Leading Factors for Weight Gain during COVID-19 Lockdown in a Spanish Population: A Cross-Sectional Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Computer-Assisted Telephone Interviewing (CATI)
2.3. Outcomes
2.4. Statistical Analysis
2.5. Statement of Ethics
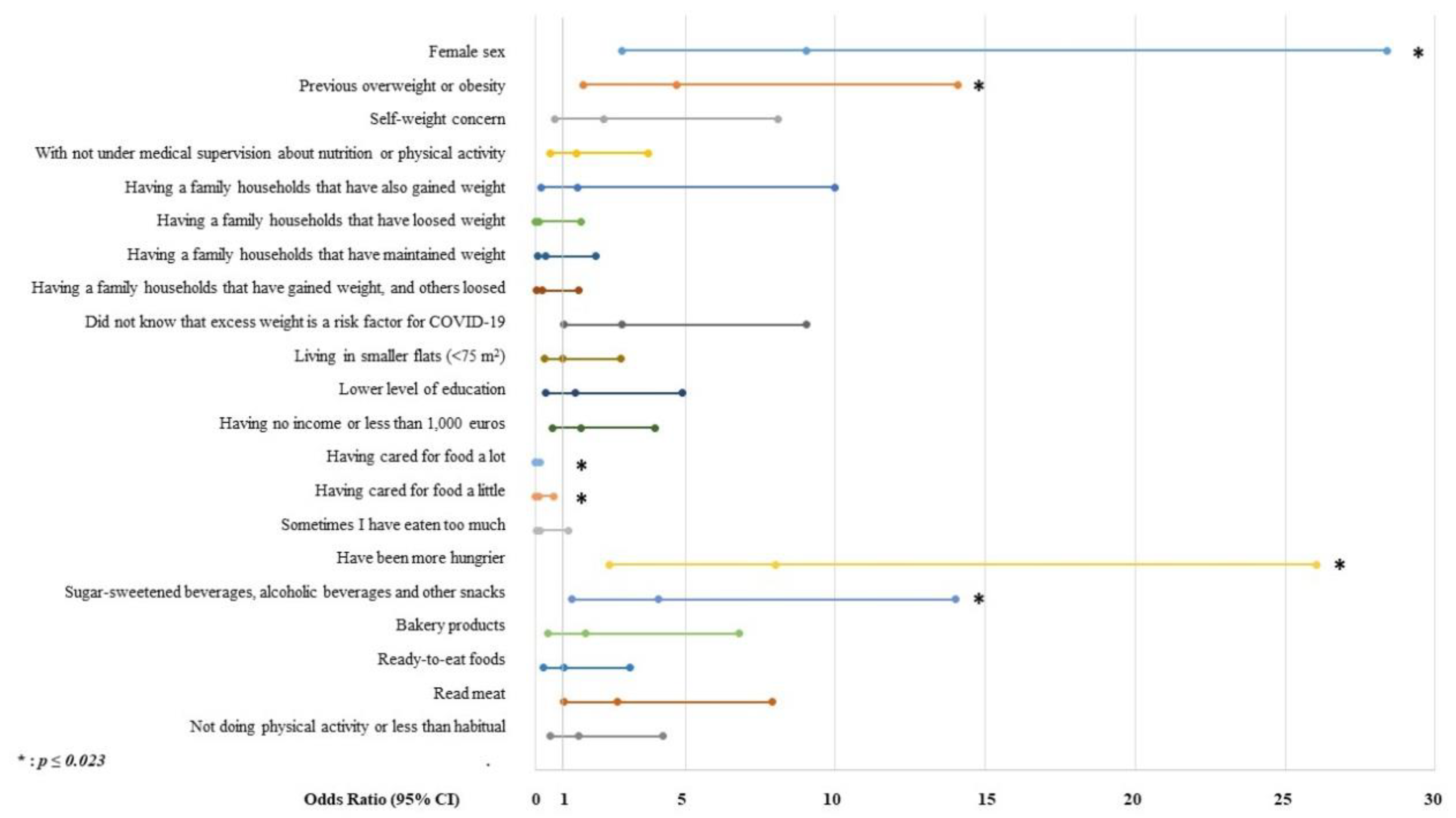
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- World Health Organization. Timeline of WHO’s Response to COVID-19. Available online: https://www.who.int/news-room/detail/29-06-2020-covidtimeline (accessed on 1 September 2020).
- Dong, E.; Du, H.; Gardner, L. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect. Dis. 2020, 20, 533–534. [Google Scholar] [CrossRef]
- Ceylan, Z. Estimation of COVID-19 prevalence in Italy, Spain, and France. Sci. Total Environ. 2020, 729, 138817. [Google Scholar] [CrossRef]
- Wu, Z.; McGoogan, J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72,314 cases from the Chinese center for disease control and prevention. JAMA 2020, 323, 1239–1242. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Flint, S.W.; A Tahrani, A. COVID-19 and obesity—lack of clarity, guidance, and implications for care. Lancet Diabetes Endocrinol. 2020, 8, 474–475. [Google Scholar] [CrossRef]
- Popkin, B.M.; Du, S.; Green, W.D.; Beck, M.A.; Algaith, T.; Herbst, C.H.; Alsukait, R.F.; Alluhidan, M.; Alazemi, N.; Shekar, M. Individuals with obesity and COVID-19: A global perspective on the epidemiology and biological relationships. Obes. Rev. 2020, 21, 13128. [Google Scholar] [CrossRef] [PubMed]
- Stefan, N.; Birkenfeld, A.L.; Schulze, M.B.; Ludwig, D.S. Obesity and impaired metabolic health in patients with COVID-19. Nat. Rev. Endocrinol. 2020, 16, 341–342. [Google Scholar] [CrossRef]
- Simonnet, A.; Chetboun, M.; Poissy, J.; Raverdy, V.; Noulette, J.; Duhamel, A.; Labreuche, J.; Mathieu, D.; Pattou, F.; Jourdain, M.; et al. High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation. Obesity 2020, 28, 1195–1199. [Google Scholar] [CrossRef]
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W.; The Northwell COVID-19 Research Consortium. Presenting Characteristics, Comorbidities, and Outcomes among 5700 Patients Hospitalized With COVID-19 in the New York City Area. JAMA 2020, 323, 2052–2059. [Google Scholar] [CrossRef] [PubMed]
- Van Kerkhove, M.D.; Vandemaele, K.A.; Shinde, V.; Jaramillo-Gutierrez, G.; Koukounari, A.; Donnelly, C.A.; Carlino, L.O.; Owen, R.; Paterson, B.; Pelletier, L.; et al. Risk factors for severe outcomes following 2009 influenza A (H1N1) infection: A global pooled analysis. PLoS Med. 2011, 8, e1001053. [Google Scholar] [CrossRef]
- Aranceta-Bartrina, J.; Pérez-Rodrigo, C.; Alberdi-Aresti, G.; Ramos-Carrera, N.; Lázaro-Masedo, S. Prevalence of general obesity and abdominal obesity in the Spanish adult population (aged 25–64 years) 2014–2015: The ENPE study. Rev. Esp. Cardiol. (Engl. Ed.) 2016, 69, 579–587. [Google Scholar] [CrossRef] [PubMed]
- Ghosal, S.; Bhattacharyya, R.; Majumder, M. Impact of complete lockdown on total infection and death rates: A hierarchical cluster analysis. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 707–711. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, E.; Tisler, A. The mortality and psychological burden caused by response to COVID-19 outbreak. Med. Hypotheses 2020, 143, 110069. [Google Scholar] [CrossRef] [PubMed]
- Ammar, A.; Brach, M.; Trabelsi, K.; Chtourou, H.; Boukhris, O.; Masmoudi, L.; Bouaziz, B.; Bentlage, E.; How, D.; Ahmed, M.; et al. Effects of COVID-19 home confinement on eating behaviour and physical activity: Results of the ECLB-COVID19 international online survey. Nutrients 2020, 12, 1583. [Google Scholar] [CrossRef]
- Di Renzo, L.; Gualtieri, P.; Pivari, F.; Soldati, L.; Attinà, A.; Cinelli, G.; Leggeri, C.; Caparello, G.; Barrea, L.; Scerbo, F.; et al. Eating habits and lifestyle changes during COVID-19 lockdown: An Italian survey. J. Transl. Med. 2020, 18, 229. [Google Scholar] [CrossRef]
- Sidor, A.; Rzymski, P. Dietary choices and habits during COVID-19 lockdown: Experience from Poland. Nutrients 2020, 12, 1657. [Google Scholar] [CrossRef] [PubMed]
- Draugalis, J.R.; Coons, S.J.; Plaza, C.M. Best practices for survey research reports: A synopsis for authors and reviewers. Am. J. Pharm. Educ. 2008, 72, 11. [Google Scholar] [CrossRef] [PubMed]
- Fowler, F.J., Jr.; Brenner, P.S.; Buskirk, T.D.; Roman, A. Telephone health survey estimates: Effects of nonresponse and sample limitations. Health Serv Res. 2019, 54, 700–706. [Google Scholar] [CrossRef]
- World Health Organization. Overweight and Obesity. Available online: https://www.who.int/gho/ncd/risk_factors/overweight/en/ (accessed on 1 September 2020).
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P.; STROBE Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE)statement: Guidelines for reporting observational studies. J. Clin. Epidemiol. 2008, 61, 344–349. [Google Scholar] [CrossRef]
- Kapoor, E.; Collazo-Clavell, M.L.; Faubion, S.S. Weight gain in women at midlife: A concise review of the pathophysiology and strategies for management. Mayo Clin. Proc. 2017, 92, 1552–1558. [Google Scholar] [CrossRef] [PubMed]
- Ghosal, S.; Sinha, B.; Majumder, M.; Misra, A. Estimation of effects of nationwide lockdown for containing coronavirus infection on worsening of glycosylated haemoglobin and increase in diabetes-related complications: A simulation model using multivariate regression analysis. Diabetes Metab. Syndr. 2020, 14, 319–323. [Google Scholar] [CrossRef]
- Dixon, A.E.; Peters, U. The effect of obesity on lung function. Expert. Rev. Respir. Med. 2018, 12, 755–767. [Google Scholar] [CrossRef]
- Pearson, N.; Biddle, S.J. Sedentary behavior and dietary intake in children, adolescents, and adults. A systematic review. Am. J. Prev. Med. 2011, 41, 178–188. [Google Scholar] [CrossRef]
- Ogden, C.L.; Carroll, M.D.; Kit, B.K.; Flegal, K.M. Prevalence of childhood and adult obesity in the United States, 2011–2012. JAMA 2014, 311, 806–814. [Google Scholar] [CrossRef]
- Hayes, A.J.; Kortt, M.A.; Clarke, P.M.; Brandrup, J.D. Estimating equations to correct self-reported height and weight: Implications for prevalence of overweight and obesity in Australia. Aust. N. Z. J. Public Health 2008, 32, 542–545. [Google Scholar] [CrossRef] [PubMed]
- Nyholm, M.; Gullberg, B.; Merlo, J.; Lundqvist-Persson, C.; Råstam, L.; Lindblad, U. The validity of obesity based on self-reported weight and height: Implications for population studies. Obesity 2007, 15, 197–208. [Google Scholar] [CrossRef]
- Waters, H.; DeVol, R. Weighing down America: The Health and Economic Impact of Obesity. Available online: https://assets1b.milkeninstitute.org/assets/Publication/ResearchReport/PDF/Weighing-Down-America-WEB.pdf (accessed on 23 July 2020).
- Meldrum, D.R.; Morris, M.A.; Gambone, J.C. Obesity pandemic: Causes, consequences, and solutions-but do we have the will? Fertil. Steril. 2017, 107, 833–839. [Google Scholar] [CrossRef]
- Santos, M.-D.; Buti, M.; López-Cano, C.; Sánchez, E.; Vidal, A.; Hernández, M.; Lafarga, A.; Gutiérrez-Carrasquilla, L.; Rius, F.; Bueno, M.; et al. Dynamics of anthropometric indices in a large paired cohort with 10 years of follow-up: Paving the way to sarcopenic obesity. Front. Endocrinol. 2020, 11, 209. [Google Scholar] [CrossRef] [PubMed]
- Lecube, A.; Sánchez, E.; Monereo, S.; Medina-Gómez, G.; Bellido, D.; García-Almeida, J.M.; De Icaya, P.M.; Malagón, M.M.; Goday, A.; Tinahones, F.J.; et al. Factors accounting for obesity and its perception among the adult Spanish population: Data from 1000 computer-assisted telephone interviews. Obes. Facts 2020, 13, 322–332. [Google Scholar] [CrossRef]
- Visscher, T.L.; Viet, A.L.; Kroesbergen, I.H.T.; Seidell, J.C. Underreporting of BMI in adults and its effect on obesity prevalence estimations in the period 1998 to 2001*. Obesity 2006, 14, 2054–2063. [Google Scholar] [CrossRef]
- Gorber, C.S.; Tremblay, M.; Moher, D.; Gorber, B. A comparison of direct vs. self-report measures for assessing height, weight and body mass index: A systematic review. Obes. Rev. 2007, 8, 307–326. [Google Scholar] [CrossRef]
- Ministerio de Agricultura. Pesca y Alimentación. Available online: https://www.mapa.gob.es/es/prensa/ultimas-noticias/ (accessed on 25 August 2020).
- Traversy, G.; Chaput, J.P. Alcohol consumption and obesity: An update. Curr. Obes. Rep. 2015, 4, 122–130. [Google Scholar] [CrossRef]
- Shelton, N.J.; Knott, C.S. Association between alcohol calorie intake and overweight and obesity in English adults. Am. J. Public Health 2014, 104, 629–631. [Google Scholar] [CrossRef] [PubMed]
- Sayon-Orea, C.; Bes-Rastrollo, M.; Nuñez-Cordoba, J.M.; Basterra-Gortari, F.J.; Beunza, J.J.; Martinez-Gonzalez, M.A. Type of alcoholic beverage and incidence of overweight/obesity in a Mediterranean cohort: The SUN project. Nutrition 2011, 27, 802–808. [Google Scholar] [CrossRef] [PubMed]
- Cabrera Escobar, M.A.; Veerman, J.L.; Tollman, S.M.; Bertram, M.Y.; Hofman, K.J. Evidence that a tax on sugar sweetened beverages reduces the obesity rate: A meta-analysis. BMC Public Health 2013, 13, 1072. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.; Scarborough, P.; Rayner, M.; Briggs, A.D. Should we tax unhealthy food and drink? Proc. Nutr. Soc. 2018, 77, 314–320. [Google Scholar] [CrossRef]
- Amaro-Gahete, F.J.; Jurado-Fasoli, L.; De-la-O, A.; Gutierrez, Á.; Castillo, M.J.; Ruiz, J.R. Accuracy and validity of resting energy expenditure predictive equations in middle-aged adults. Nutrients 2018, 10, 1635. [Google Scholar] [CrossRef]
- Ricci, F.; Izzicupo, P.; Moscucci, F.; Sciomer, S.; Maffei, S.; Di Baldassarre, A.; Mattioli, A.V.; Gallina, S. Recommendations for physical inactivity and sedentary behavior during the coronavirus disease (COVID-19) pandemic. Front. Public Health 2020, 8, 199. [Google Scholar] [CrossRef]
- Lecube, A.; on behalf of the Obesity Group of the Spanish Society of Endocrinology and Nutrition (GOSEEN); Sánchez, E.; Andrés, A.; Saldaña, C.; Morales, M.J.; Calañas, A.; Miñambres, I.; Pellitero, S.; Cordido, F.; et al. Assessing motivational stages and processes of change for weight management around bariatric surgery: A multicenter study. Obes. Surg. 2019, 29, 3348–3356. [Google Scholar] [CrossRef]
- Balanzá-Martínez, V.; Atienza-Carbonell, B.; Kapczinski, F.; De Boni, R.B. Lifestyle behaviours during the COVID-19–time to connect. Acta Psychiatr. Scand. 2020, 141, 399–400. [Google Scholar] [CrossRef] [PubMed]
- Brooks, S.K.; Webster, R.K.; Smith, L.E.; Woodland, L.; Wessely, S.; Greenberg, N. The psychological impact of quarantine and how to reduce it: Rapid review of the evidence. Lancet 2020, 395, 912–920. [Google Scholar] [CrossRef]
- Zachary, Z.; Brianna, F.; Brianna, L.; Garrett, P.; Jade, W.; Alyssa, D.; Mikayla, K. Self-quarantine and weight gain related risk factors during the COVID-19 pandemic. Obes. Res. Clin. Pract. 2020, 14, 210–216. [Google Scholar] [CrossRef]
- Rabec, C.; de Lucas Ramos, P.; Veale, D. Respiratory complications of obesity. Arch. Bronconeumol. 2011, 47, 252–261. [Google Scholar] [CrossRef]
- Park, H.S.; Park, J.Y.; Yu, R. Relationship of obesity and visceral adiposity with serum concentrations of CRP, TNF-alpha and IL-6. Diabetes Res. Clin. Pract. 2005, 69, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Merad, M.; Martin, J.C. Pathological inflammation in patients with COVID-19: A key role for monocytes and macrophages. Nat. Rev. Immunol. 2020, 20, 355–362. [Google Scholar] [CrossRef] [PubMed]
| Sex, Age, Region, Weight and Height |
|---|
| 1.-Are you worried about your weight? Yes/No |
| 2.-Thinking about before you entered lockdown, would you say that you were overweight? Yes/No |
| 3.-Were you under medical supervision regarding diet or a physical activity regime before lockdown? Yes/No |
| 4.-Have you followed the medical advice provided regarding diet and physical activity? Yes/No |
| 5.-If “no”: Why have you not followed it? It was not possible to follow up with my dietitian, primary care physician, or endocrinologist, etc./It was difficult to follow due to anxiety, stress, etc./It was difficult to purchase healthy products/It was difficult to go to the gym/Other reason |
| 6.-The professional consulted for weight loss: Pharmacist/Primary care physician/Endocrinologist/Dietitian/Friend/Internet |
| 7.-Do you think that lockdown has been an opportunity to follow a healthy lifestyle? Yes/No |
| 8.-How did your weight change during lockdown? I lost weight/I maintained my normal weight/I gained weight (<3 kg)/I gained weight (≥3 kg) |
| 9.-If you gained weight, what was the reason? Greater food intake/Less physical activity/Both factors equally |
| 10.-What happened with the rest of the household’s members? They gained weight/They lost weight/They maintained normal weight/Different weight progressions/I live alone |
| 11.-How many people do you live with? Alone/1/2/3/>3 |
| 12.-Did you know that excess weight is a risk factor for COVID-19? Yes/No |
| 13.-How many square meters does your house have? < 50 m2/50–75 m2/75–150 m2/> 150 m2 |
| 14.-What educational level have you achieved? Primary studies or lower/Secondary studies/University or higher |
| 15.-What is your monthly income? None or <€1000/€1000–2000/€2000–3000/€3000–4000/>€4000 |
| 16.-Which of the following statements is most accurate? I place high importance on the quality of of my food/I place a little importance on the quality of my food/Sometimes I have eating so much/I do not care about the quality of my food |
| 17.-Did you have been hungrier? Yes/No |
| 18.-Did you increase consumption of sugar-sweetened beverages, alcoholic beverages, or other snacks? Yes/No |
| 19.-Did you increase consumption of bakery products? Yes/No |
| 20.-Did you increase consumption of ready-to-eat- foods? Yes/No |
| 21.-Did you increase consumption of red meat? Yes/No |
| 22.-Did you increase consumption of white meat? Yes/No |
| 23.-Did you increase consumption of fish? Yes/No |
| 24.-What was the reason? I had anxiety/I was bored/I was near the fridge/All of the above |
| 25.-How many times have you eaten on average per day? 2/3/> 3/I have been eating continuously |
| 26.-Did you regularly practice physical activity? Yes/no |
| 27.-If you practiced physical activity, would you say that…? I practiced less than habitual/Equal/More than habitual |
| 28.-How many hours did you spend sitting on an average day, including when completing work or schoolwork? 1–2 h/3–4 h/5–7 h/More than 7 h |
| 29.-How many hours did you spend sitting and watching screens on an average day? <1/1–3 h/3–4 h/5–7 h/>7 h |
| All Population (n = 1000) | No Weight Gain (n = 555) | Weight Gain (n = 445) | p | |
|---|---|---|---|---|
| Female, n (%) | 515 (51.5) | 257 (46.3) | 258 (58.0) | <0.001 |
| Age (year) | 51 ± 18 | 51.2 ± 18.2 | 50.9 ± 16.8 | 0.733 |
| Body mass index, (kg/m2) | 25.3 ± 3.9 | 24.2 ± 3.7 | 26.6 ± 3.6 | <0.001 |
| Overweight or obesity, n (%) | 498 (49.8) | 199 (35.9) | 311 (69.9) | <0.001 |
| Worried about their weight, n (%) | 523 (52.3) | 182 (32.8) | 341 (76.6) | <0.001 |
| Self-perception of be overweight, n (%) | 288 (28.8) | 87 (15.7) | 201 (45.2) | <0.001 |
| Under medical supervision for diet or physical activity, n (%) | 458 (45.8) | 284 (51.1) | 174 (39.1) | 0.016 |
| Not following medical advice, n (%) | 60 (44.9) | 10 (20.8) | 50 (62.5) | <0.001 |
| The lockdown is an opportunity to follow a healthier lifestyle, n (%) | 381 (38.1) | 247 (44.5) | 134 (30.1) | <0.001 |
| Having family members who also gained weight, n (%) | 261 (26.1) | 62 (11.2) | 199 (44.7) | <0.001 |
| Did not know that excess weight is a risk factor for COVID-19, n (%) | 488 (48.8) | 226 (40.7) | 262 (58.9) | <0.001 |
| Living in housing of <50 m2, n (%) | 335 (33.5) | 178 (32.1) | 157 (35.3) | <0.001 |
| Primary studies or less? n (%) | 263 (26.3) | 138 (24.8) | 125 (28.1) | 0.002 |
| No income or < €1.000, n (%) | 333 (33.3) | 155 (27.9) | 178 (40.0) | <0.001 |
| Without worrying about food, n (%) | 147 (14.7) | 11 (2.0) | 136 (30.6) | <0.001 |
| Have been hungrier than before the lockdown, n (%) | 421 (42.1) | 94 (16.9) | 327 (73.5) | <0.011 |
| Increased consumption of sugar-sweetened and alcoholic beverages, and other snacks, n (%) | 444 (44.4) | 128 (23.1) | 316 (71.0) | <0.001 |
| Increased consumption of bakery products, n (%) | 462 (46.2) | 137 (24.7) | 325 (73.0) | <0.001 |
| Increased consumption of ready-to-eat foods, n (%) | 220 (22.0) | 59 (10.6) | 161 (32.2) | <0.001 |
| Increased consumption of red meat, n (%) | 264 (26.4) | 107 (19.3) | 157 (35.3) | <0.001 |
| Performed intermittent or irregular physical activity, n (%) | 321 (56.1) | 173 (45.9) | 148 (75.9) | <0.001 |
| Spent >5 h/day sitting watching screens, n (%) | 540 (54.0) | 231 (41.6) | 309 (69.4) | <0.001 |
| Eating continuously, n (%) | 179 (17.9) | 19 (3.4) | 160 (36.2) | <0.001 |
| Weight Gain < 3 kg (n = 327) | Weight Gain ≥ 3 kg (n = 118) | p | |
|---|---|---|---|
| Body mass index, (kg/m2) | 26.2 ± 3.7 | 27.8 ± 3.2 | <0.001 |
| Overweight or obesity, n (%) | 212 (64.8) | 99 (83.9) | <0.001 |
| Worried about their weight, n (%) | 236 (72.2) | 109 (89.0) | <0.001 |
| Self-perception of be overweight, n (%) | 130 (39.8) | 71 (60.2) | <0.001 |
| Under medical supervision for diet or physical activity, n (%) | 59 (49.4) | 21 (29.6) | 0.029 |
| Not following medical advice, n (%) | 33 (55.9) | 17 (81.0) | 0.042 |
| The lockdown is an opportunity to follow a healthier lifestyle, n (%) | 208 (63.6) | 103 (87.3) | <0.001 |
| Did not know that excess weight is a risk factor for COVID-19, n (%) | 179 (54.7) | 83 (70.3) | <0.001 |
| Without worrying about food, n (%) | 75 (22.9) | 61 (51.7) | <0.001 |
| Have been hungrier than before the lockdown, n (%) | 227 (69.4) | 100 (84.7) | <0.001 |
| Increased consumption of sugar-sweetened and alcoholic beverages, and other snacks, n (%) | 211 (64.5) | 105 (89.0) | <0.001 |
| Increased consumption of bakery products, n (%) | 223 (68.2) | 102 (86.4) | <0.001 |
| Increased consumption of ready-to-eat foods, n (%) | 103 (31.5) | 58 (49.2) | <0.001 |
| Spent >5 h/day sitting watching screens, n (%) | 28 (63.6) | 101 (85.6) | <0.001 |
| Eating continuously, n (%) | 97 (29.7) | 64 (54.2) | <0.001 |
| Group 2 (n = 177) | Group 3 (n = 134) | Group 1 (n = 134) | p | |
|---|---|---|---|---|
| Female, n (%) | 177 (100) | 81 (60.4) | 0 (0) | <0.001 |
| Age (year) | 54.1 ± 16.6 | 45.6 ± 17.1 | 52.1 ± 15.5 | <0.001 |
| Body mass index (kg/m2) | 28.1 ± 2.9 | 22.7 ± 1.4 | 28.6 ± 3.0 | <0.001 |
| Overweight or obesity, n (%) | 177 (100) | 0 (0) | 134 (100) | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez, E.; Lecube, A.; Bellido, D.; Monereo, S.; Malagón, M.M.; Tinahones, F.J.; on behalf of the Spanish Society for the Study of Obesity. Leading Factors for Weight Gain during COVID-19 Lockdown in a Spanish Population: A Cross-Sectional Study. Nutrients 2021, 13, 894. https://doi.org/10.3390/nu13030894
Sánchez E, Lecube A, Bellido D, Monereo S, Malagón MM, Tinahones FJ, on behalf of the Spanish Society for the Study of Obesity. Leading Factors for Weight Gain during COVID-19 Lockdown in a Spanish Population: A Cross-Sectional Study. Nutrients. 2021; 13(3):894. https://doi.org/10.3390/nu13030894
Chicago/Turabian StyleSánchez, Enric, Albert Lecube, Diego Bellido, Susana Monereo, María M. Malagón, Francisco J. Tinahones, and on behalf of the Spanish Society for the Study of Obesity. 2021. "Leading Factors for Weight Gain during COVID-19 Lockdown in a Spanish Population: A Cross-Sectional Study" Nutrients 13, no. 3: 894. https://doi.org/10.3390/nu13030894
APA StyleSánchez, E., Lecube, A., Bellido, D., Monereo, S., Malagón, M. M., Tinahones, F. J., & on behalf of the Spanish Society for the Study of Obesity. (2021). Leading Factors for Weight Gain during COVID-19 Lockdown in a Spanish Population: A Cross-Sectional Study. Nutrients, 13(3), 894. https://doi.org/10.3390/nu13030894








