The Probiotic Strain H. alvei HA4597® Improves Weight Loss in Overweight Subjects under Moderate Hypocaloric Diet: A Proof-of-Concept, Multicenter Randomized, Double-Blind Placebo-Controlled Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Study Design and Intervention
2.3. Measurements and Endpoints
2.4. Statistical Analysis
- Mann-Whitney-U test for comparison of independent groups (u),
- Paired Wilcoxon test for the pre-post comparisons within groups (wil),
- Exact Fisher’s test for the comparison of frequencies for independent groups (exF).
3. Results
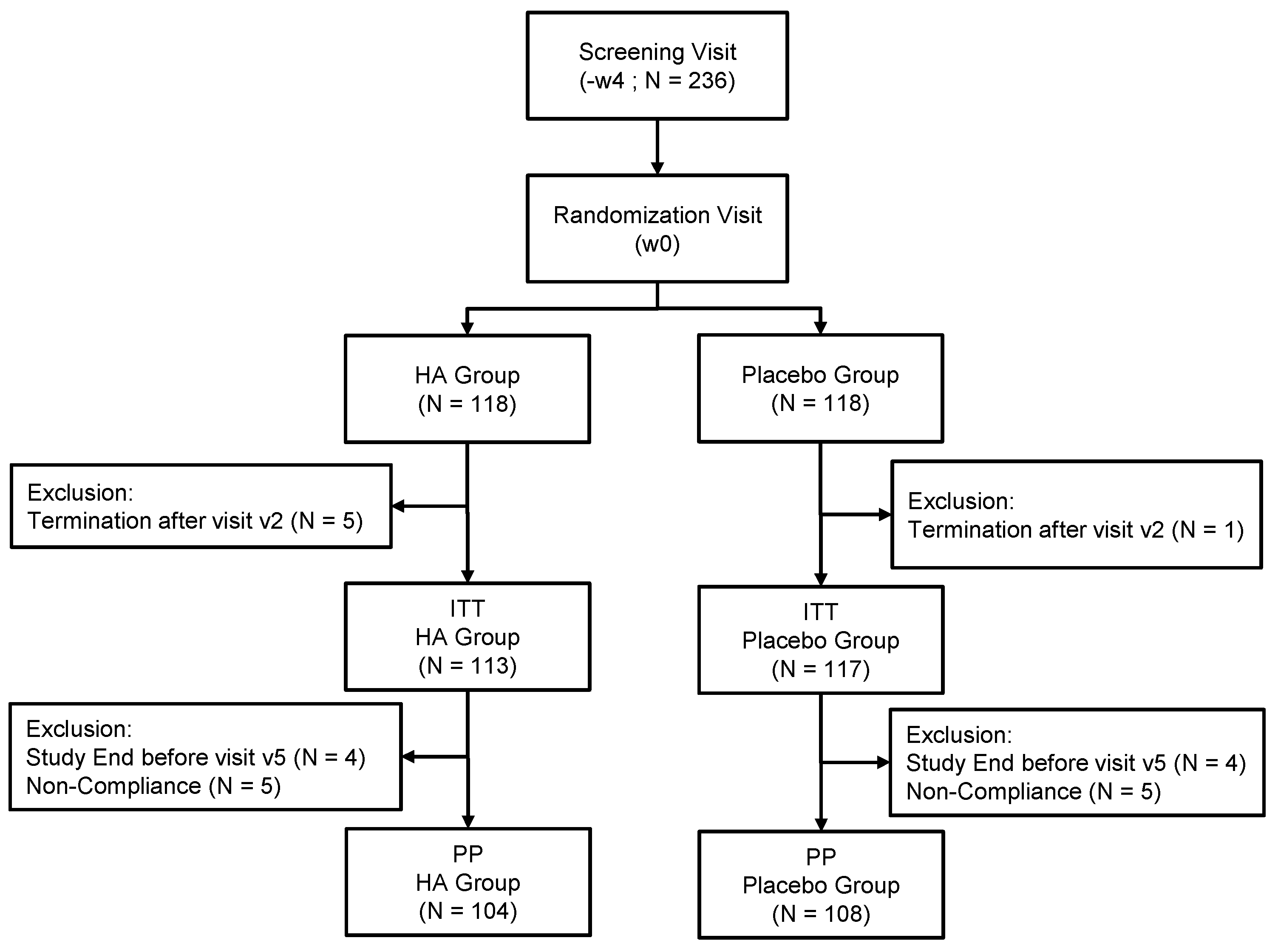
3.1. Subjects
3.2. Effects of the Probiotic strain HA4597®
3.2.1. Primary Endpoint
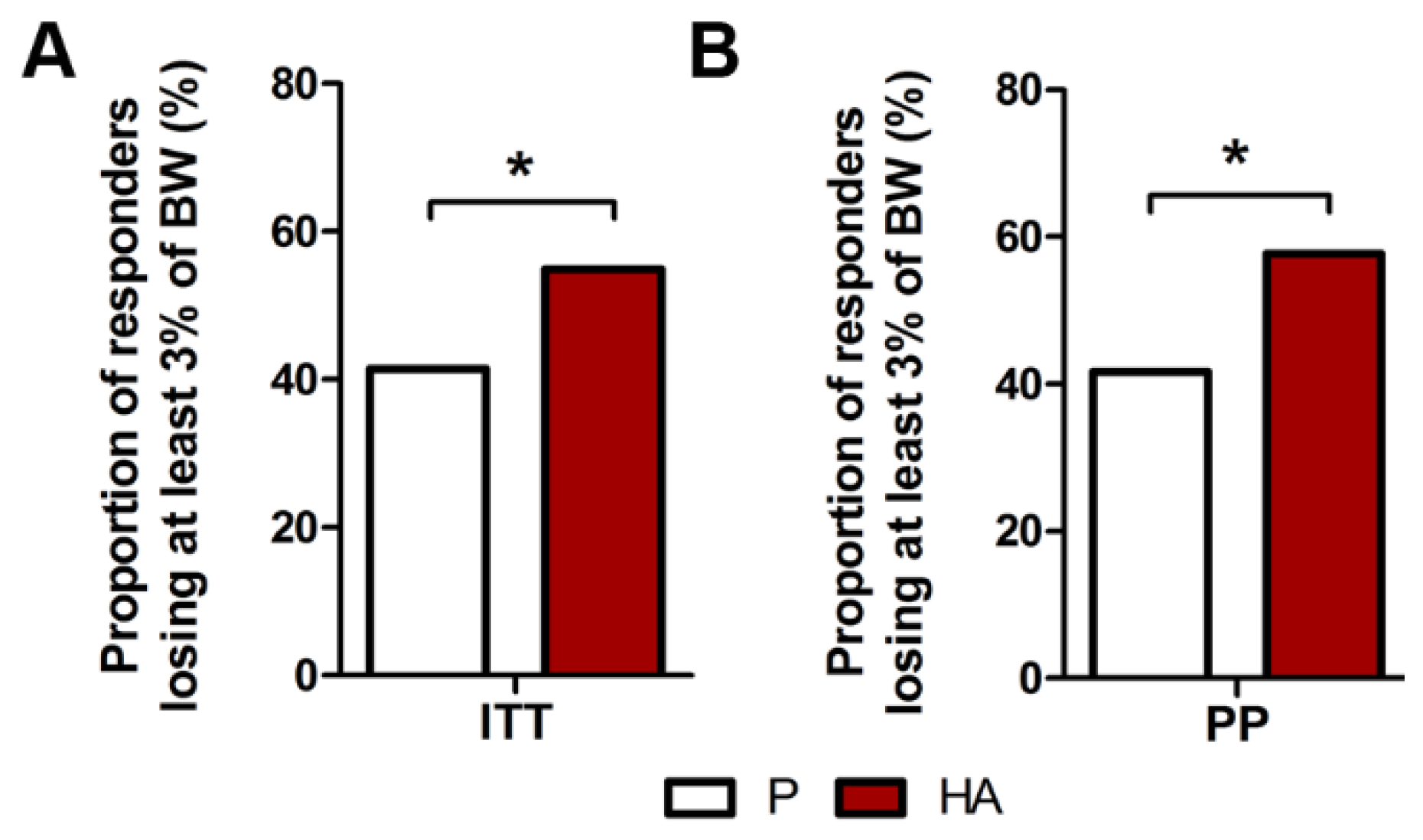
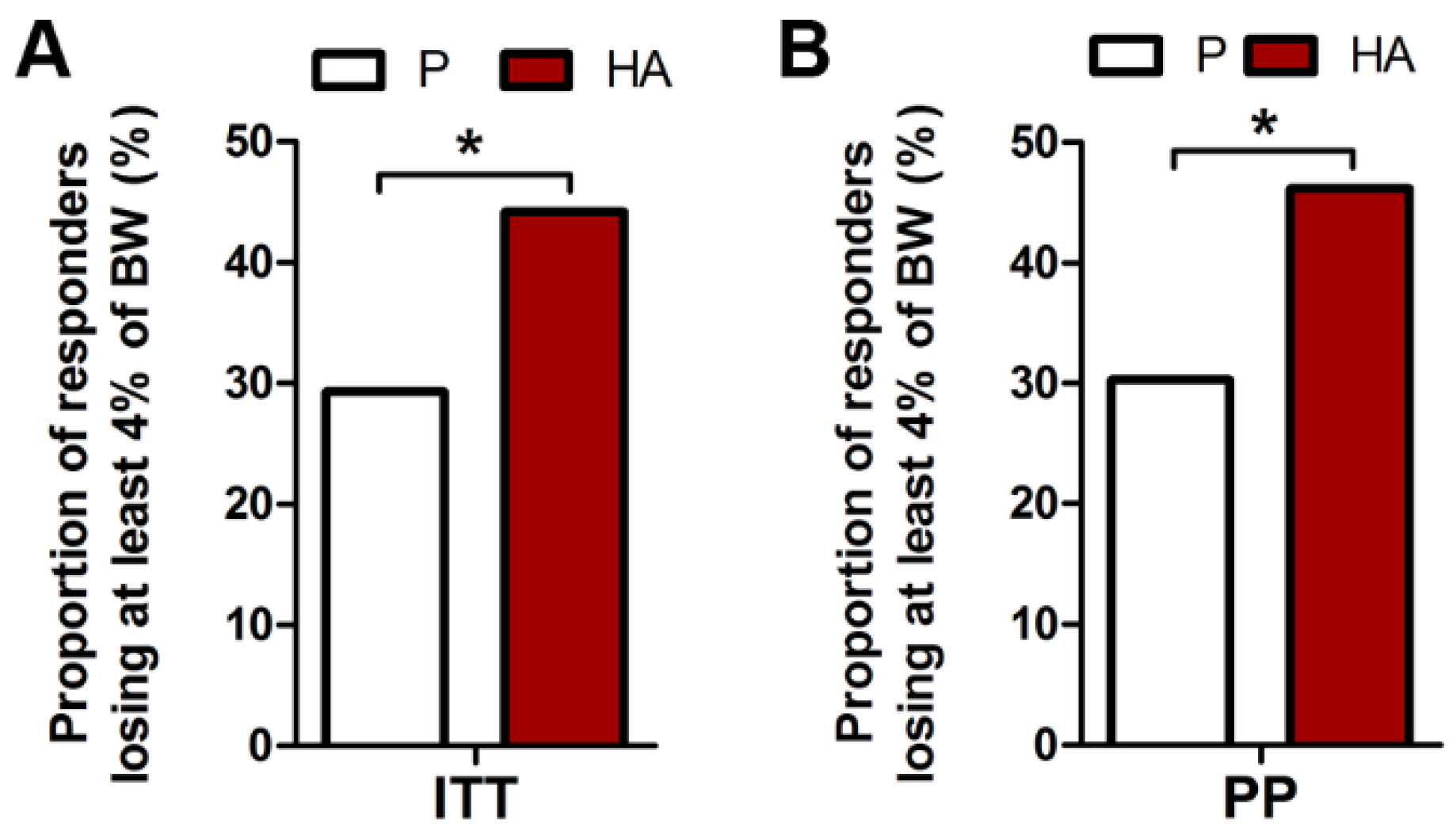
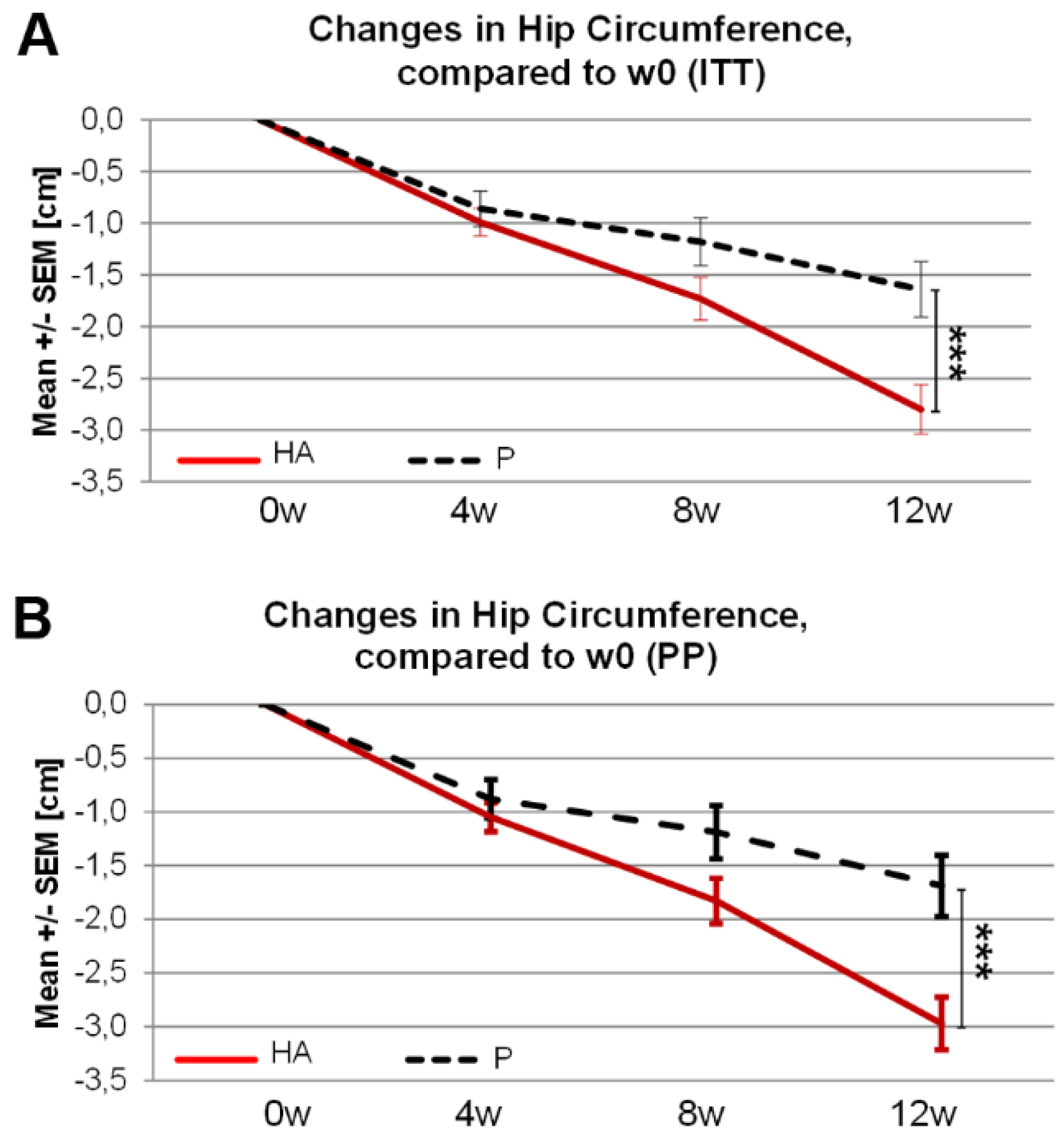
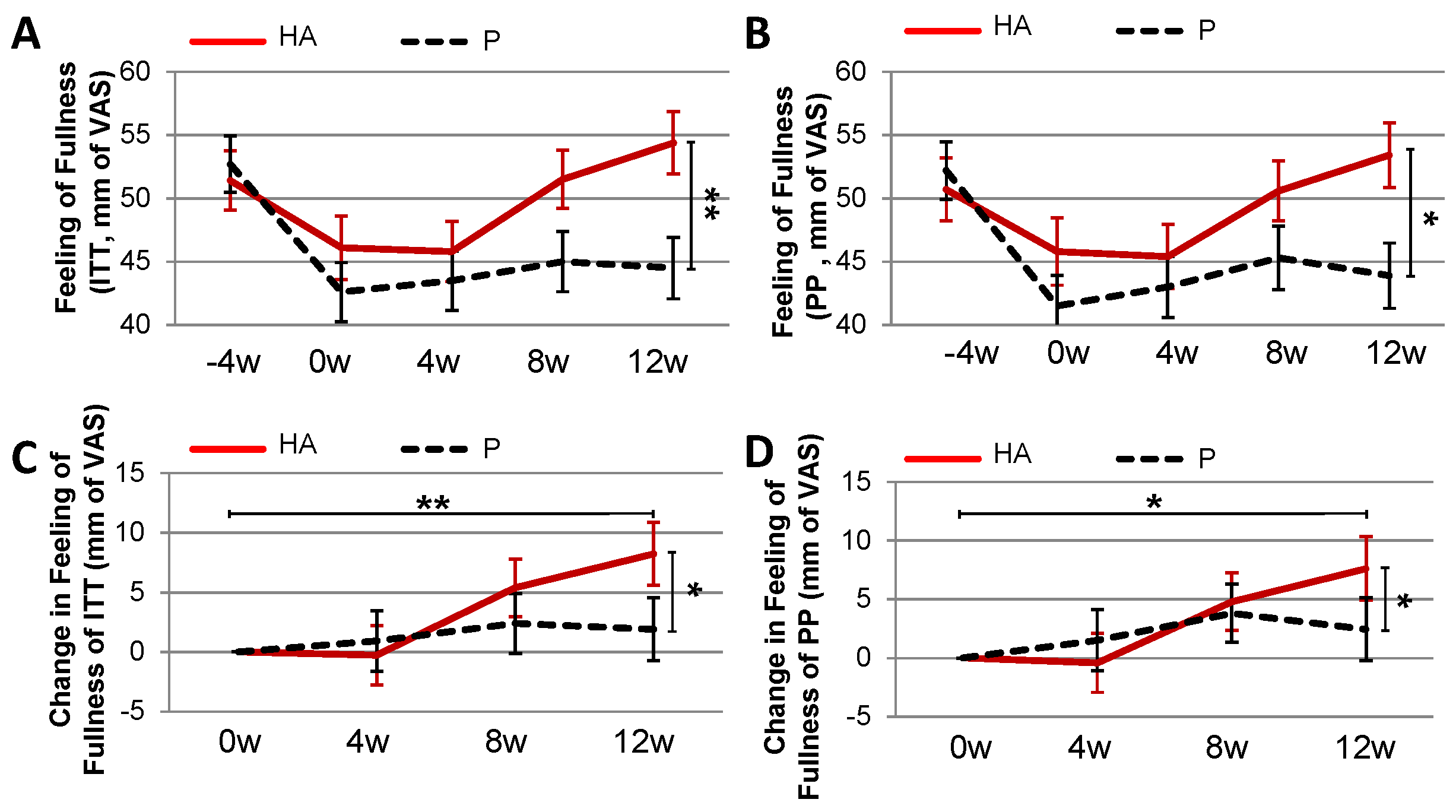
3.2.2. Secondary and Post-Hoc Endpoints
3.2.3. Clinical and Biological Safety Evaluation
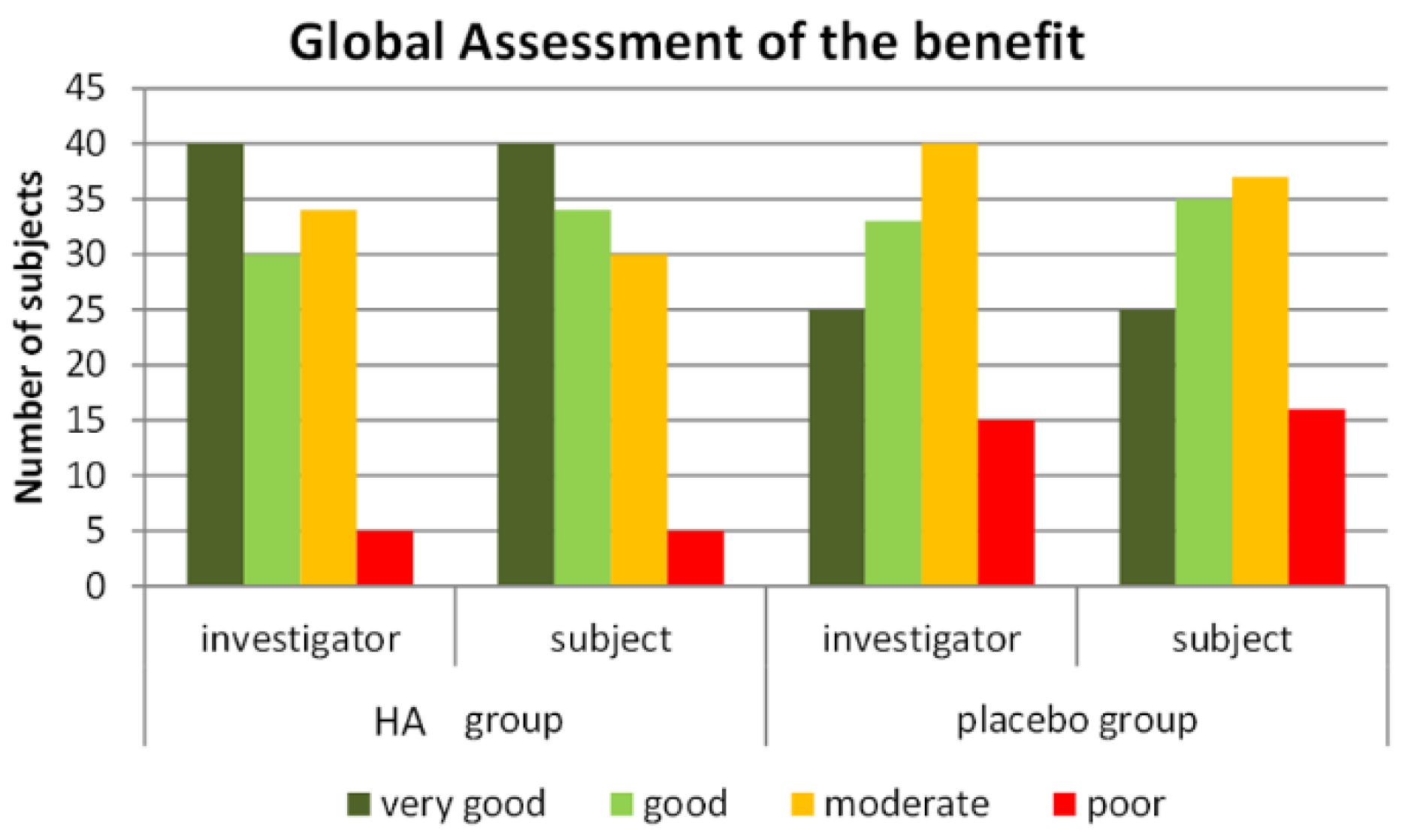
3.2.4. Global Evaluation of Efficacy and Tolerability and Adverse Events (AE)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- GBD 2015 Obesity Collaborators; Afshin, A.; Forouzanfar, M.H.; Reitsma, M.B.; Sur, P.; Estep, K.; Lee, A.; Marczak, L.; Mokdad, A.H.; Moradi-Lakeh, M.; et al. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef]
- Jensen, M.D.; Ryan, D.H.; Apovian, C.M.; Ard, J.D.; Comuzzie, A.G.; Donato, K.A.; Hu, F.B.; Hubbard, V.S.; Jakicic, J.M.; Kushner, R.F.; et al. 2013 AHA/ACC/TOS Guideline for the Management of Overweight and Obesity in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. Circulation 2014, 129, S102–S138. [Google Scholar] [CrossRef] [PubMed]
- Romo-Nava, F.; Blom, T.J.; Guerdjikova, A.; Winham, S.J.; Cuellar-Barboza, A.B.; Nunez, N.A.; Singh, B.; Biernacka, J.M.; Frye, M.A.; McElroy, S.L. Evening Chronotype, Disordered Eating Behavior, and Poor Dietary Habits in Bipolar Disorder. Acta Psychiatr. Scand. 2020, 142, 58–65. [Google Scholar] [CrossRef]
- Appolinario, J.C.; Nardi, A.E.; McElroy, S.L. Investigational Drugs for the Treatment of Binge Eating Disorder (BED): An Update. Expert Opin. Investig. Drugs 2019, 28, 1081–1094. [Google Scholar] [CrossRef]
- Cani, P.D.; Van Hul, M.; Lefort, C.; Depommier, C.; Rastelli, M.; Everard, A. Microbial Regulation of Organismal Energy Homeostasis. Nat. Metab. 2019, 1, 34–46. [Google Scholar] [CrossRef]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An Obesity-Associated Gut Microbiome with Increased Capacity for Energy Harvest. Nature 2006, 444, 1027–1031. [Google Scholar] [CrossRef]
- Fetissov, S.O. Role of the Gut Microbiota in Host Appetite Control: Bacterial Growth to Animal Feeding Behaviour. Nat. Rev. Endocrinol. 2017, 13, 11–25. [Google Scholar] [CrossRef] [PubMed]
- Carbone, E.A.; D’Amato, P.; Vicchio, G.; De Fazio, P.; Segura-Garcia, C. A Systematic Review on the Role of Microbiota in the Pathogenesis and Treatment of Eating Disorders. Eur. Psychiatry 2021, 64, e2. [Google Scholar] [CrossRef] [PubMed]
- Breton, J.; Tennoune, N.; Lucas, N.; Francois, M.; Legrand, R.; Jacquemot, J.; Goichon, A.; Guérin, C.; Peltier, J.; Pestel-Caron, M.; et al. Gut Commensal, E. Coli Proteins Activate Host Satiety Pathways Following Nutrient-Induced Bacterial Growth. Cell Metab. 2016, 23, 324–334. [Google Scholar] [CrossRef]
- Muscogiuri, G.; Cantone, E.; Cassarano, S.; Tuccinardi, D.; Barrea, L.; Savastano, S.; Colao, A. Gut Microbiota: A New Path to Treat Obesity. Int. J. Obes. Suppl. 2019, 9, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Guazzelli Marques, C.; de Piano Ganen, A.; Zaccaro de Barros, A.; Thomatieli Dos Santos, R.V.; Dos Santos Quaresma, M.V.L. Weight Loss Probiotic Supplementation Effect in Overweight and Obesity Subjects: A Review. Clin. Nutr. 2020, 39, 694–704. [Google Scholar] [CrossRef]
- Suzumura, E.A.; Bersch-Ferreira, Â.C.; Torreglosa, C.R.; da Silva, J.T.; Coqueiro, A.Y.; Kuntz, M.G.F.; Chrispim, P.P.; Weber, B.; Cavalcanti, A.B. Effects of Oral Supplementation with Probiotics or Synbiotics in Overweight and Obese Adults: A Systematic Review and Meta-Analyses of Randomized Trials. Nutr. Rev. 2019, 77, 430–450. [Google Scholar] [CrossRef]
- Koutnikova, H.; Genser, B.; Monteiro-Sepulveda, M.; Faurie, J.-M.; Rizkalla, S.; Schrezenmeir, J.; Clément, K. Impact of Bacterial Probiotics on Obesity, Diabetes and Non-Alcoholic Fatty Liver Disease Related Variables: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. BMJ Open 2019, 9, e017995. [Google Scholar] [CrossRef]
- Tennoune, N.; Chan, P.; Breton, J.; Legrand, R.; Chabane, Y.N.; Akkermann, K.; Järv, A.; Ouelaa, W.; Takagi, K.; Ghouzali, I.; et al. Bacterial ClpB Heat-Shock Protein, an Antigen-Mimetic of the Anorexigenic Peptide α-MSH, at the Origin of Eating Disorders. Transl. Psychiatry 2014, 4, e458. [Google Scholar] [CrossRef] [PubMed]
- Legrand, R.; Lucas, N.; Dominique, M.; Azhar, S.; Deroissart, C.; Le Solliec, M.-A.; Rondeaux, J.; Nobis, S.; Guérin, C.; Léon, F.; et al. Commensal Hafnia Alvei Strain Reduces Food Intake and Fat Mass in Obese Mice-a New Potential Probiotic for Appetite and Body Weight Management. Int. J. Obes. 2020, 44, 1041–1051. [Google Scholar] [CrossRef] [PubMed]
- Lucas, N.; Legrand, R.; Deroissart, C.; Dominique, M.; Azhar, S.; Le Solliec, M.-A.; Léon, F.; do Rego, J.-C.; Déchelotte, P.; Fetissov, S.O.; et al. Hafnia Alvei HA4597 Strain Reduces Food Intake and Body Weight Gain and Improves Body Composition, Glucose, and Lipid Metabolism in a Mouse Model of Hyperphagic Obesity. Microorganisms 2019, 8, 35. [Google Scholar] [CrossRef]
- Dominique, M.; Breton, J.; Guérin, C.; Bole-Feysot, C.; Lambert, G.; Déchelotte, P.; Fetissov, S. Effects of Macronutrients on the In Vitro Production of ClpB, a Bacterial Mimetic Protein of α-MSH and Its Possible Role in Satiety Signaling. Nutrients 2019, 11, 2115. [Google Scholar] [CrossRef] [PubMed]
- Chong, P.-W.; Beah, Z.-M.; Grube, B.; Riede, L. IQP-GC-101 Reduces Body Weight and Body Fat Mass: A Randomized, Double-Blind, Placebo-Controlled Study. Phytotherapy Res. 2014, 28, 1520–1526. [Google Scholar] [CrossRef] [PubMed]
- Grube, B.; Chong, P.-W.; Lau, K.-Z.; Orzechowski, H.-D. A Natural Fiber Complex Reduces Body Weight in the Overweight and Obese: A Double-Blind, Randomized, Placebo-Controlled Study. Obesity 2013, 21, 58–64. [Google Scholar] [CrossRef]
- Svedlund, J.; Sjödin, I.; Dotevall, G. GSRS—A Clinical Rating Scale for Gastrointestinal Symptoms in Patients with Irritable Bowel Syndrome and Peptic Ulcer Disease. Dig. Dis. Sci. 1988, 33, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Bäckhed, F.; Manchester, J.K.; Semenkovich, C.F.; Gordon, J.I. Mechanisms Underlying the Resistance to Diet-Induced Obesity in Germ-Free Mice. Proc. Natl. Acad. Sci. USA 2007, 104, 979–984. [Google Scholar] [CrossRef]
- Boulangé, C.L.; Neves, A.L.; Chilloux, J.; Nicholson, J.K.; Dumas, M.-E. Impact of the Gut Microbiota on Inflammation, Obesity, and Metabolic Disease. Genome Med. 2016, 8, 42. [Google Scholar] [CrossRef]
- Frost, G.; Sleeth, M.L.; Sahuri-Arisoylu, M.; Lizarbe, B.; Cerdan, S.; Brody, L.; Anastasovska, J.; Ghourab, S.; Hankir, M.; Zhang, S.; et al. The Short-Chain Fatty Acid Acetate Reduces Appetite via a Central Homeostatic Mechanism. Nat. Commun. 2014, 5, 3611. [Google Scholar] [CrossRef]
- Kerac, M.; Bunn, J.; Seal, A.; Thindwa, M.; Tomkins, A.; Sadler, K.; Bahwere, P.; Collins, S. Probiotics and Prebiotics for Severe Acute Malnutrition (PRONUT Study): A Double-Blind Efficacy Randomised Controlled Trial in Malawi. Lancet 2009, 374, 136–144. [Google Scholar] [CrossRef]
- Korpela, K.; Flint, H.J.; Johnstone, A.M.; Lappi, J.; Poutanen, K.; Dewulf, E.; Delzenne, N.; de Vos, W.M.; Salonen, A. Gut Microbiota Signatures Predict Host and Microbiota Responses to Dietary Interventions in Obese Individuals. PLoS ONE 2014, 9, e90702. [Google Scholar] [CrossRef] [PubMed]
- Salonen, A.; Lahti, L.; Salojärvi, J.; Holtrop, G.; Korpela, K.; Duncan, S.H.; Date, P.; Farquharson, F.; Johnstone, A.M.; Lobley, G.E.; et al. Impact of Diet and Individual Variation on Intestinal Microbiota Composition and Fermentation Products in Obese Men. ISME J. 2014, 8, 2218–2230. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; de Vos, W.M. Next-Generation Beneficial Microbes: The Case of Akkermansia Muciniphila. Front. Microbiol. 2017, 8, 1765. [Google Scholar] [CrossRef] [PubMed]
- Sanders, M.E. Probiotics: Definition, Sources, Selection, and Uses. Clin. Infect. Dis. 2008, 46, S58–S61. [Google Scholar] [CrossRef] [PubMed]
- Cerdó, T.; García-Santos, J.A.; Bermúdez, M.G.; Campoy, C. The Role of Probiotics and Prebiotics in the Prevention and Treatment of Obesity. Nutrients 2019, 11, 635. [Google Scholar] [CrossRef]
- Wiciński, M.; Gębalski, J.; Gołębiewski, J.; Malinowski, B. Probiotics for the Treatment of Overweight and Obesity in Humans—A Review of Clinical Trials. Microorganisms 2020, 8, 1148. [Google Scholar] [CrossRef] [PubMed]
- Rucker, D.; Padwal, R.; Li, S.K.; Curioni, C.; Lau, D.C.W. Long Term Pharmacotherapy for Obesity and Overweight: Updated Meta-Analysis. BMJ 2007, 335, 1194–1199. [Google Scholar] [CrossRef]
- Anderson, E.J.P.; Çakir, I.; Carrington, S.J.; Cone, R.D.; Ghamari-Langroudi, M.; Gillyard, T.; Gimenez, L.E.; Litt, M.J. 60 YEARS OF POMC: Regulation of Feeding and Energy Homeostasis by α-MSH. J. Mol. Endocrinol. 2016, 56, T157–T174. [Google Scholar] [CrossRef] [PubMed]
- Panaro, B.L.; Tough, I.R.; Engelstoft, M.S.; Matthews, R.T.; Digby, G.J.; Møller, C.L.; Svendsen, B.; Gribble, F.; Reimann, F.; Holst, J.J.; et al. The Melanocortin-4 Receptor Is Expressed in Enteroendocrine L Cells and Regulates the Release of Peptide YY and Glucagon-like Peptide 1 in Vivo. Cell Metab. 2014, 20, 1018–1029. [Google Scholar] [CrossRef]
- Kühnen, P.; Clément, K.; Wiegand, S.; Blankenstein, O.; Gottesdiener, K.; Martini, L.L.; Mai, K.; Blume-Peytavi, U.; Grüters, A.; Krude, H. Proopiomelanocortin Deficiency Treated with a Melanocortin-4 Receptor Agonist. N. Engl. J. Med. 2016, 375, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Arnoriaga-Rodríguez, M.; Mayneris-Perxachs, J.; Burokas, A.; Pérez-Brocal, V.; Moya, A.; Portero-Otin, M.; Ricart, W.; Maldonado, R.; Fernández-Real, J.-M. Gut Bacterial ClpB-like Gene Function Is Associated with Decreased Body Weight and a Characteristic Microbiota Profile. Microbiome 2020, 8, 59. [Google Scholar] [CrossRef]
- Million, M.; Angelakis, E.; Maraninchi, M.; Henry, M.; Giorgi, R.; Valero, R.; Vialettes, B.; Raoult, D. Correlation between Body Mass Index and Gut Concentrations of Lactobacillus Reuteri, Bifidobacterium Animalis, Methanobrevibacter Smithii and Escherichia Coli. Int. J. Obes. 2013, 37, 1460–1466. [Google Scholar] [CrossRef] [PubMed]
- Dominique, M.; Legrand, R.; Galmiche, M.; Azhar, S.; Deroissart, C.; Guérin, C.; do Rego, J.-L.; Leon, F.; Nobis, S.; Lambert, G.; et al. Changes in Microbiota and Bacterial Protein Caseinolytic Peptidase B During Food Restriction in Mice: Relevance for the Onset and Perpetuation of Anorexia Nervosa. Nutrients 2019, 11, 2514. [Google Scholar] [CrossRef]
- Breton, J.; Jacquemot, J.; Yaker, L.; Leclerc, C.; Connil, N.; Feuilloley, M.; Déchelotte, P.; Fetissov, S.O. Host Starvation and Female Sex Influence Enterobacterial ClpB Production: A Possible Link to the Etiology of Eating Disorders. Microorganisms 2020, 8, 530. [Google Scholar] [CrossRef] [PubMed]
- Maillard, F.; Pereira, B.; Boisseau, N. Effect of High-Intensity Interval Training on Total, Abdominal and Visceral Fat Mass: A Meta-Analysis. Sports Med. 2018, 48, 269–288. [Google Scholar] [CrossRef] [PubMed]
- Andreato, L.V.; Esteves, J.V.; Coimbra, D.R.; Moraes, A.J.P.; de Carvalho, T. The Influence of High-Intensity Interval Training on Anthropometric Variables of Adults with Overweight or Obesity: A Systematic Review and Network Meta-Analysis. Obes. Rev. 2019, 20, 142–155. [Google Scholar] [CrossRef] [PubMed]
- Depommier, C.; Everard, A.; Druart, C.; Plovier, H.; Van Hul, M.; Vieira-Silva, S.; Falony, G.; Raes, J.; Maiter, D.; Delzenne, N.M.; et al. Supplementation with Akkermansia Muciniphila in Overweight and Obese Human Volunteers: A Proof-of-Concept Exploratory Study. Nat. Med. 2019, 25, 1096–1103. [Google Scholar] [CrossRef] [PubMed]
- Folope, V.; Hellot, M.-F.; Kuhn, J.-M.; Ténière, P.; Scotté, M.; Déchelotte, P. Weight Loss and Quality of Life after Bariatric Surgery: A Study of 200 Patients after Vertical Gastroplasty or Adjustable Gastric Banding. Eur. J. Clin. Nutr. 2007, 62, 1022–1030. [Google Scholar] [CrossRef]
- Gordon, D.M.; FitzGibbon, F. The Distribution of Enteric Bacteria from Australian Mammals: Host and Geographical Effects. Microbiology 1999, 145, 2663–2671. [Google Scholar] [CrossRef] [PubMed]
- Tornadijo, E.; Fresno, J.M.; Carballo, J.; Martín-Sarmiento, R. Study of Enterobacteriaceae throughout the Manufacturing and Ripening of Hard Goats’ Cheese. J. Appl. Bacteriol. 1993, 75, 240–246. [Google Scholar] [CrossRef]
- Veiga, P.; Suez, J.; Derrien, M.; Elinav, E. Moving from Probiotics to Precision Probiotics. Nat. Microbiol. 2020, 5, 878–880. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Total | ITT HA group | P Group | p | Total | PP HA group | P Group | p |
|---|---|---|---|---|---|---|---|---|
| Frequency | 230 | 113 | 117 | - | 212 | 104 | 108 | - |
| Gender m/f | 103/127 | 51/62 | 52/65 | 1.000 | 97/115 | 47/57 | 50/58 | 0.891 |
| Body height [cm] | 173.1 ± 10.1 | 173.4 ± 10.9 | 172.9 ± 9.4 | 0.986 | 173.2 ± 10.1 | 173.4 ± 10.9 | 173.1 ± 9.4 | 0.791 |
| Body weight [kg] | 83.8 ± 10.8 | 84.1 ± 11.5 | 83.4 ± 10.2 | 0.817 | 83.8 ± 10.9 | 84.1 ± 11.7 | 83.5 ± 10.2 | 0.931 |
| BMI [kg/m²] | 27.8 ± 1.4 | 27.9 ± 1.4 | 27.8 ± 1.4 | 0.909 | 27.8 ± 1.4 | 27.9 ± 1.4 | 27.8 ± 1.4 | 0.744 |
| Body fat content [%] | 32.3 ± 7.3 | 31.9 ± 7.4 | 32.6 ± 7.3 | 0.414 | 32.1 ± 7.3 | 31.9 ± 7.3 | 32.4 ± 7.3 | 0.582 |
| Body fat mass [kg] | 26.7 ± 5.6 | 26.4 ± 5.3 | 26.9 ± 5.8 | 0.563 | 26.5 ± 5.5 | 26.4 ± 5.3 | 26.7 ± 5.7 | 0.730 |
| Fat free mass [kg] | 57.2 ± 11.9 | 58.0 ± 12.7 | 56.5 ± 11.1 | 0.488 | 57.3 ± 12 | 57.9 ± 12.8 | 56.8 ± 11.2 | 0.660 |
| Waist circumference [cm] | 101.3 ± 9.1 | 101.0 ± 9.4 | 101.6 ± 8.9 | 0.656 | 101.1 ± 9.3 | 100.7 ± 9.7 | 101.4 ± 8.9 | 0.568 |
| Hip circumference [cm] | 106.2 ± 7.0 | 106.1 ± 6.7 | 106.2 ± 7.3 | 0.761 | 106.0 ± 7.0 | 106.1 ± 6.7 | 105.9 ± 7.3 | 0.547 |
| Systolic blood pressure [mmHg] | 126.6 ± 10.8 | 125.8 ± 11.0 | 127.4 ± 10.7 | 0.319 | 126.7 ± 11 | 126.1 ± 11.4 | 127.4 ± 10.6 | 0.519 |
| Diastolic blood pressure [mmHg] | 79.8 ± 6.2 | 79.9 ± 6.1 | 79.6 ± 6.2 | 0.629 | 79.7 ± 6.3 | 79.9 ± 6.3 | 79.5 ± 6.3 | 0.461 |
| Pulse [bpm] | 69.7 ± 7.8 | 70.4 ± 8.0 | 69.0 ± 7.6 | 0.313 | 69.5 ± 7.9 | 70.3 ± 8.2 | 68.7 ± 7.7 | 0.313 |
| Parameters [Unit] | −4w | HA Group w12 | -4w | P Group w12 | Pu W12 (HA v P) |
|---|---|---|---|---|---|
| Haemoglobin [mmol/L] | 8.78 ± 0.82 | 8.75 ± 0.83 | 8.75 ± 0.75 | 0.82 ± 0.73 | 0.452 |
| Haematocrit [L/L] | 0.422 ± 0.036 | 0.424 ± 0.038 | 0.422 ± 0.035 | 0.426 ± 0.034 | 0.478 |
| Erythrocytes [Tpt/L] | 4.78 ± 0.44 | 4.77 ± 0.45 | 4.73 ± 0.42 | 4.68 ± 0.42 | 0.905 |
| Thrombocytes [Gpt/L] | 260.0 ± 58.1 | 265.9 ± 61.2 | 261.4 ± 48.1 | 266.8 ± 56.6 | 0.657 |
| Leukocytes [GpT/L] | 6.57 ± 1.84 | 6.60 ± 1.81 | 6.57 ± 1.92 | 6.57 ± 1.84 | 0.904 |
| ALAT [µkat/L] | 0.499 ± 0.292 | 0.468 ± 0.362 | 0.454 ± 0.223 | 0.408 ± 0.173 | 0.621 |
| ASAT [µkat/L] | 0.430 ± 0.116 | 0.439 ± 0.192 | 0.412 ± 0.138 | 0.416 ± 0.109 | 0.068 |
| AP [µkat/L] | 1.25 ± 0.343 | 1.27 ± 0.341 | 1.171 ± 0.314 | 1.170 ± 0.318 | 0.018 |
| gGT [µkat/L] | 0.406 ± 0.270 | 0.37 ± 0.22 | 0.397 ± 0.282 | 0.363 ± 0.261 | 0.479 |
| Biluribin [µmol/L] | 10.04 ± 5.2 | 10.4 ± 4.6 | 10.71 ± 5.76 | 12.00 ± 6.22 | 0.069 |
| Creatinine [µmol/L] | 74.5 ± 11.8 | 75.3 ± 12.6 | 74.9 ± 12.1 | 77.3 ± 11.8 | 0.181 |
| Urea [mmol/L] | 4.81 ± 1.3 | 4.69 ± 1.10 | 4.89 ± 1.40 | 4.84 ± 1.25 | 0.508 |
| Uric acid [µmol/L] | 311.4 ± 80.6 | 310.1 ± 83.2 | 317.4 ± 82.1 | 322.2 ± 81.8 | 0.368 |
| Glucose [mmol/L] | 5.46 ± 1.2 | 5.38 ± 0.3 | 5.49 ± 0.44 | 5.52 ± 0.53 | 0.027 |
| HbA1c [%] | 5.37 ± 0.29 | 5.38 ± 0.26 | 5.34 ± 0.24 | 5.36 ± 0.25 | 0.447 |
| Cholesterol [mmol/L] | 5.40 ± 1.16 | 5.19 ± 1.27 | 5.40 ± 1.08 | 5.28 ± 1.01 | 0.229 |
| HDL- Cholesterol [mmol/L] | 1.45 ± 0.38 | 1.43 ± 0.36 | 1.52 ± 0.34 | 1.49 ± 0.32 | 0.112 |
| LDL-cholestetol [mmol/L] | 3.53 ± 1.00 | 3.40 ± 1.09 | 3.53 ± 1.07 | 3.44 ± 0.92 | 0.423 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Déchelotte, P.; Breton, J.; Trotin-Picolo, C.; Grube, B.; Erlenbeck, C.; Bothe, G.; Fetissov, S.O.; Lambert, G. The Probiotic Strain H. alvei HA4597® Improves Weight Loss in Overweight Subjects under Moderate Hypocaloric Diet: A Proof-of-Concept, Multicenter Randomized, Double-Blind Placebo-Controlled Study. Nutrients 2021, 13, 1902. https://doi.org/10.3390/nu13061902
Déchelotte P, Breton J, Trotin-Picolo C, Grube B, Erlenbeck C, Bothe G, Fetissov SO, Lambert G. The Probiotic Strain H. alvei HA4597® Improves Weight Loss in Overweight Subjects under Moderate Hypocaloric Diet: A Proof-of-Concept, Multicenter Randomized, Double-Blind Placebo-Controlled Study. Nutrients. 2021; 13(6):1902. https://doi.org/10.3390/nu13061902
Chicago/Turabian StyleDéchelotte, Pierre, Jonathan Breton, Clémentine Trotin-Picolo, Barbara Grube, Constantin Erlenbeck, Gordana Bothe, Sergueï O. Fetissov, and Grégory Lambert. 2021. "The Probiotic Strain H. alvei HA4597® Improves Weight Loss in Overweight Subjects under Moderate Hypocaloric Diet: A Proof-of-Concept, Multicenter Randomized, Double-Blind Placebo-Controlled Study" Nutrients 13, no. 6: 1902. https://doi.org/10.3390/nu13061902
APA StyleDéchelotte, P., Breton, J., Trotin-Picolo, C., Grube, B., Erlenbeck, C., Bothe, G., Fetissov, S. O., & Lambert, G. (2021). The Probiotic Strain H. alvei HA4597® Improves Weight Loss in Overweight Subjects under Moderate Hypocaloric Diet: A Proof-of-Concept, Multicenter Randomized, Double-Blind Placebo-Controlled Study. Nutrients, 13(6), 1902. https://doi.org/10.3390/nu13061902








