EG during the baseline assessments (B1:76 vs. 65; B2: 71 vs. 65, Group: F(1,14) = 5.45, p < 0.05). | |
| Meyer et al., 1990 [14] | Experimental | EG: 8 male inpatients with alcohol dependence (AD) (DSM-III) hospitalized from 7–14 days of a 21-day alcohol rehabilitation program. 9 control subjects | Smell a real beer and then drink a “placebo” beer they believed contained alcohol. | Primary: Changes in plasma concentrations o of testosterone and Luteinizing Hormone (LH). Secondary: changes in subjective reports of anxiety and alcohol craving. | EG presented a decrease in plasma testosterone during the drink presentation period and an increase relative to the control group during the post-drink period (Group x Time: testosterone F = 4.18, 9/126 df, p < 0.001). For LH: controls showed a decrease relative to EG (Group x Time: LH F = 3.83, 1/14 df, p < 0.1). In the EG, LH decrease during the pre-drink period (while holding the beer) (Group x Time: LH F = 4.66, 1/15 df, p < 0.05). No association between Testosterone levels and secondary outcomes (at the pre-drink nor post-drink period). | |
| Corfe et al., 2020 [7] | Report examining the “NoLo drinks” market in UK, based on desk research, consumer survey, and interviews with professionals. | The online survey, taken between the 29 May 2020 and the 4 June 2020, with two samples:
| Exploring the role that NoLo products can play in improving public health outcomes, considering alcohol harm. On the report disaggregated data on moderate and heavy drinkers is offered. | Moderate and heavy drinkers appear more likely than non-drinkers and light drinkers to consume NoLo drinks on specific occasions. (such as when driving), and on top of (rather than instead of) consumption of stronger drinks. This might limit the potential health benefits that could be realized from increased use of NoLo products. | ||
| Collins et al., 1996 [15] | RCT (Balanced placebo design) | 132 young males with a moderate to heavy drinking pattern. | 30 min taste-rating task (TRT): patients’ expectations of receiving an alcoholic or a non-alcoholic beer were crossed with the receipt of an alcoholic or a non-alcoholic beer. Four groups: ENA/RNA; ENA/RA; EA/RNA; EA/RA. Taste characteristics of the beer. | Effects of drinking restraint (temptation and restriction), beverage instructions and content and self-monitoring in alcohol-related outcomes (consumption, subjective intoxication and blood alcohol concentration (BAC)). | Consumption during a 30-min taste-rating task was positively related to the temptation to drink (i.e., difficulty controlling alcohol intake, drinking in response to negative emotions. There was also an interaction between restriction (an aspect of restraint) and expected beverage with high restriction subjects tending to drink more when they expected alcohol. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caballeria, E.; Pons-Cabrera, M.T.; Balcells-Oliveró, M.; Braddick, F.; Gordon, R.; Gual, A.; Matrai, S.; López-Pelayo, H. “Doctor, Can I Drink an Alcohol-Free Beer?” Low-Alcohol and Alcohol-Free Drinks in People with Heavy Drinking or Alcohol Use Disorders: Systematic Review of the Literature. Nutrients 2022, 14, 3925. https://doi.org/10.3390/nu14193925
Caballeria E, Pons-Cabrera MT, Balcells-Oliveró M, Braddick F, Gordon R, Gual A, Matrai S, López-Pelayo H. “Doctor, Can I Drink an Alcohol-Free Beer?” Low-Alcohol and Alcohol-Free Drinks in People with Heavy Drinking or Alcohol Use Disorders: Systematic Review of the Literature. Nutrients. 2022; 14(19):3925. https://doi.org/10.3390/nu14193925
Chicago/Turabian StyleCaballeria, Elsa, Maria Teresa Pons-Cabrera, Mercedes Balcells-Oliveró, Fleur Braddick, Rebecca Gordon, Antoni Gual, Silvia Matrai, and Hugo López-Pelayo. 2022. "“Doctor, Can I Drink an Alcohol-Free Beer?” Low-Alcohol and Alcohol-Free Drinks in People with Heavy Drinking or Alcohol Use Disorders: Systematic Review of the Literature" Nutrients 14, no. 19: 3925. https://doi.org/10.3390/nu14193925
APA StyleCaballeria, E., Pons-Cabrera, M. T., Balcells-Oliveró, M., Braddick, F., Gordon, R., Gual, A., Matrai, S., & López-Pelayo, H. (2022). “Doctor, Can I Drink an Alcohol-Free Beer?” Low-Alcohol and Alcohol-Free Drinks in People with Heavy Drinking or Alcohol Use Disorders: Systematic Review of the Literature. Nutrients, 14(19), 3925. https://doi.org/10.3390/nu14193925








