α-Tocopherol Pharmacokinetics in Adults with Cystic Fibrosis: Benefits of Supplemental Vitamin C Administration
Abstract
1. Introduction
2. Materials and Methods
2.1. Participant Characteristics
2.2. Deuterium-Labeled Vitamin E
2.3. Study Design and Blood Sampling
2.4. Laboratory Analyses
2.5. Mathematical and Statistical Analyeis
3. Results
3.1. Baseline CF Subject Characteristics
3.2. Efficacy of Vitamin C Supplementation
3.3. Vitamin E Pharmacokinetics
4. Discussion
4.1. Nuances for the CF Community
4.2. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cystic Fibrosis Foundation. 2021 Cystic Fibrosis Foundation Patient Registry Highlights. Available online: https://www.cff.org/medical-professionals/patient-registry (accessed on 16 August 2022).
- Guo, J.; Garratt, A.; Hill, A. Worldwide rates of diagnosis and effective treatment for cystic fibrosis. J. Cyst. Fibros. 2022, 21, 456–462. [Google Scholar] [CrossRef]
- Office of Science (OS); Office of Genomics and Precision Public Health; Centers for Diesease Control and Prevention. Cystic Fibrosis. Available online: https://www.cdc.gov/genomics/disease/cystic_fibrosis.htm (accessed on 16 August 2022).
- Doring, G.; Gulbins, E. Cystic fibrosis and innate immunity: How chloride channel mutations provoke lung disease. Cell. Microbiol. 2009, 11, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Stoltz, D.A.; Meyerholz, D.K.; Welsh, M.J. Origins of cystic fibrosis lung disease. N. Engl. J. Med. 2015, 372, 351–362. [Google Scholar] [CrossRef] [PubMed]
- Quinn, R.A.; Adem, S.; Mills, R.H.; Comstock, W.; DeRight Goldasich, L.; Humphrey, G.; Aksenov, A.A.; Melnik, A.V.; da Silva, R.; Ackermann, G.; et al. Neutrophilic proteolysis in the cystic fibrosis lung correlates with a pathogenic microbiome. Microbiome 2019, 7, 23. [Google Scholar] [CrossRef] [PubMed]
- Bruce, M.C.; Poncz, L.; Klinger, J.D.; Stern, R.C.; Tomashefski, J.F., Jr.; Dearborn, D.G. Biochemical and pathologic evidence for proteolytic destruction of lung connective tissue in cystic fibrosis. Am. Rev. Respir Dis. 1985, 132, 529–535. [Google Scholar] [CrossRef]
- Downey, D.G.; Bell, S.C.; Elborn, J.S. Neutrophils in cystic fibrosis. Thorax 2009, 64, 81–88. [Google Scholar] [CrossRef]
- Brown, A.J. Acute effects of smoking cessation on antioxidant status. Nutr. Biochem. 1996, 7, 29–39. [Google Scholar] [CrossRef]
- van der Vliet, A.; Eiserich, J.P.; Marelich, G.P.; Halliwell, B.; Cross, C.E. Oxidative stress in cystic fibrosis: Does it occur and does it matter? Adv. Pharmacol. 1997, 38, 491–513. [Google Scholar] [CrossRef]
- Wood, L.G.; Fitzgerald, D.A.; Gibson, P.G.; Cooper, D.M.; Collins, C.E.; Garg, M.L. Oxidative stress in cystic fibrosis: Dietary and metabolic factors. J. Am. Coll. Nutr. 2001, 20, 157–165. [Google Scholar] [CrossRef]
- Back, E.I.; Frindt, C.; Nohr, D.; Frank, J.; Ziebach, R.; Stern, M.; Ranke, M.; Biesalski, H.K. Antioxidant deficiency in cystic fibrosis: When is the right time to take action? Am. J. Clin. Nutr. 2004, 80, 374–384. [Google Scholar] [CrossRef]
- Cantin, A.M. Potential for antioxidant therapy of cystic fibrosis. Curr. Opin. Pulm. Med. 2004, 10, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Cantin, A.M.; White, T.B.; Cross, C.E.; Forman, H.J.; Sokol, R.J.; Borowitz, D. Antioxidants in cystic fibrosis. Conclusions from the CF antioxidant workshop, Bethesda, Maryland, November 11–12, 2003. Free Radic. Biol. Med. 2007, 42, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Lucidi, V.; Ciabattoni, G.; Bella, S.; Barnes, P.J.; Montuschi, P. Exhaled 8-isoprostane and prostaglandin E(2) in patients with stable and unstable cystic fibrosis. Free Radic. Biol. Med. 2008, 45, 913–919. [Google Scholar] [CrossRef]
- Galli, F.; Battistoni, A.; Gambari, R.; Pompella, A.; Bragonzi, A.; Pilolli, F.; Iuliano, L.; Piroddi, M.; Dechecchi, M.C.; Cabrini, G.; et al. Oxidative stress and antioxidant therapy in cystic fibrosis. Biochim. Biophys. Acta 2012, 1822, 690–713. [Google Scholar] [CrossRef] [PubMed]
- Lezo, A.; Biasi, F.; Massarenti, P.; Calabrese, R.; Poli, G.; Santini, B.; Bignamini, E. Oxidative stress in stable cystic fibrosis patients: Do we need higher antioxidant plasma levels? J. Cyst. Fibros. 2013, 12, 35–41. [Google Scholar] [CrossRef]
- Causer, A.J.; Shute, J.K.; Cummings, M.H.; Shepherd, A.I.; Gruet, M.; Costello, J.T.; Bailey, S.; Lindley, M.; Pearson, C.; Connett, G.; et al. Circulating biomarkers of antioxidant status and oxidative stress in people with cystic fibrosis: A systematic review and meta-analysis. Redox Biol. 2020, 32, 101436. [Google Scholar] [CrossRef]
- Galiniak, S.; Molon, M.; Rachel, M. Links between Disease Severity, Bacterial Infections and Oxidative Stress in Cystic Fibrosis. Antioxidants 2022, 11, 887. [Google Scholar] [CrossRef]
- Singh, V.K.; Schwarzenberg, S.J. Pancreatic insufficiency in Cystic Fibrosis. J. Cyst. Fibros. 2017, 16 (Suppl. S2), S70–S78. [Google Scholar] [CrossRef]
- van de Peppel, I.P.; Bodewes, F.; Verkade, H.J.; Jonker, J.W. Bile acid homeostasis in gastrointestinal and metabolic complications of cystic fibrosis. J. Cyst. Fibros. 2019, 18, 313–320. [Google Scholar] [CrossRef]
- Koscik, R.L.; Farrell, P.M.; Kosorok, M.R.; Zaremba, K.M.; Laxova, A.; Lai, H.C.; Douglas, J.A.; Rock, M.J.; Splaingard, M.L. Cognitive function of children with cystic fibrosis: Deleterious effect of early malnutrition. Pediatrics 2004, 113, 1549–1558. [Google Scholar] [CrossRef]
- Huang, S.H.; Schall, J.I.; Zemel, B.S.; Stallings, V.A. Vitamin E status in children with cystic fibrosis and pancreatic insufficiency. J. Pediatr. 2006, 148, 556–559. [Google Scholar] [CrossRef] [PubMed]
- Okebukola, P.O.; Kansra, S.; Barrett, J. Vitamin E supplementation in people with cystic fibrosis. Cochrane Database Syst. Rev. 2017, 3, CD009422. [Google Scholar] [CrossRef] [PubMed]
- Packer, J.E.; Slater, T.F.; Willson, R.L. Direct observation of a free radical interaction between vitamin E and vitamin C. Nature 1979, 278, 737–738. [Google Scholar] [CrossRef] [PubMed]
- Frei, B.; England, L.; Ames, B.N. Ascorbate is an outstanding antioxidant in human blood plasma. Proc. Natl. Acad. Sci. USA 1989, 86, 6377–6381. [Google Scholar] [CrossRef]
- Buettner, G.R. The pecking order of free radicals and antioxidants: Lipid peroxidation, alpha-tocopherol, and ascorbate. Arch. Biochem. Biophys. 1993, 300, 535–543. [Google Scholar] [CrossRef]
- Buettner, C.; Phillips, R.S.; Davis, R.B.; Gardiner, P.; Mittleman, M.A. Use of dietary supplements among United States adults with coronary artery disease and atherosclerotic risks. Am. J. Cardiol. 2007, 99, 661–666. [Google Scholar] [CrossRef]
- Sato, A.; Takino, Y.; Yano, T.; Fukui, K.; Ishigami, A. Determination of tissue-specific interaction between vitamin C and vitamin E in vivo using senescence marker protein-30 knockout mice as a vitamin C synthesis deficiency model. Br. J. Nutr. 2022, 128, 993–1003. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, J.; Marquardt, D.; DiPasquale, M.; Harroun, T. From fat to bilayers: Understanding where and how vitamin E works. Free Radic. Biol. Med. 2021, 176, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Ford, E.S.; Liu, S.; Mannino, D.M.; Giles, W.H.; Smith, S.J. C-reactive protein concentration and concentrations of blood vitamins, carotenoids, and selenium among United States adults. Eur. J. Clin. Nutr. 2003, 57, 1157–1163. [Google Scholar] [CrossRef] [PubMed]
- Wannamethee, S.G.; Lowe, G.D.; Rumley, A.; Bruckdorfer, K.R.; Whincup, P.H. Associations of vitamin C status, fruit and vegetable intakes, and markers of inflammation and hemostasis. Am. J. Clin. Nutr. 2006, 83, 567–574. [Google Scholar] [CrossRef]
- Winklhofer-Roob, B.M.; Ellemunter, H.; Fruhwirth, M.; Schlegel-Haueter, S.E.; Khoschsorur, G.; van’t Hof, M.A.; Shmerling, D.H. Plasma vitamin C concentrations in patients with cystic fibrosis: Evidence of associations with lung inflammation. Am. J. Clin. Nutr. 1997, 65, 1858–1866. [Google Scholar] [CrossRef] [PubMed]
- Leonard, S.W.; Paterson, E.; Atkinson, J.K.; Ramakrishnan, R.; Cross, C.E.; Traber, M.G. Studies in humans using deuterium-labeled alpha- and gamma-tocopherols demonstrate faster plasma gamma-tocopherol disappearance and greater gamma-metabolite production. Free Radic. Biol. Med. 2005, 38, 857–866. [Google Scholar] [CrossRef] [PubMed]
- Bruno, R.S.; Leonard, S.W.; Atkinson, J.; Montine, T.J.; Ramakrishnan, R.; Bray, T.M.; Traber, M.G. Faster plasma vitamin E disappearance in smokers is normalized by vitamin C supplementation. Free Radic. Biol. Med. 2006, 40, 689–697. [Google Scholar] [CrossRef]
- Bruno, R.S.; Leonard, S.W.; Li, J.; Bray, T.M.; Traber, M.G. Lower plasma alpha-carboxyethyl-hydroxychroman after deuterium-labeled alpha-tocopherol supplementation suggests decreased vitamin E metabolism in smokers. Am. J. Clin. Nutr. 2005, 81, 1052–1059. [Google Scholar] [CrossRef]
- Bruno, R.S.; Ramakrishnan, R.; Montine, T.J.; Bray, T.M.; Traber, M.G. {alpha}-Tocopherol disappearance is faster in cigarette smokers and is inversely related to their ascorbic acid status. Am. J. Clin. Nutr. 2005, 81, 95–103. [Google Scholar] [CrossRef]
- Lei, H.; Atkinson, J. Hydrogen-deuterium exchange during the reductive deuteration of alpha- and gamma-tocopherol chromenes. J. Label. Compd. Radiopharm. 2001, 44, 215–223. [Google Scholar] [CrossRef]
- Podda, M.; Weber, C.; Traber, M.G.; Packer, L. Simultaneous determination of tissue tocopherols, tocotrienols, ubiquinols, and ubiquinones. J. Lipid Res. 1996, 37, 893–901. [Google Scholar] [CrossRef]
- Vaule, H.; Leonard, S.W.; Traber, M.G. Vitamin E delivery to human skin: Studies using deuterated alpha-tocopherol measured by APCI LC-MS. Free Radic. Biol. Med. 2004, 36, 456–463. [Google Scholar] [CrossRef]
- Lodge, J.K.; Traber, M.G.; Elsner, A.; Brigelius-Flohe, R. A rapid method for the extraction and determination of vitamin E metabolites in human urine. J. Lipid Res. 2000, 41, 148–154. [Google Scholar] [CrossRef]
- Hong, Y.L.; Yeh, S.L.; Chang, C.Y.; Hu, M.L. Total plasma malondialdehyde levels in 16 Taiwanese college students determined by various thiobarbituric acid tests and an improved high-performance liquid chromatography-based method. Clin. Biochem. 2000, 33, 619–625. [Google Scholar] [CrossRef]
- Ko, Y.H.; Pedersen, P.L. Cystic fibrosis: A brief look at some highlights of a decade of research focused on elucidating and correcting the molecular basis of the disease. J. Bioenerg. Biomembr. 2001, 33, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Food and Nutrition Board; Institute of Medicine. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids; National Academy Press: Washington, DC, USA, 2000; p. 529. [Google Scholar]
- Bruno, R.S.; Leonard, S.W.; Park, S.; Zhao, Y.Y.; Traber, M.G. Human vitamin E requirements assessed with the use of apples fortified with deuterium-labeled alpha-tocopheryl acetate. Am. J. Clin. Nutr. 2006, 83, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Traber, M.G.; Head, B. Vitamin E: How much is enough, too much and why! Free Radic. Biol. Med. 2021, 177, 212–225. [Google Scholar] [CrossRef] [PubMed]
- Zamel, R.; Khan, R.; Pollex, R.L.; Hegele, R.A. Abetalipoproteinemia: Two case reports and literature review. Orphanet J. Rare Dis. 2008, 3, 19. [Google Scholar] [CrossRef] [PubMed]
- Burnett, J.R.; Hooper, A.J.; Hegele, R.A. Abetalipoproteinemia. BTI—GeneReviews(®). In GeneReviews® [Internet]; Adam, M.P., Everman, D.B., Mirzaa, G.M., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Gripp, K.W., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 2018; [Updated 2022 May 19]. Available online: https://www.ncbi.nlm.nih.gov/books/NBK532447/ (accessed on 16 August 2022).
- Granot, E.; Kohen, R. Oxidative stress in abetalipoproteinemia patients receiving long-term vitamin E and vitamin A supplementation. Am. J. Clin. Nutr. 2004, 79, 226–230. [Google Scholar] [CrossRef]
- Di Donato, I.; Bianchi, S.; Federico, A. Ataxia with vitamin E deficiency: Update of molecular diagnosis. Neurol. Sci. 2010, 31, 511–515. [Google Scholar] [CrossRef]
- Kohlschutter, A.; Finckh, B.; Nickel, M.; Bley, A.; Hubner, C. First recognized patient with genetic vitamin E deficiency stable after 36 years of controlled supplement therapy. Neurodegener. Dis. 2020, 20, 35–38. [Google Scholar] [CrossRef]
- Traber, M.G.; Thellman, C.A.; Rindler, M.J.; Kayden, H.J. Uptake of intact TPGS (d-a-tocopheryl polyethylene glycol 1000 succinate) a water miscible form of vitamin E by human cells in vitro. Am. J. Clin. Nutr. 1988, 48, 605–611. [Google Scholar] [CrossRef]
- Winklhofer-Roob, B.M.; van’t Hof, M.A.; Shmerling, D.H. Long-term oral vitamin E supplementation in cystic fibrosis patients: RRR-alpha-tocopherol compared with all-rac-alpha-tocopheryl acetate preparations. Am. J. Clin. Nutr. 1996, 63, 722–728. [Google Scholar] [CrossRef]
- Winklhofer-Roob, B.M.; Tuchschmid, P.E.; Molinari, L.; Shmerling, D.H. Response to a single oral dose of all-rac-alpha-tocopheryl acetate in patients with cystic fibrosis and in healthy individuals. Am. J. Clin. Nutr. 1996, 63, 717–721. [Google Scholar] [CrossRef][Green Version]
- Traber, M.G.; Leonard, S.W.; Bobe, G.; Fu, X.; Saltzman, E.; Grusak, M.A.; Booth, S.L. alpha-Tocopherol disappearance rates from plasma depend on lipid concentrations: Studies using deuterium-labeled collard greens in younger and older adults. Am. J. Clin. Nutr. 2015, 101, 752–759. [Google Scholar] [CrossRef] [PubMed]
- Violet, P.C.; Ebenuwa, I.C.; Wang, Y.; Niyyati, M.; Padayatty, S.J.; Head, B.; Wilkins, K.; Chung, S.; Thakur, V.; Ulatowski, L.; et al. Vitamin E sequestration by liver fat in humans. JCI Insight 2020, 5, e133309. [Google Scholar] [CrossRef] [PubMed]
- Papas, K.; Kalbfleisch, J.; Mohon, R. Bioavailability of a novel, water-soluble vitamin E formulation in malabsorbing patients. Dig. Dis. Sci. 2007, 52, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Sagel, S.D.; Khan, U.; Jain, R.; Graff, G.; Daines, C.L.; Dunitz, J.M.; Borowitz, D.; Orenstein, D.M.; Abdulhamid, I.; Noe, J.; et al. Effects of an Antioxidant-enriched Multivitamin in Cystic Fibrosis. A Randomized, Controlled, Multicenter Clinical Trial. Am. J. Respir. Crit. Care Med. 2018, 198, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Ciofu, O.; Smith, S.; Lykkesfeldt, J. A systematic Cochrane Review of antioxidant supplementation lung disease for cystic fibrosis. Paediatr. Respir. Rev. 2020, 33, 28–29. [Google Scholar] [CrossRef]
- Farrell, P.M.; Bieri, J.G.; Fratantoni, J.F.; Wood, R.E.; di Sant’Agnese, P.A. The occurrence and effects of human vitamin E deficiency. A study in patients with cystic fibrosis. J. Clin. Investig. 1977, 60, 233–241. [Google Scholar] [CrossRef]
- Okebukola, P.O.; Kansra, S.; Barrett, J. Vitamin E supplementation in people with cystic fibrosis. Cochrane Database Syst. Rev. 2020, 9, CD009422. [Google Scholar] [CrossRef]
- Nowak, J.K.; Sobkowiak, P.; Drzymała-Czyż, S.; Krzyżanowska-Jankowska, P.; Sapiejka, E.; Skorupa, W.; Pogorzelski, A.; Nowicka, A.; Wojsyk-Banaszak, I.; Kurek, S.; et al. Fat-Soluble Vitamin Supplementation Using Liposomes, Cyclodextrins, or Medium-Chain Triglycerides in Cystic Fibrosis: A Randomized Controlled Trial. Nutrients 2021, 13, 4554. [Google Scholar] [CrossRef]
- Wagener, B.M.; Anjum, N.; Evans, C.; Brandon, A.; Honavar, J.; Creighton, J.; Traber, M.G.; Stuart, R.L.; Stevens, T.; Pittet, J.F. alpha-Tocopherol Attenuates the Severity of Pseudomonas aeruginosa-induced Pneumonia. Am. J. Respir. Cell Mol. Biol. 2020, 63, 234–243. [Google Scholar] [CrossRef]
- Cross, C.E.; Traber, M.; Eiserich, J.; van der Vliet, A. Micronutrient antioxidants and smoking. Br. Med. Bull. 1999, 55, 691–704. [Google Scholar] [CrossRef]
- Tucker, M.A.; Fox, B.M.; Seigler, N.; Rodriguez-Miguelez, P.; Looney, J.; Thomas, J.; McKie, K.T.; Forseen, C.; Davison, G.W.; Harris, R.A. Endothelial Dysfunction in Cystic Fibrosis: Role of Oxidative Stress. Oxid. Med. Cell. Longev. 2019, 2019, 1629638. [Google Scholar] [CrossRef] [PubMed]
- Lagrange-Puget, M.; Durieu, I.; Ecochard, R.; Abbas-Chorfa, F.; Drai, J.; Steghens, J.P.; Pacheco, Y.; Vital-Durand, D.; Bellon, G. Longitudinal study of oxidative status in 312 cystic fibrosis patients in stable state and during bronchial exacerbation. Pediatr. Pulmonol. 2004, 38, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Horsley, A.R.; Davies, J.C.; Gray, R.D.; Macleod, K.A.; Donovan, J.; Aziz, Z.A.; Bell, N.J.; Rainer, M.; Mt-Isa, S.; Voase, N.; et al. Changes in physiological, functional and structural markers of cystic fibrosis lung disease with treatment of a pulmonary exacerbation. Thorax 2013, 68, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Hakim, F.; Kerem, E.; Rivlin, J.; Bentur, L.; Stankiewicz, H.; Bdolach-Abram, T.; Wilschanski, M. Vitamins A and E and pulmonary exacerbations in patients with cystic fibrosis. J. Pediatr. Gastroenterol. Nutr. 2007, 45, 347–353. [Google Scholar] [CrossRef]
- Heltshe, S.L.; Goss, C.H. Optimising treatment of CF pulmonary exacerbation: A tough nut to crack. Thorax 2016, 71, 101–102. [Google Scholar] [CrossRef]
- Rowbotham, N.J.; Palser, S.C.; Smith, S.J.; Smyth, A.R. Infection prevention and control in cystic fibrosis: A systematic review of interventions. Expert Rev. Respir. Med. 2019, 13, 425–434. [Google Scholar] [CrossRef]
- Flume, P.A.; Mogayzel, P.J., Jr.; Robinson, K.A.; Goss, C.H.; Rosenblatt, R.L.; Kuhn, R.J.; Marshall, B.C.; Clinical Practice Guidelines for Pulmonary Therapies Committee. Cystic fibrosis pulmonary guidelines: Treatment of pulmonary exacerbations. Am. J. Respir. Crit. Care Med. 2009, 180, 802–808. [Google Scholar] [CrossRef]
- Teng, J.; Pourmand, A.; Mazer-Amirshahi, M. Vitamin C: The next step in sepsis management? J. Crit. Care. 2018, 43, 230–234. [Google Scholar] [CrossRef]
- Nabzdyk, C.S.; Bittner, E.A. Vitamin C in the critically ill—Indications and controversies. World J. Crit. Care Med. 2018, 7, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.J.; Ortiz-Reyes, A.; Dhaliwal, R.; Clarke, J.; Hill, A.; Stoppe, C.; Lee, Z.Y.; Heyland, D.K. IV Vitamin C in Critically Ill Patients: A Systematic Review and Meta-Analysis. Crit. Care Med. 2022, 50, e304–e312. [Google Scholar] [CrossRef]
- Rozemeijer, S.; van der Horst, F.A.L.; de Man, A.M.E. Measuring vitamin C in critically ill patients: Clinical importance and practical difficulties-Is it time for a surrogate marker? Crit. Care 2021, 25, 310. [Google Scholar] [CrossRef] [PubMed]
- Torphy, T.J.; Allen, J.; Cantin, A.M.; Konstan, M.W.; Accurso, F.J.; Joseloff, E.; Ratjen, F.A.; Chmiel, J.F.; Antiinflammatory Therapy Working, G. Considerations for the Conduct of Clinical Trials with Antiinflammatory Agents in Cystic Fibrosis. A Cystic Fibrosis Foundation Workshop Report. Ann. Am. Thorac Soc. 2015, 12, 1398–1406. [Google Scholar] [CrossRef]
- Xu, X.; Abdalla, T.; Bratcher, P.E.; Jackson, P.L.; Sabbatini, G.; Wells, J.M.; Lou, X.Y.; Quinn, R.; Blalock, J.E.; Clancy, J.P.; et al. Doxycycline improves clinical outcomes during cystic fibrosis exacerbations. Eur. Respir. J. 2017, 49, 1601102. [Google Scholar] [CrossRef]
- Hisert, K.B.; Heltshe, S.L.; Pope, C.; Jorth, P.; Wu, X.; Edwards, R.M.; Radey, M.; Accurso, F.J.; Wolter, D.J.; Cooke, G.; et al. Restoring Cystic Fibrosis Transmembrane Conductance Regulator Function Reduces Airway Bacteria and Inflammation in People with Cystic Fibrosis and Chronic Lung Infections. Am. J. Respir Crit Care Med. 2017, 195, 1617–1628. [Google Scholar] [CrossRef] [PubMed]
- Barry, P.J.; Flume, P.A. Bronchodilators in cystic fibrosis: A critical analysis. Expert Rev. Respir Med. 2017, 11, 13–20. [Google Scholar] [CrossRef]
- McCague, A.F.; Raraigh, K.S.; Pellicore, M.J.; Davis-Marcisak, E.F.; Evans, T.A.; Han, S.T.; Lu, Z.; Joynt, A.T.; Sharma, N.; Castellani, C.; et al. Correlating Cystic Fibrosis Transmembrane Conductance Regulator Function with Clinical Features to Inform Precision Treatment of Cystic Fibrosis. Am. J. Respir. Crit. Care Med. 2019, 199, 1116–1126. [Google Scholar] [CrossRef]
- Sun, T.; Sun, Z.; Jiang, Y.; Ferguson, A.A.; Pilewski, J.M.; Kolls, J.K.; Chen, W.; Chen, K. Transcriptomic Responses to Ivacaftor and Prediction of Ivacaftor Clinical Responsiveness. Am. J. Respir. Cell Mol. Biol. 2019, 61, 643–652. [Google Scholar] [CrossRef]
- Shanthikumar, S.; Ranganathan, S.; Neeland, M.R. Ivacaftor, not ivacaftor/lumacaftor, associated with lower pulmonary inflammation in preschool cystic fibrosis. Pediatr. Pulmonol. 2022. [Google Scholar] [CrossRef]
- Nichols, D.P.; Paynter, A.C.; Heltshe, S.L.; Donaldson, S.H.; Frederick, C.A.; Freedman, S.D.; Gelfond, D.; Hoffman, L.R.; Kelly, A.; Narkewicz, M.R.; et al. Clinical Effectiveness of Elexacaftor/Tezacaftor/Ivacaftor in People with Cystic Fibrosis: A Clinical Trial. Am. J. Respir. Crit. Care Med. 2022, 205, 529–539. [Google Scholar] [CrossRef]
- Veltman, M.; De Sanctis, J.B.; Stolarczyk, M.; Klymiuk, N.; Bahr, A.; Brouwer, R.W.; Oole, E.; Shah, J.; Ozdian, T.; Liao, J.; et al. CFTR Correctors and Antioxidants Partially Normalize Lipid Imbalance but not Abnormal Basal Inflammatory Cytokine Profile in CF Bronchial Epithelial Cells. Front. Physiol. 2021, 12, 619442. [Google Scholar] [CrossRef]
- Wang, Y.; Ma, B.; Li, W.; Li, P. Efficacy and Safety of Triple Combination Cystic Fibrosis Transmembrane Conductance Regulator Modulators in Patients With Cystic Fibrosis: A Meta-Analysis of Randomized Controlled Trials. Front. Pharmacol. 2022, 13, 863280. [Google Scholar] [CrossRef] [PubMed]


| # | Age (Years) | Sex | CF Genotype | BMI | Predicted FEV1 1 | CRP 2 (mg/dL) | WBC | Hb | HbA1C | Vit. A 3 (mg/L) | Vit. D 4 (ng/mL) | Sputum 5 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 28 | M | 508/508 | 20 | 38 | --- | 5000 | 14.2 | 5.1 | 0.37 | 20 | Pseudomonas |
| 2 | 32 | F | 508/508 | 20 | 34 | 2.0 | 8700 | 9.8 | 5.3 | 0.40 | 25 | Pseudomonas |
| 3 | 31 | M | 22 | 66 | 0.2 | 8000 | 12.6 | 5.4 | 0.72 | 24 | Pseudomonas/S. aureus | |
| 4 | 30 | F | 508/508 | 21 | 46 | 1.3 | 8000 | 12.7 | 6.5 | 0.34 | 29 | Pseudomonas/MRSA |
| 5 | 25 | F | 508/508 | 15 | 30 | 6.7 | 7700 | 12.8 | 6.6 | 0.20 | 34 | S. maltophilia |
| 6 | 23 | M | 20 | 47 | 0.6 | 15,600 | 12.4 | 6.1 | 0.30 | 49 | Pseudomonas/MRSA |
| Triglycerides (mmol/L) | Cholesterol (mmol/L) | Total Lipids (mmol/L) | α-Tocopherol (µmol/L) | α-T/Cholesterol (mmol/mol) | α-T/Lipids (mmol/mol) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| # | Baseline | Vitamin C | Baseline | Vitamin C | Baseline | Vitamin C | Baseline | Vitamin C | Baseline | Vitamin C | Baseline | Vitamin C |
| 1 | 0.48 | 0.42 | 2.61 | 2.39 | 3.09 | 2.82 | 6.99 | 5.03 | 2.66 | 2.11 | 2.25 | 1.80 |
| 2 | 0.41 | 0.41 | 3.62 | 3.30 | 4.03 | 3.72 | 17.91 | 16.17 | 4.96 | 4.89 | 4.45 | 4.35 |
| 3 | 0.93 | 0.87 | 3.86 | 3.40 | 4.79 | 4.27 | 20.73 | 19.08 | 5.39 | 5.61 | 4.34 | 4.47 |
| 4 | 0.32 | 0.34 | 4.34 | 3.78 | 4.65 | 4.13 | 13.41 | 15.16 | 3.09 | 4.03 | 2.88 | 3.69 |
| 5 | 0.28 | 0.27 | 2.21 | 2.15 | 2.49 | 2.42 | 7.18 | 9.51 | 3.26 | 4.42 | 2.89 | 3.93 |
| 6 | 0.35 | 0.28 | 2.50 | 2.68 | 2.86 | 2.95 | 8.61 | 7.72 | 3.47 | 2.89 | 3.05 | 2.62 |
| Intervention | ||||
|---|---|---|---|---|
| Plasma | Baseline | Vitamin C | Paired t-Test | |
| α-Tocopherol | Average concentration (µmol/L plasma) | 12.5 ± 5.9 | 12.1 ± 5.5 | NS |
| γ-Tocopherol | 0.52 ± 0.38 | 0.60 ± 0.51 | NS | |
| α-CEHC * | Average concentration (µmol/L plasma) | 0.12 ± 0.09 | 0.19 ±0.08 | NS |
| γ-CEHC | 0.47 ± 0.53 | 0.59 ± 0.80 | NS | |
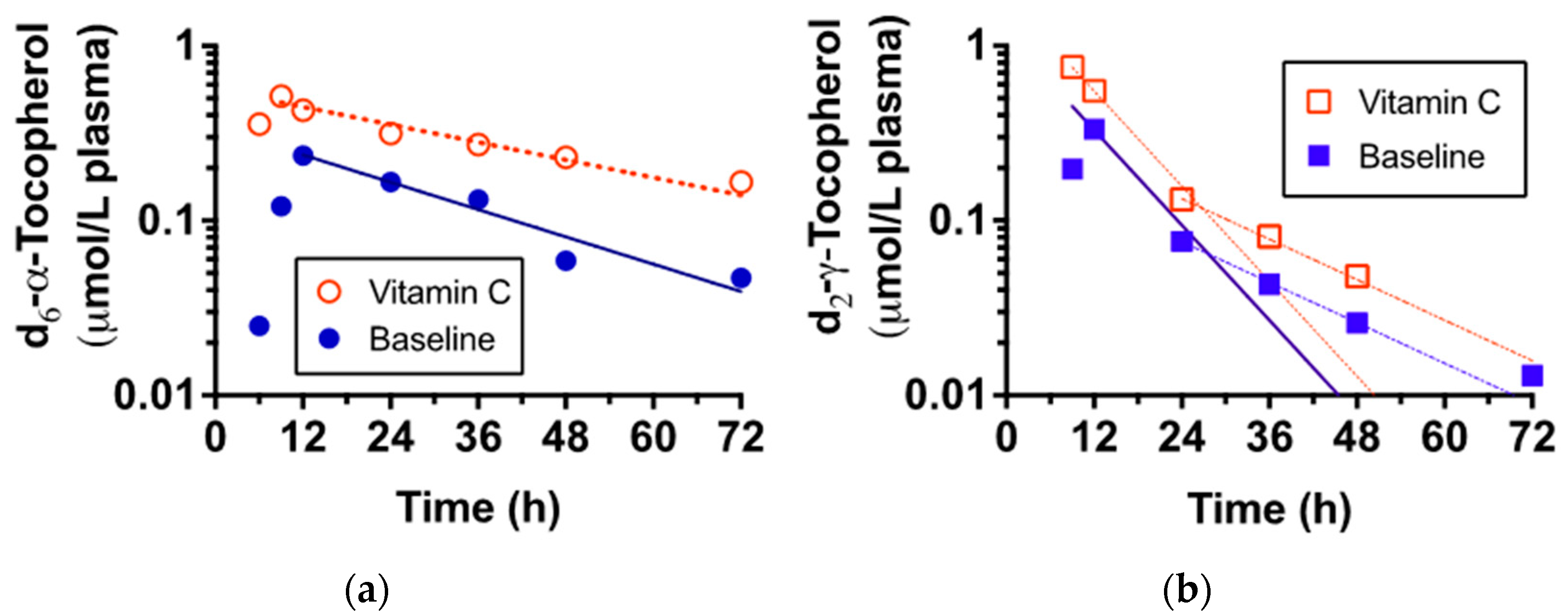
| d6-α-Tocopherol 1 | Cmax (µmol/L) | 0.27 ± 0.15 | 0.32 ± 0.13 | NS |
| Tmax (h) | 19.5 ± 7.0 | 17.5 ± 7.5 | NS | |
| Disappearance rate (pools per day) | 0.65 ± 0.14 | 0.50 ± 0.10 | p = 0.0263 | |
| AUC | 9.78 ± 5.57 | 12.04 ± 4.38 | NS | |
| d2-γ-Tocopherol | Cmax (µmol/L) | 0.20 ± 0.10 | 0.30 ± 0.22 | NS |
| Tmax (h) | 15.5 ± 6.7 | 11.5 ± 1.2 | NS | |
| Disappearance rate (pools per day) | 1.54 ± 0.50 | 1.42 ± 0.55 | NS | |
| AUC | 4.59 ± 3.14 | 5.66 ± 2.77 | NS | |
| d2-γ-CEHC n = 5 | Cmax (µmol/L) | 0.20 ± 0.14 | 0.22 ± 0.10 | NS |
| Tmax (h) | 16.2 ±7.2 | 12.0 ± 0 | NS | |
| Disappearance rate (pools per day) | 1.25 ± 0.72 | 1.61 ± 0.79 | NS | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Traber, M.G.; Leonard, S.W.; Vasu, V.T.; Morrissey, B.M.; Lei, H.; Atkinson, J.; Cross, C.E. α-Tocopherol Pharmacokinetics in Adults with Cystic Fibrosis: Benefits of Supplemental Vitamin C Administration. Nutrients 2022, 14, 3717. https://doi.org/10.3390/nu14183717
Traber MG, Leonard SW, Vasu VT, Morrissey BM, Lei H, Atkinson J, Cross CE. α-Tocopherol Pharmacokinetics in Adults with Cystic Fibrosis: Benefits of Supplemental Vitamin C Administration. Nutrients. 2022; 14(18):3717. https://doi.org/10.3390/nu14183717
Chicago/Turabian StyleTraber, Maret G., Scott W. Leonard, Vihas T. Vasu, Brian M. Morrissey, Huangshu (John) Lei, Jeffrey Atkinson, and Carroll E. Cross. 2022. "α-Tocopherol Pharmacokinetics in Adults with Cystic Fibrosis: Benefits of Supplemental Vitamin C Administration" Nutrients 14, no. 18: 3717. https://doi.org/10.3390/nu14183717
APA StyleTraber, M. G., Leonard, S. W., Vasu, V. T., Morrissey, B. M., Lei, H., Atkinson, J., & Cross, C. E. (2022). α-Tocopherol Pharmacokinetics in Adults with Cystic Fibrosis: Benefits of Supplemental Vitamin C Administration. Nutrients, 14(18), 3717. https://doi.org/10.3390/nu14183717







