Polycystic Ovary Syndrome in Insulin-Resistant Adolescents with Obesity: The Role of Nutrition Therapy and Food Supplements as a Strategy to Protect Fertility
Abstract
1. Introduction
2. Methods
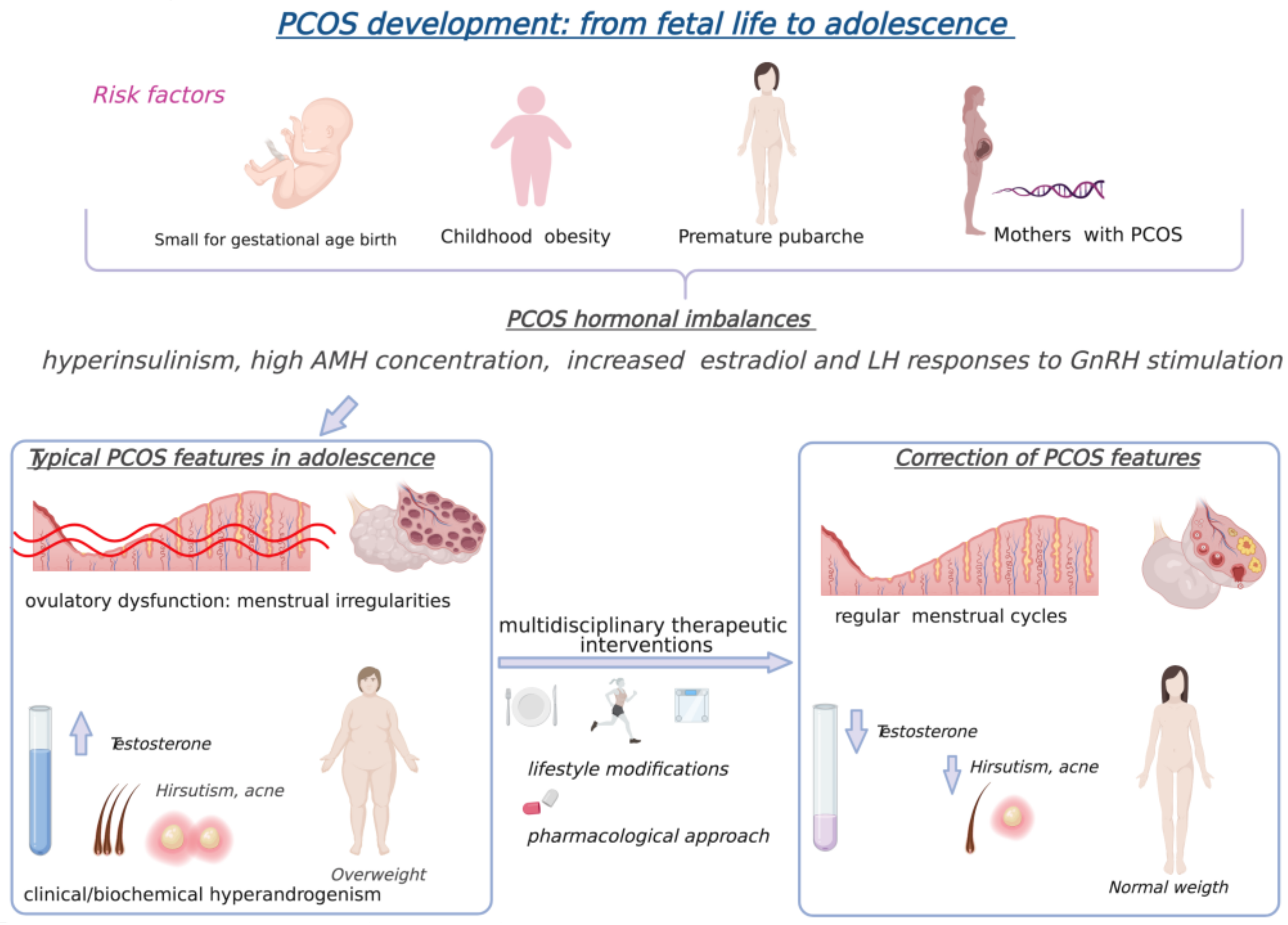
3. Polycystic Ovary Syndrome in Adolescents
4. Polycystic Ovary Syndrome and Obesity
The Adipose Tissue and Ovarian Dysfunction
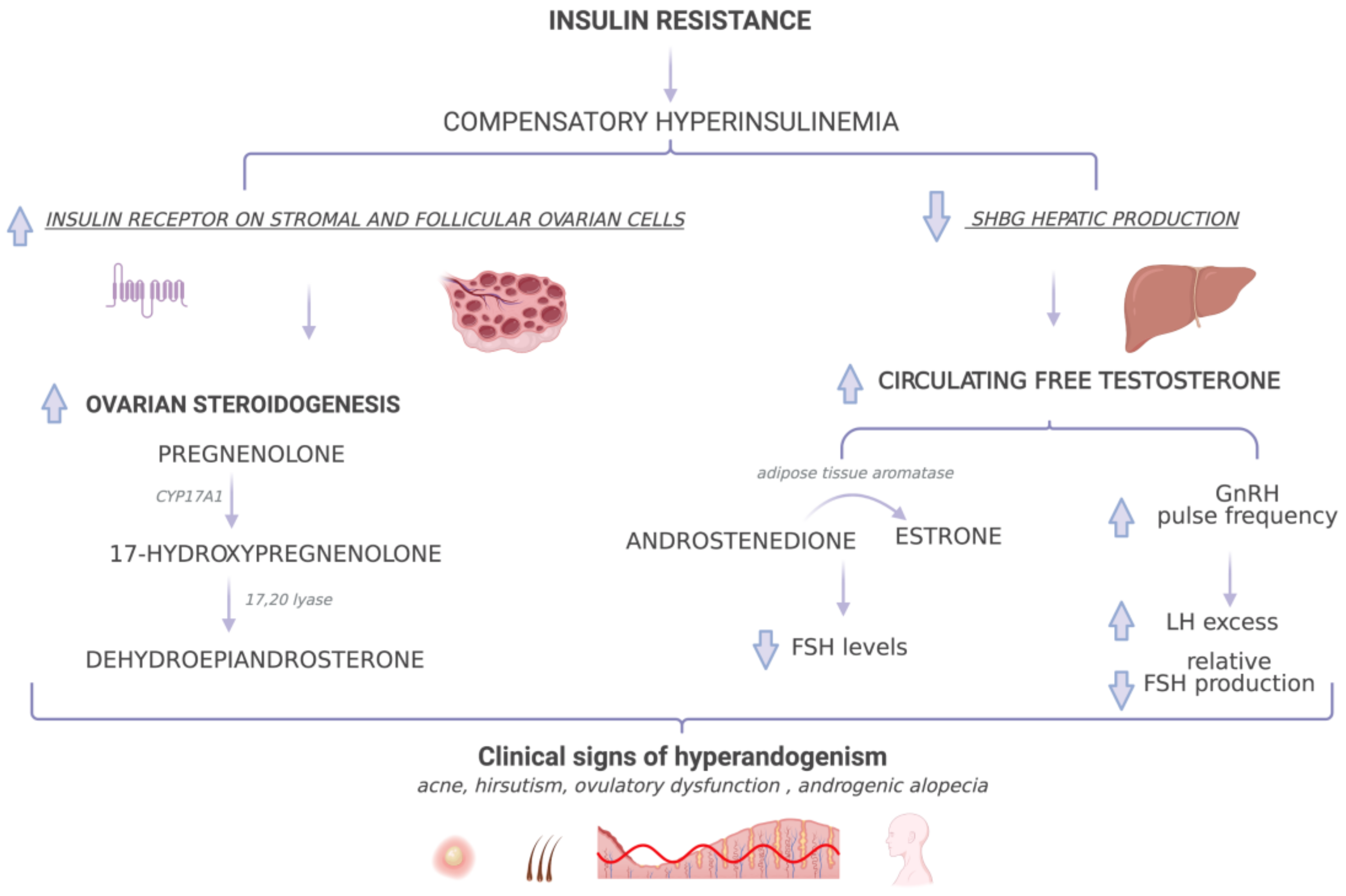
5. Polycystic Ovary Syndrome and Insulin Resistance
Molecular Mechanism of Insulin Resistance
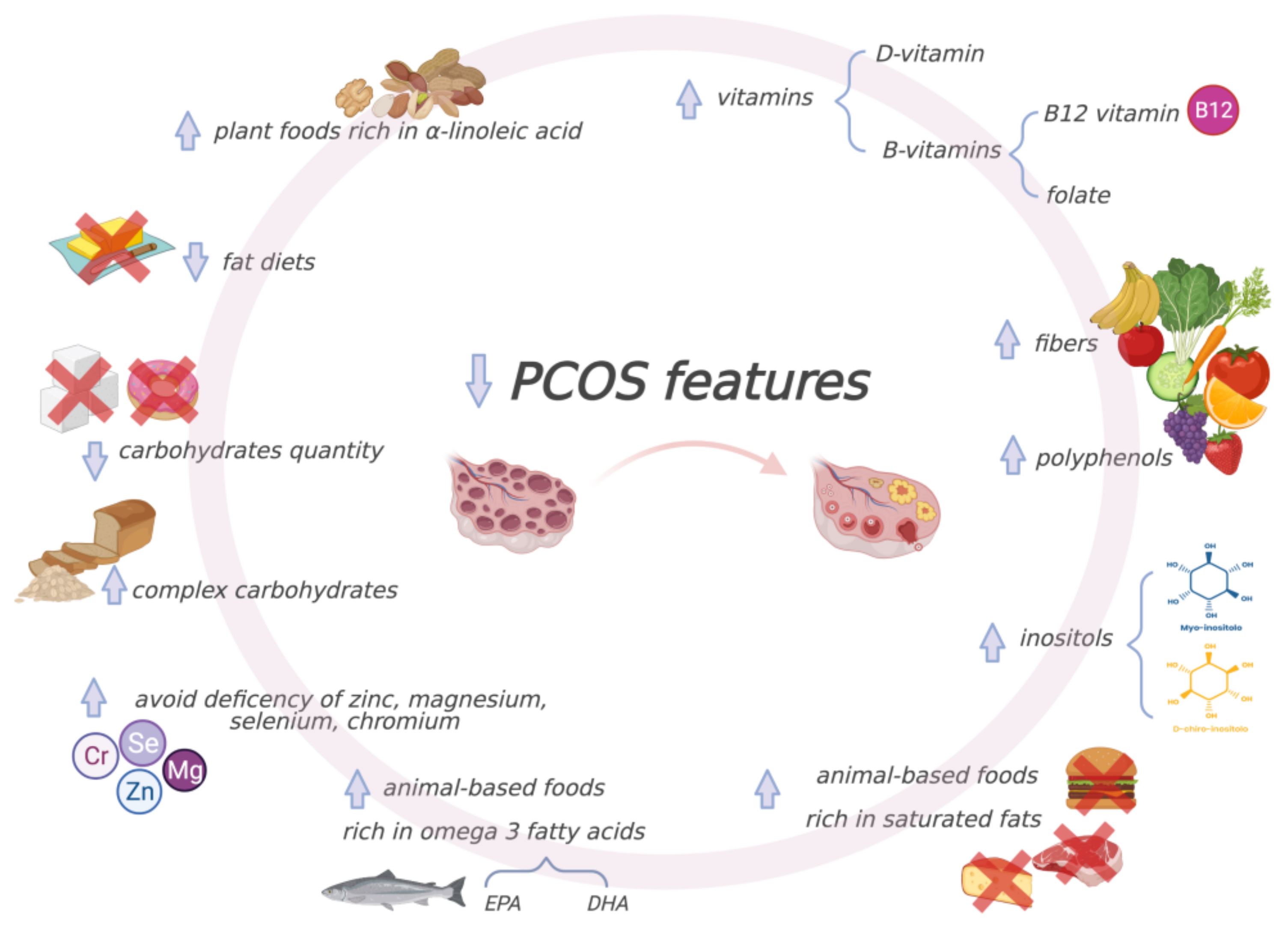
6. Medical Nutrition Therapy for Polycystic Ovary Syndrome
6.1. Role and Mechanisms of Action of Dietary Macronutrients
6.2. Role and Mechanisms of Action of Dietary Micronutrients
6.3. Diets with Beneficial Effects on Insulin Resistance
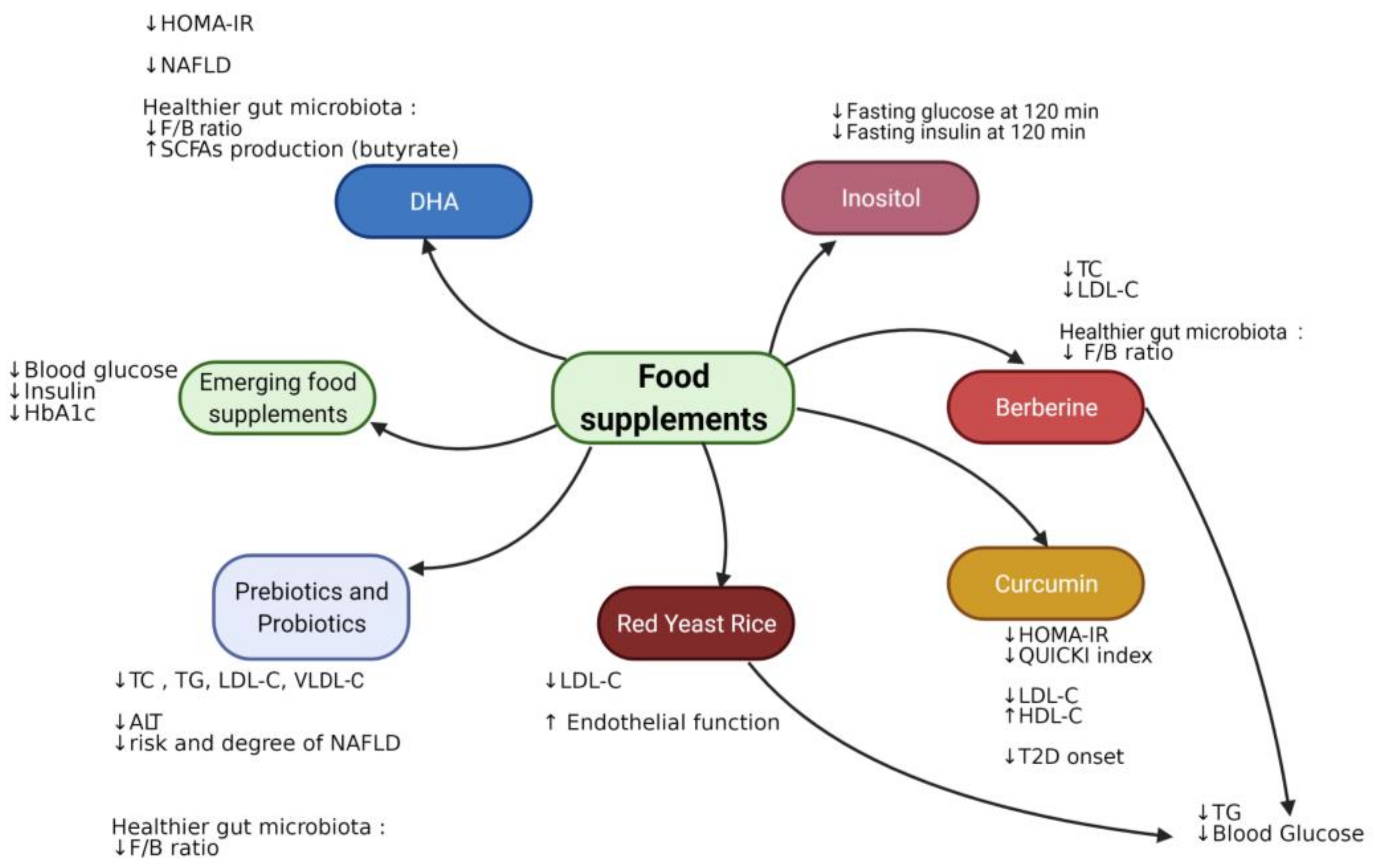
7. Food Supplements in Insulin Resistance
7.1. D-Chiro-f (DCI)
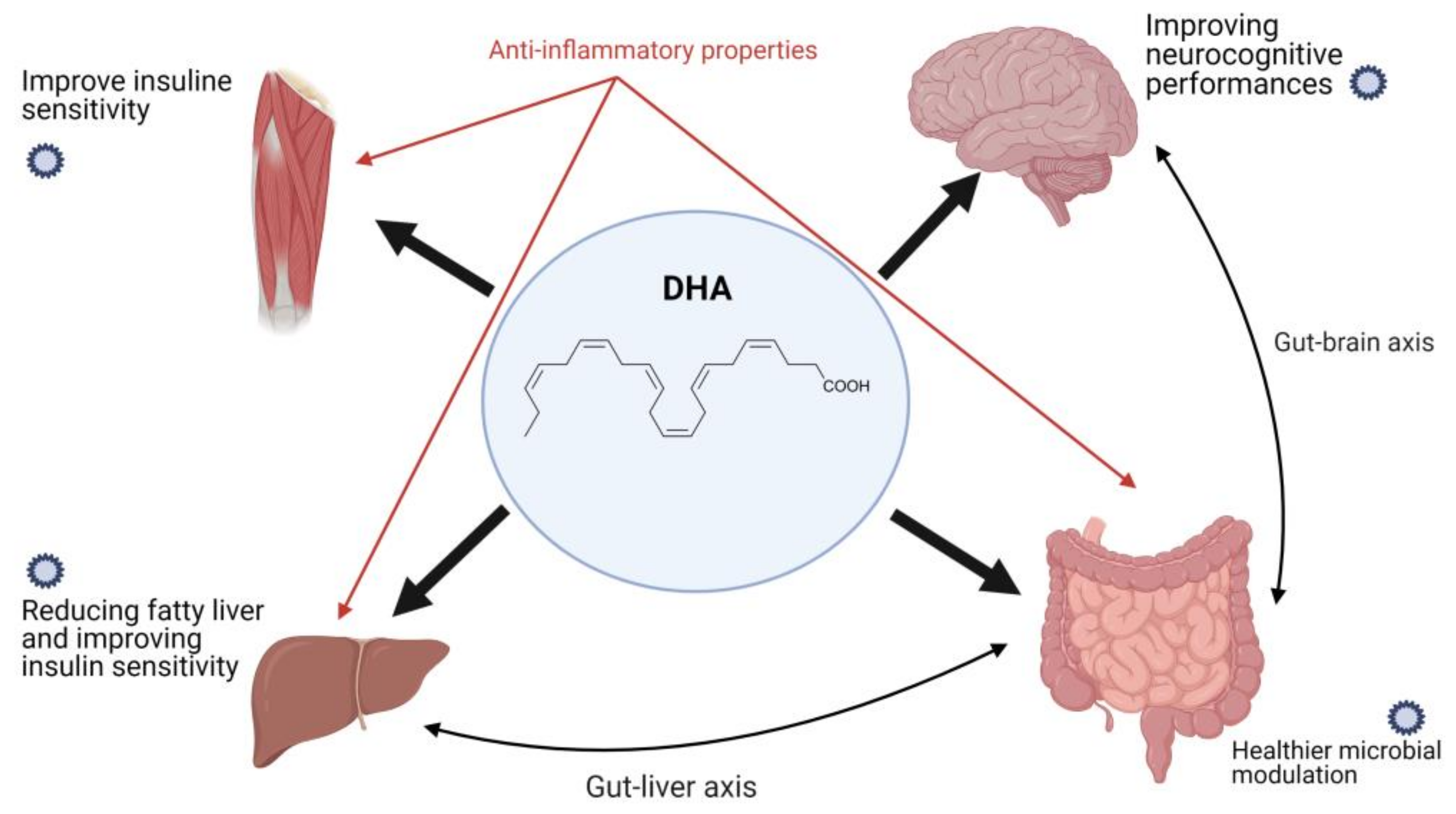
7.2. Docosahexaenoic Acid (DHA)
7.3. Red Yeast Rice (RYR)
7.4. Berberine (BBR)
7.5. Curcumin
8. Prebiotics, Probiotics and Postbiotics
9. Emerging Functional Foods and Food Supplements
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil. Steril. 2004, 81, 19–25. [CrossRef]
- Aboeldalyl, S.; James, C.; Seyam, E.; Ibrahim, E.M.; Shawki, H.E.D.; Amer, S. The role of chronic inflammation in polycystic ovarian syndrome—a systematic review and meta-analysis. Int. J. Mol. Sci. 2021, 22, 2734. [Google Scholar] [CrossRef]
- Cooney, L.G.; Dokras, A. Beyond fertility: Polycystic ovary syndrome and long-term health. Fertil. Steril. 2018, 110, 794–809. [Google Scholar] [CrossRef] [PubMed]
- Bernardo, W.M.; Nobre, M.R.C.; Jatene, F.B. Evidence-based clinical practice. Part II--Searching evidence databases. Rev. Assoc. Med. Bras. 2004, 50, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Trent, M.; Gordon, C.M. Diagnosis and management of polycystic ovary syndrome in adolescents. Pediatrics 2020, 145, S210–S218. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.; Coffler, M.S.; Dahan, M.H.; Malcom, P.J.; Deutsch, R.; Chang, R.J. Relationship of GnRH-stimulated LH release to episodic LH secretion and baseline endocrine-metabolic measures in women with polycystic ovary syndrome. Clin. Endocrinol. 2004, 60, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Franks, S.; Stark, J.; Hardy, K. Follicle dynamics and anovulation in polycystic ovary syndrome. Hum. Reprod. Update 2008, 14, 367–378. [Google Scholar] [CrossRef]
- Rosenfield, R.L.; Ehrmann, D.A. The Pathogenesis of Polycystic Ovary Syndrome (PCOS): The Hypothesis of PCOS as Functional Ovarian Hyperandrogenism Revisited. Endocr. Rev. 2016, 37, 467–520. [Google Scholar] [CrossRef] [PubMed]
- Dumesic, D.A.; Oberfield, S.E.; Stener-Victorin, E.; Marshall, J.C.; Laven, J.S.; Legro, R.S. Scientific statement on the diagnostic criteria, epidemiology, pathophysiology, and molecular genetics of polycystic ovary syndrome. Endocr. Rev. 2015, 36, 487–525. [Google Scholar] [CrossRef]
- Diamanti-Kandarakis, E.; Dunaif, A. Insulin resistance and the polycystic ovary syndrome revisited: An update on mechanisms and implications. Endocr. Rev. 2012, 33, 981–1030. [Google Scholar] [CrossRef]
- Burt Solorzano, C.M.; McCartney, C.R. Polycystic Ovary Syndrome: Ontogeny in Adolescence. Endocrinol. Metab. Clin. N. Am. 2021, 50, 25–42. [Google Scholar] [CrossRef]
- McCartney, C.R.; Campbell, R.E. Abnormal GnRH pulsatility in polycystic ovary syndrome: Recent insights. Curr. Opin. Endocr. Metab. Res. 2020, 12, 78–84. [Google Scholar] [CrossRef]
- Crisosto, N.; Echiburú, B.; Maliqueo, M.; Pérez, V.; Ladrón de Guevara, A.; Preisler, J.; Sánchez, F.; Sir-Petermann, T. Improvement of hyperandrogenism and hyperinsulinemia during pregnancy in women with polycystic ovary syndrome: Possible effect in the ovarian follicular mass of their daughters. Fertil. Steril. 2012, 97, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Crisosto, N.; Ladrón de Guevara, A.; Echiburú, B.; Maliqueo, M.; Cavada, G.; Codner, E.; Paez, F.; Sir-Petermann, T. Higher luteinizing hormone levels associated with antimüllerian hormone in postmenarchal daughters of women with polycystic ovary syndrome. Fertil. Steril. 2019, 111, 381–388. [Google Scholar] [CrossRef]
- Torchen, L.C.; Legro, R.S.; Dunaif, A. Distinctive Reproductive Phenotypes in Peripubertal Girls at Risk for Polycystic Ovary Syndrome. J. Clin. Endocrinol. Metab. 2019, 104, 3355–3361. [Google Scholar] [CrossRef] [PubMed]
- Crisosto, N.; Codner, E.; Maliqueo, M.; Echiburú, B.; Sánchez, F.; Cassorla, F.; Sir-Petermann, T. Anti-Müllerian Hormone Levels in Peripubertal Daughters of Women with Polycystic Ovary Syndrome. J. Clin. Endocrinol. Metab. 2007, 92, 2739–2743. [Google Scholar] [CrossRef] [PubMed]
- Witchel, S.F.; Oberfield, S.; Rosenfield, R.L.; Codner, E.; Bonny, A.; Ibáñez, L.; Pena, A.; Horikawa, R.; Gomez-Lobo, V.; Joel, D.; et al. The Diagnosis of Polycystic Ovary Syndrome during Adolescence. Horm. Res. Paediatr. 2015, 83, 376–389. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez, L.; Oberfield, S.E.; Witchel, S.; Auchus, R.J.; Chang, R.J.; Codner, E.; Dabadghao, P.; Darendeliler, F.; Elbarbary, N.S.; Gambineri, A.; et al. An International Consortium Update: Pathophysiology, Diagnosis, and Treatment of Polycystic Ovarian Syndrome in Adolescence. Horm. Res. Paediatr. 2017, 88, 371–395. [Google Scholar] [CrossRef]
- Teede, H.J.; Misso, M.L.; Costello, M.F.; Dokras, A.; Laven, J.; Moran, L.; Piltonen, T.; Norman, R.J.; Andersen, M.; Azziz, R.; et al. Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Fertil. Steril. 2018, 110, 364–379. [Google Scholar] [CrossRef]
- Ibáñez, L.; Potau, N.; Virdis, R. Postpubertal outcome in girls diagnosed of premature pubarche during childhood: Increased frequency of functional ovarian hyperandrogenism. J. Clin. Endocrinol. Metab. 1993, 76, 1599–1603. [Google Scholar] [CrossRef]
- Idkowiak, J.; Lavery, G.G.; Dhir, V.; Barrett, T.G.; Stewart, P.M.; Krone, N.; Arlt, W. Premature adrenarche: Novel lessons from early onset androgen excess. Eur. J. Endocrinol. 2011, 165, 189–207. [Google Scholar] [CrossRef] [PubMed]
- Paterson, W.F.; Ahmed, S.F.; Bath, L.; Donaldson, M.D.C.; Fleming, R.; Greene, S.A.; Hunter, I.; Kelnar, C.J.H.; Mayo, A.; Schulga, J.S.; et al. Exaggerated adrenarche in a cohort of Scottish children: Clinical features and biochemistry. Clin. Endocrinol. 2010, 72, 496–501. [Google Scholar] [CrossRef] [PubMed]
- Broekmans, F.J.; Visser, J.A.; Laven, J.S.E.; Broer, S.L.; Themmen, A.P.N.; Fauser, B.C. Anti-Müllerian hormone and ovarian dysfunction. Trends Endocrinol. Metab. 2008, 19, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Rosenfield, R.L. Evidence that idiopathic functional adrenal hyperandrogenism is caused by dysregulation of adrenal steroidogenesis and that hyperinsulinemia may be involved. J. Clin. Endocrinol. Metab. 1996, 81, 878–880. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez, L.; Díaz, R.; López-Bermejo, A.; Marcos, M.V. Clinical spectrum of premature pubarche: Links to metabolic syndrome and ovarian hyperandrogenism. Rev. Endocr. Metab. Disord. 2009, 10, 63–76. [Google Scholar] [CrossRef]
- Anderson, A.D.; Solorzano, C.M.B.; McCartney, C.R. Childhood Obesity and Its Impact on the Development of Adolescent PCOS. Semin. Reprod. Med. 2014, 32, 202–213. [Google Scholar] [CrossRef]
- Day, F.; Karaderi, T.; Jones, M.R.; Meun, C.; He, C.; Drong, A.; Kraft, P.; Lin, N.; Huang, H.; Broer, L.; et al. Large-scale genome-wide meta-analysis of polycystic ovary syndrome suggests shared genetic architecture for different diagnosis criteria. PLoS Genet. 2018, 14, e1007813. [Google Scholar] [CrossRef]
- Nokoff, N.; Thurston, J.; Hilkin, A.; Pyle, L.; Zeitler, P.S.; Nadeau, K.J.; Santoro, N.; Kelsey, M.M. Sex Differences in Effects of Obesity on Reproductive Hormones and Glucose Metabolism in Early Puberty. J. Clin. Endocrinol. Metab. 2019, 104, 4390–4397. [Google Scholar] [CrossRef]
- Calcaterra, V.; Cena, H.; Regalbuto, C.; Vinci, F.; Porri, D.; Verduci, E.; Chiara, M.; Zuccotti, G.V. The Role of Fetal, Infant, and Childhood Nutrition in the Timing of Sexual Maturation. Nutrients 2021, 13, 419. [Google Scholar] [CrossRef]
- Ibáñez, L.; Potau, N.; Francois, I.; de Zegher, F. Precocious Pubarche, Hyperinsulinism, and Ovarian Hyperandrogenism in Girls: Relation to Reduced Fetal Growth. J. Clin. Endocrinol. Metab. 1998, 83, 3558–3562. [Google Scholar] [CrossRef]
- Ortiz-Flores, A.E.; Luque-Ramírez, M.; Escobar-Morreale, H.F. Polycystic ovary syndrome in adult women. Med. Clínica Engl. Ed. 2019, 152, 450–457. [Google Scholar] [CrossRef]
- Rosenfield, R.L. Perspectives on the International Recommendations for the Diagnosis and Treatment of Polycystic Ovary Syndrome in Adolescence. J. Pediatr. Adolesc. Gynecol. 2020, 33, 445–447. [Google Scholar] [CrossRef]
- Kamboj, M.K.; Bonny, A.E. Polycystic ovary syndrome in adolescence: Diagnostic and therapeutic strategies. Transl. Pediatr. 2017, 6, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Senaldi, L.; Gopi, R.P.; Milla, S.; Shah, B. Is ultrasound useful in the diagnosis of adolescents with polycystic ovary syndrome? J. Pediatr. Endocrinol. Metab. 2015, 28, 605–612. [Google Scholar] [CrossRef]
- Ekramzadeh, M.; Hajivandi, L.; Noroozi, M.; Mostafavi, F. Psychological Experiences of Adolescent Girls with Polycystic Ovary Syndrome: A Qualitative Study. Iran. J. Nurs. Midwifery Res. 2020, 25, 341–347. [Google Scholar] [CrossRef]
- Ruan, X.; Li, M.; Mueck, A.O. Why does Polycystic Ovary Syndrome (PCOS) Need Long-term Management? Curr. Pharm. Des. 2018, 24, 4685–4692. [Google Scholar] [CrossRef] [PubMed]
- Moran, L.J.; Ko, H.; Misso, M.; Marsh, K.; Noakes, M.; Talbot, M.; Frearson, M.; Thondan, M.; Stepto, N.; Teede, H.J. Dietary Composition in the Treatment of Polycystic Ovary Syndrome: A Systematic Review to Inform Evidence-Based Guidelines. J. Acad. Nutr. Diet. 2013, 113, 520–545. [Google Scholar] [CrossRef] [PubMed]
- Shele, G.; Genkil, J.; Speelman, D. A Systematic Review of the Effects of Exercise on Hormones in Women with Polycystic Ovary Syndrome. J. Funct. Morphol. Kinesiol. 2020, 5, 35. [Google Scholar]
- Naderpoor, N.; Shorakae, S.; de Courten, B.; Misso, M.L.; Moran, L.J.; Teede, H.J. Metformin and lifestyle modification in polycystic ovary syndrome: Systematic review and meta-analysis. Hum. Reprod. Update 2015, 21, 560–574. [Google Scholar] [CrossRef]
- Lamos, E.M.; Malek, R.; Davis, S.N. GLP-1 receptor agonists in the treatment of polycystic ovary syndrome. Expert Rev. Clin. Pharmacol. 2017, 10, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Cena, H.; Chiovato, L.; Nappi, R.E. Obesity, Polycystic Ovary Syndrome, and Infertility: A New Avenue for GLP-1 Receptor Agonists. J. Clin. Endocrinol. Metab. 2020, 105, e2695–e2709. [Google Scholar] [CrossRef]
- Morales Camacho, W.J.; Molina Díaz, J.M.; Plata Ortiz, S.; Plata Ortiz, J.E.; Morales Camacho, M.A.; Calderón, B.P. Childhood obesity: Aetiology, comorbidities, and treatment. Diabetes. Metab. Res. Rev. 2019, 35, e3203. [Google Scholar] [CrossRef] [PubMed]
- Glueck, C.J.; Goldenberg, N. Characteristics of obesity in polycystic ovary syndrome: Etiology, treatment, and genetics. Metabolism 2019, 92, 108–120. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lim, H. The global childhood obesity epidemic and the association between socio-economic status and childhood obesity. Int. Rev. Psychiatry 2012, 24, 176–188. [Google Scholar] [CrossRef] [PubMed]
- Sommer, A.; Twig, G. The Impact of Childhood and Adolescent Obesity on Cardiovascular Risk in Adulthood: A Systematic Review. Curr. Diab. Rep. 2018, 18. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 26 May 2021).
- Syndrome, P.O.; Women, O. Prevalence and Characteristics of the Polycystic Ovary Syndrome in Overweight and Obese Women. Obstet. Gynecol. 2007, 109, 446. [Google Scholar] [CrossRef]
- Legro, R.S.; Kunselman, A.R.; Dodson, W.C.; Dunaif, A. Prevalence and Predictors of Risk for Type 2 Diabetes Mellitus and Impaired Glucose Tolerance in Polycystic Ovary Syndrome. J. Clin. Endocrinol. Metab. 1999, 84, 165–169. [Google Scholar] [CrossRef]
- Zeitler, P.S.; Nadeau, K.J.; Carreau, A.-M.; Pesant, M.-H.; Baillargeon, J.-P. Insulin resistance - Childhood Precursors of Adult Disease; Springer Nature: Basingstoke, UK, 2020; ISBN 978-3-030-25057-7. [Google Scholar]
- Ezeh, U.; Yildiz, B.O.; Azziz, R. Referral bias in defining the phenotype and prevalence of obesity in polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2013, 98, 1088–1096. [Google Scholar] [CrossRef]
- Cassar, S.; Teede, H.J.; Moran, L.J.; Joham, A.E.; Harrison, C.L.; Strauss, B.J.; Stepto, N.K. Polycystic ovary syndrome and anti-Müllerian hormone: Role of insulin resistance, androgens, obesity and gonadotrophins. Clin. Endocrinol. 2014, 81, 899–906. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Feng, Q.; Ye, M.; He, Y.; Yao, A.; Shi, K. Metabolic effect of obesity on polycystic ovary syndrome in adolescents: A meta-analysis. J. Obstet. Gynaecol. 2017, 37, 1036–1047. [Google Scholar] [CrossRef]
- Yetim Şahin, A.; Baş, F.; Yetim, Ç.; Uçar, A.; Poyrazoğlu, Ş.; Bundak, R.; Darendeliler, F. Determination of insulin resistance and its relationship with hyperandrogenemia, anti-müllerian hormone, inhibin A, inhibin B, and insulin-like peptide-3 levels in adolescent girls with polycystic ovary syndrome. Turkish J. Med. Sci. 2019, 49, 1117–1125. [Google Scholar] [CrossRef]
- Dabadghao, P. Polycystic ovary syndrome in adolescents. Best Pract. Res. Clin. Endocrinol. Metab. 2019, 33, 101272. [Google Scholar] [CrossRef]
- Hickey, M.; Doherty, D.A.; Atkinson, H.; Sloboda, D.M.; Franks, S.; Norman, R.J.; Hart, R. Clinical, ultrasound and biochemical features of polycystic ovary syndrome in adolescents: Implications for diagnosis. Hum. Reprod. 2011, 26, 1469–1477. [Google Scholar] [CrossRef]
- De Medeiros, S.F.; de Medeiros, M.A.S.; Barbosa, B.B.; Yamamoto, M.M.W.; Maciel, G.A.R. Comparison of metabolic and obesity biomarkers between adolescent and adult women with polycystic ovary syndrome. Arch. Gynecol. Obstet. 2021, 303, 739–749. [Google Scholar] [CrossRef]
- Codner, E.; Cassorla, F. Puberty and ovarian function in girls with type 1 diabetes mellitus. Horm. Res. 2009, 71, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Hannon, T.S.; Janosky, J.; Arslanian, S.A. Longitudinal study of physiologic insulin resistance and metabolic changes of puberty. Pediatr. Res. 2006, 60, 759–763. [Google Scholar] [CrossRef] [PubMed]
- Shayya, R.; Chang, R.J. Reproductive endocrinology of adolescent polycystic ovary syndrome. BJOG An Int. J. Obstet. Gynaecol. 2010, 117, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Bou Nemer, L.; Shi, H.; Carr, B.R.; Word, R.A.; Bukulmez, O. Effect of Body Weight on Metabolic Hormones and Fatty Acid Metabolism in Follicular Fluid of Women Undergoing In Vitro Fertilization: A Pilot Study. Reprod. Sci. 2019, 26, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Laitinen, J.; Taponen, S.; Martikainen, H.; Pouta, A.; Millwood, I.; Hartikainen, A.L.; Ruokonen, A.; Sovio, U.; McCarthy, M.I.; Franks, S.; et al. Body size from birth to adulthood as a predictor of self-reported polycystic ovary syndrome symptoms. Int. J. Obes. 2003, 27, 710–715. [Google Scholar] [CrossRef] [PubMed]
- Bravo, J.; Raimundo, A.M.; Santos, D.A.; Timón, R.; Sardinha, L.B. Abdominal obesity in adolescents: Development of age-specific waist circumference cut-offs linked to adult IDF criteria. Am. J. Hum. Biol. 2017, 29, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kahn, H.S.; Imperatore, G.; Cheng, Y.J. A population-based comparison of BMI percentiles and waist-to-height ratio for identifying cardiovascular risk in youth. J. Pediatr. 2005, 146, 482–488. [Google Scholar] [CrossRef] [PubMed]
- Oguz, S.H.; İdilman, I.; Helvaci, N.; Guzelce, E.C.; Eyupoglu, D.; Karcaaltincaba, M.; Yildiz, B.O. Tissue fat quantification by magnetic resonance imaging: Proton density fat fraction in polycystic ovary syndrome. Reprod. Biomed. Online 2020, 41, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Kirchengast, S.; Huber, J. Body composition characteristics and body fat distribution in lean women with polycystic ovary syndrome. Hum. Reprod. 2001, 16, 1255–1260. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E. Role of obesity and adiposity in polycystic ovary syndrome. Int. J. Obes. 2007, 31, S8–S13. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nigro, E.; Scudiero, O.; Monaco, M.L.; Palmieri, A.; Mazzarella, G.; Costagliola, C.; Bianco, A.; Daniele, A. New insight into adiponectin role in obesity and obesity-related diseases. Biomed Res. Int. 2014, 2014. [Google Scholar] [CrossRef]
- Su, X.; Peng, D. Adipokines as novel biomarkers of cardio-metabolic disorders. Clin. Chim. Acta 2020, 507, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Lainez, N.M.; Coss, D. Obesity, Neuroinflammation, and Reproductive Function. Endocrinology 2019, 160, 2719–2736. [Google Scholar] [CrossRef]
- Villa, J.; Pratley, R.E. Adipose tissue dysfunction in polycystic ovary syndrome. Curr. Diab. Rep. 2011, 11, 179–184. [Google Scholar] [CrossRef]
- Rodden, A.M.; Diaz, V.A.; Mainous, A.G.; Koopman, R.J.; Geesey, M.E. Insulin Resistance in Adolescents. J. Pediatr. 2007, 151, 275–279. [Google Scholar] [CrossRef]
- Zaccardi, F.; Webb, D.R.; Yates, T.; Davies, M.J. Pathophysiology of type 1 and type 2 diabetes mellitus: A 90-year perspective. Postgrad. Med. J. 2016, 92, 63–69. [Google Scholar] [CrossRef]
- Gustafson, B.; Hammarstedt, A.; Andersson, C.X.; Smith, U. Inflamed adipose tissue: A culprit underlying the metabolic syndrome and atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 2276–2283. [Google Scholar] [CrossRef]
- Ek, I.; Arner, P.; Rydén, M.; Holm, C.; Thörne, A.; Hoffstedt, J.; Wahrenberg, H. A unique defect in the regulation of visceral fat cell lipolysis in the polycystic ovary syndrome as an early link to insulin resistance. Diabetes 2002, 51, 484–492. [Google Scholar] [CrossRef]
- Silvestris, E.; de Pergola, G.; Rosania, R.; Loverro, G. Obesity as disruptor of the female fertility. Reprod. Biol. Endocrinol. 2018, 16, 1–13. [Google Scholar] [CrossRef]
- Sørensen, T.I.A.; Virtue, S.; Vidal-Puig, A. Obesity as a clinical and public health problem: Is there a need for a new definition based on lipotoxicity effects? Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2010, 1801, 400–404. [Google Scholar] [CrossRef] [PubMed]
- Mannerås-Holm, L.; Leonhardt, H.; Kullberg, J.; Jennische, E.; Odén, A.; Holm, G.; Hellström, M.; Lönn, L.; Olivecrona, G.; Stener-Victorin, E.; et al. Adipose tissue has aberrant morphology and function in PCOS: Enlarged adipocytes and low serum adiponectin, but not circulating sex steroids, are strongly associated with insulin resistance. J. Clin. Endocrinol. Metab. 2011, 96, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Barbe, A.; Bongrani, A.; Mellouk, N.; Estienne, A.; Kurowska, P.; Grandhaye, J.; Elfassy, Y.; Levy, R.; Rak, A.; Froment, P.; et al. Mechanisms of adiponectin action in fertility: An overview from gametogenesis to gestation in humans and animal models in normal and pathological conditions. Int. J. Mol. Sci. 2019, 20, 1526. [Google Scholar] [CrossRef]
- Panagopoulou, P.; Galli-Tsinopoulou, A.; Fleva, A.; Pavlitou-Tsiontsi, E.; Vavatsi-Christaki, N.; Nousia-Arvanitakis, S. Adiponectin and insulin resistance in childhood obesity. J. Pediatr. Gastroenterol. Nutr. 2008, 47, 356–362. [Google Scholar] [CrossRef]
- Ouchi, N.; Kihara, S.; Funahashi, T.; Matsuzawa, Y.; Walsh, K. Obesity, adiponectin and vascular inflammatory disease. Curr. Opin. Lipidol. 2003, 14, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Shi, H.; Jin, Y.; Li, X.; Pan, J.; Lai, Y.; Lin, Y.; Jiin, Y.; Roy, G.; Zhao, A.; et al. Adiponectin deficiency leads to female subfertility and ovarian dysfunctions in mice. Endocrinology 2016, 157, 4875–4887. [Google Scholar] [CrossRef]
- Drolet, R.; Bélanger, C.; Fortier, M.; Huot, C.; Mailloux, J.; Légaré, D.; Tchernof, A. Fat depot-specific impact of visceral obesity on adipocyte adiponectin release in women. Obesity 2009, 17, 424–430. [Google Scholar] [CrossRef] [PubMed]
- De Rosa, A.; Ludovica Monaco, M.; Capasso, M.; Forestieri, P.; Pilone, V.; Nardelli, C.; Buono, P.; Daniele, A. Adiponectin oligomers as potential indicators of adipose tissue improvement in obese subjects. Eur. J. Endocrinol. 2013, 169, 37–43. [Google Scholar] [CrossRef]
- Toulis, K.A.; Goulis, D.G.; Farmakiotis, D.; Georgopoulos, N.A.; Katsikis, I.; Tarlatzis, B.C.; Papadimas, I.; Panidis, D. Adiponectin levels in women with polycystic ovary syndrome: A systematic review and a meta-analysis. Hum. Reprod. Update 2009, 15, 297–307. [Google Scholar] [CrossRef] [PubMed]
- Lebovitz, H.E. Insulin resistance: Definition and consequences. Exp. Clin. Endocrinol. Diabetes 2001, 109 (Suppl. 2), S135–S148. [Google Scholar] [CrossRef]
- Philip, Z.; Kristen, N. Insulin Resistance; Humana Press: Totowa, NJ, USA, 2020; ISBN 978-3-030-25057-7. [Google Scholar]
- Moghetti, P.; Tosi, F.; Bonin, C.; Di Sarra, D.; Fiers, T.; Kaufman, J.M.; Giagulli, V.A.; Signori, C.; Zambotti, F.; Dall’Alda, M.; et al. Divergences in insulin resistance between the different phenotypes of the polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2013, 98, 628–637. [Google Scholar] [CrossRef]
- Carreau, A.M.; Baillargeon, J.P. PCOS in Adolescence and Type 2 Diabetes. Curr. Diab. Rep. 2015, 15. [Google Scholar] [CrossRef]
- Rajkhowa, M.; Bicknell, J.; Jones, M.; Clayton, R.N. Insulin sensitivity in women with polycystic ovary syndrome: Relationship to hyperandrogenemia. Fertil. Steril. 1994, 61, 605–612. [Google Scholar] [CrossRef]
- DeUgarte, C.M.; Bartolucci, A.A.; Azziz, R. Prevalence of insulin resistance in the polycystic ovary syndrome using the homeostasis model assessment. Fertil. Steril. 2005, 83, 1454–1460. [Google Scholar] [CrossRef]
- Barber, T.M.; Wass, J.A.H.; McCarthy, M.I.; Franks, S. Metabolic characteristics of women with polycystic ovaries and oligo-amenorrhoea but normal androgen levels: Implications for the management of polycystic ovary syndrome. Clin. Endocrinol. 2007, 66, 513–517. [Google Scholar] [CrossRef] [PubMed]
- Dewailly, D.; Catteau-Jonard, S.; Reyss, A.C.; Leroy, M.; Pigny, P. Oligoanovulation with polycystic ovaries but not overt hyperandrogenism. J. Clin. Endocrinol. Metab. 2006, 91, 3922–3927. [Google Scholar] [CrossRef]
- Lewy, V.D.; Danadian, K.; Witchel, S.F.; Arslanian, S. Early metabolic abnormalities in adolescent girls with polycystic ovarian syndrome. J. Pediatr. 2001, 138, 38–44. [Google Scholar] [CrossRef]
- Poretsky, L.; Kalin, M.F. The gonadotropic function of insulin. Endocr. Rev. 2021, 9, 6. [Google Scholar] [CrossRef] [PubMed]
- Cree-Green, M.; Rahat, H.; Newcomer, B.R.; Bergman, B.C.; Brown, M.S.; Coe, G.V.; Newnes, L.; Garcia-Reyes, Y.; Bacon, S.; Thurston, J.E.; et al. Insulin resistance, hyperinsulinemia, and mitochondria dysfunction in nonobese girls with polycystic ovarian syndrome. J. Endocr. Soc. 2017, 1, 931–944. [Google Scholar] [CrossRef]
- Zhao, Y.; Xu, J.J.; Zhu, M.W.; Lin, J.F. Clinical features, reproductive endocrine and metabolic abnormalities of adolescent girls with polycystic ovary syndrome. Natl. Med. J. China 2010, 90, 1729–1734. [Google Scholar]
- Poretsky, L.; Cataldo, N.A.; Rosenwaks, Z.; Giudice, L.C. The insulin-related ovarian regulatory system in health and disease. Endocr. Rev. 1999, 20, 535–582. [Google Scholar] [CrossRef] [PubMed]
- Moghetti, P.; Tosi, F. Insulin resistance and PCOS: Chicken or egg? J. Endocrinol. Investig. 2021, 44, 233–244. [Google Scholar] [CrossRef] [PubMed]
- Goodman, N.F.; Cobin, R.H.; Futterweit, W.; Glueck, J.S.; Legro, R.S.; Carmina, E. American association of clinical endocrinologists, American college of endocrinology, and androgen excess and pcos society disease state clinical review: Guide to the best practices in the evaluation and treatment of polycystic ovary syndrome - Part 2. Endocr. Pract. 2015, 21, 1415–1426. [Google Scholar] [CrossRef] [PubMed]
- Cadagan, D.; Khan, R.; Amer, S. Thecal cell sensitivity to luteinizing hormone and insulin in polycystic ovarian syndrome. Reprod. Biol. 2016, 16, 53–60. [Google Scholar] [CrossRef]
- Zeng, X.; Xie, Y.-J.; Liu, Y.-T.; Long, S.-L.; Mo, Z.-C. Polycystic ovarian syndrome: Correlation between hyperandrogenism, insulin resistance and obesity. Clin. Chim. Acta 2020, 502, 214–221. [Google Scholar] [CrossRef]
- Nelson, L.R.; Bulun, S.E. Estrogen production and action. J. Am. Acad. Dermatol. 2001, 45, S116–S124. [Google Scholar] [CrossRef]
- Chang, R.J.; Mandel, F.P.; Lu, J.K.H.; Judd, H.L. Enhanced disparity of gonadotropin secretion by estrone in women with poly cystic ovarian disease. J. Clin. Endocrinol. Metab. 1982, 54, 490–494. [Google Scholar] [CrossRef]
- McCartney, C.R. Maturation of sleep-wake gonadotrophin-releasing hormone secretion across puberty in girls: Potential mechanisms and relevance to the pathogenesis of polycystic ovary syndrome. J. Neuroendocrinol. 2010, 22, 701–709. [Google Scholar] [CrossRef]
- Cree-Green, M.; Bergman, B.C.; Coe, G.V.; Newnes, L.; Baumgartner, A.D.; Bacon, S.; Sherzinger, A.; Pyle, L.; Nadeau, K.J. Hepatic Steatosis is Common in Adolescents with Obesity and PCOS and Relates to De Novo Lipogenesis but not Insulin Resistance. Obesity 2016, 24, 2399–2406. [Google Scholar] [CrossRef] [PubMed]
- Mai, K.; Bobbert, T.; Reinecke, F.; Andres, J.; Maser-Gluth, C.; Wudy, S.A.; Möhlig, M.; Weickert, M.O.; Hartmann, M.F.; Schulte, H.M.; et al. Intravenous lipid and heparin infusion-induced elevation in free fatty acids and triglycerides modifies circulating androgen levels in women: A randomized, controlled trial. J. Clin. Endocrinol. Metab. 2008, 93, 3900–3906. [Google Scholar] [CrossRef]
- Leblanc, S.; Battista, M.C.; Noll, C.; Hallberg, A.; Gallo-Payet, N.; Carpentier, A.C.; Vine, D.F.; Baillargeon, J.P. Angiotensin II type 2 receptor stimulation improves fatty acid ovarian uptake and hyperandrogenemia in an obese rat model of polycystic ovary syndrome. Endocrinology 2014, 155, 3684–3693. [Google Scholar] [CrossRef]
- Azziz, R.; Carmina, E.; Dewailly, D.; Diamanti-Kandarakis, E.; Escobar-Morreale, H.F.; Futterweit, W.; Janssen, O.E.; Legro, R.S.; Norman, R.J.; Taylor, A.E.; et al. The Androgen Excess and PCOS Society Criteria for the Polycystic Ovary Syndrome: The Complete Task Force Report. Fertil. Steril. 2009, 91, 456–488. [Google Scholar] [CrossRef]
- Goodarzi, M.O.; Carmina, E.; Azziz, R. DHEA, DHEAS and PCOS. J. Steroid Biochem. Mol. Biol. 2015, 145, 213–225. [Google Scholar] [CrossRef]
- Book, C.B.; Dunaif, A. Selective insulin resistance in the polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 1999, 84, 3110–3116. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Pilch, P.F. The insulin receptor: Structure, function, and signaling. Am. J. Physiol. 1994, 266, C319–C334. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.; Kim, Y.B. Molecular mechanism of insulin resistance in obesity and type 2 diabetes. Korean J. Intern. Med. 2010, 25, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Dun, J.; Yang, J.; Zhang, J.; Lin, Q.; Huang, M.; Ji, F.; Huang, L.; You, X.; Lin, Y. Letrozole Rat Model Mimics Human Polycystic Ovarian Syndrome and Changes in Insulin Signal Pathways. Med. Sci. Monit. 2020, 26, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Hsueh, W.A.; Law, R.E. Cardiovascular risk continuum: Implications of insulin resistance and diabetes. Am. J. Med. 1998, 105, 4S–14S. [Google Scholar] [CrossRef]
- Griffeth, R.J.; Bianda, V.; Nef, S. The emerging role of insulin-like growth factors in testis development and function. Basic Clin. Androl. 2014, 24. [Google Scholar] [CrossRef]
- Rajkhowa, M.; Brett, S.; Cuthbertson, D.J.; Lipina, C.; Ruiz-Alcaraz, A.J.; Thomas, G.E.; Logie, L.; Petrie, J.R.; Sutherland, C. Insulin resistance in polycystic ovary syndrome is associated with defective regulation of ERK1/2 by insulin in skeletal muscle in vivo. Biochem. J. 2009, 418, 665–671. [Google Scholar] [CrossRef]
- Dunaif, A.; Xia, J.; Book, C.B.; Schenker, E.; Tang, Z. Excessive insulin receptor serine phosphorylation in cultured fibroblasts and in skeletal muscle: A potential mechanism for insulin resistance in the polycystic ovary syndrome. J. Clin. Invest. 1995, 96, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Dunaif, A.; Wu, X.; Lee, A.; Diamanti-Kandarakis, E. Defects in insulin receptor signaling in vivo in the polycystic ovary syndrome (PCOS). Am. J. Physiol. Endocrinol. Metab. 2001, 281, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Youngren, J.F.; Dunaif, A.; Goldfine, I.D.; Maddux, B.A.; Zhang, B.B.; Evans, J.L. Decreased Insulin Receptor (IR) autophosphorylation in fibroblasts from patients with PCOS: Effects of serine kinase inhibitors and IR activators. J. Clin. Endocrinol. Metab. 2002, 87, 4088–4093. [Google Scholar] [CrossRef][Green Version]
- Corbould, A.; Zhao, H.; Mirzoeva, S.; Aird, F.; Dunaif, A. Enhanced mitogenic signaling in skeletal muscle of women with polycystic ovary syndrome. Diabetes 2006, 55, 751–759. [Google Scholar] [CrossRef]
- Zhang, L.H.; Rodriguez, H.; Ohno, S.; Miller, W.L. Serine phosphorylation of human P450C17 increases 17,20-lyase activity: Implications for adrenarche and the polycystic ovary syndrome. Proc. Natl. Acad. Sci. USA 1995, 92, 10619–10623. [Google Scholar] [CrossRef] [PubMed]
- Garvey, W.T.; Mechanick, J.I.; Brett, E.M.; Garber, A.J.; Hurley, D.L.; Jastreboff, A.M.; Nadolsky, K.; Pessah-Pollack, R.; Plodkowski, R. American association of clinical endocrinologists and American college of endocrinology comprehensive clinical practice guidelines for medical care of patients with obesity. Endocr. Pract. 2016, 22, 1–203. [Google Scholar] [CrossRef]
- Gołąbek, K.D.; Regulska-Ilow, B. Dietary support in insulin resistance: An overview of current scientific reports. Adv. Clin. Exp. Med. 2019, 28, 1577–1585. [Google Scholar] [CrossRef] [PubMed]
- Salleh, S.N.; Fairus, A.A.H.; Zahary, M.N.; Raj, N.B.; Jalil, A.M.M. Unravelling the effects of soluble dietary fibre supplementation on energy intake and perceived satiety in healthy adults: Evidence from systematic review and meta-analysis of randomised-controlled trials. Foods 2019, 8, 15. [Google Scholar] [CrossRef] [PubMed]
- Aryaeian, N.; Sedehi, S.K.; Arablou, T. Polyphenols and their effects on diabetes management: A review. Med. J. Islam. Repub. Iran 2017, 31, 886–892. [Google Scholar] [CrossRef]
- Dinicola, S.; Minini, M.; Unfer, V.; Verna, R.; Cucina, A.; Bizzarri, M. Nutritional and acquired deficiencies in inositol bioavailability. Correlations with metabolic disorders. Int. J. Mol. Sci. 2017, 18, 2187. [Google Scholar] [CrossRef] [PubMed]
- Kalra, B.; Kalra, S.; Sharma, J.B. The inositols and polycystic ovary syndrome. Indian J. Endocrinol. Metab. 2016, 20, 720–724. [Google Scholar] [CrossRef]
- Zhao, X.; Jiang, Y.; Xi, H.; Chen, L.; Feng, X. Exploration of the Relationship between Gut Microbiota and Polycystic Ovary Syndrome (PCOS): A Review. Geburtshilfe Frauenheilkd. 2020, 80, 161–171. [Google Scholar] [CrossRef]
- Weickert, M.O. Nutritional Modulation of Insulin Resistance. Scientifica (Cairo) 2012, 2012, 1–15. [Google Scholar] [CrossRef] [PubMed][Green Version]
- He, F.F.; Li, Y.M. Role of gut microbiota in the development of insulin resistance and the mechanism underlying polycystic ovary syndrome: A review. J. Ovarian Res. 2020, 13, 1–13. [Google Scholar] [CrossRef]
- Giampaolino, P.; Foreste, V.; Di Filippo, C.; Gallo, A.; Mercorio, A.; Serafino, P.; Improda, F.P.; Verrazzo, P.; Zara, G.; Buonfantino, C.; et al. Microbiome and PCOS: State-of-art and future aspects. Int. J. Mol. Sci. 2021, 22, 48. [Google Scholar] [CrossRef]
- Shamasbi, S.G.; Ghanbari-Homayi, S.; Mirghafourvand, M. The effect of probiotics, prebiotics, and synbiotics on hormonal and inflammatory indices in women with polycystic ovary syndrome: a systematic review and meta-analysis. Eur. J. Nutr. 2020, 59, 433–450. [Google Scholar] [CrossRef]
- Della Guardia, L.; Thomas, M.A.; Cena, H. Insulin sensitivity and glucose homeostasis can be influenced by metabolic acid load. Nutrients 2018, 10, 618. [Google Scholar] [CrossRef]
- Acosta-Montaño, P.; García-González, V. Effects of dietary fatty acids in pancreatic beta cell metabolism, implications in homeostasis. Nutrients 2018, 10, 393. [Google Scholar] [CrossRef]
- Günalan, E.; Yaba, A.; Yılmaz, B. The effect of nutrient supplementation in the management of polycystic ovary syndrome-associated metabolic dysfunctions: A critical review. J. Turkish Ger. Gynecol. Assoc. 2018, 19, 220–232. [Google Scholar] [CrossRef]
- Wang, Y.; Lin, M.; Gao, X.; Pedram, P.; Du, J.; Vikram, C.; Gulliver, W.; Zhang, H.; Sun, G. High dietary selenium intake is associated with less insulin resistance in the Newfoundland population. PLoS ONE 2017, 12, 1–15. [Google Scholar] [CrossRef]
- Mancini, A.; Bruno, C.; Vergani, E.; D′abate, C.; Giacchi, E.; Silvestrini, A. Oxidative stress and low-grade inflammation in polycystic ovary syndrome: Controversies and new insights. Int. J. Mol. Sci. 2021, 22, 1667. [Google Scholar] [CrossRef]
- Lydic, M.L.; McNurlan, M.; Bembo, S.; Mitchell, L.; Komaroff, E.; Gelato, M. Chromium picolinate improves insulin sensitivity in obese subjects with polycystic ovary syndrome. Fertil. Steril. 2006, 86, 243–246. [Google Scholar] [CrossRef]
- Ashoush, S.; Abou-Gamrah, A.; Bayoumy, H.; Othman, N. Chromium picolinate reduces insulin resistance in polycystic ovary syndrome: Randomized controlled trial. J. Obstet. Gynaecol. Res. 2016, 42, 279–285. [Google Scholar] [CrossRef]
- Ansari, M.G.A.; Sabico, S.; Clerici, M.; Khattak, M.N.K.; Wani, K.; Al-Musharaf, S.; Amer, O.E.; Alokail, M.S.; Al-Daghri, N.M. Vitamin D supplementation is associated with increased glutathione peroxidase-1 levels in arab adults with prediabetes. Antioxidants 2020, 9, 118. [Google Scholar] [CrossRef] [PubMed]
- Łagowska, K.; Bajerska, J.; Jamka, M. The role of vitamin D oral supplementation in insulin resistance in women with polycystic ovary syndrome: A systematic review and meta-analysis of randomized controlled trials. Nutrients 2018, 10, 1637. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Tal, R.; Jiang, H.; Yuan, T.; Liu, Y. Vitamin D Supplementation Ameliorates Metabolic Dysfunction in Patients with PCOS: A SystematicReview of RCTs and Insight into the Underlying Mechanism. Int. J. Endocrinol. 2020, 2020. [Google Scholar] [CrossRef]
- Tagliaferri, S.; Porri, D.; De Giuseppe, R.; Manuelli, M.; Alessio, F.; Cena, H. The controversial role of Vitamin D as an antioxidant: Results from randomised controlled trials. Nutr. Res. Rev. 2019, 32, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Meng, Y.; Chen, X.; Peng, Z.; Liu, X.; Sun, Y.; Dai, S. Association between High Serum Homocysteine Levels and Biochemical Characteristics in Women with Polycystic Ovarian Syndrome: A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0157389. [Google Scholar] [CrossRef] [PubMed]
- De Giuseppe, R.; Braschi, V.; Bosoni, D.; Biino, G.; Stanford, F.C.; Nappi, R.E.; Cena, H. Dietary underreporting in women affected by polycystic ovary syndrome: A pilot study. Nutr. Diet. 2019, 76, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Douglas, C.C.; Gower, B.A.; Darnell, B.E.; Ovalle, F.; Oster, R.A.; Azziz, R. Role of diet in the treatment of polycystic ovary syndrome. Fertil. Steril. 2006, 85, 679–688. [Google Scholar] [CrossRef] [PubMed]
- Cena, H.; Calder, P.C. Defining a healthy diet: Evidence for the role of contemporary dietary patterns in health and disease. Nutrients 2020, 12, 334. [Google Scholar] [CrossRef] [PubMed]
- Violi, F.; Loffredo, L.; Pignatelli, P.; Angelico, F.; Bartimoccia, S.; Nocella, C.; Cangemi, R.; Petruccioli, A.; Monticolo, R.; Pastori, D.; et al. Extra virgin olive oil use is associated with improved post-prandial blood glucose and LDL cholesterol in healthy subjects. Nutr. Diabetes 2015, 5, e172. [Google Scholar] [CrossRef]
- Tsigalou, C.; Konstantinidis, T.; Paraschaki, A.; Stavropoulou, E.; Voidarou, C.; Bezirtzoglou, E. Mediterranean diet as a tool to combat inflammation and chronic diseases. An overview. Biomedicines 2020, 8, 201. [Google Scholar] [CrossRef]
- Górska, P.; Górna, I.; Przysławski, J. Mediterranean diet and oxidative stress. Nutr. Food Sci. 2020. [Google Scholar] [CrossRef]
- Lin, A.W.; Kazemi, M.; Jarrett, B.Y.; Brink, H.V.; Hoeger, K.M.; Spandorfer, S.D.; Lujan, M.E. Dietary and physical activity behaviors in women with polycystic ovary syndrome per the new international evidence-based guideline. Nutrients 2019, 11, 2711. [Google Scholar] [CrossRef]
- Esposito, K.; Maiorino, M.I.; Bellastella, G.; Chiodini, P.; Panagiotakos, D.; Giugliano, D. A journey into a Mediterranean diet and type 2 diabetes: A systematic review with meta-analyses. BMJ Open 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Marsh, K.A.; Steinbeck, K.S.; Atkinson, F.S.; Petocz, P.; Brand-Miller, J.C. Effect of a low glycemic index compared with a conventional healthy diet on polycystic ovary syndrome. Am. J. Clin. Nutr. 2010, 92, 83–92. [Google Scholar] [CrossRef]
- Zhang, X.; Zheng, Y.; Guo, Y.; Lai, Z. The Effect of Low Carbohydrate Diet on Polycystic Ovary Syndrome: A Meta-Analysis of Randomized Controlled Trials. Int. J. Endocrinol. 2019, 2019. [Google Scholar] [CrossRef] [PubMed]
- Farvid, M.S.; Homayouni, F.; Shokoohi, M.; Fallah, A.; Farvid, M.S. Glycemic index, glycemic load and their association with glycemic control among patients with type 2 diabetes. Eur. J. Clin. Nutr. 2014, 68, 459–463. [Google Scholar] [CrossRef]
- Paoli, A.; Mancin, L.; Giacona, M.C.; Bianco, A.; Caprio, M. Effects of a ketogenic diet in overweight women with polycystic ovary syndrome. J. Transl. Med. 2020, 18, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Mavropoulos, J.C.; Yancy, W.S.; Hepburn, J.; Westman, E.C. The effects of a low-carbohydrate, ketogenic diet on the polycystic ovary syndrome: A pilot study. Nutr. Metab. 2005, 2, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Shilpa, J.; Mohan, V. Ketogenic diets: Boon or bane? Indian J. Med. Res. 2018, 148, 251–253. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Xing, C.; Zhang, J.; Zhao, H.; Shi, W.; He, B. Eight-hour time-restricted feeding improves endocrine and metabolic profiles in women with anovulatory polycystic ovary syndrome. J. Transl. Med. 2021, 19, 1–9. [Google Scholar] [CrossRef]
- McDonald, T.J.W.; Cervenka, M.C. Lessons learned from recent clinical trials of ketogenic diet therapies in adults. Curr. Opin. Clin. Nutr. Metab. Care 2019, 22, 418–424. [Google Scholar] [CrossRef] [PubMed]
- Muscogiuri, G.; El Ghoch, M.; Colao, A.; Hassapidou, M.; Yumuk, V.; Busetto, L.; Obesity Management Task Force (OMTF) of the European Association for the Study of Obesity (EASO). European Guidelines for Obesity Management in Adults with a Very Low-Calorie Ketogenic Diet: A Systematic Review and Meta-Analysis. Obes. Facts 2021, 14, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Akram, T.; Hasan, S.; Imran, M.; Karim, A.; Arslan, M. Association of polycystic ovary syndrome with cardiovascular risk factors. Gynecol. Endocrinol. 2010, 26, 47–53. [Google Scholar] [CrossRef]
- Arentz, S.; Smith, C.A.; Abbott, J.; Bensoussan, A. Nutritional supplements and herbal medicines for women with polycystic ovary syndrome; a systematic review and meta-analysis. BMC Complement. Altern. Med. 2017, 17, 1–14. [Google Scholar] [CrossRef]
- Directive, E.U. Directive 2002/46/EC of the European Parliament and of the Council of 10 June 2002 on the approximation of the laws of the Member States relating to food supplements (Text with EEA relevance). Off. J. Eur. Communities Legis. 2002, 45, 51–57. [Google Scholar]
- Vettorazzi, A.; de Cerain, A.L.; Sanz-Serrano, J.; Gil, A.G.; Azqueta, A. European Regulatory Framework and Safety Assessment of Food-Related Bioactive Compounds. Nutrients 2020, 12, 613. [Google Scholar] [CrossRef]
- Gambioli, R.; Forte, G.; Aragona, C.; Bevilacqua, A.; Bizzarri, M.; Unfer, V. The use of D-chiro-Inositol in clinical practice. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 438–446. [Google Scholar] [CrossRef]
- Kamenov, Z.; Gateva, A. Inositols in PCOS. Molecules 2020, 25, 5566. [Google Scholar] [CrossRef] [PubMed]
- Sirtori, C.R.; Pavanello, C.; Calabresi, L.; Ruscica, M. Nutraceutical approaches to metabolic syndrome. Ann. Med. 2017, 49, 678–697. [Google Scholar] [CrossRef] [PubMed]
- Street, M.E.; Cirillo, F.; Catellani, C.; Dauriz, M.; Lazzeroni, P.; Sartori, C.; Moghetti, P. Current treatment for polycystic ovary syndrome: Focus on adolescence. Minerva Pediatr. 2020, 72, 288–311. [Google Scholar] [CrossRef] [PubMed]
- Bevilacqua, A.; Bizzarri, M. Inositols in insulin signaling and glucose metabolism. Int. J. Endocrinol. 2018, 2018. [Google Scholar] [CrossRef]
- Fonteles, M.C.; Almeida, M.Q.; Larner, J. Antihyperglycemic effects of 3-O-Methyl-D-Chiro-inositol and D-Chiro- inositol associated with manganese in streptozotocin diabetic rats. Horm. Metab. Res. 2000, 32, 129–132. [Google Scholar] [CrossRef]
- Gao, Y.-F.; Zhang, M.-N.; Wang, T.-X.; Wu, T.-C.; Ai, R.-D.; Zhang, Z.-S. Hypoglycemic effect of D-chiro-inositol in type 2 diabetes mellitus rats through the PI3K/Akt signaling pathway. Mol. Cell. Endocrinol. 2016, 433, 26–34. [Google Scholar] [CrossRef]
- Larner, J.; Huang, L.C.; Schwartz, C.F.; Oswald, A.S.; Shen, T.Y.; Kinter, M.; Tang, G.Z.; Zeller, K. Rat liver insulin mediator which stimulates pyruvate dehydrogenase phosphate contains galactosamine and D-chiroinositol. Biochem. Biophys. Res. Commun. 1988, 151, 1416–1426. [Google Scholar] [CrossRef]
- Ortmeyer, H.K.; Bodkin, N.L.; Hansen, B.C.; Larner, J. In vivo D-chiroinositol activates skeletal muscle glycogen synthase and inactivates glycogen phosphorylase in rhesus monkeys. J. Nutr. Biochem. 1995, 6, 499–503. [Google Scholar] [CrossRef]
- Cheang, K.I.; Baillargeon, J.P.; Essah, P.A.; Ostlund, R.E.; Apridonize, T.; Islam, L.; Nestler, J.E. Insulin-stimulated release of d-chiro-inositol-containing inositolphosphoglycan mediator correlates with insulin sensitivity in women with polycystic ovary syndrome. Metabolism 2008, 57, 1390–1397. [Google Scholar] [CrossRef] [PubMed]
- Mancini, M.; Andreassi, A.; Salvioni, M.; Pelliccione, F.; Mantellassi, G.; Banderali, G. Myoinositol and D-Chiro Inositol in Improving Insulin Resistance in Obese Male Children: Preliminary Data. Int. J. Endocrinol. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- González, F.; Rote, N.S.; Minium, J.; Kirwan, J.P. Reactive oxygen species-induced oxidative stress in the development of insulin resistance and hyperandrogenism in polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2006, 91, 336–340. [Google Scholar] [CrossRef]
- De Leo, V.; La Marca, A.; Cappelli, V.; Stendardi, A.; Focarelli, R.; Musacchio, M.C.; Piomboni, P. Evaluation of the treatment with D-chiro-inositol on levels of oxidative stress in PCOS patients. Minerva Ginecol. 2012, 64, 531–538. [Google Scholar]
- Lehner, A.; Staub, K.; Aldakak, L.; Eppenberger, P.; Rühli, F.; Martin, R.D.; Bender, N. Impact of omega-3 fatty acid DHA and EPA supplementation in pregnant or breast-feeding women on cognitive performance of children: Systematic review and meta-analysis. Nutr. Rev. 2021, 79, 585–598. [Google Scholar] [CrossRef]
- Amatruda, M.; Ippolito, G.; Vizzuso, S.; Vizzari, G.; Banderali, G.; Verduci, E. Epigenetic effects of n-3 LCPUFAs: A role in pediatric metabolic syndrome. Int. J. Mol. Sci. 2019, 20, 2118. [Google Scholar] [CrossRef]
- Scaglioni, S.; Verduci, E.; Salvioni, M.; Bruzzese, M.G.; Radaelli, G.; Zetterström, R.; Riva, E.; Agostoni, C. Plasma long-chain fatty acids and the degree of obesity in Italian children. Acta Paediatr. Int. J. Paediatr. 2006, 95, 964–969. [Google Scholar] [CrossRef]
- Verduci, E.; Radaelli, G.; Salvioni, M.; Riva, E.; Giovannini, M. Plasma long-chain fatty acids profile and metabolic outcomes in normolipidaemic obese children after one-year nutritional intervention. Acta Paediatr. Int. J. Paediatr. 2011, 100, 585–589. [Google Scholar] [CrossRef]
- Nobili, V.; Alisi, A.; Della Corte, C.; Risé, P.; Galli, C.; Agostoni, C.; Bedogni, G. Docosahexaenoic acid for the treatment of fatty liver: Randomised controlled trial in children. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 1066–1070. [Google Scholar] [CrossRef]
- Fekete, K.; Györei, E.; Lohner, S.; Verduci, E.; Agostoni, C.; Decsi, T. Long-chain polyunsaturated fatty acid status in obesity: A systematic review and meta-analysis. Obes. Rev. 2015, 16, 488–497. [Google Scholar] [CrossRef] [PubMed]
- Riva, A.; Borgo, F.; Lassandro, C.; Verduci, E.; Morace, G.; Borghi, E.; Berry, D. Pediatric obesity is associated with an altered gut microbiota and discordant shifts in Firmicutes populations. Environ. Microbiol. 2017, 19, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Borgo, F.; Verduci, E.; Riva, A.; Lassandro, C.; Riva, E.; Morace, G.; Borghi, E. Relative Abundance in Bacterial and Fungal Gut Microbes in Obese Children: A Case Control Study. Child. Obes. 2017, 13, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.D. The gut microbiome and its role in obesity. Nutr. Today 2016, 51, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Vasickova, L.; Stavek, P.; Suchanek, P. Possible effect of DHA intake on body weight reduction and lipid metabolism in obese children. Neuro Endocrinol. Lett. 2011, 32 (Suppl. 2), 64–67. [Google Scholar]
- Wu, G.D.; Chen, J.; Hoffmann, C.; Bittinger, K.; Chen, Y.-Y.; Keilbaugh, S.A.; Bewtra, M.; Knights, D.; Walters, W.A.; Knight, R.; et al. Linking Long-Term Dietary Patterns with Gut Microbial Enterotypes. Science 2011, 334, 105–109. [Google Scholar] [CrossRef]
- Magne, F.; Gotteland, M.; Gauthier, L.; Zazueta, A.; Pesoa, S.; Navarrete, P.; Balamurugan, R. The firmicutes/bacteroidetes ratio: A relevant marker of gut dysbiosis in obese patients? Nutrients 2020, 12, 1474. [Google Scholar] [CrossRef]
- Tabbaa, M.; Golubic, M.; Roizen, M.F.; Bernstein, A.M. Docosahexaenoic acid, inflammation, and bacterial dysbiosis in relation to periodontal disease, inflammatory bowel disease, and the metabolic syndrome. Nutrients 2013, 5, 3299–3310. [Google Scholar] [CrossRef]
- Amatruda, M.; Vizzuso, S.; Fabiano, V.; Borghi, E.; Zuccotti, G.V.; Verduci, E. Effect of Docosahexaenoic Acid Supplementation on Microbiota in Obese ChiLdrEn: A Pilot Study. (The DAMOCLE Study). In Proceedings of the 1st International Electronic Conference on Nutrients_Nutritional and Microbiota Effects on Chronic Disease, Basel, Switzerland, 2–15 November 2020; Volume 21, pp. 1–9. [Google Scholar]
- Fukami, H.; Higa, Y.; Hisano, T.; Asano, K.; Hirata, T.; Nishibe, S. A Review of Red Yeast Rice, a Traditional Fermented Food in Japan and East Asia: Its Characteristic Ingredients and Application in the Maintenance and Improvement of Health in Lipid Metabolism and the Circulatory System. Molecules 2021, 26, 1619. [Google Scholar] [CrossRef]
- Lu, Z.; Kou, W.; Du, B.; Wu, Y.; Zhao, S.; Brusco, O.A.; Morgan, J.M.; Capuzzi, D.M. Effect of Xuezhikang, an Extract From Red Yeast Chinese Rice, on Coronary Events in a Chinese Population With Previous Myocardial Infarction. Am. J. Cardiol. 2008, 101, 1689–1693. [Google Scholar] [CrossRef]
- Cicero, A.F.G.; Morbini, M.; Rosticci, M.; D’’Addato, S.; Grandi, E.; Borghi, C. Middle-Term Dietary Supplementation with Red Yeast Rice Plus Coenzyme Q10 Improves Lipid Pattern, Endothelial Reactivity and Arterial Stiffness in Moderately Hypercholesterolemic Subjects. Ann. Nutr. Metab. 2016, 68, 213–219. [Google Scholar] [CrossRef]
- Pirro, M.; Mannarino, M.R.; Bianconi, V.; Simental-Mendía, L.E.; Bagaglia, F.; Mannarino, E.; Sahebkar, A. The effects of a nutraceutical combination on plasma lipids and glucose: A systematic review and meta-analysis of randomized controlled trials. Pharmacol. Res. 2016, 110, 76–88. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.-W.; Zhou, J.; Gober, H.-J.; Leung, W.T.; Wang, L. Effect and mechanism of berberine against polycystic ovary syndrome. Biomed. Pharmacother. 2021, 138, 111468. [Google Scholar] [CrossRef]
- Tang, J.; Feng, Y.; Tsao, S.; Wang, N.; Curtain, R.; Wang, Y. Berberine and Coptidis Rhizoma as novel antineoplastic agents: A review of traditional use and biomedical investigations. J. Ethnopharmacol. 2009, 126, 5–17. [Google Scholar] [CrossRef]
- Wang, Y.; Fu, X.; Xu, J.; Wang, Q.; Kuang, H. Systems pharmacology to investigate the interaction of berberine and other drugs in treating polycystic ovary syndrome. Sci. Rep. 2016, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Chien, Y.J.; Chang, C.Y.; Wu, M.Y.; Chen, C.H.; Horng, Y.S.; Wu, H.C. Effects of curcumin on glycemic control and lipid profile in polycystic ovary syndrome: Systematic review with meta-analysis and trial sequential analysis. Nutrients 2021, 13, 684. [Google Scholar] [CrossRef] [PubMed]
- Chuengsamarn, S.; Rattanamongkolgul, S.; Luechapudiporn, R.; Phisalaphong, C.; Jirawatnotai, S. Curcumin extract for prevention of type 2 diabetes. Diabetes Care 2012, 35, 2121–2127. [Google Scholar] [CrossRef]
- Pari, L.; Murugan, P. Changes in glycoprotein components in streptozotocin - Nicotinamide induced type 2 diabetes: Influence of tetrahydrocurcumin from curcuma longa. Plant Foods Hum. Nutr. 2007, 62, 25–29. [Google Scholar] [CrossRef]
- Heshmati, J.; Moini, A.; Sepidarkish, M.; Morvaridzadeh, M.; Salehi, M.; Palmowski, A.; Mojtahedi, M.F.; Shidfar, F. Effects of curcumin supplementation on blood glucose, insulin resistance and androgens in patients with polycystic ovary syndrome: A randomized double-blind placebo-controlled clinical trial. Phytomedicine 2021, 80, 153395. [Google Scholar] [CrossRef]
- Lin, X.; Liu, M.-H.; Hu, H.-J.; Feng, H.; Fan, X.-J.; Zou, W.; Pan, Y.; Hu, X.; Wang, Z. Curcumin enhanced cholesterol efflux by upregulating ABCA1 expression through AMPK-SIRT1-LXRα signaling in THP-1 macrophage-derived foam cells. DNA Cell Biol. 2015, 34, 561–572. [Google Scholar] [CrossRef] [PubMed]
- Sohrevardi, S.M.; Heydari, B.; Azarpazhooh, M.R.; Teymourzadeh, M.; Simental-Mendía, L.E.; Atkin, S.L.; Sahebkar, A.; Karimi-Zarchi, M. Therapeutic Effect of Curcumin in Women with Polycystic Ovary Syndrome Receiving Metformin: A Randomized Controlled Trial. Adv. Exp. Med. Biol. 2021, 1308, 109–117. [Google Scholar] [CrossRef]
- Panahi, Y.; Khalili, N.; Hosseini, M.S.; Abbasinazari, M.; Sahebkar, A. Lipid-modifying effects of adjunctive therapy with curcuminoids-piperine combination in patients with metabolic syndrome: Results of a randomized controlled trial. Complement. Ther. Med. 2014, 22, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Sáez-Lara, M.J.; Robles-Sanchez, C.; Ruiz-Ojeda, F.J.; Plaza-Diaz, J.; Gil, A. Effects of probiotics and synbiotics on obesity, insulin resistance syndrome, type 2 diabetes and non-alcoholic fatty liver disease: A review of human clinical trials. Int. J. Mol. Sci. 2016, 17, 928. [Google Scholar] [CrossRef]
- Neyrinck, A.M.; Rodriguez, J.; Zhang, Z.; Seethaler, B.; Sánchez, C.R.; Roumain, M.; Hiel, S.; Bindels, L.B.; Cani, P.D.; Paquot, N.; et al. Prebiotic dietary fibre intervention improves fecal markers related to inflammation in obese patients: Results from the Food4Gut randomized placebo-controlled trial. Eur. J. Nutr. 2021. [Google Scholar] [CrossRef]
- Rajkumar, H.; Mahmood, N.; Kumar, M.; Varikuti, S.R.; Challa, H.R.; Myakala, S.P. Effect of probiotic (VSL#3) and omega-3 on lipid profile, insulin sensitivity, inflammatory markers, and gut colonization in overweight adults: A randomized, controlled trial. Mediators Inflamm. 2014, 2014. [Google Scholar] [CrossRef]
- Alisi, A.; Bedogni, G.; Baviera, G.; Giorgio, V.; Porro, E.; Paris, C.; Giammaria, P.; Reali, L.; Anania, F.; Nobili, V. Randomised clinical trial: The beneficial effects of VSL#3 in obese children with non-alcoholic steatohepatitis. Aliment. Pharmacol. Ther. 2014, 39, 1276–1285. [Google Scholar] [CrossRef] [PubMed]
- Larsen, N.; Vogensen, F.K.; Gøbel, R.J.; Michaelsen, K.F.; Forssten, S.D.; Lahtinen, S.J.; Jakobsen, M. Effect of Lactobacillus salivarius Ls-33 on fecal microbiota in obese adolescents. Clin. Nutr. 2013, 32, 935–940. [Google Scholar] [CrossRef] [PubMed]
- Famouri, F.; Shariat, Z.; Hashemipour, M.; Keikha, M.; Kelishadi, R. Effects of probiotics on nonalcoholic fatty liver disease in obese children and adolescents. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 413–417. [Google Scholar] [CrossRef]
- Miccheli, A.; Capuani, G.; Marini, F.; Tomassini, A.; Praticò, G.; Ceccarelli, S.; Gnani, D.; Baviera, G.; Alisi, A.; Putignani, L.; et al. Urinary (1)H-NMR-based metabolic profiling of children with NAFLD undergoing VSL#3 treatment. Int. J. Obes. 2015, 39, 1118–1125. [Google Scholar] [CrossRef]
- Vajro, P.; Mandato, C.; Licenziati, M.R.; Franzese, A.; Vitale, D.F.; Lenta, S.; Caropreso, M.; Vallone, G.; Meli, R. Effects of lactobacillus rhamnosus strain gg in pediatric obesity-related liver disease. J. Pediatr. Gastroenterol. Nutr. 2011, 52, 740–743. [Google Scholar] [CrossRef]
- Lin, H.V.; Frassetto, A.; Kowalik, E.J.J.; Nawrocki, A.R.; Lu, M.M.; Kosinski, J.R.; Hubert, J.A.; Szeto, D.; Yao, X.; Forrest, G.; et al. Butyrate and propionate protect against diet-induced obesity and regulate gut hormones via free fatty acid receptor 3-independent mechanisms. PLoS ONE 2012, 7, e35240. [Google Scholar] [CrossRef] [PubMed]
- Tsilingiri, K.; Rescigno, M. Postbiotics: What else? Benef. Microbes 2013, 4, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Toalá, J.E.; Garcia-Varela, R.; Garcia, H.S.; Mata-Haro, V.; González-Córdova, A.F.; Vallejo-Cordoba, B.; Hernández-Mendoza, A. Postbiotics: An evolving term within the functional foods field. Trends Food Sci. Technol. 2018, 75, 105–114. [Google Scholar] [CrossRef]
- De Vadder, F.; Kovatcheva-Datchary, P.; Goncalves, D.; Vinera, J.; Zitoun, C.; Duchampt, A.; Bäckhed, F.; Mithieux, G. Microbiota-generated metabolites promote metabolic benefits via gut-brain neural circuits. Cell 2014, 156, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Shirouchi, B.; Nagao, K.; Umegatani, M.; Shiraishi, A.; Morita, Y.; Kai, S.; Yanagita, T.; Ogawa, A.; Kadooka, Y.; Sato, M. Probiotic Lactobacillus gasseri SBT2055 improves glucose tolerance and reduces body weight gain in rats by stimulating energy expenditure. Br. J. Nutr. 2016, 116, 451–458. [Google Scholar] [CrossRef]
- Breton, J.; Tennoune, N.; Lucas, N.; Francois, M.; Legrand, R.; Jacquemot, J.; Goichon, A.; Guérin, C.; Peltier, J.; Pestel-Caron, M.; et al. Gut commensal E. coli proteins activate host satiety pathways following nutrient-induced bacterial growth. Cell Metab. 2016, 23, 324–334. [Google Scholar] [CrossRef] [PubMed]
- Suez, J.; Zmora, N.; Segal, E.; Elinav, E. The pros, cons, and many unknowns of probiotics. Nat. Med. 2019, 25, 716–729. [Google Scholar] [CrossRef]
- Canfora, E.E.; Meex, R.C.R.; Venema, K.; Blaak, E.E. Gut microbial metabolites in obesity, NAFLD and T2DM. Nat. Rev. Endocrinol. 2019, 15, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Vallianou, N.; Stratigou, T.; Christodoulatos, G.S.; Tsigalou, C.; Dalamaga, M. Probiotics, Prebiotics, Synbiotics, Postbiotics, and Obesity: Current Evidence, Controversies, and Perspectives. Curr. Obes. Rep. 2020, 9, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Stohs, S.J.; Miller, H.; Kaats, G.R. A review of the efficacy and safety of banaba (Lagerstroemia speciosa L.) and corosolic acid. Phyther. Res. 2012, 26, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Miura, T.; Itoh, Y.; Kaneko, T.; Ueda, N.; Ishida, T.; Fukushima, M.; Matsuyama, F.; Seino, Y. Corosolic acid induces GLUT4 translocation in genetically type 2 diabetic mice. Biol. Pharm. Bull. 2004, 27, 1103–1105. [Google Scholar] [CrossRef]
- Miura, T.; Ueda, N.; Yamada, K.; Fukushima, M.; Ishida, T.; Kaneko, T.; Matsuyama, F.; Seino, Y. Antidiabetic effects of corosolic acid in KK-Ay diabetic mice. Biol. Pharm. Bull. 2006, 29, 585–587. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pochapski, M.T.; Fosquiera, E.C.; Esmerino, L.A.; Dos Santos, E.B.; Farago, P.V.; Santos, F.A.; Groppo, F.C. Phytochemical screening, antioxidant, and antimicrobial activities of the crude leaves′ extract from Ipomoea batatas (L.) Lam. Pharmacogn. Mag. 2011, 7, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Kusano, S.; Abe, H. Antidiabetic activity of white skinned sweet potato (Ipomoea batatas L.) in obese Zucker fatty rats. Biol. Pharm. Bull 2000, 23, 23–26. [Google Scholar] [CrossRef]
- Zhao, R.; Li, Q.; Long, L.; Li, J.; Yang, R.; Gao, D. Antidiabetic activity of flavone from Ipomoea Batatas leaf in non-insulin dependent diabetic rats. Int. J. Food Sci. Technol. 2007, 42, 80–85. [Google Scholar] [CrossRef]
- Prasad, K. Secoisolariciresinol diglucoside from flaxseed delays the development of type 2 diabetes in Zucker rat. J. Lab. Clin. Med. 2001, 138, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Parikh, M.; Maddaford, T.G.; Austria, J.A.; Aliani, M.; Netticadan, T.; Pierce, G.N. Dietary Flaxseed as a Strategy for Improving Human Health. Nutrients 2019, 11, 1171. [Google Scholar] [CrossRef] [PubMed]
- MacDonald-Wicks, L.K.; Garg, M.L. Modulation of carbon tetrachloride-induced oxidative stress by dietary fat in rats(open star). J. Nutr. Biochem. 2002, 13, 87–95. [Google Scholar] [CrossRef]
- Fukumitsu, S.; Aida, K.; Ueno, N.; Ozawa, S.; Takahashi, Y.; Kobori, M. Flaxseed lignan attenuates high-fat diet-induced fat accumulation and induces adiponectin expression in mice. Br. J. Nutr. 2008, 100, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Fofana, B.; Roy, M.; Ghose, K.; Yao, X.H.; Nixon, M.S.; Nair, S.; Nyomba, G.B.L. Flaxseed lignan secoisolariciresinol diglucoside improves insulin sensitivity through upregulation of GLUT4 expression in diet-induced obese mice. J. Funct. Foods 2015, 18, 1–9. [Google Scholar] [CrossRef]
| Required |
|---|
|
| Not Recommended |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calcaterra, V.; Verduci, E.; Cena, H.; Magenes, V.C.; Todisco, C.F.; Tenuta, E.; Gregorio, C.; De Giuseppe, R.; Bosetti, A.; Di Profio, E.; et al. Polycystic Ovary Syndrome in Insulin-Resistant Adolescents with Obesity: The Role of Nutrition Therapy and Food Supplements as a Strategy to Protect Fertility. Nutrients 2021, 13, 1848. https://doi.org/10.3390/nu13061848
Calcaterra V, Verduci E, Cena H, Magenes VC, Todisco CF, Tenuta E, Gregorio C, De Giuseppe R, Bosetti A, Di Profio E, et al. Polycystic Ovary Syndrome in Insulin-Resistant Adolescents with Obesity: The Role of Nutrition Therapy and Food Supplements as a Strategy to Protect Fertility. Nutrients. 2021; 13(6):1848. https://doi.org/10.3390/nu13061848
Chicago/Turabian StyleCalcaterra, Valeria, Elvira Verduci, Hellas Cena, Vittoria Carlotta Magenes, Carolina Federica Todisco, Elisavietta Tenuta, Cristina Gregorio, Rachele De Giuseppe, Alessandra Bosetti, Elisabetta Di Profio, and et al. 2021. "Polycystic Ovary Syndrome in Insulin-Resistant Adolescents with Obesity: The Role of Nutrition Therapy and Food Supplements as a Strategy to Protect Fertility" Nutrients 13, no. 6: 1848. https://doi.org/10.3390/nu13061848
APA StyleCalcaterra, V., Verduci, E., Cena, H., Magenes, V. C., Todisco, C. F., Tenuta, E., Gregorio, C., De Giuseppe, R., Bosetti, A., Di Profio, E., & Zuccotti, G. (2021). Polycystic Ovary Syndrome in Insulin-Resistant Adolescents with Obesity: The Role of Nutrition Therapy and Food Supplements as a Strategy to Protect Fertility. Nutrients, 13(6), 1848. https://doi.org/10.3390/nu13061848











