Effects of Mediterranean Diet or Low-Fat Diet on Blood Fatty Acids in Patients with Coronary Heart Disease. A Randomized Intervention Study
Abstract
:1. Introduction
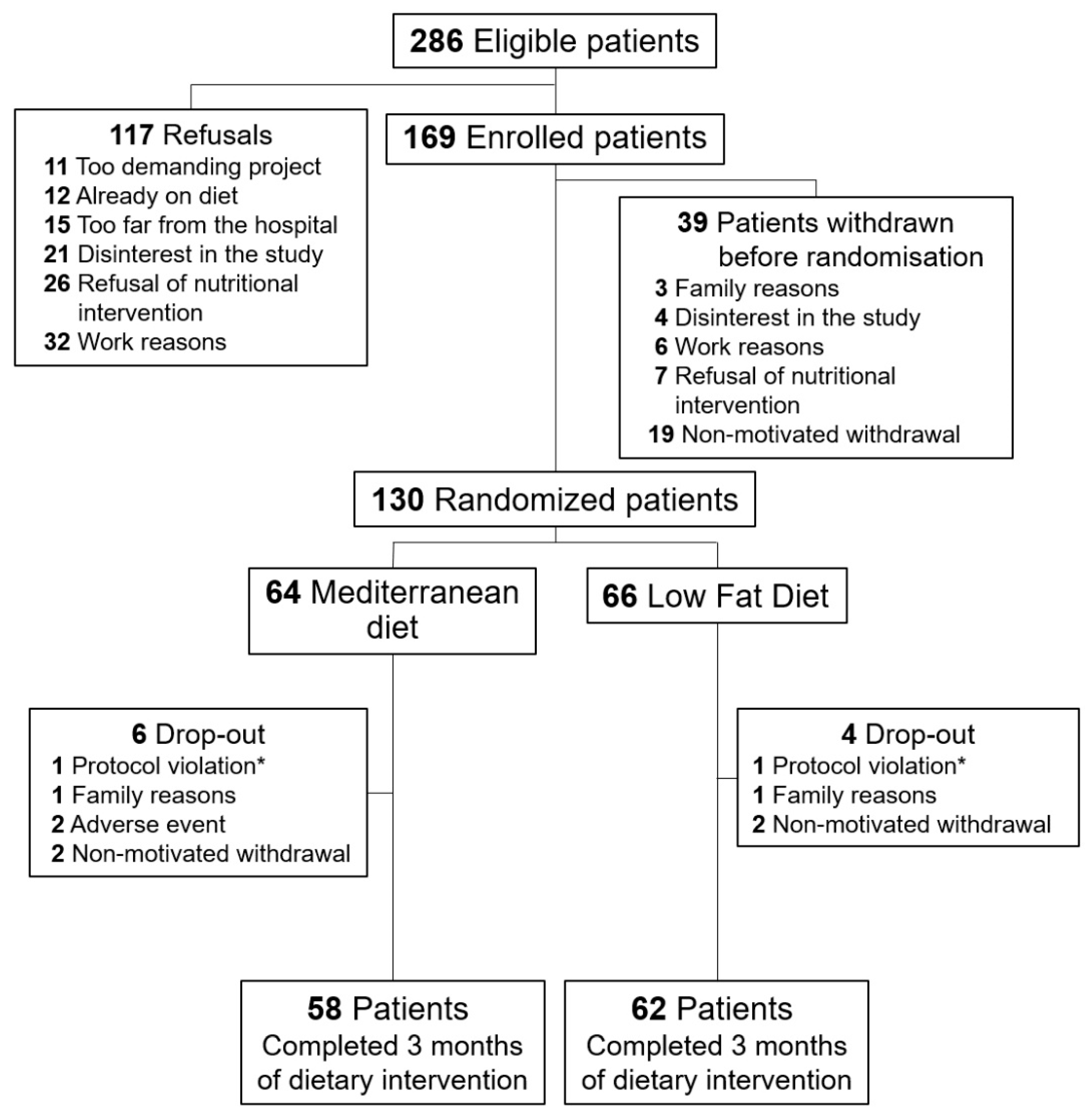
2. Materials and Methods
Statistical Analysis
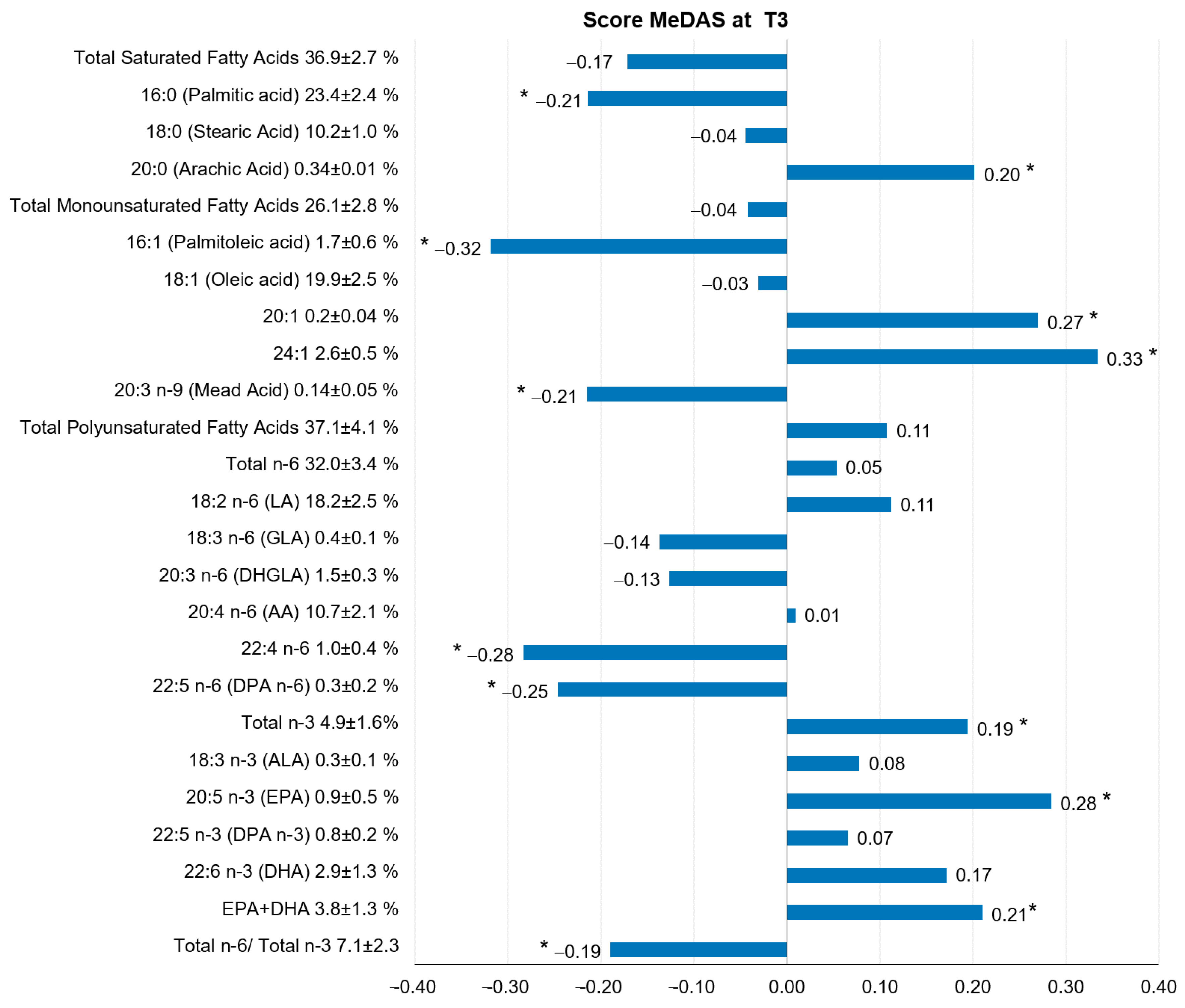
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dinu, M.; Pagliai, G.; Casini, A.; Sofi, F. Mediterranean diet and multiple health outcomes: An umbrella review of meta-analyses of observational studies and randomised trials. Eur. J. Clin. Nutr. 2018, 72, 30–43. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Marventano, S.; Yang, J.; Micek, A.; Pajak, A.; Scalfi, L.; Galvano, F.; Kales, S.N. A comprehensive meta-analysis on evidence of Mediterranean diet and cardiovascular disease: Are individual components equal? Crit. Rev. Food Sci. Nutr. 2017, 57, 3218–3232. [Google Scholar] [CrossRef] [PubMed]
- Rosato, V.; Temple, N.J.; La Vecchia, C.; Castellan, G.; Tavani, A.; Guercio, V. Mediterranean diet and cardiovascular disease: A systematic review and meta-analysis of observational studies. Eur. J. Nutr. 2019, 58, 173–191. [Google Scholar] [CrossRef] [PubMed]
- Sofi, F.; Macchi, C.; Abbate, R.; Gensini, G.F.; Casini, A. Mediterranean diet and health status: An updated meta-analysis and a proposal for a literature-based adherence score. Public Health Nutr. 2014, 17, 2769–2782. [Google Scholar] [CrossRef] [Green Version]
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.-I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Lista, J.; Perez-Martinez, P.; Garcia-Rios, A.; Alcala-Diaz, J.F.; Perez-Caballero, A.I.; Gomez-Delgado, F.; Fuentes, F.; Quintana-Navarro, G.; Lopez-Segura, F.; Ortiz-Morales, A.M.; et al. CORonary Diet Intervention with Olive oil and cardiovascular PREVention study (the CORDIOPREV study): Rationale, methods, and baseline characteristics. Am. Heart J. 2016, 177, 42–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Itsiopoulos, C.; Kucianski, T.; Mayr, H.L.; van Gaal, W.J.; Martinez-Gonzalez, M.A.; Vally, H.; Kingsley, M.; Kouris-Blazos, A.; Radcliffe, J.; Segal, L.; et al. The AUStralian MEDiterranean Diet Heart Trial (AUSMED Heart Trial): A randomized clinical trial in secondary prevention of coronary heart disease in a multiethnic Australian population: Study protocol. Am. Heart J. 2018, 203, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Tuttolomondo, A.; Simonetta, I.; Daidone, M.; Mogavero, A.; Ortello, A.; Pinto, A. Metabolic and Vascular Effect of the Mediterranean Diet. Int. J. Mol. Sci. 2019, 20, 4716. [Google Scholar] [CrossRef] [Green Version]
- Widmer, R.J.; Flammer, A.J.; Lerman, L.O.; Lerman, A. The Mediterranean Diet, its Components, and Cardiovascular Disease. Am. J. Med. 2015, 128, 229–238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Casas, R.; Castro-Barquero, S.; Estruch, R.; Sacanella, E. Nutrition and Cardiovascular Health. Int. J. Mol. Sci. 2018, 19, 3988. [Google Scholar] [CrossRef] [Green Version]
- Imamura, F.; Lemaitre, R.N.; King, I.B.; Song, X.; Lichtenstein, A.H.; Matthan, N.R.; Herrington, D.M.; Siscovick, D.S.; Mozaffarian, D. Novel circulating fatty acid patterns and risk of cardiovascular disease: The Cardiovascular Health Study. Am. J. Clin. Nutr. 2012, 96, 1252–1261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giroli, M.G.; Laguzzi, F.; Amato, M.; Risé, P.; Vigo, L.; Bonomi, A.; Veglia, F.; Werba, J.P.E.T. Determinants of the Fatty Acid Profile in Patients with and without Coronary Heart Disease. JSM Nutr. Disord. 2017, 1, 1005. [Google Scholar]
- Marangoni, F.; Novo, G.; Perna, G.; Filardi, P.P.; Pirelli, S.; Ceroti, M.; Querci, A.; Poli, A. Omega-6 and omega-3 polyunsaturated fatty acid levels are reduced in whole blood of Italian patients with a recent myocardial infarction: The AGE-IM study. Atherosclerosis 2014, 232, 334–338. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Féart, C.; Torres, M.J.M.; Samieri, C.; Jutand, M.-A.; Peuchant, E.; Simopoulos, A.P.; Barberger-Gateau, P. Adherence to a Mediterranean diet and plasma fatty acids: Data from the Bordeaux sample of the Three-City study. Br. J. Nutr. 2011, 106, 149–158. [Google Scholar] [CrossRef] [Green Version]
- Mayneris-Perxachs, J.; Sala-Vila, A.; Chisaguano, M.; Castellote, A.I.; Estruch, R.; Covas, M.I.; Fíto, M.; Salas-Salvadó, J.; Martinez-Gonzalez, M.A.; Lamuela-Raventos, R.M.; et al. Effects of 1-Year Intervention with a Mediterranean Diet on Plasma Fatty Acid Composition and Metabolic Syndrome in a Population at High Cardiovascular Risk. PLoS ONE 2014, 9, e85202. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Risé, P.; Pazzucconi, F.; Sirtori, C.R.; Galli, C.; Risé, P.; Pazzucconi, F.; Sirtori, C.R.; Galli, C. Statins enhance arachidonic acid synthesis in hypercholesterolemic patients. Nutr. Metab. Cardiovasc. Dis. 2001, 11, 88–94. [Google Scholar] [PubMed]
- Martínez-González, M.A.; García-Arellano, A.; Toledo, E.; Salas-Salvadó, J.; Buil-Cosiales, P.; Corella, D.; Covas, M.I.; Schroder, H.; Arós, F.; Gómez-Gracia, E.; et al. A 14-Item Mediterranean Diet Assessment Tool and Obesity Indexes among High-Risk Subjects: The PREDIMED Trial. PLoS ONE 2012, 7, e43134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marangoni, F.; Colombo, C.; Galli, C. A method for the direct evaluation of the fatty acid status in a drop of blood from a fingertip in humans: Applicability to nutritional and epidemiological studies. Anal. Biochem. 2004, 326, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Perk, J.; De Backer, G.; Gohlke, H.; Graham, I.; Reiner, Ž.; Verschuren, M.; Albus, C.; Benlian, P.; Boysen, G.; Cifkova, R.; et al. European Guidelines on cardiovascular disease prevention in clinical practice (version 2012): The Fifth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of nine societies and by invited experts). Eur. Hearth J. 2012, 33, 1635–1701. [Google Scholar] [CrossRef] [Green Version]
- Ristic-Medic, D.; Kovacic, M.; Takic, M.; Arsic, A.; Petrovic, S.; Paunovic, M.; Jovicic, M.; Vucic, V. Calorie-Restricted Mediterranean and Low-Fat Diets Affect Fatty Acid Status in Individuals with Nonalcoholic Fatty Liver Disease. Nutrients 2020, 13, 15. [Google Scholar] [CrossRef] [PubMed]
- Shearer, G.C.; Pottala, J.V.; Spertus, J.A.; Harris, W.S. Red Blood Cell Fatty Acid Patterns and Acute Coronary Syndrome. PLoS ONE 2009, 4, e5444. [Google Scholar] [CrossRef] [Green Version]
- Chei, C.-L.; Yamagishi, K.; Kitamura, A.; Kiyama, M.; Sankai, T.; Okada, T.; Imano, H.; Ohira, T.; Cui, R.; Umesawa, M.; et al. Serum Fatty Acid and Risk of Coronary Artery Disease―Circulatory Risk in Communities Study (CIRCS). Circ. J. 2018, 82, 3013–3020. [Google Scholar] [CrossRef] [Green Version]
- Liu, M.; Zuo, L.-S.-Y.; Sun, T.-Y.; Wu, Y.-Y.; Liu, Y.-P.; Zeng, F.-F.; Chen, Y.-M. Circulating Very-Long-Chain Saturated Fatty Acids Were Inversely Associated with Cardiovascular Health: A Prospective Cohort Study and Meta-Analysis. Nutrients 2020, 12, 2709. [Google Scholar] [CrossRef] [PubMed]
- Visioli, F.; Poli, A. Fatty Acids and Cardiovascular Risk. Evidence, Lack of Evidence, and Diligence. Nutrients 2020, 12, 3782. [Google Scholar] [CrossRef]
- Lovegrove, J.A. Dietary dilemmas over fats and cardiometabolic risk. Proc. Nutr. Soc. 2019, 79, 11–21. [Google Scholar] [CrossRef]
- Lai, H.; Otto, M.C.D.O.; Lee, Y.; Wu, J.; Song, X.; King, I.B.; Psaty, B.M.; Lemaitre, R.N.; McKnight, B.; Siscovick, D.S.; et al. Serial Plasma Phospholipid Fatty Acids in the De Novo Lipogenesis Pathway and Total Mortality, Cause-Specific Mortality, and Cardiovascular Diseases in the Cardiovascular Health Study. J. Am. Heart Assoc. 2019, 8, e012881. [Google Scholar] [CrossRef] [PubMed]
- Tapiero, H.; Ba, G.N.; Couvreur, P.; Tew, K. Polyunsaturated fatty acids (PUFA) and eicosanoids in human health and pathologies. Biomed. Pharmacother. 2002, 56, 215–222. [Google Scholar] [CrossRef]
- Wu, J.H.; Lemaitre, R.N.; King, I.B.; Song, X.; Psaty, B.M.; Siscovick, D.S.; Mozaffarian, D. Circulating Omega-6 Polyunsaturated Fatty Acids and Total and Cause-Specific Mortality. Circulation 2014, 130, 1245–1253. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marklund, M.; Wu, J.H.; Imamura, F.; Del Gobbo, L.C.; Fretts, A.; De Goede, J.; Shi, P.; Tintle, N.; Wennberg, M.; Aslibekyan, S.; et al. Biomarkers of Dietary Omega-6 Fatty Acids and Incident Cardiovascular Disease and Mortality. Circulation 2019, 139, 2422–2436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, L.; Folsom, A.; Eckfeldt, J.; the ARIC Study Investigators. Plasma fatty acid composition and incidence of coronary heart disease in middle aged adults: The Atherosclerosis Risk in Communities (ARIC) Study. Nutr. Metab. Cardiovasc. Dis. 2003, 13, 256–266. [Google Scholar] [CrossRef]
- Del Gobbo, L.C.; Imamura, F.; Aslibekyan, S.; Marklund, M.; Virtanen, J.K.; Wennberg, M.; Yakoob, M.Y.; Chiuve, S.E.; Cruz, L.D.; Frazier-Wood, A.C.; et al. ω-3 Polyunsaturated Fatty Acid Biomarkers and Coronary Heart Disease. JAMA Intern. Med. 2016, 176, 1155–1166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Q.; Matthan, N.R.; Manson, J.E.; Howard, B.V.; Tinker, L.F.; Neuhouser, M.L.; Van Horn, L.V.; Rossouw, J.E.; Allison, M.A.; Martin, L.W.; et al. Plasma Phospholipid Fatty Acids and Coronary Heart Disease Risk: A Matched Case-Control Study within the Women’s Health Initiative Observational Study. Nutrients 2019, 11, 1672. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kleber, M.E.; Delgado, G.E.; Lorkowski, S.; März, W.; Von Schacky, C. Omega-3 fatty acids and mortality in patients referred for coronary angiography. The Ludwigshafen Risk and Cardiovascular Health Study. Atherosclerosis 2016, 252, 175–181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mozaffarian, D.; Lemaitre, R.N.; King, I.B.; Song, X.; Huang, H.; Sacks, F.M.; Rimm, E.B.; Wang, M.; Siscovick, D.S. Plasma Phospholipid Long-Chain ω-3 Fatty Acids and Total and Cause-Specific Mortality in Older Adults. Ann. Intern. Med. 2013, 158, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Stark, K.D.; Henao, J.J.A.; Metherel, A.H.; Pilote, L. Translating plasma and whole blood fatty acid compositional data into the sum of eicosapentaenoic and docosahexaenoic acid in erythrocytes. Prostaglandins Leukot. Essent. Fat. Acids 2016, 104, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; von Schacky, C. The Omega-3 Index: A new risk factor for death from coronary heart disease? Prev. Med. 2004, 39, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S. The omega-3 index as a risk factor for coronary heart disease. Am. J. Clin. Nutr. 2008, 87, 1997S–2002S. [Google Scholar] [CrossRef]
- Morioka, T.; Emoto, M.; Imamura, S.; Kakutani, Y.; Yamazaki, Y.; Motoyama, K.; Mori, K.; Fukumoto, S.; Shioi, A.; Shoji, T.; et al. Plasma polyunsaturated fatty acid profile is associated with vascular endothelial function in patients with type 2 diabetes. Diabetes Vasc. Dis. Res. 2018, 15, 352–355. [Google Scholar] [CrossRef] [Green Version]
- Shibabaw, T. Omega-3 polyunsaturated fatty acids: Anti-inflammatory and anti-hypertriglyceridemia mechanisms in cardiovascular disease. Mol. Cell. Biochem. 2021, 476, 993–1003. [Google Scholar] [CrossRef]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Nut consumption and risk of cardiovascular disease, total cancer, all-cause and cause-specific mortality: A systematic review and dose-response meta-analysis of prospective studies. BMC Med. 2016, 14, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Zhang, B.; Xiong, K.; Cai, J.; Ma, A. Fish Consumption and Coronary Heart Disease: A Meta-Analysis. Nutrients 2020, 12, 2278. [Google Scholar] [CrossRef] [PubMed]
- Mohan, D.; Mente, A.; Dehghan, M.; Rangarajan, S.; O’Donnell, M.; Hu, W.; Dagenais, G.; Wielgosz, A.; Lear, S.; Wei, L.; et al. Associations of Fish Consumption With Risk of Cardiovascular Disease and Mortality Among Individuals With or Without Vascular Disease From 58 Countries. JAMA Intern. Med. 2021, 181, 631. [Google Scholar] [CrossRef]
- Burr, M.; Gilbert, J.; Holliday, R.; Elwood, P.; Fehily, A.; Rogers, S.; Sweetnam, P.; Deadman, N. Effects of changes in fat, fish, and fibre intakes on death and myocardial reinfarction: Diet and reinfarction trial (DART). Lancet 1989, 334, 757–761. [Google Scholar] [CrossRef]
- De Souza, R.J.; Dehghan, M.; Mente, A.; Bangdiwala, S.I.; Ahmed, S.H.; Alhabib, K.F.; Altuntas, Y.; Basiak-Rasała, A.; Dagenais, G.-R.; Diaz, R.; et al. Association of nut intake with risk factors, cardiovascular disease, and mortality in 16 countries from 5 continents: Analysis from the Prospective Urban and Rural Epidemiology (PURE) study. Am. J. Clin. Nutr. 2020, 112, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Guasch-Ferré, M.; Liu, X.; Malik, V.S.; Sun, Q.; Willett, W.C.; Manson, J.E.; Rexrode, K.; Li, Y.; Hu, F.B.; Bhupathiraju, S.N. Nut Consumption and Risk of Cardiovascular Disease. J. Am. Coll. Cardiol. 2017, 70, 2519–2532. [Google Scholar] [CrossRef] [PubMed]
- Risé, P.; Eligini, S.; Ghezzi, S.; Colli, S.; Galli, C. Fatty acid composition of plasma, blood cells and whole blood: Relevance for the assessment of the fatty acid status in humans. Prostaglandins Leukot. Essent. Fat. Acids 2007, 76, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Baylin, A.; Kim, M.K.; Donovan-Palmer, A.; Siles, X.; Dougherty, L.; Tocco, P.; Campos, H. Fasting Whole Blood as a Biomarker of Essential Fatty Acid Intake in Epidemiologic Studies: Comparison with Adipose Tissue and Plasma. Am. J. Epidemiol. 2005, 162, 373–381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drzymała-Czyż, S.; Janich, S.; Klingler, M.; Demmelmair, J.; Walkowiak, J.; Koletzko, B. Whole blood glycerophospholipids in dried blood spots—A reliable marker for the fatty acid status. Chem. Phys. Lipids 2017, 207, 1–9. [Google Scholar] [CrossRef]
- Shen, W.; Weaver, A.M.; Salazar, C.; Samet, J.M.; Diaz-Sanchez, D.; Tong, H. Validation of a Dietary Questionnaire to Screen Omega-3 Fatty Acids Levels in Healthy Adults. Nutrients 2019, 11, 1470. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| ALL | MD | LFD | p-Value | |
|---|---|---|---|---|
| N = 130 | n = 64 | n = 66 | ||
| Men (n%) | 113 (86.9%) | 54 (84.4%) | 59 (89.4%) | 0.40 |
| Age (years) | 62.2 ± 9.5 | 63.0 ± 6.8 | 61.5 ± 11.6 | 0.35 |
| Height (m) | 1.70 ± 0.09 | 1.69 ± 0.09 | 1.70 ± 0.08 | 0.77 |
| Body Weight (Kg) | 79.2 ± 12.5 | 77.6 ± 12.2 | 80.8 ± 12.6 | 0.18 |
| BMI (Kg/m2) | 27.47 ± 3.46 | 27.1 ± 3.3 | 27.8 ± 3.6 | 0.31 |
| Waist circumference (cm) | 98.0 ± 9.8 | 97.0 ± 9.8 | 99.0 ± 9.8 | 0.33 |
| Smoking (n%) | 0.26 | |||
| former smoker | 77 (59.2%) | 37 (57.8%) | 40 (60.6%) | |
| current smoker | 15 (11.5%) | 5 (7.8%) | 10 (15.2%) | |
| non-smoker | 38 (29.2%) | 22 (34.4%) | 16 (24.2%) | |
| Physical activity (n%) | 0.47 | |||
| intense | 29 (22.3%) | 14 (21.9%) | 15 (22.7%) | |
| average | 41 (31.5%) | 18 (28.1%) | 23 (34.9%) | |
| low | 60 (46.2%) | 32 (50.0%) | 28 (42.4%) | |
| Total cholesterol (mg/dL) | 167.6 ± 29.2 | 172.7 ± 29.1 | 162.6 ± 28.5 | 0.05 |
| HDL-cholesterol (mg/dL) | 50.9 ± 13.0 | 49.8 ± 12.5 | 51.9 ± 13.5 | 0.45 |
| LDL-cholesterol (mg/dL) | 93.8 ± 25.5 | 99.3 ± 26.3 | 88.5 ± 23.6 | 0.02 |
| Triglycerides (mg/dL) | 114.4 ± 57.7 | 117.9 ± 63.2 | 111.1 ± 52.0 | 0.93 |
| Fasting blood glucose (mg/dL) | 101.9 ± 10.4 | 100.1 ± 9.1 | 103.7 ± 11.3 | 0.14 |
| SBP (mmHg) | 132.4 ± 18.0 | 132.8 ± 16.8 | 131.9 ± 19.3 | 0.63 |
| DBP (mmHg) | 79.4 ± 10.2 | 79.0 ± 10.9 | 79.9 ± 9.5 | 0.63 |
| On lipid-lowering therapy (n%) | 119 (91.5%) | 55 (85.9%) | 64 (97.0%) | 0.02 |
| On antihypertensive therapy (n%) | 119 (91.5%) | 60 (93.8%) | 59 (89.4%) | 0.37 |
| On antiplatelet therapy (n%) | 123 (94.6%) | 61 (95.3%) | 62 (93.9%) | 0.73 |
| Total medicines (median [min; max]) | 5 [3;9] | 5 [3; 9] | 5 [3; 9] | 0.94 |
| MeDAS Score | 7.25 ± 1.6 | 7.36 ± 1.4 | 7.14 ± 1.8 | 0.47 |
| T0 | T3 | T3-T0 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| MD | LFD | p-Value | MD | LFD | p-Value | MD | p-Value | LFD | p-Value | p-Value | |
| n = 58 | n = 62 | n = 58 | n = 62 | n = 58 | vs. T0 | n = 62 | vs. T0 | Between Groups | |||
| MeDAS Score | 7.3 ± 1.4 | 7.1 ± 1.8 | 0.55 | 9.9 ± 1.7 | 8.2 ± 1.7 | <0.0001 | 2.6 ± 2.2 | <0.0001 | 1.0 ± 1.8 | <0.0001 | <0.0001 |
| Fatty acids levels (% of total fatty acids): | |||||||||||
| Total Saturated Fatty Acids | 38.5 ± 4.33 | 38.52 ± 4.07 | 0.77 | 36.39 ± 2.7 | 37.29 ± 2.72 | 0.03 | −1.88 ± 3.98 | 0.001 | −1.25 ± 3.66 | 0.009 | 0.4 |
| 16:0 (Palmitic Acid) | 24.15 ± 3.21 | 24.35 ± 2.96 | 0.54 | 23 ± 2.45 | 23.71 ± 2.23 | 0.04 | −0.89 ± 2.94 | 0.025 | −0.65 ± 2.81 | 0.07 | 0.42 |
| 18:0 (Stearic Acid) | 10.81 ± 1.64 | 10.77 ± 1.43 | 0.78 | 10.13 ± 0.96 | 10.32 ± 0.98 | 0.33 | −0.71 ± 1.49 | 0.001 | −0.45 ± 1.07 | 0.002 | 0.33 |
| 20:0 (Arachic Acid) | 0.43 ± 0.27 | 0.43 ± 0.24 | 0.94 | 0.34 ± 0.09 | 0.33 ± 0.1 | 0.59 | −0.08 ± 0.23 | 0.012 | −0.1 ± 0.21 | <0.001 | 0.26 |
| Total Monounsaturated Fatty Acids | 25.39 ± 3.65 | 24.81 ± 3.27 | 0.28 | 26.22 ± 3 | 25.89 ± 2.65 | 0.82 | 0.74 ± 3.4 | 0.1 | 1.08 ± 3.18 | 0.01 | 0.42 |
| 16:1 (Palmitoleic Acid) | 1.76 ± 0.64 | 1.99 ± 0.81 | 0.09 | 1.59 ± 0.53 | 1.8 ± 0.68 | 0.08 | −0.1 ± 0.48 | 0.1 | −0.19 ± 0.58 | 0.012 | 0.9 |
| 18:1 (Oleic Acid) | 19.3 ± 3.64 | 18.43 ± 3.24 | 0.12 | 20.13 ± 2.75 | 19.61 ± 2.31 | 0.51 | 0.7 ± 3.36 | 0.12 | 1.14 ± 3.31 | 0.009 | 0.34 |
| 20:1 | 0.2 ± 0.12 | 0.22 ± 0.19 | 0.84 | 0.19 ± 0.05 | 0.18 ± 0.03 | 0.66 | 0 ± 0.1 | 0.77 | −0.04 ± 0.19 | 0.08 | 0.49 |
| 24:1 | 2.5 ± 0.56 | 2.46 ± 0.46 | 0.96 | 2.66 ± 0.47 | 2.62 ± 0.44 | 0.59 | 0.11 ± 0.58 | 0.17 | 0.17 ± 0.45 | 0.004 | 0.7 |
| 20:3 n−9 (Mead Acid) | 0.21 ± 0.16 | 0.16 ± 0.11 | 0.03 | 0.14 ± 0.06 | 0.14 ± 0.04 | 0.82 | −0.03 ± 0.11 | 0.05 | −0.07 ± 0.14 | 0.001 | 0.12 |
| Total Polyunsaturated Fatty Acids | 36.11 ± 4.04 | 36.67 ± 4.78 | 0.38 | 37.39 ± 4.13 | 36.81 ± 4.11 | 0.31 | 1.14 ± 3.73 | 0.023 | 0.17 ± 4.14 | 0.75 | 0.1 |
| Total n−6 | 31.06 ± 3.36 | 31.3 ± 4.23 | 0.56 | 32.15 ± 3.29 | 31.88 ± 3.51 | 0.58 | 0.95 ± 3.21 | 0.028 | 0.56 ± 3.56 | 0.22 | 0.4 |
| 18:2 n−6 (LA) | 17.52 ± 2.99 | 17.1 ± 3.56 | 0.45 | 18.23 ± 2.15 | 18.09 ± 2.83 | 0.76 | 0.58 ± 2.8 | 0.12 | 0.91 ± 2.74 | 0.011 | 0.53 |
| 18:3 n−6 (GLA) | 0.51 ± 0.29 | 0.54 ± 0.25 | 0.15 | 0.42 ± 0.15 | 0.42 ± 0.13 | 0.42 | −0.07 ± 0.29 | 0.08 | −0.12 ± 0.27 | 0.002 | 0.14 |
| 20:3 n−6 (DHGLA) | 1.62 ± 0.35 | 1.54 ± 0.3 | 0.13 | 1.53 ± 0.3 | 1.43 ± 0.29 | 0.01 | −0.11 ± 0.27 | 0.004 | −0.11 ± 0.24 | 0.001 | 0.86 |
| 20:4 n−6 (AA) | 10.13 ± 2.37 | 10.75 ± 2.44 | 0.13 | 10.73 ± 2.21 | 10.68 ± 1.99 | 0.69 | 0.6 ± 1.86 | 0.017 | −0.01 ± 1.96 | 0.95 | 0.05 |
| 22:4 n−6 | 1.01 ± 0.42 | 1.08 ± 0.4 | 0.25 | 0.97 ± 0.35 | 1 ± 0.35 | 0.78 | −0.06 ± 0.38 | 0.27 | −0.07 ± 0.34 | 0.09 | 0.85 |
| 22:5 n−6 (DPA n−6) | 0.27 ± 0.15 | 0.3 ± 0.16 | 0.25 | 0.27 ± 0.17 | 0.26 ± 0.12 | 0.54 | 0 ± 0.22 | 0.99 | −0.04 ± 0.19 | 0.14 | 0.42 |
| Total n−3 | 4.88 ± 1.85 | 5.17 ±1.83 | 0.24 | 5.11 ± 1.46 | 4.79 ± 1.54 | 0.2 | 0.22 ± 1.67 | 0.32 | −0.32 ± 1.48 | 0.09 | 0.03 |
| 18:3 n−3 (ALA) | 0.39 ± 0.24 | 0.39 ± 0.28 | 0.65 | 0.32 ± 0.11 | 0.34 ± 0.16 | 0.98 | −0.03 ± 0.23 | 0.27 | −0.05 ± 0.3 | 0.19 | 0.9 |
| 20:5 n−3 (EPA) | 0.8 ± 0.47 | 0.88 ± 0.48 | 0.26 | 0.91 ± 0.5 | 0.83 ± 0.44 | 0.27 | 0.09 ± 0.56 | 0.22 | −0.04 ± 0.41 | 0.417 | 0.07 |
| 22:5 n−3 (DPA n−3) | 0.95 ± 0.53 | 1.02 ± 0.53 | 0.27 | 0.84 ± 0.24 | 0.81 ± 0.25 | 0.4 | −0.11 ± 0.51 | 0.11 | −0.2 ± 0.46 | 0.001 | 0.11 |
| 22:6 n−3 (DHA) | 2.74 ± 1.06 | 2.88 ± 1.03 | 0.43 | 3.04 ± 0.87 | 2.82 ± 0.95 | 0.13 | 0.27 ± 0.89 | 0.023 | −0.03 ± 0.92 | 0.77 | 0.12 |
| EPA + DHA | 3.58 ± 1.37 | 3.72 ± 1.42 | 0.38 | 3.94 ± 1.21 | 3.64 ± 1.30 | 0.18 | 0.36 ± 1.17 | 0.02 | −0.07 ± 1.16 | 0.6 | 0.04 |
| Total n−6/Total n−3 | 7.38 ± 3.01 | 7.01 ± 3 | 0.24 | 6.84 ± 2.16 | 7.35 ± 2.37 | 0.18 | −0.62 ± 2.76 | 0.1 | 0.24 ± 2.36 | 0.42 | 0.04 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giroli, M.G.; Werba, J.P.; Risé, P.; Porro, B.; Sala, A.; Amato, M.; Tremoli, E.; Bonomi, A.; Veglia, F. Effects of Mediterranean Diet or Low-Fat Diet on Blood Fatty Acids in Patients with Coronary Heart Disease. A Randomized Intervention Study. Nutrients 2021, 13, 2389. https://doi.org/10.3390/nu13072389
Giroli MG, Werba JP, Risé P, Porro B, Sala A, Amato M, Tremoli E, Bonomi A, Veglia F. Effects of Mediterranean Diet or Low-Fat Diet on Blood Fatty Acids in Patients with Coronary Heart Disease. A Randomized Intervention Study. Nutrients. 2021; 13(7):2389. https://doi.org/10.3390/nu13072389
Chicago/Turabian StyleGiroli, Monica Gianna, José Pablo Werba, Patrizia Risé, Benedetta Porro, Angelo Sala, Manuela Amato, Elena Tremoli, Alice Bonomi, and Fabrizio Veglia. 2021. "Effects of Mediterranean Diet or Low-Fat Diet on Blood Fatty Acids in Patients with Coronary Heart Disease. A Randomized Intervention Study" Nutrients 13, no. 7: 2389. https://doi.org/10.3390/nu13072389
APA StyleGiroli, M. G., Werba, J. P., Risé, P., Porro, B., Sala, A., Amato, M., Tremoli, E., Bonomi, A., & Veglia, F. (2021). Effects of Mediterranean Diet or Low-Fat Diet on Blood Fatty Acids in Patients with Coronary Heart Disease. A Randomized Intervention Study. Nutrients, 13(7), 2389. https://doi.org/10.3390/nu13072389







