Guidelines on Standard and Therapeutic Diets for Adults in Hospitals by the French Association of Nutritionist Dieticians (AFDN) and the French Speaking Society of Clinical Nutrition and Metabolism (SFNCM)
Abstract
:1. Introduction
1.1. Hospital Food Provision: Interest and Societal Challenges
1.2. Constraints Related to Meals
1.3. Nourishing the Ill Patients
1.4. Objectives of the Present Guidelines
- To ensure the nutritional needs of the patients and adapt to their pathophysiological condition as well as to the needs associated with hospitalization (acute care, follow-up and rehabilitation care, long-term stay).
- To show consideration for the patient by conferring food provision its full meaning: both nutrition-wise as well as personal-wise (taking into account choices, mealtimes, etc.).
- To rationalize and harmonize practices related to nutritional diet prescriptions.
- To lighten the constraints that could induce restrictions on meal provision (choice of food for the preparation of dishes, implications on meal costs, consequences on the palatability and consumption of meals/dishes by patients).
2. Materials and Methods
2.1. Description of the Method
- A preliminary survey was carried out by the French speaking Society of Clinical Nutrition and Metabolism (SFNCM, Société Francophone de Nutrition Clinique et Métabolisme) in 2017, with the goal of establishing an inventory of food practices and diets in French healthcare establishments [3]. This survey revealed a wide heterogeneity in the various nutritional regimens offered, whereby the same diet name could correspond to different contents in terms of authorized or prohibited foods as well as threshold levels in the case of restrictions. This discordance, induced by historical practices or prescribing habits, has direct consequences on the food intake of patients and their nutritional status.
- This inventory led to the creation of a working group comprised equally of members of the French Association of Nutritionist Dieticians (AFDN, Association Française des Diététiciens Nutritionnistes) and the SFNCM. Proposals for recommendations were put forward based on the results of the survey and data from the literature.
- These proposals were submitted to a group of 50 national experts (25 dieticians and 25 physicians). The experts were drawn from all regions of the country and chosen for their expertise in the various specialties pertaining to nutrition (gastroenterology, nephrology, obesity, etc.). The experts were consulted on the basis of the DELPHI method, as proposed by the French Health Authority (HAS) [4].
2.2. Procedural Design of the DELPHI Method
- Analyzed, synthesized and debated the literature on hospital food provision and nutritional diets.
- Produced 22 initial proposals stemming from the baseline survey of 2017.
- Submitted the 22 version 1 recommendations online for a 1st assessment rating (score ranging from 0 to 9) by the 50 experts.
- Analyzed the responses from this first round: the comments were used to upgrade the proposals and ultimately led to proposing a 23rd recommendation to complete version 1.
- Submitted the 23rd recommendation as well as the version 2 recommendations (with accompanying arguments) for a second assessment rating of the version 1 proposals which had an initial rating below 7.
- Analyzed the responses of the 2nd round. All of the recommendations were deemed appropriate (median ≥ 7) with strong agreement for 18 of the latter (scores between 7 and 9), and relative agreement (scores between 5 and 9) for the remaining 5. The experts who gave a score less than 7 on these 5 recommendations with relative agreement were contacted individually for a 3rd rating and comments.
- Following the 3rd round, the 23 recommendations were all accepted with strong agreement.
2.3. The 23 Recommendations on Standard and Therapeutic Diets
2.3.1. Lexical Scope of the Recommendations
- Prefer the term “standard” to “normal” with regard to standard food offered in health establishments.
- Only retain therapeutic diets—i.e., that imply restrictions or modifications on foods—that fall under prescribed medical settings.
2.3.2. Structuring of the Recommendations
- Define food provision (quantitative and qualitative) in hospital establishments, both in terms of adaptation to the patient’s needs as well as to promote food intake (recommendations 1, 2, 3, 4).
- Highlight the indications/importance of nutritional prescription and its re-assessment (recommendations 5, 6, 7).
- Specify the food provision modalities that must sometimes be implemented in order to adapt the diet to the patient’s capabilities and appetite (recommendations 8, 9).
- Streamline and limit the restrictions in therapeutic diets which exclude certain nutrients, in order to only retain the indications based on scientific evidence and to define these indications with regard to the benefit/risk balance of undernutrition (recommendations 10 to 22).
- Highlight the importance of the place of specifically adapted diets for undernourished patients and those at risk of undernutrition (recommendation 23).
3. Results
3.1. Recommendations on Food Provision
- Recommendation 1: It is recommended that food provision offer several choices to the patient.
- Recommendation 2: It is recommended that the standard diet meet the guidelines for prevention and health promotion.
- Recommendation 3: Outside of a personalized adaptation, it is recommended that the standard diet provide a minimum of 2000 kcal/d.
- Recommendation 4: It is recommended that the nutritional values (energy, proteins, fats, carbohydrates) of provided food be accessible, in particular to the prescribing physician and the dietician.
- For dishes prepared by catering services: knowledge per serving served, at minimum—proteins, lipids, carbohydrates and energy.
- For therapeutic diets, the nutritional values of these dishes can target salt, fiber, potassium, calcium intake, etc.
- For prepared products originating directly from the food industry, suppliers must be able to provide the nutritional value of the dishes.
- Special attention is required for each change in supplier, for both finished dishes and raw foodstuffs. An update of the calculations is then necessary.
- Given that the calculations are based on recipe technical sheets as well as technical sheets of raw foodstuffs from suppliers, approximations are frequent.
- Regular nutritional analyses by appropriate analysis laboratories are to be integrated into the follow-up and quality control of the catering service. They are the guarantors of the regularity and adequacy of food provision along with the nutritional target. They also allow strategies to be adjusted in terms of menus, palatability, and the reaching of nutritional goals.
3.2. Prescription of Nutritional Diets at the Hospital
- Recommendation 5: It is recommended that all diets (standard and therapeutic) be prescribed upon admission of the patient and adapted according to his or her clinical condition.
- Recommendation 6: It is recommended that all types of therapeutic diets be reassessed during hospitalization and upon discharge by the prescriber, and if necessary, by a dietician and/or a physician nutritionist.
- Recommendation 7: It is recommended not to combine more than two restrictive therapeutic diets due to the risk of undernutrition.
3.3. Adaptation of Food Provision
- Recommendation 8: It is recommended that food provision can be partitioned by offering more than three meals a day.
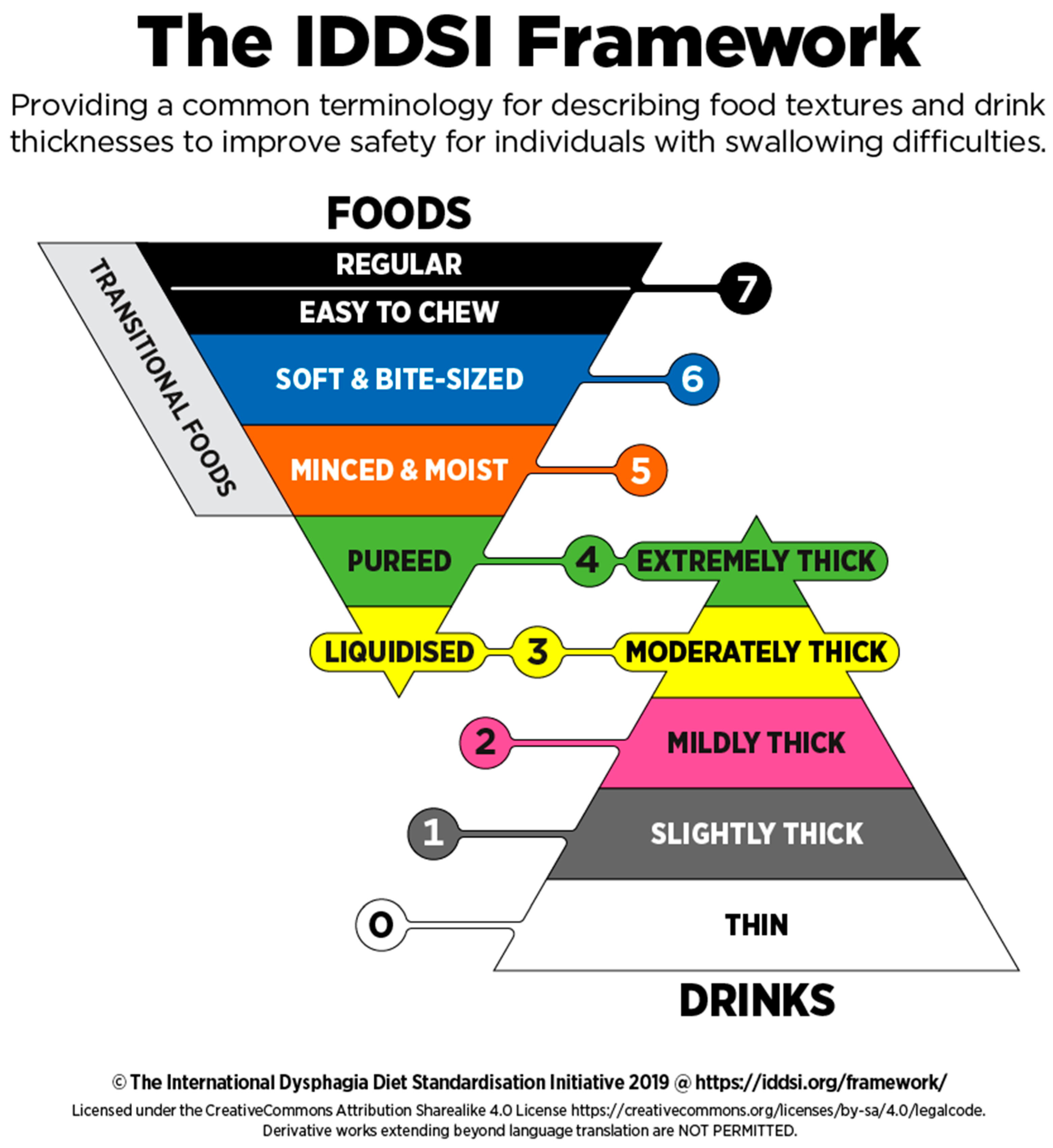
- Recommendation 9: It is recommended that the adaptation of food texture be established according to the International Dysphagia Diet Standardization Initiative (IDDSI) recommendations.
3.4. Therapeutic Diets
- Recommendation 10: During hospitalization, aside from a specialized indication, the prescription of a therapeutic diet aimed at weight loss is not recommended.
- Recommendation 11: It is recommended to adapt protein intake according to Chronic Kidney Disease stage and nutritional status.
- Normal renal function with increased risk of CKD (diabetes, hypertension, solitary kidney, etc.): <1 g of protein/kg/day or on adjusted weight if BMI > 30.
- Increase the proportion of vegetable proteins while maintaining a satisfactory animal protein/vegetable protein ratio.
- Mild to moderate CKD (stages II and III): 0.8 g protein/kg/day
- Consider 0.6–0.8 g protein/kg/day if the glomerular filtration rate (GFR) <45 mL/min/1.73 m2 (stage IIIB) or if progression is rapid.
- Severe and end-stage CKD (stages IV and V): 0.6 g to 0.8 g/kg/d including 50% high biological value protein, or <0.6 g/kg/d with the addition of essential amino acids or keto-analogs.
- Recommendation 12: It is not recommended to prescribe a low-fat therapeutic diet < 35% of the total energy intake, with the exception of major primary hypertriglyceridemia and chylous effusions (chylothorax, chylous ascites and chyluria) in which a strict fat restriction is required (<30 g per day, excluding medium chain triglycerides (MCTs)).
- The lack of consensus and scientific literature regarding the value of a low-fat diet in instances of hypertriglyceridemia, other than its primary form. There is no consensus as to the definition of major hypertriglyceridemia or of the threshold beyond which there is risk of acute pancreatitis.
- In the case of acute alcoholic pancreatitis, often associated with hypertriglyceridemia, the value of such a diet and the triglyceridemia threshold requiring a low-fat diet has not been established.
- Substitution with medium chain triglycerides is not addressed in this recommendation.
- The value of a low-fat diet (<35% of energy intake) is now questioned in its classic indications: prevention of obesity, prevention of cardiovascular disease [59]. The Mediterranean-type diet (40–45% fat, rich in monounsaturated fats and omega 3) appears more effective in terms of cardiovascular risk prevention, high blood pressure [60], prevention of type 2 diabetes [61], NASH (non-alcoholic steatohepatitis), etc.
- Hypertriglyceridemia (hyperTG):
- Acute pancreatitis:
- Exocrine pancreatic insufficiency: chronic pancreatitis:
- Following supramesocolic surgery (cephalic duodenopancreatectomy, gastrectomy and superior polar esogastrectomy):
- Chylothorax:
- Chyluria:
- Chylous ascites:
- Bile acid malabsorption:
- Obesity and cardiovascular risk:
- Cancer:
- NASH (non-alcoholic steatohepatitis):
- Recommendation 13: The standard diet is suitable for diabetic patients without exclusion of foods and desserts containing sucrose.
- Recommendation 14: It is recommended that the standard diet provide a regular carbohydrate content for each meal.
- Recommendation 15: It is recommended that the amount of carbohydrates be known and accessible for each dish served.
- Recommendation 16: If a low-salt therapeutic diet is indicated, it is recommended not to restrict salt intake (NaCl) from the diet to less than 5 g/day (or approx. 2 g of sodium/d), except in severe acute decompensation and for a very short duration.
- Recommendation 17: The diet designated as “acid-free and/or spice-free” has no indication (except oral or digestive hypersensitivity or food allergy).
3.5. Glossary
3.6. Beliefs and Misconceptions
- In irritable bowel syndrome: no study has established a link between spicy food and symptoms.
- In gastroesophageal reflux disease (GERD): spices may induce burns, but not GERD. Spices can indeed trigger pain when there are existing lesions, such as esophagitis or gastric ulceration. Spices are not involved in the pathophysiology of lesions, but may reveal these lesions by their hyperemic effects [126].
- Spices have no involvement in the pathophysiology of ulcers. Some studies have shown that spices may increase mucus secretion and have a protective role. Capsaicin has been reported to inhibit acid secretion, stimulate mucus secretion and gastric mucosal blood flow, thereby helping in the prevention and healing of gastric ulcers [127]. The metabolic pathway involves prostaglandin E2 and prostacyclins in conjunction with EP1 and the IP receptor [128,129]. Spices (pepper, chili) thus appear to have a protective effect on the gastric mucosa.
3.7. Oral Mucositis
3.8. Regarding the Acidity of Food
- Recommendation 18: It is recommended not to exclude pulp-free fruit juices, potatoes, white bread, milk and dairy products from a “strict low-fiber” diet (10–14 g fiber/d, commonly called low-residue or no-residue diet).
3.9. Definition and Semantic Choices
- Edible carbohydrate polymers, naturally occurring in the food as consumed;
- Edible carbohydrate polymers which have been obtained from raw food material by physical, enzymatic or chemical means and have a beneficial physiological effect demonstrated by generally accepted scientific evidence;
- Edible synthetic carbohydrate polymers which have a beneficial physiological effect demonstrated by generally accepted scientific evidence.
- The elimination of the designation “no residue”: this designation should no longer be used because all foods generate residues.
- The term “low-fiber” replaces that of “low-residue”. In some countries such as the United States, the learned societies in charge of reflecting on these themes have proposed since 2011 to prefer the term “low-fiber” to that of “low-residue”; indeed, unlike fiber, there is no consensus scientific methodology to accurately assess the residue content of a meal [132,138]. Similarly to this search for harmonization across the Atlantic, and anxious to base itself on a quantifiable scientific definition, it is proposed to no longer use the designation “low-residue” diet.
- To define fiber intake thresholds according to the level of restriction:
- “strict low-fiber” corresponds to intakes of 10 to 14 g/d of fiber;
- “low-fiber” corresponds to intakes of 15 to 20 g/d of fiber;
- As a reminder, most countries recommend that healthy adults, as part of a balanced diet, consume 25 to 35 g/d of dietary fiber daily [134]. In France, ANSES recommends a consumption of 30 g/d while the average consumption among French adults according to the INCA3 survey is 20 g/d [140,141]. The “strict low-fiber” diet (10 to 14 g/d):
- Recommendation 19: It is recommended to reserve the “strict low-fiber” diet (10 to 14 g/d of fiber) for therapeutic purposes in symptomatic intestinal strictures; for diagnostic purposes in certain GI explorations (colonoscopy, CT colonography, MR enterography (MRE), etc.); or for symptomatic purposes.
- Recommendation 20: Per medical prescription, a low-fiber diet (15–20 g fiber/day) may be indicated during hospital stay, in terms of digestive symptoms.
- Recommendation 21: It is recommended not to exclude all dairy products in the context of lactose intolerance.
- Recommendation 22: Aside from medically diagnosed celiac disease, a gluten-free diet is not recommended.
- Recommendation 23: It is recommended to propose, in terms of food provision, an energy- and/or protein-enriched diet in order to meet the requirements of prevention and management of undernutrition.
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nardin, A.; Flandrin, J.L.; Loux, F.; Beauvalet-Boutouyrie, S.; Maes, G.; Corbeau, J.P.; Apfelbaum, M.; Arnould, P.; Tronchon, P. L’Appétit Vient en Mangeant! Histoire de l’Alimentation à L’Hôpital. XVe-XXe Siècles. Exposition, Musée de L’assistance Publique, 7 Novembre 1997–26 Avril 1998; Musée de l’Assistance Publique-Hôpitaux de Paris, Editions Doin: Paris, France, 1997; p. 179. [Google Scholar]
- Preiser, J.C.; Hiesmayr, M.; Beau De Luca, A.P. Épidémiologie de la dénutrition chez le malade hospitalisé. In Traité de Nutrition Clinique; SFNEP: Paris, France, 2016; pp. 2071–2115. [Google Scholar]
- Vaillant, M.-F.; Hennequin, V.; Duval, C.; Rossi, F.; Fontaine, E.; Thibault, R.; Quilliot, D. État des lieux des pratiques alimentaires et régimes dans les établissements de santé français: Enquête nationale 2017. Nutr. Clin. Metab. 2019, 33, 131–138. [Google Scholar] [CrossRef]
- Haute Autorité de Santé. Elaboration de Recommandations de Bonne Pratique. Méthode «Recommandations par Consensus Formalisé» (Internet). 2010. Available online: https://www.has-sante.fr/upload/docs/application/pdf/2011-11/guide_methodologique_cf_40_pages_2011-11-03_15-40-2_278.pdf (accessed on 1 September 2018).
- Keller, U.; Lüthy, J.; Meier, R.; Rosé, B.; Sterchi, A.B. La Dénutrition à L’hôpital: Conclusions d’un Groupe D’experts du Conseil de L’europe et Recommandations de la Commission Fédérale de L’alimentation. (Internet). Office Fédéral de la Santé Publique. 2006. Available online: https://www.eek.admin.ch/dam/eek/fr/dokumente/.../mangelernaehrung-spital.pdf (accessed on 12 May 2019).
- Conseil National de l’Alimentation. Avis N°78 du 04 Juillet 2017: Alimentation en Milieu Hospitalier (Internet). 2017. Available online: http://www.cna-alimentation.fr/v2/wp-content/uploads/2017/07/CNA_Avis78.pdf (accessed on 12 May 2019).
- Hiesmayr, M.; Schindler, K.; Pernicka, E.; Schuh, C.; Schoeniger-Hekele, A.; Bauer, P.; Laviano, A.; Lovell, A.D.; Mouhieddine, M.; Schuetz, T.; et al. Decreased food intake is a risk factor for mortality in hospitalised patients: The NutritionDay survey 2006. Clin. Nutr. 2009, 28, 484–491. [Google Scholar] [CrossRef]
- Agarwal, E.; Ferguson, M.; Banks, M.; Batterham, M.; Bauer, J.; Capra, S.; Insering, E. Malnutrition and poor food intake are associated with prolonged hospital stay, frequent readmissions, and greater in-hospital mortality: Results from the Nutrition Care Day Survey 2010. Clin. Nutr. 2013, 32, 737–745. [Google Scholar] [CrossRef] [Green Version]
- Dupertuis, Y.M.; Kossovsky, M.P.; Kyle, U.G.; Raguso, C.A.; Genton, L.; Pichard, C. Food intake in 1707 hospitalised patients: A prospective comprehensive hospital survey. Clin. Nutr. Edinb. Scotl. 2003, 22, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Thibault, R.; Chikhi, M.; Clerc, A.; Darmon, P.; Chopard, P.; Genton, L.; Kossovsky, M.P.; Pichard, C. Assessment of food intake in hospitalised patients: A 10-year comparative study of a prospective hospital survey. Clin. Nutr. 2011, 30, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Zazzo, J.-F. Évaluation des apports alimentaires des malades hospitalisés. Nutr. Clin. Metab. 2003, 17, 213–217. [Google Scholar] [CrossRef]
- Coti Bertrand, P.; Lafaille, M.; Depraz-Cissoko, M.-P. Lutte contre les régimes restrictifs à l’hôpital: l’expérience du centre hospitalier universitaire de Lausanne. Nutr. Clin. Metab. 2015, 29, 54–57. [Google Scholar] [CrossRef]
- Rossi-Pacini, F.; Monier, L.; Agnetti, R.; Guex, E.; Genton, L.; Pichard, C. Conseils diététiques et adaptation alimentaire. In Traité de Nutrition Clinique; Springer: Paris, France, 2016; pp. 797–814. [Google Scholar]
- Guy Grand, B. Alimentation en Milieu Hospitalier: Rapport de Mission à Monsieur le Ministre Chargé de la Santé (Internet). 1997. Available online: https://www.ladocumentationfrancaise.fr/var/storage/rapports-publics/974060600.pdf (accessed on 12 May 2019).
- Crenn, P.; Poisson-Salomon, A.-S.; Puissant, M.-C. Prescription Médicale Diététique Pratique chez L’adulte à L’hôpital; Lamarre éditeur: Rueil-Malmaison, France, 2005. [Google Scholar]
- Jarrin, I.; Poiroux, F.; Mulhen, D.; Debellis, N.; Alric, P.; Augizeau, E.; Sauban, L.; Andrianmanantenasoa, M.; Drymon, S.; Berreby, L.; et al. Effet du choix des menus sur la prise alimentaire et la satisfaction des patients hospitalisés dans un service de médecine interne. Cah. Nutr. Diet. 2014, 49, 113–119. [Google Scholar] [CrossRef]
- Hugol-Gential, C. Donner le choix du repas à l’hôpital: Effet sur le gaspillage alimentaire et les représentations du patient. In Bien et bon à Manger: Penser Notre Alimentation du Quotidien à L’institution de Santé; Universitaires de Dijon: Dijon, France, 2018; p. 277. [Google Scholar]
- Stanga, Z. Hospital food: A survey of patients’ perceptions. Clin. Nutr. 2003, 22, 241–246. [Google Scholar] [CrossRef]
- Mosqueira, C.; Harris, L.R. Spoken Menu: A Menu Process Catered to The Patients Food Choices and Present Appetite. J. Am. Diet. Assoc. 1996, 96, A14. [Google Scholar] [CrossRef]
- Folio, D.; O’Sullivan-Maillet, J.; Touger-Decker, R. The spoken menu concept of patient foodservice delivery systems increases overall patient satisfaction, therapeutic and tray accuracy, and is cost neutral for food and labor. J. Am. Diet. Assoc. 2002, 102, 546–548. [Google Scholar] [CrossRef]
- Beck, A.M.; Balkn, U.N.; Fürst, P.; Hasunen, K.; Jones, L.; Keller, U.; Melchior, J.C.; Mikkelsen, B.E.; Schauder, P.; Sivonen, P.; et al. Food and nutritional care in hospitals: How to prevent undernutrition–report and guidelines from the Council of Europe. Clin. Nutr. 2001, 20, 455–460. [Google Scholar] [CrossRef]
- Dias-Ferreira, C.; Santos, T.; Oliveira, V. Hospital food waste and environmental and economic indicators—A Portuguese case study. Waste Manag. 2015, 46, 146–154. [Google Scholar] [CrossRef] [PubMed]
- ADEME et Ministère de l’Écologie, du Développement Durable et de l’Énergie. Guide Pratique «Réduire le Gaspillage Alimentaire en Restauration Collective» (Internet). 2015. Available online: https://www.ademe.fr/sites/default/files/assets/documents/reduire-gaspillage-alimentaire-en-restauration-collective_010256.pdf (accessed on 12 May 2019).
- Haut Conseil de la Santé Publique. Avis Relatif aux Objectifs de Santé Publique Quantifiés Pour la Politique Nutritionnelle de Santé Publique (PNNS) 2018–2022. (Internet). 2018. Available online: https://www.hcsp.fr/Explore.cgi/Telecharger?NomFichier=hcspa20180209_avisrelaauxobjequanpourlapolinut.pdf (accessed on 2 February 2019).
- Ministère Chargé de la Santé. PNNS–Un Plan Lancé Pour Améliorer L’état de Santé de la Population. (Internet). Santé Publique France. Available online: http://www.mangerbouger.fr/PNNS (accessed on 1 February 2019).
- Ministère de L’économie, de L’industrie et du Numérique. Recommandation Nutrition. Groupe D’étude des Marchés de Restauration Collective et Nutrition GEM-RCN (Internet). 2015. Available online: https://www.economie.gouv.fr/files/directions_services/daj/marches_publics/oeap/gem/nutrition/nutrition.pdf (accessed on 19 January 2019).
- Volkert, D.; Beck, A.M.; Cederholm, T.; Cruz-Jentoft, A.; Goisser, S.; Hooper, L.; Kiesswetter, E.; Maggio, M.; Raynaud-Simon, A.; Sieber, C.C.; et al. ESPEN guideline on clinical nutrition and hydration in geriatrics. Clin. Nutr. 2019, 38, 10–47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moran, A.; Lederer, A.; Johnson Curtis, C. Use of Nutrition Standards to Improve Nutritional Quality of Hospital Patient Meals: Findings from New York City’s Healthy Hospital Food Initiative. J. Acad. Nutr. Diet. 2015, 115, 1847–1854. [Google Scholar] [CrossRef] [PubMed]
- Direction de L’hospitalisation et de L’organisation des Soins (DHOS). Circulaire DHOS/E 1 n° 2002-186 du 29 Mars 2002 Relative à L’alimentation et à la Nutrition dans les Etablissements de Santé (Internet). Available online: https://solidarites-sante.gouv.fr/fichiers/bo/2002/02-16/a0161475.htm (accessed on 18 January 2019).
- Lowenfels, A.; Pattison, M.J.; Martin, A.M.; Ferrari, C. Improving the Food Environment in Hospitals and Senior Meal Programs. Prev. Chronic Dis. 2018, 15, 170429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ricour, C.; Zazzo, F. Mise en Place d’une Politique Nutritionnelle dans les Etablissements de Santé. (Internet). 2002. Available online: http://www.mangerbouger.fr/pro/IMG/pdf/RapportRicour-Nutritrion-EtablissementsSante.pdf (accessed on 12 May 2019).
- Bertin, E.; Delarue, J.; Piquet, M.A.; Schneider, S.; Laville, M.; Ziegler, O.; Pradignac, A.; Fontaine, E.; Joly, F.; Oppert, J.-M.; et al. Prescription d’un Régime Diététique; Item 179; Elsevier Masson: Paris, France, 2011; pp. 258–263. [Google Scholar]
- Schlienger, J.L. Prescription du Régime Alimentaire. In EMC Endocrinologie-Nutrition (Internet); Elsevier: Paris, France, 2014. [Google Scholar] [CrossRef]
- AFDN. Recommandations Pour la Pratique Clinique: Le Soin Diététique Réalisé par un Diététicien en Etablissement de Santé (Internet). 2011. Available online: http://www.afdn.org/fileadmin/pdf/soins-etbs-sante/1105-reco-soin-dietetique-ets-de-sante.pd (accessed on 10 January 2019).
- Ministère des Affaires Sociales et de la Santé-Direction Générale de L’offre de Soins. Synthèse Pédagogique sur L’organisation Transversale de la Nutrition au Sein des Etablissements de Santé et Médicosociaux (Internet). 2012. Available online: https://solidarites-sante.gouv.fr/IMG/pdf/synthese_pedagogique_organisation_transversale_nutrition_etablissements_de_sante_et_medico-sociaux.pdf (accessed on 19 January 2019).
- Code de la Santé Publique. Article 4371-1: Définition de la Profession de Diététicien. (Internet). Available online: https://www.legifrance.gouv.fr/affichCodeArticle.do?cidTexte=LEGITEXT000006072665&idArticle=LEGIARTI000006689536&dateTexte=&categorieLien=cid (accessed on 14 February 2019).
- Haute Autorité de Santé. Critère 19b-Troubles de l’état Nutritionnel. Manuel de Certification des Etablissements de Santé V2010 (Internet). 2014, p. 62. Available online: https://www.has-sante.fr/portail/upload/docs/application/pdf/2014-03/manuel_v2010_janvier2014.pdf (accessed on 12 May 2019).
- Rossi-Pacini, F.; Parmentier, I. Le soin diététique dans la prise en charge globale. In Traité de Nutrition Clinique; SFNEP: Paris, France, 2016; pp. 829–839. [Google Scholar]
- Kondrup, J.; Allison, S.P.; Elia, M.; Vellas, B.; Plauth, M. Educational and Clinical Practice Committee, European Society of Parenteral and Enteral Nutrition (ESPEN). ESPEN guidelines for nutrition screening 2002. Clin. Nutr. Edinb. Scotl. 2003, 22, 415–421. [Google Scholar] [CrossRef]
- Darmon, P.; Gorin-Gottraux, S.; Pichard, C. Y a-t-il une Place Pour les Régimes Alimentaires Restrictifs chez la Personne Agée? In Traité de Nutrition de la Personne Agée (Internet); Springer: Paris, France, 2009; pp. 131–140. Available online: http://link.springer.com/10.1007/978-2-287-98117-3_16 (accessed on 29 June 2019).
- ANSES. Avis de l’ANSES Relatif à L’actualisation des Repères Alimentaires du PNNS-Femmes Enceintes et Allaitantes (Internet). 2019. Available online: https://www.anses.fr/fr/system/files/NUT2017SA0141.pdf (accessed on 12 May 2019).
- Haute Autorité de Santé. Stratégie de Prise en Charge en cas de Dénutrition Protéino-Energétique chez la Personne Agée. (Internet). 2007. Available online: https://www.has-sante.fr/portail/upload/docs/application/pdf/denutrition_personne_agee_2007_-_recommandations.pdf (accessed on 11 January 2019).
- Cichero, J.; Lam, P.; Steele, C.M.; Hanson, B.; Chen, J.; Dantas, R.O.; Duivestein, J.; Kayashita, J.; Lecko, C.; Murray, J.; et al. Development of International Terminology and Definitions for Texture-Modified Foods and Thickened Fluids Used in Dysphagia Management: The IDDSI Framework. Dysphagia 2017, 32, 293–314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- European Society for Swallowing Disorders. ESSD Position Statements: Oropharyngeal Dysphagia in Adult Patients. Available online: http://www.myessd.org/docs/position_statements/ESSD_Position_Statements_on_OD_in_adult_patients_for_web.pdf (accessed on 15 July 2021).
- Cichero, J.; Steele, C.; Duivestein, J.; Clavé, P.; Chen, J.; Kayashita, J.; Dantas, R.; Lecko, C.; Speyer, R.; Lam, P.; et al. The Need for International Terminology and Definitions for Texture-Modified Foods and Thickened Liquids Used in Dysphagia Management: Foundations of a Global Initiative. Curr. Phys. Med. Rehabil. Rep. 2013, 1, 280–291. [Google Scholar] [CrossRef] [Green Version]
- Steele, C.M.; Alsanei, W.A.; Ayanikalath, S.; Barbon, C.E.A.; Chen, J.; Cichero, J.; Julie, A.Y.; Coutts, K.; Dantas, R.O.; Duivestein, J.; et al. The influence of food texture and liquid consistency modification on swallowing physiology and function: A systematic review. Dysphagia 2015, 30, 2–26. [Google Scholar] [CrossRef] [Green Version]
- Matta, J.; Zins, M.; Feral-Pierssens, A.L.; Carette, C.; Ozguler, A.; Goldberg, M.; Czernichow, S. Prévalence du surpoids, de l’obésité et des facteurs de risque cardio-métaboliques dans la cohorte Constances. Bull. Epidémiol. Hebd. 2016, 35, 640–646. [Google Scholar]
- Tardif, N.; Salles, J.; Guillet, C.; Gadéa, E.; Boirie, Y.; Walrand, S. Obésité sarcopénique et altérations du métabolisme protéique musculaire. Nutr. Clin. Metab. 2011, 25, 138–151. [Google Scholar] [CrossRef]
- Vaduva, P.; Tamion, F.; Thibault, R. Nutrition de l’obèse agressé. Nutr. Clin. Metab. 2017, 31, 294–302. [Google Scholar] [CrossRef]
- Jésus, P.; Coëffier, M. Comment évaluer les besoins énergétiques et protéiques du sujet obèse ? Nutr. Clin. Metab. 2017, 31, 260–267. [Google Scholar] [CrossRef]
- Batsis, J.A.; Villareal, D.T. Sarcopenic obesity in older adults: Aetiology, epidemiology and treatment strategies. Nat. Rev. Endocrinol. 2018, 14, 513–537. [Google Scholar] [CrossRef] [PubMed]
- Chambrier, C.; Sztark, F. Recommandations de bonnes pratiques cliniques sur la nutrition périopératoire. Actualisation 2010 de la conférence de consensus de 1994 sur la «Nutrition artificielle périopératoire en chirurgie programmée de l’adulte». Nutr. Clin. Metab. 2010, 24, 145–156. [Google Scholar] [CrossRef]
- Guex, E.; Kouadio, A.; Fierz, Y.; Coti Bertrand, P. Prise en charge diététique du sujet obèse à l’hôpital: Quels régimes ? Nutr. Clin. Metab. 2018, 32, 85–89. [Google Scholar] [CrossRef]
- Fierz, Y.; Kouadio, A.; Guex, E.; Larche, M.; Raya, A.; Pralong, F.P.; Coti-Bertrand, P. P062, La surcharge pondérale et l’obésité sont des facteurs de risque d’une non-couverture des besoins protéino-caloriques des patients hospitaliers. Nutr. Clin. Metab. 2014, 28, S100–S101. [Google Scholar] [CrossRef]
- Fouque, D.; Aparicio, M. Eleven reasons to control the protein intake of patients with chronic kidney disease. Nat. Clin. Pract. Nephrol. 2007, 3, 383–392. [Google Scholar] [CrossRef]
- Fouque, D.; Laville, M.; Boissel, J.P. Low protein diets for chronic kidney disease in non diabetic adults. Cochrane Database Syst. Rev. 2006, 2, CD001892. [Google Scholar]
- Garneata, L.; Stancu, A.; Dragomir, D.; Stefan, G.; Mircescu, G. Ketoanalogue-Supplemented Vegetarian Very Low-Protein Diet and CKD Progression. J. Am. Soc. Nephrol. JASN 2016, 27, 2164–2176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalantar-Zadeh, K.; Fouque, D. Nutritional Management of Chronic Kidney Disease. N. Engl. J. Med. 2017, 377, 1765–1776. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, D.S. Lowering the Bar on the Low-Fat Diet. JAMA 2016, 316, 2087. [Google Scholar] [CrossRef]
- Larstorp, A.C.K.; Tonstad, S. Effect of a Low Fat Diet Intervention on Blood Pressure and Hypertension: Rather Switch to a Mediterranean Diet? Am. J. Hypertens. 2016, 29, 900–903. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Esposito, K.; Maiorino, M.I.; Ciotola, M.; Di Palo, C.; Scognamiglio, P.; Gicchino, M.; Petrizzo, M.; Saccomanno, F.; Beneduce, F.; Ceriello, A.; et al. Effects of a Mediterranean-style diet on the need for antihyperglycemic drug therapy in patients with newly diagnosed type 2 diabetes: A randomized trial. Ann. Intern. Med. 2009, 151, 306–314. [Google Scholar] [CrossRef]
- Cugnet-Anceau, C.; Moret, M.; Moulin, P. Hypertriglyceridemia: Therapeutic strategy. Rev. Prat. 2011, 61, 1110–1116. [Google Scholar]
- Athyros, V.G.; Giouleme, O.I.; Nikolaidis, N.L.; Vasiliadis, T.V.; Bouloukos, V.I.; Kontopoulos, A.G.; Eugenidis, N.P. Long-term follow-up of patients with acute hypertriglyceridemia-induced pancreatitis. J. Clin. Gastroenterol. 2002, 34, 472–475. [Google Scholar] [CrossRef] [PubMed]
- Scherer, J.; Singh, V.P.; Pitchumoni, C.S.; Yadav, D. Issues in Hypertriglyceridemic Pancreatitis: An Update. J. Clin. Gastroenterol. 2014, 48, 195–203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bălănescu, N.R.; Topor, L.; Ulici, A.; Djendov, F.B. Acute pancreatitis secondary to hyperlipidemia in an 11-year-old girl: A case report and review of literature. J. Med. Life 2013, 6, 2–6. [Google Scholar]
- Dominguez-Muñoz, J.E.; Malfertheiner, P.; Ditschuneit, H.H.; Blanco-Chavez, J.; Uhl, W.; Büchler, M.; Ditschuneit, H. Hyperlipidemia in acute pancreatitis. Relationship with etiology, onset, and severity of the disease. Int. J. Pancreatol. Off. J. Int. Assoc. Pancreatol. 1991, 10, 261–267. [Google Scholar]
- Shah, A.S.; Wilson, D.P. Primary hypertriglyceridemia in children and adolescents. J. Clin. Lipidol. 2015, 9, S20–S28. [Google Scholar] [CrossRef] [PubMed]
- Pongprasobchai, S. Maldigestion from pancreatic exocrine insufficiency. J. Gastroenterol. Hepatol. 2013, 28 (Suppl. 4), 99–102. [Google Scholar] [CrossRef]
- Takuwa, T.; Yoshida, J.; Ono, S.; Hishida, T.; Nishimura, M.; Aokage, K.; Nagai, K. Low-fat diet management strategy for chylothorax after pulmonary resection and lymph node dissection for primary lung cancer. J. Thorac. Cardiovasc. Surg. 2013, 146, 571–574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Graziani, G.; Cucchiari, D.; Verdesca, S.; Balzarini, L.; Montanelli, A.; Ponticelli, C. Chyluria associated with nephrotic-range proteinuria: Pathophysiology, clinical picture and therapeutic options. Nephron. Clin. Pract. 2011, 119, c248–c253. [Google Scholar] [CrossRef]
- Vandenbos, F.; Rakotoarisoa, C.; Zeanandin, G.; Mourani, A. Un cas de chylothorax d’origine cirrhotique. Rev. Pneumol. Clin. 2005, 61, 369–371. [Google Scholar] [CrossRef]
- Bhardwaj, R.; Vaziri, H.; Gautam, A.; Ballesteros, E.; Karimeddini, D.; Wu, G.Y. Chylous Ascites: A Review of Pathogenesis, Diagnosis and Treatment. J. Clin. Transl. Hepatol. 2018, 6, 105–113. [Google Scholar] [CrossRef] [Green Version]
- Lizaola, B.; Bonder, A.; Trivedi, H.D.; Tapper, E.B.; Cardenas, A. Review article: The diagnostic approach and current management of chylous ascites. Aliment. Pharmacol. Ther. 2017, 46, 816–824. [Google Scholar] [CrossRef] [Green Version]
- Al-Busafi, S.A.; Ghali, P.; Deschênes, M.; Wong, P. Chylous Ascites: Evaluation and Management. ISRN Hepatol. 2014, 2014, 240473. [Google Scholar] [CrossRef] [Green Version]
- Zeanandin, G.; Thibault, R.; Caldari, D.; Coti, P.; Guex, E.; Bachmann, P.; Quilliot, D. Prise en charge nutritionnelle d’une ascite chyleuse. Nutr. Clin. Metab. 2016, 30, 83–87. [Google Scholar] [CrossRef]
- Jackson, A.; Lalji, A.; Kabir, M.; Muls, A.; Gee, C.; Vyoral, S.; Shaw, C.; Andreyev, H.J.N. The efficacy of a low-fat diet to manage the symptoms of bile acid malabsorption-outcomes in patients previously treated for cancer. Clin. Med. Lond. Engl. 2017, 17, 412–418. [Google Scholar] [CrossRef] [Green Version]
- Sackner-Bernstein, J.; Kanter, D.; Kaul, S. Dietary Intervention for Overweight and Obese Adults: Comparison of Low-Carbohydrate and Low-Fat Diets. A Meta-Analysis. PLoS ONE. 2015, 10, e0139817. [Google Scholar] [CrossRef]
- Bueno, N.B.; de Melo, I.S.V.; de Oliveira, S.L.; da Rocha Ataide, T. Very-low-carbohydrate ketogenic diet v. low-fat diet for long-term weight loss: A meta-analysis of randomised controlled trials. Br. J. Nutr. 2013, 110, 1178–1187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bazzano, L.A.; Hu, T.; Reynolds, K.; Yao, L.; Bunol, C.; Liu, Y.; Chen, C.S.; Klag, M.J.; Whelton, P.K.; He, J. Effects of low-carbohydrate and low-fat diets: A randomized trial. Ann. Intern. Med. 2014, 161, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Tobias, D.K.; Chen, M.; Manson, J.E.; Ludwig, D.S.; Willett, W.; Hu, F.B. Effect of low-fat diet interventions versus other diet interventions on long-term weight change in adults: A systematic review and meta-analysis. Lancet Diabetes Endocrinol. 2015, 3, 968–979. [Google Scholar] [CrossRef] [Green Version]
- Wu, L.; Ma, D.; Walton-Moss, B.; He, Z. Effects of low-fat diet on serum lipids in premenopausal and postmenopausal women: A meta-analysis of randomized controlled trials. Menopause 2014, 21, 89–99. [Google Scholar] [CrossRef]
- Yancy, W.S.; Olsen, M.K.; Guyton, J.R.; Bakst, R.P.; Westman, E.C. A low-carbohydrate, ketogenic diet versus a low-fat diet to treat obesity and hyperlipidemia: A randomized, controlled trial. Ann. Intern. Med. 2004, 140, 769–777. [Google Scholar] [CrossRef]
- Davis, N.J.; Tomuta, N.; Schechter, C.; Isasi, C.R.; Segal-Isaacson, C.J.; Stein, D.; Zonszein, J.; Wylie-Rosett, J. Comparative study of the effects of a 1-year dietary intervention of a low-carbohydrate diet versus a low-fat diet on weight and glycemic control in type 2 diabetes. Diabetes Care 2009, 32, 1147–1152. [Google Scholar] [CrossRef] [Green Version]
- Guldbrand, H.; Lindström, T.; Dizdar, B.; Bunjaku, B.; Östgren, C.J.; Nystrom, F.H.; Bachrach-Lindström, M. Randomization to a low-carbohydrate diet advice improves health related quality of life compared with a low-fat diet at similar weight-loss in Type 2 diabetes mellitus. Diabetes Res. Clin. Pract. 2014, 106, 221–227. [Google Scholar] [CrossRef] [Green Version]
- Chlebowski, R.T.; Aragaki, A.K.; Anderson, G.L.; Thomson, C.A.; Manson, J.E.; Simon, M.S.; Howard, B.V.; Rohan, T.E.; Snetselar, L.; Lane, D.; et al. Low-Fat Dietary Pattern and Breast Cancer Mortality in the Women’s Health Initiative Randomized Controlled Trial. J. Clin. Oncol. 2017, 35, 2919–2926. [Google Scholar] [CrossRef] [Green Version]
- Romero-Gómez, M.; Zelber-Sagi, S.; Trenell, M. Treatment of NAFLD with diet, physical activity and exercise. J. Hepatol. 2017, 67, 829–846. [Google Scholar] [CrossRef] [Green Version]
- Zelber-Sagi, S.; Salomone, F.; Mlynarsky, L. The Mediterranean dietary pattern as the diet of choice for non-alcoholic fatty liver disease: Evidence and plausible mechanisms. Liver. Int. 2017, 37, 936–949. [Google Scholar] [CrossRef] [Green Version]
- Papamiltiadous, E.S.; Roberts, S.K.; Nicoll, A.J.; Ryan, M.C.; Itsiopoulos, C.; Salim, A.; Tierney, A.C. A randomised controlled trial of a Mediterranean Dietary Intervention for Adults with Non Alcoholic Fatty Liver Disease (MEDINA): Study protocol. BMC Gastroenterol. 2016, 16, 14. [Google Scholar] [CrossRef] [Green Version]
- Gibson, P.S.; Lang, S.; Dhawan, A.; Fitzpatrick, E.; Blumfield, M.L.; Truby, H.; Hart, K.H.; Moore, J.B. Systematic Review: Nutrition and Physical Activity in the Management of Paediatric Nonalcoholic Fatty Liver Disease. J. Pediatr. Gastroenterol. Nutr. 2017, 65, 141–149. [Google Scholar] [CrossRef]
- Errazuriz, I.; Dube, S.; Slama, M.; Visentin, R.; Nayar, S.; O’Connor, H.; Cobelli, C.; Das, S.K.; Basu, A.; Kremers, W.K.; et al. Randomized Controlled Trial of a MUFA or Fiber-Rich Diet on Hepatic Fat in Prediabetes. J. Clin. Endocrinol. Metab. 2017, 102, 1765–1774. [Google Scholar]
- van Herpen, N.A.; Schrauwen-Hinderling, V.B.; Schaart, G.; Mensink, R.P.; Schrauwen, P. Three weeks on a high-fat diet increases intrahepatic lipid accumulation and decreases metabolic flexibility in healthy overweight men. J. Clin. Endocrinol. Metab. 2011, 96, E691–E695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Westerbacka, J.; Lammi, K.; Häkkinen, A.-M.; Rissanen, A.; Salminen, I.; Aro, A.; Yki-Järvinen, H. Dietary fat content modifies liver fat in overweight nondiabetic subjects. J. Clin. Endocrinol. Metab. 2005, 90, 2804–2809. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herman, W.H.; Kalyani, R.R.; Cherrington, A.L.; Coustan, D.R.; de Boer, I.; Dudl, R.J.; Feldman, H.; Florez, H.J.; Koliwad, S.; Maryniuk, M.; et al. Introduction. Diabetes Care 2017, 40 (Suppl. 1), S1–S2. [Google Scholar]
- ANSES. Actualisation des Repères du PNNS: Établissement de Recommandations D’apport de Sucres (Internet). 2016. Available online: https://www.anses.fr/fr/system/files/NUT2012SA0186Ra.pdf (accessed on 12 May 2019).
- Référentiel de bonnes Pratiques-Nutrition & Diététique Diabète de type 2 de l’adulte-Nutrition-Alimentation-Comportement alimentaire-Éducation Thérapeutique-Évaluation des Pratiques. Med. Mal. Metab. 2014, 8, 1–75.
- Bertoglio, J.; Martineau, C. Regard nouveau sur les glucides pour la pratique en Diabétologie. Med. Mal. Metab. 2017, 11, 564–565. [Google Scholar] [CrossRef]
- Ley, S.H.; Hamdy, O.; Mohan, V.; Hu, F.B. Prevention and management of type 2 diabetes: Dietary components and nutritional strategies. Lancet 2014, 383, 1999–2007. [Google Scholar] [CrossRef] [Green Version]
- Trumbo, P.R.; Rivers, C.R. Systematic review of the evidence for an association between sugar-sweetened beverage consumption and risk of obesity. Nutr. Rev. 2014, 72, 566–574. [Google Scholar] [CrossRef] [Green Version]
- Slama, G.; Jean-Joseph, P.; Goicolea, I.; Elgrably, F.; Haardt, M.J.; Costagliola, D.; Bornet, F.; Tchobroutsky, G. Sucrose taken during mixed meal has no additional hyperglycaemic action over isocaloric amounts of starch in well-controlled diabetics. Lancet 1984, 324, 122–125. [Google Scholar] [CrossRef]
- Filardi, T.; Panimolle, F.; Crescioli, C.; Lenzi, A.; Morano, S. Gestational Diabetes Mellitus: The Impact of Carbohydrate Quality in Diet. Nutrients 2019, 11, 1549. [Google Scholar] [CrossRef] [Green Version]
- Mahtani, K.R.; Heneghan, C.; Onakpoya, I.; Tierney, S.; Aronson, J.K.; Roberts, N.; Richard Hobbs, F.D.; Nunan, D. Reduced Salt Intake for Heart Failure: A Systematic Review. JAMA Intern. Med. 2018, 178, 1693. [Google Scholar] [CrossRef]
- Hummel, S.L.; Karmally, W.; Gillespie, B.W.; Helmke, S.T.; Teruya, S.; Wessler, J.D.; Wells, J.M.; Jimenez, O.; Trumble, E.; Cornellier, M.; et al. Geriatric Out-of-Hospital Randomized Meal Trial in Heart Failure (GOURMET-HF) Pilot Randomized Trial: Primary Results. J. Card. Fail. 2017, 23, 830. [Google Scholar] [CrossRef]
- Colin-Ramirez, E.; Ezekowitz, J.A. Rationale and design of the Study of Dietary Intervention Under 100 MMOL in Heart Failure (SODIUM-HF). Am. Heart J. 2018, 205, 87–96. [Google Scholar] [CrossRef]
- Butler, J.; Papadimitriou, L.; Georgiopoulou, V.; Skopicki, H.; Dunbar, S.; Kalogeropoulos, A. Comparing Sodium Intake Strategies in Heart Failure: Rationale and Design of the Prevent Adverse Outcomes in Heart Failure by Limiting Sodium (PROHIBIT) Study. Circ. Heart Fail. 2015, 8, 636–645. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, D.; Georgiopoulou, V.V.; Kalogeropoulos, A.P.; Dunbar, S.B.; Reilly, C.M.; Sands, J.M.; Fonarow, G.C.; Jessup, M.; Gheorghiade, M.; Yancy, C.; et al. Dietary Sodium Intake in Heart Failure. Circulation 2012, 126, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.F.; Coats, A.J.S.; Falk, V.; Gonzales-Juanetey, J.R.; Harjola, V.P.; Jankowska, E.A.; et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC)Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2016, 37, 2129–2200. [Google Scholar] [PubMed]
- McMurray, J.J.V.; Adamopoulos, S.; Anker, S.D.; Auricchio, A.; Bohm, M.; Dicksktein, K.; Falk, V.; Filippatos, G.; Fonseca, C.; Gomez-Sanchez, M.A.; et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012, The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2012, 33, 1787–1847. [Google Scholar] [PubMed]
- Atherton, J.J.; Sindone, A.; De Pasquale, C.G.; Driscoll, A.; MacDonald, P.S.; Hopper, I.; Kistler, P.M.; Briffa, T.; Wong, J.; Abhayaratna, W.; et al. National Heart Foundation of Australia and Cardiac Society of Australia and New Zealand: Guidelines for the Prevention, Detection, and Management of Heart Failure in Australia 2018. Heart Lung Circ. 2018, 27, 1123–1208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- National Heart Foundation of Australia and the Cardiac Society of Australia and New Zealand (Chronic Heart Failure Guidelines Expert Writing). Guidelines for the Prevention, Detection and Management of Chronic Heart Failure in Australia. (Internet). 2011. Available online: https://www.heartfoundation.org.au/images/uploads/publications/Chronic_Heart_Failure_Guidelines_2011.pdf (accessed on 3 February 2019).
- Angeli, P.; Bernardi, M.; Villanueva, C.; Francoz, C.; Mookerjee, R.P.; Trebicka, J.; Krag, A.; Laleman, W.; Gines, P. EASL Clinical Practice Guidelines for the management of patients with decompensated cirrhosis. J. Hepatol. 2018, 69, 406–460. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rondanelli, M.; Opizzi, A.; Perna, S.; Faliva, M.; Solerte, S.B.; Fioravanti, M.; Klersy, C.; Edda, C.; Maddalena, P.; Luciano, S.; et al. Acute Effect on Satiety, Resting Energy Expenditure, Respiratory Quotient, Glucagon-Like Peptide-1, Free Fatty Acids, and Glycerol Following Consumption of a Combination of Bioactive Food Ingredients in Overweight Subjects. J. Am. Coll. Nutr. 2013, 32, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, A.; Arguin, H.; Panahi, S. Capsaicinoids: A spicy solution to the management of obesity? Int. J. Obes. 2016, 40, 1198–1204. [Google Scholar] [CrossRef] [PubMed]
- Prakash, U.N.S.; Srinivasan, K. Enhanced intestinal uptake of iron, zinc and calcium in rats fed pungent spice principles--piperine, capsaicin and ginger (Zingiber officinale). J. Trace Elem. Med. Biol. 2013, 27, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Veda, S.; Srinivasan, K. Influence of dietary spices on the in vivo absorption of ingested β-carotene in experimental rats. Br. J. Nutr. 2011, 105, 1429–1438. [Google Scholar] [CrossRef] [Green Version]
- Reddy, A.C.; Lokesh, B.R. Studies on spice principles as antioxidants in the inhibition of lipid peroxidation of rat liver microsomes. Mol. Cell Biochem. 1992, 111, 117–124. [Google Scholar]
- Joe, B.; Lokesh, B.R. Role of capsaicin, curcumin and dietary n-3 fatty acids in lowering the generation of reactive oxygen species in rat peritoneal macrophages. Biochim. Biophys. Acta 1994, 1224, 255–263. [Google Scholar] [CrossRef]
- Xue, Y.; He, T.; Yu, K.; Zhao, A.; Zheng, W.; Zhang, Y.; Zhy, B. Association between spicy food consumption and lipid profiles in adults: A nationwide population-based study. Br. J. Nutr. 2017, 118, 144–153. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.; Chen, Z.-Y. Roles of Spicy Foods and Their Bioactive Compounds in Management of Hypercholesterolemia. J. Agric. Food Chem. 2018, 66, 8662–8671. [Google Scholar] [CrossRef]
- Li, Q.; Cui, Y.; Jin, R.; Lang, H.; Yu, H.; Sun, F.; He, C.; Ma, T.; Li, Y.; Zhou, X.; et al. Enjoyment of Spicy Flavor Enhances Central Salty-Taste Perception and Reduces Salt Intake and Blood Pressure. Hypertension 2017, 70, 1291–1299. [Google Scholar] [CrossRef]
- Friedman, J.R.; Nolan, N.A.; Brown, K.C.; Miles, S.L.; Akers, A.T.; Colclough, K.W.; Seidler, J.M.; Rimoldi, J.M.; Valentovic, M.A.; Dasgupta, P. Anticancer Activity of Natural and Synthetic Capsaicin Analogs. J. Pharmacol. Exp. Ther. 2018, 364, 462–473. [Google Scholar] [CrossRef]
- Banik, U.; Parasuraman, S.; Adhikary, A.K.; Othman, N.H. Curcumin: The spicy modulator of breast carcinogenesis. J. Exp. Clin. Cancer Res. 2017, 36, 98. [Google Scholar] [CrossRef] [Green Version]
- Suresh, D.; Srinivasan, K. Influence of curcumin, capsaicin, and piperine on the rat liver drug-metabolizing enzyme system in vivo and in vitro. Can. J. Physiol. Pharmacol. 2006, 84, 1259–1265. [Google Scholar] [CrossRef] [PubMed]
- Shamsi, S.; Tran, H.; Tan, R.S.J.; Tan, Z.J.; Lim, L.Y. Curcumin, Piperine, and Capsaicin: A Comparative Study of Spice-Mediated Inhibition of Human Cytochrome P450 Isozyme Activities. Drug Metab. Dispos. Biol. Fate Chem. 2017, 45, 49–55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jensen-Jarolim, E.; Gajdzik, L.; Haberl, I.; Kraft, D.; Scheiner, O.; Graf, J. Hot spices influence permeability of human intestinal epithelial monolayers. J. Nutr. 1998, 128, 577–581. [Google Scholar] [CrossRef] [Green Version]
- Deng, Y.; Huang, X.; Wu, H.; Zhao, M.; Lu, Q.; Israeli, E.; Dahan, S.; Blank, M.; Shoenfeld, Y. Some like it hot: The emerging role of spicy food (capsaicin) in autoimmune diseases. Autoimmun. Rev. 2016, 15, 451–456. [Google Scholar] [CrossRef]
- Surdea-Blaga, T.; Negrutiu, D.E.; Palage, M.; Dumitrascu, D.L. Food and Gastroesophageal Reflux Disease. Curr. Med. Chem. 2019, 26, 3497–3511. [Google Scholar] [CrossRef]
- Srinivasan, K. Biological Activities of Red Pepper (Capsicum annuum) and Its Pungent Principle Capsaicin: A Review. Crit. Rev. Food Sci. Nutr. 2016, 56, 1488–1500. [Google Scholar] [CrossRef]
- Takeuchi, K. Gastric cytoprotection by prostaglandin E₂ and prostacyclin: Relationship to EP1 and IP receptors. J. Physiol. Pharmacol. 2014, 65, 3–14. [Google Scholar] [PubMed]
- Takeuchi, K. Prostaglandin EP receptors and their roles in mucosal protection and ulcer healing in the gastrointestinal tract. Adv. Clin. Chem. 2010, 51, 121–144. [Google Scholar] [PubMed]
- Chan, C.W.H.; Chang, A.M.; Molassiotis, A.; Lee, I.Y.M.; Lee, G.C.T. Oral complications in Chinese cancer patients undergoing chemotherapy. Support. Care Cancer 2003, 11, 48–55. [Google Scholar] [CrossRef]
- Shen, P.; Walker, G.D.; Yuan, Y.; Reynolds, C.; Stacey, M.A.; Reynolds, E.C. Food acid content and erosive potential of sugar-free confections. Aust. Dent. J. 2017, 62, 215–222. [Google Scholar] [CrossRef] [Green Version]
- Vanhauwaert, E.; Matthys, C.; Verdonck, L.; De Preter, V. Low-Residue and Low-Fiber Diets in Gastrointestinal Disease Management. Adv. Nutr. 2015, 6, 820–827. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Règlement (UE) n° 1169/2011 du Parlement Européen et du Conseil du 25 Octobre 2011 Concernant L’Information des Consommateurs sur les Denrées Alimentaires. (Internet). Available online: http://data.europa.eu/eli/reg/2011/1169/oj (accessed on 10 January 2019).
- EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA). Scientific Opinion on Dietary Reference Values for Carbohydrates and Dietary Fibre. EFSA J. 2010, 8, 1462. [Google Scholar]
- O’Grady, J.; O’Connor, E.M.; Shanahan, F. Review article: Dietary fibre in the era of microbiome science. Aliment. Pharmacol. Ther. 2019, 49, 506–515. [Google Scholar] [CrossRef] [Green Version]
- Stephen, A.M.; Champ, M.M.-J.; Cloran, S.J.; Fleith, M.; van Lieshout, L.; Mejborn, H.; Burley, V. Dietary fibre in Europe: Current state of knowledge on definitions, sources, recommendations, intakes and relationships to health. Nutr. Res. Rev. 2017, 30, 149–190. [Google Scholar] [CrossRef] [PubMed]
- Gomila, S.; Romon, M. Le régime sans résidus. Cah. Nutr. Diet. 2004, 39, 290–292. [Google Scholar] [CrossRef]
- Cunningham, E. Are low-residue diets still applicable? J. Acad. Nutr. Diet. 2012, 112, 960. [Google Scholar] [CrossRef]
- Leszczynski, A.M.; MacArthur, K.L.; Nelson, K.P.; Schueler, S.A.; Quatromoni, P.A.; Jacobson, B.C. The association among diet, dietary fiber, and bowel preparation at colonoscopy. Gastrointest. Endosc. 2018, 88, 685–694. [Google Scholar] [CrossRef]
- Agence Nationale de Sécurité Sanitaire, Alimentation, Environnement, Travail (ANSES). Chapitre 3 : Equilibre Entre les Macronutriments Recommandations D’Apport en Fibres. In Actualisation des Repères du PNNS : Elaboration des Références Nutritionnelles (Internet); ANSES: Maisons-Alfort, France, 2016; 196p, Available online: https://www.anses.fr/fr/system/files/NUT2012SA0103Ra-1.pdf (accessed on 10 January 2019).
- ANSES. Étude Individuelle Nationale des Consommations Alimentaires: Avis de l’ANSES, Rapport D’expertise Collective (Internet). ANSES Edition. 2017. Available online: https://www.anses.fr/fr/system/files/NUT2014SA0234Ra.pdf (accessed on 10 January 2019).
- Flourié, B. Régimes sans résidus: Quelle définition, quelles indications ? Gastroentérol. Clin. Biol. 1999, 23, 124–129. [Google Scholar]
- Morin, M.C. Evolution du régime sans résidu. Inf. Diet. 2001, 4, 2–5. [Google Scholar]
- Frexinos, J. Alimentation et troubles fonctionnels digestifs. Cah. Nutr. Diet. 2004, 39, 307–310. [Google Scholar] [CrossRef]
- Lairon, D.; Cherbut, C.; Barry, J.L. Fibres alimentaires. In Apports Nutritionnels Conseillés Pour la Population Française, 3rd ed.; Tec et Doc Lavoisier: Paris, France, 2014; pp. 99–108. [Google Scholar]
- Melchior, C.; Maccarone, M.; Lemaitre, C.; Ducrotté, P. Que reste-t-il en 2015 des régimes en hépatogastroentérologie ? Nutr. Clin. Metab. 2015, 29, 101–108. [Google Scholar] [CrossRef]
- Saltzman, J.R.; Cash, B.D.; Pasha, S.F.; Early, D.S.; Muthusamy, V.R.; Khashab, M.A.; Chathadi, K.V.; Fanelli, R.D.; Chandrasekhara, V.; Lightdale, J.R.; et al. Bowel preparation before colonoscopy. Gastrointest. Endosc. 2015, 81, 781–794. [Google Scholar] [CrossRef] [PubMed]
- Haute Autorité de Santé. Prise en Charge Médicale et Chirurgicale Diverticulite (Internet). 2017. Available online: https://www.has-sante.fr/portail/upload/docs/application/pdf/2017-12/prise_en_charge_medicale_et_chirurgicale_-_argumentaire.pdf (accessed on 10 January 2019).
- Tarleton, S.; DiBaise, J.K. Low-residue diet in diverticular disease: Putting an end to a myth. Nutr. Clin. Pract. 2011, 26, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Forbes, A.; Escher, J.; Hébuterne, X.; Kłęk, S.; Krznaric, Z.; Schneider, S.; Shamir, R.; Stardelova, K.; Wierdsma, N.; Wiskin, A.E.; et al. ESPEN guideline: Clinical nutrition in inflammatory bowel disease. Clin. Nutr. 2017, 36, 321–347. [Google Scholar] [CrossRef] [Green Version]
- Pironi, L.; Arends, J.; Bozzetti, F.; Cuerda, C.; Gillanders, L.; Jeppesen, P.B.; Joly, F.; Kelly, D.; Lal, S.; Staun, M.; et al. ESPEN guidelines on chronic intestinal failure in adults. Clin. Nutr. 2016, 35, 247–307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beau, P. Traitement diététique et médicamenteux du syndrome du grêle court. Nutr. Clin. Metab. 2000, 14, 289–295. [Google Scholar] [CrossRef]
- Strate, L.L.; Morris, A.M. Epidemiology, Pathophysiology and Treatment of Diverticulitis. Gastroenterology 2019, 156, 1282–1298.e1. [Google Scholar] [CrossRef] [Green Version]
- Swanson, S.M.; Strate, L.L. Acute Colonic Diverticulitis. Ann. Intern. Med. 2018, 168, ITC65. [Google Scholar] [CrossRef]
- Dahl, C.; Crichton, M.; Jenkins, J.; Nucera, R.; Mahoney, S.; Marx, W.; Marshall, S. Evidence for Dietary Fibre Modification in the Recovery and Prevention of Reoccurrence of Acute, Uncomplicated Diverticulitis: A Systematic Literature Review. Nutrients 2018, 10, 137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Règlement CE n°1924/2006 du Parlement Européen et du Conseil du 20 Décembre 2006 Concernant les Allégations Nutritionnelles et de Santé Portant sur les Denrées Alimentaires (Internet). Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2007,012,0003,0018,FR:PDF (accessed on 10 January 2019).
- Table de Composition du CIQUAL (Internet). Available online: https://ciqual.anses.fr/#/constituants/34100/fibres-alimentaires-(g-100g) (accessed on 10 January 2019).
- Bharadwaj, S.; Meka, K.; Tandon, P.; Rathur, A.; Rivas, J.M.; Vallabh, H.; Jevenn, A.; Guirguis, J.; Sunesara, I.; Nischnick, A.; et al. Management of gastroparesis-associated malnutrition. J. Dig. Dis. 2016, 17, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Camilleri, M.; Parkman, H.P.; Shafi, M.A.; Abell, T.L.; Gerson, L. American College of Gastroenterology. Clinical guideline: Management of gastroparesis. Am. J. Gastroenterol. 2013, 108, 18–37. [Google Scholar] [CrossRef] [Green Version]
- Ducrotté, P.; Gourcerol, G. Gastroparésie: Physiopathologie et traitement. Presse Med. 2012, 41, 721–729. [Google Scholar] [CrossRef]
- Szilagyi, A. Review article: Lactose—A potential prebiotic. Aliment. Pharmacol. Ther. 2002, 16, 1591–1602. [Google Scholar] [CrossRef]
- Szilagyi, A. Adaptation to Lactose in Lactase Non Persistent People: Effects on Intolerance and the Relationship between Dairy Food Consumption and Evalution of Diseases. Nutrients 2015, 7, 6751–6779. [Google Scholar] [CrossRef] [Green Version]
- Lule, V.K.; Garg, S.; Tomar, S.K.; Khedkar, C.D.; Nalage, D.N. Food Intolerance: Lactose Intolerance. In Encyclopedia of Food and Health (Internet); Elsevier: Amsterdam, The Netherlands, 2016; pp. 43–48. Available online: https://linkinghub.elsevier.com/retrieve/pii/B9780123849472003123 (accessed on 29 June 2019).
- Rezaie, A.; Buresi, M.; Lembo, A.; Lin, H.; McCallum, R.; Rao, S.; Schmulson, M.; Valdovinos, M.; Zakko, S.; Pimentel, M. Hydrogen and Methane-Based Breath Testing in Gastrointestinal Disorders: The North American Consensus. Am. J. Gastroenterol. 2017, 112, 775–784. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dainese-Plichon, R.; Schneider, S.; Piche, T.; Hébuterne, X. Malabsorption et intolérance au lactose chez l’adulte. Nutr. Clin. Metab. 2014, 28, 46–51. [Google Scholar] [CrossRef]
- Levitt, M.; Wilt, T.; Shaukat, A. Clinical Implications of Lactose Malabsorption Versus Lactose Intolerance. J. Clin. Gastroenterol. 2013, 47, 471–480. [Google Scholar] [CrossRef]
- Wedlake, L.; Thomas, K.; McGough, C.; Andreyev, H.J.N. Small bowel bacterial overgrowth and lactose intolerance during radical pelvic radiotherapy: An observational study. Eur. J. Cancer 2008, 44, 2212–2217. [Google Scholar] [CrossRef] [PubMed]
- Marteau, P.; Messing, B.; Arrigoni, E.; Briet, F.; Flourié, B.; Morin, M.-C.; Rambaud, J.C. Do patients with short-bowel syndrome need a lactose-free diet? Nutrition 1997, 13, 13–16. [Google Scholar] [CrossRef]
- Wahlqvist, M.L. Lactose Nutrition in Lactase Nonpersisters. Asia Pac. J. Clin. Nutr. 2015, 24, s21–s25. [Google Scholar] [PubMed]
- Lukito, W.; Malik, S.G.; Surono, I.S.; Wahlqvist, M.L. From «Lactose Intolerance » to « Lactose Nutrition». Asia Pac. J. Clin. Nutr. 2015, 24, s1–s8. [Google Scholar] [PubMed]
- Fardellone, P.; Séjourné, A.; Blain, H.; Cortet, B.; Thomas, T. Ostéoporose: Avec ou sans lait ? Rev. Rhum. 2016, 83, 334–340. [Google Scholar] [CrossRef]
- Abrahamsen, B.; Vestergaard, P. Lactose avoidance and bone. Maturitas 2018, 109, A1–A2. [Google Scholar] [CrossRef]
- Elli, L.; Ferretti, F.; Orlando, S.; Vecchi, M.; Monguzzi, E.; Roncoroni, L.; Schuppan, D. Management of celiac disease in daily clinical practice. Eur. J. Intern. Med. 2019, 61, 15–24. [Google Scholar] [CrossRef]
- Cellier, C.; Malamut, G. Les nouveaux contours de la maladie cœliaque. Prat. Nutr. 2014, 10, 31–35. [Google Scholar] [CrossRef]
- Ciacci, C.; Ciclitira, P.; Hadjivassiliou, M.; Kaukinen, K.; Ludvigsson, J.F.; McGough, N.; Sanders, D.S.; Woodward, J.; Leonard, J.N.; Swift, G.L. The gluten-free diet and its current application in coeliac disease and dermatitis herpetiformis. United Eur. Gastroenterol. J. 2015, 3, 121–135. [Google Scholar] [CrossRef]
- FFAS. Etat des Lieux Gluten (Internet). Available online: http://alimentation-sante.org/wp-content/uploads/2016/11/2016__EtatDesLieux-GlutenWeb_Pages-1.pdf (accessed on 12 May 2019).
- Catassi, C.; Elli, L.; Bonaz, B.; Bouma, G.; Carroccio, A.; Castillejo, G.; Cellier, C.; Cristofori, F.; De Magistris, L.; Dolinsek, J.; et al. Diagnosis of Non-Celiac Gluten Sensitivity (NCGS): The Salerno Experts’ Criteria. Nutrients 2015, 7, 4966–4977. [Google Scholar] [CrossRef]
- Catassi, C.; Alaedini, A.; Bojarski, C.; Bonaz, B.; Bouma, G.; Carroccio, A.; Castillejo, G.; De Magistris, L.; Dieterich, W.; Di Liberto, D.; et al. The Overlapping Area of Non-Celiac Gluten Sensitivity (NCGS) and Wheat-Sensitive Irritable Bowel Syndrome (IBS): An Update. Nutrients 2017, 9, 1268. [Google Scholar] [CrossRef] [Green Version]
- Henggeler, J.C.; Veríssimo, M.; Ramos, F. Non-coeliac gluten sensitivity: A review of the literature. Trends Food Sci. Technol. 2017, 66, 84–92. [Google Scholar] [CrossRef]
- Bouteloup, C. Les pathologies digestives liées au blé ou au gluten: Certitudes et doutes. Cah. Nutr. Diet. 2016, 51, 248–258. [Google Scholar] [CrossRef]
- Gibson, P.R.; Skodje, G.I.; Lundin, K.E.A. Non-coeliac gluten sensitivity: Non-coeliac gluten sensitivity. J. Gastroenterol. Hepatol. 2017, 32, 86–89. [Google Scholar] [CrossRef] [PubMed]
- Reilly, N.R. The Gluten-Free Diet: Recognizing Fact, Fiction, and Fad. J. Pediatr. 2016, 175, 206–210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saintot, M.; Flabbee, J.-A.; Ziegler, O.; Schmutz, J.-L.; Barbaud, A. Manifestations digestives des intolérances au blé. Rev. Fr. Allergol. 2017, 57, 317–326. [Google Scholar] [CrossRef]
- Bouteloup, C. Le Régime Sans Gluten: Pourquoi, Comment, Quels Risques? Corresp. Metab. Horm. Diabetes Nutr. 2018, 1. Available online: http://www.edimark.fr/correspondances-mhdn/regime-sans-gluten-pourquoi-comment-quels-risques (accessed on 12 May 2019).
- Lebwohl, B.; Cao, Y.; Zong, G.; Hu, F.B.; Green, P.H.R.; Neugut, A.I.; Rimm, E.B.; Sampson, L.; Dougherty, L.W.; Giovannucci, E.; et al. Long term gluten consumption in adults without celiac disease and risk of coronary heart disease: Prospective cohort study. BMJ 2017, 357, j1892. [Google Scholar] [CrossRef] [Green Version]
- Gaesser, G.A.; Angadi, S.S. Gluten-free diet: Imprudent dietary advice for the general population? J. Acad. Nutr. Diet. 2012, 112, 1330–1333. [Google Scholar] [CrossRef]
- Trabal, J.; Farran-Codina, A. Effects of dietary enrichment with conventional foods on energy and protein intake in older adults: A systematic review. Nutr. Rev. 2015, 73, 624–633. [Google Scholar] [CrossRef]
- Mills, S.R.; Wilcox, C.R.; Ibrahim, K.; Roberts, H.C. Can fortified foods and snacks increase the energy and protein intake of hospitalised older patients? A systematic review. J. Hum. Nutr. Diet. 2018, 31, 379–389. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Odlund Olin, A.; Armyr, I.; Soop, M.; Jerstrom, S.; Classon, I.; Cederholm, T.; Ljungren, G.; Ljungqvist, O. Energy-dense meals improve energy intake in elderly residents in a nursing home. Clin. Nutr. Edinb. Scotl. 2003, 22, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Hope, K.; Ferguson, M.; Reidlinger, D.P.; Agarwal, E. «I don’t eat when I’m sick»: Older people’s food and mealtime experiences in hospital. Maturitas 2017, 97, 6–13. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vaillant, M.-F.; Alligier, M.; Baclet, N.; Capelle, J.; Dousseaux, M.-P.; Eyraud, E.; Fayemendy, P.; Flori, N.; Guex, E.; Hennequin, V.; et al. Guidelines on Standard and Therapeutic Diets for Adults in Hospitals by the French Association of Nutritionist Dieticians (AFDN) and the French Speaking Society of Clinical Nutrition and Metabolism (SFNCM). Nutrients 2021, 13, 2434. https://doi.org/10.3390/nu13072434
Vaillant M-F, Alligier M, Baclet N, Capelle J, Dousseaux M-P, Eyraud E, Fayemendy P, Flori N, Guex E, Hennequin V, et al. Guidelines on Standard and Therapeutic Diets for Adults in Hospitals by the French Association of Nutritionist Dieticians (AFDN) and the French Speaking Society of Clinical Nutrition and Metabolism (SFNCM). Nutrients. 2021; 13(7):2434. https://doi.org/10.3390/nu13072434
Chicago/Turabian StyleVaillant, Marie-France, Maud Alligier, Nadine Baclet, Julie Capelle, Marie-Paule Dousseaux, Evelyne Eyraud, Philippe Fayemendy, Nicolas Flori, Esther Guex, Véronique Hennequin, and et al. 2021. "Guidelines on Standard and Therapeutic Diets for Adults in Hospitals by the French Association of Nutritionist Dieticians (AFDN) and the French Speaking Society of Clinical Nutrition and Metabolism (SFNCM)" Nutrients 13, no. 7: 2434. https://doi.org/10.3390/nu13072434
APA StyleVaillant, M.-F., Alligier, M., Baclet, N., Capelle, J., Dousseaux, M.-P., Eyraud, E., Fayemendy, P., Flori, N., Guex, E., Hennequin, V., Lavandier, F., Martineau, C., Morin, M.-C., Mokaddem, F., Parmentier, I., Rossi-Pacini, F., Soriano, G., Verdier, E., Zeanandin, G., & Quilliot, D. (2021). Guidelines on Standard and Therapeutic Diets for Adults in Hospitals by the French Association of Nutritionist Dieticians (AFDN) and the French Speaking Society of Clinical Nutrition and Metabolism (SFNCM). Nutrients, 13(7), 2434. https://doi.org/10.3390/nu13072434






