Alcohol Consumption Patterns and Risk of Early-Onset Vasomotor Symptoms in Premenopausal Women
Abstract
1. Introduction
2. Methods
2.1. Study Population
2.2. Data Collection
2.3. Definition of Early-Onset VMS
2.4. Definition of Alcohol Consumption
2.5. Statistical Analysis
3. Results
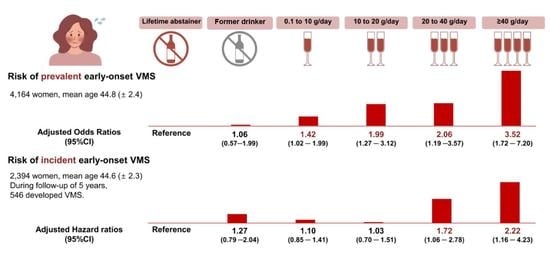
3.1. Alcohol Consumption and Prevalent Early-Onset VMS
3.2. Alcohol Consumption and Incidence of Early-Onset VMS
3.3. Effect Modification by Alcohol Flushing Status
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thurston, R.C.; Joffe, H. Vasomotor symptoms and menopause: Findings from the Study of Women’s Health across the Nation. Obstet. Gynecol. Clin. 2011, 38, 489–501. [Google Scholar] [CrossRef] [PubMed]
- Kronenberg, F. Hot flashes: Epidemiology and physiology. Ann. N. Y. Acad. Sci. 1990, 592, 52–86; discussion 123–133. [Google Scholar] [CrossRef] [PubMed]
- Avis, N.E.; Crawford, S.L.; Greendale, G.; Bromberger, J.T.; Everson-Rose, S.A.; Gold, E.B.; Hess, R.; Joffe, H.; Kravitz, H.M.; Tepper, P.G.; et al. Duration of Menopausal Vasomotor Symptoms Over the Menopause Transition. JAMA Intern. Med. 2015, 175, 531. [Google Scholar] [CrossRef] [PubMed]
- Thurston, R.C.; El Khoudary, S.R.; Tepper, P.G.; Jackson, E.A.; Joffe, H.; Chen, H.-Y.; Matthews, K.A.; Harlow, S.; Sowers, M. Trajectories of vasomotor symptoms and carotid intima media thickness in the Study of Women’s Health Across the Nation. Stroke 2016, 47, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Chung, H.F.; Dobson, A.J.; Pandeya, N.; Anderson, D.J.; Kuh, D.; Hardy, R.; Brunner, E.J.; Avis, N.E.; Gold, E.B.; et al. Vasomotor menopausal symptoms and risk of cardiovascular disease: A pooled analysis of six prospective studies. Am. J. Obstet. Gynecol. 2020, 223, 898.e1–898.e16. [Google Scholar] [CrossRef]
- Griswold, M.G.; Fullman, N.; Hawley, C.; Arian, N.; Zimsen, S.R.; Tymeson, H.D.; Venkateswaran, V.; Tapp, A.D.; Forouzanfar, M.H.; Salama, J.S. Alcohol use and burden for 195 countries and territories, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2018, 392, 1015–1035. [Google Scholar] [CrossRef]
- Erol, A.; Ho, A.M.C.; Winham, S.J.; Karpyak, V.M. Sex hormones in alcohol consumption: A systematic review of evidence. Addict. Biol. 2019, 24, 157–169. [Google Scholar] [CrossRef]
- Mendelson, J.H.; Lukas, S.E.; Mello, N.K.; Amass, L.; Ellingboe, J.; Skupny, A. Acute alcohol effects on plasma estradiol levels in women. Psychopharmacology 1988, 94, 464–467. [Google Scholar] [CrossRef]
- Sarkola, T.; Fukunaga, T.; Mäkisalo, H.; Peter Eriksson, C. Acute effect of alcohol on androgens in premenopausal women. Alcohol Alcohol. 2000, 35, 84–90. [Google Scholar] [CrossRef]
- Frias, J.; Torres, J.; Miranda, M.; Ruiz, E.; Ortega, E. Effects of acute alcohol intoxication on pituitary–gonadal axis hormones, pituitary–adrenal axis hormones, β-endorphin and prolactin in human adults of both sexes. Alcohol Alcohol. 2002, 37, 169–173. [Google Scholar] [CrossRef]
- Li, N.; Fu, S.; Zhu, F.; Deng, X.; Shi, X. Alcohol intake induces diminished ovarian reserve in childbearing age women. J. Obstet. Gynaecol. Res. 2013, 39, 516–521. [Google Scholar] [CrossRef] [PubMed]
- Moilanen, J.; Aalto, A.M.; Hemminki, E.; Aro, A.R.; Raitanen, J.; Luoto, R. Prevalence of menopause symptoms and their association with lifestyle among Finnish middle-aged women. Maturitas 2010, 67, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Freeman, E.W.; Sammel, M.D.; Grisso, J.A.; Battistini, M.; Garcia-Espagna, B.; Hollander, L. Hot flashes in the late reproductive years: Risk factors for African American and Caucasian women. J. Women’s Health Gend. Based Med. 2001, 10, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Schwingl, P.J.; Hulka, B.S.; Harlow, S.D. Risk factors for menopausal hot flashes. Obstet. Gynecol. 1994, 84, 29–34. [Google Scholar] [PubMed]
- Gold, E.B.; Block, G.; Crawford, S.; Lachance, L.; FitzGerald, G.; Miracle, H.; Sherman, S. Lifestyle and demographic factors in relation to vasomotor symptoms: Baseline results from the Study of Women’s Health Across the Nation. Am. J. Epidemiol. 2004, 159, 1189–1199. [Google Scholar] [CrossRef] [PubMed]
- Gallicchio, L.; Miller, S.R.; Kiefer, J.; Greene, T.; Zacur, H.A.; Flaws, J.A. Risk factors for hot flashes among women undergoing the menopausal transition: Baseline results from the Midlife Women’s Health Study. Menopause 2015, 22, 1098–1107. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.L.; Gallicchio, L.; Miller, S.R.; Zacur, H.A.; Flaws, J.A. Risk factors for extended duration and timing of peak severity of hot flashes. PLoS ONE 2016, 11, e0155079. [Google Scholar] [CrossRef]
- Chang, Y.; Ryu, S.; Sung, K.-C.; Cho, Y.K.; Sung, E.; Kim, H.-N.; Jung, H.-S.; Yun, K.E.; Ahn, J.; Shin, H. Alcoholic and non-alcoholic fatty liver disease and associations with coronary artery calcification: Evidence from the Kangbuk Samsung Health Study. Gut 2019, 68, 1667–1675. [Google Scholar] [CrossRef]
- Han, S.Y.; Chang, Y.; Kim, Y.; Choi, C.Y.; Ryu, S. A Dose–Response Relationship of Alcohol Consumption with Risk of Visual Impairment in Korean Adults: The Kangbuk Samsung Health Study. Nutrients 2022, 14, 791. [Google Scholar] [CrossRef]
- Choi, H.R.; Chang, Y.; Kim, Y.; Cho, Y.; Kang, J.; Kwon, M.J.; Kwon, R.; Lim, G.; Kim, K.H.; Kim, H.; et al. Ideal cardiovascular health metrics and risk of incident early-onset vasomotor symptoms among premenopausal women. J. Clin. Endocrinol. Metab. 2022, dgac327. [Google Scholar] [CrossRef]
- Namgoung, S.; Chang, Y.; Woo, C.Y.; Kim, Y.; Kang, J.; Kwon, R.; Lim, G.; Choi, H.R.; Kim, K.H.; Kim, H.; et al. Metabolically healthy and unhealthy obesity and risk of vasomotor symptoms in premenopausal women: Cross-sectional and cohort studies. BJOG Int. J. Obstet. Gynaecol. 2022. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. The Asia-Pacific Perspective: Redefining Obesity and Its Treatment; World Health Organization: Geneva, Switzerland, 2000. [Google Scholar]
- Craig, C.L.; Marshall, A.L.; Sjöström, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [PubMed]
- Forde, C. Scoring the International Physical Activity Questionnaire (IPAQ); University of Dublin: Dublin, Ireland, 2018; Volume 3. [Google Scholar]
- Cho, I.Y.; Chang, Y.; Kang, J.H.; Kim, Y.; Sung, E.; Shin, H.; Wild, S.H.; Byrne, C.D.; Ryu, S. Long or Irregular Menstrual Cycles and Risk of Prevalent and Incident Nonalcoholic Fatty Liver Disease. J. Clin. Endocrinol. Metab. 2022, 107, e2309–e2317. [Google Scholar] [CrossRef] [PubMed]
- Park, J.-H.; Bae, S.H.; Jung, Y.-M. Validity and Reliability of the Korean Version of the Menopause-Specific Quality of Life. J. Korean Acad. Nurs. 2020, 50, 487–500. [Google Scholar] [CrossRef] [PubMed]
- Nappi, R.E.; Kroll, R.; Siddiqui, E.; Stoykova, B.; Rea, C.; Gemmen, E.; Schultz, N.M. Global cross-sectional survey of women with vasomotor symptoms associated with menopause: Prevalence and quality of life burden. Menopause 2021, 28, 875. [Google Scholar] [CrossRef]
- Whiteley, J.; Wagner, J.-S.; Bushmakin, A.; Kopenhafer, L.; DiBonaventura, M.; Racketa, J. Impact of the severity of vasomotor symptoms on health status, resource use, and productivity. Menopause 2013, 20, 518–524. [Google Scholar] [CrossRef]
- Saunders, J.B.; Aasland, O.G.; Babor, T.F.; De la Fuente, J.R.; Grant, M. Development of the alcohol use disorders identification test (AUDIT): WHO collaborative project on early detection of persons with harmful alcohol consumption-II. Addiction 1993, 88, 791–804. [Google Scholar] [CrossRef]
- Kim, K.; Chang, Y.; Ahn, J.; Yang, H.-J.; Ryu, S. Low levels of alcohol consumption and risk of intestinal metaplasia: A cohort study. Cancer Epidemiol. Prev. Biomark. 2020, 29, 2633–2641. [Google Scholar] [CrossRef]
- Anderson, D.J.; Chung, H.-F.; Seib, C.A.; Dobson, A.J.; Kuh, D.; Brunner, E.J.; Crawford, S.L.; Avis, N.E.; Gold, E.B.; Greendale, G.A. Obesity, smoking, and risk of vasomotor menopausal symptoms: A pooled analysis of eight cohort studies. Am. J. Obstet. Gynecol. 2020, 222, 478.e1–478.e17. [Google Scholar] [CrossRef]
- Melby, M.K.; Anderson, D.; Sievert, L.L.; Obermeyer, C.M. Methods used in cross-cultural comparisons of vasomotor symptoms and their determinants. Maturitas 2011, 70, 110–119. [Google Scholar] [CrossRef]
- Herber-Gast, G.-C.M.; Mishra, G.D.; van der Schouw, Y.T.; Brown, W.J.; Dobson, A.J. Risk factors for night sweats and hot flushes in midlife: Results from a prospective cohort study. Menopause 2013, 20, 953–959. [Google Scholar] [CrossRef] [PubMed]
- Royston, P.; Parmar, M.K. Flexible parametric proportional-hazards and proportional-odds models for censored survival data, with application to prognostic modelling and estimation of treatment effects. Stat. Med. 2002, 21, 2175–2197. [Google Scholar] [CrossRef] [PubMed]
- Heeb, J.-L.; Gmel, G. Measuring alcohol consumption: A comparison of graduated frequency, quantity frequency, and weekly recall diary methods in a general population survey. Addict. Behav. 2005, 30, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Hyde Riley, E.; Inui, T.S.; Kleinman, K.; Connelly, M.T. Differential association of modifiable health behaviors with hot flashes in perimenopausal and postmenopausal women. J. Gen. Intern. Med. 2004, 19, 740–746. [Google Scholar] [CrossRef] [PubMed]
- Herber-Gast, G.; Brown, W.J.; Mishra, G.D. Hot flushes and night sweats are associated with coronary heart disease risk in midlife: A longitudinal study. BJOG Int. J. Obstet. Gynaecol. 2015, 122, 1560–1567. [Google Scholar] [CrossRef] [PubMed]
- Gast, G.C.; Pop, V.J.; Samsioe, G.N.; Grobbee, D.E.; Nilsson, P.M.; Keyzer, J.J.; Wijnands-van Gent, C.J.; van der Schouw, Y.T. Vasomotor menopausal symptoms are associated with increased risk of coronary heart disease. Menopause 2011, 18, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, S.; Peeters, P.; Bezemer, I.; Dossus, L.; Biessy, C.; Sacerdote, C.; Berrino, F.; Panico, S.; Palli, D.; Tumino, R. Relationship of alcohol intake and sex steroid concentrations in blood in pre-and post-menopausal women: The European Prospective Investigation into Cancer and Nutrition. Cancer Causes Control 2006, 17, 1033–1043. [Google Scholar] [CrossRef]
- Ginsburg, E.S.; Walsh, B.W.; Shea, B.F.; Gao, X.; Gleason, R.E.; Barbieri, R.L. The effects of ethanol on the clearance of estradiol in postmenopausal women. Fertil. Steril. 1995, 63, 1227–1230. [Google Scholar] [CrossRef]
- Randolph, J.F., Jr.; Sowers, M.; Bondarenko, I.; Gold, E.B.; Greendale, G.A.; Bromberger, J.T.; Brockwell, S.E.; Matthews, K.A. The relationship of longitudinal change in reproductive hormones and vasomotor symptoms during the menopausal transition. J. Clin. Endocrinol. Metab. 2005, 90, 6106–6112. [Google Scholar] [CrossRef]
- Schilling, C.; Gallicchio, L.; Miller, S.R.; Langenberg, P.; Zacur, H.; Flaws, J.A. Genetic polymorphisms, hormone levels, and hot flashes in midlife women. Maturitas 2007, 57, 120–131. [Google Scholar] [CrossRef][Green Version]
- Yoda, T.; Crawshaw, L.I.; Nakamura, M.; Saito, K.; Konishi, A.; Nagashima, K.; Uchida, S.; Kanosue, K. Effects of alcohol on thermoregulation during mild heat exposure in humans. Alcohol 2005, 36, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Kronenberg, F.; Downey, J.A. Thermoregulatory physiology of menopausal hot flashes: A review. Can. J. Physiol. Pharmacol. 1987, 65, 1312–1324. [Google Scholar] [CrossRef] [PubMed]
- Jones, H.; Bailey, T.G.; Barr, D.A.; France, M.; Lucas, R.A.; Crandall, C.G.; Low, D.A. Is core temperature the trigger of a menopausal hot flush? Menopause 2019, 26, 1016–1023. [Google Scholar] [CrossRef] [PubMed]
- Pachman, D.R.; Jones, J.M.; Loprinzi, C.L. Management of menopause-associated vasomotor symptoms: Current treatment options, challenges and future directions. Int. J. Women’s Health 2010, 2, 123. [Google Scholar]
- Berendsen, H.H. The role of serotonin in hot flushes. Maturitas 2000, 36, 155–164. [Google Scholar] [CrossRef]
- Stockwell, T.; Zhao, J.; Sherk, A.; Rehm, J.; Shield, K.; Naimi, T. Underestimation of alcohol consumption in cohort studies and implications for alcohol’s contribution to the global burden of disease. Addiction 2018, 113, 2245–2249. [Google Scholar] [CrossRef]

| Characteristics | Total | Drinking Status | |||||
|---|---|---|---|---|---|---|---|
| Lifetime Abstainer | Former Drinker | 0 to <10 g/day | 10 to <20 g/day | 20 to <40 g/day | ≥40 g/day | ||
| Number (%) | 4164 | 569 (13.7) | 172 (4.1) | 2879 (69.1) | 328 (7.9) | 160 (3.8) | 56 (1.3) |
| Age (years) * | 44.8 ± 2.4 | 45.6 ± 2.6 | 45.3 ± 2.8 | 44.6 ± 2.3 | 44.6 ± 2.4 | 44.8 ± 2.5 | 44.7 ± 2.4 |
| Age at menarche (years) * | 13.9 ± 1.4 | 14.0 ± 1.4 | 13.9 ± 1.5 | 13.9 ± 1.4 | 14.0 ± 1.5 | 14.1 ± 1.6 | 14.0 ± 1.5 |
| Obesity (%) a | 16.2 | 16.3 | 20.9 | 15.7 | 16.5 | 18.8 | 19.6 |
| High physical activity (%) b | 14.8 | 13.5 | 17.4 | 14.1 | 18.3 | 19.4 | 21.4 |
| Current smoker (%) | 1.9 | 0.7 | 0.6 | 1.2 | 3.7 | 11.4 | 17.9 |
| High education (%) c | 80.8 | 80.9 | 78.4 | 82.8 | 73.7 | 66.9 | 66.1 |
| Hypertension (%) d | 4.5 | 5.3 | 3.5 | 3.7 | 6.7 | 11.3 | 10.7 |
| Systolic BP (mmHg) * | 103.9 ± 11.7 | 104.2 ± 11.8 | 104.1 ± 12.0 | 103.2 ± 11.4 | 105.5 ± 11.4 | 109.0 ± 13.9 | 109.8 ± 13.3 |
| Diastolic BP (mmHg) * | 66.8 ± 9.2 | 66.3 ± 8.8 | 65.5 ± 9.6 | 66.5 ± 8.9 | 68.5 ± 9.7 | 71.6 ± 11.0 | 72.4 ± 11.3 |
| Diabetes (%) e | 1.7 | 1.2 | 0.6 | 1.6 | 2.1 | 3.8 | 1.8 |
| Glucose (mg/dL) * | 93.0 ± 12.0 | 92.4 ± 10.4 | 94.0 ± 20.9 | 92.7 ± 11.7 | 94.0 ± 8.8 | 96.1 ± 15.7 | 93.2 ± 15.7 |
| LDLC (mg/dL) * | 119.2 ± 28.7 | 123.5 ± 29.0 | 120.2 ± 27.7 | 118.7 ± 28.1 | 119.8 ± 30.6 | 115.5 ± 33.0 | 108.2 ± 30.5 |
| HDLC (mg/dL) * | 67.0 ± 15.9 | 65.0 ± 15.4 | 66.0 ± 15.1 | 66.6 ± 15.6 | 69.3 ± 16.3 | 74.1 ± 18.8 | 77.0 ± 20.4 |
| Triglycerides (mg/dL) † | 74.0 (57.0–100.0) | 77.0 (58.0–105.0) | 73.5 (56.0–100.0) | 73.0 (57.0–98.0) | 78.5 (57.0–106.0) | 78.5 (62.0–105.0) | 77.0 (63.5–109.5) |
| AST (U/l) † | 18.0 (15.0–20.0) | 18.0 (15.0–20.0) | 18.0 (16.0–21.0) | 17.0 (15.0–20.0) | 18.0 (15.0–21.0) | 18.0 (16.0–21.0) | 18.0 (16.0–20.0) |
| ALT (U/l) † | 13.0 (11.0–17.0) | 14.0 (11.0–17.0) | 14.5 (12.0–19.0) | 13.0 (11.0–17.0) | 13.0 (11.0–17.0) | 13.0 (11.0–17.0) | 13.0 (10.5–15.5) |
| GGT (U/l) † | 13.0 (11.0–18.0) | 13.0 (10.0–17.0) | 13.0 (11.0–16.0) | 13.0 (11.0–18.0) | 15.0 (12.0–21.0) | 17.0 (13.0–21.0) | 18.0 (14.0–30.0) |
| HOMA-IR † | 1.1 (0.7–1.6) | 1.1 (0.7–1.6) | 1.1 (0.8–1.9) | 1.1 (0.7–1.6) | 1.2 (0.8–1.6) | 1.0 (0.7–1.4) | 0.9 (0.6–1.2) |
| hsCRP (mg/L) † | 0.03 (0.02–0.06) | 0.03 (0.02–0.06) | 0.03 (0.02–0.07) | 0.03 (0.02–0.06) | 0.03 (0.02–0.05) | 0.04 (0.02–0.06) | 0.03 (0.02–0.05) |
| Alcohol Consumption Patterns | Vasomotor Symptoms | Hot Flash Symptoms | Night Sweat Symptoms | |||
|---|---|---|---|---|---|---|
| Age-Adjusted OR (95% CI) | Multivariable-Adjusted OR * (95% CI) | Age-Adjusted OR (95% CI) | Multivariable-Adjusted OR * (95% CI) | Age-Adjusted OR (95% CI) | Multivariable-Adjusted OR * (95% CI) | |
| Drinking status | ||||||
| Lifetime abstainer | Reference | Reference | Reference | Reference | Reference | Reference |
| Current drinker | ||||||
| 0.1 to <10 g/day | 1.41 (1.01–1.97) | 1.42 (1.02–1.99) | 1.30 (0.91–1.84) | 1.31 (0.92–1.86) | 1.48 (0.85–2.58) | 1.50 (0.86–2.63) |
| 10 to <20 g/day | 2.05 (1.32–3.19) | 1.99 (1.27–3.12) | 1.83 (1.14–2.92) | 1.73 (1.07–2.78) | 2.82 (1.43–5.58) | 2.82 (1.42–5.63) |
| 20 to <40 g/day | 2.24 (1.32–3.83) | 2.06 (1.19–3.57) | 1.33 (0.70–2.51) | 1.17 (0.61–2.25) | 4.82 (2.34–9.93) | 4.51 (2.15–9.48) |
| ≥40 g/day | 3.89 (1.94–7.80) | 3.52 (1.72–7.20) | 2.32 (1.03–5.25) | 2.04 (0.88–4.72) | 8.98 (3.80–21.26) | 8.30 (3.40–20.28) |
| p trend | <0.01 | <0.01 | 0.01 | 0.06 | <0.01 | <0.01 |
| Former drinker | 1.08 (0.58–2.03) | 1.06 (0.56–1.99) | 1.10 (0.57–2.11) | 1.06 (0.55–2.05) | 1.62 (0.65–4.04) | 1.59 (0.63–3.99) |
| Frequency of drinking (days/week) | ||||||
| 0 | Reference | Reference | Reference | Reference | Reference | Reference |
| 1–2 | 1.43 (1.05–1.95) | 1.44 (1.05–1.96) | 1.31 (0.95–1.81) | 1.31 (0.95–1.82) | 1.54 (0.94–2.52) | 1.54 (0.94–2.53) |
| ≥3 | 2.15 (1.41–3.27) | 2.10 (1.37–3.22) | 1.63 (1.03–2.59) | 1.59 (0.99–2.54) | 3.23 (1.75–5.96) | 3.08 (1.64–5.75) |
| p trend | <0.01 | <0.01 | 0.03 | 0.04 | <0.01 | <0.01 |
| Number of drinks drinking day | ||||||
| 0 | Reference | Reference | Reference | Reference | Reference | Reference |
| 1–2 | 1.37 (0.99–1.89) | 1.39 (1.01–1.92) | 1.28 (0.91–1.80) | 1.31 (0.93–1.84) | 1.29 (0.77–2.15) | 1.29 (0.77–2.17) |
| 3–5 | 1.77 (1.24–2.52) | 1.74 (1.21–2.49) | 1.57 (1.08–2.28) | 1.53 (1.05–2.23) | 2.14 (1.24–3.68) | 2.11 (1.22–5.65) |
| ≥6 | 2.40 (1.54–3.76) | 2.14 (1.34–3.38) | 1.75 (1.06–2.87) | 1.53 (0.92–2.54) | 3.50 (1.85–6.62) | 3.01 (1.56–5.80) |
| p trend | <0.01 | <0.01 | <0.01 | 0.03 | <0.01 | <0.01 |
| Alcohol Drinking Patterns | Vasomotor Symptoms | Hot Flash Symptoms | Night Sweat Symptoms | |||
|---|---|---|---|---|---|---|
| Age-Adjusted HRs (95% CI) | Multivariable-Adjusted HR (95% CI) * | Age-Adjusted HRs (95% CI) | Multivariable-Adjusted HR (95% CI) * | Age-Adjusted HRs (95% CI) | Multivariable-Adjusted HR (95% CI) * | |
| Drinking status | ||||||
| Lifetime abstainer | Reference | Reference | Reference | Reference | Reference | Reference |
| Current drinker | ||||||
| 0.1 to <10 g/day | 1.09 (0.85–1.41) | 1.10 (0.85–1.41) | 0.99 (0.74–1.34) | 1.00 (0.75–1.35) | 1.21 (0.89–1.64) | 1.21 (0.89–1.64) |
| 10 to <20 g/day | 1.09 (0.74–1.59) | 1.03 (0.70–1.51) | 0.86 (0.53–1.39) | 0.82 (0.51–1.35) | 1.19 (0.75–1.88) | 1.12 (0.71–1.77) |
| 20 to <40 g/day | 1.74 (1.09–2.79) | 1.72 (1.06–2.78) | 1.34 (0.74–2.41) | 1.28 (0.70–2.34) | 2.05 (1.19–3.52) | 1.95 (1.12–3.40) |
| ≥40 g/day | 2.23 (1.18–4.24) | 2.22 (1.16–4.23) | 1.30 (0.52–3.26) | 1.32 (0.52–3.32) | 3.20 (1.61–6.34) | 3.12 (1.56–6.23) |
| p trend | <0.01 | 0.02 | 0.59 | 0.70 | <0.01 | <0.01 |
| Former drinker | 1.29 (0.80–2.05) | 1.27 (0.79–2.04) | 1.09 (0.62–1.94) | 1.08 (0.61–1.91) | 1.17 (0.65–2.12) | 1.15 (0.63–2.09) |
| Frequency of drinking (days/week) | ||||||
| 0 | Reference | Reference | Reference | Reference | Reference | Reference |
| 1–2 | 1.09 (0.86–1.38) | 1.08 (0.85–1.37) | 0.99 (0.74–1.31) | 1.00 (0.75–1.32) | 1.19 (0.89–1.59) | 1.18 (0.89–1.57) |
| ≥3 | 1.50 (1.05–2.13) | 1.48 (1.03–2.12) | 1.30 (0.84–1.99) | 1.30 (0.84–2.02) | 1.65 (1.09–2.51) | 1.61 (1.06–2.46) |
| p trend | 0.05 | 0.06 | 0.39 | 0.37 | 0.03 | 0.04 |
| Number of drinks a drinking day | ||||||
| 0 | Reference | Reference | Reference | Reference | Reference | Reference |
| 1–2 | 1.04 (0.82–1.33) | 1.04 (0.81–1.33) | 0.95 (0.72–1.27) | 0.96 (0.72–1.28) | 1.15 (0.86–1.55) | 1.14 (0.85–1.54) |
| 3–5 | 1.21 (0.91–1.61) | 1.20 (0.90–1.59) | 0.98 (0.70–1.40) | 0.98 (0.70–1.38) | 1.43 (1.02–2.00) | 1.41 (1.00–1.97) |
| ≥6 | 1.55 (1.05–2.28) | 1.47 (1.00–2.17) | 1.37 (0.86–2.17) | 1.31 (0.82–2.09) | 1.79 (1.15–2.80) | 1.66 (1.05–2.62) |
| p trend | 0.01 | 0.03 | 0.34 | 0.45 | <0.01 | <0.01 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwon, R.; Chang, Y.; Kim, Y.; Cho, Y.; Choi, H.R.; Lim, G.-Y.; Kang, J.; Kim, K.-H.; Kim, H.; Hong, Y.S.; et al. Alcohol Consumption Patterns and Risk of Early-Onset Vasomotor Symptoms in Premenopausal Women. Nutrients 2022, 14, 2276. https://doi.org/10.3390/nu14112276
Kwon R, Chang Y, Kim Y, Cho Y, Choi HR, Lim G-Y, Kang J, Kim K-H, Kim H, Hong YS, et al. Alcohol Consumption Patterns and Risk of Early-Onset Vasomotor Symptoms in Premenopausal Women. Nutrients. 2022; 14(11):2276. https://doi.org/10.3390/nu14112276
Chicago/Turabian StyleKwon, Ria, Yoosoo Chang, Yejin Kim, Yoosun Cho, Hye Rin Choi, Ga-Young Lim, Jeonggyu Kang, Kye-Hyun Kim, Hoon Kim, Yun Soo Hong, and et al. 2022. "Alcohol Consumption Patterns and Risk of Early-Onset Vasomotor Symptoms in Premenopausal Women" Nutrients 14, no. 11: 2276. https://doi.org/10.3390/nu14112276
APA StyleKwon, R., Chang, Y., Kim, Y., Cho, Y., Choi, H. R., Lim, G.-Y., Kang, J., Kim, K.-H., Kim, H., Hong, Y. S., Park, J., Zhao, D., Rampal, S., Cho, J., Guallar, E., Park, H.-Y., & Ryu, S. (2022). Alcohol Consumption Patterns and Risk of Early-Onset Vasomotor Symptoms in Premenopausal Women. Nutrients, 14(11), 2276. https://doi.org/10.3390/nu14112276