Arsenic Exposure through Dietary Intake and Associated Health Hazards in the Middle East
Abstract
:1. Introduction
2. Facts of Arsenic Exposure
2.1. Arsenic Ingestion through Water Ecosystem
2.2. Arsenic Ingestion through Dietary Contaminant Food
3. Arsenic Cellular Metabolism
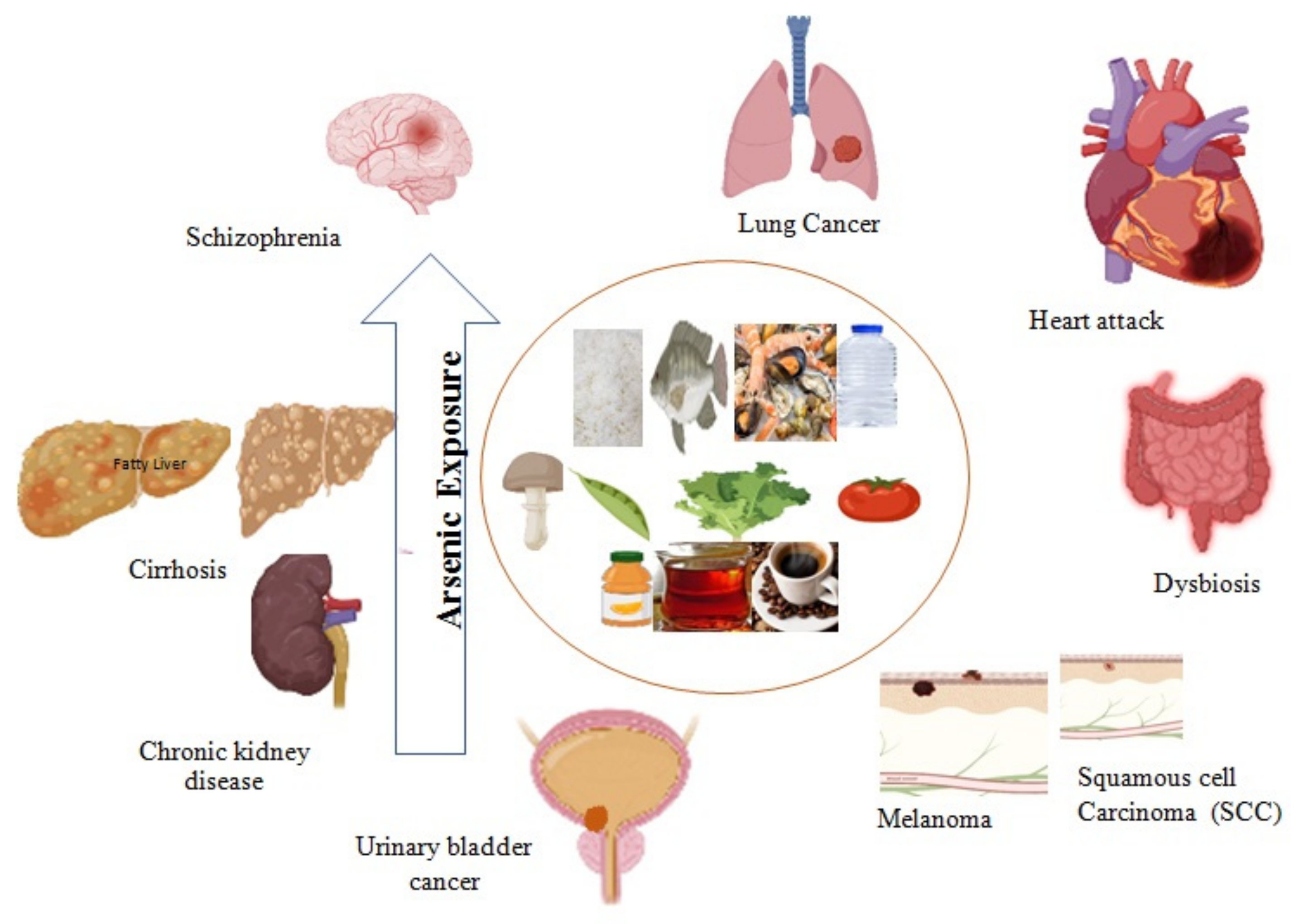
4. Arsenic-induced Health Hazards
4.1. Major Organ Damage and Chronic Disease Development
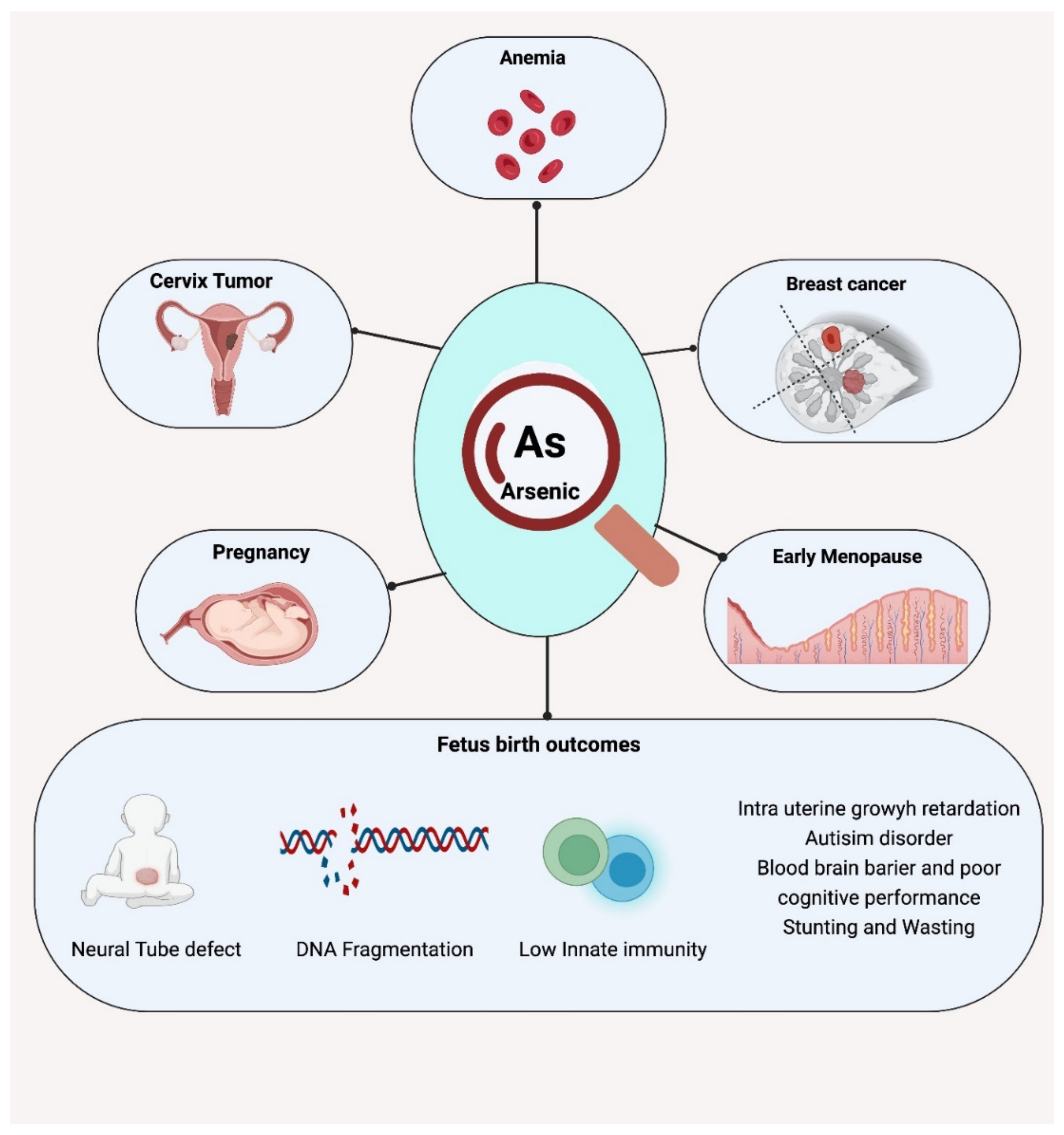
4.2. Effects on Maternal Health
4.3. Effects on Fetal and Neonatal Health
5. Arsenic Screening
6. Food Safety and Policy Interventions
7. Conclusions and Recommendation
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ezemonye, L.I.; Adebayo, P.O.; Enuneku, A.A.; Tongo, I.; Ogbomida, E. Potential health risk consequences of heavy metal concentrations in surface water, shrimp (Macrobrachium macrobrachion) and fish (Brycinus longipinnis) from Benin River, Nigeria. Toxicol. Rep. 2019, 6, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hill-Briggs, F.; Adler, N.E.; Berkowitz, S.A.; Chin, M.H.; Gary-Webb, T.L.; Navas-Acien, A.; Thornton, P.L.; Haire-Joshu, D. Social determinants of health and diabetes: A scientific review. Diabetes Care 2021, 44, 258–279. [Google Scholar] [CrossRef] [PubMed]
- Authority, E.F.S.; Arcella, D.; Cascio, C.; Gómez Ruiz, J.Á. Chronic dietary exposure to inorganic arsenic. EFSA J. 2021, 19, e06380. [Google Scholar]
- Chou, C.-H.; Harper, C. Toxicological Profile for Arsenic. 2007. Available online: https://hero.epa.gov/hero/index.cfm/reference/details/reference_id/657856 (accessed on 1 March 2022).
- ATSDR (Agency for Toxic Substances and Disease Registry). Prepared by Clement International Corp., under contract 2000, 205, 88-0608.
- Wang, J.; Hu, W.; Yang, H.; Chen, F.; Shu, Y.; Zhang, G.; Liu, J.; Liu, Y.; Li, H.; Guo, L. Arsenic concentrations, diversity and co-occurrence patterns of bacterial and fungal communities in the feces of mice under sub-chronic arsenic exposure through food. Environ. Int. 2020, 138, 105600. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Hirano, S. Metabolism of arsenic and its toxicological relevance. Arch. Toxicol. 2013, 87, 969–979. [Google Scholar] [CrossRef] [PubMed]
- Eckstein, M.; Eleazer, R.; Rea, M.; Fondufe-Mittendorf, Y. Epigenomic reprogramming in inorganic arsenic-mediated gene expression patterns during carcinogenesis. Rev. Environ. Health 2017, 32, 93–103. [Google Scholar] [CrossRef] [Green Version]
- Alexander, J.; Benford, D.; Boobis, A.; Ceccatelli, S.; Cravedi, J.; di Domenico, A.; Doerge, D.; Dogliotti, E.; Edler, L.; Farmer, P.; et al. Scientific Opinion on marine biotoxins in shellfish—Palytoxin group. EFSA J. 2009, 7, 1393. [Google Scholar]
- Signes-Pastor, A.J.; Woodside, J.V.; McMullan, P.; Mullan, K.; Carey, M.; Karagas, M.R.; Meharg, A.A. Levels of infants’ urinary arsenic metabolites related to formula feeding and weaning with rice products exceeding the EU inorganic arsenic standard. PLoS ONE 2017, 12, e0176923. [Google Scholar] [CrossRef]
- Molin, M.; Ulven, S.M.; Meltzer, H.M.; Alexander, J. Arsenic in the human food chain, biotransformation and toxicology–Review focusing on seafood arsenic. J. Trace Elem. Med. Biol. 2015, 31, 249–259. [Google Scholar] [CrossRef] [Green Version]
- Yoshinaga, J.; Narukawa, T. Dietary intake and urinary excretion of methylated arsenicals of Japanese adults consuming marine foods and rice. Food Addit. Contam. Part A 2021, 38, 622–629. [Google Scholar] [CrossRef]
- Tibon, J.; Silva, M.; Sloth, J.J.; Amlund, H.; Sele, V. Speciation analysis of organoarsenic species in marine samples: Method optimization using fractional factorial design and method validation. Anal. Bioanal. Chem. 2021, 413, 3909–3923. [Google Scholar] [CrossRef] [PubMed]
- Taylor, V.F.; Jackson, B.P. Concentrations and speciation of arsenic in New England seaweed species harvested for food and agriculture. Chemosphere 2016, 163, 6–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiong, C.; Stiboller, M.; Glabonjat, R.A.; Rieger, J.; Paton, L.; Francesconi, K.A. Transport of arsenolipids to the milk of a nursing mother after consuming salmon fish. J. Trace Elem. Med. Biol. 2020, 61, 126502. [Google Scholar] [CrossRef] [PubMed]
- Bencko, V.; Foong, F.Y.L. The history of arsenical pesticides and health risks related to the use of Agent Blue. Ann. Agric. Environ. Med. 2017, 24, 312–316. [Google Scholar] [CrossRef]
- Gilbert-Diamond, D.; Cottingham, K.L.; Gruber, J.F.; Punshon, T.; Sayarath, V.; Gandolfi, A.J.; Baker, E.R.; Jackson, B.P.; Folt, C.L.; Karagas, M.R. Rice consumption contributes to arsenic exposure in US women. Proc. Natl. Acad. Sci. USA 2011, 108, 20656–20660. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, M.K.; Shaheen, N.; Islam, M.S.; Habibullah-Al-Mamun, M.; Islam, S.; Islam, M.M.; Kundu, G.K.; Bhattacharjee, L. A comprehensive assessment of arsenic in commonly consumed foodstuffs to evaluate the potential health risk in Bangladesh. Sci. Total Environ. 2016, 544, 125–133. [Google Scholar] [CrossRef]
- Kurzius-Spencer, M.; O’rourke, M.K.; Hsu, C.-H.; Hartz, V.; Harris, R.B.; Burgess, J.L. Measured versus modeled dietary arsenic and relation to urinary arsenic excretion and total exposure. J. Expo. Sci. Environ. Epidemiol. 2013, 23, 442–449. [Google Scholar] [CrossRef]
- Guillod-Magnin, R.; Brüschweiler, B.J.; Aubert, R.; Haldimann, M. Arsenic species in rice and rice-based products consumed by toddlers in Switzerland. Food Addit. Contam. Part A 2018, 35, 1164–1178. [Google Scholar] [CrossRef] [Green Version]
- Yamauchi, H.; Takata, A. Arsenic metabolism differs between child and adult patients during acute arsenic poisoning. Toxicol. Appl. Pharmacol. 2021, 410, 115352. [Google Scholar] [CrossRef]
- Shibata, T.; Meng, C.; Umoren, J.; West, H. Risk assessment of arsenic in rice cereal and other dietary sources for infants and toddlers in the US. Int. J. Environ. Res. Public Health 2016, 13, 361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cubadda, F.; Jackson, B.P.; Cottingham, K.L.; Van Horne, Y.O.; Kurzius-Spencer, M. Human exposure to dietary inorganic arsenic and other arsenic species: State of knowledge, gaps and uncertainties. Sci. Total Environ. 2017, 579, 1228–1239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hughes, M.F.; Beck, B.D.; Chen, Y.; Lewis, A.S.; Thomas, D.J. Arsenic exposure and toxicology: A historical perspective. Toxicol. Sci. 2011, 123, 305–332. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- IARC. Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC: Lyon, France, 2012; Volume 100, p. 11. [Google Scholar]
- Lee, C.-H.; Yu, C.-L.; Liao, W.-T.; Kao, Y.-H.; Chai, C.-Y.; Chen, G.-S.; Yu, H.-S. Effects and interactions of low doses of arsenic and UVB on keratinocyte apoptosis. Chem. Res. Toxicol. 2004, 17, 1199–1205. [Google Scholar] [CrossRef]
- Chen, W.-J.; Davis, E.M.; Stoner, J.A.; Robledo, C.; Goodman, J.R.; Garwe, T.; Janitz, A.E.; Xu, C.; Hwang, J.; Peck, J.D. Urinary total arsenic and arsenic methylation capacity in pregnancy and gestational diabetes mellitus: A case-control study. Chemosphere 2021, 271, 129828. [Google Scholar] [CrossRef]
- Sarkar, M.; Pal, S.C. Human health hazard assessment for high groundwater arsenic and fluoride intact in Malda district, Eastern India. Groundw. Sustain. Dev. 2021, 13, 100565. [Google Scholar] [CrossRef]
- Mahmoud, M.T.; Hamouda, M.A.; Al Kendi, R.R.; Mohamed, M.M. Health risk assessment of household drinking water in a district in the UAE. Water 2018, 10, 1726. [Google Scholar] [CrossRef] [Green Version]
- Ali, A.; Chidambaram, S. Assessment of trace inorganic contaminates in water and sediment to address its impact on common fish varieties along Kuwait Bay. Environ. Geochem. Health 2021, 43, 855–883. [Google Scholar] [CrossRef]
- Okati, N.; Moghadam, M.S.; Einollahipeer, F. Mercury, arsenic and selenium concentrations in marine fish species from the Oman Sea, Iran, and health risk assessment. Toxicol. Environ. Health Sci. 2021, 13, 25–36. [Google Scholar] [CrossRef]
- Sabarathinam, C.; Bhandary, H.; Al-Khalid, A. A geochemical analogy between the metal sources in Kuwait Bay and territorial sea water of Kuwait. Environ. Monit. Assess. 2019, 191, 1–19. [Google Scholar] [CrossRef]
- Molamohyeddin, N.; Ghafourian, H.; Sadatipour, S.M. Contamination assessment of mercury, lead, cadmium and arsenic in surface sediments of Chabahar Bay. Mar. Pollut. Bull. 2017, 124, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Al-Kalbani, M.S.; Price, M.F.; Ahmed, M.; Abahussain, A.; O’Higgins, T.; Argyll, U. Water quality assessment of Aflaj in the Mountains of Oman. Environ. Nat. Resour. Res. 2016, 6, 99. [Google Scholar] [CrossRef] [Green Version]
- Al-Farraj, A.S.; Al-Wabel, M.I.; El-Saeid, M.H.; El-Naggar, A.H.; Ahmed, Z. Evaluation of groundwater for arsenic contamination using hydrogeochemical properties and multivariate statistical methods in Saudi Arabia. J. Chem. 2013, 2013, 812365. [Google Scholar] [CrossRef] [Green Version]
- Pakzad, D.; Akbari, V.; Sepand, M.R.; Aliomrani, M. Risk of neurodegenerative disease due to tau phosphorylation changes and arsenic exposure via drinking water. Toxicol. Res. 2021, 10, 325–333. [Google Scholar] [CrossRef]
- Al-Forkan, M.; Wali, F.B.; Khaleda, L.; Alam, M.J.; Chowdhury, R.H.; Datta, A.; Rahman, M.Z.; Hosain, N.; Maruf, M.F.; Chowdhury, M.A.Q. Association of arsenic-induced cardiovascular disease susceptibility with genetic polymorphisms. Sci. Rep. 2021, 11, 6263. [Google Scholar] [CrossRef]
- Ghrefat, H.; El Waheidi, M.; Batayneh, A.; Nazzal, Y.; Zumlot, T.; Mogren, S. Pollution assessment of arsenic and other selected elements in the groundwater and soil of the Gulf of Aqaba, Saudi Arabia. Environ. Earth Sci. 2016, 75, 229. [Google Scholar] [CrossRef]
- Bu-Olayan, A.; Thomas, B. Arsenic levels in the marine ecosystem off the Kuwait coast, Arabian Gulf. Environmentalist 2001, 21, 71–75. [Google Scholar] [CrossRef]
- Al-Taani, A.A.; Batayneh, A.; Nazzal, Y.; Ghrefat, H.; Elawadi, E.; Zaman, H. Status of trace metals in surface seawater of the Gulf of Aqaba, Saudi Arabia. Mar. Pollut. Bull. 2014, 86, 582–590. [Google Scholar] [CrossRef]
- De Mora, S.; Fowler, S.W.; Wyse, E.; Azemard, S. Distribution of heavy metals in marine bivalves, fish and coastal sediments in the Gulf and Gulf of Oman. Mar. Pollut. Bull. 2004, 49, 410–424. [Google Scholar] [CrossRef]
- Agah, H.; Bastami, K.D.; Fumani, N.S. Ecological risk, source and preliminary assessment of metals in the surface sediments of Chabahar Bay, Oman Sea. Mar. Pollut. Bull. 2016, 107, 383–388. [Google Scholar] [CrossRef]
- El-Sorogy, A.S.; Youssef, M.; Al-Kahtany, K.; Al-Otaiby, N. Assessment of arsenic in coastal sediments, seawaters and molluscs in the Tarut Island, Arabian Gulf, Saudi Arabia. J. Afr. Earth Sci. 2016, 113, 65–72. [Google Scholar] [CrossRef]
- Youssef, M.; El-Sorogy, A.; Al Kahtany, K.; Al Otiaby, N. Environmental assessment of coastal surface sediments at Tarut Island, Arabian Gulf (Saudi Arabia). Mar. Pollut. Bull. 2015, 96, 424–433. [Google Scholar] [CrossRef] [PubMed]
- El Maghraby, M.M.; El Nasr, A.K.O.A.; Hamouda, M.S. Quality assessment of groundwater at south Al Madinah Al Munawarah area, Saudi Arabia. Environ. Earth Sci. 2013, 70, 1525–1538. [Google Scholar] [CrossRef]
- Juma, H.A.; Al-Madany, I.M. Concentration of heavy metals in the territorial sea water of the Kingdom of Bahrain, Arabian Gulf. Arab. Gulf J. Sci. Res. 2008, 26, 19–32. [Google Scholar]
- Al-Awadi, E.; Mukhopadhyay, A.; Akber, A.; Hadi, K. Distribution of selected trace constituents in the ground water of Kuwait. Adv. Environ. Res. 2003, 7, 367–380. [Google Scholar] [CrossRef]
- Gu, Z.; de Silva, S.; Reichman, S.M. Arsenic concentrations and dietary exposure in rice-based infant food in Australia. Int. J. Environ. Res. Public Health 2020, 17, 415. [Google Scholar] [CrossRef] [Green Version]
- Hernández-Martínez, R.; Navarro-Blasco, I. Survey of total mercury and arsenic content in infant cereals marketed in Spain and estimated dietary intake. Food Control 2013, 30, 423–432. [Google Scholar] [CrossRef] [Green Version]
- Juskelis, R.; Li, W.; Nelson, J.; Cappozzo, J.C. Arsenic speciation in rice cereals for infants. J. Agric. Food Chem. 2013, 61, 10670–10676. [Google Scholar] [CrossRef]
- Shraim, A.M. Rice is a potential dietary source of not only arsenic but also other toxic elements like lead and chromium. Arab. J. Chem. 2017, 10, S3434–S3443. [Google Scholar] [CrossRef] [Green Version]
- Rashid, M.H.; Rahman, M.M.; Correll, R.; Naidu, R. Arsenic and other elemental concentrations in mushrooms from Bangladesh: Health risks. Int. J. Environ. Res. Public Health 2018, 15, 919. [Google Scholar] [CrossRef] [Green Version]
- Shaheen, N.; Irfan, N.M.; Khan, I.N.; Islam, S.; Islam, M.S.; Ahmed, M.K. Presence of heavy metals in fruits and vegetables: Health risk implications in Bangladesh. Chemosphere 2016, 152, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Edirisinghe, E.; Jinadasa, B. Arsenic and cadmium concentrations in legumes and cereals grown in the North Central Province, Sri Lanka and assessment of their health risk. Int. J. Food Contam. 2019, 6, 3. [Google Scholar] [CrossRef]
- Kowalska, G.; Pankiewicz, U.; Kowalski, R. Determination of the level of selected elements in canned meat and fish and risk assessment for consumer health. J. Anal. Methods Chem. 2020, 2020, 2148794. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szkoda, J. Arsenic in food of animal origin-exposure assessment. Ochr. Sr. I Zasobów Nat. 2009, 41, 128–134. [Google Scholar]
- Ashraf, M.W.; Mian, A. Levels of mercury and arsenic contamination in popular fish and shrimp brands consumed in Saudi Arabia. Bull. Chem. Soc. Ethiop. 2019, 33, 573–578. [Google Scholar] [CrossRef]
- Krishnakumar, P.K.; Qurban, M.A.; Stiboller, M.; Nachman, K.E.; Joydas, T.V.; Manikandan, K.P.; Mushir, S.A.; Francesconi, K.A. Arsenic and arsenic species in shellfish and finfish from the western Arabian Gulf and consumer health risk assessment. Sci. Total Environ. 2016, 566, 1235–1244. [Google Scholar] [CrossRef]
- Simsek, O.; Gültekin, R.; Öksüz, O.; Kurultay, S. The effect of environmental pollution on the heavy metal content of raw milk. Food 2000, 44, 360–363. [Google Scholar] [CrossRef]
- Gebeyehu, H.R.; Bayissa, L.D. Levels of heavy metals in soil and vegetables and associated health risks in Mojo area, Ethiopia. PLoS ONE 2020, 15, e0227883. [Google Scholar] [CrossRef] [Green Version]
- Massadeh, A.M.; Allah, A.; Al-Massaedh, T.; Kharibeh, S. Determination of selected elements in canned food sold in Jordan markets. Environ. Sci. Pollut. Res. 2018, 25, 3501–3509. [Google Scholar] [CrossRef]
- Salama, K.F.; Randhawa, M.A.; Al Mulla, A.A.; Labib, O.A. Heavy metals in some date palm fruit cultivars in Saudi Arabia and their health risk assessment. Int. J. Food Prop. 2019, 22, 1684–1692. [Google Scholar] [CrossRef] [Green Version]
- Maduabuchi, J.-M.; Adigba, E.; Nzegwu, C.; Oragwu, C.; Okonkwo, I.; Orisakwe, O.E. Arsenic and chromium in canned and non-canned beverages in Nigeria: A potential public health concern. Int. J. Environ. Res. Public Health 2007, 4, 28–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fathabad, A.E.; Shariatifar, N.; Moazzen, M.; Nazmara, S.; Fakhri, Y.; Alimohammadi, M.; Azari, A.; Khaneghah, A.M. Determination of heavy metal content of processed fruit products from Tehran’s market using ICP-OES: A risk assessment study. Food Chem. Toxicol. 2018, 115, 436–446. [Google Scholar] [CrossRef]
- Savić, S.R.; Petrović, S.M.; Stamenković, J.J.; Petronijević, Ž.B. The presence of minerals in clear orange juices. Adv. Technol. 2015, 4, 71–78. [Google Scholar] [CrossRef] [Green Version]
- Bazeyad, A.Y.; Al-Sarar, A.S.; Rushdi, A.I.; Hassanin, A.S.; Abobakr, Y. Levels of heavy metals in a multifloral Saudi honey. Environ. Sci. Pollut. Res. 2019, 26, 3946–3953. [Google Scholar] [CrossRef] [PubMed]
- Schwalfenberg, G.; Genuis, S.J.; Rodushkin, I. The benefits and risks of consuming brewed tea: Beware of toxic element contamination. J. Toxicol. 2013, 2013, 370460. [Google Scholar] [CrossRef] [Green Version]
- Al Nouri, D.; Al Abdulkarim, B.; Arzoo, S.; Bakeet, Z. Quality characteristics of commonly consumed drinking water in Riyadh and effect of domestic treatments on its chemical constituents. J. Food Nutr. Res. 2014, 2, 25–33. [Google Scholar]
- Shraim, A.; Alsuhaimi, A.; Al-Muzaini, K.; Kurdi, K.; Al-Ameen, H. Quality assessment of groundwater of Almadinah Almunawarah City. Global NEST J. 2013, 15, 374–383. [Google Scholar]
- Stanton, B.A.; Caldwell, K.; Congdon, C.B.; Disney, J.; Donahue, M.; Ferguson, E.; Flemings, E.; Golden, M.; Guerinot, M.L.; Highman, J. MDI Biological Laboratory arsenic summit: Approaches to limiting human exposure to arsenic. Curr. Environ. Health Rep. 2015, 2, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Authority, E. Dietary exposure to inorganic arsenic in the European population. EFSA J. 2014, 8, 36–37. [Google Scholar]
- Lynch, H.N.; Greenberg, G.I.; Pollock, M.C.; Lewis, A.S. A comprehensive evaluation of inorganic arsenic in food and considerations for dietary intake analyses. Sci. Total Environ. 2014, 496, 299–313. [Google Scholar] [CrossRef]
- Lu, Y.; Dong, F.; Deacon, C.; Chen, H.-j.; Raab, A.; Meharg, A.A. Arsenic accumulation and phosphorus status in two rice (Oryza sativa L.) cultivars surveyed from fields in South China. Environ. Pollut. 2010, 158, 1536–1541. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.F. Ganoderma lucidum: A rational pharmacological approach to surmount the cancer. J. Ethnopharmacol. 2020, 260, 113047. [Google Scholar] [CrossRef]
- Ahmad, M.F. Ganoderma lucidum: Persuasive biologically active constituents and their health endorsement. Biomed. Pharmacother. 2018, 107, 507–519. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.F. Ganoderma lucidum: A Macro Fungus with Phytochemicals and Their Pharmacological Properties. In Plant and Human Health; Springer: Berlin, Germany, 2019; Volume 2, pp. 491–515. [Google Scholar]
- Wahab, S.; Alshahrani, M.Y.; Ahmad, M.F.; Abbas, H. Current trends and future perspectives of nanomedicine for the management of colon cancer. Eur. J. Pharmacol. 2021, 910, 174464. [Google Scholar] [CrossRef] [PubMed]
- Sattar, A.; Xie, S.; Hafeez, M.A.; Wang, X.; Hussain, H.I.; Iqbal, Z.; Pan, Y.; Iqbal, M.; Shabbir, M.A.; Yuan, Z. Metabolism and toxicity of arsenicals in mammals. Environ. Toxicol. Pharmacol. 2016, 48, 214–224. [Google Scholar] [CrossRef]
- Wang, D.; Shimoda, Y.; Wang, S.; Wang, Z.; Liu, J.; Liu, X.; Jin, H.; Gao, F.; Tong, J.; Yamanaka, K. Total arsenic and speciation analysis of saliva and urine samples from individuals living in a chronic arsenicosis area in China. Environ. Health Prev. Med. 2017, 22, 45. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Li, Y.; Liu, J.; Wang, D.; Zheng, Q.; Sun, G. Differences of urinary arsenic metabolites and methylation capacity between individuals with and without skin lesions in Inner Mongolia, Northern China. Int. J. Environ. Res. Public Health 2014, 11, 7319–7332. [Google Scholar] [CrossRef] [Green Version]
- Roggenbeck, B.A.; Banerjee, M.; Leslie, E.M. Cellular arsenic transport pathways in mammals. J. Environ. Sci. 2016, 49, 38–58. [Google Scholar] [CrossRef]
- Moe, B.; Peng, H.; Lu, X.; Chen, B.; Chen, L.W.; Gabos, S.; Li, X.-F.; Le, X.C. Comparative cytotoxicity of fourteen trivalent and pentavalent arsenic species determined using real-time cell sensing. J. Environ. Sci. 2016, 49, 113–124. [Google Scholar] [CrossRef]
- Hubaux, R.; Becker-Santos, D.D.; Enfield, K.S.; Rowbotham, D.; Lam, S.; Lam, W.L.; Martinez, V.D. Molecular features in arsenic-induced lung tumors. Mol. Cancer 2013, 12, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Dheeman, D.S.; Packianathan, C.; Pillai, J.K.; Rosen, B.P. Pathway of human AS3MT arsenic methylation. Chem. Res. Toxicol. 2014, 27, 1979–1989. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roggenbeck, B.A.; Leslie, E.M.; Walk, S.T.; Schmidt, E.E. Redox metabolism of ingested arsenic: Integrated activities of microbiome and host on toxicological outcomes. Curr. Opin. Toxicol. 2019, 13, 90–98. [Google Scholar] [CrossRef]
- Flora, S.J. Toxic metals: Health effects, and therapeutic measures. J. Biomed. Ther. Sci. 2014, 1, 48–64. [Google Scholar]
- Styblo, M.; Serves, S.V.; Cullen, W.R.; Thomas, D.J. Comparative inhibition of yeast glutathione reductase by arsenicals and arsenothiols. Chem. Res. Toxicol. 1997, 10, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Mishra, D.; Mehta, A.; Flora, S.J. Reversal of arsenic-induced hepatic apoptosis with combined administration of DMSA and its analogues in guinea pigs: Role of glutathione and linked enzymes. Chem. Res. Toxicol. 2008, 21, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Chakraborti, D.; Rahman, M.M.; Ahamed, S.; Dutta, R.N.; Pati, S.; Mukherjee, S.C. Arsenic groundwater contamination and its health effects in Patna district (capital of Bihar) in the middle Ganga plain, India. Chemosphere 2016, 152, 520–529. [Google Scholar] [CrossRef]
- Hu, Y.; Xiao, T.; Zhang, A. Associations between and risks of trace elements related to skin and liver damage induced by arsenic from coal burning. Ecotoxicol. Environ. Saf. 2021, 208, 111719. [Google Scholar] [CrossRef]
- Chervona, Y.; Arita, A.; Costa, M. Carcinogenic metals and the epigenome: Understanding the effect of nickel, arsenic, and chromium. Metallomics 2012, 4, 619–627. [Google Scholar] [CrossRef] [Green Version]
- Calatayud, M.; Barrios, J.A.; Vélez, D.; Devesa, V. In vitro study of transporters involved in intestinal absorption of inorganic arsenic. Chem. Res. Toxicol. 2012, 25, 446–453. [Google Scholar] [CrossRef]
- Frediani, J.K.; Naioti, E.A.; Vos, M.B.; Figueroa, J.; Marsit, C.J.; Welsh, J.A. Arsenic exposure and risk of nonalcoholic fatty liver disease (NAFLD) among US adolescents and adults: An association modified by race/ethnicity, NHANES 2005–2014. Environ. Health 2018, 17, 6. [Google Scholar] [CrossRef] [Green Version]
- Bunderson, M.; Coffin, J.D.; Beall, H.D. Arsenic induces peroxynitrite generation and cyclooxygenase-2 protein expression in aortic endothelial cells: Possible role in atherosclerosis. Toxicol. Appl. Pharmacol. 2002, 184, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Factor-Litvak, P.; Howe, G.R.; Graziano, J.H.; Brandt-Rauf, P.; Parvez, F.; Van Geen, A.; Ahsan, H. Arsenic exposure from drinking water, dietary intakes of B vitamins and folate, and risk of high blood pressure in Bangladesh: A population-based, cross-sectional study. Am. J. Epidemiol. 2007, 165, 541–552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bustaffa, E.; Stoccoro, A.; Bianchi, F.; Migliore, L. Genotoxic and epigenetic mechanisms in arsenic carcinogenicity. Arch. Toxicol. 2014, 88, 1043–1067. [Google Scholar] [CrossRef] [PubMed]
- Tao, X.; Wang, N.; Qin, W. Gut microbiota and hepatocellular carcinoma. Gastrointest. Tumors 2015, 2, 33–40. [Google Scholar] [CrossRef]
- Lu, K.; Abo, R.P.; Schlieper, K.A.; Graffam, M.E.; Levine, S.; Wishnok, J.S.; Swenberg, J.A.; Tannenbaum, S.R.; Fox, J.G. Arsenic exposure perturbs the gut microbiome and its metabolic profile in mice: An integrated metagenomics and metabolomics analysis. Environ. Health Perspect. 2014, 122, 284–291. [Google Scholar] [CrossRef]
- Sinha, D.; Roy, M. Antagonistic role of tea against sodium arsenite-induced oxidative DNA damage and inhibition of DNA repair in Swiss albino mice. J. Environ. Pathol. Toxicol. Oncol. 2011, 30, 311–312. [Google Scholar] [CrossRef]
- D’Ippoliti, D.; Santelli, E.; De Sario, M.; Scortichini, M.; Davoli, M.; Michelozzi, P. Arsenic in drinking water and mortality for cancer and chronic diseases in Central Italy, 1990-2010. PLoS ONE 2015, 10, e0138182. [Google Scholar] [CrossRef]
- Abdul, K.S.M.; Jayasinghe, S.S.; Chandana, E.P.; Jayasumana, C.; de Silva, P.M.C. Arsenic and human health effects: A review. Environ. Toxicol. Pharmacol. 2015, 40, 828–846. [Google Scholar] [CrossRef]
- Dangleben, N.L.; Skibola, C.F.; Smith, M.T. Arsenic immunotoxicity: A review. Environ. Health 2013, 12, 73. [Google Scholar] [CrossRef] [Green Version]
- Rehman, M.Y.A.; Briedé, J.J.; van Herwijnen, M.; Krauskopf, J.; Jennen, D.G.; Malik, R.N.; Kleinjans, J.C. Integrating SNPs-based genetic risk factor with blood epigenomic response of differentially arsenic-exposed rural subjects reveals disease-associated signaling pathways. Environ. Pollut. 2022, 292, 118279. [Google Scholar] [CrossRef]
- Martinez, V.D.; Vucic, E.A.; Adonis, M.; Gil, L.; Lam, W.L. Arsenic biotransformation as a cancer promoting factor by inducing DNA damage and disruption of repair mechanisms. Mol. Biol. Int. 2011, 2011, 718974. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soni, M.; Prakash, C.; Sehwag, S.; Kumar, V. Protective effect of hydroxytyrosol in arsenic-induced mitochondrial dysfunction in rat brain. J. Biochem. Mol. Toxicol. 2017, 31, e21906. [Google Scholar] [CrossRef] [PubMed]
- Akbal, A.; Yılmaz, H.; Tutkun, E. Arsenic exposure associated with decreased bone mineralization in male. Aging Male 2014, 17, 256–258. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.K.; Dwivedi, S.; Yadav, S.S.; Yadav, R.S.; Khattri, S. Anti-diabetic effect of Emblica officinalis (Amla) against arsenic induced metabolic disorder in mice. Indian J. Clin. Biochem. 2020, 35, 179–187. [Google Scholar] [CrossRef]
- Carlson, P.; van Beneden, R.J. Arsenic exposure alters expression of cell cycle and lipid metabolism genes in the liver of adult zebrafish (Danio rerio). Aquat. Toxicol. 2014, 153, 66–72. [Google Scholar] [CrossRef]
- Afolabi, O.K.; Wusu, A.D.; Ogunrinola, O.O.; Abam, E.O.; Babayemi, D.O.; Dosumu, O.; Onunkwor, O.; Balogun, E.; Odukoya, O.O.; Ademuyiwa, O. Arsenic-induced dyslipidemia in male albino rats: Comparison between trivalent and pentavalent inorganic arsenic in drinking water. BMC Pharmacol. Toxicol. 2015, 16, 15. [Google Scholar] [CrossRef] [Green Version]
- Souza, A.; Bastos, D.; Sertorio, M.; Santos, F.; Ervilha, L.; de Oliveira, L.; Machado-Neves, M. Combined effects of arsenic exposure and diabetes on male reproductive functions. Andrology 2019, 7, 730–740. [Google Scholar] [CrossRef]
- Wang, X.; Wu, Y.; Sun, X.; Guo, Q.; Xia, W.; Wu, Y.; Li, J.; Xu, S.; Li, Y. Arsenic exposure and metabolism in relation to blood pressure changes in pregnant women. Ecotoxicol. Environ. Saf. 2021, 222, 112527. [Google Scholar] [CrossRef]
- Faita, F.; Cori, L.; Bianchi, F.; Andreassi, M.G. Arsenic-induced genotoxicity and genetic susceptibility to arsenic-related pathologies. Int. J. Environ. Res. Public Health 2013, 10, 1527–1546. [Google Scholar] [CrossRef] [Green Version]
- Tokar, E.J.; Benbrahim-Tallaa, L.; Ward, J.M.; Lunn, R.; Sams, R.L.; Waalkes, M.P. Cancer in experimental animals exposed to arsenic and arsenic compounds. Crit. Rev. Toxicol. 2010, 40, 912–927. [Google Scholar] [CrossRef]
- Fatoki, J.O.; Badmus, J.A. Arsenic as an environmental and human health antagonist: A review of its toxicity and disease initiation. J. Hazard. Mater. Adv. 2022, 100052. [Google Scholar] [CrossRef]
- Xu, P.; Liu, A.; Li, F.; Tinkov, A.A.; Liu, L.; Zhou, J.-C. Associations between metabolic syndrome and four heavy metals: A systematic review and meta-analysis. Environ. Pollut. 2021, 273, 116480. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Sun, G.-X.; Williams, P.N.; Nunes, L.; Zhu, Y.-G. Inorganic arsenic in Chinese food and its cancer risk. Environ. Int. 2011, 37, 1219–1225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biswas, R.; Ghosh, P.; Banerjee, N.; Das, J.; Sau, T.; Banerjee, A.; Roy, S.; Ganguly, S.; Chatterjee, M.; Mukherjee, A. Analysis of T-cell proliferation and cytokine secretion in the individuals exposed to arsenic. Hum. Exp. Toxicol. 2008, 27, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Islam, L.N.; Nurun Nabi, A.; Rahman, M.M.; Zahid, M.S.H. Association of respiratory complications and elevated serum immunoglobulins with drinking water arsenic toxicity in human. J. Environ. Sci. Health Part A 2007, 42, 1807–1814. [Google Scholar] [CrossRef]
- Robles-Osorio, M.L.; Sabath-Silva, E.; Sabath, E. Arsenic-mediated nephrotoxicity. Ren. Fail. 2015, 37, 542–547. [Google Scholar] [CrossRef]
- Li, Z.; Piao, F.; Liu, S.; Wang, Y.; Qu, S. Subchronic exposure to arsenic trioxide-induced oxidative DNA damage in kidney tissue of mice. Exp. Toxicol. Pathol. 2010, 62, 543–547. [Google Scholar] [CrossRef]
- Hossain, E.; Ota, A.; Takahashi, M.; Karnan, S.; Damdindorj, L.; Konishi, Y.; Konishi, H.; Hosokawa, Y. Arsenic upregulates the expression of angiotensin II Type I receptor in mouse aortic endothelial cells. Toxicol. Lett. 2013, 220, 70–75. [Google Scholar] [CrossRef]
- Li, L.; Bi, Z.; Wadgaonkar, P.; Lu, Y.; Zhang, Q.; Fu, Y.; Thakur, C.; Wang, L.; Chen, F. Metabolic and Epigenetic Reprogramming in the Arsenic-Induced Cancer Stem Cells. In Proceedings of the Seminars in Cancer Biology; Academic Press: Cambridge, MA, USA, 2019; pp. 10–18. [Google Scholar]
- Muzaffar, S.; Khan, J.; Srivastava, R.; Gorbatyuk, M.S.; Athar, M. Mechanistic understanding of the toxic effects of arsenic and warfare arsenicals on human health and environment. Cell Biol. Toxicol. 2022, 1–26. [Google Scholar] [CrossRef]
- Xu, L.; Polya, D.A.; Li, Q.; Mondal, D. Association of low-level inorganic arsenic exposure from rice with age-standardized mortality risk of cardiovascular disease (CVD) in England and Wales. Sci. Total Environ. 2020, 743, 140534. [Google Scholar] [CrossRef]
- Kulshrestha, A.; Jarouliya, U.; Prasad, G.; Flora, S.; Bisen, P.S. Arsenic-induced abnormalities in glucose metabolism: Biochemical basis and potential therapeutic and nutritional interventions. World J. Transl. Med. 2014, 3, 96–111. [Google Scholar] [CrossRef]
- Sung, T.-C.; Huang, J.-W.; Guo, H.-R. Association between arsenic exposure and diabetes: A meta-analysis. BioMed Res. Int. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Cárdenas-González, M.; Osorio-Yáñez, C.; Gaspar-Ramírez, O.; Pavković, M.; Ochoa-Martinez, A.; López-Ventura, D.; Medeiros, M.; Barbier, O.; Pérez-Maldonado, I.; Sabbisetti, V. Environmental exposure to arsenic and chromium in children is associated with kidney injury molecule-1. Environ. Res. 2016, 150, 653–662. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hsu, L.-I.; Hsieh, F.-I.; Wang, Y.-H.; Lai, T.-S.; Wu, M.-M.; Chen, C.-J.; Chiou, H.-Y.; Hsu, K.-H. Arsenic exposure from drinking water and the incidence of CKD in low to moderate exposed areas of Taiwan: A 14-year prospective study. Am. J. Kidney Dis. 2017, 70, 787–797. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wu, L.; Sun, Q.; Yang, Q.; Xue, J.; Shi, M.; Tang, H.; Zhang, J.; Liu, Q. MicroRNA-191 blocking the translocation of GLUT4 is involved in arsenite-induced hepatic insulin resistance through inhibiting the IRS1/AKT pathway. Ecotoxicol. Environ. Saf. 2021, 215, 112130. [Google Scholar] [CrossRef] [PubMed]
- López-Carrillo, L.; Hernández-Ramírez, R.U.; Gandolfi, A.J.; Ornelas-Aguirre, J.M.; Torres-Sánchez, L.; Cebrian, M.E. Arsenic methylation capacity is associated with breast cancer in northern Mexico. Toxicol. Appl. Pharmacol. 2014, 280, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Smith, N.K.; Keltie, E.; Sweeney, E.; Weerasinghe, S.; MacPherson, K.; Kim, J.S. Toenail speciation biomarkers in arsenic-related disease: A feasibility study for investigating the association between arsenic exposure and chronic disease. Ecotoxicol. Environ. Saf. 2022, 232, 113269. [Google Scholar] [CrossRef]
- Díaz-Villaseñor, A.; Burns, A.L.; Hiriart, M.; Cebrián, M.E.; Ostrosky-Wegman, P. Arsenic-induced alteration in the expression of genes related to type 2 diabetes mellitus. Toxicol. Appl. Pharmacol. 2007, 225, 123–133. [Google Scholar] [CrossRef]
- Sarker, M.; Tony, S.R.; Siddique, A.E.; Karim, M.; Haque, N.; Islam, Z.; Islam, M.; Khatun, M.; Islam, J.; Hossain, S. Arsenic secondary methylation capacity is inversely associated with arsenic exposure-related muscle mass reduction. Int. J. Environ. Res. Public Health 2021, 18, 9730. [Google Scholar] [CrossRef]
- Ambrosio, F.; Brown, E.; Stolz, D.; Ferrari, R.; Goodpaster, B.; Deasy, B.; Distefano, G.; Roperti, A.; Cheikhi, A.; Garciafigueroa, Y. Arsenic induces sustained impairment of skeletal muscle and muscle progenitor cell ultrastructure and bioenergetics. Free. Radic. Biol. Med. 2014, 74, 64–73. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.; Ferrari, R.; Beezhold, K.; Stearns-Reider, K.; D’Amore, A.; Haschak, M.; Stolz, D.; Robbins, P.D.; Barchowsky, A.; Ambrosio, F. Arsenic promotes NF-κB-mediated fibroblast dysfunction and matrix remodeling to impair muscle stem cell function. Stem Cells 2016, 34, 732–742. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calatayud, M.; Devesa, V.; Vélez, D. Differential toxicity and gene expression in Caco-2 cells exposed to arsenic species. Toxicol. Lett. 2013, 218, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Calatayud, M.; Gimeno-Alcañiz, J.V.; Devesa, V.; Vélez, D. Proinflammatory effect of trivalent arsenical species in a co-culture of Caco-2 cells and peripheral blood mononuclear cells. Arch. Toxicol. 2015, 89, 555–564. [Google Scholar] [CrossRef] [PubMed]
- Schreibelt, G.; Kooij, G.; Reijerkerk, A.; van Doorn, R.; Gringhuis, S.I.; van der Pol, S.; Weksler, B.B.; Romero, I.A.; Couraud, P.O.; Piontek, J. Reactive oxygen species alter brain endothelial tight junction dynamics via RhoA, PI3 kinase, and PKB signaling. FASEB J. 2007, 21, 3666–3676. [Google Scholar] [CrossRef] [Green Version]
- Capaldo, C.T.; Nusrat, A. Cytokine regulation of tight junctions. Biochim. Biophys. Acta Biomembr. 2009, 1788, 864–871. [Google Scholar] [CrossRef] [Green Version]
- Groschwitz, K.R.; Hogan, S.P. Intestinal barrier function: Molecular regulation and disease pathogenesis. J. Allergy Clin. Immunol. 2009, 124, 3–20. [Google Scholar] [CrossRef] [Green Version]
- Ashraf, S.A.; Elkhalifa, A.E.O.; Ahmad, M.F.; Patel, M.; Adnan, M.; Sulieman, A.M.E. Probiotic Fermented Foods and Health Promotion. In African Fermented Food Products-New Trends; Springer: Berlin/Heildeberg, Germany, 2022; pp. 59–88. [Google Scholar]
- Dong, X.; Shulzhenko, N.; Lemaitre, J.; Greer, R.L.; Peremyslova, K.; Quamruzzaman, Q.; Rahman, M.; Hasan, O.S.I.; Joya, S.A.; Golam, M. Arsenic exposure and intestinal microbiota in children from Sirajdikhan, Bangladesh. PLoS ONE 2017, 12, e0188487. [Google Scholar] [CrossRef] [Green Version]
- Wu, F.; Yang, L.; Islam, M.T.; Jasmine, F.; Kibriya, M.G.; Nahar, J.; Barmon, B.; Parvez, F.; Sarwar, G.; Ahmed, A. The role of gut microbiome and its interaction with arsenic exposure in carotid intima-media thickness in a Bangladesh population. Environ. Int. 2019, 123, 104–113. [Google Scholar] [CrossRef]
- Yadav, R.S.; Shukla, R.K.; Sankhwar, M.L.; Patel, D.K.; Ansari, R.W.; Pant, A.B.; Islam, F.; Khanna, V.K. Neuroprotective effect of curcumin in arsenic-induced neurotoxicity in rats. Neurotoxicology 2010, 31, 533–539. [Google Scholar] [CrossRef]
- Ramos-Chávez, L.A.; Rendón-López, C.R.; Zepeda, A.; Silva-Adaya, D.; Del Razo, L.M.; Gonsebatt, M.E. Neurological effects of inorganic arsenic exposure: Altered cysteine/glutamate transport, NMDA expression and spatial memory impairment. Front. Cell. Neurosci. 2015, 2015, 21. [Google Scholar] [CrossRef] [Green Version]
- Salmeri, N.; Villanacci, R.; Ottolina, J.; Bartiromo, L.; Cavoretto, P.; Dolci, C.; Lembo, R.; Schimberni, M.; Valsecchi, L.; Viganò, P. Maternal arsenic exposure and gestational diabetes: A systematic review and meta-analysis. Nutrients 2020, 12, 3094. [Google Scholar] [CrossRef] [PubMed]
- Tseng, C.-H. The potential biological mechanisms of arsenic-induced diabetes mellitus. Toxicol. Appl. Pharmacol. 2004, 197, 67–83. [Google Scholar] [CrossRef] [PubMed]
- Seshadri, N.; Jonasson, M.E.; Hunt, K.L.; Xiang, B.; Cooper, S.; Wheeler, M.B.; Dolinsky, V.W.; Doucette, C.A. Uncoupling protein 2 regulates daily rhythms of insulin secretion capacity in MIN6 cells and isolated islets from male mice. Mol. Metab. 2017, 6, 760–769. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, M.G.; Queen, Z.J.; Cherry, N. Histopathology of cervical cancer and arsenic concentration in well water: An ecological analysis. Int. J. Environ. Res. Public Health 2017, 14, 1185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kile, M.L.; Faraj, J.M.; Ronnenberg, A.G.; Quamruzzaman, Q.; Rahman, M.; Mostofa, G.; Afroz, S.; Christiani, D.C. A cross sectional study of anemia and iron deficiency as risk factors for arsenic-induced skin lesions in Bangladeshi women. BMC Public Health 2016, 16, 158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yunus, F.M.; Rahman, M.J.; Alam, M.Z.; Hore, S.K.; Rahman, M. Relationship between arsenic skin lesions and the age of natural menopause. BMC Public Health 2014, 14, 419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Surdu, S.; Bloom, M.S.; Neamtiu, I.A.; Pop, C.; Anastasiu, D.; Fitzgerald, E.F.; Gurzau, E.S. Consumption of arsenic-contaminated drinking water and anemia among pregnant and non-pregnant women in northwestern Romania. Environ. Res. 2015, 140, 657–660. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Breton, C.V.; Houseman, E.A.; Kile, M.L.; Quamruzzaman, Q.; Rahman, M.; Mahiuddin, G.; Christiani, D.C. Gender-specific protective effect of hemoglobin on arsenic-induced skin lesions. Cancer Epidemiol. Prev. Biomark. 2006, 15, 902–907. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmed, S.; Khoda, S.M.-e.; Rekha, R.S.; Gardner, R.M.; Ameer, S.S.; Moore, S.; Ekström, E.-C.; Vahter, M.; Raqib, R. Arsenic-associated oxidative stress, inflammation, and immune disruption in human placenta and cord blood. Environ. Health Perspect. 2011, 119, 258–264. [Google Scholar] [CrossRef] [Green Version]
- Davey, J.C.; Bodwell, J.E.; Gosse, J.A.; Hamilton, J.W. Arsenic as an endocrine disruptor: Effects of arsenic on estrogen receptor–mediated gene expression in vivo and in cell culture. Toxicol. Sci. 2007, 98, 75–86. [Google Scholar] [CrossRef] [Green Version]
- Chatterjee, A.; Chatterji, U. Arsenic abrogates the estrogen-signaling pathway in the rat uterus. Reprod. Biol. Endocrinol. 2010, 8, 80. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aquino, N.B.; Sevigny, M.B.; Sabangan, J.; Louie, M.C. The role of cadmium and nickel in estrogen receptor signaling and breast cancer: Metalloestrogens or not? J. Environ. Sci. Health Part C 2012, 30, 189–224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marciniak, W.; Derkacz, R.; Muszyńska, M.; Baszuk, P.; Gronwald, J.; Huzarski, T.; Cybulski, C.; Jakubowska, A.; Falco, M.; Dębniak, T. Blood arsenic levels and the risk of familial breast cancer in Poland. Int. J. Cancer 2020, 146, 2721–2727. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, L.; Liu, B.; Zhang, L.; Wu, M.; Wang, L.; Cao, Z.; Zhang, B.; Li, Y.; Wang, Y.; Xu, S. Association of prenatal exposure to arsenic with newborn telomere length: Results from a birth cohort study. Environ. Res. 2019, 175, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Pilsner, J.R.; Hall, M.N.; Liu, X.; Ilievski, V.; Slavkovich, V.; Levy, D.; Factor-Litvak, P.; Yunus, M.; Rahman, M.; Graziano, J.H. Influence of prenatal arsenic exposure and newborn sex on global methylation of cord blood DNA. PLoS ONE 2012, 7, e37147. [Google Scholar] [CrossRef] [Green Version]
- Milton, A.H.; Hussain, S.; Akter, S.; Rahman, M.; Mouly, T.A.; Mitchell, K. A review of the effects of chronic arsenic exposure on adverse pregnancy outcomes. Int. J. Environ. Res. Public Health 2017, 14, 556. [Google Scholar] [CrossRef] [Green Version]
- He, Y.; Pan, A.; Hu, F.B.; Ma, X. Folic acid supplementation, birth defects, and adverse pregnancy outcomes in Chinese women: A population-based mega-cohort study. Lancet 2016, 388, S91. [Google Scholar] [CrossRef]
- Mazumdar, M.; Hasan, M.O.S.I.; Hamid, R.; Valeri, L.; Paul, L.; Selhub, J.; Rodrigues, E.G.; Silva, F.; Mia, S.; Mostofa, M.G. Arsenic is associated with reduced effect of folic acid in myelomeningocele prevention: A case control study in Bangladesh. Environ. Health 2015, 14, 34. [Google Scholar] [CrossRef] [Green Version]
- Parajuli, R.P.; Fujiwara, T.; Umezaki, M.; Watanabe, C. Association of cord blood levels of lead, arsenic, and zinc with neurodevelopmental indicators in newborns: A birth cohort study in Chitwan Valley, Nepal. Environ. Res. 2013, 121, 45–51. [Google Scholar] [CrossRef]
- Demir, N.; Başaranoğlu, M.; Huyut, Z.; Değer, İ.; Karaman, K.; Şekeroğlu, M.R.; Tuncer, O. The relationship between mother and infant plasma trace element and heavy metal levels and the risk of neural tube defect in infants. J. Matern. Fetal Neonatal Med. 2019, 32, 1433–1440. [Google Scholar] [CrossRef]
- Tolins, M.; Ruchirawat, M.; Landrigan, P. The developmental neurotoxicity of arsenic: Cognitive and behavioral consequences of early life exposure. Ann. Glob. Health 2014, 80, 303–314. [Google Scholar] [CrossRef] [PubMed]
- Nadeau, K.C.; Li, Z.; Farzan, S.; Koestler, D.; Robbins, D.; Fei, D.L.; Malipatlolla, M.; Maecker, H.; Enelow, R.; Korrick, S. In utero arsenic exposure and fetal immune repertoire in a US pregnancy cohort. Clin. Immunol. 2014, 155, 188–197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farzan, S.F.; Korrick, S.; Li, Z.; Enelow, R.; Gandolfi, A.J.; Madan, J.; Nadeau, K.; Karagas, M.R. In utero arsenic exposure and infant infection in a United States cohort: A prospective study. Environ. Res. 2013, 126, 24–30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, C.-B.; Hsi, H.-C.; Fan, C.-H.; Chien, L.-C. Fetal exposure to environmental neurotoxins in Taiwan. PLoS ONE 2014, 9, e109984. [Google Scholar] [CrossRef] [Green Version]
- Vahter, M. Effects of arsenic on maternal and fetal health. Annu. Rev. Nutr. 2009, 29, 381–399. [Google Scholar] [CrossRef]
- Bae, S.; Kamynina, E.; Farinola, A.F.; Caudill, M.A.; Stover, P.J.; Cassano, P.A.; Berry, R.; Peña-Rosas, J.P. Provision of folic acid for reducing arsenic toxicity in arsenic-exposed children and adults. Cochrane Database Syst. Rev. 2017, CD012649. [Google Scholar] [CrossRef]
- Arroyo, H.A.; Fernández, M.C. Tóxicos ambientales y su efecto sobre el neurodesarrollo. Medicina 2013, 73. [Google Scholar]
- Yorifuji, T.; Kato, T.; Ohta, H.; Bellinger, D.C.; Matsuoka, K.; Grandjean, P. Neurological and neuropsychological functions in adults with a history of developmental arsenic poisoning from contaminated milk powder. Neurotoxicol. Teratol. 2016, 53, 75–80. [Google Scholar] [CrossRef] [Green Version]
- Normandin, L.; Ayotte, P.; Levallois, P.; Ibanez, Y.; Courteau, M.; Kennedy, G.; Chen, L.; Le, X.C.; Bouchard, M. Biomarkers of arsenic exposure and effects in a Canadian rural population exposed through groundwater consumption. J. Expo. Sci. Environ. Epidemiol. 2014, 24, 127–134. [Google Scholar] [CrossRef] [Green Version]
- Barr, D.B.; Wilder, L.C.; Caudill, S.P.; Gonzalez, A.J.; Needham, L.L.; Pirkle, J.L. Urinary creatinine concentrations in the US population: Implications for urinary biologic monitoring measurements. Environ. Health Perspect. 2005, 113, 192–200. [Google Scholar] [CrossRef] [Green Version]
- Pullella, K.; Kotsopoulos, J. Arsenic exposure and breast cancer risk: A re-evaluation of the literature. Nutrients 2020, 12, 3305. [Google Scholar] [CrossRef] [PubMed]
- Arabia, S. Grain and Feed Annual 2016, United States Department of Agriculture. Foreign Agriculture Service. Available online: www.gain.fas.usda.gov (accessed on 3 March 2022).
- Menon, M.; Sarkar, B.; Hufton, J.; Reynolds, C.; Reina, S.V.; Young, S. Do arsenic levels in rice pose a health risk to the UK population? Ecotoxicol. Environ. Saf. 2020, 197, 110601. [Google Scholar] [CrossRef] [PubMed]
- Meharg, A.A.; Williams, P.N.; Adomako, E.; Lawgali, Y.Y.; Deacon, C.; Villada, A.; Cambell, R.C.; Sun, G.; Zhu, Y.-G.; Feldmann, J. Geographical variation in total and inorganic arsenic content of polished (white) rice. Environ. Sci. Technol. 2009, 43, 1612–1617. [Google Scholar] [CrossRef] [PubMed]
- Islam, S.; Rahman, M.M.; Duan, L.; Islam, M.; Kuchel, T.; Naidu, R. Variation in arsenic bioavailability in rice genotypes using swine model: An animal study. Sci. Total Environ. 2017, 599, 324–331. [Google Scholar] [CrossRef]
- Mwale, T.; Rahman, M.M.; Mondal, D. Risk and benefit of different cooking methods on essential elements and arsenic in rice. Int. J. Environ. Res. Public Health 2018, 15, 1056. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Menon, M.; Dong, W.; Chen, X.; Hufton, J.; Rhodes, E.J. Improved rice cooking approach to maximise arsenic removal while preserving nutrient elements. Sci. Total Environ. 2021, 755, 143341. [Google Scholar] [CrossRef]
- Bundschuh, J.; Nath, B.; Bhattacharya, P.; Liu, C.-W.; Armienta, M.A.; López, M.V.M.; Lopez, D.L.; Jean, J.-S.; Cornejo, L.; Macedo, L.F.L. Arsenic in the human food chain: The Latin American perspective. Sci. Total Environ. 2012, 429, 92–106. [Google Scholar] [CrossRef]
- Sun, G.; Williams, P.; Carey, A.; Zhu, Y.; Deacon, C.; Raab, A.; Feldmann, J.; Meharg, A.; Islam, R. Elevated arsenic in rice bran products used as food supplements and in food aid programs. Environ. Sci. Technol. 2008, 42, 7542–7546. [Google Scholar] [CrossRef]
- Raab, A.; Baskaran, C.; Feldmann, J.; Meharg, A.A. Cooking rice in a high water to rice ratio reduces inorganic arsenic content. J. Environ. Monit. 2009, 11, 41–44. [Google Scholar] [CrossRef]
- Carey, M.; Jiujin, X.; Gomes Farias, J.; Meharg, A.A. Rethinking rice preparation for highly efficient removal of inorganic arsenic using percolating cooking water. PLoS ONE 2015, 10, e0131608. [Google Scholar] [CrossRef]
- Gray, P.J.; Conklin, S.D.; Todorov, T.I.; Kasko, S.M. Cooking rice in excess water reduces both arsenic and enriched vitamins in the cooked grain. Food Addit. Contam. Part A 2016, 33, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, N.R.; Das, A.; Joardar, M.; De, A.; Mridha, D.; Das, R.; Rahman, M.M.; Roychowdhury, T. Flow of arsenic between rice grain and water: Its interaction, accumulation and distribution in different fractions of cooked rice. Sci. Total Environ. 2020, 731, 138937. [Google Scholar] [CrossRef] [PubMed]
- Sanyal, T.; Bhattacharjee, P.; Paul, S. Recent advances in arsenic research: Significance of differential susceptibility and sustainable strategies for mitigation. Front. Public Health 2020, 8, 464. [Google Scholar] [CrossRef]
- Moxness Reksten, A.; Rahman, Z.; Kjellevold, M.; Garrido Gamarro, E.; Thilsted, S.H.; Pincus, L.M.; Aakre, I.; Ryder, J.; Ariyawansa, S.; Nordhagen, A. Metal Contents in Fish from the Bay of Bengal and Potential Consumer Exposure—The EAF-Nansen Programme. Foods 2021, 10, 1147. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, M.K.; Shukla, A.; Yadav, P.; Srivastava, S. A review of arsenic in crops, vegetables, animals and food products. Food Chem. 2019, 276, 608–618. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 1993. [Google Scholar]
- Nachman, K.E.; Ginsberg, G.L.; Miller, M.D.; Murray, C.J.; Nigra, A.E.; Pendergrast, C.B. Mitigating dietary arsenic exposure: Current status in the United States and recommendations for an improved path forward. Sci. Total Environ. 2017, 581, 221–236. [Google Scholar] [CrossRef]
| Different Species of Arsenic | Abbreviation | Distribution | References |
|---|---|---|---|
| Arsenocholine | AC | Arsenic species generally found in seafood and oxidized to arsenobetaine in a biological system. | [6] |
| In organic arsenic | iAs | Found in most foods and its presence in water is in low amounts. | [7] |
| Arsenite | As (III) | It is highly toxic in nature but present in lesser amounts in most foods. | [7] |
| Arsenate | As (V) | It is highly toxic in nature but present in lesser amounts in most foods and water. | [8,9] |
| Dimethylarsinate | DMA | Found in seafood and terrestrial foods and is a urine metabolite of iAs arsenosugars. | [10] |
| Dimethylarsinite | DMA (III) | It cannot be detected in food samples. It is a metabolite of iAs and can be seen in human urine samples but is highly toxic in nature. | [11] |
| Methylarsonate | MA | Found in seafood and terrestrial foods in very low amounts and is a metabolite of iAs that can be seen in urine. | [12] |
| Methylarsonite | MA (III) | It cannot be detected in food samples. It is a metabolite of iAs that can be seen in human urine samples but it is a toxic metabolite. | [13] |
| Arsenobetaine Arsenosugar | AB | It is a major arsenic species and commonly found in seafood but is non-toxic in nature. | [7] |
| Trimethylarsonio propionate | TMAP | Present in most foods. It is one of the major arsenic species. | [14] |
| Trimethylarsine oxide | TMAO | It is generally found in seafood and distributed in small amounts. | [11] |
| Study Area | Source | As Concentration Range (μg/L) | References |
|---|---|---|---|
| Kuwait | Marine water and sediment | 0–43 | [30] |
| Oman Sea | Fish species | 0.74 ± 0.37 to 3.30 ± 1.39 μg/g | [31] |
| Kuwait | Kuwait Bay and the Open Sea | [32] | |
| District of Baniyas, Abu Dhabi/UAE | Household drinking water | Max. 95.04 | [29] |
| Avg. 12.75 | |||
| Min. 8.9 | |||
| Chabahar Bay/Oman | Surface sediments | 5 and 22 ppm | [33] |
| Saudi Arabia/Al-Kharj agricultural region | Boreholes | Max.122 | [35] |
| Avg.31.18 | |||
| Min. 2 | |||
| Chabahar Bay/Oman | Surface sediments | 8.67–21 ppm | [42] |
| Al Jabal Al Akhdar/Oman. | Aflaj | [34] | |
| Saudi Arabia/Gulf of Aqaba | Groundwater samples (wells) | Max. 2.2 | [38] |
| Avg. 0.63 | |||
| Min. < 0.1 | |||
| Saudi Arabian Gulf coast, Tarut | Sediments, seawater, and gastropod and bivalve specimens | 53–342 sediments | [43] |
| 8.55–14.88 seawaters | |||
| 16.3–38.1 Molluscs | |||
| Saudi Arabia/Arabian Gulf | Surface sediments | 148 | [44] |
| Al Munawarah area, Al Madinah/Saudi Arabia | Groundwater samples (wells) | <detection limit | [45] |
| Saudi Arabia/Gulf of Aqaba | Surface seawater and sediments | 12.2–15.1 sediments | [40] |
| 0.46–1.55 Sea water samples | |||
| Bahrain | Water fishing areas | 0.85–2.75 | [46] |
| Gulf and Oman’s Gulf | Marine biota (fish and various bivalves) and coastal sediments | Coastal Sediments | [41] |
| Qatar 1.0–6.3 | |||
| UAE 0.7–9.6 Bahrain 3.16–6.88 | |||
| Oman 0.74–5.01 | |||
| Molluscs | |||
| Gulf of Oman and Arabian Gulf 11.1–156 | |||
| Kuwait | Wells | <detection limit, 0.005 mg/L | [47] |
| Kuwait coast | Seawater, microplankton, shrimp, mollusk, fish | 0.01–0.06 Water | [39] |
| 0.01–0.10 Particulate matter | |||
| 0.01–0.04 Phytoplankton | |||
| 0.08–0.42 Shrimp | |||
| 0.15–0.43 Mollusc | |||
| 0.21–2.10 Fin Fish |
| Different Dietary Products | Toxic Limits | Food Source | Total as Detected | References |
|---|---|---|---|---|
| Rice species | (0.10 mg/kg–0.30 mg/kg) | American Rice | 0.25 mg/kg | [51] |
| Thai rice | 0.2 mg/kg | |||
| Pakistani rice | 0.14 mg/kg | |||
| Indian rice | 0.103 mg/kg | |||
| Egyptian rice | 0.097 mg/kg | |||
| Infant cereals | (0.1 mg/kg) | Rice | 0.160 mg/day | [33,34,52,53] |
| Milk powder | 20 μg/kg | |||
| Fruit and vegetable mix | 49 μg/kg | |||
| Mixed cereals | 55 μg/kg–158 μg/kg | |||
| Beans | Cowpea/Black-eyed pea | [54] | ||
| Meat products | Liver; 0.20 mg/kg Kidney; 0.50 mg/kg Fish; 4.0 mg/kg | Canned Meat | 0.002 mg/kg | [55,56] |
| Canned Fish | 0.857 mg/kg | |||
| Sea food/fish | 6.0 μg/g | Shrimps Clams and pearl oyster Mollusk shells Saudi Arabia | 0.19–0.53 μg/g | [57,58] |
| 11–134 mg/kg | ||||
| 16.3–38.1 mg/g | ||||
| Dairy products | Milk | 0.0002–0.05 mg/kg | [59] | |
| Vegetables | 0.1 mg/kg 1.0 mg/kg (China) | Fresh vegetables | 1.93–5.73 mg/kg | [52,53,60,61] |
| Canned vegetables | 2.50–5.10 mg/kg | |||
| Mushroom (Bangladesh) | 0.51 mg/kg | |||
| Dates | 0.1 mg/kg | Eklas (Al-Hasaa) | 0.584 mg/kg | [62] |
| Barny (Al-adina) | 0.078 mg/kg | |||
| Sakay mabroum (Al-Karj), | 0.095 mg/kg | |||
| Sakay Nomal (Al-Karj), | 0.109 mg/kg | |||
| Kadary (Al-Qaseem) | 0.121 mg/kg | |||
| Juices and beverages | 0.01 mg/L | Canned beverages Non-canned beverages Juices Orange juice | 0.003–0.161 mg/L | [63,64,65] |
| 0.002–0.261 mg/L | ||||
| 3.76 μg/kg | ||||
| (1.137–18.36) | ||||
| 2.01–2.56 mg/kg | ||||
| Honey | 15 μg/kg | Albaha Saudi Arabia | 0.02–533.7 μg/kg | [66] |
| Tea and coffee | Organic oolong tea (China) | 0.06–1.12 μg/L | [67] | |
| Granulated black tea (India) | 2.680 mg/kg | |||
| House hold water | 10 μg/L | Commercial botteled Riyadh | 0.574 ± 0.748 μg/L | [68,69] |
| Groundwater samples | Madina Al Munawwarah | 12.0–29.0 μg/L |
| Arsenic Chemical Forms | Health Effects | References |
|---|---|---|
| Inorganic arsenic (AsIII and AsV) | Cancer | [100] |
| Chronic diseases | [101,102] | |
| Mutation | [103] | |
| DNA damage | [104] | |
| Mitochondrial dysfunction | [105] | |
| Reduces bone mineralization | [106] | |
| Hyperglycemia | [107] | |
| Lipid disorders | [108] | |
| Coronary heart disease | [109] | |
| Liver toxicity | [110] | |
| Hypertension | [111] | |
| Genotoxicity | [112] | |
| Arsenite (AsIII) | Cancer | [113] |
| Fatty liver | [114] | |
| Hepatotoxicity | [115] | |
| Arsenic trioxide | Breast cancer | [116] |
| Arsenic Species | Direct Toxic Effect and Target Organ Damage (TOD) | Molecular Mechanisms | References |
|---|---|---|---|
| Inorganic arsenic in drinking water and rice | Skin cancer | Differentiation and generation of cancer stem cells | [122,123] |
| Coronary artery disease and cardiac muscle damage | Cardiac tissue hypoxia and inflammation | [124] | |
| Diabetes and insulin resistance | Inhibition of glycolysis, Krebs’s cycle, and ATP synthesis | [125,126] | |
| Acute Kidney Injury (AKI) | Kidney injury molecule-1 (KIM1) | [127] | |
| Chronic kidney disease (CKD) | Decreased glomerular filtration rate | [128] | |
| Arsenite (AsIII) | Insulin resistance and metabolic syndrome | Diminished translocation of GLUT4 | [129] |
| MMA (Monomethylarsonic acid) | Breast cancer | Endocrine disruptor | [130] |
| Lung cancer | DNA damage | [104,131] | |
| Kidney cancer | DNA damage | [131] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, M.I.; Ahmad, M.F.; Ahmad, I.; Ashfaq, F.; Wahab, S.; Alsayegh, A.A.; Kumar, S.; Hakeem, K.R. Arsenic Exposure through Dietary Intake and Associated Health Hazards in the Middle East. Nutrients 2022, 14, 2136. https://doi.org/10.3390/nu14102136
Khan MI, Ahmad MF, Ahmad I, Ashfaq F, Wahab S, Alsayegh AA, Kumar S, Hakeem KR. Arsenic Exposure through Dietary Intake and Associated Health Hazards in the Middle East. Nutrients. 2022; 14(10):2136. https://doi.org/10.3390/nu14102136
Chicago/Turabian StyleKhan, Mohammad Idreesh, Md Faruque Ahmad, Irfan Ahmad, Fauzia Ashfaq, Shadma Wahab, Abdulrahman A. Alsayegh, Sachil Kumar, and Khalid Rehman Hakeem. 2022. "Arsenic Exposure through Dietary Intake and Associated Health Hazards in the Middle East" Nutrients 14, no. 10: 2136. https://doi.org/10.3390/nu14102136
APA StyleKhan, M. I., Ahmad, M. F., Ahmad, I., Ashfaq, F., Wahab, S., Alsayegh, A. A., Kumar, S., & Hakeem, K. R. (2022). Arsenic Exposure through Dietary Intake and Associated Health Hazards in the Middle East. Nutrients, 14(10), 2136. https://doi.org/10.3390/nu14102136







