Vitamin D Level Trajectories of Adolescent Patients with Anorexia Nervosa at Inpatient Admission, during Treatment, and at One Year Follow Up: Association with Depressive Symptoms
Abstract
1. Introduction
- patients with AN are at risk of 25(OH)D deficiency over the course of AN treatment;
- supplementation of vitamin D during (in-)patient treatment reduces the risk of vitamin D deficiency at discharge;
- 25(OH)D levels are inversely associated with depressive symptoms.
2. Materials and Methods
2.1. Study Sample
2.2. Data Assessments
2.3. Statistical Analysis
3. Results
3.1. Study Sample Characteristics
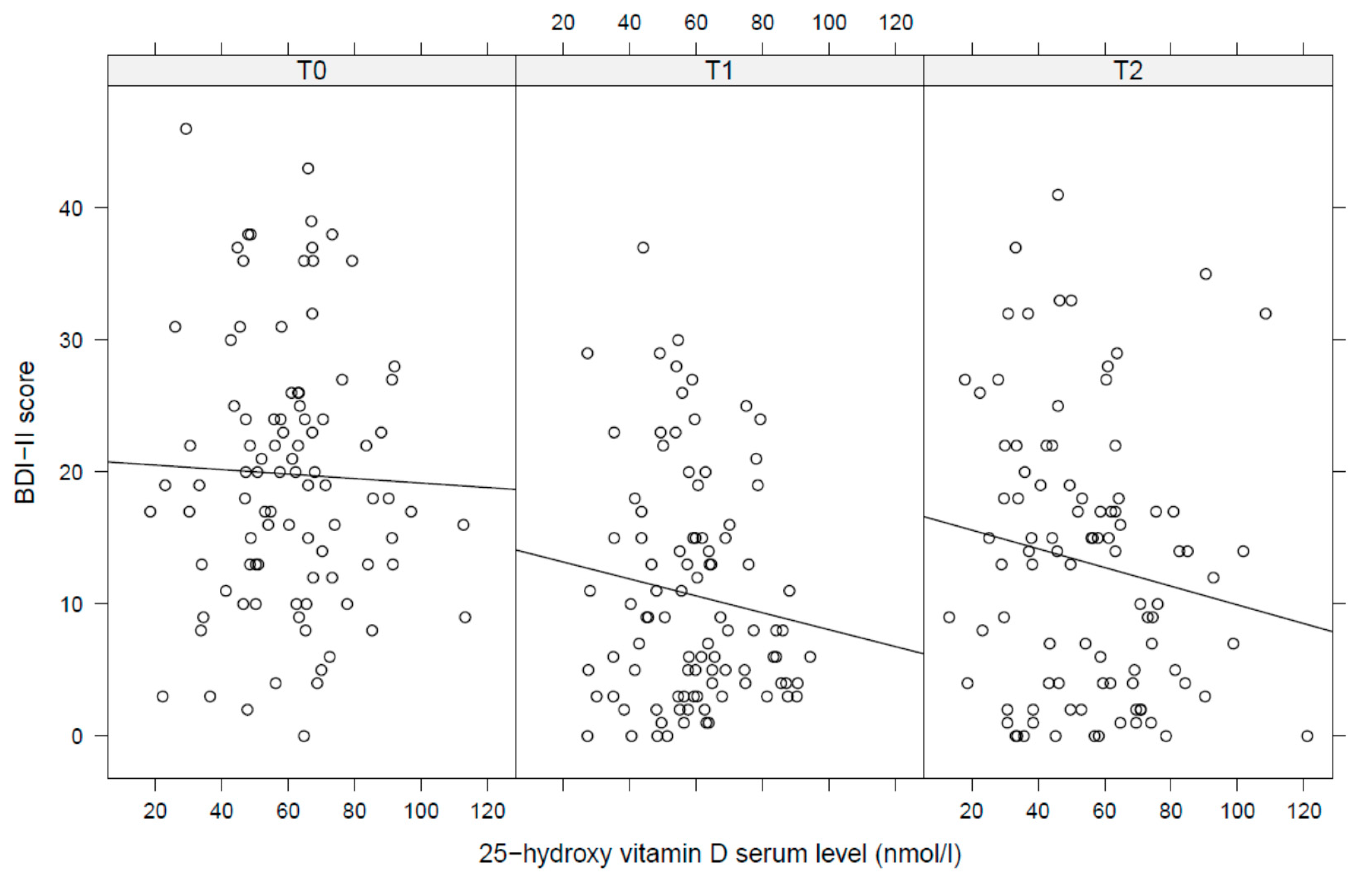
3.2. Trajectories of 25(OH)D Levels and Associations with Depressive Symptoms
4. Discussion
- Vitamin D deficiency (<50 nmol/L) was found in only 30% of patients with AN at admission, but 25(OH)D levels decreased until 1 year follow up. Approximately 50% of the patients were vitamin D deficient after 1 year follow up (T1).
- Vitamin D supplementation during treatment was associated with a lower risk of vitamin D deficiency at discharge.
- 25(OH)D levels were not associated with depressive symptoms, neither cross-sectionally, prospectively, nor longitudinally.
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tasegian, A.; Curcio, F.; Dalla Ragione, L.; Rossetti, F.; Cataldi, S.; Codini, M.; Ambesi-Impiombato, F.S.; Beccari, T.; Albi, E. Hypovitaminosis D3, Leukopenia, and Human Serotonin Transporter Polymorphism in Anorexia Nervosa and Bulimia Nervosa. Mediat. Inflamm. 2016, 2016, 8046479. [Google Scholar] [CrossRef]
- Veronese, N.; Solmi, M.; Rizza, W.; Manzato, E.; Sergi, G.; Santonastaso, P.; Caregaro, L.; Favaro, A.; Correll, C.U. Vitamin D status in anorexia nervosa: A meta-analysis. Int. J. Eat. Disord. 2015, 48, 803–813. [Google Scholar] [CrossRef]
- Mehler, P.S.; Blalock, D.V.; Walden, K.; Kaur, S.; McBride, J.; Walsh, K.; Watts, J. Medical findings in 1026 consecutive adult inpatient-residential eating disordered patients. Int. J. Eat. Disord. 2018, 51, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Hanachi, M.; Dicembre, M.; Rives-Lange, C.; Ropers, J.; Bemer, P.; Zazzo, J.F.; Poupon, J.; Dauvergne, A.; Melchior, J.C. Micronutrients Deficiencies in 374 Severely Malnourished Anorexia Nervosa Inpatients. Nutrients 2019, 11, 792. [Google Scholar] [CrossRef] [PubMed]
- Schleicher, R.L.; Sternberg, M.R.; Lacher, D.A.; Sempos, C.T.; Looker, A.C.; Durazo-Arvizu, R.A.; Yetley, E.A.; Chaudhary-Webb, M.; Maw, K.L.; Pfeiffer, C.M.; et al. The vitamin D status of the US population from 1988 to 2010 using standardized serum concentrations of 25-hydroxyvitamin D shows recent modest increases. Am. J. Clin. Nutr. 2016, 104, 454–461. [Google Scholar] [CrossRef] [PubMed]
- Carlsson, M.; Brudin, L.; Wanby, P. Directly measured free 25-hydroxy vitamin D levels show no evidence of vitamin D deficiency in young Swedish women with anorexia nervosa. Eat. Weight Disord. 2018, 23, 247–254. [Google Scholar] [CrossRef]
- Svedlund, A.; Pettersson, C.; Tubic, B.; Magnusson, P.; Swolin-Eide, D. Vitamin D status in young Swedish women with anorexia nervosa during intensive weight gain therapy. Eur. J. Nutr. 2017, 56, 2061–2067. [Google Scholar] [CrossRef]
- Giollo, A.; Idolazzi, L.; Caimmi, C.; Fassio, A.; Bertoldo, F.; Dalle Grave, R.; El Ghoch, M.; Calugi, S.; Bazzani, P.V.; Viapiana, O.; et al. Vitamin D levels strongly influence bone mineral density and bone turnover markers during weight gain in female patients with anorexia nervosa. Int. J. Eat. Disord. 2017, 50, 1041–1049. [Google Scholar] [CrossRef]
- Schorr, M.; Drabkin, A.; Rothman, M.S.; Meenaghan, E.; Lashen, G.T.; Mascolo, M.; Watters, A.; Holmes, T.M.; Santoso, K.; Yu, E.W.; et al. Bone mineral density and estimated hip strength in men with anorexia nervosa, atypical anorexia nervosa and avoidant/restrictive food intake disorder. Clin. Endocrinol. 2019, 90, 789–797. [Google Scholar] [CrossRef]
- Focker, M.; Antel, J.; Ring, S.; Hahn, D.; Kanal, O.; Ozturk, D.; Hebebrand, J.; Libuda, L. Vitamin D and mental health in children and adolescents. Eur. Child Adolesc. Psychiatry 2017, 26, 1043–1066. [Google Scholar] [CrossRef]
- Tolppanen, A.M.; Sayers, A.; Fraser, W.D.; Lewis, G.; Zammit, S.; Lawlor, D.A. The association of serum 25-hydroxyvitamin D3 and D2 with depressive symptoms in childhood--a prospective cohort study. J. Child Psychol. Psychiatry 2012, 53, 757–766. [Google Scholar] [CrossRef] [PubMed]
- Husmann, C.; Frank, M.; Schmidt, B.; Jockel, K.H.; Antel, J.; Reissner, V.; Libuda, L.; Hebebrand, J.; Focker, M. Low 25(OH)-vitamin D concentrations are associated with emotional and behavioral problems in German children and adolescents. PLoS ONE 2017, 12, e0183091. [Google Scholar] [CrossRef]
- Ju, S.Y.; Lee, Y.J.; Jeong, S.N. Serum 25-hydroxyvitamin D levels and the risk of depression: A systematic review and meta-analysis. J. Nutr. Health Aging 2013, 17, 447–455. [Google Scholar] [CrossRef]
- Spedding, S. Vitamin D and depression: A systematic review and meta-analysis comparing studies with and without biological flaws. Nutrients 2014, 6, 1501–1518. [Google Scholar] [CrossRef] [PubMed]
- Gowda, U.; Mutowo, M.P.; Smith, B.J.; Wluka, A.E.; Renzaho, A.M. Vitamin D supplementation to reduce depression in adults: Meta-analysis of randomized controlled trials. Nutrition 2015, 31, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Libuda, L.; Timmesfeld, N.; Antel, J.; Hirtz, R.; Bauer, J.; Fuhrer, D.; Zwanziger, D.; Ozturk, D.; Langenbach, G.; Hahn, D.; et al. Effect of vitamin D deficiency on depressive symptoms in child and adolescent psychiatric patients: Results of a randomized controlled trial. Eur. J. Nutr. 2020. [Google Scholar] [CrossRef] [PubMed]
- Buhren, K.; Schwarte, R.; Fluck, F.; Timmesfeld, N.; Krei, M.; Egberts, K.; Pfeiffer, E.; Fleischhaker, C.; Wewetzer, C.; Herpertz-Dahlmann, B. Comorbid psychiatric disorders in female adolescents with first-onset anorexia nervosa. Eur. Eat. Disord. Rev. 2014, 22, 39–44. [Google Scholar] [CrossRef]
- Herpertz-Dahlmann, B.; Schwarte, R.; Krei, M.; Egberts, K.; Warnke, A.; Wewetzer, C.; Pfeiffer, E.; Fleischhaker, C.; Scherag, A.; Holtkamp, K.; et al. Day-patient treatment after short inpatient care versus continued inpatient treatment in adolescents with anorexia nervosa (ANDI): A multicentre, randomised, open-label, non-inferiority trial. Lancet 2014, 383, 1222–1229. [Google Scholar] [CrossRef]
- APA. Diagnostic and Statistical Manual of Mental Disorders; American Psychiatric Press: Washington, DC, USA, 1994. [Google Scholar]
- Fichter, M.; Quadflieg, N. The structured interview for anorexic and bulimic disorders for DSM-IV and ICD-10 (SIAB-EX): Reliability and validity. Eur. Psychiatry 2001, 16, 38–48. [Google Scholar] [CrossRef]
- DGE. 10 Guidelines of the German Nutrition Society (DGE) for a Wholesome Diet. Available online: https://www.dge.de/fileadmin/public/doc/en/10-guidelines-wholesome-diet-dge.pdf (accessed on 30 June 2021).
- Kumar, G.; Steer, R.A.; Teitelman, K.B.; Villacis, L. Effectiveness of Beck Depression Inventory-II subscales in screening for major depressive disorders in adolescent psychiatric inpatients. Assessment 2002, 9, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Committee to Review Dietary Reference Intakes for Vitamin D and Calcium; Food and Nutrition Board; Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D; National Academy Press: Washington, DC, USA, 2010. [Google Scholar]
- Kromeyer-Hauschild, K.; Jaeger, U. Growth studies in Jena, Germany: Changes in body size and subcutaneous fat distribution between 1975 and 1995. Am. J. Hum. Biol. 1998, 10, 579–587. [Google Scholar] [CrossRef]
- Pereira, M.A.; Jacobs, D.R., Jr. Sugar-sweetened beverages, weight gain and nutritional epidemiological study design. Br. J. Nutr. 2008, 99, 1169–1170. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Thierfelder, W.; Dortschy, R.; Hintzpeter, B.; Kahl, H.; Scheidt-Nave, C. Biochemical measures in the German Health Interview and Examination Survey for Children and Adolescents (KiGGS). Bundesgesundh. Gesundh. Gesundh. 2007, 50, 757–770. [Google Scholar] [CrossRef] [PubMed]
- Saggese, G.; Vierucci, F.; Boot, A.M.; Czech-Kowalska, J.; Weber, G.; Camargo, C.A., Jr.; Mallet, E.; Fanos, M.; Shaw, N.J.; Holick, M.F. Vitamin D in childhood and adolescence: An expert position statement. Eur. J. Pediatr. 2015, 174, 565–576. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Misra, M.; Tsai, P.; Anderson, E.J.; Hubbard, J.L.; Gallagher, K.; Soyka, L.A.; Miller, K.K.; Herzog, D.B.; Klibanski, A. Nutrient intake in community-dwelling adolescent girls with anorexia nervosa and in healthy adolescents. Am. J. Clin. Nutr. 2006, 84, 698–706. [Google Scholar] [CrossRef]
- Lim, H.S.; Byun, D.W.; Suh, K.I.; Park, H.K.; Kim, H.J.; Kim, T.H.; Lee, H.H. Is There a Difference in Serum Vitamin D Levels and Bone Mineral Density According to Body Mass Index in Young Adult Women? J. Bone Metab. 2019, 26, 145–150. [Google Scholar] [CrossRef]
- Dura-Trave, T.; Gallinas-Victoriano, F.; Chueca-Guindulain, M.J.; Berrade-Zubiri, S.; Urretavizcaya-Martinez, M.; Ahmed-Mohamed, L. Assessment of vitamin D status and parathyroid hormone during a combined intervention for the treatment of childhood obesity. Nutr. Diabetes 2019, 9, 18. [Google Scholar] [CrossRef]
- Jaksic, M.; Martinovic, M.; Gligorovic-Barhanovic, N.; Vujacic, A.; Djurovic, D.; Nedovic-Vukovic, M. Association between inflammation, oxidative stress, vitamin D, copper and zinc with pre-obesity and obesity in school children from the city of Podgorica, Montenegro. J. Pediatr. Endocrinol. Metab. 2019, 32, 951–957. [Google Scholar] [CrossRef]
- Resmark, G.; Herpertz, S.; Herpertz-Dahlmann, B.; Zeeck, A. Treatment of Anorexia Nervosa-New Evidence-Based Guidelines. J. Clin. Med. 2019, 8, 153. [Google Scholar] [CrossRef]
- Wong, S.K.; Chin, K.Y.; Ima-Nirwana, S. Vitamin D and Depression: The Evidence from an Indirect Clue to Treatment Strategy. Curr. Drug Targets 2018, 19, 888–897. [Google Scholar] [CrossRef]
- Rejnmark, L.; Bislev, L.S.; Cashman, K.D.; Eiriksdottir, G.; Gaksch, M.; Grubler, M.; Grimnes, G.; Gudnason, V.; Lips, P.; Pilz, S.; et al. Non-skeletal health effects of vitamin D supplementation: A systematic review on findings from meta-analyses summarizing trial data. PLoS ONE 2017, 12, e0180512. [Google Scholar] [CrossRef]
- Li, H.; Sun, D.; Wang, A.; Pan, H.; Feng, W.; Ng, C.H.; Ungvari, G.S.; Tao, L.; Li, X.; Wang, W.; et al. Serum 25-Hydroxyvitamin D Levels and Depression in Older Adults: A Dose-Response Meta-Analysis of Prospective Cohort Studies. Am. J. Geriatr. Psychiatry 2019, 27, 1192–1202. [Google Scholar] [CrossRef]
- Milaneschi, Y.; Peyrot, W.J.; Nivard, M.G.; Mbarek, H.; Boomsma, D.I.; Penninx, B.W. A role for vitamin D and omega-3 fatty acids in major depression? An exploration using genomics. Transl. Psychiatry 2019, 9, 219. [Google Scholar] [CrossRef]
- Libuda, L.; Laabs, B.H.; Ludwig, C.; Buhlmeier, J.; Antel, J.; Hinney, A.; Naaresh, R.; Focker, M.; Hebebrand, J.; Konig, I.R.; et al. Vitamin D and the Risk of Depression: A Causal Relationship? Findings from a Mendelian Randomization Study. Nutrients 2019, 11, 1085. [Google Scholar] [CrossRef]
- Autier, P.; Boniol, M.; Pizot, C.; Mullie, P. Vitamin D status and ill health—Author’s reply. Lancet Diabetes Endocrinol. 2014, 2, 275–276. [Google Scholar] [CrossRef]
- Seitz, J.; Trinh, S.; Herpertz-Dahlmann, B. The Microbiome and Eating Disorders. Psychiatr. Clin. N. Am. 2019, 42, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Lipsman, N.; Woodside, D.B.; Lozano, A.M. Neurocircuitry of limbic dysfunction in anorexia nervosa. Cortex 2015, 62, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Himmerich, H.; Bentley, J.; Kan, C.; Treasure, J. Genetic risk factors for eating disorders: An update and insights into pathophysiology. Ther. Adv. Psychopharmacol. 2019, 9, 2045125318814734. [Google Scholar] [CrossRef] [PubMed]
- King, J.A.; Frank, G.K.W.; Thompson, P.M.; Ehrlich, S. Structural Neuroimaging of Anorexia Nervosa: Future Directions in the Quest for Mechanisms Underlying Dynamic Alterations. Biol. Psychiatry 2018, 83, 224–234. [Google Scholar] [CrossRef]
- Lawson, E.A.; Donoho, D.; Miller, K.K.; Misra, M.; Meenaghan, E.; Lydecker, J.; Wexler, T.; Herzog, D.B.; Klibanski, A. Hypercortisolemia is associated with severity of bone loss and depression in hypothalamic amenorrhea and anorexia nervosa. J. Clin. Endocrinol. Metab. 2009, 94, 4710–4716. [Google Scholar] [CrossRef]
- Lawson, E.A.; Holsen, L.M.; Santin, M.; Meenaghan, E.; Eddy, K.T.; Becker, A.E.; Herzog, D.B.; Goldstein, J.M.; Klibanski, A. Oxytocin secretion is associated with severity of disordered eating psychopathology and insular cortex hypoactivation in anorexia nervosa. J. Clin. Endocrinol. Metab. 2012, 97, E1898–E1908. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hebebrand, J.; Milos, G.; Wabitsch, M.; Teufel, M.; Fuhrer, D.; Buhlmeier, J.; Libuda, L.; Ludwig, C.; Antel, J. Clinical Trials Required to Assess Potential Benefits and Side Effects of Treatment of Patients With Anorexia Nervosa With Recombinant Human Leptin. Front. Psychol. 2019, 10, 769. [Google Scholar] [CrossRef] [PubMed]
- Milos, G.; Antel, J.; Kaufmann, L.K.; Barth, N.; Koller, A.; Tan, S.; Wiesing, U.; Hinney, A.; Libuda, L.; Wabitsch, M.; et al. Short-term metreleptin treatment of patients with anorexia nervosa: Rapid on-set of beneficial cognitive, emotional, and behavioral effects. Transl. Psychiatry 2020, 10, 303. [Google Scholar] [CrossRef] [PubMed]
- Holtkamp, K.; Herpertz-Dahlmann, B.; Hebebrand, K.; Mika, C.; Kratzsch, J.; Hebebrand, J. Physical activity and restlessness correlate with leptin levels in patients with adolescent anorexia nervosa. Biol. Psychiatry 2006, 60, 311–313. [Google Scholar] [CrossRef]
- Schneider, N.; Salbach-Andrae, H.; Merle, J.V.; Hein, J.; Pfeiffer, E.; Lehmkuhl, U.; Ehrlich, S. Psychopathology in underweight and weight-recovered females with anorexia nervosa. Eat. Weight Disord. 2009, 14, e205–e211. [Google Scholar] [CrossRef] [PubMed]
- Gatti, D.; El Ghoch, M.; Viapiana, O.; Ruocco, A.; Chignola, E.; Rossini, M.; Giollo, A.; Idolazzi, L.; Adami, S.; Dalle Grave, R. Strong relationship between vitamin D status and bone mineral density in anorexia nervosa. Bone 2015, 78, 212–215. [Google Scholar] [CrossRef]
- Emslie, G.J.; Heiligenstein, J.H.; Wagner, K.D.; Hoog, S.L.; Ernest, D.E.; Brown, E.; Nilsson, M.; Jacobson, J.G. Fluoxetine for acute treatment of depression in children and adolescents: A placebo-controlled, randomized clinical trial. J. Am. Acad. Child Adolesc. Psychiatry 2002, 41, 1205–1215. [Google Scholar] [CrossRef]
- Van der Ende, J.; Verhulst, F.C.; Tiemeier, H. Agreement of informants on emotional and behavioral problems from childhood to adulthood. Psychol. Assess. 2012, 24, 293–300. [Google Scholar] [CrossRef]
- Salbach-Andrae, H.; Klinkowski, N.; Lenz, K.; Pfeiffer, E.; Lehmkuhl, U.; Ehrlich, S. Correspondence between self-reported and parent-reported psychopathology in adolescents with eating disorders. Psychopathology 2008, 41, 307–312. [Google Scholar] [CrossRef]
| Characteristics at Inpatient Admission | |||
|---|---|---|---|
| Age (years) | 15.3 (±1.5) | ||
| Duration of illness (weeks) | 47.3 (±36.3) | ||
| Premorbid BMI kg/m2 | 20.2 (±2.8) | ||
| AN-subtype (N (%)) | |||
| Restrictive | 79 (85%) | ||
| Binge-Purge | 14 (15%) | ||
| Diagnosis of affective disorder (N (%)) * | 25 (31%) | ||
| Vitamin D supplementation (N (%)) | 50 (54%) | ||
| Characteristics During Treatment | |||
| T0 | T1 | T2 | |
| BMI (kg/m2) | 15.1 (±1.3) | 18.2 (±0.9) | 18.4 (±1.9) |
| BMI percentile | 1.9 (±3.2) | 19.3 (±7.9) | 20.5 (±20.8) |
| BMI-SDS | −2.67 (±0.91) | −0.93 (±0.38) | −1.04 (±0.83) |
| BDI-II score | 19.8 (±10.1) | 10.7 (±8.6) | 13.1 (±10.4) |
| 25(OH)D (nmol/L) | 60.3 (±19.0) | 59.2 (±16.2) | 55.1 (±21.6) |
| Vitamin D deficiency (<20 ng/mL (50 nmol/L)) (N (%)) | 28 (30%) | 27 (29%) | 42 (45%) |
| Season at measuring time point (N (%)): | |||
| Winter (Nov–Mar) | 37 (40%) | 34 (37%) | 34 (37%) |
| Summer (Jul–Oct) | 28 (30%) | 40 (43%) | 28 (30%) |
| Spring (Apr–Jun) | 28 (30%) | 19 (20%) | 31 (33%) |
| Antidepressants | 10 (10.8%) | 18 (19.4%) | |
| Outcome: BDI-II Score at Each Measuring Time Point (T0, T1, T2) | |||
|---|---|---|---|
| ß | 95% CI | p | |
| Intercept | 7.45 | −9.80, 24.71 | 0.412 |
| 25(OH)D estimates | |||
| Cross-sectional: 25(OH)D T0 | −0.02 | −0.12, 0.08 | 0.682 |
| Prospective: 25(OH)D at T0 on change in BDI between T0 × T1 | −0.05 | −0.18, 0.07 | 0.388 |
| Prospective: 25(OH)D at T0 on change in BDI between T0 × T2 | −0.02 | −0.14, 0.09 | 0.684 |
| Longitudinal: Change of 25(OH)D on concurrent change in BDI | −0.04 | −0.11, 0.04 | 0.385 |
| Covariates | |||
| Measuring time point: T1 | −2.15 | −12.71, 8.38 | 0.695 |
| Measuring time point: T2 | −3.27 | −13.63, 6.96 | 0.541 |
| Age | 0.91 | −0.16, 1.99 | 0.109 |
| AN Subtype (Binge–Purge) | 6.28 | 2.27, 10.30 | 0.004 |
| Antidepressant usage | 5.50 | 1.87, 9.29 | 0.004 |
| Duration of illness | −0.02 | −0.05, 0.02 | 0.462 |
| BMI-SDS T0 | 0.23 | −1.84, 2.30 | 0.833 |
| Prospective: BMI-SDS at T0 on change in BDI between T0 × T1 | 1.49 | −1.37, 4.35 | 0.318 |
| Prospective: BMI-SDS at T0 on change in BDI between T0 × T2 | 1.09 | −1.68, 3.84 | 0.45 |
| Longitudinal: Change of BMI-SDS on concurrent change in BDI | −0.22 | −2.32, 1.90 | 0.845 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Föcker, M.; Timmesfeld, N.; Bühlmeier, J.; Zwanziger, D.; Führer, D.; Grasemann, C.; Ehrlich, S.; Egberts, K.; Fleischhaker, C.; Wewetzer, C.; et al. Vitamin D Level Trajectories of Adolescent Patients with Anorexia Nervosa at Inpatient Admission, during Treatment, and at One Year Follow Up: Association with Depressive Symptoms. Nutrients 2021, 13, 2356. https://doi.org/10.3390/nu13072356
Föcker M, Timmesfeld N, Bühlmeier J, Zwanziger D, Führer D, Grasemann C, Ehrlich S, Egberts K, Fleischhaker C, Wewetzer C, et al. Vitamin D Level Trajectories of Adolescent Patients with Anorexia Nervosa at Inpatient Admission, during Treatment, and at One Year Follow Up: Association with Depressive Symptoms. Nutrients. 2021; 13(7):2356. https://doi.org/10.3390/nu13072356
Chicago/Turabian StyleFöcker, Manuel, Nina Timmesfeld, Judith Bühlmeier, Denise Zwanziger, Dagmar Führer, Corinna Grasemann, Stefan Ehrlich, Karin Egberts, Christian Fleischhaker, Christoph Wewetzer, and et al. 2021. "Vitamin D Level Trajectories of Adolescent Patients with Anorexia Nervosa at Inpatient Admission, during Treatment, and at One Year Follow Up: Association with Depressive Symptoms" Nutrients 13, no. 7: 2356. https://doi.org/10.3390/nu13072356
APA StyleFöcker, M., Timmesfeld, N., Bühlmeier, J., Zwanziger, D., Führer, D., Grasemann, C., Ehrlich, S., Egberts, K., Fleischhaker, C., Wewetzer, C., Wessing, I., Seitz, J., Herpertz-Dahlmann, B., Hebebrand, J., & Libuda, L. (2021). Vitamin D Level Trajectories of Adolescent Patients with Anorexia Nervosa at Inpatient Admission, during Treatment, and at One Year Follow Up: Association with Depressive Symptoms. Nutrients, 13(7), 2356. https://doi.org/10.3390/nu13072356







