Short-Term RCT of Increased Dietary Potassium from Potato or Potassium Gluconate: Effect on Blood Pressure, Microcirculation, and Potassium and Sodium Retention in Pre-Hypertensive-to-Hypertensive Adults
Abstract
1. Introduction
2. Methods
2.1. Subjects
2.2. Dietary Intervention
2.3. Blood Pressure and Microvascular Measures
2.4. Mineral Balance and Net Absorption
Statistical Methods
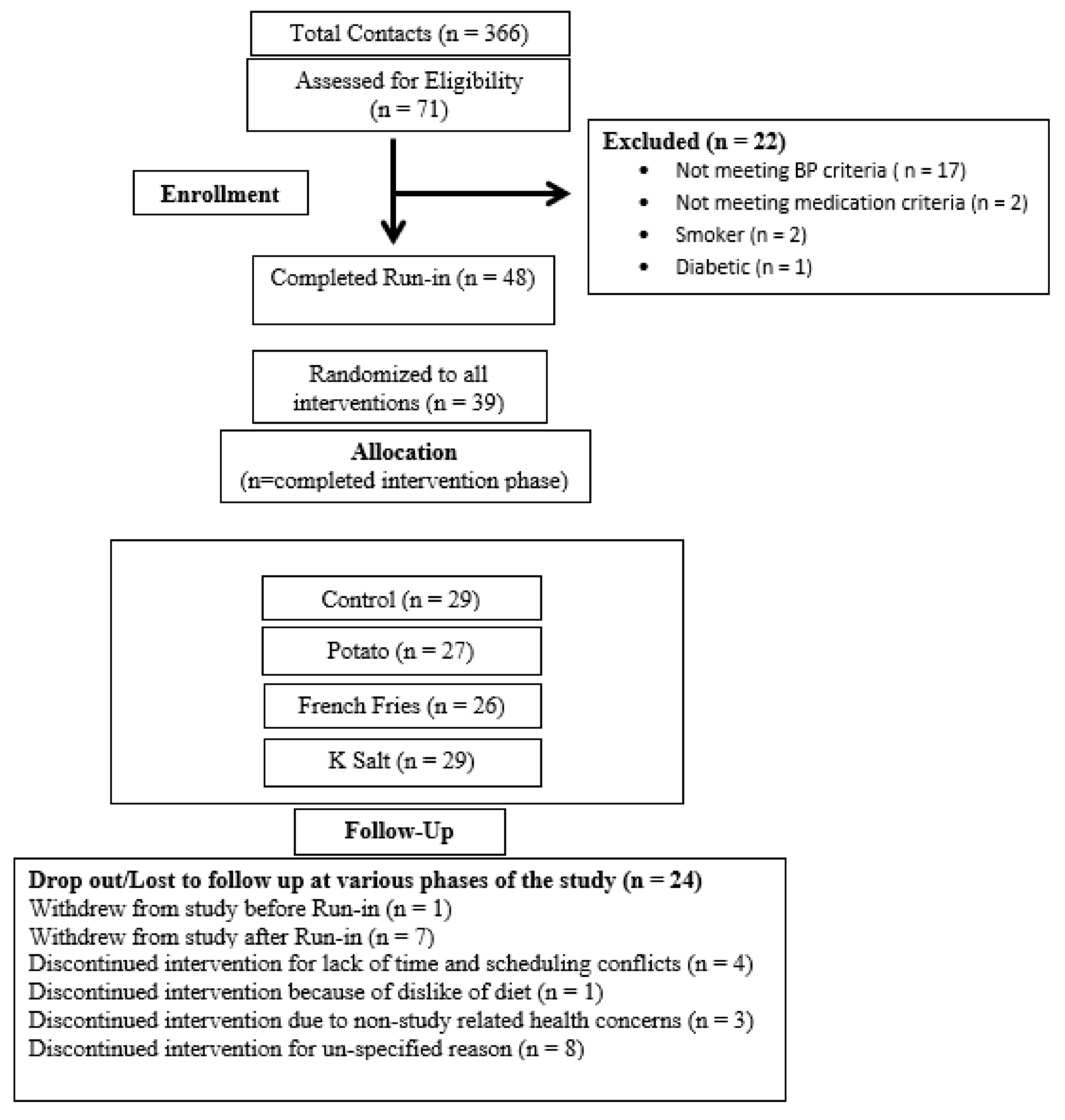
3. Results
3.1. Baseline Characteristics of Study Subjects
3.2. Subject Adherence
3.3. Chemical Analysis of Controlled Diets
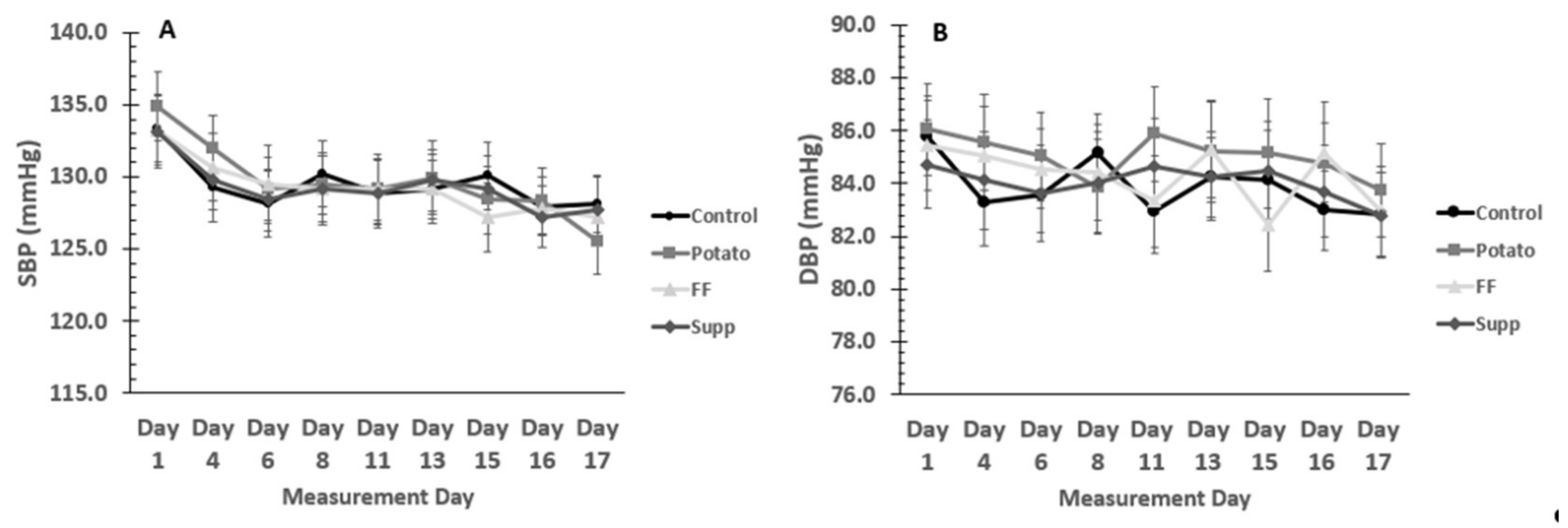
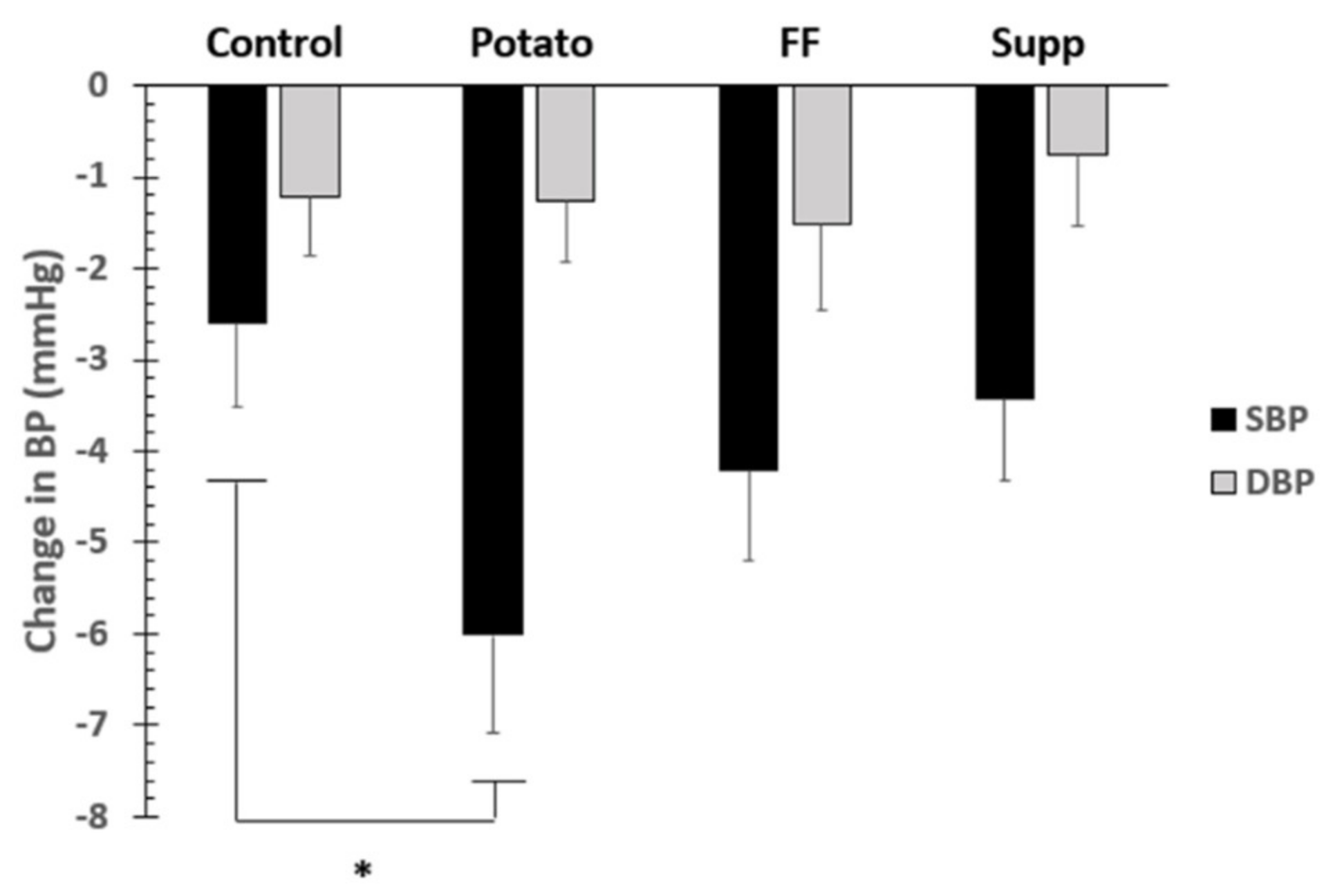
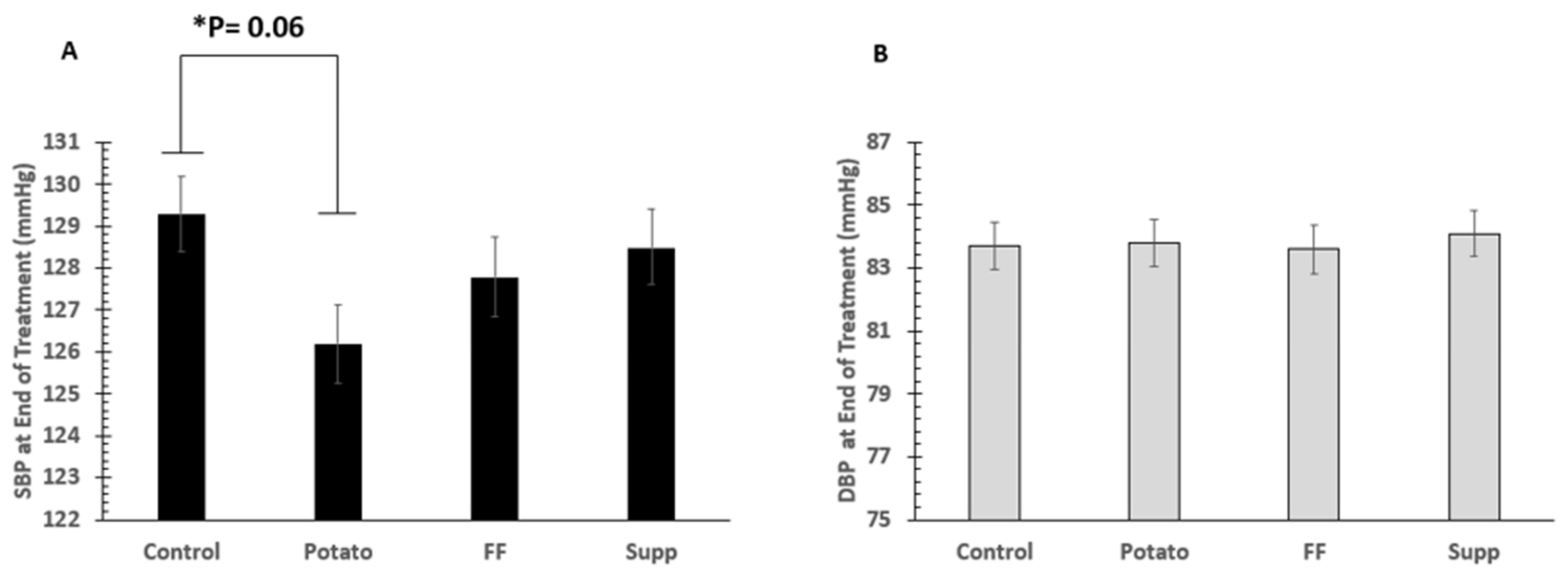
3.4. Blood Pressure
3.5. Urinary Potassium and Sodium Excretion
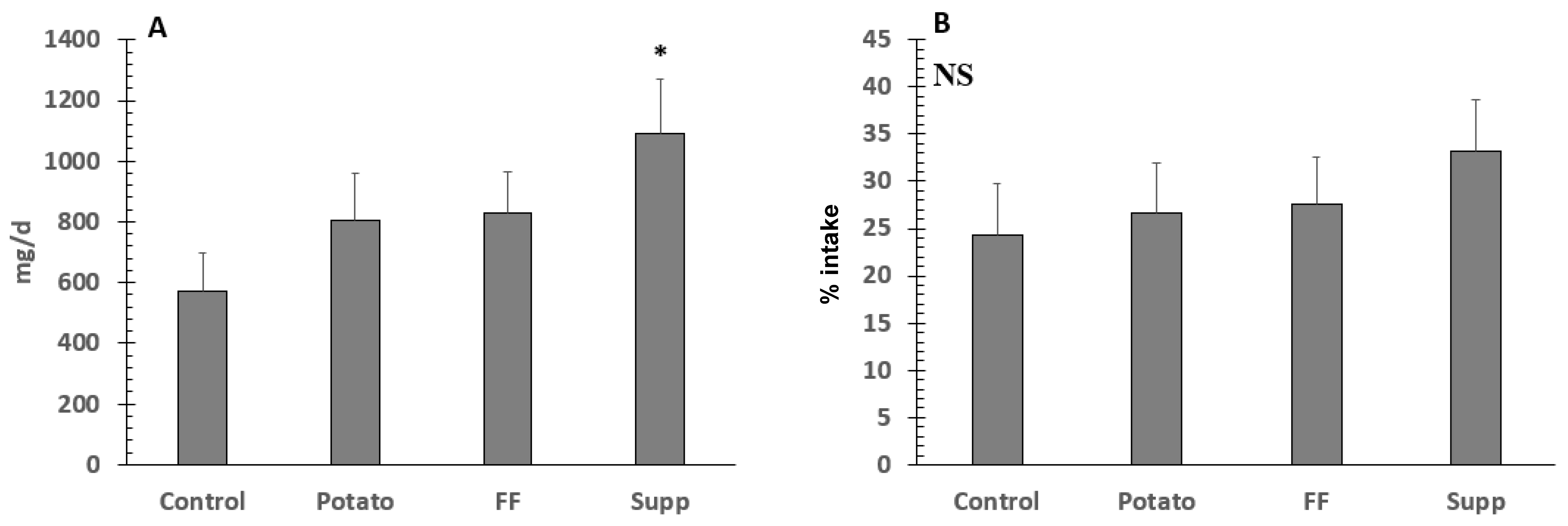
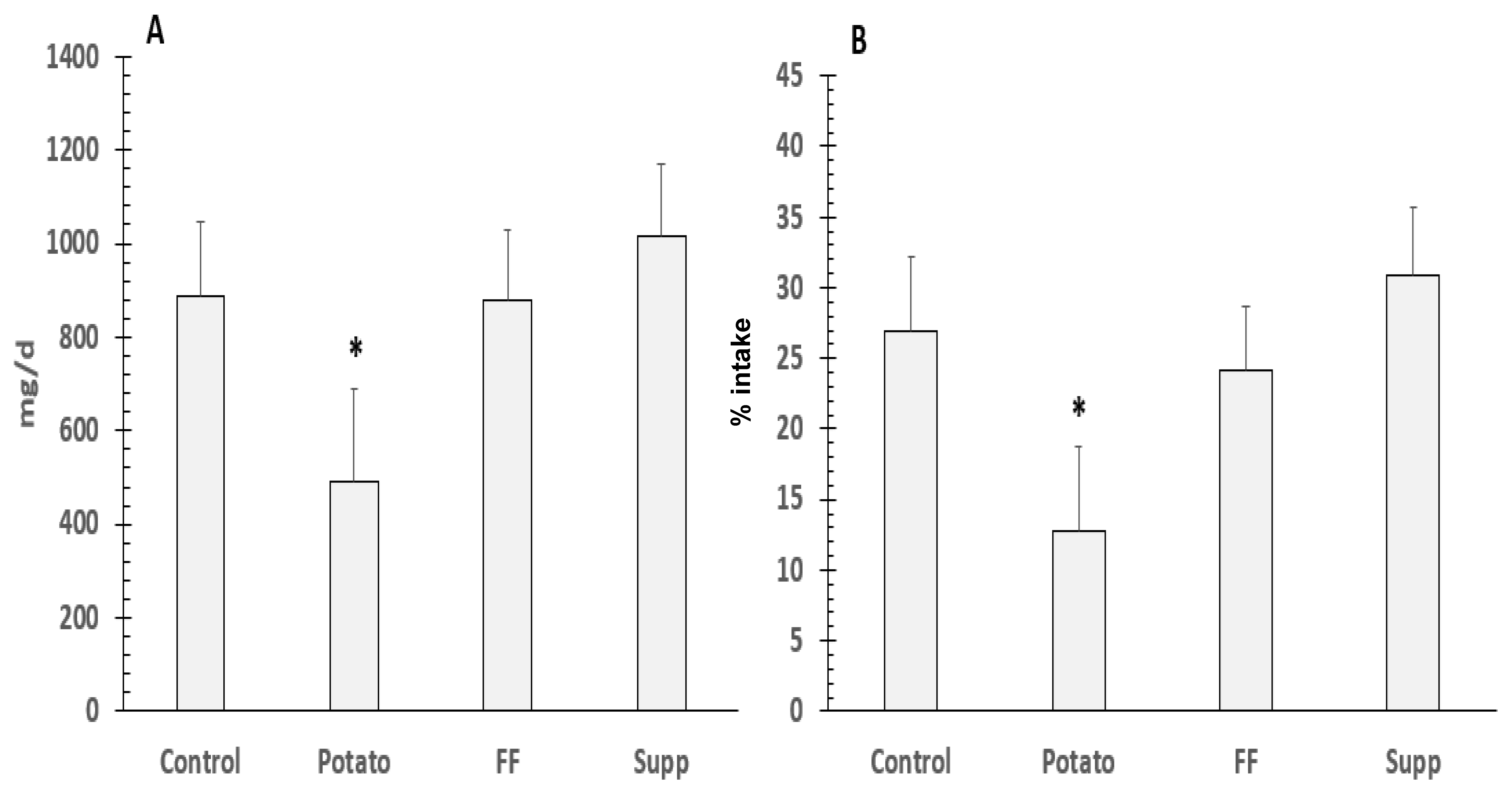
3.6. Potassium and Sodium Fecal Excretion, Balance, and % Absorption
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dietary Guidelines Advisory Committee. Report of the Dietary Guidelines Advisory Committee on the Dietary Guidelines for Americans, 2010; U.S. Department of Agriculture, Agricultural Research Service: Washington, DC, USA, 2011. [Google Scholar]
- U.S. Department of Health; Human Services; U.S. Department of Agriculture. 2015–2020 Dietary Guidelines for Americans, 8th ed.; U.S. Department of Agriculture, Agricultural Research Service: Washington, DC, USA, 2015. [Google Scholar]
- Dietary Guidelines Advisory Committee. Scientific Report of the 2020 Dietary Guidelines Advisory Report to the Secretary of Agriculture and the Secretary of Health and Human Services; U.S. Department of Agriculture, Agriculture Research Services: Washington, DC, USA, 2020. [Google Scholar]
- Roger, V.L.; Go, A.S.; Lloyd-Jones, D.M.; Benjamin, E.J.; Berry, J.D.; Borden, W.B.; Bravata, D.M.; Dai, S.; Ford, E.S.; Fox, C.S.; et al. Executive summary: Heart disease and stroke statistics—2012 update: A report from the American Heart Association. Circulation 2012, 125, 188–197. [Google Scholar] [PubMed]
- Lopez, A.D.; Mathers, C.D. Measuring the global burden of disease and epidemiological transitions: 2002–2030. Ann. Trop. Med. Parasitol. 2006, 100, 481–499. [Google Scholar] [CrossRef] [PubMed]
- Newberry, S.J.; Chung, M.; Anderson, C.A.; Chen, C.; Fu, Z.; Tang, A.; Zhao, N.; Booth, M.; Marks, J.; Hollands, S.; et al. Sodium and Potassium Intake: Effects on Chronic Disease Outcomes and Risks; Agency for Healthcare and Research Quality (US): Rockville, MD, USA, 2018. [Google Scholar]
- Aburto, N.J.; Hanson, S.; Gutierrez, H.; Hooper, L.; Elliott, P.; Cappuccio, F.P. Effect of increased potassium intake on cardiovascular risk factors and disease: Systematic review and meta-analyses. BMJ 2013, 346, f1378. [Google Scholar] [CrossRef]
- Whelton, P.K. Effects of oral potassium on blood pressure. Meta-analysis of randomized controlled clinical trials. JAMA 1997, 277, 1624–1632. [Google Scholar] [CrossRef] [PubMed]
- Cappuccio, F.P.; MacGregor, G.A. Does potassium supplementation lower blood pressure? A meta-analysis of published trials. J. Hypertens. 1991, 9, 465–473. [Google Scholar] [CrossRef]
- Geleijnse, J.M.; Kok, F.J.; Grobbee, D.E. Blood pressure response to changes in sodium and potassium intake: A metaregression analysis of randomised trials. J. Hum. Hypertens. 2003, 17, 471–480. [Google Scholar] [CrossRef]
- Binia, A.; Jaeger, J.; Hu, Y.; Singh, A.; Zimmermann, D. Daily potassium intake and sodium-to-potassium ratio in the reduction of blood pressure: A meta-analysis of randomized controlled trials. J. Hypertens. 2015, 33, 1509–1520. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, H.O.; Nicolson, D.J.; Campbell, F.; Beyer, F.R.; Mason, J. Potassium supplementation for the management of primary hypertension in adults. Cochrane Database Syst. Rev. 2006, 19, CD004641. [Google Scholar] [CrossRef]
- Chalmers, J.; Morgan, T.; Doyle, A.; Dickson, B.; Hopper, J.; Mathews, J.; Matthews, G.; Moulds, R.; Myers, J.; Nowson, C. Australian National Health and Medical Research Council dietary salt study in mild hypertension. J. Hypertens. Suppl. 1986, 4, 629–637. [Google Scholar]
- Berry, S.E.; Mulla, U.Z.; Chowienczyk, P.J.; Sanders, T.A.B. Increased potassium intake from fruit and vegetables or supplements does not lower blood pressure or improve vascular function in UK men and women with early hypertension: A randomised controlled trial. Br. J. Nutr. 2010, 104, 1839–1847. [Google Scholar] [CrossRef]
- Miller, E.R., 3rd; Cooper, L.A.; Carson, K.A.; Wang, N.Y.; Appel, L.J.; Gayles, D.; Charleston, J.; White, K.; You, N.; Weng, Y.; et al. A Dietary Intervention in Urban African Americans: Results of the “Five Plus Nuts and Beans” Randomized Trial. Am. J. Prev. Med. 2016, 50, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Appel, L.J.; Moore, T.J.; Obarzanek, E.; Vollmer, W.M.; Svetkey, L.P.; Sacks, F.M.; Bray, G.A.; Vogt, T.M.; Cutler, J.A.; Windhauser, M.M.; et al. A Clinical Trial of the Effects of Dietary Patterns on Blood Pressure. N. Engl. J. Med. 1997, 336, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Sacks, F.M.; Svetkey, L.P.; Vollmer, W.M.; Appel, L.J.; Bray, G.A.; Harsha, D.; Obarzanek, E.; Conlin, P.R.; Miller, E.R.; Simons-Morton, D.G.; et al. Effects on Blood Pressure of Reduced Dietary Sodium and the Dietary Approaches to Stop Hypertension (DASH) Diet. N. Engl. J. Med. 2001, 344, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Svetkey, L.P.; Simons-Morton, D.G.; Proschan, M.A.; Sacks, F.M.; Conlin, P.R.; Harsha, D.; Moore, T.J. DASH-Sodium Collaborative Research Group Effect of the Dietary Approaches to Stop Hypertension Diet and Reduced Sodium Intake on Blood Pressure Control. J. Clin. Hypertens. 2004, 6, 373–381. [Google Scholar] [CrossRef]
- Weaver, C.M.; Stone, M.S.; Lobene, A.J.; Cladis, D.P.; Hodges, J.K. What Is the Evidence Base for a Potassium Requirement? Nutr. Today 2018, 53, 184–195. [Google Scholar] [CrossRef]
- Hené, R.J.; Koomans, H.A.; Boer, P.; Mees, E.J.D. Adaptation to chronic potassium loading in normal man. Miner. Electrolyte Metab. 1986, 12, 165–172. [Google Scholar]
- Holbrook, J.T.; Patterson, K.Y.; Bodner, J.E.; Douglas, L.W.; Veillon, C.; Kelsay, J.L.; Mertz, W.; Smith, J.C. Sodium and potassium intake and balance in adults consuming self-selected diets. Am. J. Clin. Nutr. 1984, 40, 786–793. [Google Scholar] [CrossRef]
- Rosa, R.M.; De Jesus, E.; Sperling, K.; Suh, A.; Gmurczyk, A.; Myrie, K.A.; Rosner, K.; Lerma, E.; Yu, W.; Breuer, R.; et al. Gastrointestinal and renal excretion of potassium in African–Americans and White Americans. J. Hypertens. 2012, 30, 2373–2377. [Google Scholar] [CrossRef]
- Palacios, C.; Wigertz, K.; Martin, B.R.; Braun, M.; Pratt, J.H.; Peacock, M.; Weaver, C.M. Racial differences in potassium homeostasis in response to differences in dietary sodium in girls. Am. J. Clin. Nutr. 2009, 91, 597–603. [Google Scholar] [CrossRef]
- Consolazio, C.F.; Matoush, L.O.; Nelson, R.A.; Harding, R.S.; Canham, J.E. Excretion of Sodium, Potassium, Magnesium and Iron in Human Sweat and the Relation of Each to Balance and Requirements. J. Nutr. 1963, 79, 407–415. [Google Scholar] [CrossRef]
- Costill, D.L.; Côté, R.; Fink, W.J. Dietary potassium and heavy exercise: Effects on muscle water and electrolytes. Am. J. Clin. Nutr. 1982, 36, 266–275. [Google Scholar] [CrossRef]
- Fukumoto, T.; Tanaka, T.; Fujioka, H.; Yoshihara, S.; Ochi, T.; Kuroiwa, A. Differences in composition of sweat induced by thermal exposure and by running exercise. Clin. Cardiol. 1988, 11, 707–709. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, L.E.; Hubbard, R.W.; Szlyk, P.C.; Matthew, W.T.; Sils, I.V. Voluntary dehydration and electrolyte losses during prolonged exercise in the heat. Aviat. Space Environ. Med. 1985, 56, 765–770. [Google Scholar]
- Malhotra, M.S.; Sridharan, K.; Venkataswamy, Y.; Rai, R.M.; Pichan, G.; Radhakrishnan, U.; Grover, S.K. Effect of restricted potassium intake on its excretion and on physiological responses during heat stress. Graefe’s Arch. Clin. Exp. Ophthalmol. 1981, 47, 169–179. [Google Scholar] [CrossRef] [PubMed]
- Khaw, K.T.; Barrett-Connor, E. The association between blood pressure, age, and dietary sodium and potassium: A population study. Circulation 1988, 77, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.-Y.; Hu, Y.-W.; Yue, C.-S.J.; Wen, Y.-W.; Yeh, W.-T.; Hsu, L.-S.; Tsai, S.-Y.; Pan, W.-H. Effect of potassium-enriched salt on cardiovascular mortality and medical expenses of elderly men. Am. J. Clin. Nutr. 2006, 83, 1289–1296. [Google Scholar] [CrossRef]
- Nowson, C.A.; Morgan, T.O.; Gibbons, C. Decreasing Dietary Sodium While Following a Self-Selected Potassium-Rich Diet Reduces Blood Pressure. J. Nutr. 2003, 133, 4118–4123. [Google Scholar] [CrossRef][Green Version]
- Hypertension Prevention Trial Research Group. The Hypertension Prevention Trial: Three-year effects of dietary changes on blood pressure. Arch. Intern. Med. 1990, 150, 153–162. [Google Scholar] [CrossRef]
- Whelton, P.K.; Appel, L.; Charleston, J.; Dalcin, A.T.; Ewart, C.; Fried, L.; Kaidy, D.; Klag, M.J.; Kumanyika, S.; Steffen, L.; et al. The Effects of Nonpharmacologic Interventions on Blood Pressure of Persons with High Normal Levels. JAMA 1992, 267, 1213–1220. [Google Scholar] [CrossRef] [PubMed]
- Alba, B.K.; Stanhewicz, A.E.; Kenney, W.L.; Alexander, L.M. Acute dairy milk ingestion does not improve nitric oxide-dependent vasodilation in the cutaneous microcirculation. Br. J. Nutr. 2016, 116, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Stanhewicz, A.E.; Alexander, L.M.; Kenney, W.L. Oral sapropterin acutely augments reflex vasodilation in aged human skin through nitric oxide-dependent mechanisms. J. Appl. Physiol. 2013, 115, 972–978. [Google Scholar] [CrossRef] [PubMed]
- Spence, L.A.; Lipscomb, E.R.; Cadogan, J.; Martin, B.; Wastney, M.E.; Peacock, M.; Weaver, C.M. The effect of soy protein and soy isoflavones on calcium metabolism in postmenopausal women: A randomized crossover study. Am. J. Clin. Nutr. 2005, 81, 916–922. [Google Scholar] [CrossRef]
- Gorostidi, M.; Vinyoles, E.; Banegas, J.R.; De La Sierra, A. Prevalence of white-coat and masked hypertension in national and international registries. Hypertens. Res. 2015, 38, 1–7. [Google Scholar] [CrossRef]
- Brunt, V.E.; Minson, C.T. Cutaneous thermal hyperemia: More than skin deep. J. Appl. Physiol. 2011, 111, 5–7. [Google Scholar] [CrossRef] [PubMed]
- Fujii, N.; Brunt, V.E.; Minson, C.T. Tempol improves cutaneous thermal hyperemia through increasing nitric oxide bioavailability in young smokers. Am. J. Physiol. Circ. Physiol. 2014, 306, H1507–H1511. [Google Scholar] [CrossRef] [PubMed]
- Cracowski, J.-L.; Minson, C.T.; Salvat-Melis, M.; Halliwill, J.R. Methodological issues in the assessment of skin microvascular endothelial function in humans. Trends Pharmacol. Sci. 2006, 27, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Palacios, C.; Wigertz, K.; Martin, B.R.; Jackman, L.; Pratt, J.H.; Peacock, M.; McCabe, G.; Weaver, C.M. Sodium Retention in Black and White Female Adolescents in Response to Salt Intake. J. Clin. Endocrinol. Metab. 2004, 89, 1858–1863. [Google Scholar] [CrossRef] [PubMed]
- Squires, R.D.; Huth, E.J. Experimental potassium depletion in normal human subjects. I. Relation of ionic intakes to the renal conservation of potassium. J. Clin. Investig. 1959, 38, 1134–1148. [Google Scholar] [CrossRef] [PubMed]
- Kodama, N.; Morikuni, E.; Matsuzaki, N.; Yoshioka, Y.H.; Takeyama, H.; Yamada, H.; Kitajima, H.; Nishimuta, M. Sodium and Potassium Balances in Japanese Young Adults. J. Nutr. Sci. Vitaminol. 2005, 51, 161–168. [Google Scholar] [CrossRef]
- Soleimani, M.; Bergman, J.A.; Hosford, M.A.; McKinney, T.D. Potassium depletion increases luminal Na+/H+ exchange and basolateral Na+:CO3=:HCO3- cotransport in rat renal cortex. J. Clin. Investig. 1990, 86, 1076–1083. [Google Scholar] [CrossRef] [PubMed]
- Veiras, L.C.; Han, J.; Ralph, D.L.; McDonough, A.A. Potassium Supplementation Prevents Sodium Chloride Cotransporter Stimulation During Angiotensin II Hypertension. Hypertension 2016, 68, 904–912. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, M.V.; Grossmann, S.; Roesinger, M.; Gresko, N.; Todkar, A.P.; Barmettler, G.; Ziegler, U.; Odermatt, A.; Loffing-Cueni, D.; Loffing, J. Rapid dephosphorylation of the renal sodium chloride cotransporter in response to oral potassium intake in mice. Kidney Int. 2013, 83, 811–824. [Google Scholar] [CrossRef]
- Juraschek, S.P.; Woodward, M.; Sacks, F.M.; Carey, V.J.; Miller, I.E.R.; Appel, L.J. Time Course of Change in Blood Pressure From Sodium Reduction and the DASH Diet. Hypertension 2017, 70, 923–929. [Google Scholar] [CrossRef] [PubMed]

| All Mean (SD) | Male Mean (SD) | Female Mean (SD) | |
|---|---|---|---|
| n | 30 | 15 | 15 |
| Age (year) | 48.2 (15.0) | 43.8 (13.7) | 52.7 (15.4) |
| Height (cm) | 172.2 (10.2) | 179.4 (7.1) | 165.0 (7.2) |
| Weight (kg) | 93.9 (22.9) | 99.1 (20.7) | 88.6 (24.4) |
| BMI (kg/m2) | 31.4 (6.1) | 30.5 (4.8) | 32.3 (7.2) |
| Systolic Blood Pressure (SBP) (mmHg) | 133.6 (12.2) | 133.8 (13.8) | 133.3 (10.4) |
| Diastolic Blood Pressure (DBP) (mmHg) | 85.5 (8.6) | 86.1 (8.1) | 84.9 (9.2) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stone, M.S.; Martin, B.R.; Weaver, C.M. Short-Term RCT of Increased Dietary Potassium from Potato or Potassium Gluconate: Effect on Blood Pressure, Microcirculation, and Potassium and Sodium Retention in Pre-Hypertensive-to-Hypertensive Adults. Nutrients 2021, 13, 1610. https://doi.org/10.3390/nu13051610
Stone MS, Martin BR, Weaver CM. Short-Term RCT of Increased Dietary Potassium from Potato or Potassium Gluconate: Effect on Blood Pressure, Microcirculation, and Potassium and Sodium Retention in Pre-Hypertensive-to-Hypertensive Adults. Nutrients. 2021; 13(5):1610. https://doi.org/10.3390/nu13051610
Chicago/Turabian StyleStone, Michael S., Berdine R. Martin, and Connie M. Weaver. 2021. "Short-Term RCT of Increased Dietary Potassium from Potato or Potassium Gluconate: Effect on Blood Pressure, Microcirculation, and Potassium and Sodium Retention in Pre-Hypertensive-to-Hypertensive Adults" Nutrients 13, no. 5: 1610. https://doi.org/10.3390/nu13051610
APA StyleStone, M. S., Martin, B. R., & Weaver, C. M. (2021). Short-Term RCT of Increased Dietary Potassium from Potato or Potassium Gluconate: Effect on Blood Pressure, Microcirculation, and Potassium and Sodium Retention in Pre-Hypertensive-to-Hypertensive Adults. Nutrients, 13(5), 1610. https://doi.org/10.3390/nu13051610







