Egg White Protein Feeding Facilitates Skeletal Muscle Gain in Young Rats with/without Clenbuterol Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Experimental Diets
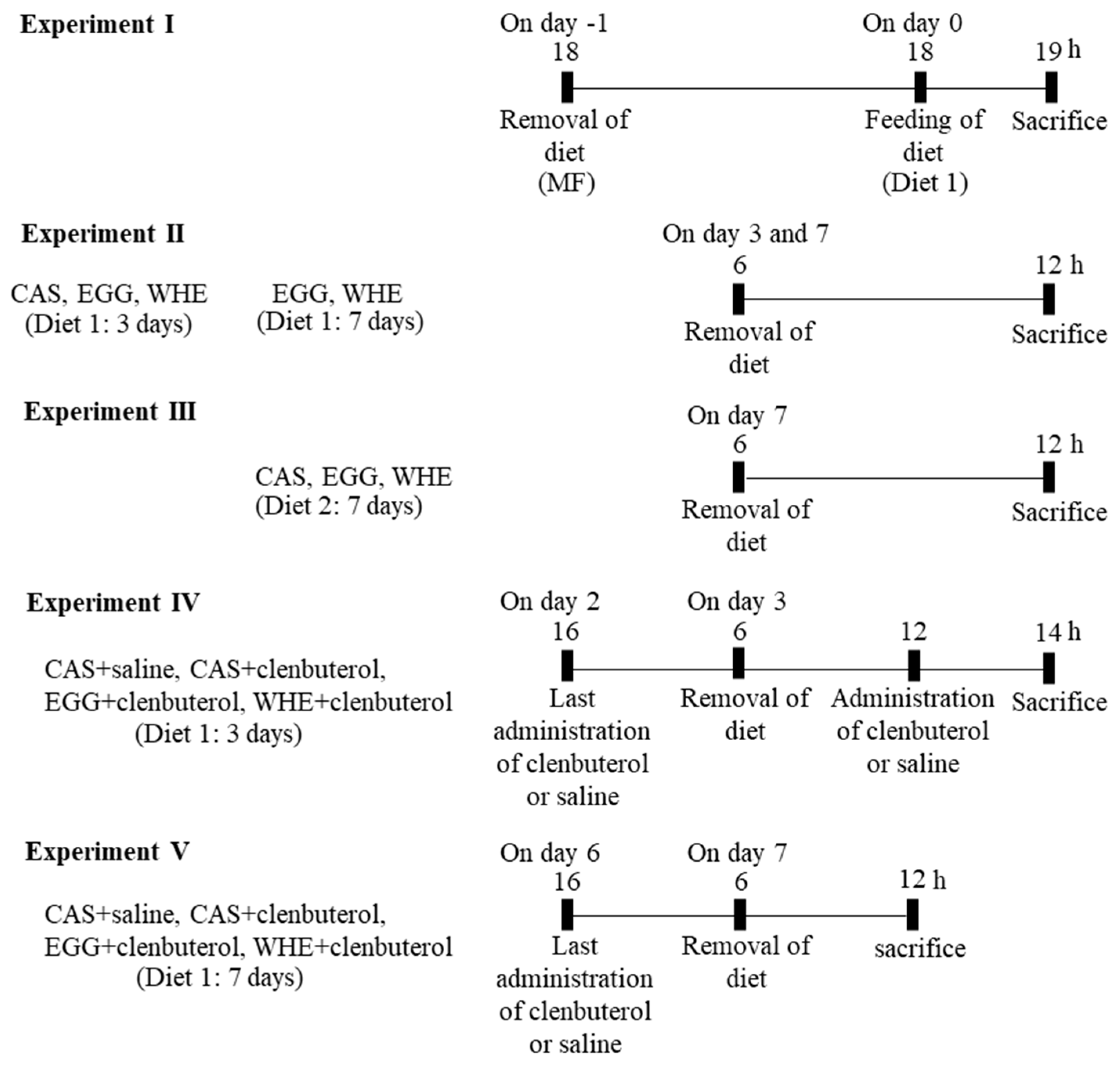
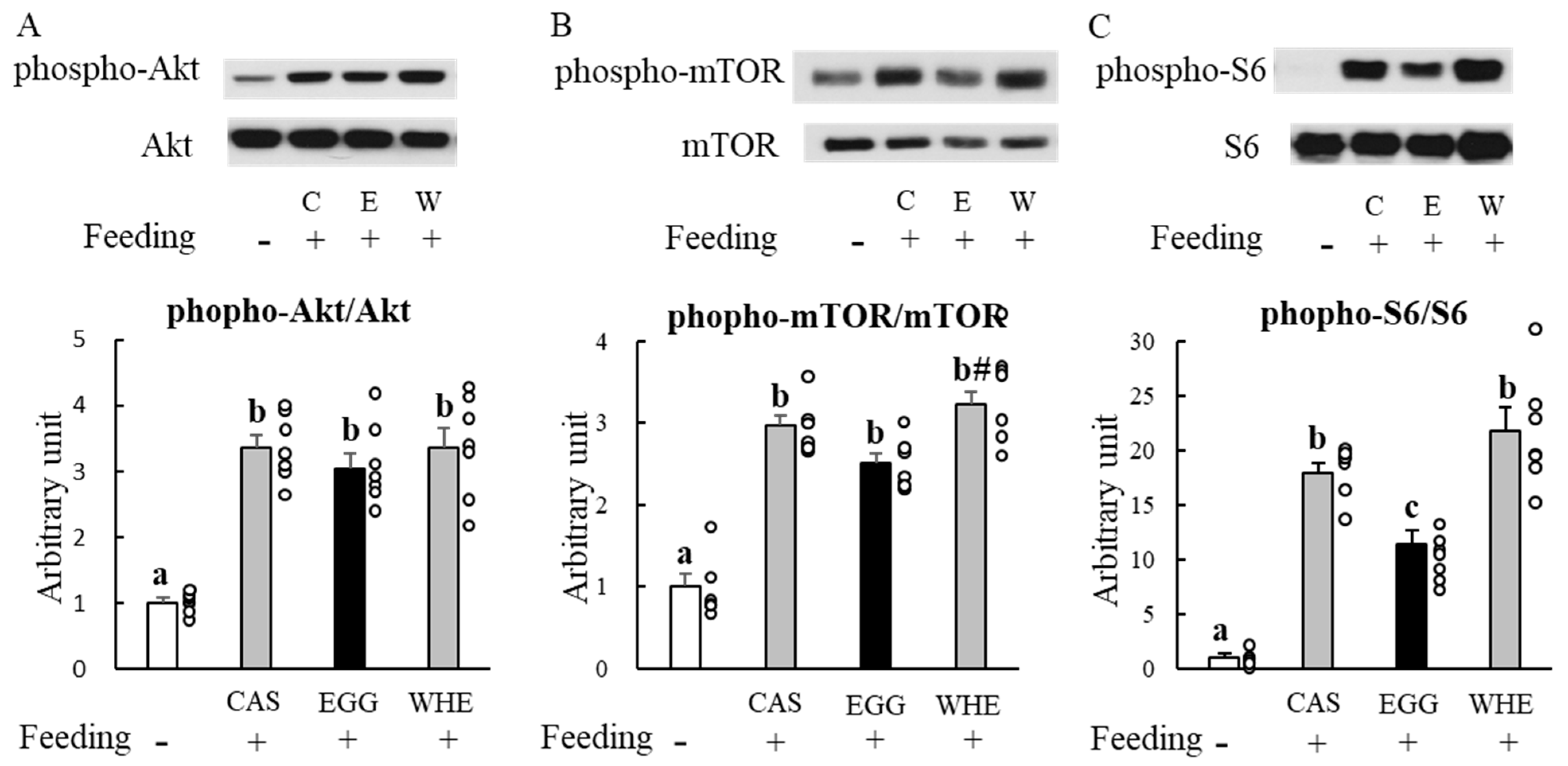
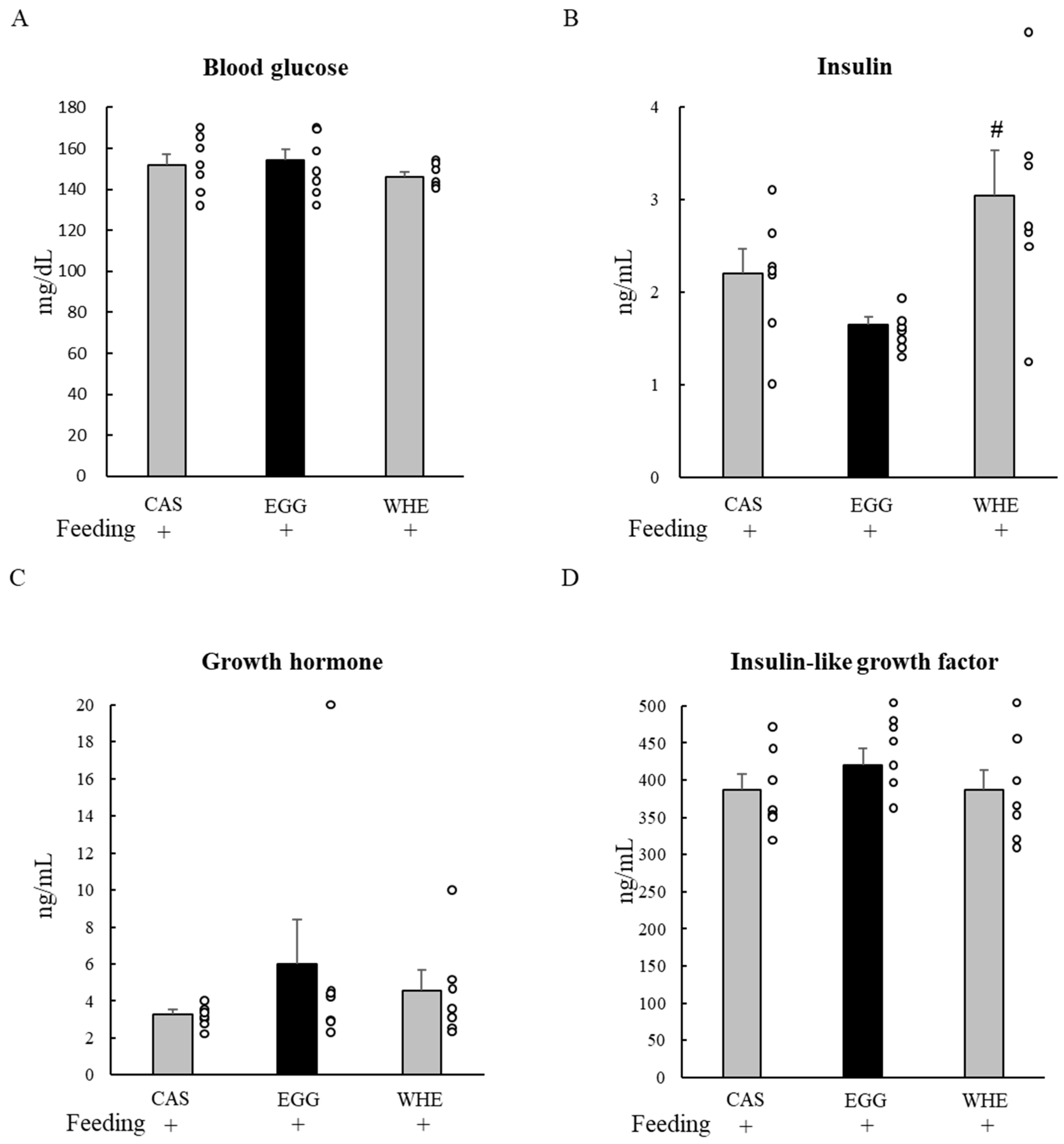
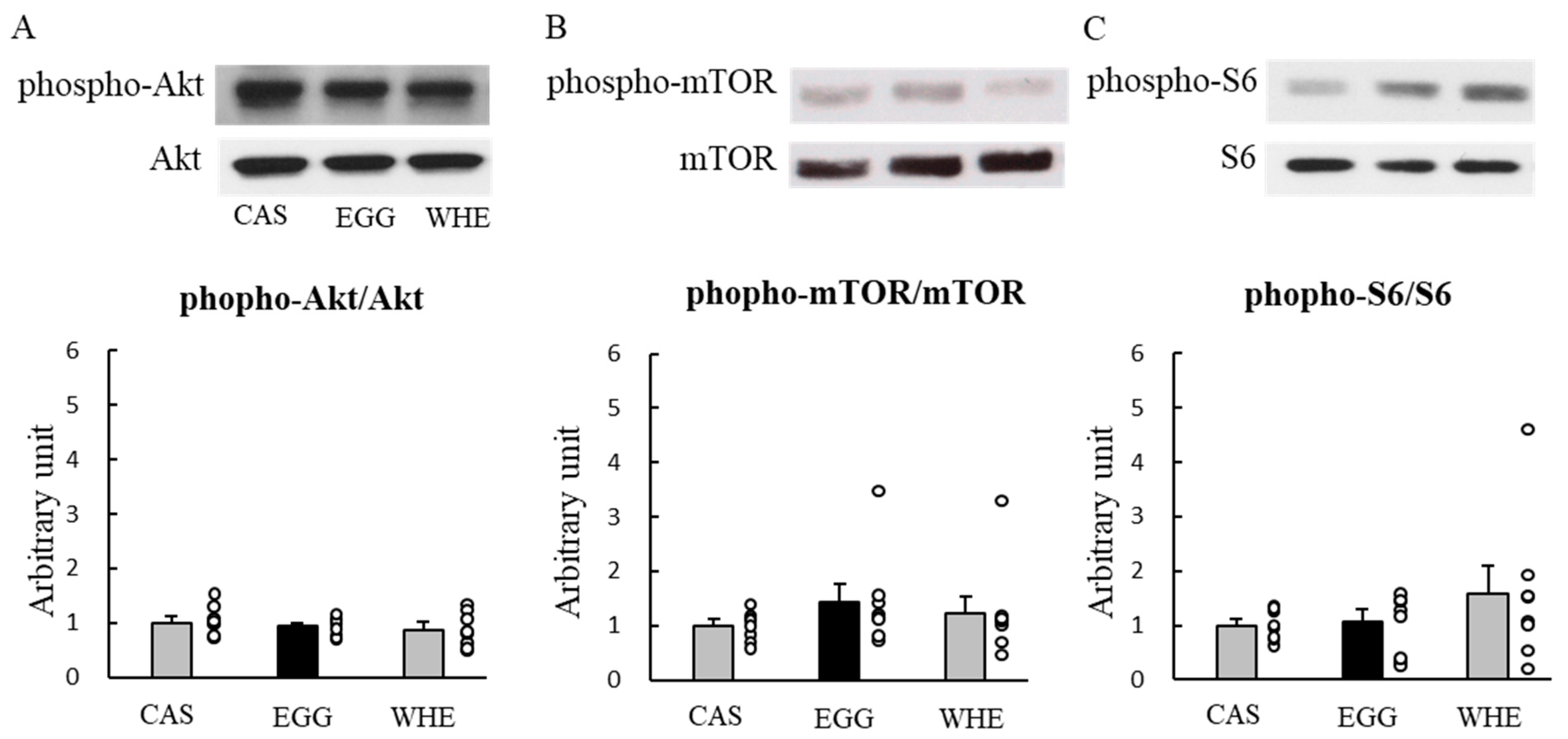
2.3. Experiment I: Acute Effect of EGG Feeding on Muscle Signal Transduction (on Day 0) under Normal Conditions (without Clenbuterol Treatment)
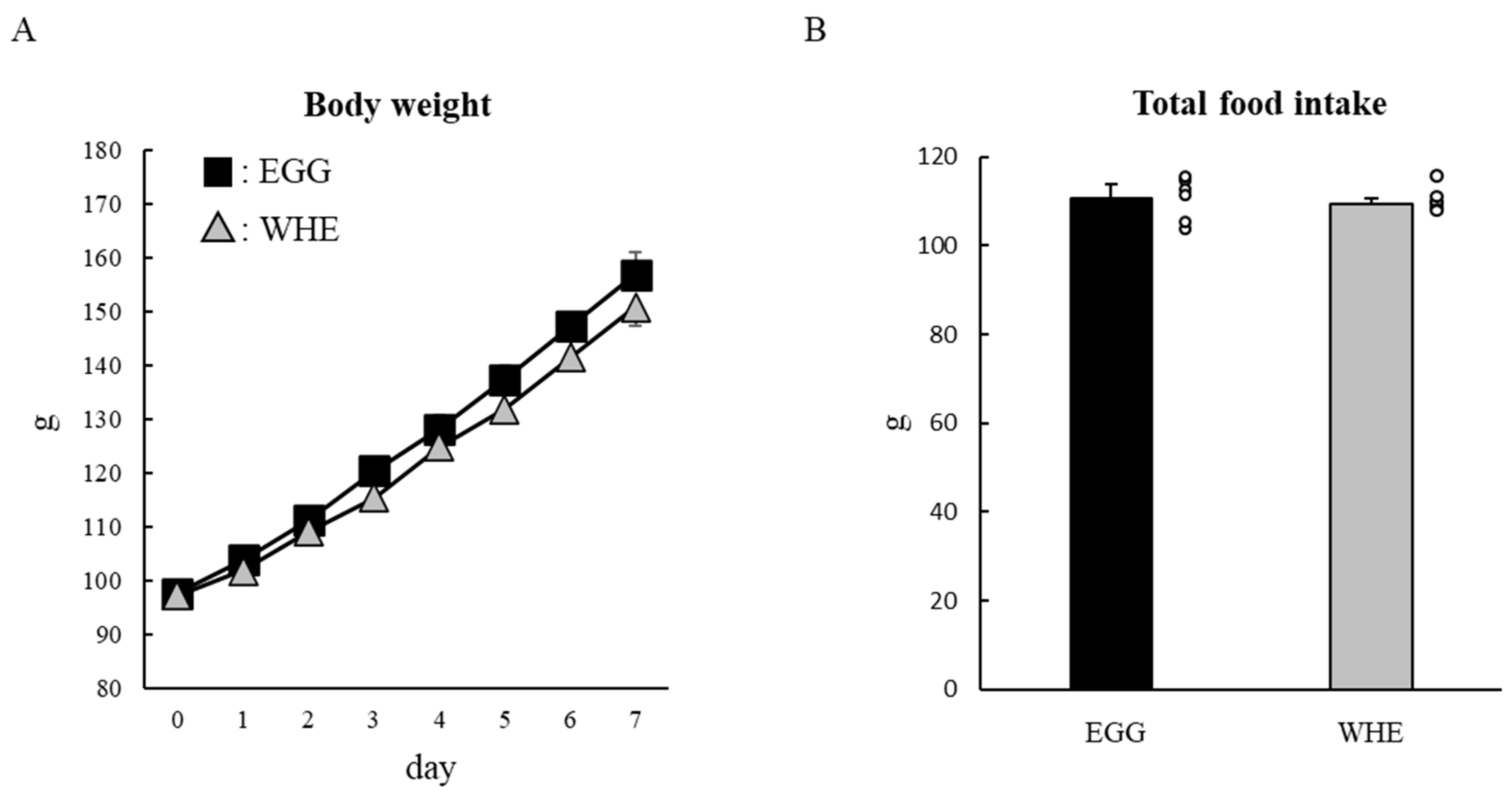
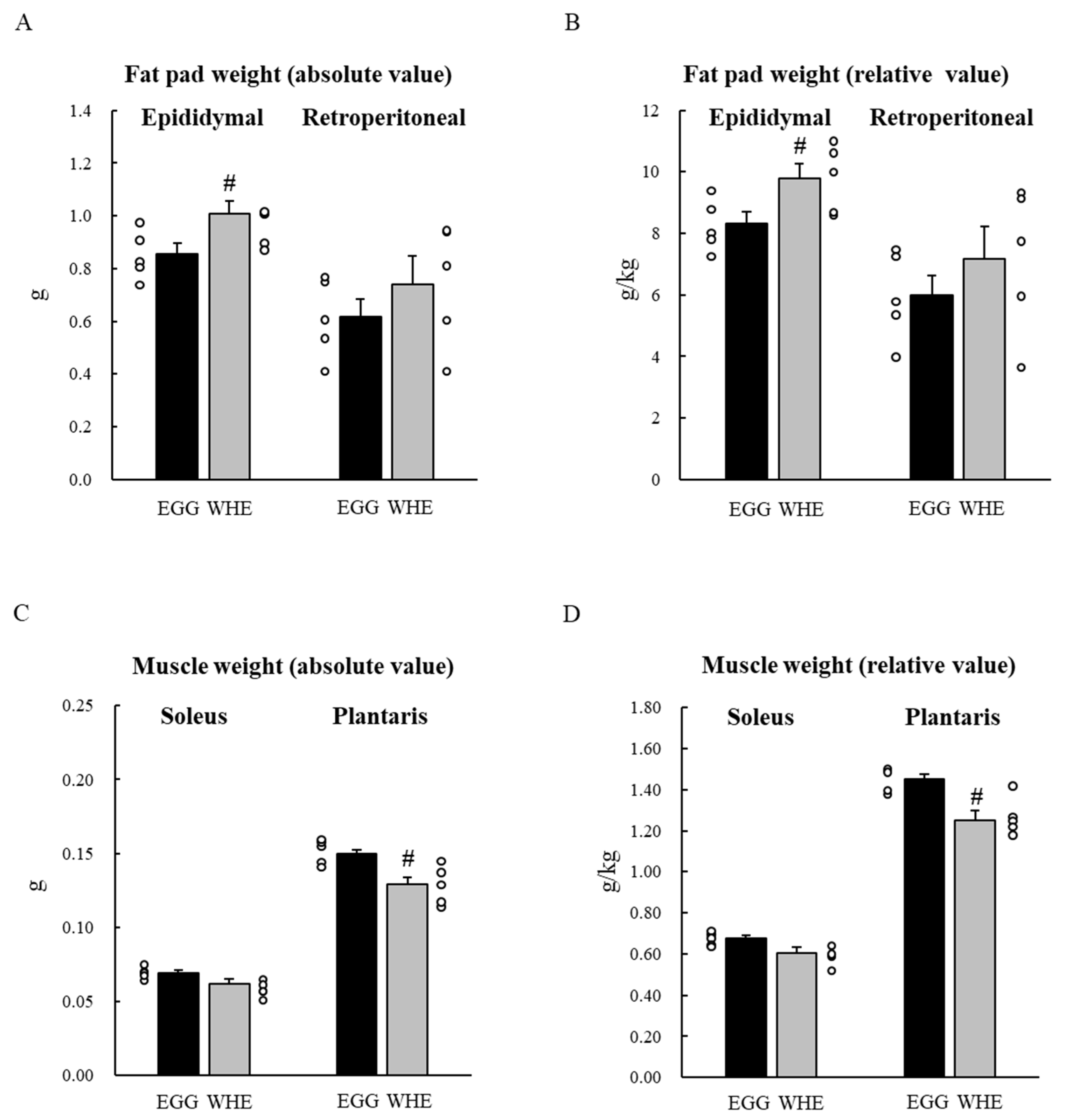
2.4. Experiment II: Chronic (7-Day) Effect of EGG Feeding under Normal Conditions (without Clenbuterol Treatment)
2.5. Experiment III: Chronic Effect of EGG Feeding on Arginine Modification (without Clenbuterol Treatment)
2.6. Experiment IV: Acute Effect of Clenbuterol on Muscle Signal Transduction on Day 3
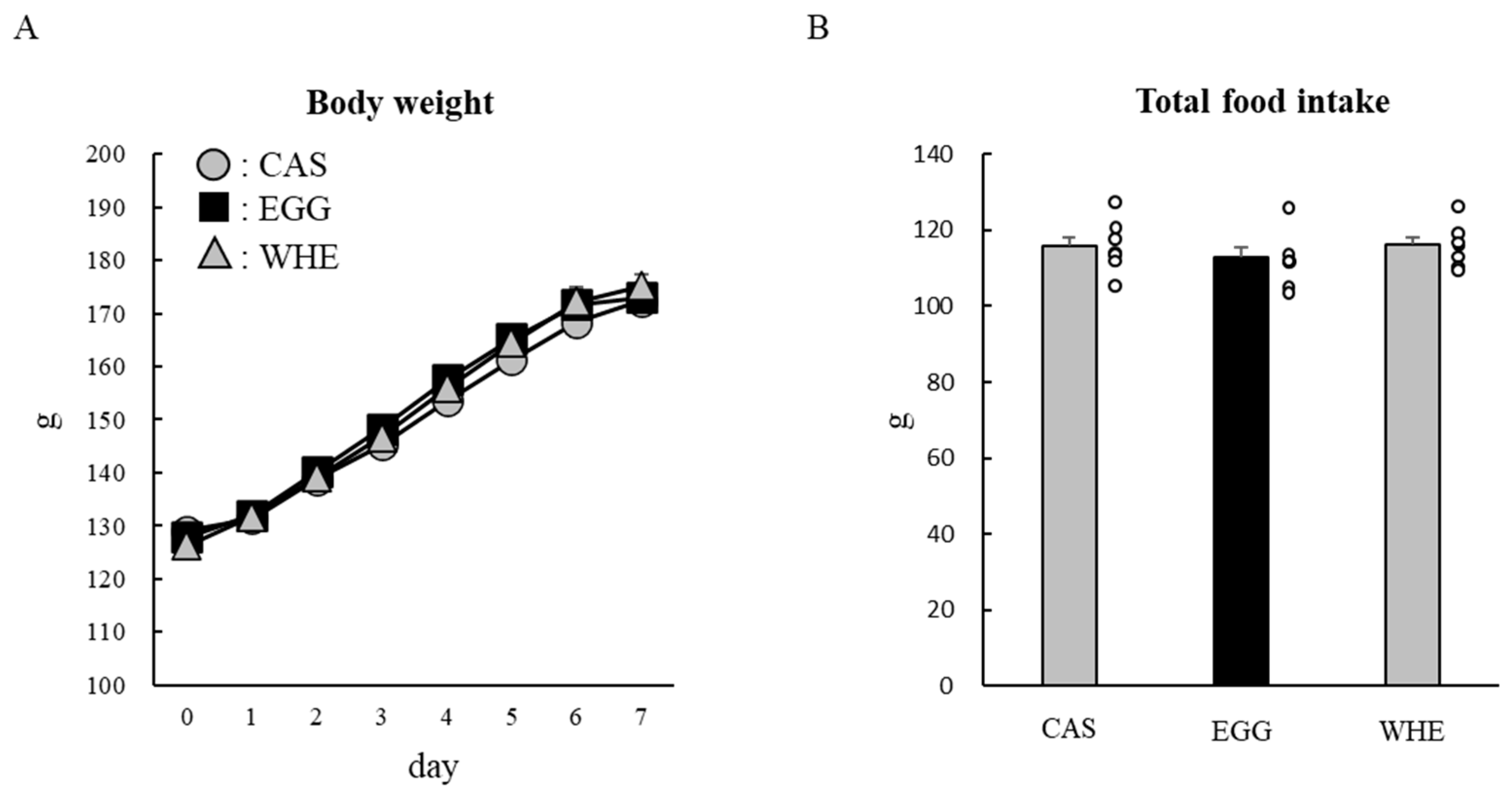
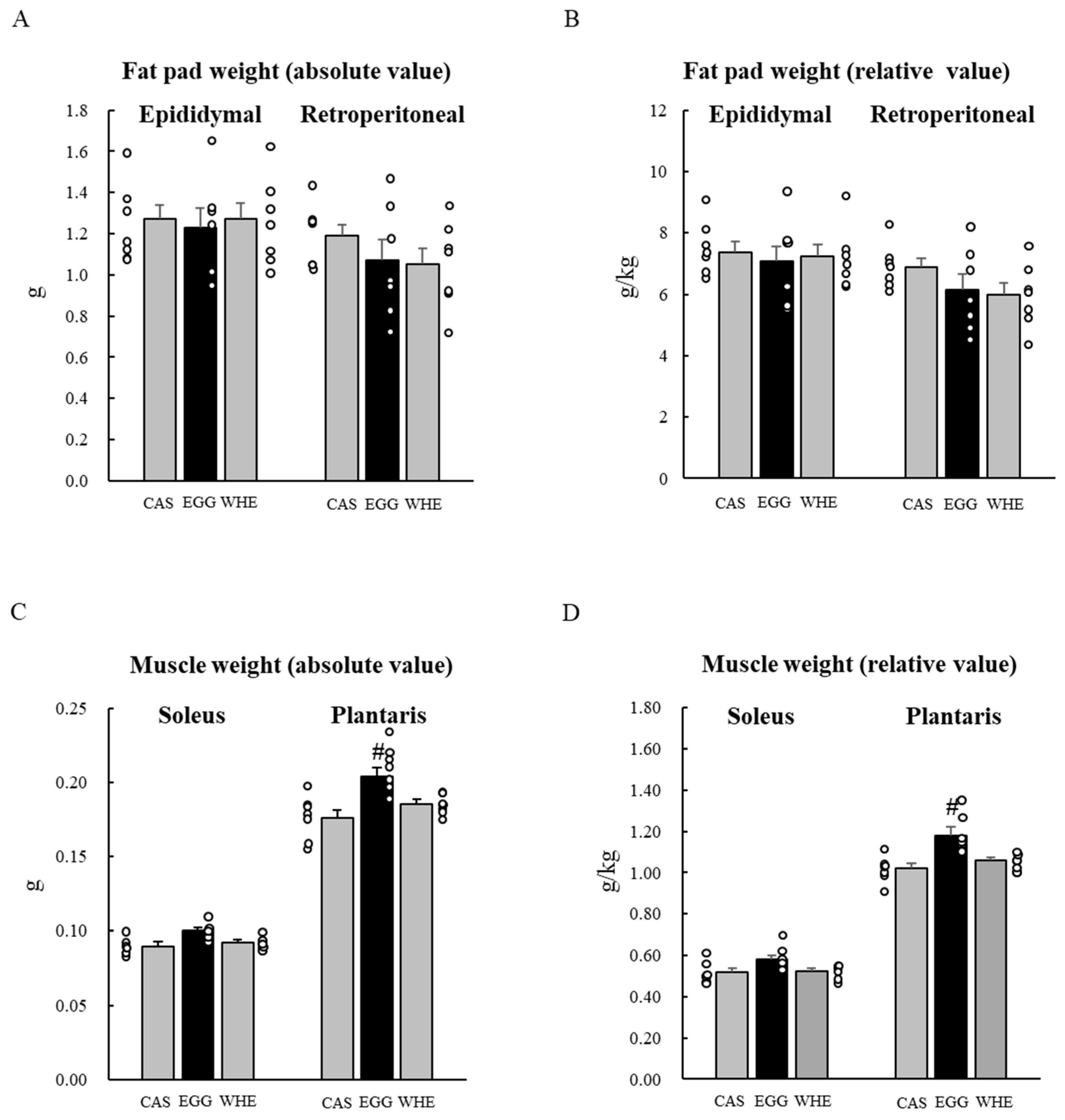
2.7. Experiment V: Chronic (7-Day) Effect of EGG Feeding with Clenbuterol
2.8. Western Blot Analysis
2.9. Blood Parameters
2.10. Amino Acid and Protein Compositions
2.11. Statistics
3. Results
3.1. Experiment I
3.2. Experiment II
3.3. Experiment III
3.4. Experiment IV
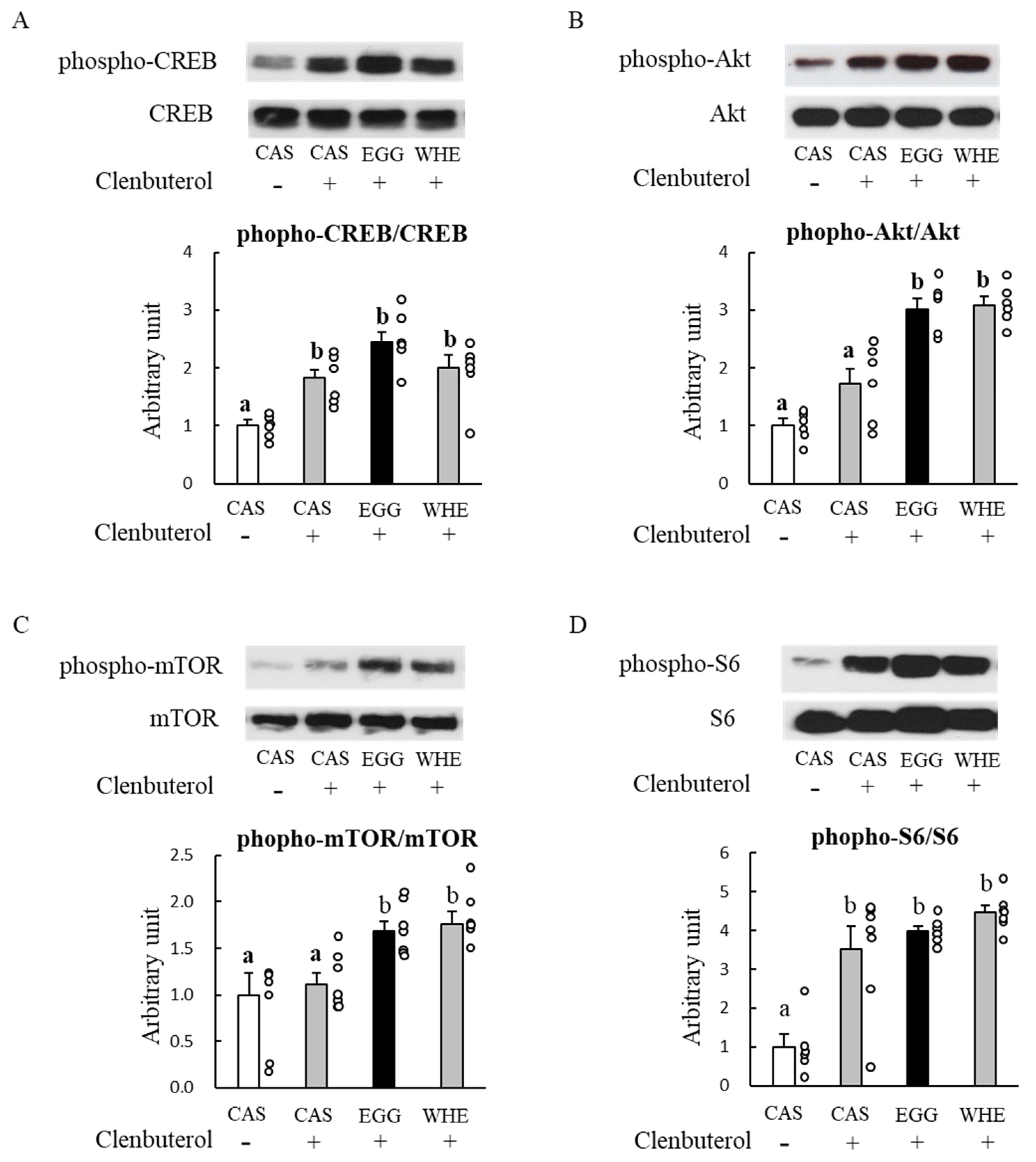
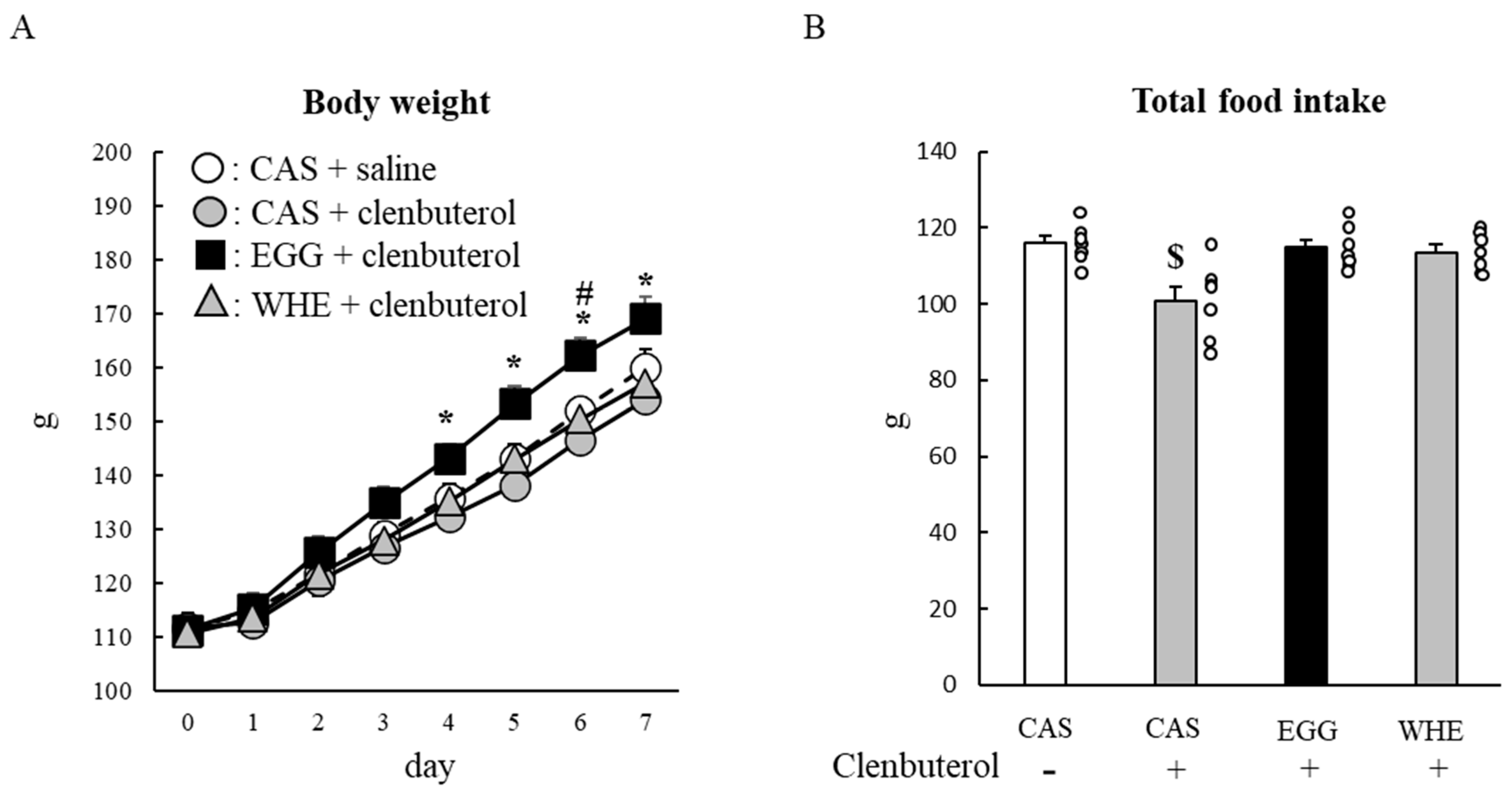
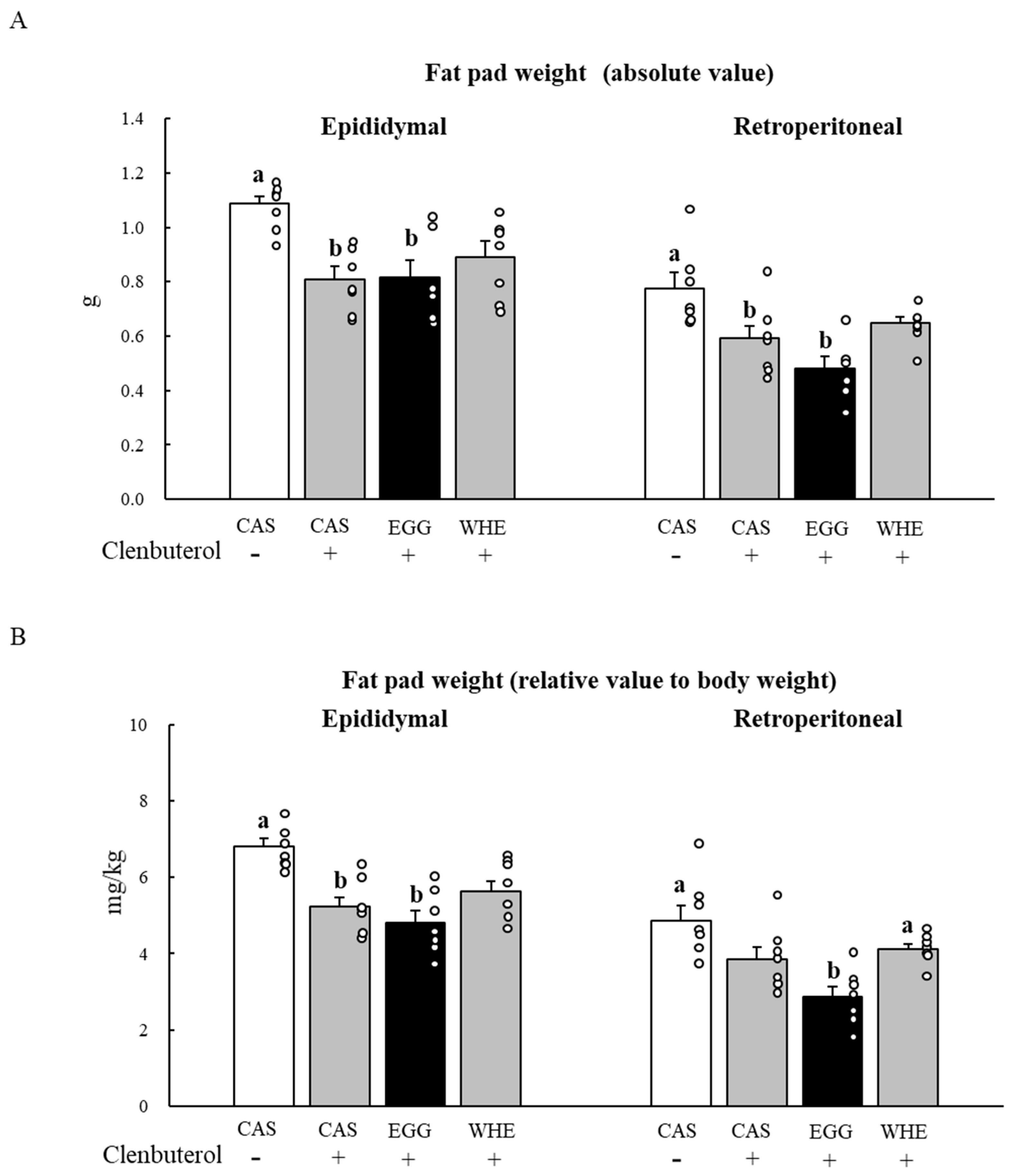
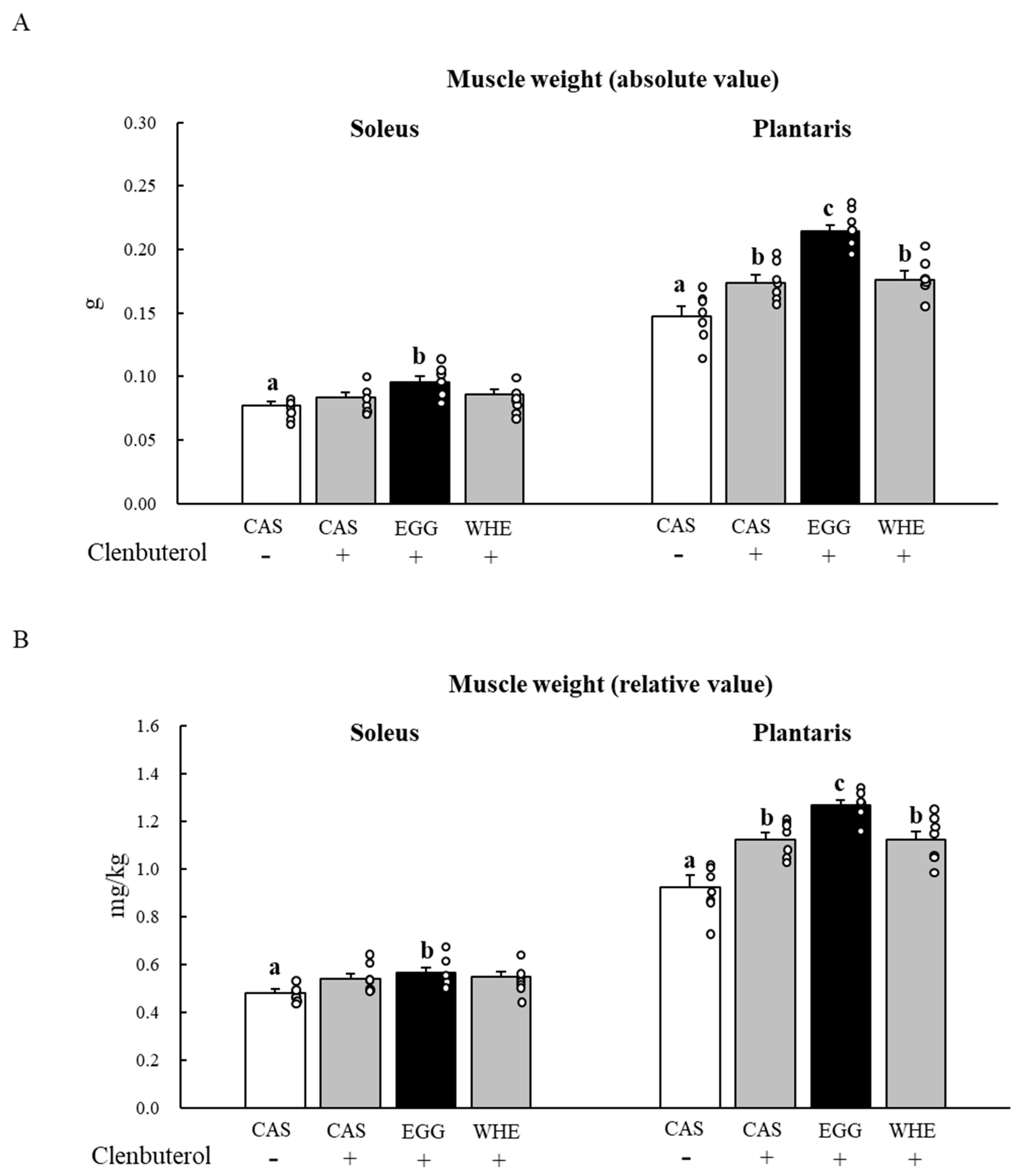
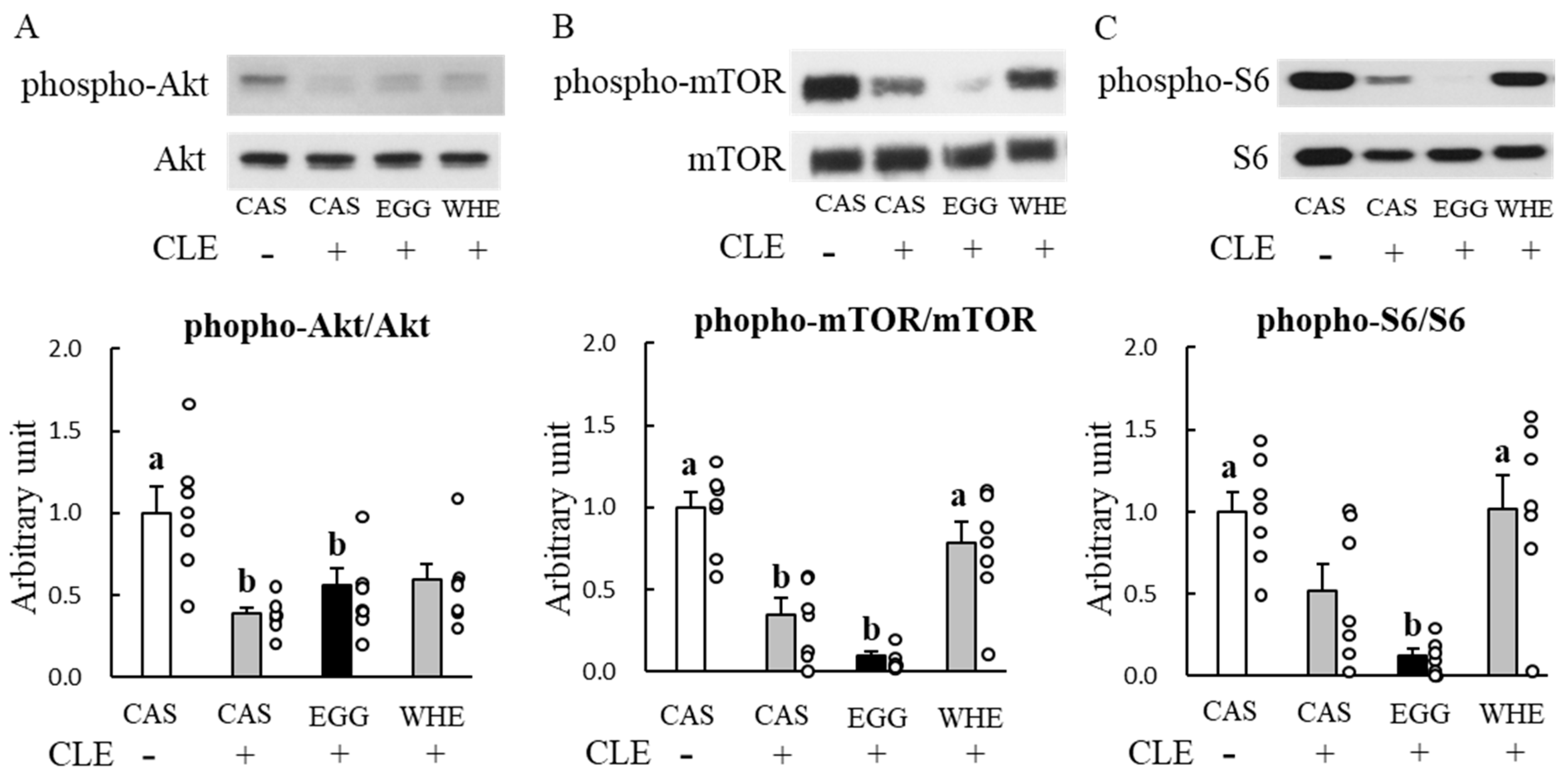
3.5. Experiment V
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Atherton, P.J.; Smith, K. Muscle protein synthesis in response to nutrition and exercise. J. Physiol. 2012, 590, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Jäger, R.; Kerksick, C.M.; Campbell, B.I.; Cribb, P.J.; Wells, S.D.; Skwiat, T.M.; Purpura, M.; Ziegenfuss, T.N.; Ferrando, A.A.; Arent, S.M.; et al. International Society of Sports Nutrition Position Stand: Protein and exercise. J. Int. Soc. Sports Nutr. 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Putra, C.; Konow, N.; Gage, M.; York, C.G.; Mangano, K.M. Protein source and muscle health in older adults: A literature review. Nutrients 2021, 13, 743. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, J.R.; Falvo, M.J. Protein—Which is best? J. Sports Sci. Med. 2004, 3, 118–130. [Google Scholar] [PubMed]
- Mathai, J.K.; Liu, Y.; Stein, H.H. Values for digestible indispensable amino acid scores (DIAAS) for some dairy and plant proteins may better describe protein quality than values calculated using the concept for protein digestibility-corrected amino acid scores (PDCAAS). Br. J. Nutr. 2017, 117, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.E.; Moore, D.R.; Kujbida, G.W.; Tamopolsky, M.A.; Phillips, S.M. Ingestion of whey hydrolysate, casein, or soy protein isolate: Effects on mixed muscle protein synthesis at rest and following resistance exercise in young man. J. Appl. Physiol. 2009, 107, 987–992. [Google Scholar] [CrossRef]
- Yang, Y.; Churchward-Venne, T.A.; Burd, N.A.; Breen, L.; Tarnopolsky, M.A.; Phillips, S.M. Myofibrillar protein synthesis following ingestion of soy protein isolate at rest and after resistance exercise in elderly man. Nutr. Metab. 2012, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Volek, J.S.; Volk, B.M.; Gómez, A.L.; Kunces, L.J.; Kupchak, B.R.; Freidenreich, D.J.; Aristizabal, J.C.; Saenz, C.; Dunn-Lewis, C.; Ballard, K.D.; et al. Whey protein supplementation during resistance training augments lean body mass. J. Am. Coll. Nutr. 2013, 32, 122–135. [Google Scholar] [CrossRef]
- Ham, D.J.; Caldow, M.K.; Lynch, G.S.; Koopman, R. Leucine as a treatment for muscle wasting: A critical review. Clin. Nutr. 2014, 33, 937–945. [Google Scholar] [CrossRef]
- Crozier, S.J.; Kimball, S.R.; Emmert, S.W.; Anthony, J.C.; Jefferson, L.S. Oral leucine administration stimulates protein synthesis in rat skeletal muscle. J. Nutr. 2005, 135, 376–382. [Google Scholar] [CrossRef]
- Wilkinson, D.J.; Hossain, T.; Hill, D.S.; Phillips, B.E.; Crossland, H.; Williams, J.; Loughna, P.; Churchward-Venne, T.A.; Breen, L.; Phillips, S.M.; et al. Effects of leucine and its metabolite β-hydroxy-β-methylbutyrate on human skeletal muscle protein metabolism. J. Physiol. 2013, 591, 2911–2923. [Google Scholar] [CrossRef] [PubMed]
- Churchward-Venne, T.A.; Burd, N.A.; Mitchell, C.J.; West, D.W.D.; Philp, A.; Marcotte, G.R.; Baker, S.K.; Baar, K.; Phillips, S.M. Supplementation of a suboptimal protein dose with leucine or essential amino acids: Effects on myofibrillar protein synthesis at rest and following resistance exercise in men. J. Physiol. 2012, 590, 2751–2765. [Google Scholar] [CrossRef]
- Devries, M.C.; McGlory, C.; Bolster, D.R.; Kamil, A.; Rahn, M.; Harkness, L.; Baker, S.K.; Phillips, S.M. Leucine, not total protein, content of a supplement is the primary determinant of muscle protein anabolic responses in healthy older women. J. Nutr. 2018, 148, 1088–1095. [Google Scholar] [CrossRef] [PubMed]
- Gorissen, S.H.M.; Crombag, J.J.R.; Senden, J.M.G.; Waterval, W.A.H.; Bierau, J.; Verdjik, L.B.; van Loon, L.J.C. Protein content and amino acid composition of commercially available plant-based protein isolates. Amino Acid 2018, 50, 1685–1695. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, R.; Kurihara, H.; Nishijima, N.; Oda, Y.; Handa, A. Egg White hydrolysate retains the nutritional value of proteins and is quickly absorbed in Rats. Sci. World J. 2019. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Sawada, A.; Numao, S.; Suzuki, M. chronic effect of light resistance exercise after ingestion of a high-protein snack on increase of skeletal muscle mass and strength in young adults. J. Nutr. Sci. Vitaminol. 2011, 57, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Ochiai, M.; Kuroda, T.; Matsuo, T. Increased muscular triglyceride content and hyperglycemia in Goto-Kakizaki rat are decreased by egg white hydrolysate. Int. J. Food Sci. Nutr. 2014, 65, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Ochiai, M.; Matsuo, T. Effect of egg white and its hydrolysate on stearoyl-CoA desaturase index and fat accumulation in rat tissues. Int. J. Food Sci. Nutr. 2014, 65, 948–952. [Google Scholar] [CrossRef] [PubMed]
- Ochiai, M.; Misaki, K.; Takeuchi, T.; Narumi, R.; Azuma, Y.; Matsuo, T. Egg White hydrolysate can be a low-allergenic food material to suppress ectopic fat accumulation in rats fed an equicaloric diet. J. Nutr. Sci. Vitaminol. 2017, 63, 111–119. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Matsuoka, R.; Takahashi, Y.; Kimura, M.; Masuda, Y.; Kunou, M. Heating has no effect on the net protein utilization from egg whites in rats. Sci. World J. 2017. [Google Scholar] [CrossRef]
- Parmer, J.P.; Benson, J.W.; Walter, R.M.; Ensinck, J.W. Arginine-stimulated acute phase of insulin and glucagon secretion in diabetic subjects. J. Clin. Investig. 1976, 58, 565–570. [Google Scholar]
- Alba-Roth, J.; Müller, O.A.; Schopohl, J.; von Werder, K. Arginine stimulates growth hormone secretion by suppressing endogenous somatostatin secretion. J. Clin. Endocrinol. Metab. 1988, 67, 1186–1189. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.J.; Haynes, T.E.; Kohli, R.; Hu, J.; Shi, W.; Spencer, T.E.; Carroll, R.J.; Meininger, C.J.; Wu, G. Dietary L-arginine supplementation reduces fat mass in Zucker diabetic fatty rats. J. Nutr. 2005, 135, 714–721. [Google Scholar] [CrossRef] [PubMed]
- Kitaura, T. How β2-adrenergic agonists induce skeletal muscle hypertrophy? J. Phys. Fitness Sports Med. 2013, 2, 423–428. [Google Scholar] [CrossRef][Green Version]
- Miranda, J.M.; Anton, X.; Redondo-Valbuena, C.; Roca-Saavedra, P.; Rodriguez, J.A.; Lamas, A.; Franco, C.M.; Cepeda, A. Egg and egg-derived foods: Effects on human health and use as functional foods. Nutrients 2015, 7, 706–729. [Google Scholar] [CrossRef]
- Tapiero, H.; Mathé, G.; Couvreur, P.; Tew, K.D. I. Arginine. Biomed. Pharmacother. 2002, 56, 439–445. [Google Scholar] [CrossRef]
- Milner, J.A.; Wakeling, A.E.; Visek, W.J. Effect of arginine deficiency on growth and intermediary metabolism in rats. J. Nutr. 1974, 104, 1681–1689. [Google Scholar] [CrossRef] [PubMed]
- Suzumura, S.; Tujioka, K.; Yamada, T.; Yokogoshi, H.; Akizuki, S.; Hishida, Y.; Tsutsui, K.; Hayase, K. Comparison of the Effects of Ornithine and Arginine on the Brain Protein Synthesis Rate in Young Rats. J. Nutr. Sci. Vitam. 2015, 61, 417–421. [Google Scholar] [CrossRef] [PubMed]
- Maltin, C.A.; Delday, M.L.; Hay, S.M.; Smith, F.G.; Lobley, G.E.; Reeds, P.J. The effect of the anabolic agent, clenbuterol, on overloaded rat skeletal muscle. Biosci. Rep. 1987, 7, 143–149. [Google Scholar] [CrossRef]
- Maltin, C.A.; Hay, S.M.; McMillan, D.N.; Delday, M.I. Tissue specific responses to clenbuterol; temporal changes in protein metabolism of striated muscle and visceral tissues from rats. Growth Regul. 1992, 2, 161–166. [Google Scholar] [PubMed]
- Sneddon, A.A.; Delday, M.I.; Steven, J.; Maltin, C.A. Elevated IGF-II mRNA and phosphorylation of 4E-BP1 and p70S6k in muscle showing clenbuterol-induced anabolism. Am. J. Physiol. Endocrinol. Metab. 2001, E676–E682. [Google Scholar] [CrossRef] [PubMed]
- Kline, W.O.; Panaro, F.J.; Yang, H.; Bodine, S.C. Rapamycin inhibits the growth and muscle-sparing effects of clenbuterol. J. Appl. Physiol. 2007, 102, 740–747. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Yoshikawa, Y.; Tsujimoto, H.; Kitaura, T.; Muraoka, I. Clenbuterol accelerates recovery after immobilization-induced atrophy of rat hindlimb muscle. Acta Histochem. 2020, 122, 151453. [Google Scholar] [CrossRef] [PubMed]
- Maltin, C.A.; Delday, M.I.; Hay, S.M.; Innes, G.M.; Williams, P.E. Effects of bovine pituitary growth hormone alone or in combination with the beta-agonist clenbuterol on muscle growth and composition in veal calves. Br. J. Nutr. 1990, 63, 535–545. [Google Scholar] [CrossRef] [PubMed]
- Berdeaux, R.; Stewart, R. cAMP signaling in skeletal muscle adaptation: Hypertrophy, metabolism, and regeneration. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E1–E17. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.R.; Robinson, M.J.; Fry, J.L.; Tang, J.E.; Glover, E.I.; Wilkinson, S.B.; Prior, T.; Tarnopolsky, M.A.; Phillips, S.M. Ingested protein dose response of muscle and albumin protein synthesis after resistance exercise in young men. Am. J. Clin. Nutr. 2009, 89, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Son, M.; Meram, C.D.; Wu, J. Mechanism and potential of egg consumption and egg bioactive components on type-2 diabetes. Nutrients 2019, 11, 357. [Google Scholar] [CrossRef]
| CAS | EGG | WHE | |
|---|---|---|---|
| In each protein source (g/100g) | |||
| Protein | 91 | 81 | 78 |
| Fat | 1 | 1 | 6 |
| In each diet (Diet 1) (g/100g) | |||
| Casein protein | 22 | - | - |
| Egg white protein | - | 25 | - |
| Whey protein | - | - | 26 |
| Sucrose | 5 | 5 | 5 |
| α-Corn starch | 46 | 47 | 47 |
| Corn oil | 11 | 7 | 6 |
| Vitamin mix (AIN-76) | 2 | 2 | 2 |
| Mineral mix (AIN-76) | 6 | 6 | 6 |
| Cellulose | 8 | 8 | 8 |
| Final protein content | 20 | 20 | 20 |
| Final fat content | 12 | 12 | 12 |
| Original (Diet 1) | Arginine-modified (Diet 2) | |||||
|---|---|---|---|---|---|---|
| (mg/100g) | CAS | EGG | WHE | CAS | EGG | WHE |
| Arginine | 610 | 1070 | 438 | 1063 | 1063 | 1063 |
| Lysine | 1400 | 1290 | 1790 | 1391 | 1282 | 1749 |
| Histidine | 457 | 385 | 332 | 454 | 383 | 330 |
| Phenylalanine | 935 | 1130 | 605 | 929 | 1123 | 601 |
| Tyrosine | 924 | 722 | 596 | 918 | 717 | 592 |
| Leucine | 1760 | 1600 | 2210 | 1749 | 1590 | 2196 |
| Isoleucine | 611 | 643 | 957 | 607 | 639 | 951 |
| Methionine | 554 | 750 | 452 | 551 | 745 | 449 |
| Valine | 788 | 844 | 877 | 783 | 839 | 871 |
| Alanine | 616 | 1240 | 1120 | 612 | 1232 | 1113 |
| Glycine | 357 | 730 | 383 | 355 | 725 | 381 |
| Proline | 2080 | 714 | 1280 | 2238 | 1338 | 1272 |
| Glutamic acid | 4370 | 2720 | 3770 | 4343 | 2703 | 3746 |
| Serine | 1120 | 1500 | 1160 | 1113 | 1491 | 1153 |
| Threonine | 764 | 907 | 1380 | 759 | 901 | 1371 |
| Aspartic acid | 1410 | 2150 | 2290 | 1401 | 2136 | 2276 |
| Tryptophan | 228 | 296 | 367 | 227 | 294 | 365 |
| Cysteine | 87 | 598 | 508 | 86 | 594 | 505 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koshinaka, K.; Honda, A.; Iizumi, R.; Miyazawa, Y.; Kawanaka, K.; Sato, A. Egg White Protein Feeding Facilitates Skeletal Muscle Gain in Young Rats with/without Clenbuterol Treatment. Nutrients 2021, 13, 2042. https://doi.org/10.3390/nu13062042
Koshinaka K, Honda A, Iizumi R, Miyazawa Y, Kawanaka K, Sato A. Egg White Protein Feeding Facilitates Skeletal Muscle Gain in Young Rats with/without Clenbuterol Treatment. Nutrients. 2021; 13(6):2042. https://doi.org/10.3390/nu13062042
Chicago/Turabian StyleKoshinaka, Keiichi, Asuka Honda, Rihei Iizumi, Yuto Miyazawa, Kentaro Kawanaka, and Akiko Sato. 2021. "Egg White Protein Feeding Facilitates Skeletal Muscle Gain in Young Rats with/without Clenbuterol Treatment" Nutrients 13, no. 6: 2042. https://doi.org/10.3390/nu13062042
APA StyleKoshinaka, K., Honda, A., Iizumi, R., Miyazawa, Y., Kawanaka, K., & Sato, A. (2021). Egg White Protein Feeding Facilitates Skeletal Muscle Gain in Young Rats with/without Clenbuterol Treatment. Nutrients, 13(6), 2042. https://doi.org/10.3390/nu13062042





