1. Introduction
The World Health Organization (WHO) reports that cardiovascular disease (CVD) is the leading cause of death worldwide (31% of all deaths), with an estimate of 17.3 million deaths a year [
1]. Atherosclerosis is a relevant factor contributing to CVD development [
2], and several factors can contribute to its development (i.e., endothelial dysfunction) [
3]. Thus, strategies to prevent atherosclerosis and CVD could be greatly beneficial for the general population. Recently, the gut microbiota-derived metabolite trimethylamine N-oxide (TMAO) has emerged as a pro-atherogenic molecule associated with higher cardiovascular risk [
4,
5,
6].
TMAO is formed due to consecutive gut bacterial and host metabolism of substrates such as choline and L-carnitine. The first metabolic step occurs in the gut, where bacteria oxidize these substrates into trimethylamine (TMA) through TMA-lyase enzymes. Specifically, TMA is formed from choline mainly through CutC/D enzyme complex activity [
7], and from L-carnitine through CntA/B [
8]. Following absorption into circulation, the second step occurs in the liver, where TMA is further oxidized by flavin-containing monooxygenase 3 (FMO3) into TMAO [
9]. Various strategies have been proposed to control TMAO formation as a means to reduce CVD risk [
10]. Reducing TMAO formation through TMA-lyase inhibition has shown promising anti-atherosclerotic effects in mice [
11]. Another strategy would be to reduce the expression of the CutC/D or CntA/B enzyme complexes by altering the composition of the gut microbiota. Thus, the development of new strategies that target TMA-lyase(s) could be useful to manage and/or reduce TMAO formation, atherosclerosis and CVD development.
It is believed that the reported cardioprotective effects of fruit and vegetal consumption are attributed, at least to some extent, to their bioactive phytochemicals [
12,
13]. While various mechanisms of action have been proposed, an emerging theory is that phytochemicals may also mediate their cardioprotective effects in part through the inhibition of TMAO formation [
10]. Some intervention strategies based on phytochemical supplementation (i.e., allicin and 3,3-dimethyl-1-butanol) have been successful to reduce TMA and/or TMAO formation in animal models [
11,
14,
15] and humans [
16,
17]. High-throughput inhibitor screening studies have demonstrated their utility for quickly identifying synthetic CutC/D inhibitors [
18], and these types of approaches could be used to narrow down a vast array of hundreds or thousands of potential phytochemicals to a few promising candidate compounds with inhibitory activity to be subsequently tested in vivo. In this sense, Heng et al. have recently proposed a methodology to identify TMA-lyase-containing bacteria, isolate them, and use them to screen for potential compounds that inhibit choline metabolism to TMA in silico and in vitro [
19]. In their approach, in silico docking studies suggested a few phytochemicals as potential TMA-lyase inhibitors, but the
in silico approach alone does not take into account the diversity of bacteria in the gut (i.e., interaction between bacteria). Additionally, no cytotoxic tests are reported, which poses the question of whether the results are due to a TMA-lyase inhibitory effect of phytochemicals or a cytotoxic one. Thus, there still exists a lack of in vitro studies that screen the potential of phytochemicals as TMAO-reducing agents, and phytochemicals are currently being tested in vivo one at a time in a non-systematic fashion. To date, Bresciani et al. [
20] are the only authors that have screened the TMA-reducing properties of several phytochemicals in vitro. Specifically, their study showed that some phenolic compounds from orange juice were able to modulate choline and L-carnitine microbial conversion to TMA in a fecal fermentation model. Of note, in vitro studies that target the gut microbiome profile and/or function can potentially provide a reasonable estimation of the activity of tested conditions in vivo, as no absorption of the tested compound is required [
10]. A high-throughput in vitro screening assay could significantly advance pre-clinical data on the use of phytochemicals for reducing TMA production in a rapid, systematic, and inexpensive manner.
Thus, the aims of this study were to optimize the experimental conditions for a high-throughput, low-volume fecal fermentation method capable of screening the TMA-lowering potential of phytochemicals, and demonstrate the efficacy of selected test compounds. Optimized conditions included fecal slurry to growth media ratio, fermentation time, choline concentration, positive TMA-lyase inhibitor (DMB) concentration, and phytochemical (gallic acid and chlorogenic acid) range of concentrations.
4. Discussion
Elevated TMAO circulating levels have been associated with CVD and atherosclerosis development [
4,
5,
6], and their reduction has been shown to produce cardioprotective and anti-atherosclerotic effects [
11]. The inhibition of TMAO formation can be achieved by reducing the conversion of choline and carnitine to TMA in the gut, thus reducing the amount of TMA available for oxidation to TMAO [
11]. Phytochemicals are known to exert cardioprotective and anti-atherosclerotic functions [
13,
27], and some of these compounds have also been reported to reduce TMAO formation [
11,
14,
15,
16,
17]. It is thus possible that phytochemical inhibition of TMA formation may be an effective cardioprotective mechanism via lowering TMAO. However, so far there have been relatively few studies in this area, the majority of which are in animal models, which are inefficient for rapidly screening large numbers of potentially effective compounds [
10]. Thus, the development of a high-throughput in vitro screening method to evaluate the inhibition of TMA formation in the gut by phytochemicals could be highly useful to select promising lead compounds, for subsequent focused in vivo studies. In this study, we performed in vitro anaerobic fecal fermentations using fecal samples from healthy humans and optimized parameters including the proportion of fecal slurry, choline concentration, fermentation time, positive control (DMB) concentrations, and concentration range of relevant phytochemicals (gallic acid and chlorogenic acid).
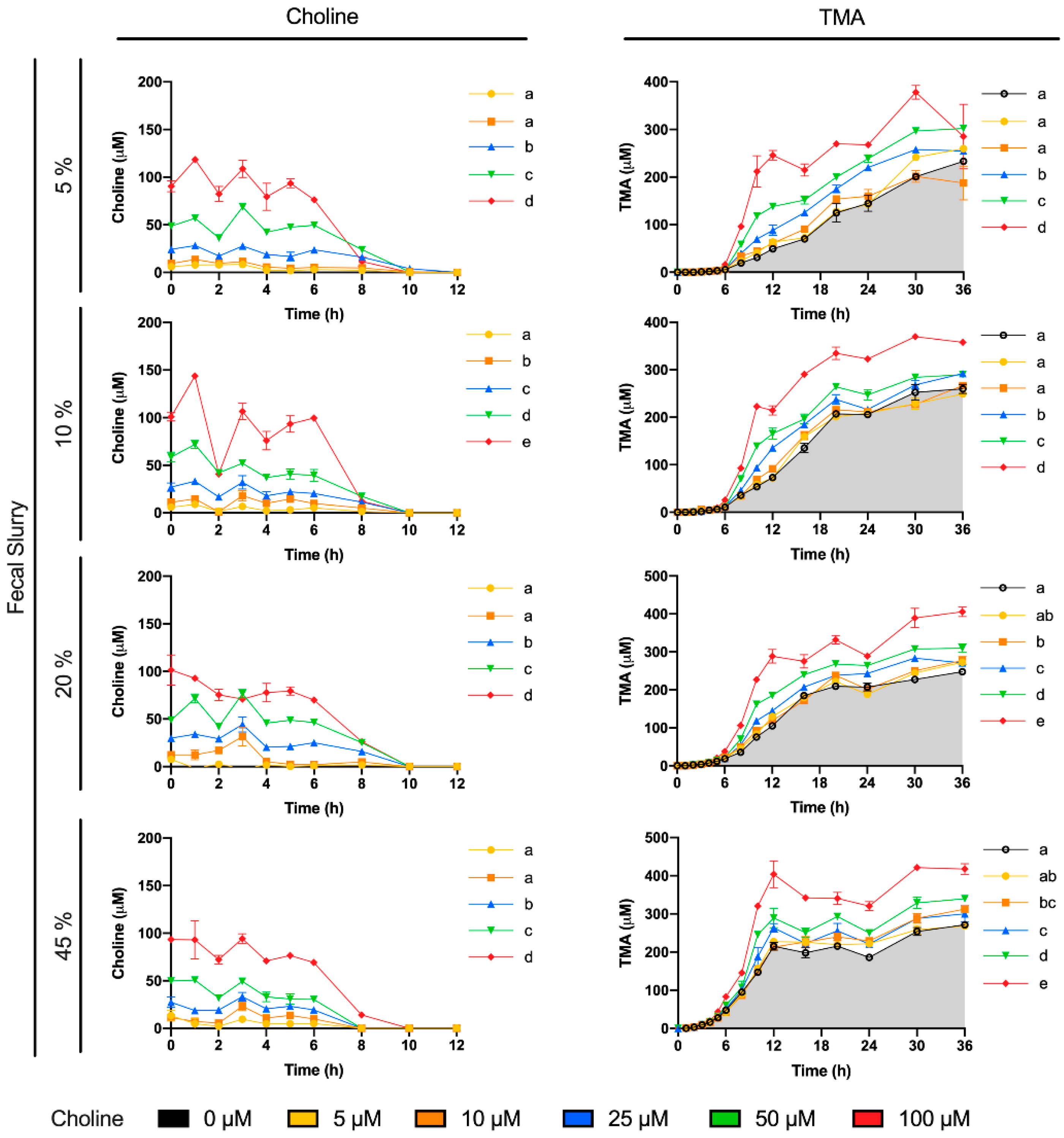
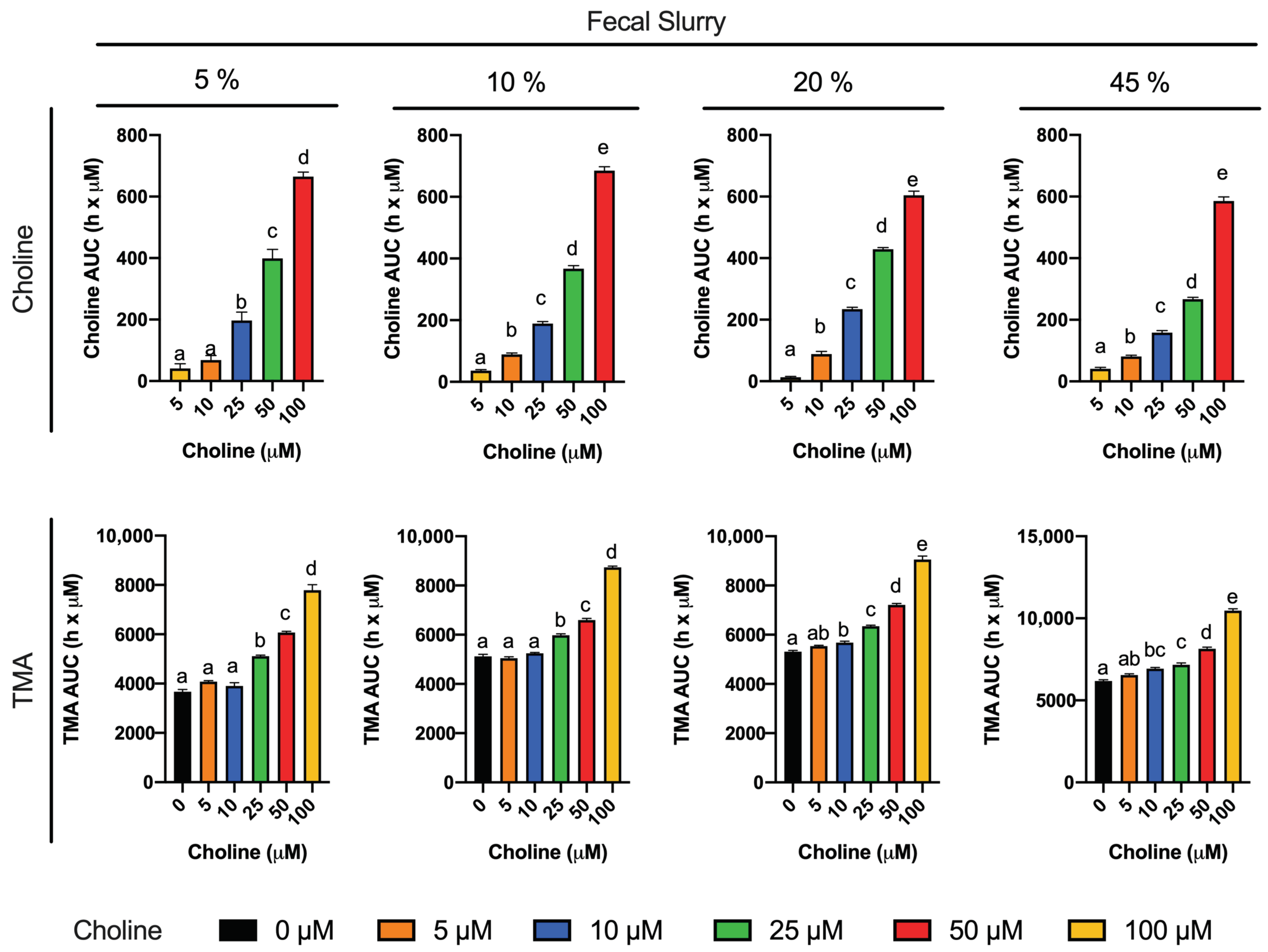
To reach our goal, we first studied choline (0–100 µM) utilization and TMA production in fermentations containing 5–45% fecal slurry (1:10 in PBS 1X) for 36 h (
Figure 1 and
Figure 2). Choline consumption curves indicated that choline-metabolizing bacteria became active around 6 h after fecal slurry inoculation, and that bacterial choline use occurred predominantly from 6 to 12 h. This is also corroborated by observed TMA production, which started to increase at 6 h in fecal slurry concentrations ≤ 20%. The plateau in TMA concentrations reached by fecal slurry concentrations ≥ 10%, as well as the final similar TMA concentration reached by each level of initial choline conditions regardless of fecal slurry concentration, demonstrate that all compounds capable of being metabolized into TMA were generally consumed and oxidized within the 36-h timeframe. Background (i.e., when 0 µM choline was added) concentrations of TMA production reached significant levels in all cases (>200 µM), suggesting that there are significant TMA precursors in the background (media and/or fecal samples). Indeed, TMA precursors have been reported in fermentation growth media, human fecal starters [
20] and human feces [
28]. In our study, when fermentation media + fecal slurry 1:10 (20%) was screened for TMA precursors, it showed ~225 µM choline and ~1120 µM betaine, both of which could contribute to background TMA production. Furthermore, endogenous phospholipids likely contributed to background TMA production. Based on background TMA production levels, it seems that endogenous choline is the primary compound giving rise to TMA background levels. For example, background levels in fecal fermentations with 20% fecal slurry report ~225 µM choline (
Table 1), and TMA production at 36 h is quite proportional (~250 µM TMA). Thus, background production of TMA can likely be attributed predominantly to background choline levels. Oddly, the administration of 100 µM exogenous choline did not result in a proportional increase in TMA levels when compared to the background. For example, at 36 h in fecal slurry 20%, TMA levels reached by choline-free and choline 100 µM were, respectively, of 247.7 ± 2.9 µM and 405.6 ± 12.5 µM (a difference of ~158 µM), when we would have expected a maximal possible increase of ~100 µM in TMA between choline 100 µM and choline-free conditions (as 1 mol choline produces a maximum of 1 mol TMA). This is, however, in line with the study of Bresciani et al. [
20]. When an omnivore human fecal starter was used to ferment 100 µM choline, TMA reached concentrations > 500 µM (initial background concentrations of choline of 45.6 ± 17.7 µM). Presumably, the addition of choline might cause bacterial shifts to promote the use of choline and other TMA precursors, causing an increase in TMA production capacity, and the lack of proportionality between the background and choline-supplemented conditions (i.e., differences are not just due to increases in substrate, but also increases in metabolic capacity, enhancing TMA production from background substrates as well). Overall, the speed of the changes in choline and TMA concentrations, and the time at which TMA concentrations reached a plateau were dependent on fecal slurry concentrations: the higher the fecal slurry concentration, the earlier the TMA concentration plateau was reached and the faster the kinetics. This suggests that choline oxidation of TMA is mediated by bacteria rather than spontaneous chemical oxidation.
To study the inhibition of choline microbial conversion to TMA in an in vitro assay, TMA production should be significantly different from the background in the assay. Choline concentrations of 5 and 10 µM were discarded because of their lack of statistical difference in kinetic curve and AUCs compared to the background. Although choline concentrations of 25 and 50 µM generated statistically different kinetic curves and AUCs against the background, the absolute differences in TMA production in at 36 h, and at time to reach TMA plateau were low. For example, in fecal slurry 10%, at 20 h (time to reach plateau in TMA) and at 36 h, the fold-change in TMA production between choline 25 and 50 µM and the background ranged between 1.1 and 1.3. However, this fold-change ranged between 1.4 and 1.6 in choline 100 µM. Due to the fact that the 5% fecal slurry percentage did not report a TMA concentration plateau, and that fecal slurry 45% required a high proportion of fecal slurry and reported the fastest kinetics, both conditions were not selected for further experiments. Fast kinetics might involve practical issues (missing relevant sample collection timepoints), and slower kinetics are also inconvenient due to longer experimental conditions. For these reasons, fecal slurry 20% was preferred over 10%. Thus, we selected 20% fecal slurry (1:10 in PBS 1X) and choline 100 µM for further experiments. Additionally, based on the observed TMA plateau profile, fermentation time was shortened to 24 h in follow-up experiments. These experimental conditions are in line with the ones used in similar fermentation experiments [
11,
20].
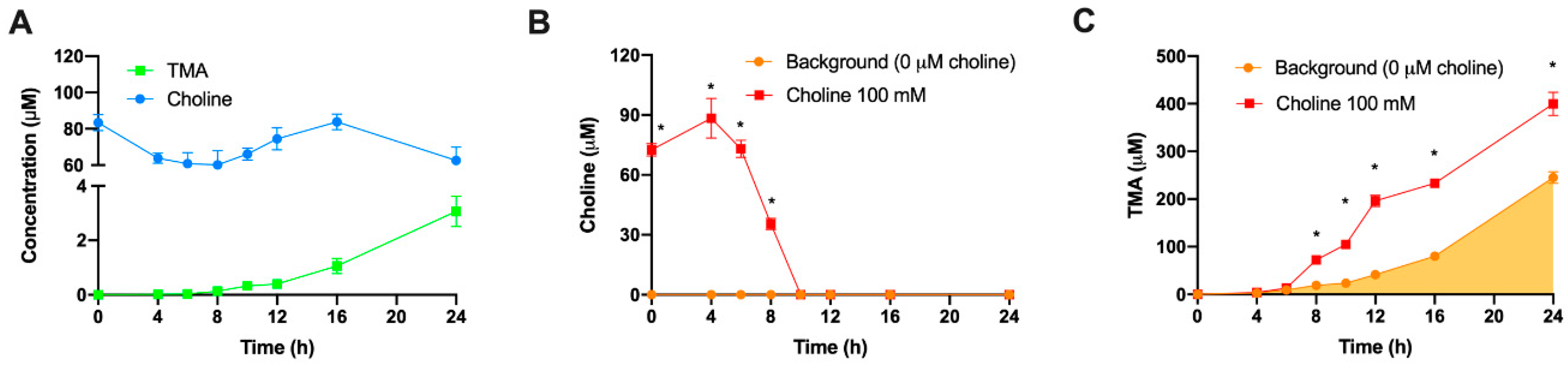
The first set of experiments was used to select optimal choline and fecal slurry fermentation conditions. Some questions still remained. For example, does non-microbial conversion of choline contribute significantly to TMA formation in this model? To study this contribution, choline (100 µM) was incubated in fermentation media (45% growth media and 55% PBS 1X) for 24 h under anaerobic conditions in the absence of fecal slurry, which was substituted by PBS 1X (
Figure 3). Overall, choline concentrations remained > 70% of initial concentration throughout the incubation, and spontaneous TMA production barely occurred (reaching 3.07 ± 0.28 µM at time 24 h, a small fraction of TMA production observed during fecal incubations). Thus, the vast majority of observed TMA formation in fermentations with fecal slurry does in fact appear to originate from the action of the microbiota on relevant substrates. Nevertheless, the fecal fermentation inoculated with fecal slurry itself contains choline and betaine, and may even contain other TMA precursors such as phosphatidyl choline, which can be metabolized to TMA [
7,
8,
26]. To corroborate that TMA was in fact produced from the exogenous choline rather than only endogenous background choline levels, choline and TMA were tracked for 24 h in fermentations containing 20% fecal slurry (1:10 in PBS 1X) with and without choline added at 100 µM (
Figure 4). TMA production from the background reached ~245 µM, which further demonstrates that the presence of TMA-producing substrates in background fermentation conditions, understood as everything except added choline (growth media + PBS + fecal slurries), contributes significantly to TMA production. This is in line with our previous results and the study of Bresciani et al. [
20]. Of note, we reported relevant concentrations of choline and betaine in our background, suggesting that fecal slurries and media contribute to TMA formation through intrinsic metabolism of endogenous choline, and potentially betaine. Other compounds, such as L-carnitine [
8,
26] or TMAO through retro-conversion to TMA [
25], seem not to contribute to TMA formation in the background as they are not detected in fermentation media + fecal slurry. The differences in TMA production between the choline-free and the choline-supplemented conditions (~154 µM) indicate that exogenous choline is being oxidized into TMA by the action of the bacteria present in the fecal slurry. The lack of proportionality (154 µM increase in TMA from addition of 100 µM choline) again suggests that the addition of exogenous choline promotes a shift in bacteria communities towards a higher TMA-producing profile, which would explain the lack of proportionality between the levels of choline added and the generated TMA compared to the background. Thus, further studies will include the analysis of background conditions.
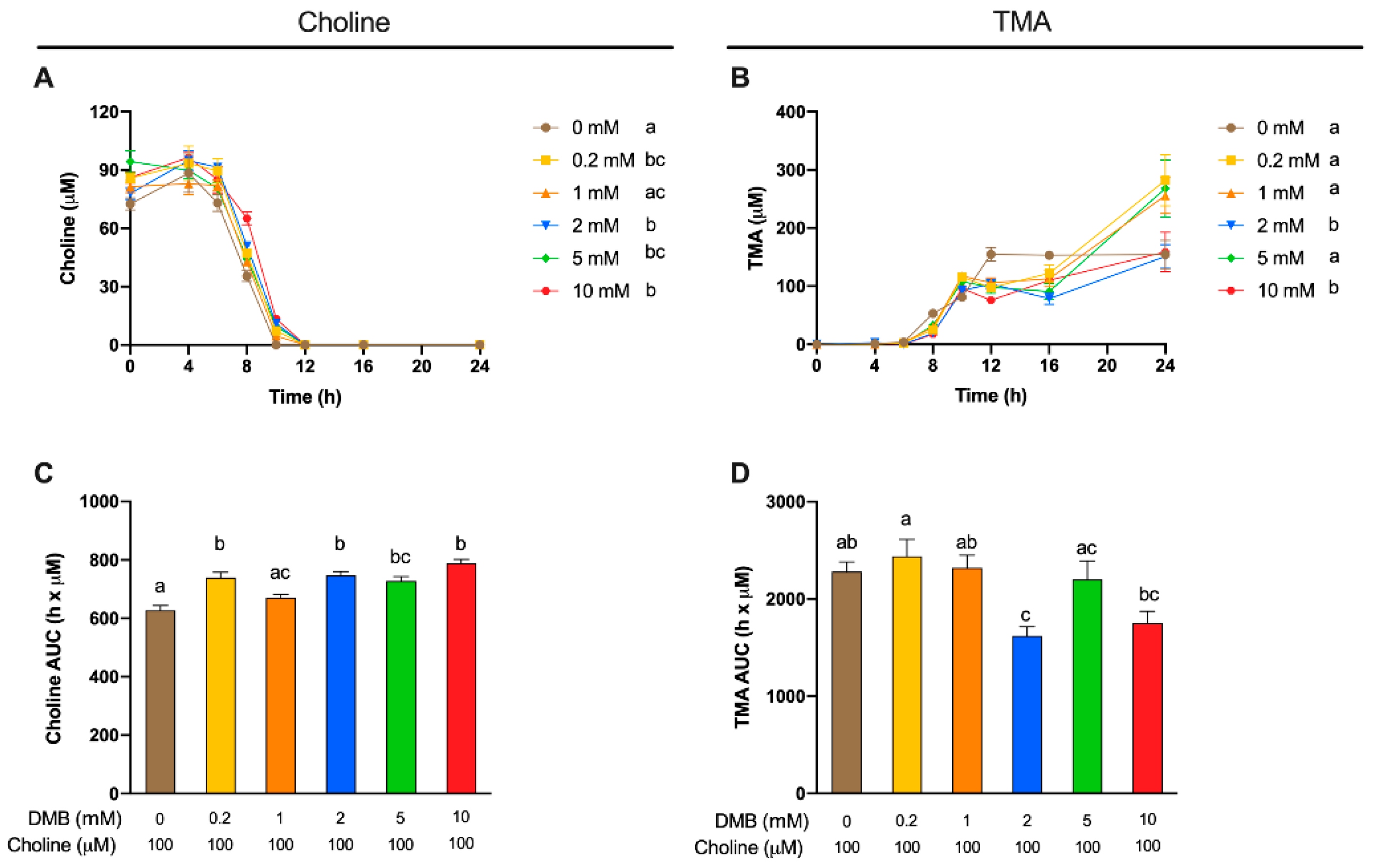
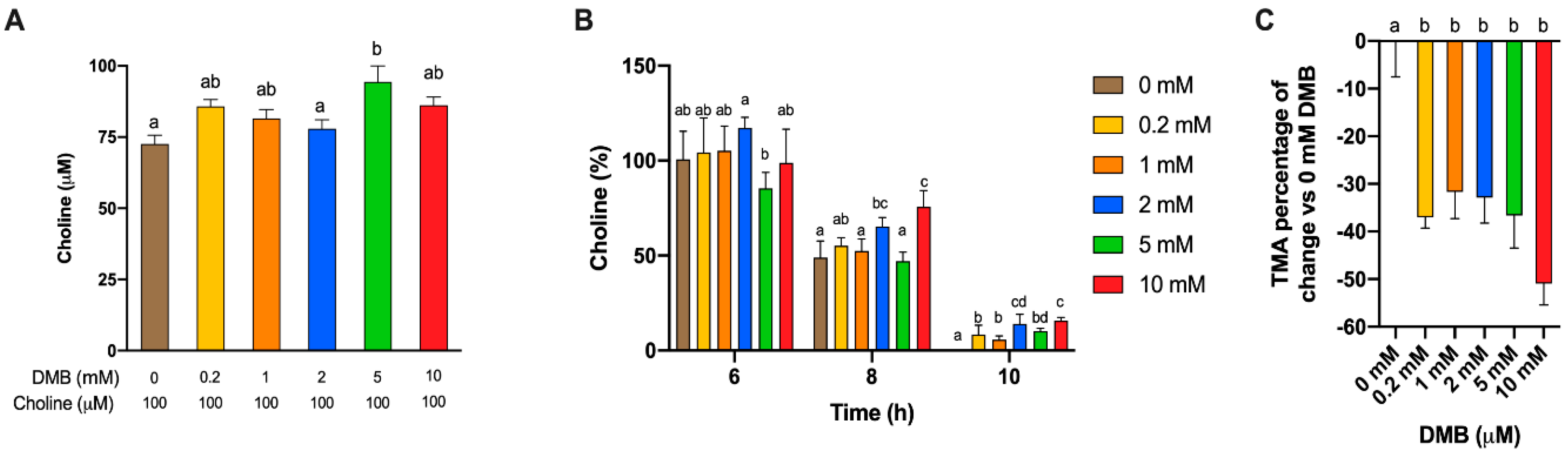
A range of DMB concentrations between 0.2 and 10 mM were tested to optimize the concentrations of a known TMA-lyase inhibitor (
Figure 5). This range was selected based on preliminary experiments (data not shown). The kinetic curves corroborate our previous results that the metabolism of choline by fecal slurry microbiota occurs primarily from 6 to 10 h in DMB-free conditions, and show that this occurs from 6 to 12 in DMB-supplemented conditions. Our data thus suggest that DMB is delaying the microbial oxidation of exogenous choline into TMA by inhibiting TMA-lyase enzymes. Previous studies using DMB have already reported such inhibitory effects. For example, DMB (0–2 mM) reduced TMA-lyase activity by ~20% in
E. coli cells transfected with
D. alaskensis CutC/D genes [
11]. Of note, the microbial oxidation of exogenous choline occurred at up to 12 h (
Figure 4A). Thus, the exogenous substrate is not present after this time, and any TMA formation after 12 h (
Figure 4B) is originating from other TMA precursors. Considering all data, the DMB concentrations that demonstrate optimal performance as a TMA-lyase inhibitor are 2 and 10 mM.
The odd behavior of DMB 5 mM compared to 2 and 10 mM could be attributed to the higher initial measured choline concentration in that condition (
Figure 5A), or to U- and/or J-shape dose–response curves (hormetic effect) [
29]. DMB 5 mM seems to follow the trend of lower (1 and 0.2 mM) DMB concentrations. To consider this variability in the inhibitory effect of DMB, choline concentrations relativized to initial (0 h) choline concentrations were evaluated at relevant metabolically active time points (6–12 h) (
Figure 5B). The higher the DMB concentration, the higher the percentage of choline detected, which indicates that choline is not being metabolized by bacteria, potentially due to a TMA-lyase inhibitory effect of DMB. To further investigate the inhibitory effect of DMB, we analyzed the relative change in TMA of DMB-treated groups in comparison to DMB-free conditions. At 12 h, a time point at which choline is no longer quantifiable (above the background) in any groups, DMB-treated groups report a >30% reduction in TMA concentration when compared to DMB-free conditions (
Figure 5C), which is in line with the percentages of inhibition of 40–50% reported by Wang et al. [
11] in human fecal intact cells. Overall, our results reinforce the idea that DMB inhibits choline metabolism, probably due to a TMA-lyase inhibitory effect already reported by Wang et al. [
11].
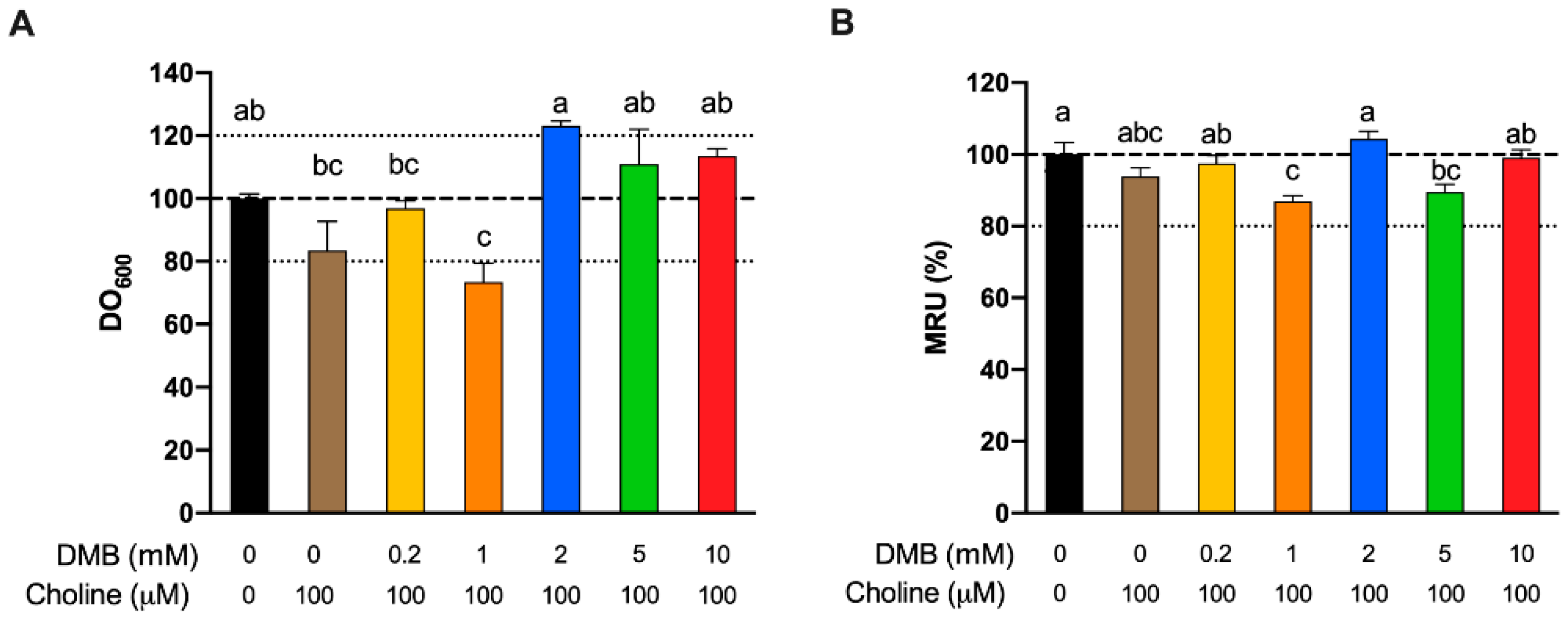
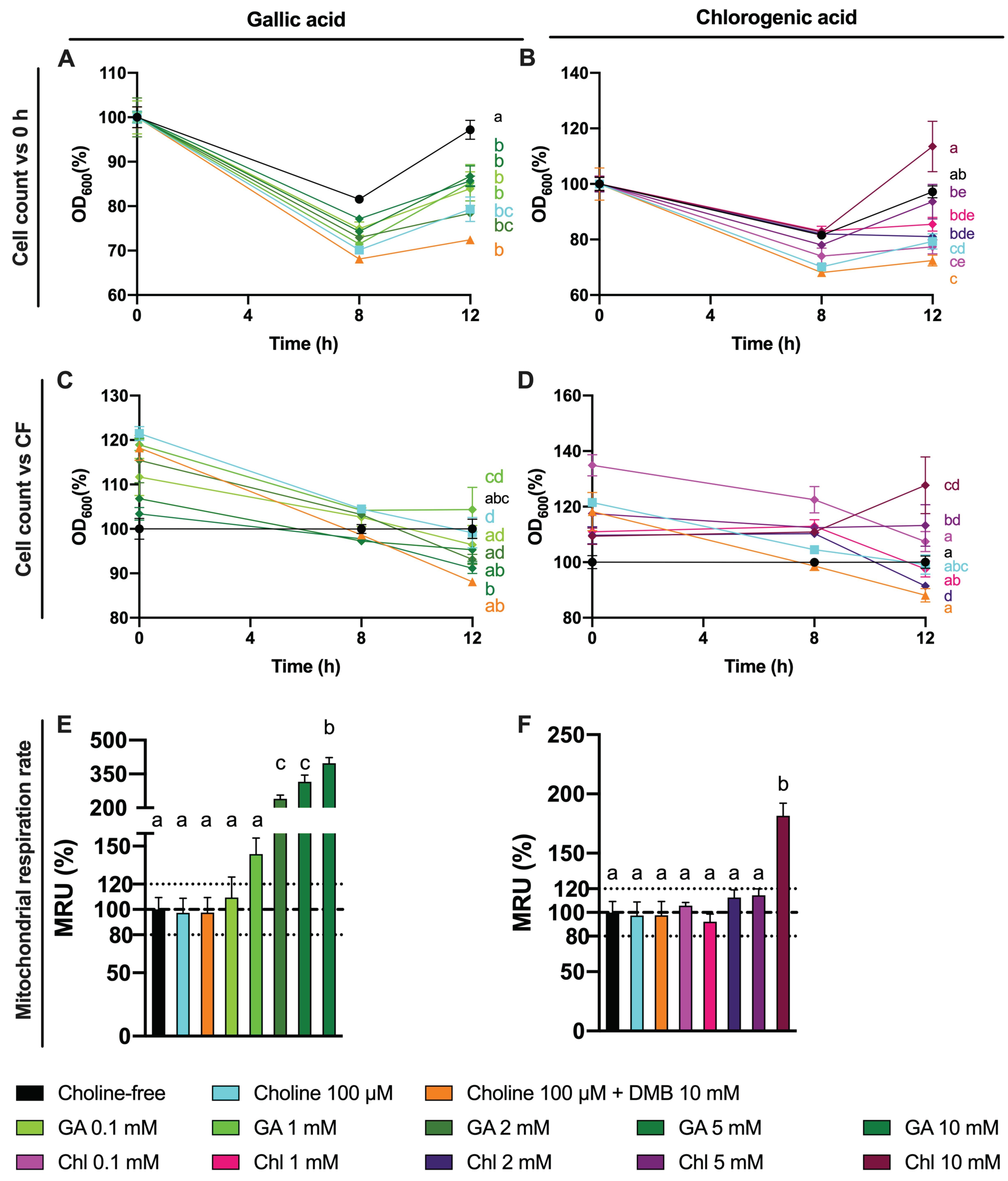
It is clear now that DMB does inhibit TMA production, and this is linked with a reduced usage of choline. However, whether or not these changes in TMA and choline are attributed to a TMA-lyase inhibitory effect of DMB or a cytotoxic/cytostatic effect in our study are unknown. Wang et al. [
11] have already defined DMB as a TMA-lyase inhibitor using cell lysates, intact cells, and microbiota from mice and humans. In their study, they also corroborated that the growth of certain bacterial strains (
P. mirabillis,
P. penneri and
E. fergusonii) under ~8 mM DMB was not compromised. We analyzed the cell content and mitochondrial respiration rate of our fermentations with or without DMB (0.2–10 mM) at the end (24 H) of the fermentations (
Figure 6). No significant differences were reported between background and choline +/DMB-free conditions, which suggests that choline administration is not toxic to bacteria. Although DMB seems to have some slight effects on bacterial growth, it does not have a significant impact on biochemical activity. Overall, these results indicate that DMB administration to choline-supplemented bacteria does not alter their growth or biochemical activity in a way that could explain the observed changes in TMA and choline metabolism. Thus, DMB effect on TMA and choline concentrations does not predominantly arise from an inhibition of bacterial growth or toxic effect, which agrees with the TMA-lyase inhibitory effect reported by Wang et al. [
11]. To our knowledge, this is the first attempt to study mitochondrial respiration rate in the context of choline microbial oxidation into TMA. However, these data represent the viability of the whole microbial species present in the fermentations. Overall, the highest inhibition of microbial choline oxidation into TMA with low effect on cell growth and mitochondrial respiration is the dose of 10 mM DMB. The inhibition of microbial choline oxidation into TMA through the DMB-mediated inhibition of TMA-lyases in human fecal samples is ~50% in our study (DMB 10 mM), and ~70% in the study of Wang et al. (DMB 2 mM). DMB can thus be used as a positive control in the inhibition of TMA-lyases. Thus, 10 mM DMB was selected as a positive TMA-lyase control treatment in further studies. However, its reported low levels in foodstuffs (maximum levels of 25 mM in balsamic vinegars, red wine and cold-pressed extra virgin olive oils [
11]) make the inhibition of TMA-lyases virtually impossible in a normal diet.
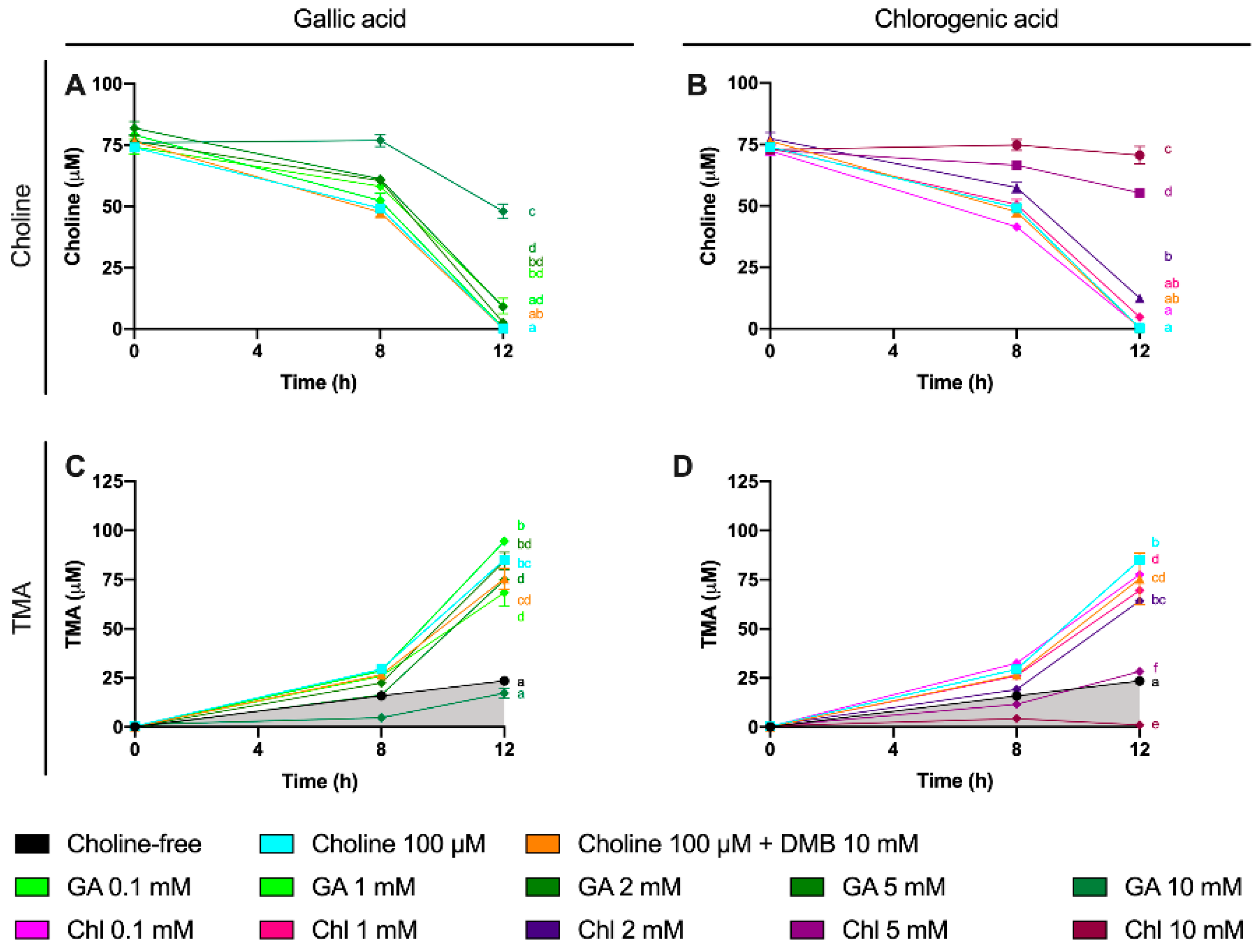
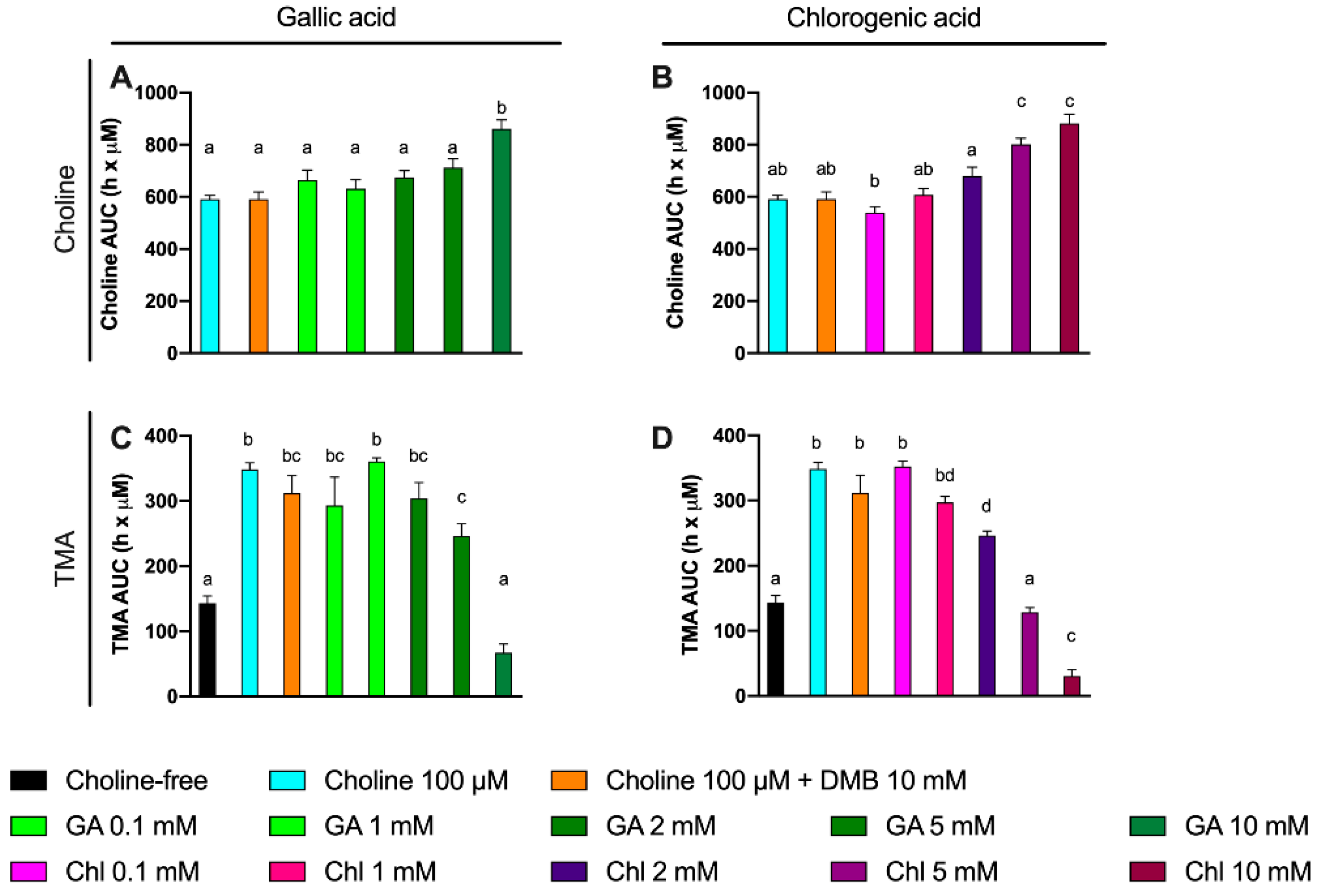
Some phenolic compounds have shown their ability to reduce TMAO levels in different models [
10]. Thus, we selected two phenolic compounds highly consumed by humans: gallic acid and chlorogenic acid [
30]. Moreover, gallic acid as well as chlorogenic acid and their derivatives have reported relevant cardioprotective functions [
31,
32]. We thus studied the potential use of these two phenolic compounds to inhibit TMA production in a range of concentration within 0.1–10 mM at relevant time points (8 and 12 h). Both gallic acid and chlorogenic acid reported a dose-dependent effect on choline use and TMA production. The most effective doses were gallic acid 10 mM and chlorogenic acid 10 mM, both at reducing choline use and TMA production. Of note, the percentage of TMA production inhibition at 12 h was >80% for those conditions, which is superior to the results reported by DMB. Interestingly, TMA production at the 10 mM of gallic acid was equivalent to the background, and it was lower than the background for chlorogenic acid at 10 mM, indicating that chlorogenic acid inhibited background TMA production as well. Overall, between both compounds, chlorogenic acid seems to be a better inhibitor of TMA production. This is reflected in lower IC
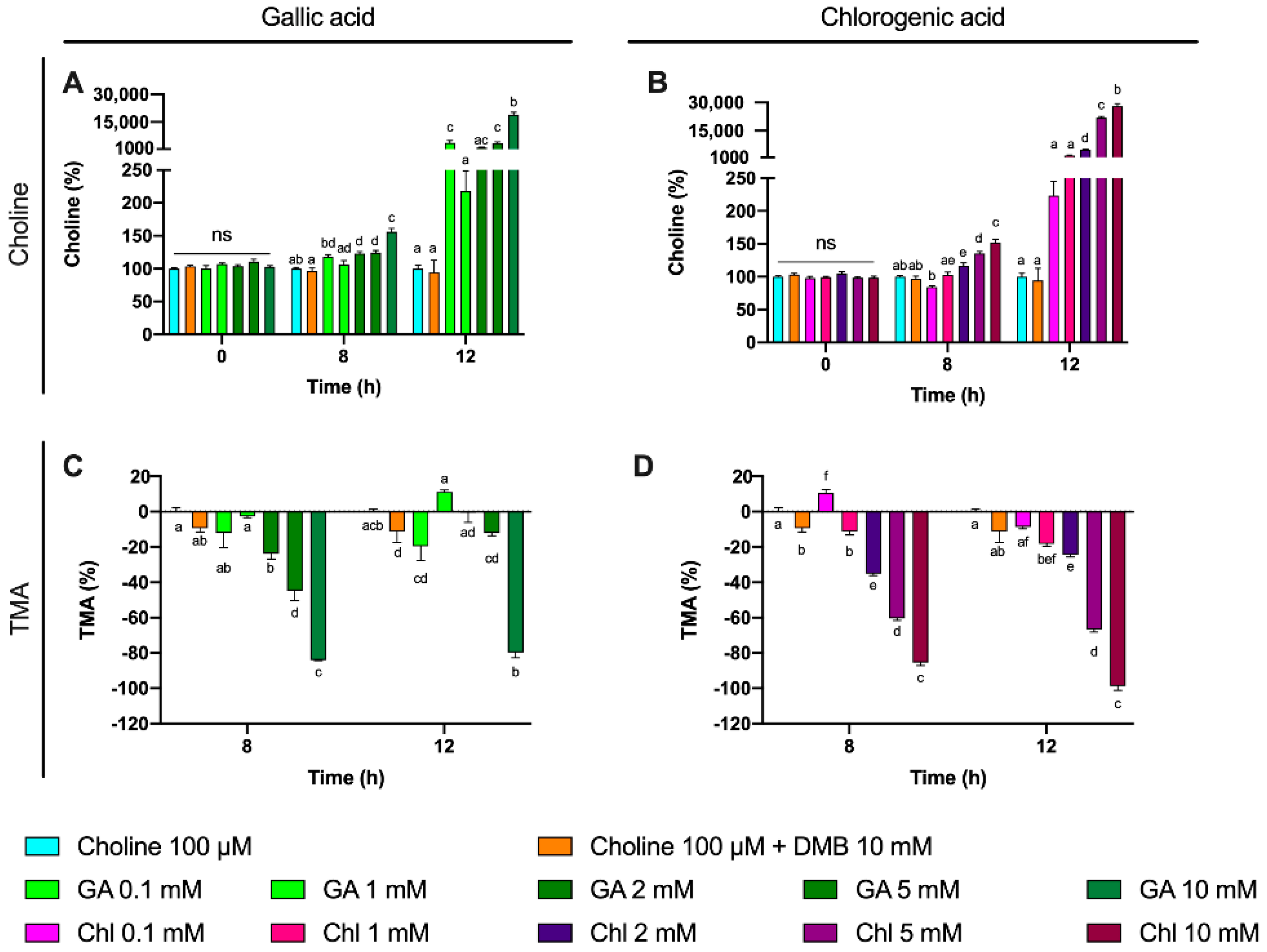
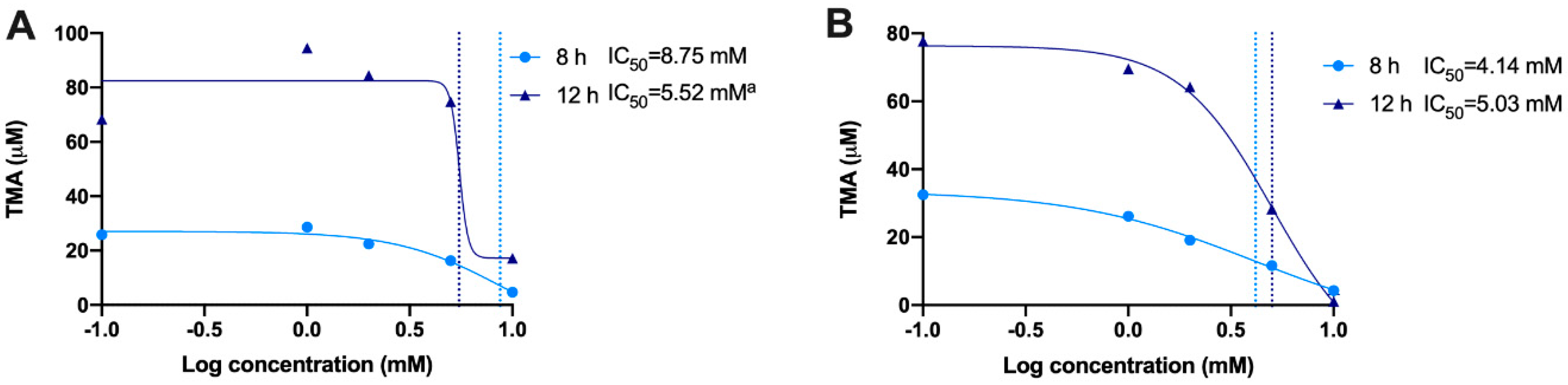
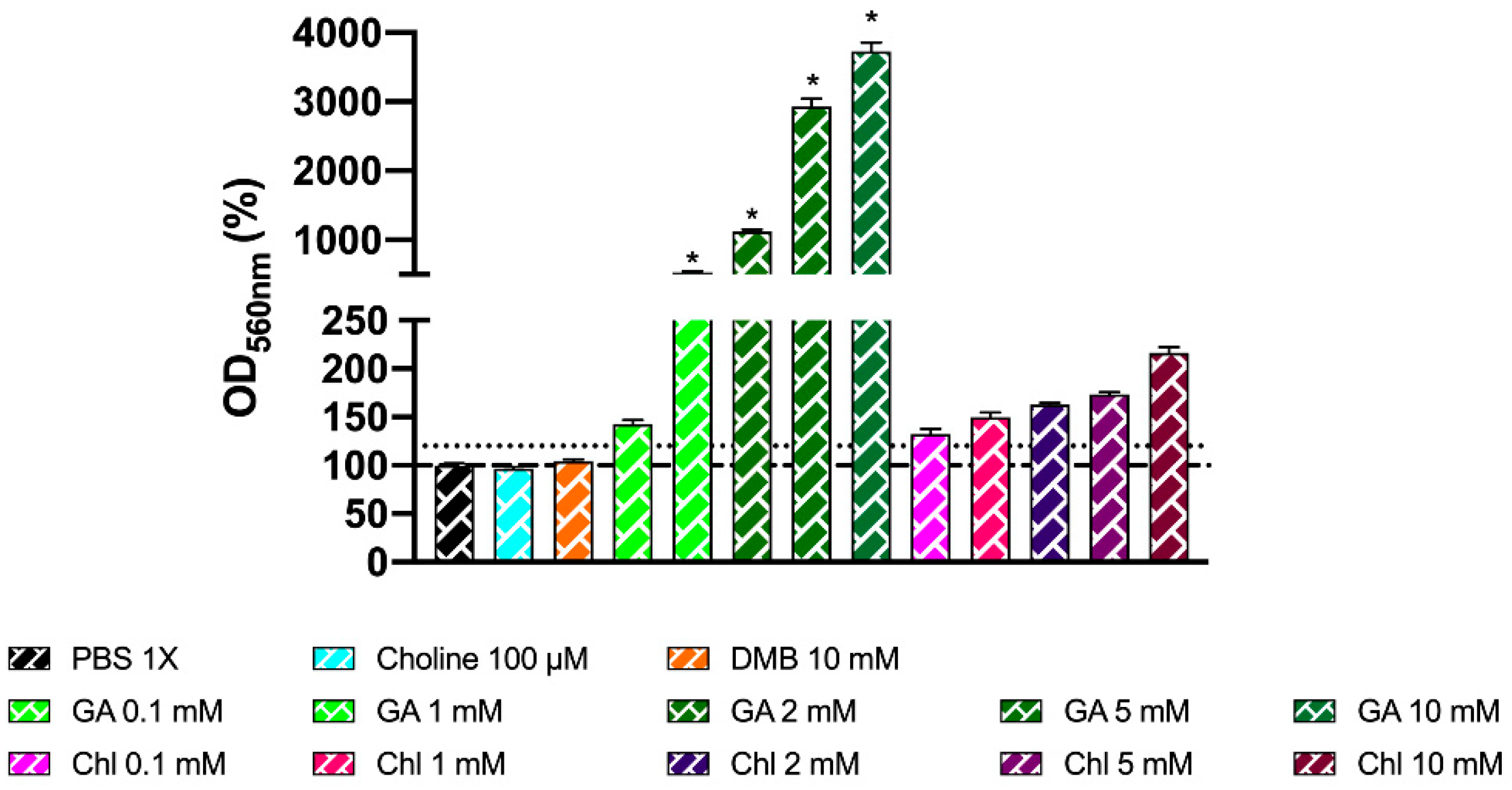
50 for chlorogenic at 8 h. Remarkably, these inhibitory effects were not attributed to a reduction in cell number, as cell count data demonstrate a lack of significant reduction in growth between phenolic compound-treated fermentations compared to choline-free and choline 100 µM conditions. Due to gallic acid and chlorogenic acid interference with MTT, cell viability in these specific conditions could not be accurately assessed. The greater MTT-reactive activity of gallic acid compared to chlorogenic acid reflects the superior reducing activity of gallic acid, due to its three phenolic hydroxyl groups on the same ring. Due to the antioxidant/reducing activities of many phytochemicals (particularly phenolics), MTT will not be a generally valid approach to assess cellular viability when these compounds are present. Other approaches of assessing cell viability will need to be employed.
To further study the effect of gallic acid and chlorogenic acid on TMA production inhibition, other potential sources of TMA were studied. Besides choline, L-carnitine, γ-butyrobetaine and TMAO were not detected or not quantified at 0 h in all samples. However, betaine was present in fermentation media. Betaine levels were monitored in this third experiment, and showed interesting results. Given the high concentration of betaine in fermentation media, its quantification through the addition of standard betaine would provide inaccurate values. Rather, we monitored the relative changes of betaine in fermentation conditions relativized to choline 100 µM fermentation conditions. We also plotted relativized betaine/betaine-d
9 peak areas. Of note, no differences were reported at time 0 h between treatment groups, but relative betaine levels versus choline-free (0 µM) increased over time in gallic acid- and chlorogenic acid-supplemented groups. This effect was most relevant for chlorogenic acid treatments, especially at 8 h (all concentrations) and at 12 h for 5 mM. Choline is primarily metabolized to TMA through its direct oxidation mediated by the bacterial CutC/D enzyme complex [
7]. Alternatively, choline can be metabolized to TMA in a series of catalytic steps involving choline dehydrogenase, betaine aldehyde dehydrogenase and betaine reductase, which generate betaine as an intermediate product [
33]. Our data seem to indicate that gallic acid, and especially chlorogenic acid, could be modulating the metabolic route by which choline is metabolized, which could potentially indicate a TMA-lyase inhibitory effect of these compounds. DMB, however, is not reporting these effects. Substrate analogues tend to inhibit enzymes by competing for enzyme’s active site. This is the case of a cyclic choline analogue that inhibits CutC [
34], and in silico docking studies have suggested this as a potential mechanism of action of some other synthetic choline analogues [
18]. However, other compounds can inhibit enzymes by interacting with other enzyme pockets and shifting enzyme structure in a way that it loses enzymatic activity. Thus, this could be a potential explanation between the differences in betaine production between DMB and the tested phenolic compounds when compared to choline 100 µM, as DMB is structurally very close to both choline and betaine. The shunting of choline to betaine by phenolic compounds warrants further investigation.
Overall, gallic acid and chlorogenic acid reduce TMA formation, either through a direct TMA-lyase inhibitory effect or another non-toxic one. In fact, the data presented herein do not prove that gallic acid and/or chlorogenic acid reduce TMA production through direct TMA-lyase inhibitory effects. For example, in the study of Bresciani et al. [
20], the most potent treatments to reduce TMA production were those that involved the supplementation of growth media with mono or disaccharides (i.e., sucrose, fructose and glucose). This suggests that some treatments reduce TMA production simply by providing better energy substrates than choline. Put simply, in our experiment, bacteria could be using gallic acid and chlorogenic acid as a more preferent source of energy than choline, thus leading to a reduction in choline use and TMA production. Indeed, both gallic acid and chlorogenic acid can be metabolized by the gut microbiota [
35,
36]. For example, chlorogenic acid can be rapidly metabolized (7 h) in human fecal fermentation into 3-(3,4-dihydroxyphenyl)propionic acid [
35], which opens the door to new mechanistic questions such as what is the bioactive compound, chlorogenic acid or one of its microbial metabolites. Of note, the fact that IC
50 for chlorogenic acid is higher at 12 h than at 8 h could indicate that chlorogenic acid is being degraded to a more potent inhibitory metabolite. Moreover, these compounds could also inhibit choline bacterial uptake. Future studies using cell lysates and the analysis of choline and TMA within bacterial cell should shed some light onto those mechanisms. Furthermore, the mechanisms of action may be to reduce the abundance of bacterial species that express the TMA-lyase gene. Further studies are necessary in order to investigate this possibility.
Concentrations of gallic acid and chlorogenic acid at 5–10 mM have herein been demonstrated to be efficient at inhibiting the microbial conversion of choline into TMA in vitro, although the mechanisms by which this occurs are still unknown. Although these doses can be perceived as high for phenolic compounds, in the context of TMA production inhibition and selected phenolic compounds, those doses are likely relevant. First, the poor bioavailability and extensive phase-II metabolism of phenolics, which is a main limiting factor in systemic bioavailability and effects in peripheral organs and tissues such as the liver and adipose tissue of these compounds [
37], are not anticipated to play a crucial limiting role in the inhibition of choline microbial conversion into TMA. Indeed, over 95% of ingested phenolic compounds reach the colon lumen [
38], which is the localization where their target of this particular bioactivity is located: the gut microbiota. Thus, considering the systemic low bioavailability of phenolic compounds, gallic acid and chlorogenic reach this specific target destination at the highest possible doses achieved anywhere in the body. Second, gallic acid and chlorogenic acid, as well as their derivates, can be found in high concentrations in commonly consumed fruits and vegetables [
39,
40], and the consumption of these compounds is high [
30]. For example, coffee is a rich source of chlorogenic acid (a cup of coffee of ~200 mL contains between 50 and 150 mg chlorogenic acid) [
41], and ~65% of all phenolic compounds consumed by coffee consumers come from coffee [
42]. Although large differences exist in human colon volumes, the mean value approximates to 560 mL [
43]. This suggests that average chlorogenic acid concentrations, at least in the proximal colon where chlorogenic acid is starting to be metabolized by the gut microbiota and right after its arrival, could range up to 0.25–0.76 mM after consumption of a single cup of coffee. Daily gallic acid consumption has been estimated at 25 mg/day [
44], which suggests that the maximum concentration could be 0.26 mM. However, gallic acid can be produced by the gut microbiota metabolism of gallated flavan-3-ols and anthocyanins [
45,
46,
47]. Tea contains both gallic acid and gallated flavan-3-ols, as well as lower quantities of chlorogenic acid [
48]. For coffee and tea consumers, ~80% of all phenolic compounds consumed come from these two beverages [
49]. It thus seems that concentrations on the lower millimolar scale, such as those determined to inhibit microbial TMA production from choline, are on the order of physiologically relevant concentrations achieved in the colon. Furthermore, common food-derived supplements (functional foods supplemented with chlorogenic acid and/or gallic acid) as well as nutraceuticals could easily achieve the levels employed here.
Overall, we have reported that: (1) DMB 10 mM can be used as a control substance in TMA production inhibition studies; (2) gallic acid and chlorogenic acid (10 mM) are phenolic compounds able to reduce TMA formation at a higher proportion than DMB 10 mM; and (3) the effects reported by DMB, gallic acid and chlorogenic acid (10 mM) are due to non-toxic effects within a period of 12 h. These results open the door to using this developed methodology to study other phenolic compounds in a high-throughput way. However, the main limitation of this study is the fecal slurry itself. Further studies using fecal starters from high TMAO and/or TMA producers should be selected to study the inhibition potential of phenolic compounds, or any other treatments for that matter. Additionally, the source of the fecal slurry can have a great impact on other parameters, such as capacity to metabolize phenolic compounds such as chlorogenic acid [
35,
50], which ultimately could affect choline microbial conversion to TMA. The concentrations of betaine and choline in the fermentation media + fecal slurry are also a drawback in this experimental design. The quantification of choline in all samples in this study was achieved by spiking fermentation media (45% growth media +55% PBS 1X) without fecal slurry with different concentrations of choline to prepare external standard curves. Given the presence of choline in the fermentation media (~225 µM), only exogenously added choline was quantifiable. Thus, the results presented herein do not take into account endogenous choline levels. For example, when we report 100 µM choline in fermentations containing 20% fecal slurry, the background levels have been subtracted, and the real concentration would approximate to 325 µM total choline. This effect was not reported for TMA, as TMA levels were non-detectable in the original fecal slurry and the fermentation media inoculated with fecal slurry 1:10 (20%). This was also true for L-carnitine, γ-butyrobetaine and TMAO, but not for betaine. As a matter of fact, betaine levels were so high in the fecal fermentation inoculated with the fecal slurry 1:10 (20%) that addition of betaine would not produce a linear curve, hence making its absolute quantification problematic. Simply running calibration curves in water does not take into account the matrix effect, which could modulate the quantification of choline and betaine. Rather, calibration curves should be constructed with a matrix as similar as possible. To potentially bypass this issue, results could be presented as percentage of change versus background analyte concentrations, as performed by Bresciani et al. [
20]; use deuterated choline, as performed by Wang et al. [
11]; or study the inhibitory effect of phytochemicals on background choline microbial metabolism into TMA. Finally, other cell viability tests than MTT should be considered for screening studies.
The method presented herein facilitates efficient, rapid screening of large compounds libraries for potential TMA-reducing activity. This will allow rational selection of particularly effective lead compounds for focused animal studies, potentially leading to the development of strategies to benefit specific population groups with elevated TMAO circulating levels. This approach should prove much more effective at identifying lead compounds for human trials, as opposed to the scattered approach that is currently being employed.