Effectiveness of Therapeutic Patient Education Interventions in Obesity and Diabetes: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Abstract
1. Introduction
- (a)
- Present a critical synthesis of the theoretical basis and development of TPE interventions for obesity and diabetes.
- (b)
- Present quantitative evidence for the efficacy of these interventions across a range of biomedical, psychosocial and psychological outcomes.
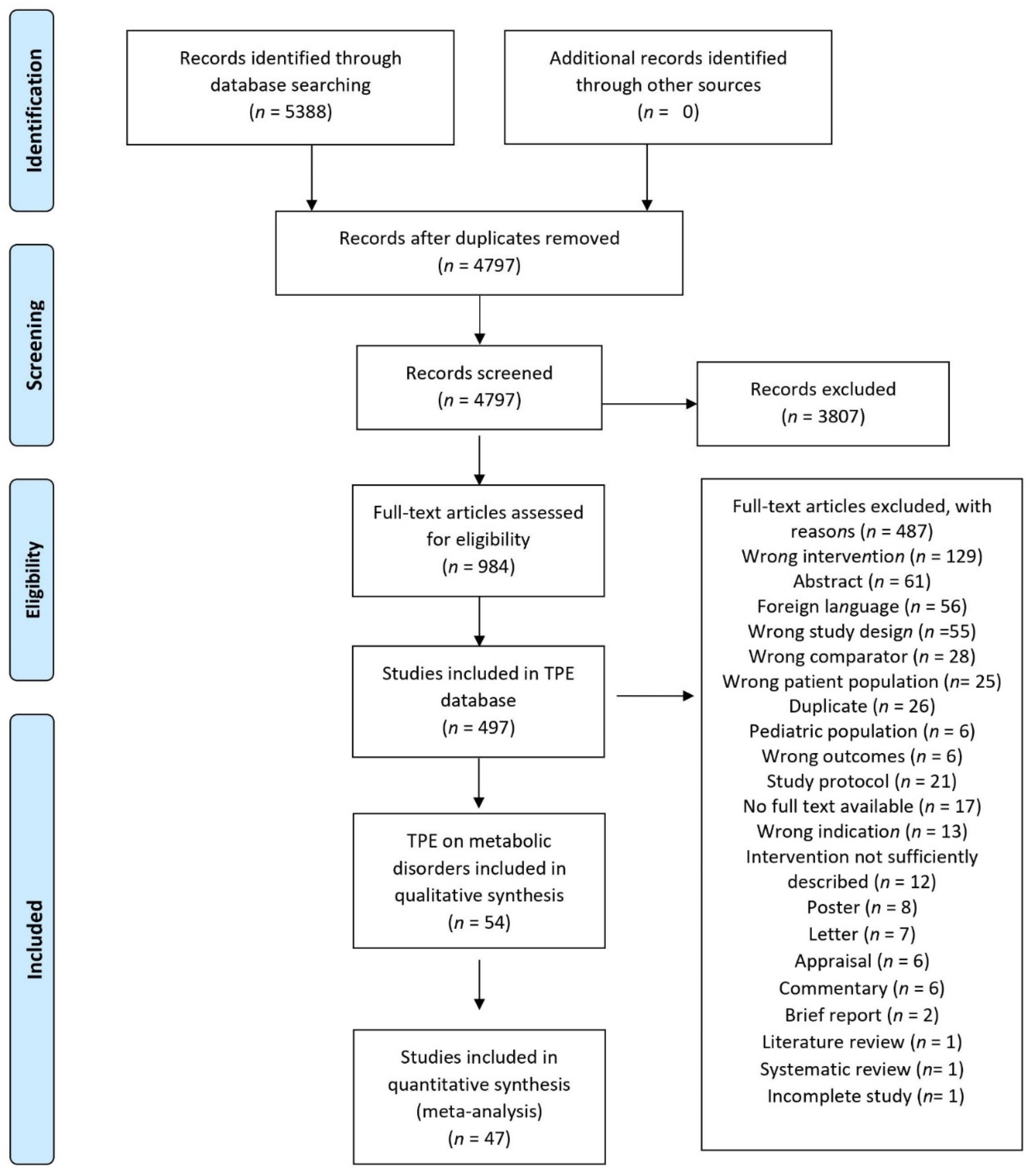
2. Materials and Methods
2.1. Information Sources and Search Strategy
2.2. Eligibility Criteria
2.3. Data Extraction
2.4. Meta-Analysis
2.5. Risk of Bias Assessment
3. Results
3.1. Characteristics of Interventions
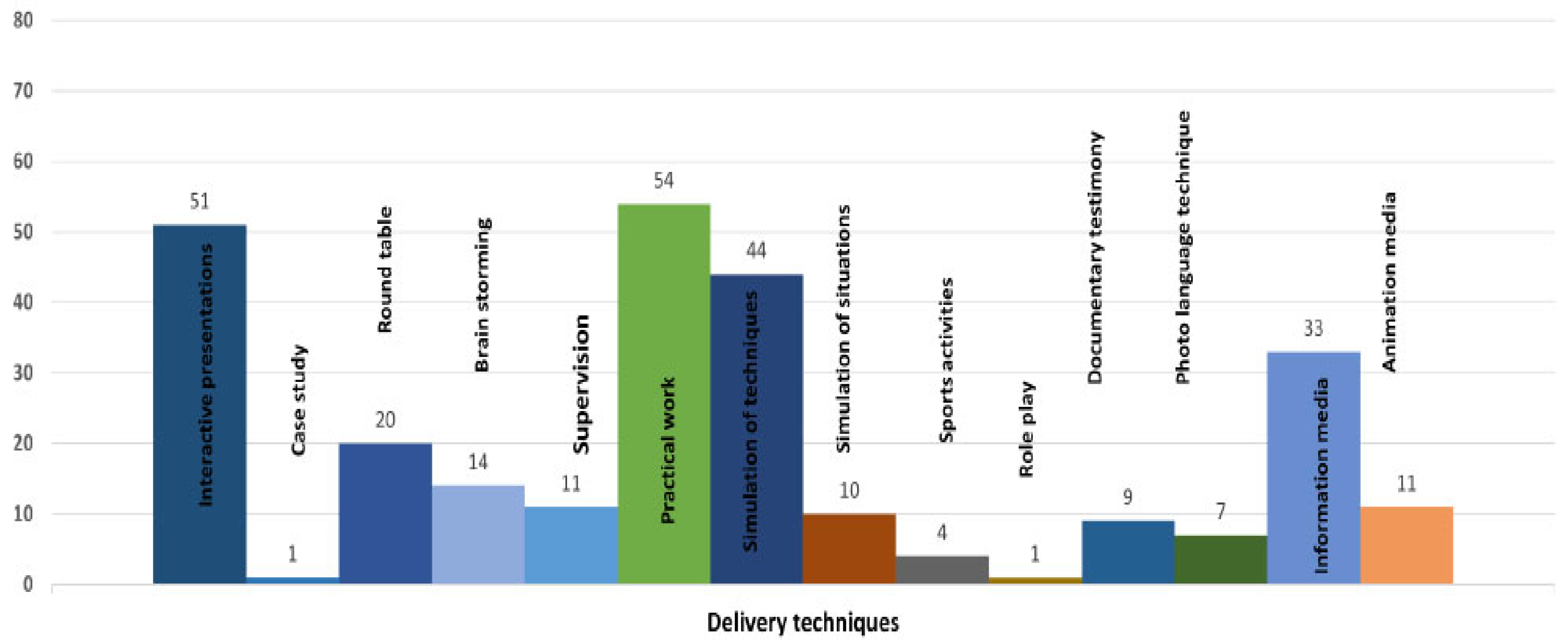
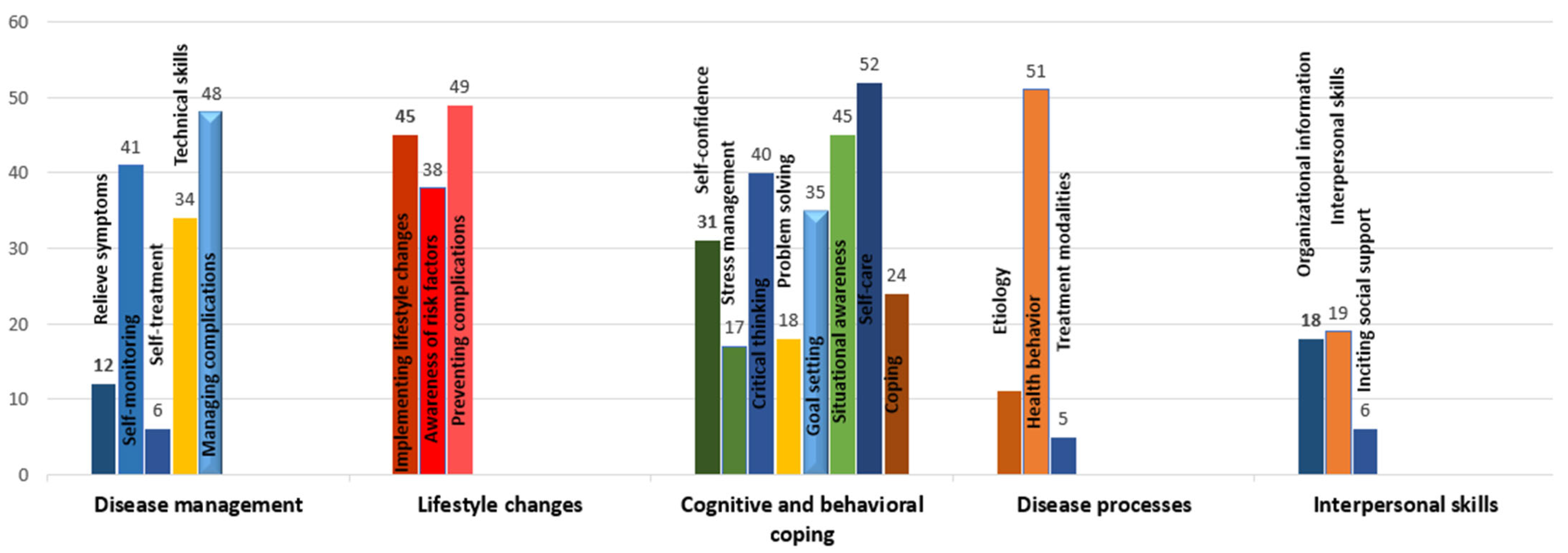
3.2. Ingredients of Interventions
3.3. Description of Interventions According to Delivery Format
3.3.1. Electronic Interventions
3.3.2. Group Interventions
3.3.3. Individually Delivered Interventions
3.3.4. Multimodal Interventions
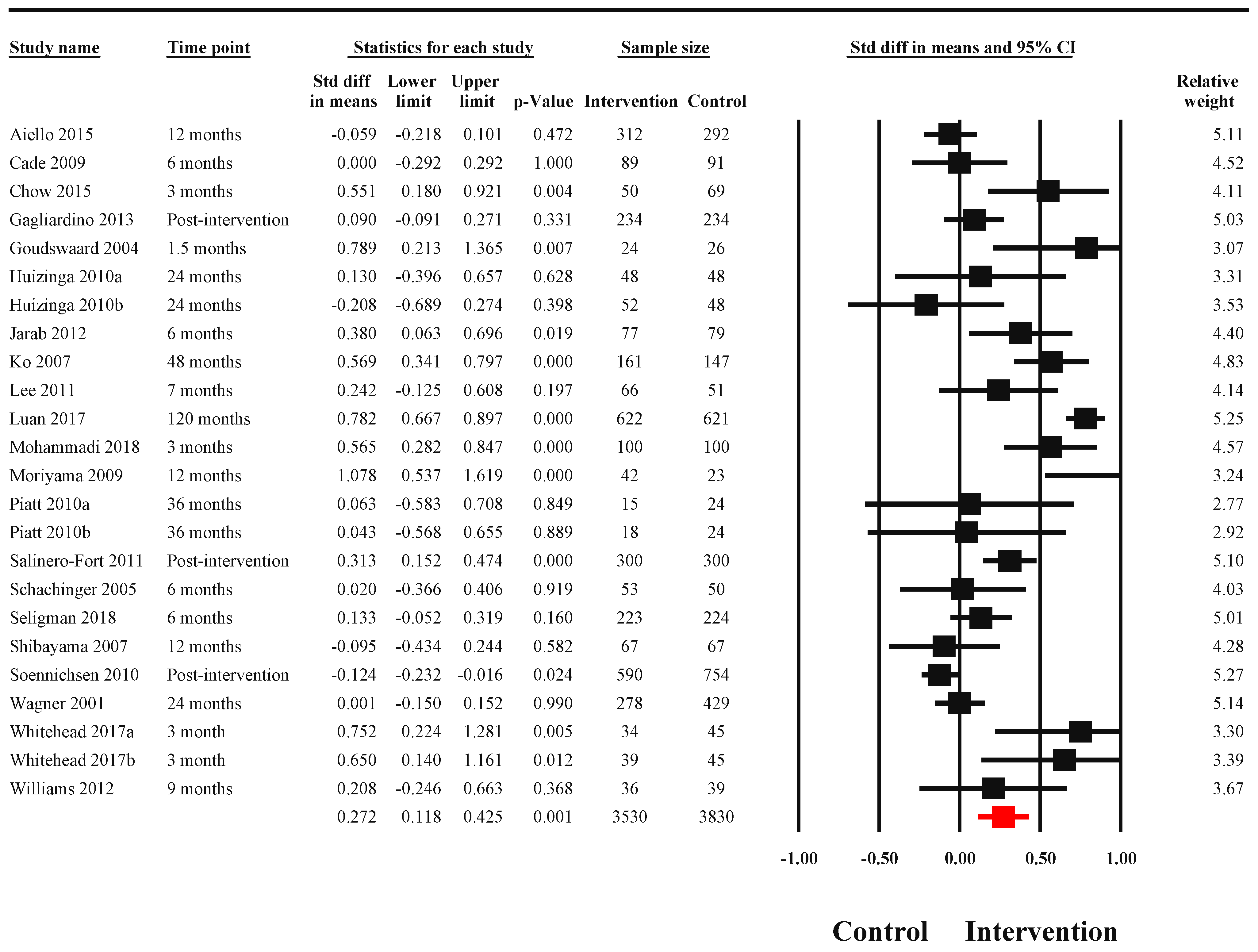
3.4. Efficacy of Interventions
3.4.1. Biological Outcomes
3.4.2. Adherence
3.4.3. Knowledge
3.4.4. Quality of Life
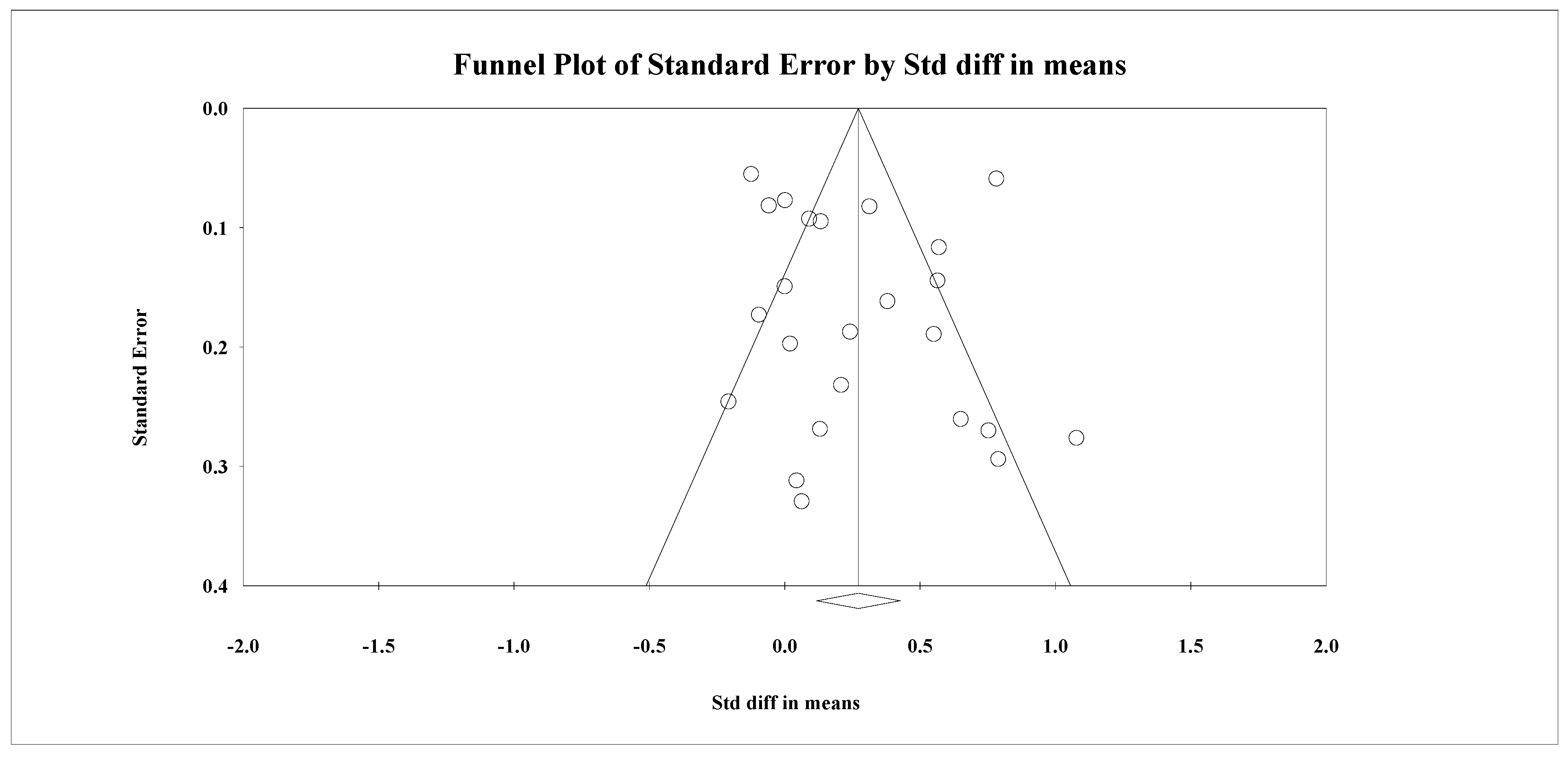
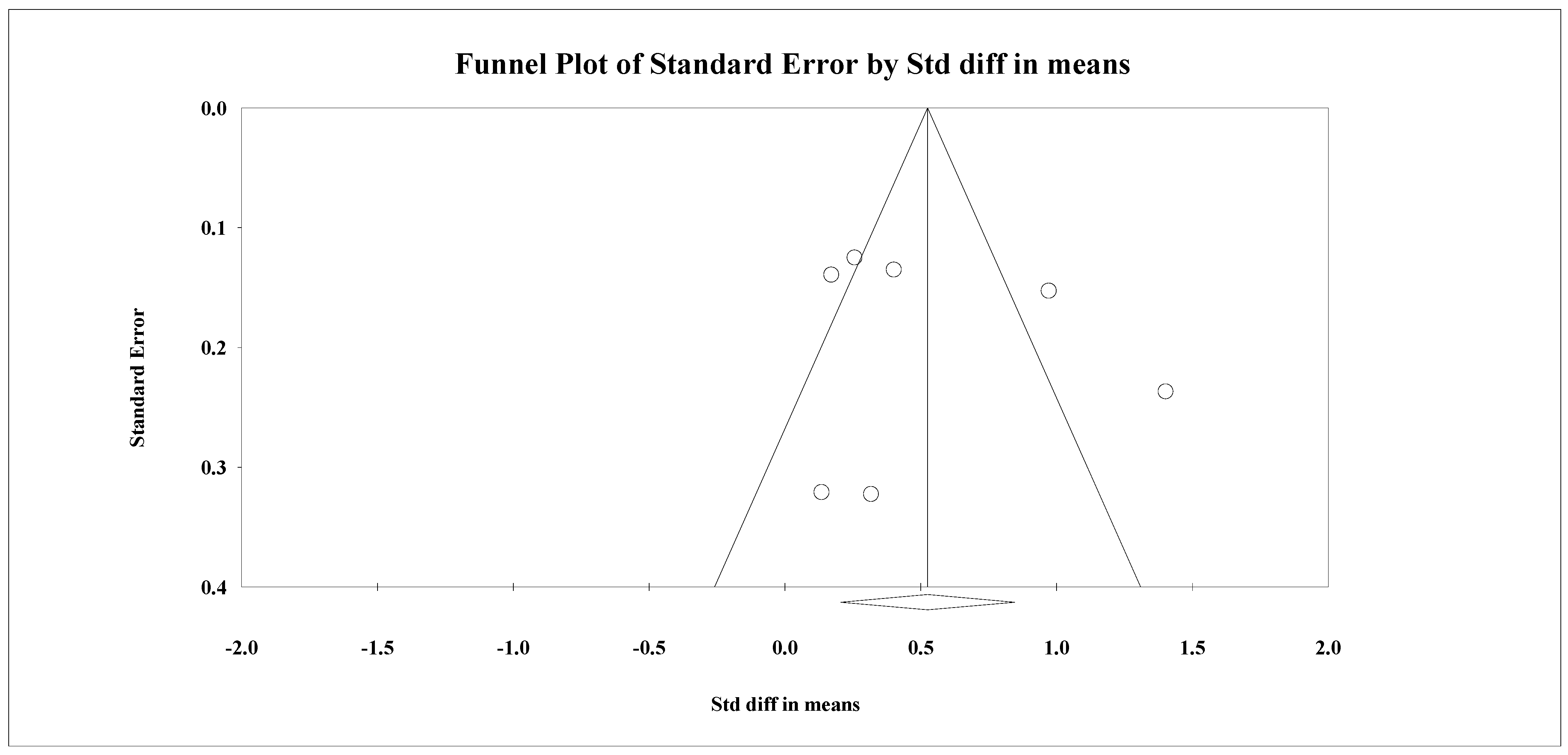
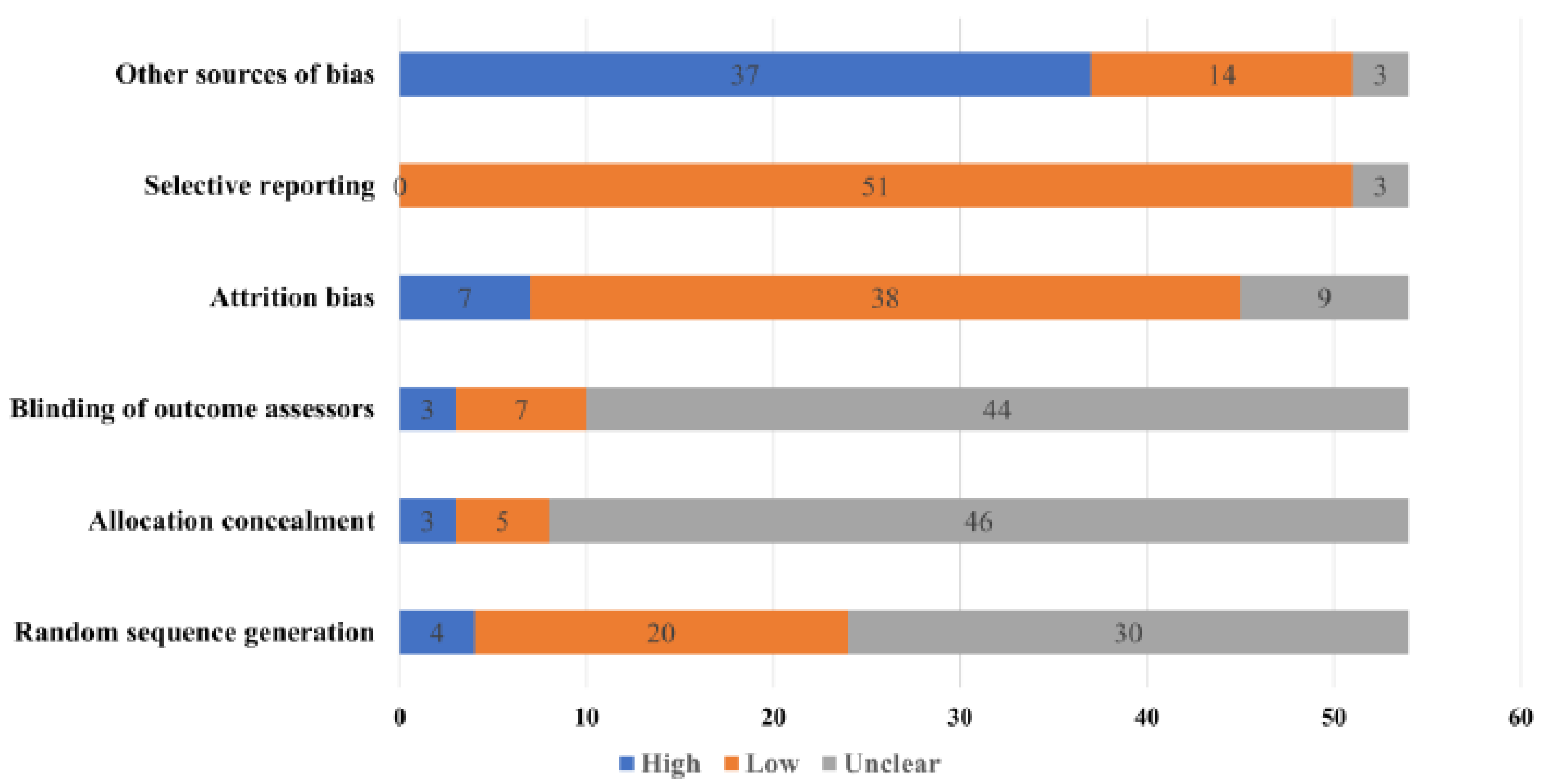
3.4.5. Risk of Bias
4. Discussion
5. Strengths and Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wong, M.C.S.; Huang, J.; Wang, J.; Chan, P.S.F.; Lok, V.; Chen, X.; Leung, C.; Wang, H.H.X.; Lao, X.Q.; Zheng, Z.J. Global, regional and time-trend prevalence of central obesity: A systematic review and meta-analysis of 13.2 million subjects. Eur. J. Epidemiol. 2020, 35, 673–683. [Google Scholar] [CrossRef] [PubMed]
- Emerging Risk Factors Collaboration; Sarwar, N.; Gao, P.; Seshasai, S.R.; Gobin, R.; Kaptoge, S.; Di Angelantonio, E.; Ingelsson, E.; Lawlor, D.A.; Selvin, E.; et al. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: A collaborative meta-analysis of 102 prospective studies. Lancet 2010, 375, 2215–2222. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Economic Costs of Diabetes in the U.S. in 2017. Diabetes Care 2018, 41, 917–928. [Google Scholar] [CrossRef]
- Hugh, W.; Marlon, G. America’s Obesity Crisis: The Health and Economic Costs of Excess Weight; Milken Institute: Santa Monica, CA, USA, 2018. [Google Scholar]
- Moriyama, M.; Nakano, M.; Kuroe, Y.; Nin, K.; Niitani, M.; Nakaya, T. Efficacy of a self-management education program for people with type 2 diabetes: Results of a 12 month trial. Jpn. J. Nurs. Sci. 2009, 6, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Hardhono Susanto, A.; Sawitri, D.R.; Widiyati, S.; Daely, L.W. The Effect of Diabetes Self- Management Education and Support (DSME/S) on Self-Efficacy in Type 2 Diabetes Mellitus Patients. J. Med. Sci. Clin. Res. 2019, 7, 635–641. [Google Scholar] [CrossRef]
- Trouilloud, D.; Regnier, J. Therapeutic education among adults with type 2 diabetes: Effects of a three-day intervention on perceived competence, self-management behaviours and glycaemic control. Glob. Health Promot. 2013, 20, 94–98. [Google Scholar] [CrossRef]
- Webb, E.M.; Rheeder, P. A cluster-randomized trial to estimate the effect of mobile screening and treatment feedback on HbA1c and diabetes-related complications in Tshwane primary health care clinics, South Africa. Prim. Care Diabetes 2017, 11, 546–554. [Google Scholar] [CrossRef]
- Whitehead, L.C.; Crowe, M.T.; Carter, J.D.; Maskill, V.R.; Carlyle, D.; Bugge, C.; Frampton, C.M.A. A nurse-led education and cognitive behaviour therapy-based intervention among adults with uncontrolled type 2 diabetes: A randomised controlled trial. J. Eval. Clin. Pract. 2017, 23, 821–829. [Google Scholar] [CrossRef]
- World Health Organization Regional Office for Europe Copenhagen. Therapeutic Patient Education: Continuing Education Programmes for Health Care Providers in the Field of Prevention of Chronic Diseases; World Health Organization Regional Office for Europe: Copenhagen, Denmark, 1998. [Google Scholar]
- Correia, J.C.; Waqas, A.; Aujoulat, I.; Davies, M.J.; Assal, J.-P.; Golay, A.; Pataky, Z. Evolution of Therapeutic Patient Education: A Systematic Scoping Review and Scientometric Analysis. Int. J. Environ. Res. Public Health 2022, 19, 6128. [Google Scholar] [CrossRef]
- Tanaka, R.; Shibayama, T.; Sugimoto, K.; Hidaka, K. Diabetes self-management education and support for adults with newly diagnosed type 2 diabetes mellitus: A systematic review and meta-analysis of randomized controlled trials. Diabetes Res. Clin. Pract. 2020, 169, 108480. [Google Scholar] [CrossRef]
- Sonal Sekhar, M.; Unnikrishnan, M.K.; Vijayanarayana, K.; Rodrigues, G.S. Impact of patient-education on health related quality of life of diabetic foot ulcer patients: A randomized study. Clin. Epidemiol. Glob. Health 2019, 7, 382–388. [Google Scholar] [CrossRef]
- Anderson, L.J.; Nuckols, T.K.; Coles, C.; Le, M.M.; Schnipper, J.L.; Shane, R.; Jackevicius, C.; Lee, J.; Pevnick, J.M.; Members of the PHARM-DC Group. A systematic overview of systematic reviews evaluating medication adherence interventions. Am. J. Health Syst. Pharm. 2020, 77, 138–147. [Google Scholar] [CrossRef]
- Nkhoma, D.E.; Soko, C.J.; Bowrin, P.; Manga, Y.B.; Greenfield, D.; Househ, M.; Li Jack, Y.C.; Iqbal, U. Digital interventions self-management education for type 1 and 2 diabetes: A systematic review and meta-analysis. Comput. Methods Programs Biomed. 2021, 210, 106370. [Google Scholar] [CrossRef]
- Correia, J.C.; Somers, F.; Golay, A.; Pataky, Z. Therapeutic patient education effectiveness: A systematic review and meta-analysis. Prospero 2019, CRD42019141294. Available online: https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42019141294 (accessed on 3 July 2022).
- Page, M.J.; Moher, D.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. PRISMA 2020 explanation and elaboration: Updated guidance and exemplars for reporting systematic reviews. BMJ 2021, 372, n160. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions; Wiley: Chichester, UK, 2019. [Google Scholar] [CrossRef]
- Duval, S.; Tweedie, R. A Nonparametric “Trim and Fill” Method of Accounting for Publication Bias in Meta-Analysis. J. Am. Stat. Assoc. 2000, 95, 89–98. [Google Scholar] [CrossRef]
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.T.; Rothstein, H.R. Introduction to Meta-Analysis; Wiley: Chichester, UK, 2021. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Oxman, A.D.; Schünemann, H.J.; Tugwell, P.; Knottnerus, A. GRADE guidelines: A new series of articles in the Journal of Clinical Epidemiology. J. Clin. Epidemiol. 2011, 64, 380–382. [Google Scholar] [CrossRef]
- Correia, J.; Waqas, A.; Assal, J.; Davies, M.; Somers, F.; Golay, A.; Pataky, Z. Effectiveness of therapeutic patient education interventions for chronic diseases: A systematic review and meta-analyses of randomized controlled trials. Preprints 2022, in press. [Google Scholar] [CrossRef]
- Carter, E.L.; Nunlee-Bland, G.; Callender, C. A patient-centric, provider-assisted diabetes telehealth self-management intervention for urban minorities. Perspect. Health Inf. Manag. 2011, 8, 1b. [Google Scholar]
- McKay, H.G.; King, D.; Eakin, E.G.; Seeley, J.R.; Glasgow, R.E. The Diabetes Network Internet-Based Physical Activity Intervention. Diabetes Care 2001, 24, 1328–1334. [Google Scholar] [CrossRef]
- Blomfield, R.L.; Collins, C.E.; Hutchesson, M.J.; Young, M.D.; Jensen, M.E.; Callister, R.; Morgan, P.J. Impact of self-help weight loss resources with or without online support on the dietary intake of overweight and obese men: The SHED-IT randomised controlled trial. Obes. Res. Clin. Pract. 2014, 8, e476–e487. [Google Scholar] [CrossRef]
- Ramadas, A.; Chan, C.K.Y.; Oldenburg, B.; Hussein, Z.; Quek, K.F. Randomised-controlled trial of a web-based dietary intervention for patients with type 2 diabetes: Changes in health cognitions and glycemic control. BMC Public Health 2018, 18, 716. [Google Scholar] [CrossRef]
- Shea, S.; Consortium, I.D. The Informatics for Diabetes and Education Telemedicine (IDEATel) project. Trans. Am. Clin. Climatol. Assoc. 2007, 118, 289–304. [Google Scholar]
- Huizinga, M.M.; Gebretsadik, T.; Garcia Ulen, C.; Shintani, A.K.; Michon, S.R.; Shackleford, L.O.; Wolff, K.L.; Brown, A.W.; Rothman, R.L.; Elasy, T.A. Preventing glycaemic relapse in recently controlled type 2 diabetes patients: A randomised controlled trial. Diabetologia 2010, 53, 832–839. [Google Scholar] [CrossRef]
- Williams, E.D.; Bird, D.; Forbes, A.W.; Russell, A.; Ash, S.; Friedman, R.; Scuffham, P.A.; Oldenburg, B. Randomised controlled trial of an automated, interactive telephone intervention (TLC Diabetes) to improve type 2 diabetes management: Baseline findings and six-month outcomes. BMC Public Health 2012, 12, 602. [Google Scholar] [CrossRef]
- Al-Haj Mohd, M.M.M.; Phung, H.; Sun, J.; Morisky, D.E. Improving adherence to medication in adults with diabetes in the United Arab Emirates. BMC Public Health 2016, 16, 857. [Google Scholar] [CrossRef]
- Sönnichsen, A.C.; Winkler, H.; Flamm, M.; Panisch, S.; Kowatsch, P.; Klima, G.; Fürthauer, B.; Weitgasser, R. The effectiveness of the Austrian disease management programme for type 2 diabetes: A cluster-randomised controlled trial. BMC Fam. Pract. 2010, 11, 86. [Google Scholar] [CrossRef]
- Chao, Y.-H.; Usher, K.; Buettner, P.G.; Holmes, C. Cluster randomised controlled trial: Educational self-care intervention with older Taiwanese patients with Type 2 diabetes mellitus––Impact on blood glucose levels and diabetic complications. Collegian 2014, 21, 43–51. [Google Scholar] [CrossRef]
- Kruger, S.; Guthrie, D. Foot Care: Knowledge Retention and Self-care Practices. Diabetes Educ. 1992, 18, 487–490. [Google Scholar] [CrossRef]
- Chaveepojnkamjorn, W.; Pichainarong, N.; Schelp, F.P.; Mahaweerawat, U. A randomized controlled trial to improve the quality of life of type 2 diabetic patients using a self-help group program. Southeast Asian J. Trop. Med. Public Health 2009, 2009, 169–176. [Google Scholar]
- Gagliardino, J.J.; Lapertosa, S.; Pfirter, G.; Villagra, M.; Caporale, J.E.; Gonzalez, C.D.; Elgart, J.; González, L.; Cernadas, C.; Rucci, E.; et al. Clinical, metabolic and psychological outcomes and treatment costs of a prospective randomized trial based on different educational strategies to improve diabetes care (PRODIACOR). Diabet. Med. 2013, 30, 1102–1111. [Google Scholar] [CrossRef] [PubMed]
- Gillett, M.; Dallosso, H.M.; Dixon, S.; Brennan, A.; Carey, M.E.; Campbell, M.J.; Heller, S.; Khunti, K.; Skinner, T.C.; Davies, M.J. Delivering the diabetes education and self management for ongoing and newly diagnosed (DESMOND) programme for people with newly diagnosed type 2 diabetes: Cost effectiveness analysis. BMJ 2010, 341, c4093. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.; Siu, C.F.; Leung, K.T.; Lau, L.C.H.; Chan, C.C.M.; Wong, K.K. General practice and social service partnership for better clinical outcomes, patient self efficacy and lifestyle behaviours of diabetic care: Randomised control trial of a chronic care model. Postgrad. Med. J. 2011, 87, 688–693. [Google Scholar] [CrossRef][Green Version]
- Riddell, M.A.; Dunbar, J.A.; Absetz, P.; Wolfe, R.; Li, H.; Brand, M.; Aziz, Z.; Oldenburg, B.; Australasian Peers for Progress Diabetes Project Investigators. Cardiovascular risk outcome and program evaluation of a cluster randomised controlled trial of a community-based, lay peer led program for people with diabetes. BMC Public Health 2016, 16, 864. [Google Scholar] [CrossRef]
- McRobbie, H.; Hajek, P.; Peerbux, S.; Kahan, B.C.; Eldridge, S.; Trépel, D.; Parrott, S.; Griffiths, C.; Snuggs, S.; Myers Smith, K. Tackling obesity in areas of high social deprivation: Clinical effectiveness and cost-effectiveness of a task-based weight management group programme—A randomised controlled trial and economic evaluation. Health Technol. Assess. 2016, 20, 1–150. [Google Scholar] [CrossRef]
- Ko, S.H.; Song, K.H.; Kim, S.R.; Lee, J.M.; Kim, J.S.; Shin, J.H.; Cho, Y.K.; Park, Y.M.; Jeong, J.H.; Yoon, K.H.; et al. Long-term effects of a structured intensive diabetes education programme (SIDEP) in patients with Type 2 diabetes mellitus-a 4-year follow-up study. Diabet. Med. 2007, 24, 55–62. [Google Scholar] [CrossRef]
- Schachinger, H.; Hegar, K.; Hermanns, N.; Straumann, M.; Keller, U.; Fehm-Wolfsdorf, G.; Berger, W.; Cox, D. Randomized Controlled Clinical Trial of Blood Glucose Awareness Training (BGAT III) in Switzerland and Germany. J. Behav. Med. 2005, 28, 587–594. [Google Scholar] [CrossRef]
- Mohammadi, S.; Karim, N.A.; Talib, R.A.; Amani, R. The impact of self-efficacy education based on the health belief model in Iranian patients with type 2 diabetes: A randomised controlled intervention study. Asia Pac. J. Clin. Nutr. 2018, 27, 546–555. [Google Scholar] [CrossRef]
- Cade, J.E.; Kirk, S.F.; Nelson, P.; Hollins, L.; Deakin, T.; Greenwood, D.C.; Harvey, E.L. Can peer educators influence healthy eating in people with diabetes? Results of a randomized controlled trial. Diabet. Med. 2009, 26, 1048–1054. [Google Scholar] [CrossRef]
- McGowan, P. The Relative Effectiveness of Self-Management Programs for Type 2 Diabetes. Can. J. Diabetes 2015, 39, 411–419. [Google Scholar] [CrossRef]
- Cox, D.J.; Kovatchev, B.; Kovatchev, B.; Koev, D.; Koeva, L.; Dachev, S.; Tcharaktchiev, D.; Protopopova, A.; Gonder-Frederick, L.; Clarke, W. Hypoglycemia anticipation, awareness and treatment training (HAATT) reduces occurrence of severe hypoglycemia among adults with type 1 diabetes mellitus. Int. J. Behav. Med. 2004, 11, 212–218. [Google Scholar] [CrossRef]
- Goudswaard, A.N.; Stolk, R.P.; Zuithoff, N.P.A.; de Valk, H.W.; Rutten, G.E.H.M. Long-term effects of self-management education for patients with Type 2 diabetes taking maximal oral hypoglycaemic therapy: A randomized trial in primary care. Diabet. Med. 2004, 21, 491–496. [Google Scholar] [CrossRef]
- Nejhaddadgar, N.; Darabi, F.; Rohban, A.; Solhi, M.; Kheire, M. The effectiveness of self-management program for people with type 2 diabetes mellitus based on PRECEDE-PROCEDE model. Diabetes Metab. Syndr. Clin. Res. Rev. 2019, 13, 440–443. [Google Scholar] [CrossRef]
- Nichter, M.; Mini, G.K.; Thankappan, K.R. Low-level smoking among diabetes patients in India: A smoking cessation challenge. Clin. Epidemiol. Glob. Health 2018, 6, 176–180. [Google Scholar] [CrossRef]
- Piatt, G.A.; Anderson, R.M.; Brooks, M.M.; Songer, T.; Siminerio, L.M.; Korytkowski, M.M.; Zgibor, J.C. 3-Year Follow-up of Clinical and Behavioral Improvements Following a Multifaceted Diabetes Care Intervention. Diabetes Educ. 2010, 36, 301–309. [Google Scholar] [CrossRef]
- Reid, R.D.; Malcolm, J.; Wooding, E.; Geertsma, A.; Aitken, D.; Arbeau, D.; Blanchard, C.; Gagnier, J.-A.; Gupta, A.; Mullen, K.-A.; et al. Prospective, Cluster-Randomized Trial to Implement the Ottawa Model for Smoking Cessation in Diabetes Education Programs in Ontario, Canada. Diabetes Care 2017, 41, 406–412. [Google Scholar] [CrossRef]
- Salinero-Fort, M.A.; Carrillo-de Santa Pau, E.; Arrieta-Blanco, F.J.; Abanades-Herranz, J.C.; Martín-Madrazo, C.; Rodés-Soldevila, B.; de Burgos-Lunar, C. Effectiveness of PRECEDE model for health education on changes and level of control of HbA1c, blood pressure, lipids, and body mass index in patients with type 2 diabetes mellitus. BMC Public Health 2011, 11, 267. [Google Scholar] [CrossRef]
- Shibayama, T.; Kobayashi, K.; Takano, A.; Kadowaki, T.; Kazuma, K. Effectiveness of lifestyle counseling by certified expert nurse of Japan for non-insulin-treated diabetic outpatients: A 1-year randomized controlled trial. Diabetes Res. Clin. Pract. 2007, 76, 265–268. [Google Scholar] [CrossRef]
- Rachmani, R.; Slavacheski, I.; Berla, M.; Frommer-Shapira, R.; Ravid, M. Treatment of High-Risk Patients with Diabetes: Motivation and Teaching Intervention: A Randomized, Prospective 8-Year Follow-Up Study. J. Am. Soc. Nephrol. 2005, 16, S22–S26. [Google Scholar] [CrossRef]
- Seligman, H.K.; Smith, M.; Rosenmoss, S.; Marshall, M.B.; Waxman, E. Comprehensive Diabetes Self-Management Support From Food Banks: A Randomized Controlled Trial. Am. J. Public Health 2018, 108, 1227–1234. [Google Scholar] [CrossRef]
- Lutes, L.D.; Winett, R.A.; Barger, S.D.; Wojcik, J.R.; Herbert, W.G.; Nickols-Richardson, S.M.; Anderson, E.S. Small Changes in Nutrition and Physical Activity Promote Weight Loss and Maintenance: 3-Month Evidence from the ASPIRE Randomized Trial. Ann. Behav. Med. 2008, 35, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Tourkmani, A.M.; Abdelhay, O.; Alkhashan, H.I.; Alaboud, A.F.; Bakhit, A.; Elsaid, T.; Alawad, A.; Alobaikan, A.; Alqahtani, H.; Alqahtani, A.; et al. Impact of an integrated care program on glycemic control and cardiovascular risk factors in patients with type 2 diabetes in Saudi Arabia: An interventional parallel-group controlled study. BMC Fam. Pract. 2018, 19, 1. [Google Scholar] [CrossRef] [PubMed]
- Kampan, P. Effects of counseling and implementation of clinical pathway on diabetic patients hospitalized with hypoglycemia. J. Med. Assoc. Thai. 2006, 89, 619–625. [Google Scholar] [PubMed]
- Aiello, L.P.; Ayala, A.R.; Antoszyk, A.N.; Arnold-Bush, B.; Baker, C.; Bressler, N.M.; Elman, M.J.; Glassman, A.R.; Jampol, L.M.; Melia, M.; et al. Assessing the Effect of Personalized Diabetes Risk Assessments During Ophthalmologic Visits on Glycemic Control: A Randomized Clinical Trial. JAMA Ophthalmol. 2015, 133, 888–896. [Google Scholar] [CrossRef]
- Luan, L.L.; Yang, J.; He, Y.F.; Huang, Z.X.; Liu, L.; Huabg, Z.S. Impact of diabetes education and self-management support on the 4D series of diabetes patients. Biomed. Res. 2017, 28, 1172–1177. [Google Scholar]
- Farmanbar, R.; Moghadam, M.Z.; Omidi, S.; Abolhasan, A. The effects of intervention based on the trans-theoretical model on physical activity and metabolic control of diabetic women. Gazi Med. J. 2019, 30, 36–139. [Google Scholar]
- Chen, J.-H.; Ou, H.-T.; Lin, T.-C.; Lai, E.C.-C.; Yang Kao, Y.-H. Pharmaceutical care of elderly patients with poorly controlled type 2 diabetes mellitus: A randomized controlled trial. Int. J. Clin. Pharm. 2015, 38, 88–95. [Google Scholar] [CrossRef]
- Chow, E.P.; Hassali, M.A.; Saleem, F.; Aljadhey, H. Effects of pharmacist-led patient education on diabetes-related knowledge and medication adherence: A home-based study. Health Educ. J. 2015, 75, 421–433. [Google Scholar] [CrossRef]
- Jarab, A.S.; Alqudah, S.G.; Mukattash, T.L.; Shattat, G.; Al-Qirim, T. Randomized Controlled Trial of Clinical Pharmacy Management of Patients with Type 2 Diabetes in an Outpatient Diabetes Clinic in Jordan. J. Manag. Care Pharm. 2012, 18, 516–526. [Google Scholar] [CrossRef]
- Assunção, M.C.F.; Gigante, D.P.; Cardoso, M.A.; Sartorelli, D.S.; Santos, I.S. Randomized, controlled trial promotes physical activity and reduces consumption of sweets and sodium among overweight and obese adults. Nutr. Res. 2010, 30, 541–549. [Google Scholar] [CrossRef]
- Perri, M.G.; Limacher, M.C.; Durning, P.E.; Janicke, D.M.; Lutes, L.D.; Bobroff, L.B.; Dale, M.S.; Daniels, M.J.; Radcliff, T.A.; Martin, A.D. Extended-care programs for weight management in rural communities: The treatment of obesity in underserved rural settings (TOURS) randomized trial. Arch. Intern. Med. 2008, 168, 2347–2354. [Google Scholar] [CrossRef]
- Wadden, T.A.; Volger, S.; Sarwer, D.B.; Vetter, M.L.; Tsai, A.G.; Berkowitz, R.I.; Kumanyika, S.; Schmitz, K.H.; Diewald, L.K.; Barg, R.; et al. A two-year randomized trial of obesity treatment in primary care practice. N. Engl. J. Med. 2011, 365, 1969–1979. [Google Scholar] [CrossRef]
- Korhonen, T.; Huttunen, J.K.; Aro, A.; Hentinen, M.; Ihalainen, O.; Majander, H.; Siitonen, O.; Uusitupa, M.; Pyörälä, K. A Controlled Trial on the Effects of Patient Education in the Treatment of Insulin-Dependent Diabetes. Diabetes Care 1983, 6, 256–261. [Google Scholar] [CrossRef]
- Schillinger, D.; Handley, M.; Wang, F.; Hammer, H. Effects of self-management support on structure, process, and outcomes among vulnerable patients with diabetes: A three-arm practical clinical trial. Diabetes Care 2009, 32, 559–566. [Google Scholar] [CrossRef]
- Chao, J.; Yang, L.; Xu, H.; Yu, Q.; Jiang, L.; Zong, M. The effect of integrated health management model on the health of older adults with diabetes in a randomized controlled trial. Arch. Gerontol. Geriatr. 2015, 60, 82–88. [Google Scholar] [CrossRef]
- Wagner, E.H.; Grothaus, L.C.; Sandhu, N.; Galvin, M.S.; McGregor, M.; Artz, K.; Coleman, E.A. Chronic care clinics for diabetes in primary care: A system-wide randomized trial. Diabetes Care 2001, 24, 695–700. [Google Scholar] [CrossRef]
- Glasgow, R.E.; La Chance, P.-A.; Toobert, D.J.; Brown, J.; Hampson, S.E.; Riddle, M.C. Long term effects and costs of brief behavioural dietary intervention for patients with diabetes delivered from the medical office. Patient Educ. Couns. 1997, 32, 175–184. [Google Scholar] [CrossRef]
- Howorka, K.; Pumprla, J.; Wagner-Nosiska, D.; Grillmayr, H.; Schlusche, C.; Schabmann, A. Empowering diabetes out-patients with structured education: Short-term and long-term effects of functional insulin treatment on perceived control over diabetes. J. Psychosom. Res. 2000, 48, 37–44. [Google Scholar] [CrossRef]
- Norris, S.L.; Lau, J.; Smith, S.J.; Schmid, C.H.; Engelgau, M.M. Self-Management Education for Adults With Type 2 Diabetes: A Meta-Analysis of the Effect on Glycemic Control. Diabetes Care 2002, 25, 1159–1171. [Google Scholar] [CrossRef]
- Miller, T.A. Health literacy and adherence to medical treatment in chronic and acute illness: A meta-analysis. Patient Educ. Couns. 2016, 99, 1079–1086. [Google Scholar] [CrossRef]
- Devine, E.C.; Cook, T.D. Clinical and cost-saving effects of psychoeducational interventions with surgical patients: A meta-analysis. Res. Nurs. Health 1986, 9, 89–105. [Google Scholar] [CrossRef]
- Petre, B.; Gagnayre, R.; De Andrade, V.; Ziegler, O.; Guillaume, M. From therapeutic patient education principles to educative attitude: The perceptions of health care professionals—A pragmatic approach for defining competencies and resources. Patient Prefer. Adherence 2017, 11, 603–617. [Google Scholar] [CrossRef]
- Ahmad, W.; Krupat, E.; Asma, Y.; Fatima, N.E.; Attique, R.; Mahmood, U.; Waqas, A. Attitudes of medical students in Lahore, Pakistan towards the doctor-patient relationship. PeerJ 2015, 3, e1050. [Google Scholar] [CrossRef]
- Moreno, L.; Dale, S.B.; Chen, A.Y.; Magee, C.A. Costs to Medicare of the Informatics for Diabetes Education and Telemedicine (IDEATel) home telemedicine demonstration: Findings from an independent evaluation. Diabetes Care 2009, 32, 1202–1204. [Google Scholar] [CrossRef][Green Version]

| Group | n | SMD | 95% CI | I-Squared | Q | df | p | |
|---|---|---|---|---|---|---|---|---|
| Serum HbA1c levels among patients with diabetes | ||||||||
| Delivery agents | ||||||||
| Allied health workers | 12 | 0.26 | 0.10 | 0.43 | 59.98 | 21.49 | 3 | <0.001 |
| Doctors | 1 | −0.12 | −0.23 | −0.02 | 0.00 | |||
| Multidisciplinary | 10 | 0.35 | 0.09 | 0.61 | 92.33 | |||
| Peers | 1 | 0.00 | −0.29 | 0.29 | 0.00 | |||
| Format of delivery | ||||||||
| Group | 9 | 0.27 | 0.05 | 0.50 | 85.38 | 2.32 | 3 | 0.51 |
| Individual | 9 | 0.33 | 0.04 | 0.62 | 92.08 | |||
| Mixed | 3 | 0.28 | −0.08 | 0.64 | 80.28 | |||
| Telephone | 3 | 0.05 | −0.23 | 0.33 | 0.00 | |||
| Body weight among patients with obesity | ||||||||
| Delivery agents | ||||||||
| Allied health workers | 4 | 0.44 | 0.11 | 0.77 | 83.32 | 15.02 | 2 | <0.001 |
| Multidisciplinary | 2 | 0.23 | −0.22 | 0.67 | 0.00 | |||
| Research staff | 1 | 1.40 | 0.94 | 1.87 | 0.00 | |||
| Format of delivery | ||||||||
| Group | 1 | 0.26 | 0.01 | 0.50 | 0.00 | 19.61 | 3.00 | <0.001 |
| Individual | 2 | 0.23 | −0.22 | 0.67 | 0.00 | |||
| Internet | 1 | 1.40 | 0.94 | 1.87 | 0.00 | |||
| Mixed | 3 | 0.51 | 0.06 | 0.96 | 87.21 | |||
| Covariate | B | SE | t | p-Value |
|---|---|---|---|---|
| Intercept | −0.402 | 0.341 | −1.18 | 0.25 |
| Disease management | 0.061 | 0.063 | 0.97 | 0.34 |
| Lifestyle | 0.016 | 0.136 | 0.11 | 0.91 |
| Coping total | 0.009 | 0.041 | 0.22 | 0.83 |
| Disease processes | 0.19 | 0.114 | 1.67 | 0.11 |
| Interpersonal skill | 0.15 | 0.092 | 1.64 | 0.12 |
| R2 = 0.37 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Correia, J.C.; Waqas, A.; Huat, T.S.; Gariani, K.; Jornayvaz, F.R.; Golay, A.; Pataky, Z. Effectiveness of Therapeutic Patient Education Interventions in Obesity and Diabetes: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2022, 14, 3807. https://doi.org/10.3390/nu14183807
Correia JC, Waqas A, Huat TS, Gariani K, Jornayvaz FR, Golay A, Pataky Z. Effectiveness of Therapeutic Patient Education Interventions in Obesity and Diabetes: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients. 2022; 14(18):3807. https://doi.org/10.3390/nu14183807
Chicago/Turabian StyleCorreia, Jorge C., Ahmed Waqas, Teoh Soo Huat, Karim Gariani, François R. Jornayvaz, Alain Golay, and Zoltan Pataky. 2022. "Effectiveness of Therapeutic Patient Education Interventions in Obesity and Diabetes: A Systematic Review and Meta-Analysis of Randomized Controlled Trials" Nutrients 14, no. 18: 3807. https://doi.org/10.3390/nu14183807
APA StyleCorreia, J. C., Waqas, A., Huat, T. S., Gariani, K., Jornayvaz, F. R., Golay, A., & Pataky, Z. (2022). Effectiveness of Therapeutic Patient Education Interventions in Obesity and Diabetes: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients, 14(18), 3807. https://doi.org/10.3390/nu14183807














