Living with Food Hypersensitivity as an Adolescent Impairs Health Related Quality of Life Irrespective of Disease Severity: Results from a Population-Based Birth Cohort
Abstract
1. Introduction
2. Material and Methods
2.1. Study Design and Population
2.2. Assessment of HRQoL
2.3. Assessments of Food Hypersensitivity
- Common foods (milk, egg, fish and wheat).
- Peanuts and tree nuts eanut, hazelnut, almond, walnut/pecan, cashew/pistachio andBrazil nut).
2.4. Statistics
3. Results
3.1. Background Characteristics and Self-Reported HRQoL in the Study Population
3.2. Background Characteristics and Self-Reported HRQoL in Relation to FHS
3.3. Self-Reported HRQoL in Relation to Phenotypes of FHS
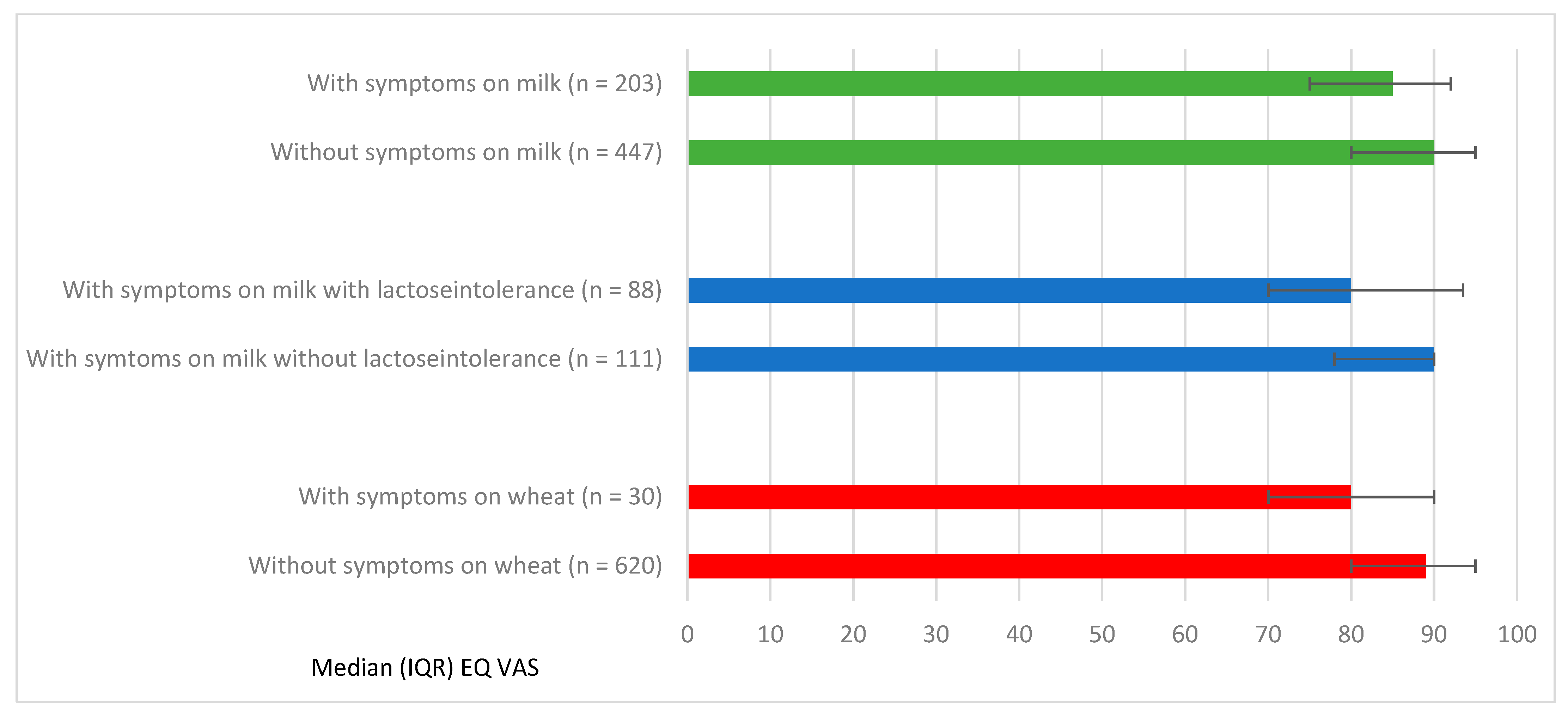
3.4. Self-Reported HRQoL in Relation to Symptoms on Specific Foods among Adolescents with FHS
3.5. Sensitivity Analyses
4. Discussion
4.1. Strength and Limitations
4.2. Impact on HRQoL among Adolescents with FHS
4.3. The Importance of Correct Diagnoses and Appropriate Care for Adolescents with FHS
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BAMSE | Swedish abbreviation for Children, Allergy, Milieu, Stockholm, Epidemiology |
| FHS | Food hypersensitivity |
| HRQoL | Health Related Quality of Life |
| IgE sensitization | Immunoglobulin E sensitization |
References
- Nwaru, B.I.; Hickstein, L.; Panesar, S.S.; Roberts, G.; Muraro, A.; Sheikh, A.; Allergy, E.F.; Anaphylaxis Guidelines, G. Prevalence of common food allergies in Europe: A systematic review and meta-analysis. Allergy 2014, 69, 992–1007. [Google Scholar] [CrossRef]
- Deschildre, A.; Lejeune, S.; Cap, M.; Flammarion, S.; Jouannic, L.; Amat, F.; Just, J. Food allergy phenotypes: The key to personalized therapy. Clin. Exp. Allergy 2017, 47, 1125–1137. [Google Scholar] [CrossRef] [PubMed]
- Sicherer, S.H.; Sampson, H.A. Food allergy: A review and update on epidemiology, pathogenesis, diagnosis, prevention, and management. J. Allergy Clin. Immunol. 2018, 141, 41–58. [Google Scholar] [CrossRef] [PubMed]
- Venter, C.; Meyer, R.W.; Nwaru, B.I.; Roduit, C.; Untersmayr, E.; Adel-Patient, K.; Agache, I.; Agostoni, C.; Akdis, C.A.; Bischoff, S.C.; et al. EAACI position paper: Influence of dietary fatty acids on asthma, food allergy, and atopic dermatitis. Allergy 2019, 74, 1429–1444. [Google Scholar] [CrossRef] [PubMed]
- Tuck, C.J.; Biesiekierski, J.R.; Schmid-Grendelmeier, P.; Pohl, D. Food Intolerances. Nutrients 2019, 11, 1684. [Google Scholar] [CrossRef]
- Ballardini, N.; Kull, I.; Lind, T.; Hallner, E.; Almqvist, C.; Ostblom, E.; Melen, E.; Pershagen, G.; Lilja, G.; Bergstrom, A.; et al. Development and comorbidity of eczema, asthma and rhinitis to age 12: Data from the BAMSE birth cohort. Allergy 2012, 67, 537–544. [Google Scholar] [CrossRef]
- Pereira, B.; Venter, C.; Grundy, J.; Clayton, C.B.; Arshad, S.H.; Dean, T. Prevalence of sensitization to food allergens, reported adverse reaction to foods, food avoidance, and food hypersensitivity among teenagers. J. Allergy Clin. Immunol. 2005, 116, 884–892. [Google Scholar] [CrossRef] [PubMed]
- Rona, R.J.; Keil, T.; Summers, C.; Gislason, D.; Zuidmeer, L.; Sodergren, E.; Sigurdardottir, S.T.; Lindner, T.; Goldhahn, K.; Dahlstrom, J.; et al. The prevalence of food allergy: A meta-analysis. J. Allergy Clin. Immunol. 2007, 120, 638–646. [Google Scholar] [CrossRef]
- Savage, J.; Johns, C.B. Food allergy: Epidemiology and natural history. Immunol. Allergy Clin. N. Am. 2015, 35, 45–59. [Google Scholar] [CrossRef]
- Roehr, C.C.; Edenharter, G.; Reimann, S.; Ehlers, I.; Worm, M.; Zuberbier, T.; Niggemann, B. Food allergy and non-allergic food hypersensitivity in children and adolescents. Clin. Exp. Allergy 2004, 34, 1534–1541. [Google Scholar] [CrossRef]
- Protudjer, J.L.; Vetander, M.; Kull, I.; Hedlin, G.; van Hage, M.; Wickman, M.; Bergstrom, A. Food-Related Symptoms and Food Allergy in Swedish Children from Early Life to Adolescence. PLoS ONE 2016, 11, e0166347. [Google Scholar] [CrossRef]
- Boyce, J.A.; Assa’ad, A.; Burks, A.W.; Sampson, H.A.; Jones, S.M.; Wood, R.A.; Plaut, M.; Cooper, S.F.; Fenton, M.J.; Arshad, S.H.; et al. NIAD-sponsored expert panel. Guidelines for the diagnoses and management of food allergy in United States: Summary of NIAID-sponsered expert panel report. J. Allergy Clin. Immunol. 2010, 126, 1105–1118. [Google Scholar] [CrossRef] [PubMed]
- Iweala, O.I.; Choudhary, S.K.; Commins, S.P. Food Allergy. Curr. Gastroenterol. Rep. 2018, 20, 17. [Google Scholar] [CrossRef]
- Begen, F.M.; Barnett, J.; Payne, R.; Gowland, M.H.; DunnGalvin, A.; Lucas, J.S. Eating out with a food allergy in the UK: Change in the eating out practices of consumers with food allergy following introduction of allergen information legislation. Clin. Exp. 2018, 48, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Begen, F.M.; Barnett, J.; Barber, M.; Payne, R.; Gowland, M.H.; Lucas, J.S. Parents’ and caregivers’ experiences and behaviours when eating out with children with a food hypersensitivity. BMC Public Health 2017, 18, 38. [Google Scholar] [CrossRef]
- De Schryver, S.; Clarke, A.; La Vieille, S.; Eisman, H.; Morris, J.; Lim, R.; Gravel, J.; Ben-Shoshan, M. Food-induced anaphylaxis to a known food allergen in children often occurs despite adult supervision. Pediatric Allergy Immunol. 2017, 28, 715–717. [Google Scholar] [CrossRef] [PubMed]
- Miles, L.M.; Gabrielli, S.; Clarke, A.E.; Morris, J.; Eisman, H.; Gravel, J.; Lim, R.; Hochstadter, E.; Gerdts, J.; Upton, J.; et al. When and how pediatric anaphylaxis cases reach the emergency department: Findings from the Cross-Canada Anaphylaxis Registry. J. Allergy Clin. Immunol. Pract. 2020, 8, 1406–1409.e2. [Google Scholar] [CrossRef]
- Lagercrantz, B.; Persson, A.; Kull, I. “Healthcare seems to vary a lot”: A focus group study among parents of children with severe allergy. J. Asthma 2017, 54, 1–7. [Google Scholar] [CrossRef]
- Michaud, P.A.; Suris, J.C.; Viner, R. The adolescent with a chronic condition. Part II: Healthcare provision. Arch. Dis. Child. 2004, 89, 943–949. [Google Scholar] [CrossRef]
- Jonsson, M.; Schuster, M.; Protudjer, J.L.P.; Bergstrom, A.; Egmar, A.C.; Kull, I. Experiences of Daily Life Among Adolescents with Asthma—A Struggle with Ambivalence. J. Pediatric Nurs. 2017, 35, 23–29. [Google Scholar] [CrossRef]
- Haraldstad, K.; Wahl, A.; Andenaes, R.; Andersen, J.R.; Andersen, M.H.; Beisland, E.; Borge, C.R.; Engebretsen, E.; Eisemann, M.; Halvorsrud, L.; et al. A systematic review of quality of life research in medicine and health sciences. Qual. Life Res. 2019, 28, 2641–2650. [Google Scholar] [CrossRef] [PubMed]
- Greenhawt, M. Food allergy quality of life. Ann. Allergy Asthma Immunol. 2014, 113, 506–512. [Google Scholar] [CrossRef]
- Haraldstad, K.; Kvarme, L.G.; Christophersen, K.A.; Helseth, S. Associations between self-efficacy, bullying and health-related quality of life in a school sample of adolescents: A cross-sectional study. BMC Public Health 2019, 19, 757. [Google Scholar] [CrossRef]
- Jonsson, M.; Bergstrom, A.; Egmar, A.C.; Hedlin, G.; Lind, T.; Kull, I. Asthma during adolescence impairs health-related quality of life. J. Allergy Clin. Immunol. Pract. 2016, 4, 144–146 e142. [Google Scholar] [CrossRef]
- Covaciu, C.; Bergstrom, A.; Lind, T.; Svartengren, M.; Kull, I. Childhood allergies affect health-related quality of life. J. Asthma 2013, 50, 522–528. [Google Scholar] [CrossRef]
- Marklund, B.; Ahlstedt, S.; Nordstrom, G. Health-related quality of life in food hypersensitive schoolchildren and their families: Parents’ perceptions. Health Qual. Life Outcomes 2006, 4, 48. [Google Scholar] [CrossRef]
- Avery, N.J.; King, R.M.; Knight, S.; Hourihane, J.O. Assessment of quality of life in children with peanut allergy. Pediatric Allergy Immunol. 2003, 14, 378–382. [Google Scholar] [CrossRef] [PubMed]
- Flokstra-de Blok, B.M.; DunnGalvin, A.; Vlieg-Boerstra, B.J.; Oude Elberink, J.N.; Duiverman, E.J.; Hourihane, J.O.; Dubois, A.E. Development and validation of the self-administered Food Allergy Quality of Life Questionnaire for adolescents. J. Allergy Clin. Immunol. 2008, 122, 139–144.e2. [Google Scholar] [CrossRef] [PubMed]
- Protudjer, J.L.; Jansson, S.A.; Middelveld, R.; Ostblom, E.; Dahlen, S.E.; Arnlind, M.H.; Bengtsson, U.; Kallstrom-Bengtsson, I.; Marklund, B.; Rentzos, G.; et al. Impaired health-related quality of life in adolescents with allergy to staple foods. Clin. Transl. Allergy 2016, 6, 37. [Google Scholar] [CrossRef]
- Thornqvist, V.; Middelveld, R.; Wai, H.M.; Ballardini, N.; Nilsson, E.; Stromquist, J.; Ahlstedt, S.; Nilsson, L.J.; Protudjer, J.L.P. Health-related quality of life worsens by school age amongst children with food allergy. Clin. Transl. Allergy 2019, 9, 10. [Google Scholar] [CrossRef]
- Arik Yilmaz, E.; Cavkaytar, O.; Buyuktiryaki, B.; Soyer, O.; Sahiner, U.M.; Sekerel, B.E.; DunnGalvin, A.; Karabulut, E.; Sackesen, C. Factors Affecting Food Allergy-Related Quality of Life From Parents’ Perception in Turkish Children. Allergy Asthma Immunol. Res. 2018, 10, 379–386. [Google Scholar] [CrossRef]
- Howe, L.; Franxman, T.; Teich, E.; Greenhawt, M. What affects quality of life among caregivers of food-allergic children? Ann. Allergy Asthma Immunol. 2014, 113, 69–74 e62. [Google Scholar] [CrossRef]
- Strinnholm, A.; Hedman, L.; Winberg, A.; Jansson, S.A.; Lindh, V.; Ronmark, E. Health Related Quality of Life among schoolchildren aged 12–13 years in relation to food hypersensitivity phenotypes: A population-based study. Clin. Transl. Allergy 2017, 7, 20. [Google Scholar] [CrossRef][Green Version]
- Wickman, M.; Kull, I.; Pershagen, G.; Nordvall, S.L. The BAMSE project: Presentation of a prospective longitudinal birth cohort study. Pediatric Allergy Immunol. 2002, 13 (Suppl. S15), 11–13. [Google Scholar] [CrossRef] [PubMed]
- EQ-5D a Standardised Instrument for Use as a Measure of Health Outcome 2010. Available online: www.euroqol.org (accessed on 16 September 2017).
- The EuroQol Group. EuroQol—A new facility for the measurement of health-related quality of life. Health Policy 1990, 16, 199–208. [Google Scholar] [CrossRef]
- Rabin, R.; de Charro, F. EQ-5D: A measure of health status from the EuroQol Group. Ann. Med. 2001, 33, 337–343. [Google Scholar] [CrossRef]
- Swedish Association for Allergy. Anaphylaxis Document; Recommendations for Care and Treatment. Prepared on Behalf of Svenska Association for Allergology (SFFA) 2015. Available online: http://www.sffa.nu/wp-content/uploads/2015/12/Anafylaxi_sept_2015.pdf (accessed on 11 October 2020).
- Dahlen, E.; Almqvist, C.; Bergstrom, A.; Wettermark, B.; Kull, I. Factors associated with concordance between parental-reported use and dispensed asthma drugs in adolescents: Findings from the BAMSE birth cohort. Pharmacoepidemiol. Drug Saf. 2014, 23, 942–949. [Google Scholar] [CrossRef] [PubMed]
- WHO Collaborating Centre for Drug Statistics Methodology. Guidelines for ATC Classification and DDD Assignment 2018. Available online: https://www.whocc.no/filearchive/publications/guidelines.pdf (accessed on 1 July 2021).
- Finch, A.P.; Brazier, J.E.; Mukuria, C. What is the evidence for the performance of generic preference-based measures? A systematic overview of reviews. Eur. J. Health Econ. 2018, 19, 557–570. [Google Scholar] [CrossRef]
- Robles, L.; Priefer, R. Lactose Intolerance: What Your Breath Can Tell You. Diagnostics 2020, 10, 412. [Google Scholar] [CrossRef]
- Ferro, M.A.; Van Lieshout, R.J.; Ohayon, J.; Scott, J.G. Emotional and behavioral problems in adolescents and young adults with food allergy. Allergy 2016, 71, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Fong, A.T.; Katelaris, C.H.; Wainstein, B. Bullying and quality of life in children and adolescents with food allergy. J. Paediatr. Child Health 2017, 53, 630–635. [Google Scholar] [CrossRef]
- Flokstra-de Blok, B.M.; van der Velde, J.L.; Vlieg-Boerstra, B.J.; Oude Elberink, J.N.; DunnGalvin, A.; Hourihane, J.O.; Duiverman, E.J.; Dubois, A.E. Health-related quality of life of food allergic patients measured with generic and disease-specific questionnaires. Allergy 2010, 65, 1031–1038. [Google Scholar] [CrossRef] [PubMed]
- Verboom, C.E.; Sijtsema, J.J.; Verhulst, F.C.; Penninx, B.W.; Ormel, J. Longitudinal associations between depressive problems, academic performance, and social functioning in adolescent boys and girls. Dev. Psychol. 2014, 50, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Dunn Galvin, A.; Hourihane, J.O. Health-related quality of life in food allergy: Impact, correlates, and predictors. Bundesgesundheitsblatt-Gesundheitsforschung-Gesundheitsschutz 2016, 59, 841–848. [Google Scholar] [CrossRef]
- Stukus, D.R.; Nassef, M.; Rubin, M. Leaving home: Helping teens with allergic conditions become independent. Ann. Allergy Asthma Immunol. 2016, 116, 388–391. [Google Scholar] [CrossRef] [PubMed]
- Stensgaard, A.; Bindslev-Jensen, C.; Nielsen, D.; Munch, M.; DunnGalvin, A. Quality of life in childhood, adolescence and adult food allergy: Patient and parent perspectives. Clin. Exp. Allergy 2017, 47, 530–539. [Google Scholar] [CrossRef] [PubMed]
- Venter, C.; Sommer, I.; Moonesinghe, H.; Grundy, J.; Glasbey, G.; Patil, V.; Dean, T. Health-related quality of life in children with perceived and diagnosed food hypersensitivity. Pediatric Allergy Immunol. 2015, 26, 126–132. [Google Scholar] [CrossRef]
- Wassenberg, J.; Cochard, M.M.; Dunngalvin, A.; Ballabeni, P.; Flokstra-de Blok, B.M.; Newman, C.J.; Hofer, M.; Eigenmann, P.A. Parent perceived quality of life is age-dependent in children with food allergy. Pediatric Allergy Immunol. 2012, 23, 412–419. [Google Scholar] [CrossRef]
- Protudjer, J.L.P.; Middelveld, R.; Dahlen, S.E.; Ahlstedt, S.; Food, H.E.I. Food allergy-related concerns during the transition to self-management. Allergy Asthma Clin. Immunol. 2019, 15, 54. [Google Scholar] [CrossRef]
- Hamshaw, R.J.T.; Barnett, J.; Gavin, J.; Lucas, J.S. Perceptions of Food Hypersensitivity Expertise on Social Media: Qualitative Study. Interact. J. Med. Res. 2019, 8, e10812. [Google Scholar] [CrossRef]
- Hart, A.; Henwood, F.; Wyatt, S. The role of the Internet in patient-practitioner relationships: Findings from a qualitative research study. J. Med. Internet Res. 2004, 6, e36. [Google Scholar] [CrossRef] [PubMed]
- Haahtela, T.; Valovirta, E.; Bousquet, J.; Makela, M.; Allergy Programme Steering, G. The Finnish Allergy Programme 2008–2018 works. Eur. Respir. J. 2017, 49, 1700470. [Google Scholar] [CrossRef] [PubMed]
- Quigley, J.; Sanders, G.M. Food Allergy in Patients Seeking Mental Health Care: What the Practicing Psychiatrist Should Know. Curr. Psychiatry Rep. 2017, 19, 99. [Google Scholar] [CrossRef] [PubMed]
| Dimensions | No FHS n = 2340 (%) | FHS n = 650 (%) | Crude OR a (95% CI) | Crude p-Value c,d | Adjusted b,d OR (95% CI) | Adjusted b,d p-Value |
|---|---|---|---|---|---|---|
| Mobility | 1.64 (0.77–3.49) | 0.19 | 1.29 (0.54–3.13) | 0.57 | ||
| No Problems | 2318 (99.06) | 640 (98.46) | ||||
| Some Problems | 21 (0.90) | 10 (1.54) | ||||
| Extreme Problems | 1 (0.04) | 0 (0.00) | ||||
| Self-Care | 0.56 (0.17–1.92) | 0.35 | 0.43 (0.10–1.93) | 0.27 | ||
| No Problems | 2321 (99.19) | 647 (99.54) | ||||
| Some Problems | 10 (0.43) | 1 (0.15) | ||||
| Extreme Problems | 9 (0.38) | 2 (0.31) | ||||
| Usual Activities | 1.54 (1.01–2.35) | 0.04 | 1.51 (0.94–2.44) | 0.08 | ||
| No Problems | 2264 (96.75) | 618 (95.08) | ||||
| Some Problems | 68 (2.91) | 32 (4.92) | ||||
| Extreme Problems | 8 (0.34) | 0 (0.00) | ||||
| Pain or Discomfort | 1.49 (1.22–1.85) | <0.001 | 1.37 (1.08–1.72) | 0.007 | ||
| No Problems | 1930 (82.48) | 493 (75.85) | ||||
| Some Problems | 402 (17.18) | 156 (24.00) | ||||
| Extreme Problems | 8 (0.34) | 1 (0.15) | ||||
| Anxiety or Depression | 1.31 (1.08–1.60) | 0.007 | 1.24 (0.99–1.55) | 0.05 | ||
| No Problems | 1831 (78.25) | 476 (73.23) | ||||
| Some Problems | 479 (20.47) | 164 (25.23) | ||||
| Extreme Problems | 30 (1.28) | 10 (1.54) | ||||
| EQ VAS | Crude Beta-coefficient (95% CI) | Crude p-value | Adjusted c Beta-coefficient (95% CI) | Adjusted c p-value | ||
| EQ VAS Mean (SD) | 85.68 (13.31) | 83.91 (14.19) | 0.003 e | −1.4 (−2.64−0.19) | 0.023 | |
| No FHS | FHS | Crude Beta-coefficient (95% CI) | Adjusted c Beta-coefficient (95% CI) | |||
| EQ VAS Median (IQR) | 90 (80–95) | 89 (80–94.5) | 0.001 f | 0 (−0.97−0.98) | 1.00 |
| Dimensions | Females with FHS n = 363 (%) | Males with FHS n = 287 (%) | Crude OR a (95% CI) | Crude p-Value | Adjusted b OR a (95% CI) | Adjusted b p-Value | ||
|---|---|---|---|---|---|---|---|---|
| Mobility | 0.84 (0.24–3.01) | 0.79 | 0.86 (0.18–3.98) | 0.846 | ||||
| No Problems | 357 | (98.35) | 283 | (98.61) | ||||
| Some Problems | 6 | (1.65) | 4 | (1.39) | ||||
| Extreme Problems | 0 | (0.00) | 0 | (0.00) | ||||
| Self-Care | 2.54 (0.23–28.16) | 0.45 | 1.38 (0.08–22.19) | 0.821 | ||||
| No Problems | 362 | (99.72) | 285 | (99.30) | ||||
| Some Problems | 0 | (0.00) | 1 | (0.35) | ||||
| Extreme Problems | 1 | (0.28) | 1 | (0.35) | ||||
| Usual Activities | 0.75 (0.36–1.56) | 0.78 | 0.79 (0.35–1.79) | 0.57 | ||||
| No Problems | 343 | (94.49) | 275 | (95.82) | ||||
| Some Problems | 20 | (5.51) | 12 | (4.18) | ||||
| Extreme Problems | 0 | (0.00) | 0 | (0.00) | ||||
| Pain or Discomfort | 0.49 (0.33–0.71) | <0.001 | 0.51 (0.34–0.77) | 0.001 | ||||
| No Problems | 255 | (70.25) | 238 | (82.93) | ||||
| Some Problems | 107 | (29.48) | 49 | (17.07) | ||||
| Extreme Problems | 1 | (0.28) | 0 | (0.00) | ||||
| Anxiety or Depression | 0.54 (0.38–0.78) | <0.001 | 0.55 (0.37–0.82) | 0.003 | ||||
| No Problems | 247 | (68.04) | 229 | (79.79) | ||||
| Some Problems | 111 | (30.58) | 53 | (18.47) | ||||
| Extreme Problems | 5 | (1.38) | 5 | (1.74) | ||||
| EQ VAS | Female | Male | Crude Beta-coefficient (95% CI) | Crude p-value | Adjusted Beta-coefficient (95%CI) | Adjusted p-value | ||
| Mean (SD) | 82.62 (14.49) | 85.53 (13.67) | 2.91 (0.72–5.10) | 0.009 | 2.88 (0.64–5.12) | 0.012 | ||
| Crude Beta-coefficient (95%CI) | Adjusted c Beta-coefficient (95%CI) | |||||||
| Median (IQR) | 85 (75–90) | 90 (80–95) | 5.0 (2.31–7.69) | 0.002 | 2.0 (1.24–6.76) | 0.005 | ||
| Dimensions | FHS with Mild-to-Moderate Allergic Symptoms a n = 438 | FHS with Severe Allergic Symptoms b n = 155 | p-Value c,d |
|---|---|---|---|
| Mobility | 0.62 | ||
| No Problems | 432 (98.63) | 152 (98.06) | |
| Some Problems | 6 (1.37) | 3 (1.94) | |
| Extreme Problems | 0 (0.00) | 0 (0.00) | |
| Self-Care | 0.30 | ||
| No Problems | 435 (99.32) | 155 (100) | |
| Some Problems | 0 (0.00) | 0 (0.00) | |
| Extreme Problems | 1 (0.00) | 0 (0.00) | |
| Usual Activities | 0.89 | ||
| No Problems | 417 (95.21) | 148 (95.48) | |
| Some Problems | 21 (4.79) | 7 (4.52) | |
| Extreme Problems | 0 (0.00) | 0 (0.00) | |
| Pain or Discomfort | 0.64 | ||
| No Problems | 331 (75.57) | 120 (77.42) | |
| Some Problems | 106 (24.20) | 35 (22.58) | |
| Extreme Problem | 1 (0.23) | 0 (0.00) | |
| Anxiety or Depression | 0.39 | ||
| No Problems | 321 (73.29) | 119 (76.77) | |
| Some Problems | 108 (24.66) | 36 (23.23) | |
| Extreme Problems | 9 (2.05) | 0 (0.00) | |
| EQ VAS Mean (SD) | 84.0 (14.5) | 83.8 (11.9) | 0.87 e |
| EQ VAS Median (IQR) | 89.5 (80–95) | 86 (80–90) | 0.27 f |
| Dimensions | FHS with IgE Sensitization n = 164 (%) | FHS without IgE Sensitization n = 397 (%) | p-Value a,b |
|---|---|---|---|
| Mobility | 0.10 | ||
| No Problems | 160 (97.56) | 394 (99.24) | |
| Some Problems | 4 (2.44) | 3 (0.76) | |
| Extreme Problems | 0 (0.00) | 0 (0.00) | |
| Self-Care | 0.52 | ||
| No Problems | 164 (100.00) | 396 (99.75) | |
| Some Problems | 0 (0.00) | 1 (0.25) | |
| Extreme Problems | 1 (0.28) | 0 (0.00) | |
| Usual Activities | 0.70 | ||
| No Problems | 157 (95.73) | 377 (94.96) | |
| Some Problems | 7 (4.27) | 20 (5.04) | |
| Extreme Problems | 0 (0.00) | 0 (0.00) | |
| Pain or Discomfort | 0.81 | ||
| No Problems | 126 (76.83) | 303 (76.32) | |
| Some Problems | 38 (23.17) | 93 (23.43) | |
| Extreme Problems | 0 (0.00) | 1 (0.25) | |
| Anxiety or Depression | 0.65 | ||
| No Problems | 117 (71.34) | 297 (74.81) | |
| Some Problems | 44 (26.83) | 95 (23.93) | |
| Extreme Problems | 3 (1.83) | 5 (1.26) | |
| EQ VAS Mean (SD) | 84.47(12.91) | 83.95 (13.9) | 0.68 c |
| EQ VAS Median (IQR) | 88 (80–90) | 87 (80–95) | 0.75 d |
| Parent-Reported FHS a | ||
|---|---|---|
| Adolescent-Reported Food Reactions b | No | Yes |
| No | 2174 | 280 |
| Yes | 159 | 370 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jonsson, M.; Ekström, S.; Protudjer, J.L.P.; Bergström, A.; Kull, I. Living with Food Hypersensitivity as an Adolescent Impairs Health Related Quality of Life Irrespective of Disease Severity: Results from a Population-Based Birth Cohort. Nutrients 2021, 13, 2357. https://doi.org/10.3390/nu13072357
Jonsson M, Ekström S, Protudjer JLP, Bergström A, Kull I. Living with Food Hypersensitivity as an Adolescent Impairs Health Related Quality of Life Irrespective of Disease Severity: Results from a Population-Based Birth Cohort. Nutrients. 2021; 13(7):2357. https://doi.org/10.3390/nu13072357
Chicago/Turabian StyleJonsson, Marina, Sandra Ekström, Jennifer L. P. Protudjer, Anna Bergström, and Inger Kull. 2021. "Living with Food Hypersensitivity as an Adolescent Impairs Health Related Quality of Life Irrespective of Disease Severity: Results from a Population-Based Birth Cohort" Nutrients 13, no. 7: 2357. https://doi.org/10.3390/nu13072357
APA StyleJonsson, M., Ekström, S., Protudjer, J. L. P., Bergström, A., & Kull, I. (2021). Living with Food Hypersensitivity as an Adolescent Impairs Health Related Quality of Life Irrespective of Disease Severity: Results from a Population-Based Birth Cohort. Nutrients, 13(7), 2357. https://doi.org/10.3390/nu13072357






