Psychological and Gastrointestinal Symptoms of Patients with Irritable Bowel Syndrome Undergoing a Low-FODMAP Diet: The Role of the Intestinal Barrier
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patient Recruitment
2.2. Psychological Questionnaire
2.2.1. Symptom Checklist-90-Revised (SCL-90-R)
2.2.2. Psychophysiological Questionnaire—QPF/R
2.3. Quality of Life Questionnaire
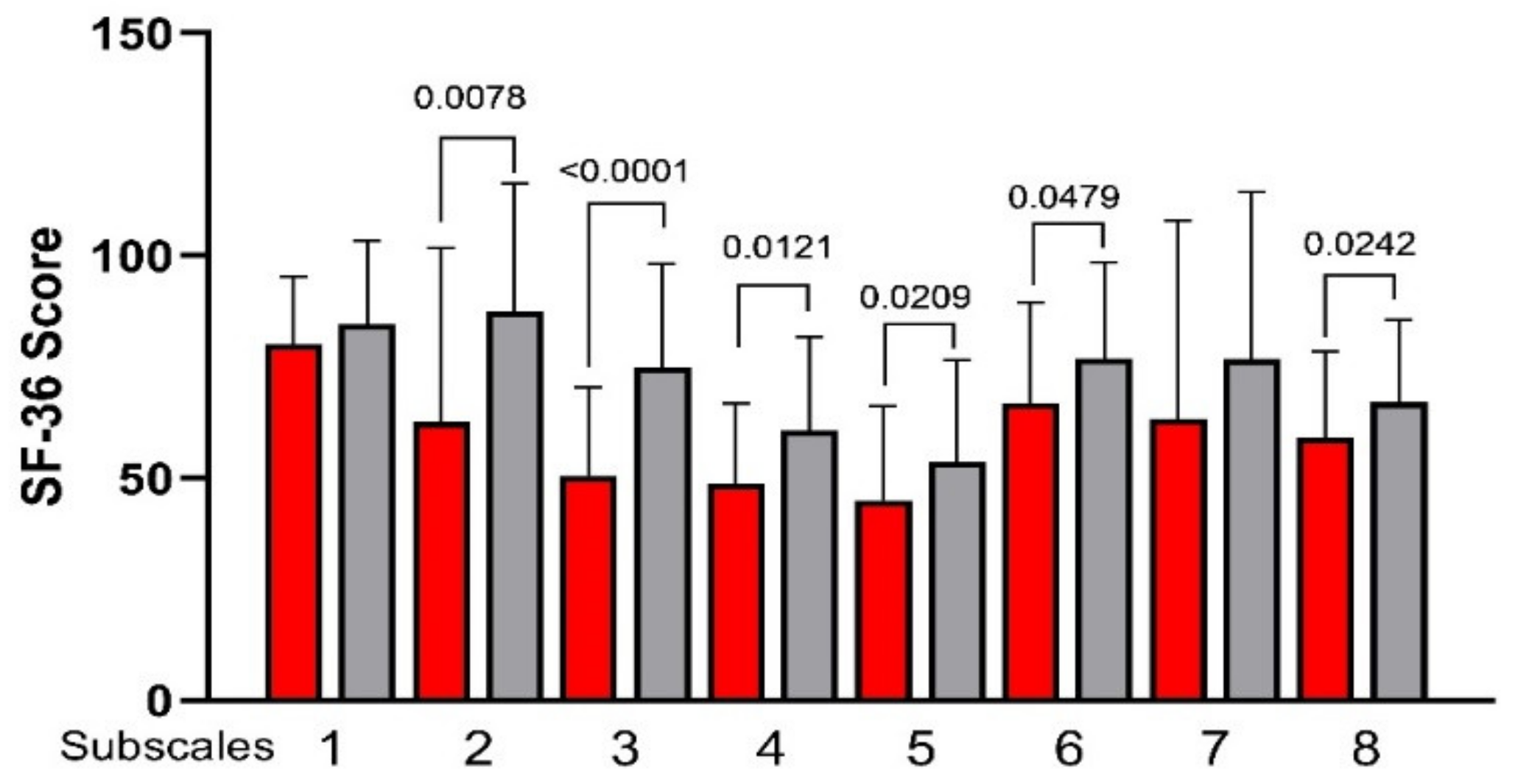
2.3.1. 36-Item Short-Form Health Survey (SF-36)
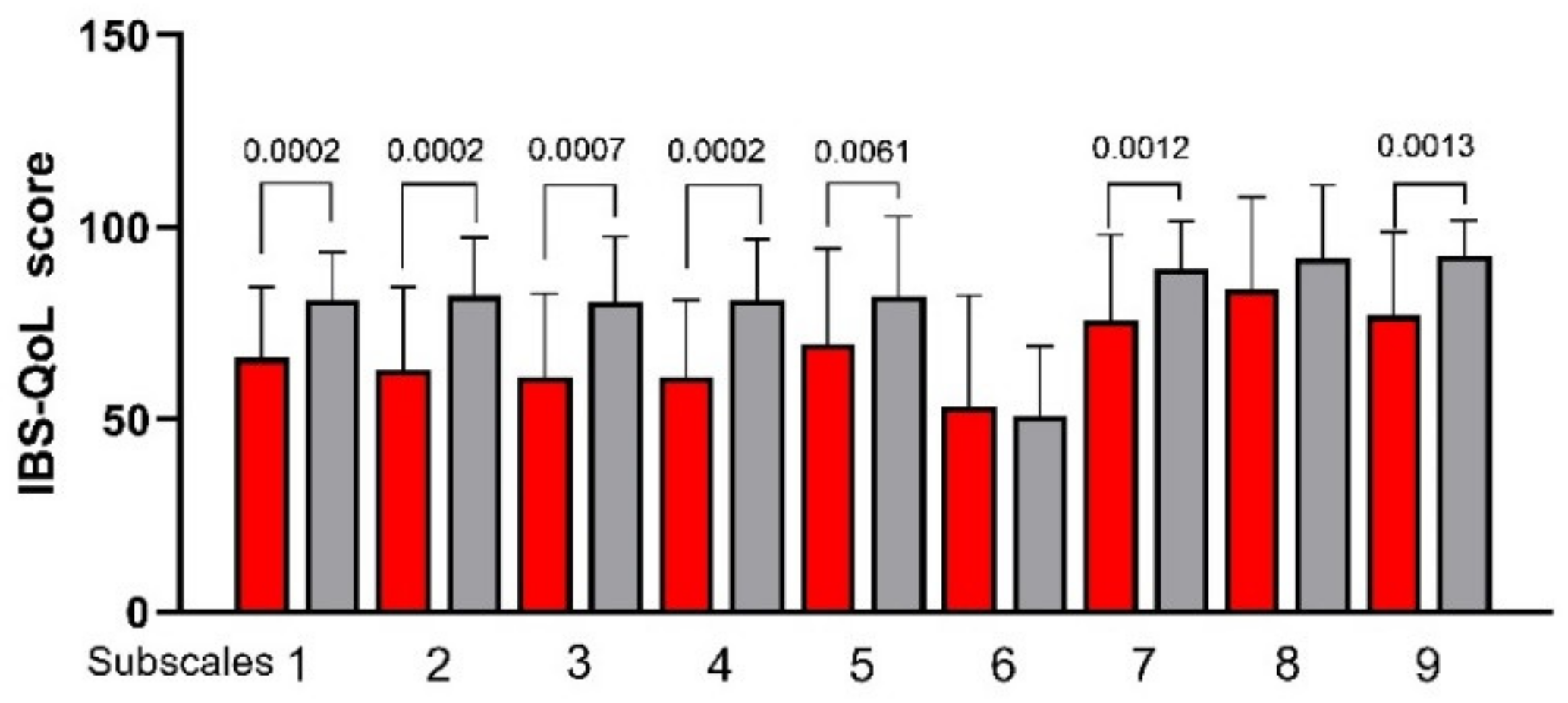
2.3.2. IBS Quality of Life Questionnaire
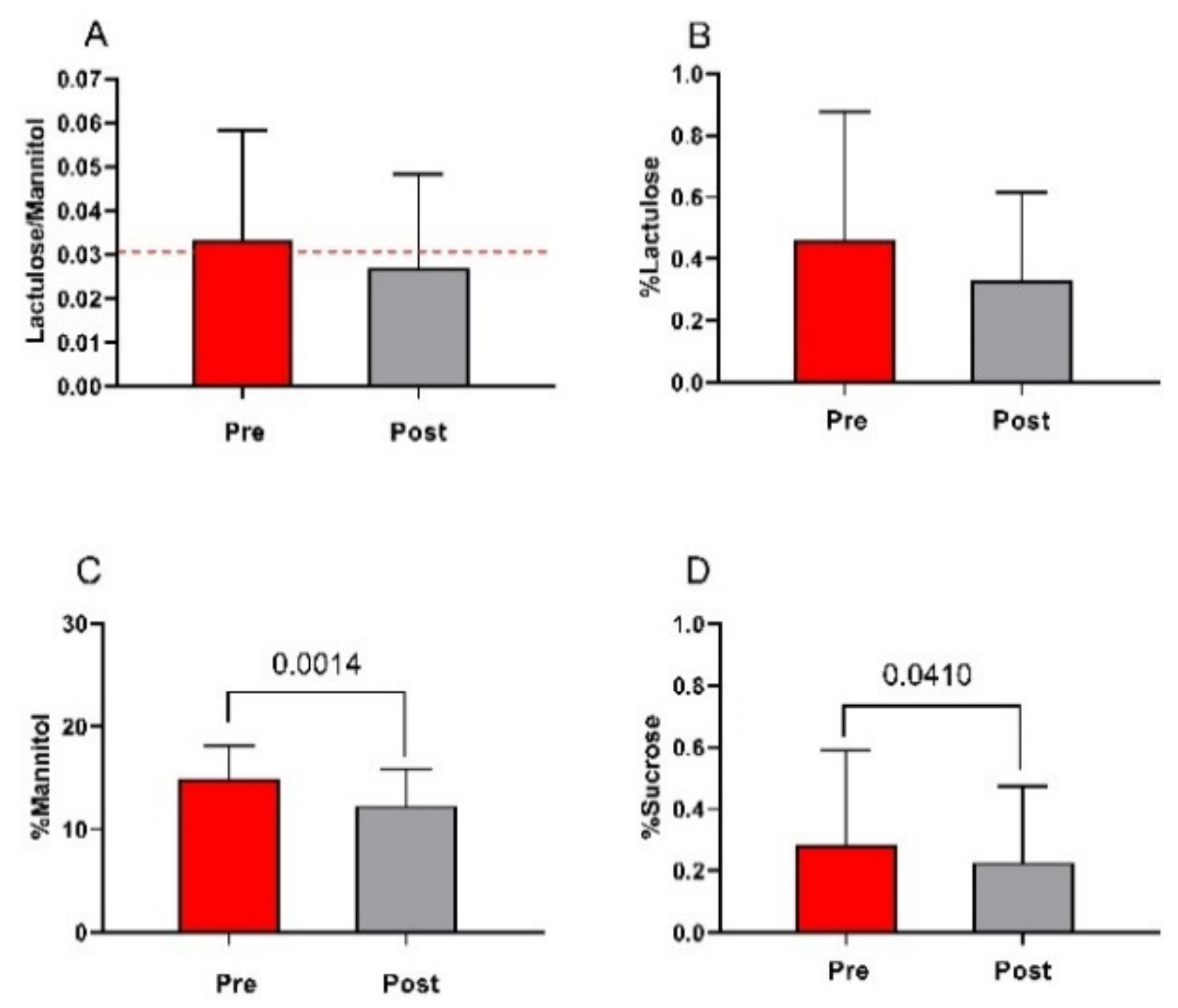
2.4. Sugar Absorption Test
2.5. Biochemical Analyses
2.6. Indican and Skatole Evaluation
2.7. Statistical Procedures
3. Results
3.1. Anthropometric Characteristics of the Patients
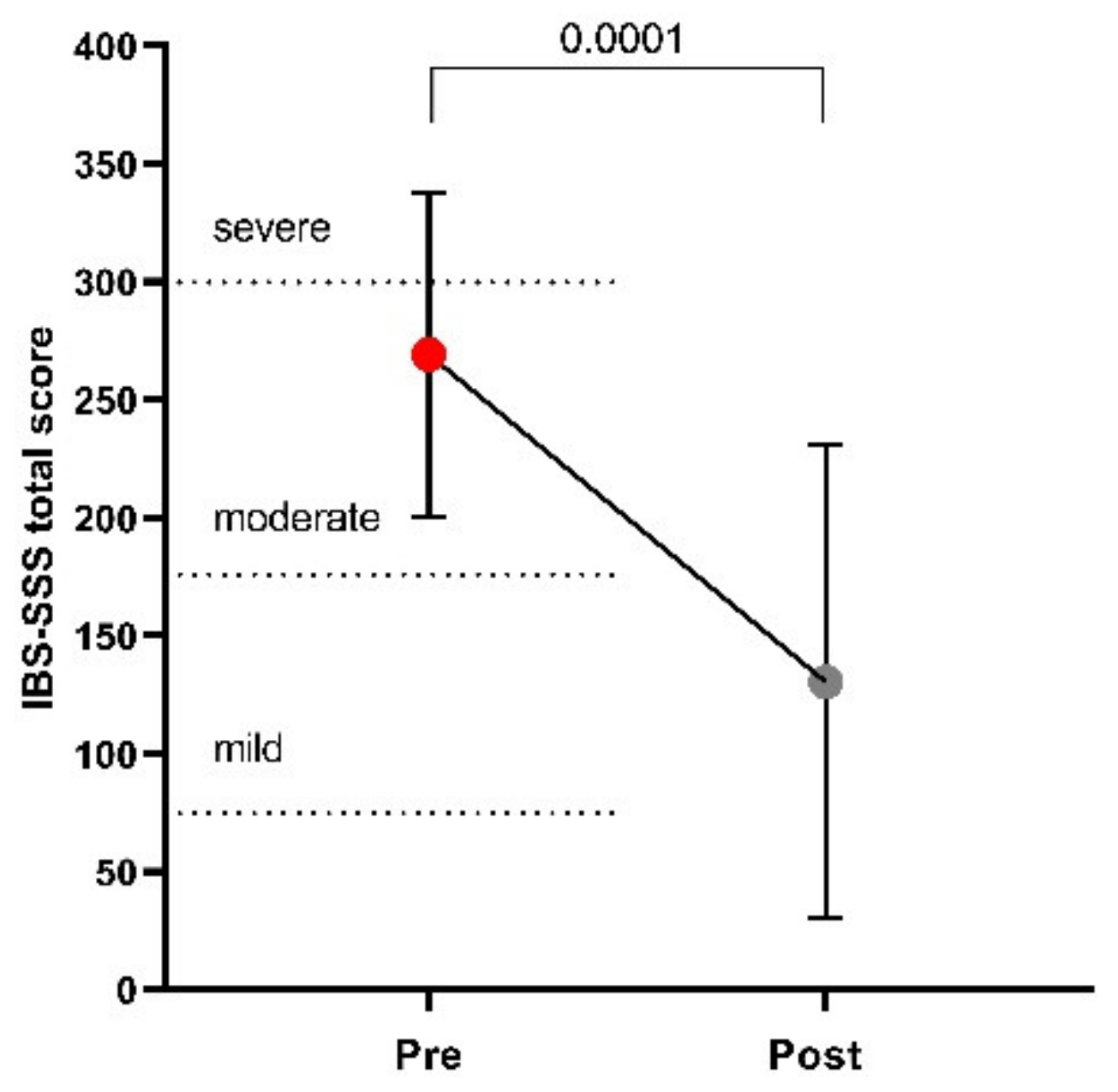
3.2. Symptom Profile
3.3. Psychological and QoL Profiles
3.4. The Small Intestinal Permeability (s-IP)
3.5. Markers of Function and Integrity of the Intestinal Barrier
3.6. Indices of Inflammation
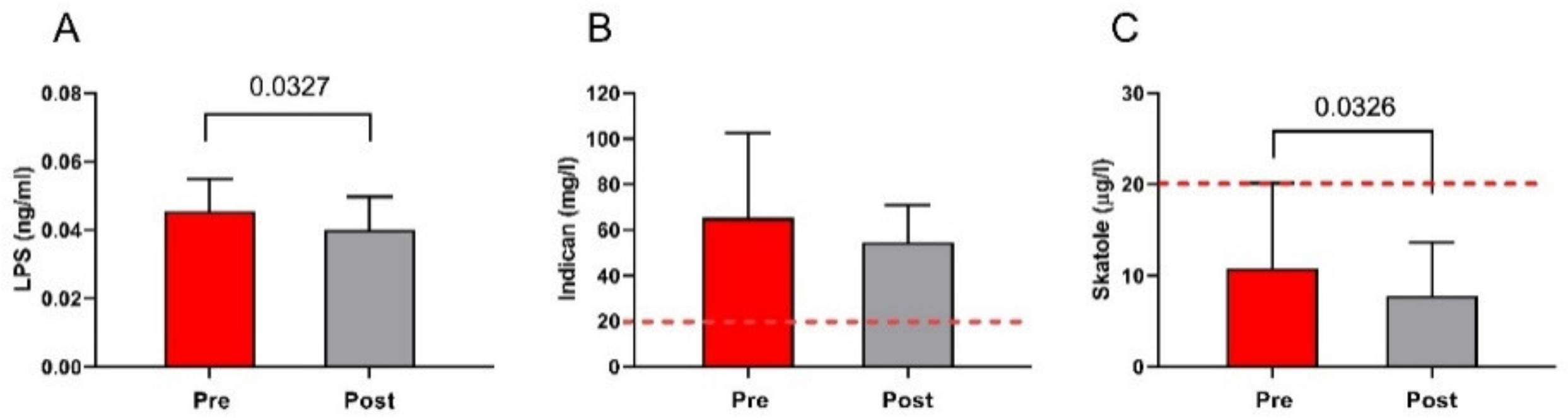
3.7. The Markers of Bacterial Translocation and Intestinal Dysbiosis
3.8. Regression Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chey, W.D.; Kurlander, J.; Eswaran, S. Irritable bowel syndrome: A clinical review. JAMA 2015, 313, 949–958. [Google Scholar] [CrossRef] [PubMed]
- The Rome Foundation. Available online: https://theromefoundation.org/rome-iv/rome-iv-criteria/ (accessed on 1 December 2017).
- Whitehead, W.E.; Palsson, O.; Jones, K.R. Systematic review of the comorbidity of irritable bowel syndrome with other disorders: What are the causes and implications? Gastroenterology 2002, 122, 1140–1156. [Google Scholar] [CrossRef]
- Hu, Z.; Li, M.; Yao, L.; Wang, Y.; Wang, E.; Yuan, J.; Wang, F.; Yang, K.; Bian, Z.; Zhong, L.L.D. The level and prevalence of depression and anxiety among patients with different subtypes of irritable bowel syndrome: A network meta-analysis. BMC Gastroenterol. 2021, 21, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Zamani, M.; Alizadeh-Tabari, S.; Zamani, V. Systematic review with meta-analysis: The prevalence of anxiety and depression in patients with irritable bowel syndrome. Aliment. Pharmacol. Ther. 2019, 50, 132–143. [Google Scholar] [CrossRef] [Green Version]
- Kabra, N.; Nadkarni, A. Prevalence of depression and anxiety in irritable bowel syndrome: A clinic based study from India. Indian J. Psychiatry 2013, 55, 77–80. [Google Scholar] [CrossRef]
- Tanaka, Y.; Kanazawa, M.; Fukudo, S.; Drossman, D.A. Biopsychosocial model of irritable bowel syndrome. J. Neurogastroenterol. Motil. 2011, 17, 131–139. [Google Scholar] [CrossRef] [Green Version]
- Bai, T.; Xia, J.; Jiang, Y.; Cao, H.; Zhao, Y.; Zhang, L.; Wang, H.; Song, J.; Hou, X. Comparison of the Rome IV and Rome III criteria for IBS diagnosis: A cross-sectional survey. J. Gastroenterol. Hepatol. 2017, 32, 1018–1025. [Google Scholar] [CrossRef]
- Tornblom, H.; Van Oudenhove, L.; Sadik, R.; Abrahamsson, H.; Tack, J.; Simren, M. Colonic transit time and IBS symptoms: What’s the link? Am. J. Gastroenterol. 2012, 107, 754–760. [Google Scholar] [CrossRef] [Green Version]
- Simren, M.; Tornblom, H.; Palsson, O.S.; van Tilburg, M.A.L.; Van Oudenhove, L.; Tack, J.; Whitehead, W.E. Visceral hypersensitivity is associated with GI symptom severity in functional GI disorders: Consistent findings from five different patient cohorts. Gut 2018, 67, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Martinez, C.; Gonzalez-Castro, A.; Vicario, M.; Santos, J. Cellular and molecular basis of intestinal barrier dysfunction in the irritable bowel syndrome. Gut Liver 2012, 6, 305–315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martin-Vinas, J.J.; Quigley, E.M. Immune response in irritable bowel syndrome: A systematic review of systemic and mucosal inflammatory mediators. J. Dig. Dis. 2016, 17, 572–581. [Google Scholar] [CrossRef]
- Lee, K.N.; Lee, O.Y. Intestinal microbiota in pathophysiology and management of irritable bowel syndrome. World J. Gastroenterol. 2014, 20, 8886–8897. [Google Scholar] [CrossRef]
- Powell, N.; Walker, M.M.; Talley, N.J. The mucosal immune system: Master regulator of bidirectional gut-brain communications. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 143–159. [Google Scholar] [CrossRef]
- Mayer, E.A. Gut feelings: The emerging biology of gut-brain communication. Nat. Rev. Neurosci. 2011, 12, 453–466. [Google Scholar] [CrossRef]
- Bashashati, M.; Rezaei, N.; Shafieyoun, A.; McKernan, D.P.; Chang, L.; Ohman, L.; Quigley, E.M.; Schmulson, M.; Sharkey, K.A.; Simren, M. Cytokine imbalance in irritable bowel syndrome: A systematic review and meta-analysis. Neurogastroenterol. Motil. 2014, 26, 1036–1048. [Google Scholar] [CrossRef] [PubMed]
- Liebregts, T.; Adam, B.; Bredack, C.; Roth, A.; Heinzel, S.; Lester, S.; Downie-Doyle, S.; Smith, E.; Drew, P.; Talley, N.J.; et al. Immune activation in patients with irritable bowel syndrome. Gastroenterology 2007, 132, 913–920. [Google Scholar] [CrossRef] [PubMed]
- Orlando, A.; Tutino, V.; Notarnicola, M.; Riezzo, G.; Linsalata, M.; Clemente, C.; Prospero, L.; Martulli, M.; D’Attoma, B.; De Nunzio, V.; et al. Improved Symptom Profiles and Minimal Inflammation in IBS-D Patients Undergoing a Long-Term Low-FODMAP Diet: A Lipidomic Perspective. Nutrients 2020, 12, 1652. [Google Scholar] [CrossRef] [PubMed]
- Wells, J.M.; Brummer, R.J.; Derrien, M.; MacDonald, T.T.; Troost, F.; Cani, P.D.; Theodorou, V.; Dekker, J.; Meheust, A.; de Vos, W.M.; et al. Homeostasis of the gut barrier and potential biomarkers. Am. J. Physiol. Gastrointest. Liver Physiol. 2017, 312, G171–G193. [Google Scholar] [CrossRef]
- Hughes, P.A.; Zola, H.; Penttila, I.A.; Blackshaw, L.A.; Andrews, J.M.; Krumbiegel, D. Immune activation in irritable bowel syndrome: Can neuroimmune interactions explain symptoms? Am. J. Gastroenterol. 2013, 108, 1066–1074. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Linsalata, M.; Riezzo, G.; D’Attoma, B.; Clemente, C.; Orlando, A.; Russo, F. Noninvasive biomarkers of gut barrier function identify two subtypes of patients suffering from diarrhoea predominant-IBS: A case-control study. BMC Gastroenterol. 2018, 18, 167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stevens, B.R.; Goel, R.; Seungbum, K.; Richards, E.M.; Holbert, R.C.; Pepine, C.J.; Raizada, M.K. Increased human intestinal barrier permeability plasma biomarkers zonulin and FABP2 correlated with plasma LPS and altered gut microbiome in anxiety or depression. Gut 2018, 67, 1555–1557. [Google Scholar] [CrossRef] [PubMed]
- Camilleri, M.; Lyle, B.J.; Madsen, K.L.; Sonnenburg, J.; Verbeke, K.; Wu, G.D. Role for diet in normal gut barrier function: Developing guidance within the framework of food-labeling regulations. Am. J. Physiol. Gastrointest. Liver Physiol. 2019, 317, G17–G39. [Google Scholar] [CrossRef] [PubMed]
- Tuck, C.J.; Reed, D.E.; Muir, J.G.; Vanner, S.J. Implementation of the low FODMAP diet in functional gastrointestinal symptoms: A real-world experience. Neurogastroenterol. Motil. 2020, 32, e13730. [Google Scholar] [CrossRef] [PubMed]
- Linsalata, M.; Riezzo, G.; Orlando, A.; D’Attoma, B.; Prospero, L.; Tutino, V.; Notarnicola, M.; Russo, F. The Relationship between Low Serum Vitamin D Levels and Altered Intestinal Barrier Function in Patients with IBS Diarrhoea Undergoing a Long-Term Low-FODMAP Diet: Novel Observations from a Clinical Trial. Nutrients 2021, 13, 1011. [Google Scholar] [CrossRef]
- Kortlever, T.L.; Ten Bokkel Huinink, S.; Offereins, M.; Hebblethwaite, C.; O’Brien, L.; Leeper, J.; Mulder, C.J.J.; Barrett, J.S.; Gearry, R.B. Low-FODMAP Diet Is Associated With Improved Quality of Life in IBS Patients-A Prospective Observational Study. Nutr. Clin. Pract. 2019, 34, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Hayes, P.A.; Fraher, M.H.; Quigley, E.M. Irritable bowel syndrome: The role of food in pathogenesis and management. Gastroenterol. Hepatol. 2014, 10, 164–174. [Google Scholar]
- Mearin, F.; Lacy, B.E.; Chang, L.; Chey, W.D.; Lembo, A.J.; Simren, M.; Spiller, R. Bowel Disorders. Gastroenterology 2016, 150, 1393–1407. [Google Scholar] [CrossRef] [Green Version]
- Francis, C.Y.; Morris, J.; Whorwell, P.J. The irritable bowel severity scoring system: A simple method of monitoring irritable bowel syndrome and its progress. Aliment. Pharmacol. Ther. 1997, 11, 395–402. [Google Scholar] [CrossRef] [Green Version]
- Miller, L.E. Study design considerations for irritable bowel syndrome clinical trials. Ann. Gastroenterol. 2014, 27, 338–345. [Google Scholar]
- Blake, M.R.; Raker, J.M.; Whelan, K. Validity and reliability of the Bristol Stool Form Scale in healthy adults and patients with diarrhoea-predominant irritable bowel syndrome. Aliment. Pharmacol. Ther. 2016, 44, 693–703. [Google Scholar] [CrossRef] [Green Version]
- Derogatis, L.R.; Lipman, R.S.; Covi, L. SCL-90: An outpatient psychiatric rating scale--preliminary report. Psychopharmacol. Bull. 1973, 9, 13–28. [Google Scholar] [PubMed]
- Jensen, H.H.; Mortensen, E.L.; Lotz, M. Scl-90-R symptom profiles and outcome of short-term psychodynamic group therapy. ISRN Psychiatry 2013, 2013, 540134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bertolotti, G.; Zotti, A.M.; Michielin, P.; Vidotto, G.; Sanavio, E. A computerized approach to cognitive behavioural assessment: An introduction to CBA-2.0 primary scales. J. Behav. Ther. Exp. psychiatry 1990, 21, 21–27. [Google Scholar] [CrossRef]
- Ware, J.E., Jr.; Sherbourne, C.D. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med. Care 1992, 30, 473–483. [Google Scholar] [CrossRef]
- Patrick, D.L.; Drossman, D.A.; Frederick, I.O.; DiCesare, J.; Puder, K.L. Quality of life in persons with irritable bowel syndrome: Development and validation of a new measure. Dig. Dis. Sci. 1998, 43, 400–411. [Google Scholar] [CrossRef]
- Hilsden, R.J.; Meddings, J.B.; Sutherland, L.R. Intestinal permeability changes in response to acetylsalicylic acid in relatives of patients with Crohn’s disease. Gastroenterology 1996, 110, 1395–1403. [Google Scholar] [CrossRef]
- Hendrikx, T.; Schnabl, B. Indoles: Metabolites produced by intestinal bacteria capable of controlling liver disease manifestation. J. Intern. Med. 2019, 286, 32–40. [Google Scholar] [CrossRef] [Green Version]
- Garcia Regueiro, J.A.; Rius, M.A. Rapid determination of skatole and indole in pig back fat by normal-phase liquid chromatography. J. Chromatography. A 1998, 809, 246–251. [Google Scholar] [CrossRef]
- Zhou, Q.; Verne, M.L.; Fields, J.Z.; Lefante, J.J.; Basra, S.; Salameh, H.; Verne, G.N. Randomised placebo-controlled trial of dietary glutamine supplements for postinfectious irritable bowel syndrome. Gut 2019, 68, 996–1002. [Google Scholar] [CrossRef]
- Prospero, L.; Riezzo, G.; Linsalata, M.; Orlando, A.; D’Attoma, B.; Di Masi, M.; Martulli, M.; Russo, F. Somatization in patients with predominant diarrhoea irritable bowel syndrome: The role of the intestinal barrier function and integrity. BMC Gastroenterol. 2021, 21, 235. [Google Scholar] [CrossRef] [PubMed]
- Van Lanen, A.S.; de Bree, A.; Greyling, A. Efficacy of a low-FODMAP diet in adult irritable bowel syndrome: A systematic review and meta-analysis. Eur. J. Nutr. 2021. [Google Scholar] [CrossRef]
- Cingolani, A.; Paduano, D.; Vecchiarelli, V.; Demelas, M.; Corrias, P.T.; Casula, L.; Usai, P. Feasibility of Low Fermentable Oligosaccharide, Disaccharide, Monosaccharide, and Polyol Diet and Its Effects on Quality of Life in an Italian Cohort. Nutrients 2020, 12, 716. [Google Scholar] [CrossRef] [Green Version]
- Kumar, S.; Shukla, R.; Ranjan, P.; Kumar, A. Interleukin-10: A Compelling Therapeutic Target in Patients With Irritable Bowel Syndrome. Clin. Ther. 2017, 39, 632–643. [Google Scholar] [CrossRef]
- Holzer, P.; Hassan, A.M.; Jain, P.; Reichmann, F.; Farzi, A. Neuroimmune pharmacological approaches. Curr. Opin. Pharmacol. 2015, 25, 13–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gonsalkorale, W.M.; Perrey, C.; Pravica, V.; Whorwell, P.J.; Hutchinson, I.V. Interleukin 10 genotypes in irritable bowel syndrome: Evidence for an inflammatory component? Gut 2003, 52, 91–93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, C.C.; Gan, L.; Tan, Y.; Yan, M.Z.; Liu, X.M.; Chang, Q.; Pan, R.L. Chronic restraint stress induced changes in colonic homeostasis-related indexes and tryptophan-kynurenine metabolism in rats. J. Proteom. 2021, 240, 104190. [Google Scholar] [CrossRef]
- Zhou, S.Y.; Gillilland, M., III; Wu, X.; Leelasinjaroen, P.; Zhang, G.; Zhou, H.; Ye, B.; Lu, Y.; Owyang, C. FODMAP diet modulates visceral nociception by lipopolysaccharide-mediated intestinal inflammation and barrier dysfunction. J. Clin. Investig. 2018, 128, 267–280. [Google Scholar] [CrossRef] [PubMed]
- Sperandeo, P.; Martorana, A.M.; Polissi, A. Lipopolysaccharide Biosynthesis and Transport to the Outer Membrane of Gram-Negative Bacteria. Bact. Cell Walls Membr. 2019, 92, 9–37. [Google Scholar] [CrossRef]
- Wang, Z.; Xu, C.M.; Liu, Y.X.; Wang, X.Q.; Zhang, L.; Li, M.; Zhu, S.W.; Xie, Z.J.; Wang, P.H.; Duan, L.P.; et al. Characteristic dysbiosis of gut microbiota of Chinese patients with diarrhea-predominant irritable bowel syndrome by an insight into the pan-microbiome. Chin. Med. J. 2019, 132, 889–904. [Google Scholar] [CrossRef]
- Maes, M.; Kubera, M.; Leunis, J.C. The gut-brain barrier in major depression: Intestinal mucosal dysfunction with an increased translocation of LPS from gram negative enterobacteria (leaky gut) plays a role in the inflammatory pathophysiology of depression. Neuroendocrinol. Lett. 2008, 29, 117–124. [Google Scholar]
- Rogers, G.B.; Keating, D.J.; Young, R.L.; Wong, M.L.; Licinio, J.; Wesselingh, S. From gut dysbiosis to altered brain function and mental illness: Mechanisms and pathways. Mol. Psychiatry 2016, 21, 738–748. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hollier, J.M.; van Tilburg, M.A.L.; Liu, Y.; Czyzewski, D.I.; Self, M.M.; Weidler, E.M.; Heitkemper, M.; Shulman, R.J. Multiple psychological factors predict abdominal pain severity in children with irritable bowel syndrome. Neurogastroenterol. Motil. 2019, 31, e13509. [Google Scholar] [CrossRef] [PubMed]
- Boem, F.; Amedei, A. Healthy axis: Towards an integrated view of the gut-brain health. World J. Gastroenterol. 2019, 25, 3838–3841. [Google Scholar] [CrossRef] [PubMed]



| Pre | Post | p | |
|---|---|---|---|
| Age (years) | 42.65 ± 9.66 | // | |
| Sex (M/F) | 4/16 | // | |
| Height (m) | 1.63 ± 0.10 | // | |
| Weight (kg) | 65.16 ± 13.22 | 60.99 ± 12.22 | <0.0001 |
| BMI (kg/m2) | 24.30 ± 4.16 | 22.84 ± 4.02 | <0.0001 |
| Mid-upper arm circumference (cm) | 28.21 ± 3.67 | 26.68 ± 3.48 | <0.0001 |
| Shoulder circumference (cm) | 109.90 ± 9.55 | 101.00 ± 8.49 | <0.0001 |
| Abdominal circumference (cm) | 86.78 ± 10.25 | 83.79 ± 10.08 | <0.0001 |
| Hip circumference (cm) | 77.78 ± 11.60 | 74.70 ± 10.04 | 0.0001 |
| Waist circumference (cm) | 97.18 ± 9.39 | 93.27 ± 8.53 | <0.0001 |
| Pre | Post | p | |
|---|---|---|---|
| Abdominal pain intensity | 47.60 ± 22.15 | 20.25 ± 21.18 | 0.0003 |
| Abdominal pain frequency | 47.00 ± 28.12 | 20.00 ± 25.75 | 0.0001 |
| Abdominal distension | 51.63 ± 23.60 | 22.90 ± 20.29 | 0.0007 |
| Dissatisfaction of bowel habit | 61.45 ± 23.16 | 30.45 ± 22.92 | 0.0032 |
| Interference with life in general | 61.45 ± 18.43 | 36.80 ± 29.03 | 0.0054 |
| Stool frequency (Bowel movements per day) | 1.96 ± 0.78 | 1.18 ± 0.60 | 0.0012 |
| Stool frequency (last 10 days) | 1.79 ± 0.59 | 1.20 ± 0.61 | 0.0032 |
| β | Std. Error (β) | p | 95% CI | |
|---|---|---|---|---|
| DAO | 10.58 | 4.42 | 0.029 | 2.88–18.27 |
| Somatization (SCL-90) | 4.65 | 1.41 | 0.005 | 2.11–7.20 |
| Total score (IBS-QoL) | −3.13 | 1.12 | 0.013 | −4.95–−1.31 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prospero, L.; Riezzo, G.; Linsalata, M.; Orlando, A.; D’Attoma, B.; Russo, F. Psychological and Gastrointestinal Symptoms of Patients with Irritable Bowel Syndrome Undergoing a Low-FODMAP Diet: The Role of the Intestinal Barrier. Nutrients 2021, 13, 2469. https://doi.org/10.3390/nu13072469
Prospero L, Riezzo G, Linsalata M, Orlando A, D’Attoma B, Russo F. Psychological and Gastrointestinal Symptoms of Patients with Irritable Bowel Syndrome Undergoing a Low-FODMAP Diet: The Role of the Intestinal Barrier. Nutrients. 2021; 13(7):2469. https://doi.org/10.3390/nu13072469
Chicago/Turabian StyleProspero, Laura, Giuseppe Riezzo, Michele Linsalata, Antonella Orlando, Benedetta D’Attoma, and Francesco Russo. 2021. "Psychological and Gastrointestinal Symptoms of Patients with Irritable Bowel Syndrome Undergoing a Low-FODMAP Diet: The Role of the Intestinal Barrier" Nutrients 13, no. 7: 2469. https://doi.org/10.3390/nu13072469
APA StyleProspero, L., Riezzo, G., Linsalata, M., Orlando, A., D’Attoma, B., & Russo, F. (2021). Psychological and Gastrointestinal Symptoms of Patients with Irritable Bowel Syndrome Undergoing a Low-FODMAP Diet: The Role of the Intestinal Barrier. Nutrients, 13(7), 2469. https://doi.org/10.3390/nu13072469








