Essential Amino Acid Ingestion Facilitates Leucine Retention and Attenuates Myofibrillar Protein Breakdown following Bodyweight Resistance Exercise in Young Adults in a Home-Based Setting
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
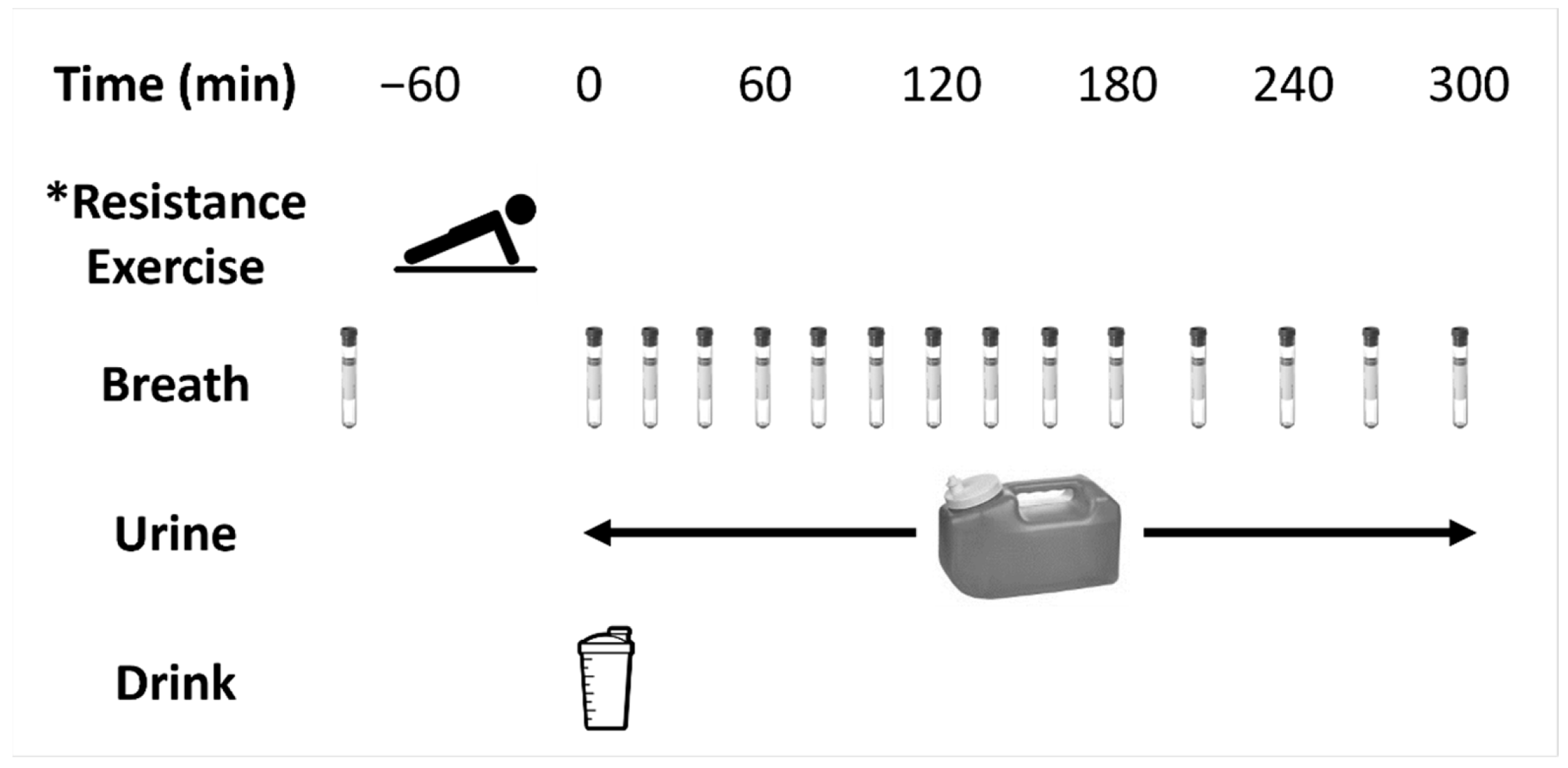
2.2. Metabolic Trials
2.3. Resistance Exercise Protocol
2.4. Trial Supplements
2.5. Analysis of Breath Samples
2.6. Analysis of Urine Samples
2.7. Calculations
2.8. Statistical Analysis
3. Results
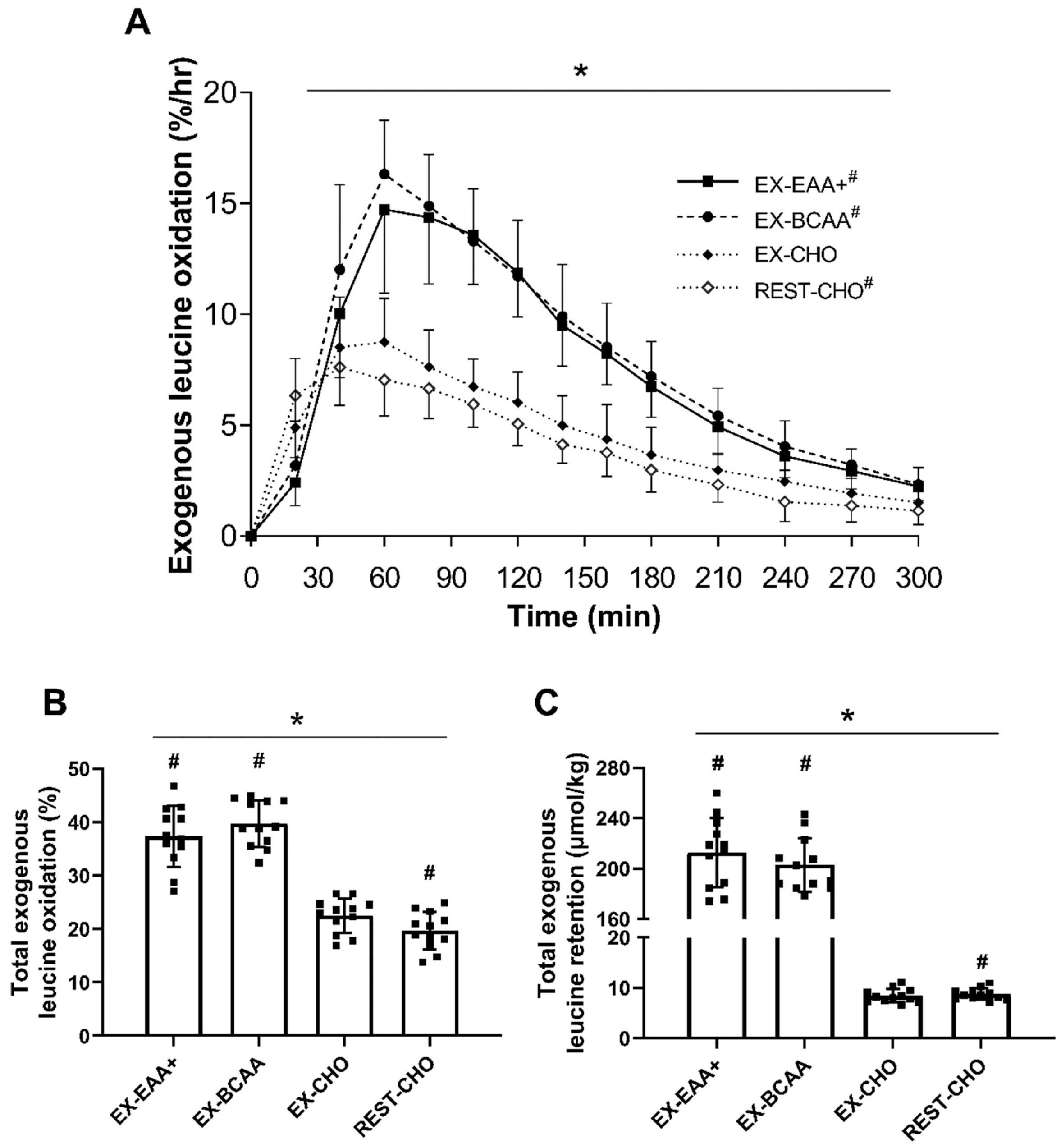
3.1. Relative Exogenous Leucine Oxidation
3.2. Absolute Exogenous Leucine Retention
3.3. Urinary 3-Methylhistidine:Creatinine
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wolfe, R.R. The underappreciated role of muscle in health and disease. Am. J. Clin. Nutr. 2006, 84, 475–482. [Google Scholar] [CrossRef]
- Phillips, S.M. Dietary protein requirements and adaptive advantages in athletes. Br. J. Nutr. 2012, 108, S158–S167. [Google Scholar] [CrossRef]
- Biolo, G. Protein metabolism and requirements. World Rev. Nutr. Diet. 2013, 105, 12–20. [Google Scholar] [CrossRef]
- Frontera, W.R.; Ochala, J. Skeletal muscle: A brief review of structure and function. Calcif. Tissue Int. 2015, 96, 183–195. [Google Scholar] [CrossRef]
- Mitchell, C.J.; Churchward-Venne, T.A.; West, D.W.D.; Burd, N.A.; Breen, L.; Baker, S.K.; Phillips, S.M. Resistance exercise load does not determine training-mediated hypertrophic gains in young men. J. Appl. Physiol. 2012, 113, 71–77. [Google Scholar] [CrossRef]
- Morton, R.W.; Oikawa, S.Y.; Wavell, C.G.; Mazara, N.; McGlory, C.; Quadrilatero, J.; Baechler, B.L.; Baker, S.K.; Phillips, S.M. Neither load nor systemic hormones determine resistance training-mediated hypertrophy or strength gains in resistance-trained young men. J. Appl. Physiol. 2016, 121, 129–138. [Google Scholar] [CrossRef]
- Jessee, M.B.; Buckner, S.L.; Mouser, J.G.; Mattocks, K.T.; Dankel, S.J.; Abe, T.; Bell, Z.W.; Bentley, J.P.; Loenneke, J.P. Muscle Adaptations to High-Load Training and Very Low-Load Training With and Without Blood Flow Restriction. Front. Physiol. 2018, 9, 1448. [Google Scholar] [CrossRef]
- Wagenmakers, A.J. Tracers to investigate protein and amino acid metabolism in human subjects. Proc. Nutr. Soc. 1999, 58, 987–1000. [Google Scholar] [CrossRef]
- Rennie, M.J.; Tipton, K.D. Protein and amino acid metabolism during and after exercise and the effects of nutrition. Annu. Rev. Nutr. 2000, 20, 457–483. [Google Scholar] [CrossRef]
- Phillips, S.M.; Tipton, K.D.; Aarsland, A.; Wolf, S.E.; Wolfe, R.R. Mixed muscle protein synthesis and breakdown after resistance exercise in humans. Am. J. Physiol. 1997, 273, E99–E107. [Google Scholar] [CrossRef]
- Thompson, W.R. Worldwide Survey of Fitness Trends for 2021. ACSMs Health Fit. J. 2021, 25, 10–19. [Google Scholar] [CrossRef]
- Malowany, J.M.; West, D.W.D.; Williamson, E.; Volterman, K.A.; Abou Sawan, S.; Mazzulla, M.; Moore, D.R. Protein to Maximize Whole-Body Anabolism in Resistance-trained Females after Exercise. Med. Sci. Sports Exerc. 2019, 51, 798–804. [Google Scholar] [CrossRef]
- Mazzulla, M.; Abou Sawan, S.; Williamson, E.; Hannaian, S.J.; Volterman, K.A.; West, D.W.D.; Moore, D.R. Protein Intake to Maximize Whole-Body Anabolism during Postexercise Recovery in Resistance-Trained Men with High Habitual Intakes is Severalfold Greater than the Current Recommended Dietary Allowance. J. Nutr. 2020, 150, 505–511. [Google Scholar] [CrossRef]
- Tinline-Goodfellow, C.T.; West, D.W.D.; Malowany, J.M.; Gillen, J.B.; Moore, D.R. An Acute Reduction in Habitual Protein Intake Attenuates Post Exercise Anabolism and May Bias Oxidation-Derived Protein Requirements in Resistance Trained Men. Front. Nutr. 2020, 7, 55. [Google Scholar] [CrossRef]
- Matthews, D.E. An overview of phenylalanine and tyrosine kinetics in humans. J. Nutr. 2007, 137, 1549S–1555S, discussion 1573S–1575S. [Google Scholar] [CrossRef]
- Boirie, Y.; Gachon, P.; Corny, S.; Fauquant, J.; Maubois, J.; Beaufrere, B. Acute postprandial changes in leucine metabolism as assessed with an intrinsically labeled milk protein. Am. J. Physiol. -Endocrinol. Metab. 1996, 271, E1083–E1091. [Google Scholar] [CrossRef]
- Areta, J.L.; Burke, L.M.; Ross, M.L.; Camera, D.M.; West, D.W.; Broad, E.M.; Jeacocke, N.A.; Moore, D.R.; Stellingwerff, T.; Phillips, S.M. Timing and distribution of protein ingestion during prolonged recovery from resistance exercise alters myofibrillar protein synthesis. J. Physiol. 2013, 591, 2319–2331. [Google Scholar] [CrossRef]
- Matthews, D.E.; Marano, M.A.; Campbell, R.G. Splanchnic bed utilization of leucine and phenylalanine in humans. Am. J. Physiol. -Endocrinol. Metab. 1993, 264, E109–E118. [Google Scholar] [CrossRef]
- Mazzulla, M.; Parel, J.T.; Beals, J.W.; Van Vliet, S.; Abou Sawan, S.; West, D.W.D.; Paluska, S.A.; Ulanov, A.V.; Moore, D.R.; Burd, N.A. Endurance Exercise Attenuates Postprandial Whole-Body Leucine Balance in Trained Men. Med. Sci. Sports Exerc. 2017, 49. [Google Scholar] [CrossRef]
- Mazzulla, M.; Hodson, N.; West, D.W.; Kumbhare, D.A.; Moore, D.R. A non-invasive 13CO2 breath test detects differences in anabolic sensitivity with feeding and heavy resistance exercise in healthy young males: A randomized control trial. Appl. Physiol. Nutr. Metab. 2022. [Google Scholar] [CrossRef]
- Neinast, M.D.; Jang, C.; Hui, S.; Murashige, D.S.; Chu, Q.; Morscher, R.J.; Li, X.; Zhan, L.; White, E.; Anthony, T.G.; et al. Quantitative Analysis of the Whole-Body Metabolic Fate of Branched-Chain Amino Acids. Cell Metab. 2019, 29, 417–429.e4. [Google Scholar] [CrossRef] [PubMed]
- Berg, J.M.; Tymoczko, J.L.; Stryer, L. Biochemistry; WH Freeman: New York, NY, USA, 2002. [Google Scholar]
- Moore, D.R.; Robinson, M.J.; Fry, J.L.; Tang, J.E.; Glover, E.I.; Wilkinson, S.B.; Prior, T.; Tarnopolsky, M.A.; Phillips, S.M. Ingested protein dose response of muscle and albumin protein synthesis after resistance exercise in young men. Am. J. Clin. Nutr. 2009, 89, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Evenepoel, P.; Hiele, M.; Luypaerts, A.; Geypens, B.; Buyse, J.; Decuypere, E.; Rutgeerts, P.; Ghoos, Y. Production of egg proteins, enriched with L-leucine-13C1, for the study of protein assimilation in humans using the breath test technique. J. Nutr. 1997, 127, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Nolles, J.; Verreijen, A.; Koopmanschap, R.; Verstegen, M.; Schreurs, V. Postprandial oxidative losses of free and protein-bound amino acids in the diet: Interactions and adaptation. J. Anim. Physiol. Anim. Nutr. 2009, 93, 431–438. [Google Scholar] [CrossRef]
- Wolfe, R.R. Branched-chain amino acids and muscle protein synthesis in humans: Myth or reality? J. Int. Soc. Sports Nutr. 2017, 14, 30. [Google Scholar] [CrossRef]
- Young, V.R.; Munro, H.N. Ntau-methylhistidine (3-methylhistidine) and muscle protein turnover: An overview. Fed. Proc. 1978, 37, 2291–2300. [Google Scholar]
- Long, C.L.; Haverberg, L.N.; Young, V.R.; Kinney, J.M.; Munro, H.N.; Geiger, J.W. Metabolism of 3-methylhistidine in man. Metab. Clin. Exp. 1975, 24, 929–935. [Google Scholar] [CrossRef]
- Elia, M.; Carter, A.; Bacon, S.; Winearls, C.G.; Smith, R. Clinical usefulness of urinary 3-methylhistidine excretion in indicating muscle protein breakdown. Br. Med. J. 1981, 282, 351–354. [Google Scholar] [CrossRef]
- Bird, S.P.; Tarpenning, K.M.; Marino, F.E. Independent and combined effects of liquid carbohydrate/essential amino acid ingestion on hormonal and muscular adaptations following resistance training in untrained men. Eur. J. Appl. Physiol. 2006, 97, 225–238. [Google Scholar] [CrossRef]
- Bird, S.P.; Tarpenning, K.M.; Marino, F.E. Liquid carbohydrate/essential amino acid ingestion during a short-term bout of resistance exercise suppresses myofibrillar protein degradation. Metab. Clin. Exp. 2006, 55, 570–577. [Google Scholar] [CrossRef]
- Thomas, S.; Reading, J.; Shephard, R.J. Revision of the Physical Activity Readiness Questionnaire (PAR-Q). Can. J. Sport Sci. J. Can. Des Sci. Du Sport 1992, 17, 338–345. [Google Scholar]
- Craig, C.L.; Marshall, A.L.; Sjöström, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borg, G. Borg’s Perceived Exertion and Pain Scales; Human Kinetics: Champaign, IL, USA, 1998. [Google Scholar]
- Schwedhelm, E.; Maas, R.; Freese, R.; Jung, D.; Lukacs, Z.; Jambrecina, A.; Spickler, W.; Schulze, F.; Böger, R.H. Pharmacokinetic and pharmacodynamic properties of oral L-citrulline and L-arginine: Impact on nitric oxide metabolism. Br. J. Clin. Pharm. 2008, 65, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Guisado, J.; Jakeman, P.M. Citrulline Malate Enhances Athletic Anaerobic Performance and Relieves Muscle Soreness. J. Strength Cond. Res. 2010, 24, 1215–1222. [Google Scholar] [CrossRef]
- Ham, D.J.; Gleeson, B.G.; Chee, A.; Baum, D.M.; Caldow, M.K.; Lynch, G.S.; Koopman, R. L-Citrulline Protects Skeletal Muscle Cells from Cachectic Stimuli through an iNOS-Dependent Mechanism. PLoS ONE 2015, 10, e0141572. [Google Scholar] [CrossRef]
- Ham, D.J.; Lynch, G.S.; Koopman, R. Amino acid sensing and activation of mechanistic target of rapamycin complex 1: Implications for skeletal muscle. Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 67–73. [Google Scholar] [CrossRef]
- Legault, Z.; Bagnall, N.; Kimmerly, D.S. The Influence of Oral L-Glutamine Supplementation on Muscle Strength Recovery and Soreness Following Unilateral Knee Extension Eccentric Exercise. Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 417–426. [Google Scholar] [CrossRef]
- Cho, S.; Hong, R.; Yim, P.; Yeom, M.; Lee, B.; Yang, W.M.; Hong, J.; Lee, H.S.; Hahm, D.H. An herbal formula consisting of Schisandra chinensis (Turcz.) Baill, Lycium chinense Mill and Eucommia ulmoides Oliv alleviates disuse muscle atrophy in rats. J. Ethnopharmacol. 2018, 213, 328–339. [Google Scholar] [CrossRef]
- Kim, C.H.; Shin, J.-H.; Hwang, S.J.; Choi, Y.H.; Kim, D.-S.; Kim, C.M. Schisandrae fructus enhances myogenic differentiation and inhibits atrophy through protein synthesis in human myotubes. Int. J. Nanomed. 2016, 11, 2407–2415. [Google Scholar] [CrossRef]
- Bucci, L.; Blackman, G.; Defoyd, W.; Kaufmann, R.; Mandel-Tayes, C.; Sparks, W.; Stiles, J.; Hickson, J. Effect of ferulate on strength and body composition of weightlifters. J. Appl. Sports Sci. Res. 1990, 4, 110. [Google Scholar]
- Neuhäuser, M.; Bässler, K.H. Endogenous 3-methylhistidine excretion in healthy women and men with reference to muscle protein metabolism. Z. Für Ernährungswissenschaft 1984, 23, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Sjölin, J.; Stjernström, H.; Henneberg, S.; Hambraeus, L.; Friman, G. Evaluation of urinary 3-methylhistidine excretion in infection by measurements of 1-methylhistidine and the creatinine ratios. Am. J. Clin. Nutr. 1989, 49, 62–70. [Google Scholar] [CrossRef] [PubMed]
- van Hall, G. Correction factors for 13C-labelled substrate oxidation at whole-body and muscle level. Proc. Nutr. Soc. 1999, 58, 979–986. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Péronnet, F.; Massicotte, D.; Brisson, G.; Hillaire-Marcel, C. Use of 13C substrates for metabolic studies in exercise: Methodological considerations. J. Appl. Physiol. 1990, 69, 1047–1052. [Google Scholar] [CrossRef]
- Hoerr, R.A.; Yu, Y.M.; Wagner, D.A.; Burke, J.F.; Young, V.R. Recovery of 13C in breath from NaH13CO3 infused by gut and vein: Effect of feeding. Am. J. Physiol. 1989, 257, E426–E438. [Google Scholar] [CrossRef]
- Reckman, G.A.R.; Navis, G.J.; Krijnen, W.P.; Vonk, R.J.; Jager-Wittenaar, H.; van der Schans, C.P. Aerobic exercise increases post-exercise exogenous protein oxidation in healthy young males. PLoS ONE 2019, 14, e0225803. [Google Scholar] [CrossRef]
- Shreeve, W.; Cerasi, E.; Luft, R. Metabolism of [2-14C] pyruvate in normal, acromegalic and HGH-treated human subjects. Eur. J. Endocrinol. 1970, 65, 155–169. [Google Scholar] [CrossRef]
- Haycock, G.B.; Schwartz, G.J.; Wisotsky, D.H. Geometric method for measuring body surface area: A height-weight formula validated in infants, children, and adults. J. Pediatr. 1978, 93, 62–66. [Google Scholar] [CrossRef]
- Wingfield, H.L.; Smith-Ryan, A.E.; Melvin, M.N.; Roelofs, E.J.; Trexler, E.T.; Hackney, A.C.; Weaver, M.A.; Ryan, E.D. The acute effect of exercise modality and nutrition manipulations on post-exercise resting energy expenditure and respiratory exchange ratio in women: A randomized trial. Sports Med.-Open 2015, 1, 11. [Google Scholar] [CrossRef]
- Pate, L. A Comparison of High-Intensity Interval Running and TABATA on Post-Exercise Metabolism: A Pilot Analysis. 2020. Available online: https://scholarworks.sfasu.edu/cgi/viewcontent.cgi?article=1369&context=urc (accessed on 29 March 2022).
- Ghosh, D.; Vogt, A. Outliers: An evaluation of methodologies. In Proceedings of the Joint Statistical Meetings, San Diego, CA, USA, 28 July–2 August 2012; pp. 3455–3460. [Google Scholar]
- Dangin, M.; Boirie, Y.; Garcia-Rodenas, C.; Gachon, P.; Fauquant, J.; Callier, P.; Ballèvre, O.; Beaufrère, B. The digestion rate of protein is an independent regulating factor of postprandial protein retention. Am. J. Physiol.-Endocrinol. Metab. 2001, 280, E340–E348. [Google Scholar] [CrossRef]
- Reckman, G.A.; Koehorst, M.; Priebe, M.; Schierbeek, H.; Vonk, R.J. 13C protein oxidation in breath: Is it relevant for the whole body protein status? J. Biomed. Sci. Eng. 2016, 9, 160–169. [Google Scholar] [CrossRef]
- Reckman, G.A.R.; Navis, G.J.; Krijnen, W.P.; van der Schans, C.P.; Vonk, R.J.; Jager-Wittenaar, H. Whole Body Protein Oxidation Unaffected after a Protein Restricted Diet in Healthy Young Males. Nutrients 2019, 11, 115. [Google Scholar] [CrossRef] [PubMed]
- Reckman, G.A.R.; Koehorst, M.; Schierbeek, H.; Vonk, R.J. Milk protein oxidation in healthy subjects: A preliminary study. Int. Dairy J. 2020, 111, 104826. [Google Scholar] [CrossRef]
- Bujko, J.; Schreurs, V.V.; Nolles, J.A.; Verreijen, A.M.; Koopmanschap, R.E.; Verstegen, M.W. Application of a [13CO2] breath test to study short-term amino acid catabolism during the postprandial phase of a meal. Br. J. Nutr. 2007, 97, 891–897. [Google Scholar] [CrossRef]
- Bowtell, J.L.; Leese, G.P.; Smith, K.; Watt, P.W.; Nevill, A.; Rooyackers, O.; Wagenmakers, A.J.; Rennie, M.J. Modulation of whole body protein metabolism, during and after exercise, by variation of dietary protein. J. Appl. Physiol. 1998, 85, 1744–1752. [Google Scholar] [CrossRef] [PubMed]
- Garlick, P.J. The Role of Leucine in the Regulation of Protein Metabolism. J. Nutr. 2005, 135, 1553S–1556S. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.J.; Chinkes, D.L.; Wolfe, R.R. Measurement of muscle protein fractional synthesis and breakdown rates from a pulse tracer injection. Am. J. Physiol. Endocrinol. Metab. 2002, 283, E753–E764. [Google Scholar] [CrossRef]
- Burd, N.A.; West, D.W.; Staples, A.W.; Atherton, P.J.; Baker, J.M.; Moore, D.R.; Holwerda, A.M.; Parise, G.; Rennie, M.J.; Baker, S.K. Low-load high volume resistance exercise stimulates muscle protein synthesis more than high-load low volume resistance exercise in young men. PLoS ONE 2010, 5, e12033. [Google Scholar] [CrossRef]
- Seaborne, R.A.; Sharples, A.P. The interplay between exercise metabolism, epigenetics, and skeletal muscle remodeling. Exerc. Sport Sci. Rev. 2020, 48, 188–200. [Google Scholar] [CrossRef]
- Brown, G.A.; Ray, M.W.; Abbey, B.M.; Shaw, B.S.; Shaw, I. Oxygen Consumption, Heart Rate, and Blood Lactate Responses to an Acute Bout of Plyometric Depth Jumps in College-Aged Men and Women. J. Strength Cond. Res. 2010, 24, 2475–2482. [Google Scholar] [CrossRef]
- Wolfe, R.R.; Goodenough, R.D.; Wolfe, M.H.; Royle, G.T.; Nadel, E.R. Isotopic analysis of leucine and urea metabolism in exercising humans. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1982, 52, 458–466. [Google Scholar] [CrossRef] [PubMed]
- Tarnopolsky, M. Protein requirements for endurance athletes. Eur. J. Sport Sci. 2004, 4, 1–15. [Google Scholar] [CrossRef]
- Schranner, D.; Kastenmüller, G.; Schönfelder, M.; Römisch-Margl, W.; Wackerhage, H. Metabolite Concentration Changes in Humans After a Bout of Exercise: A Systematic Review of Exercise Metabolomics Studies. Sports Med.-Open 2020, 6, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kato, H.; Suzuki, K.; Bannai, M.; Moore, D.R. Branched-Chain Amino Acids Are the Primary Limiting Amino Acids in the Diets of Endurance-Trained Men after a Bout of Prolonged Exercise. J. Nutr. 2018, 148, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Volterman, K.A.; West, D.W.D.; Suzuki, K.; Moore, D.R. Nutritionally non-essential amino acids are dispensable for whole-body protein synthesis after exercise in endurance athletes with an adequate essential amino acid intake. Amino Acids 2018, 50, 1679–1684. [Google Scholar] [CrossRef] [PubMed]
- Pasiakos, S.M.; Carbone, J.W. Assessment of skeletal muscle proteolysis and the regulatory response to nutrition and exercise. IUBMB Life 2014, 66, 478–484. [Google Scholar] [CrossRef]
- Tipton, K.D.; Hamilton, D.L.; Gallagher, I.J. Assessing the Role of Muscle Protein Breakdown in Response to Nutrition and Exercise in Humans. Sports Med. 2018, 48, 53–64. [Google Scholar] [CrossRef]
- Kuci, O.; Archambault, E.; Dodacki, A.; Nubret, E.; De Bandt, J.-P.; Cynober, L. Effect of citrulline on muscle protein turnover in an in vitro model of muscle catabolism. Nutrition 2020, 71, 110597. [Google Scholar] [CrossRef]
- Cruzat, V.; Macedo Rogero, M.; Noel Keane, K.; Curi, R.; Newsholme, P. Glutamine: Metabolism and Immune Function, Supplementation and Clinical Translation. Nutrients 2018, 10, 1564. [Google Scholar] [CrossRef]
- Zduńska, K.; Dana, A.; Kolodziejczak, A.; Rotsztejn, H. Antioxidant Properties of Ferulic Acid and Its Possible Application. Ski. Pharmacol. Physiol. 2018, 31, 332–336. [Google Scholar] [CrossRef]
- Guo, L.Y.; Hung, T.M.; Bae, K.H.; Shin, E.M.; Zhou, H.Y.; Hong, Y.N.; Kang, S.S.; Kim, H.P.; Kim, Y.S. Anti-inflammatory effects of schisandrin isolated from the fruit of Schisandra chinensis Baill. Eur. J. Pharmacol. 2008, 591, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.-M.; Chan, E.; Kwok, C.-Y.; Lee, Y.-K.; Wu, J.-H.; Wan, C.-W.; Chan, R.Y.-K.; Yu, P.H.-F.; Chan, S.-W. A review of the anticancer and immunomodulatory effects of Lycium barbarum fruit. Inflammopharmacology 2012, 20, 307–314. [Google Scholar] [CrossRef] [PubMed]

| Set | Exercise | Duration |
|---|---|---|
| Legs | Close stance squats | 1 min |
| Leaning Romanian deadlifts | 1 min | |
| Reverse lunges with kickback | 1 min | |
| Squat with calf jump | 1 min | |
| Lunge pulses | 1 min | |
| Chest | Side-to-side push-ups | 1 min |
| Decline push-ups | 1 min | |
| Reverse grip push-ups | 1 min | |
| Push-up holds | 1 min | |
| Pectoral crushers | 1 min | |
| Back | Reverse snow angels | 1 min |
| Seal push-ups | 1 min | |
| Pulse rows | 1 min | |
| Forward reaches | 1 min | |
| Supermans | 1 min | |
| Shoulders | Front twist raises | 1 min |
| Switch side pulses | 1 min | |
| Front air drivers | 1 min | |
| Reverse Arnolds | 1 min | |
| Bent triplexes | 1 min | |
| Abdominals | Rising flutters | 30 s |
| Abdominal rockers | 30 s | |
| Toe touchers | 30 s | |
| Penguin ankle taps | 30 s | |
| Legs extended crunches | 30 s | |
| Touch and go crunches | 30 s | |
| Hip thrusts | 30 s | |
| Bicycle crunches | 30 s | |
| Leg crunches | 30 s | |
| Buster crunches | 30 s | |
| Quadriceps | Explosive squat jumps | 1 min |
| Close to wide squat jumps | 1 min | |
| Speed squats | 1 min | |
| Squat pulses | 1 min | |
| Squat hold | 1 min |
| Variable | Mean ± SD |
|---|---|
| Age (years) | 26.92 ± 3.34 |
| Estimated height (cm) | 172.42 ± 8.37 |
| Estimated weight (kg) | 71.00 ± 9.06 |
| Body mass index (kg/m2) | 23.81 ± 2.40 |
| Habitual dietary protein (g/kg/day) | 1.76 ± 0.65 |
| Habitual dietary energy (kcal/day) | 2170.75 ± 599.70 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Waskiw-Ford, M.; Hodson, N.; Fung, H.J.W.; West, D.W.D.; Apong, P.; Bashir, R.; Moore, D.R. Essential Amino Acid Ingestion Facilitates Leucine Retention and Attenuates Myofibrillar Protein Breakdown following Bodyweight Resistance Exercise in Young Adults in a Home-Based Setting. Nutrients 2022, 14, 3532. https://doi.org/10.3390/nu14173532
Waskiw-Ford M, Hodson N, Fung HJW, West DWD, Apong P, Bashir R, Moore DR. Essential Amino Acid Ingestion Facilitates Leucine Retention and Attenuates Myofibrillar Protein Breakdown following Bodyweight Resistance Exercise in Young Adults in a Home-Based Setting. Nutrients. 2022; 14(17):3532. https://doi.org/10.3390/nu14173532
Chicago/Turabian StyleWaskiw-Ford, Marcus, Nathan Hodson, Hugo J. W. Fung, Daniel W. D. West, Philip Apong, Raza Bashir, and Daniel R. Moore. 2022. "Essential Amino Acid Ingestion Facilitates Leucine Retention and Attenuates Myofibrillar Protein Breakdown following Bodyweight Resistance Exercise in Young Adults in a Home-Based Setting" Nutrients 14, no. 17: 3532. https://doi.org/10.3390/nu14173532
APA StyleWaskiw-Ford, M., Hodson, N., Fung, H. J. W., West, D. W. D., Apong, P., Bashir, R., & Moore, D. R. (2022). Essential Amino Acid Ingestion Facilitates Leucine Retention and Attenuates Myofibrillar Protein Breakdown following Bodyweight Resistance Exercise in Young Adults in a Home-Based Setting. Nutrients, 14(17), 3532. https://doi.org/10.3390/nu14173532






