Baseline Levels of Vitamin D in a Healthy Population from a Region with High Solar Irradiation
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Size and Representativeness
2.2. Study Design
2.3. Measurement of Blood 25(OH)D Levels
2.4. Statistical Analysis
3. Results
3.1. Study Population
3.2. Levels and Distribution of 25(OH)D, Calcium and Phosphate by Gender, Age, Place of Residence and BMI in the Healthy Population
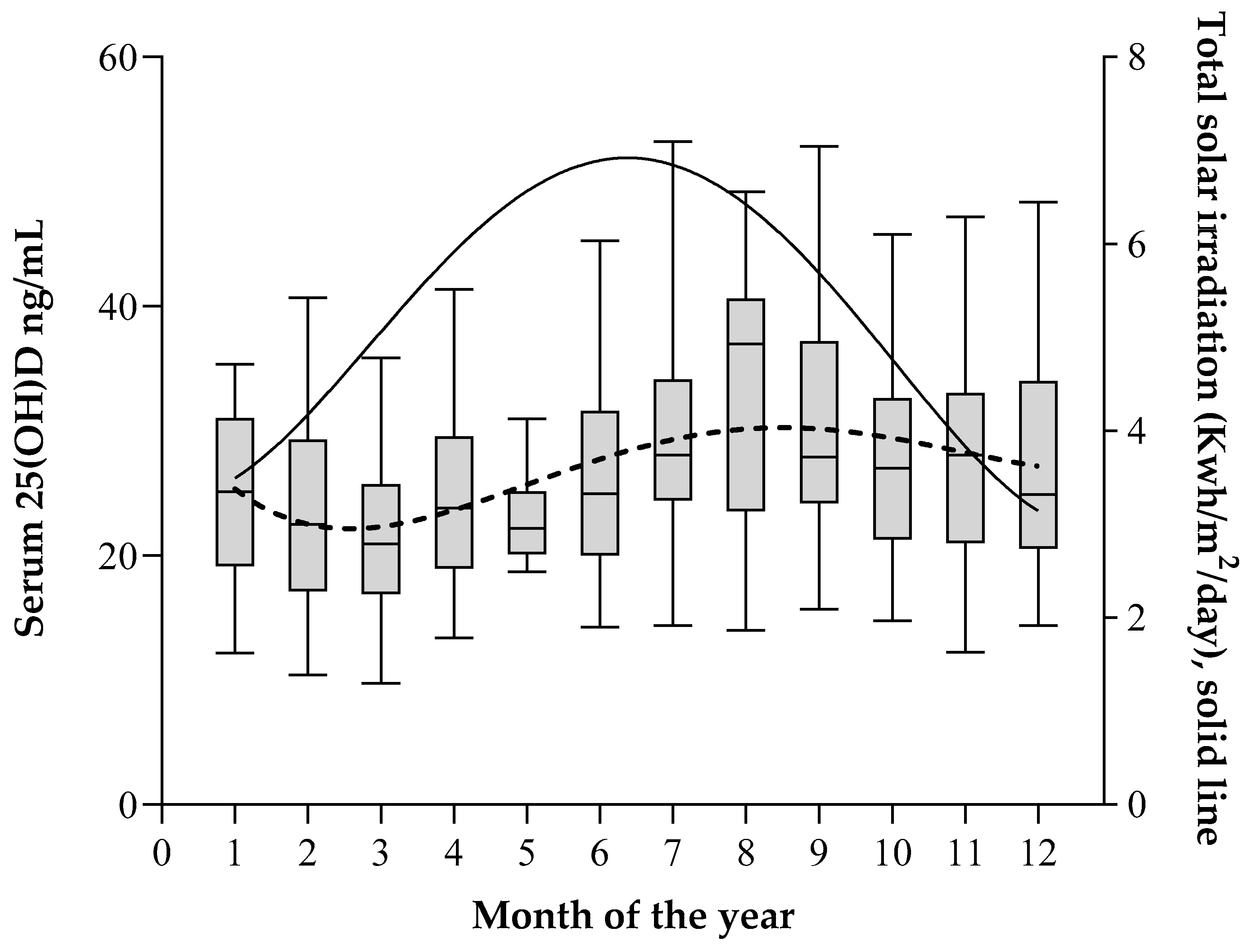
3.3. Seasonal Variation in Serum 25(OH)D
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jones, G. Vitamin D. In Modern Nutrition in Health and Disease, 11th ed.; Ross, A.C., Caballero, B., Cousins, R.J., Tucker, K.L., Ziegler, T.R., Eds.; Lippincott Williams and Wilkins: Philadelphia, PA, USA, 2014. [Google Scholar]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Engelsen, O. The relationship between ultraviolet radiation exposure and vitamin D status. Nutrients 2010, 2, 482–495. [Google Scholar] [CrossRef] [PubMed]
- Calvo, M.S.; Whiting, S.J.; Barton, C.N. Vitamin D fortification in the United States and Canada: Current status and data needs. Am. J. Clin. Nutr. 2004, 80, 1710S–1716S. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D: A d-lightful solution for health. J. Investig. Med. 2011, 59, 872–880. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Environmental factors that influence the cutaneous production of vitamin D. Am. J. Clin. Nutr. 1995, 61, 638S–645S. [Google Scholar] [CrossRef]
- Webb, A.R. Who, what, where and when-influences on cutaneous vitamin D synthesis. Prog. Biophys. Mol. Biol. 2006, 92, 17–25. [Google Scholar] [CrossRef]
- Webb, A.R.; Engelsen, O. Ultraviolet exposure scenarios: Risks of erythema from recommendations on cutaneous vitamin D synthesis. Adv. Exp. Med. Biol. 2008, 624, 72–85. [Google Scholar] [CrossRef]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M.; Endocrine, S. Evaluation, treatment, and prevention of vitamin D deficiency: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef]
- Giustina, A.; Adler, R.A.; Binkley, N.; Bouillon, R.; Ebeling, P.R.; Lazaretti-Castro, M.; Marcocci, C.; Rizzoli, R.; Sempos, C.T.; Bilezikian, J.P. Controversies in vitamin D: Summary statement from an international conference. J. Clin. Endocrinol. Metab. 2019, 104, 234–240. [Google Scholar] [CrossRef]
- Trajanoska, K.; Morris, J.A.; Oei, L.; Zheng, H.F.; Evans, D.M.; Kiel, D.P.; Ohlsson, C.; Richards, J.B.; Rivadeneira, F.; Consortium, G.G.; et al. Assessment of the genetic and clinical determinants of fracture risk: Genome wide association and mendelian randomisation study. BMJ 2018, 362, k3225. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.G.; Zeng, X.T.; Wang, J.; Liu, L. Association between calcium or vitamin D supplementation and fracture incidence in community-dwelling older adults: A systematic review and meta-analysis. JAMA 2017, 318, 2466–2482. [Google Scholar] [CrossRef]
- Khaw, K.T.; Stewart, A.W.; Waayer, D.; Lawes, C.M.M.; Toop, L.; Camargo, C.A., Jr.; Scragg, R. Effect of monthly high-dose vitamin D supplementation on falls and non-vertebral fractures: Secondary and post-hoc outcomes from the randomised, double-blind, placebo-controlled ViDA trial. Lancet Diabetes Endocrinol. 2017, 5, 438–447. [Google Scholar] [CrossRef]
- LeBoff, M.S.; Chou, S.H.; Murata, E.M.; Donlon, C.M.; Cook, N.R.; Mora, S.; Lee, I.M.; Kotler, G.; Bubes, V.; Buring, J.E.; et al. Effects of supplemental vitamin D on bone health outcomes in women and men in the VITamin D and OmegA-3 TriaL (VITAL). J. Bone Miner. Res. 2020, 35, 883–893. [Google Scholar] [CrossRef]
- Medicine, I.O. Dietary Reference Intakes for Calcium and Vitamin D; The National Academies Press: Washington, DC, USA, 2011; p. 1132. [Google Scholar]
- Souberbielle, J.C.; Massart, C.; Brailly-Tabard, S.; Cavalier, E.; Chanson, P. Prevalence and determinants of vitamin D deficiency in healthy French adults: The VARIETE study. Endocrine 2016, 53, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.C.; Manson, J.E.; Abrams, S.A.; Aloia, J.F.; Brannon, P.M.; Clinton, S.K.; Durazo-Arvizu, R.A.; Gallagher, J.C.; Gallo, R.L.; Jones, G.; et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the institute of medicine: What clinicians need to know. J. Clin. Endocrinol. Metab. 2011, 96, 53–58. [Google Scholar] [CrossRef]
- Anglin, R.E.; Samaan, Z.; Walter, S.D.; McDonald, S.D. Vitamin D deficiency and depression in adults: Systematic review and meta-analysis. Br. J. Psychiatry 2013, 202, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Feldman, D.; Krishnan, A.V.; Swami, S.; Giovannucci, E.; Feldman, B.J. The role of vitamin D in reducing cancer risk and progression. Nat. Rev. Cancer 2014, 14, 342–357. [Google Scholar] [CrossRef] [PubMed]
- McCullough, M.L.; Zoltick, E.S.; Weinstein, S.J.; Fedirko, V.; Wang, M.; Cook, N.R.; Eliassen, A.H.; Zeleniuch-Jacquotte, A.; Agnoli, C.; Albanes, D.; et al. Circulating vitamin D and colorectal cancer risk: An international pooling project of 17 cohorts. J. Natl. Cancer Inst. 2019, 111, 158–169. [Google Scholar] [CrossRef] [PubMed]
- Ginde, A.A.; Mansbach, J.M.; Camargo, C.A., Jr. Association between serum 25-hydroxyvitamin D level and upper respiratory tract infection in the Third National Health and Nutrition Examination Survey. Arch. Intern. Med. 2009, 169, 384–390. [Google Scholar] [CrossRef]
- Jovanovich, A.J.; Ginde, A.A.; Holmen, J.; Jablonski, K.; Allyn, R.L.; Kendrick, J.; Chonchol, M. Vitamin D level and risk of community-acquired pneumonia and sepsis. Nutrients 2014, 6, 2196–2205. [Google Scholar] [CrossRef]
- Zhang, R.; Li, B.; Gao, X.; Tian, R.; Pan, Y.; Jiang, Y.; Gu, H.; Wang, Y.; Wang, Y.; Liu, G. Serum 25-hydroxyvitamin D and the risk of cardiovascular disease: Dose-response meta-analysis of prospective studies. Am. J. Clin. Nutr. 2017, 105, 810–819. [Google Scholar] [CrossRef] [PubMed]
- Gowda, U.; Mutowo, M.P.; Smith, B.J.; Wluka, A.E.; Renzaho, A.M. Vitamin D supplementation to reduce depression in adults: Meta-analysis of randomized controlled trials. Nutrition 2015, 31, 421–429. [Google Scholar] [CrossRef]
- Manson, J.E.; Cook, N.R.; Lee, I.M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Gordon, D.; Copeland, T.; D’Agostino, D.; et al. Vitamin D supplements and prevention of cancer and cardiovascular disease. N. Engl. J. Med. 2019, 380, 33–44. [Google Scholar] [CrossRef]
- Ng, K.; Nimeiri, H.S.; McCleary, N.J.; Abrams, T.A.; Yurgelun, M.B.; Cleary, J.M.; Rubinson, D.A.; Schrag, D.; Miksad, R.; Bullock, A.J.; et al. Effect of high-dose vs standard-dose vitamin D3 supplementation on progression-free survival among patients with advanced or metastatic colorectal cancer: The SUNSHINE randomized clinical trial. JAMA 2019, 321, 1370–1379. [Google Scholar] [CrossRef]
- Urashima, M.; Ohdaira, H.; Akutsu, T.; Okada, S.; Yoshida, M.; Kitajima, M.; Suzuki, Y. Effect of vitamin D supplementation on relapse-free survival among patients with digestive tract cancers: The AMATERASU randomized clinical trial. JAMA 2019, 321, 1361–1369. [Google Scholar] [CrossRef]
- Langlois, P.L.; Szwec, C.; D’Aragon, F.; Heyland, D.K.; Manzanares, W. Vitamin D supplementation in the critically ill: A systematic review and meta-analysis. Clin. Nutr. 2018, 37, 1238–1246. [Google Scholar] [CrossRef]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef]
- Torjesen, I. Evidence does not support vitamin D for reducing respiratory infections, reviews conclude. BMJ 2020, 369, m2629. [Google Scholar] [CrossRef]
- Pathak, K.; Soares, M.J.; Calton, E.K.; Zhao, Y.; Hallett, J. Vitamin D supplementation and body weight status: A systematic review and meta-analysis of randomized controlled trials. Obes. Rev. 2014, 15, 528–537. [Google Scholar] [CrossRef] [PubMed]
- Pittas, A.G.; Dawson-Hughes, B.; Sheehan, P.; Ware, J.H.; Knowler, W.C.; Aroda, V.R.; Brodsky, I.; Ceglia, L.; Chadha, C.; Chatterjee, R.; et al. Vitamin D Supplementation and Prevention of Type 2 Diabetes. N. Engl. J. Med. 2019, 381, 520–530. [Google Scholar] [CrossRef]
- Lagunova, Z.; Porojnicu, A.C.; Lindberg, F.; Hexeberg, S.; Moan, J. The dependency of vitamin D status on body mass index, gender, age and season. Anticancer Res. 2009, 29, 3713–3720. [Google Scholar] [CrossRef] [PubMed]
- Jorde, R.; Sneve, M.; Emaus, N.; Figenschau, Y.; Grimnes, G. Cross-sectional and longitudinal relation between serum 25-hydroxyvitamin D and body mass index: The Tromso study. Eur. J. Nutr. 2010, 49, 401–407. [Google Scholar] [CrossRef] [PubMed]
- ISTAC: Estadísticas de la Comunidad Autónoma de Canarias. 2020. Available online: http://www.gobiernodecanarias.org/istac/estadisticas/demografia/ (accessed on 5 April 2021).
- Sancho Ávila, J.M.; Jesús Riesco Martín, J.; Jiménez Alonso, C.; Sánchez de Cos Escuin, M.C.; Montero Cadalso, J.; López Bartolomé, M. Atlas de Radiación Solar en España Utilizando Datos del SAF de Clima de EUMETSAT; AEMET: Madrid, Spain, 2012; Available online: http://www.aemet.es/documentos/es/serviciosclimaticos/datosclimatologicos/atlas_radiacion_solar/atlas_de_radiacion_24042012.pdf (accessed on 5 April 2021).
- Woodford, H.J.; Barrett, S.; Pattman, S. Vitamin D: Too much testing and treating? Clin. Med. 2018, 18, 196–200. [Google Scholar] [CrossRef]
- Sanders, K.M.; Stuart, A.L.; Williamson, E.J.; Simpson, J.A.; Kotowicz, M.A.; Young, D.; Nicholson, G.C. Annual high-dose oral vitamin D and falls and fractures in older women: A randomized controlled trial. JAMA 2010, 303, 1815–1822. [Google Scholar] [CrossRef] [PubMed]
- Korgavkar, K.; Xiong, M.; Weinstock, M.A. Review: Higher vitamin D status and supplementation may be associated with risks. Eur. J. Dermatol. 2014, 24, 428–434. [Google Scholar] [CrossRef]
- Galior, K.; Grebe, S.; Singh, R. Development of vitamin D yoxicity from overcorrection of vitamin D deficiency: A review of case reports. Nutrients 2018, 10, 953. [Google Scholar] [CrossRef]
- Jiang, W.; Wu, D.B.; Xiao, G.B.; Ding, B.; Chen, E.Q. An epidemiology survey of vitamin D deficiency and its influencing factors. Med. Clin. 2020, 154, 7–12. [Google Scholar] [CrossRef]
- Flores, C.; Maca-Meyer, N.; Perez, J.A.; Gonzalez, A.M.; Larruga, J.M.; Cabrera, V.M. A predominant European ancestry of paternal lineages from Canary Islanders. Ann. Hum. Genet. 2003, 67, 138–152. [Google Scholar] [CrossRef]
- Terushkin, V.; Bender, A.; Psaty, E.L.; Engelsen, O.; Wang, S.Q.; Halpern, A.C. Estimated equivalency of vitamin D production from natural sun exposure versus oral vitamin D supplementation across seasons at two US latitudes. J. Am. Acad. Dermatol. 2010, 62, e921–e929. [Google Scholar] [CrossRef] [PubMed]
- Petersen, B.; Wulf, H.C.; Triguero-Mas, M.; Philipsen, P.A.; Thieden, E.; Olsen, P.; Heydenreich, J.; Dadvand, P.; Basagana, X.; Liljendahl, T.S.; et al. Sun and ski holidays improve vitamin D status, but are associated with high levels of DNA damage. J. Investig. Dermatol. 2014, 134, 2806–2813. [Google Scholar] [CrossRef] [PubMed]
- Young, A.R.; Narbutt, J.; Harrison, G.I.; Lawrence, K.P.; Bell, M.; O’Connor, C.; Olsen, P.; Grys, K.; Baczynska, K.A.; Rogowski-Tylman, M.; et al. Optimal sunscreen use, during a sun holiday with a very high ultraviolet index, allows vitamin D synthesis without sunburn. Br. J. Dermatol. 2019, 181, 1052–1062. [Google Scholar] [CrossRef]
- Klingberg, E.; Olerod, G.; Konar, J.; Petzold, M.; Hammarsten, O. Seasonal variations in serum 25-hydroxy vitamin D levels in a Swedish cohort. Endocrine 2015, 49, 800–808. [Google Scholar] [CrossRef] [PubMed]
- Giustina, A.; Bouillon, R.; Binkley, N.; Sempos, C.; Adler, R.A.; Bollerslev, J.; Dawson-Hughes, B.; Ebeling, P.R.; Feldman, D.; Heijboer, A.; et al. Controversies in vitamin D: A statement from the third international conference. JBMR Plus 2020, 4. [Google Scholar] [CrossRef] [PubMed]
- WHO Scientific Group on the Prevention and Management of Osteoporosis. Prevention and Management of Osteoporosis: Report of a WHO Scientific Group; World Health Organization: Geneva, Switzerland, 2003; Available online: https://apps.who.int/iris/handle/10665/42841 (accessed on 5 April 2021).
- Chapuy, M.C.; Preziosi, P.; Maamer, M.; Arnaud, S.; Galan, P.; Hercberg, S.; Meunier, P.J. Prevalence of vitamin D insufficiency in an adult normal population. Osteoporos. Int. 1997, 7, 439–443. [Google Scholar] [CrossRef] [PubMed]
- Steingrimsdottir, L.; Gunnarsson, O.; Indridason, O.S.; Franzson, L.; Sigurdsson, G. Relationship between serum parathyroid hormone levels, vitamin D sufficiency, and calcium intake. JAMA 2005, 294, 2336–2341. [Google Scholar] [CrossRef]
- Heaney, R.P. The Vitamin D requirement in health and disease. J. Steroid Biochem. Mol. Biol. 2005, 97, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Wills, K.; Laslett, L.L.; Oldenburg, B.; Seibel, M.J.; Jones, G.; Winzenberg, T. Cut-points for associations between vitamin D status and multiple musculoskeletal outcomes in middle-aged women. Osteoporos. Int. 2017, 28, 505–515. [Google Scholar] [CrossRef] [PubMed]
- LeBoff, M.S.; Murata, E.M.; Cook, N.R.; Cawthon, P.; Chou, S.H.; Kotler, G.; Bubes, V.; Buring, J.E.; Manson, J.E. VITamin D and OmegA-3 TriaL (VITAL): Effects of Vitamin D Supplements on Risk of Falls in the US Population. J. Clin. Endocrinol. Metab. 2020, 105. [Google Scholar] [CrossRef]
- Scragg, R.; Waayer, D.; Stewart, A.W.; Lawes, C.M.M.; Toop, L.; Murphy, J.; Khaw, K.T.; Camargo, C.A., Jr. The Vitamin D Assessment (ViDA) Study: Design of a randomized controlled trial of vitamin D supplementation for the prevention of cardiovascular disease, acute respiratory infection, falls and non-vertebral fractures. J. Steroid Biochem. Mol. Biol. 2016, 164, 318–325. [Google Scholar] [CrossRef]
- Manson, J.E.; Brannon, P.M.; Rosen, C.J.; Taylor, C.L. Vitamin D deficiency—Is there really a pandemic? N. Engl. J. Med. 2016, 375, 1817–1820. [Google Scholar] [CrossRef]
- Pittas, A.; Dawson-Hughes, B.; Staten, M. Vitamin D supplementation and prevention of type 2 diabetes. Reply. N. Engl. J. Med. 2019, 381, 1785–1786. [Google Scholar] [CrossRef]
- Manios, Y.; Moschonis, G.; Hulshof, T.; Bourhis, A.S.; Hull, G.L.J.; Dowling, K.G.; Kiely, M.E.; Cashman, K.D. Prevalence of vitamin D deficiency and insufficiency among schoolchildren in Greece: The role of sex, degree of urbanisation and seasonality. Br. J. Nutr. 2017, 118, 550–558. [Google Scholar] [CrossRef][Green Version]
- Chailurkit, L.; Aekplakorn, W.; Ongphiphadhanakul, B. Serum C3 epimer of 25-hydroxyvitamin D and its determinants in adults: A national health examination survey in Thais. Osteoporos. Int. 2015, 26, 2339–2344. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, J.C. Vitamin D and aging. Endocrinol. Metab. Clin. N. Am. 2013, 42, 319–332. [Google Scholar] [CrossRef] [PubMed]
- Farrell, C.J.; Martin, S.; McWhinney, B.; Straub, I.; Williams, P.; Herrmann, M. State-of-the-art vitamin D assays: A comparison of automated immunoassays with liquid chromatography-tandem mass spectrometry methods. Clin. Chem. 2012, 58, 531–542. [Google Scholar] [CrossRef] [PubMed]
- Volmer, D.A.; Mendes, L.R.; Stokes, C.S. Analysis of vitamin D metabolic markers by mass spectrometry: Current techniques, limitations of the “gold standard” method, and anticipated future directions. Mass. Spectrom. Rev. 2015, 34, 2–23. [Google Scholar] [CrossRef]
- Seida, J.C.; Mitri, J.; Colmers, I.N.; Majumdar, S.R.; Davidson, M.B.; Edwards, A.L.; Hanley, D.A.; Pittas, A.G.; Tjosvold, L.; Johnson, J.A. Clinical review: Effect of vitamin D3 supplementation on improving glucose homeostasis and preventing diabetes: A systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 2014, 99, 3551–3560. [Google Scholar] [CrossRef] [PubMed]

| Healthy Individuals n = 876 (92.3%) | Excluded * n = 73 (7.7%) | p | Total n = 949 | |
|---|---|---|---|---|
| Sex | <0.001 | |||
| Male | 461 (53%) | 13 (18%) | 474 (50%) | |
| Female | 415 (47%) | 60 (82%) | 475 (50%) | |
| Age (years) | 43.3 ± 15.8 | 59.1 ± 14.9 | <0.001 | 44.5 ± 16.3 |
| Age (years) | <0.001 | |||
| 20–44 | 527 (60%) | 10 (14%) | 537 (57%) | |
| 45–64 | 233 (27%) | 33 (45%) | 266 (28%) | |
| ≥65 | 116 (13%) | 30 (41%) | 146 (15%) | |
| Area | 0.478 | |||
| Rural | 134 (15%) | 14 (19%) | 148 (16%) | |
| Urban | 742 (85%) | 59 (81%) | 801 (84%) | |
| BMI (kg/m2) | 26.74 ± 4.67 | 27.79 ± 4.76 | 0.070 | 26.82 ± 4.69 |
| 25(OH)D (ng/mL) | 26.30 (14.30; 45.84) | 23.70 (11.50; 47.7) | 0.351 | 26.20 (14.20; 45.8) |
| Phosphate (mg/dL) | 3.50 (2.54; 5.25) | 3.48 (2.35; 4.65) | 0.425 | 3.50 (2.53; 5.19) |
| Calcium (mg/dL) | 9.34 (8.15; 10.28) | 9.50 (8.13; 11.12) | 0.072 | 9.35 (8.15; 10.31) |
| Male n = 461 | Female n = 415 | p | Age (Years) | p | Total n = 876 | |||
|---|---|---|---|---|---|---|---|---|
| 20–44 n = 527 | 45–64 n = 233 | ≥65 n = 116 | ||||||
| Sex | 0.558 | |||||||
| Male | 271 (51%) | 130 (56%) | 60 (52%) | 461 (53%) | ||||
| Female | 256 (49%) | 103 (44%) | 56 (48%) | 415 (47%) | ||||
| Age (years) | 43.9 ± 15.9 | 42.7 ± 15.7 | 0.259 | 43.3 ± 15.8 | ||||
| Age (years) | 0.558 | |||||||
| 20–44 | 271 (59%) | 256 (62%) | 527 (60%) | |||||
| 45–64 | 130 (28%) | 103 (25%) | 233 (27%) | |||||
| ≥65 | 60 (13%) | 56 (13%) | 146 (13%) | |||||
| Area | 0.854 | 0.459 | ||||||
| Rural | 72 (16%) | 62 (15%) | 74 (14%) | 39 (16%) | 21 (18%) | 134 (15%) | ||
| Urban | 389 (84%) | 353 (85%) | 453 (86%) | 194 (84%) | 95 (82%) | 742 (85%) | ||
| BMI (kg/m2) | 26.92 ± 4.13 | 26.53 ± 5.21 | 0.235 | 25.42 ± 4.39 | 28.68 ± 4.62 | 28.14 ± 4.11 | <0.001 | 26.74 ± 4.67 |
| 25(OH)D (ng/mL) | 27.4 (15.8; 46.7) | 25.0 (12.8; 45.3) | <0.001 | 27.4 (15.6; 48.1) | 25.0 (13.4; 43.7) | 23.0 (12.4; 36.6) | <0.001 | 26.3 (14.3; 45.8) |
| Phosphate (mg/dL) | 3.48 (2.4; 4.8) | 3.51 (2.6; 6) | 0.180 | 3.58 (2.6; 5.7) | 3.46 (2.5; 5.) | 3.23 (2.5; 4.8) | 0.001 | 3.5 (2.5; 5.2) |
| Calcium (mg/dL) | 9.35 (8.; 10.3) | 9.32 (8.2; 10.2) | 0.584 | 9.36 (8.2; 10.2) | 9.30 (8.1; 10.4) | 9.19 (7.9; 10.3) | 0.031 | 9.3 (8.1; 10.2) |
| Parameter | Coefficient | SE | Student’s t | p | 95% CI |
|---|---|---|---|---|---|
| Intersection | 33.607 | 2.326 | 14.447 | <0.001 | (29.041; 38.174) |
| Age (ref ≥ 65) | |||||
| 20–44 | 4.372 | 1.045 | 4.183 | <0.001 | (2.320; 6.424) |
| 45–64 | 2.483 | 1.056 | 2.350 | 0.019 | (0.409; 4.557) |
| Female sex | −2.689 | 0.719 | −3.742 | <0.001 | (−4.099; −1.278) |
| Rural area | −1.438 | 1.166 | −1.234 | 0.218 | (−3.727; 0.850) |
| BMI (kg/m2) | −0.298 | 0.075 | −3.969 | <0.001 | (−0.446; −0.151) |
| Baseline 25(OH)D (ng/mL) | Male (Age, Years) | Female (Age, Years) | Gender | Age (Years) | Total | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 20–44 * | 45–64 | ≥65 | 20–44 * | 45–64 | ≥65 | Male | Female | 20–44 * | 45–64 | ≥65 | ||
| 12 | 0.3% | 1.3% | 0.8% | 2.1% | 3.2% | 7.4% | 0.6% | 3.2% | 1.0% | 2.1% | 3.6% | 1.6% |
| 13 | 0.6% | 2.3% | 1.6% | 3.4% | 5.2% | 11.2% | 1.1% | 5.0% | 1.7% | 3.5% | 5.8% | 2.7% |
| 14 | 1.2% | 3.7% | 2.9% | 5.2% | 7.9% | 15.8% | 2.0% | 7.3% | 2.9% | 5.5% | 8.8% | 4.3% |
| 15 | 2.0% | 5.6% | 4.7% | 7.4% | 11.2% | 21.1% | 3.3% | 10.2% | 4.4% | 8.0% | 12.4% | 6.4% |
| 16 | 3.2% | 8.1% | 7.2% | 10.0% | 15.0% | 26.8% | 5.0% | 13.5% | 6.4% | 11.1% | 16.5% | 8.9% |
| 17 | 4.8% | 11.1% | 10.3% | 13.1% | 19.4% | 32.9% | 7.2% | 17.2% | 8.8% | 14.7% | 21.2% | 12.0% |
| 18 | 6.9% | 14.6% | 14.0% | 16.5% | 24.1% | 39.0% | 9.9% | 21.3% | 11.6% | 18.8% | 26.3% | 15.4% |
| 19 | 9.5% | 18.4% | 18.2% | 20.2% | 29.1% | 45.1% | 13.1% | 25.6% | 14.9% | 23.2% | 31.6% | 19.3% |
| 20 | 12.5% | 22.7% | 22.9% | 24.2% | 34.3% | 51.0% | 16.7% | 30.1% | 18.5% | 27.8% | 37.0% | 23.4% |
| 21 | 16.0% | 27.2% | 28.0% | 28.3% | 39.5% | 56.5% | 20.7% | 34.6% | 22.4% | 32.7% | 42.4% | 27.7% |
| 22 | 19.8% | 31.8% | 33.2% | 32.5% | 44.6% | 61.7% | 25.0% | 39.2% | 26.5% | 37.5% | 47.7% | 32.2% |
| 23 | 23.9% | 36.6% | 38.6% | 36.8% | 49.6% | 66.5% | 29.5% | 43.7% | 30.7% | 42.4% | 52.8% | 36.7% |
| 24 | 28.2% | 41.3% | 43.9% | 41.0% | 54.4% | 70.8% | 34.1% | 48.1% | 35.1% | 47.2% | 57.6% | 41.3% |
| 25 | 32.7% | 46.0% | 49.2% | 45.2% | 58.9% | 74.7% | 38.7% | 52.3% | 39.4% | 51.8% | 62.2% | 45.7% |
| 26 | 37.3% | 50.5% | 54.2% | 49.2% | 63.1% | 78.2% | 43.4% | 56.4% | 43.7% | 56.2% | 66.4% | 50.0% |
| 27 | 41.9% | 54.9% | 59.0% | 53.2% | 67.1% | 81.3% | 47.9% | 60.2% | 47.9% | 60.4% | 70.2% | 54.2% |
| 28 | 46.4% | 59.0% | 63.5% | 56.9% | 70.7% | 83.9% | 52.3% | 63.8% | 52.0% | 64.3% | 73.7% | 58.2% |
| 29 | 50.8% | 62.9% | 67.7% | 60.4% | 74.0% | 86.3% | 56.6% | 67.1% | 55.9% | 67.9% | 76.9% | 61.9% |
| 30 | 55.0% | 66.6% | 71.5% | 63.8% | 77.0% | 88.3% | 60.6% | 70.3% | 59.7% | 71.3% | 79.8% | 65.5% |
| Quarters Months | Number of Subjects | 25(OH)D ng/mL | 25(OH)D ng/mL | 25(OH)D ≤ 15 ng/mL | 25(OH)D ≤ 20 ng/mL | 25(OH)D ≤ 30 ng/mL |
|---|---|---|---|---|---|---|
| Q1 January–March | 187 | 23.1 ± 8.4 | 22.0 (10.0; 37.0) | 23 (12.3%) | 71 (38.0%) | 147 (78.6%) |
| Q2 April–June | 275 | 27.0 ± 9.1 | 25.4 (14.0; 44.0) | 14 (5.1%) | 67 (24.4%) | 183 (66.5%) |
| Q3 July–September | 175 | 31.1 ± 10.4 | 28.1 (15.3; 52.2) | 8 (4.6%) | 17 (9.7%) | 96 (54.9%) |
| Q4 October–December | 239 | 28.8 ± 10.6 | 27.5 (15.3; 47.6) | 10 (4.2%) | 39 (22.1%) | 144 (60.3%) |
| All | 876 | 27.6 ± 10.0 | 26.3 (14.3; 45.8) | 55 (6.3%) | 194 (22.1%) | 570 (65.1%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Dorta, A.; Medina-Vega, L.; Villacampa-Jiménez, J.J.; Hernández-Díaz, M.; Bustabad-Reyes, S.; González-Dávila, E.; Díaz-González, F. Baseline Levels of Vitamin D in a Healthy Population from a Region with High Solar Irradiation. Nutrients 2021, 13, 1647. https://doi.org/10.3390/nu13051647
García-Dorta A, Medina-Vega L, Villacampa-Jiménez JJ, Hernández-Díaz M, Bustabad-Reyes S, González-Dávila E, Díaz-González F. Baseline Levels of Vitamin D in a Healthy Population from a Region with High Solar Irradiation. Nutrients. 2021; 13(5):1647. https://doi.org/10.3390/nu13051647
Chicago/Turabian StyleGarcía-Dorta, Alicia, Lillian Medina-Vega, Jacobo Javier Villacampa-Jiménez, Marta Hernández-Díaz, Sagrario Bustabad-Reyes, Enrique González-Dávila, and Federico Díaz-González. 2021. "Baseline Levels of Vitamin D in a Healthy Population from a Region with High Solar Irradiation" Nutrients 13, no. 5: 1647. https://doi.org/10.3390/nu13051647
APA StyleGarcía-Dorta, A., Medina-Vega, L., Villacampa-Jiménez, J. J., Hernández-Díaz, M., Bustabad-Reyes, S., González-Dávila, E., & Díaz-González, F. (2021). Baseline Levels of Vitamin D in a Healthy Population from a Region with High Solar Irradiation. Nutrients, 13(5), 1647. https://doi.org/10.3390/nu13051647






