Nutrient Requirements during Pregnancy and Lactation
Abstract
1. Introduction
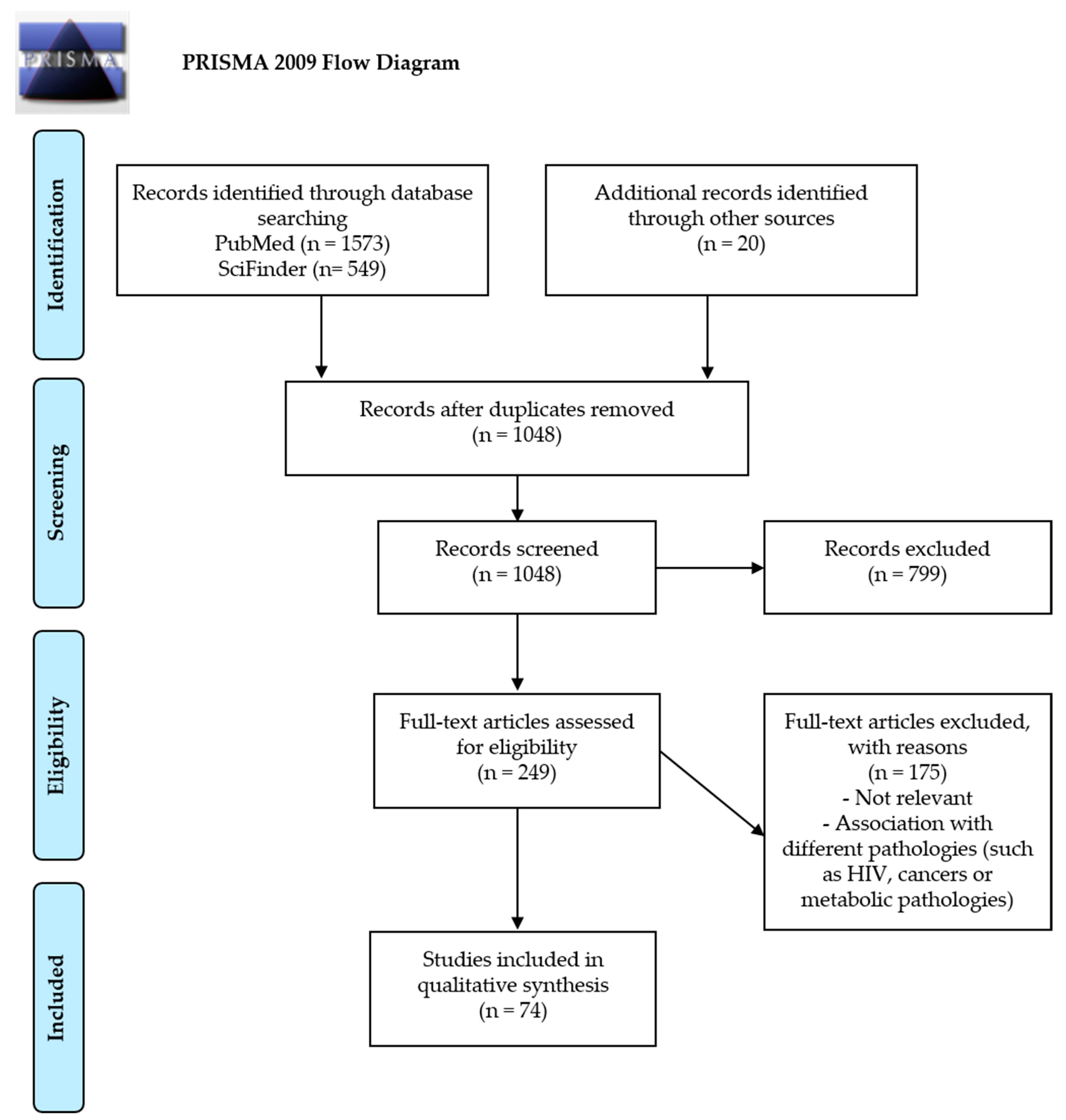
2. Methods
- Recommended dietary allowance (RDA): the average daily dietary intake level that is sufficient to meet the nutrient requirement of nearly all healthy individuals in a group.
- Adequate intake (AI): a value based on observed or experimentally determined approximations of nutrient intake by a group (or groups) of healthy people—used when an RDA cannot be determined.
- Tolerable upper intake level (UL): the highest level of daily nutrient intake that is likely to pose no risk of adverse health effects to almost all individuals in the general population. As intake increases above the UL, the risk of adverse effects increases.
- Estimated average requirement (EAR): a nutrient intake value that is estimated to meet the requirement of half the healthy individuals in a group.
3. Results and Discussion
3.1. Role of Minerals, Vitamins, and Omega-3 Fatty Acids in Pregnancy—Needs and Benefits/Risks of Supplementation
3.1.1. Iron and Vitamin B9
- Hb level <11 g/dL in the first and third trimesters;
- Hb level <10.5 g/dL in the second trimester;
- ferritin level <30 μg/L: insufficient iron reserve.
- Daily supplementation
- Intermittent supplementation
3.1.2. Calcium
- O’Brien et al. [34] highlighted that intakes of dietary calcium <1000 mg/day, particularly for pregnancies in winter (where vitamin D stocks are low due to low sunshine) were associated with increased bone resorption. They concluded that calcium supplementation improves bone resorption at the end of pregnancy, particularly for winter pregnancies.
- A recent Cochrane review [35] on the effects of calcium supplementation during pregnancy in preventing hypertensive disorders and related problems has identified 24 studies that evaluated the effects of high-dose supplementation (≥ 1000 mg/day versus placebo) or low-dose supplementation (<1000 mg/day versus placebo). The authors conclude that high-dose calcium supplementation may reduce the risk of pre-eclampsia and preterm births, especially for populations with low-calcium diets. It also appears that supplementation with low doses would reduce the risk of pre-eclampsia and hypertension. They moderate these conclusions by specifying that additional research with larger and better-quality clinical trials is necessary not only to confirm these effects, but also to verify that this supplementation does not have any adverse effects, particularly for the fetus.
3.1.3. Magnesium
3.1.4. Iodine
3.1.5. Zinc
3.1.6. Vitamin D
- Probably reduces the risk of pre-eclampsia, the risk of gestational diabetes, and the risk of having a baby with low birthweight (less than 2500 g).
- May make little or no difference to the risk of preterm birth.
- In terms of maternal adverse events, vitamin D supplementation may reduce the risk of severe postpartum hemorrhage, although it should be noted that this result is based on findings from a single trial and was an unexpected finding that has not been documented before by any other study.
3.1.7. Vitamin A
3.1.8. Other B-Group Vitamins
3.1.9. Vitamins E and C Supplementation
3.1.10. Omega-3 Fatty Acids
3.2. Micronutrients and Omega-3 Fatty Acid Requirements for Breastfeeding Women
4. Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Heidemann, B.H.; McClure, J.H. Changes in maternal physiology during pregnancy. BJA CEPD Rev. 2003, 3, 65–68. [Google Scholar] [CrossRef]
- Soma-Pillay, P.; Nelson-Piercy, C.; Tolppanen, H.; Mebazaa, A. Physiological changes in pregnancy. Cardiovasc. J. Afr. 2016, 27, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Weight Gain during Pregnancy: Reexamining the Guidelines; National Academies Press: Washington, DC, USA, 2009. [Google Scholar]
- Magon, N.; Kumar, P. Hormones in pregnancy. Niger. Med. J. 2012, 53, 179–183. [Google Scholar] [CrossRef]
- Tkachenko, O.; Shchekochikhin, D.; Schrier, R.W. Hormones and Hemodynamics in Pregnancy. Int. J. Endocrinol. Metab. 2014, 12, e14098. [Google Scholar] [CrossRef]
- Truchet, S.; Honvo-Houéto, E. Physiology of milk secretion. Pract. Res. Clin. Endocrinol. Metab. 2017, 31, 367–384. [Google Scholar] [CrossRef]
- Mensink, G.B.M.; Fletcher, R.; Gurinovic, M.; Huybrechts, I.; Lafay, L.; Serra-Majem, L.; Szponar, L.; Tetens, I.; Verkaik-Kloosterman, J.; Baka, A.; et al. Mapping low intake of micronutrients across Europe. Br. J. Nutr. 2013, 110, 755–773. [Google Scholar] [CrossRef]
- Lips, P.; Cashman, K.D.; Lamberg-Allardt, C.; Bischoff-Ferrari, H.A.; Obermayer-Pietsch, B.; Bianchi, M.L.; Stepan, J.; El–Hajj, F.G.; Bouillon, R. Current vitamin D status in European and Middle East countries and strategies to prevent vitamin D deficiency: A position statement of the European Calcified Tissue Society. Eur. J. Endocrinol. 2019, 180, 23–54. [Google Scholar] [CrossRef] [PubMed]
- Serra-Majem, L.; Aranceta, J. Nutritional objectives for the Spanish population. Consensus from the Spanish Society of Community Nutrition. Public Health Nutr. 2001, 4, 1409–1413. [Google Scholar] [CrossRef]
- Milman, N.; Taylor, C.L.; Merkel, J.; Brannon, P.M. Iron status in pregnant women and women of reproductive age in Europe. Am. J. Clin. Nutr. 2017, 106, 1655S–1662S. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. The PRISMA Group PRISMA 2009 Flow Diagram. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Institute of Medicine. Dietary Reference Intakes. A Risk Assessment Model for Establishing Upper Intake Levels for Nutrients; The National Academies Press: Washington, DC, USA, 1998. [Google Scholar]
- WHO. WHO Recommendations on Antenatal Care for a Positive Pregnancy Experience; WHO Press: Geneva, Switzerland, 2016; ISBN 9789241549912. [Google Scholar]
- Kocyłowski, R.; Lewicka, I.; Grzesiak, M.; Gaj, Z.; Sobanska, A.; Poznaniak, J.; Von Kaisenberg, C.; Suliburska, J. Assessment of dietary intake and mineral status in pregnant women. Arch. Gynecol. Obstet. 2018, 297, 1433–1440. [Google Scholar] [CrossRef]
- Beucher, G.; Grossetti, E.; Simonet, T.; Leporrier, M.; Dreyfus, M. Anémie par carence martiale et grossesse. Prévention et traitement. J. Gynécol. Obs. Biol. Reprod. 2011, 40, 185–200. [Google Scholar] [CrossRef]
- Ayoubi, J.M.; Hirt, R.; Badiou, W.; Hininger-Favier, I.; Favier, M.; Zraik-Ayoubi, F.; Berrebi, A.; Pons, J.C. Nutrition et femmes enceintes. EMC Gyn. Obst. 2012. [Google Scholar] [CrossRef]
- Kominiarek, M.A.; Rajan, P. Nutrition Recommendations in Pregnancy and Lactation. Med. Clin. N. Am. 2016, 100, 1199–1215. [Google Scholar] [CrossRef]
- Zijp, I.M.; Korver, O.; Tijburg, L.B.M. Effect of Tea and Other Dietary Factors on Iron Absorption. Crit. Rev. Food Sci. Nutr. 2000, 40, 371–398. [Google Scholar] [CrossRef]
- Fuzi, S.F.A.; Koller, D.; Bruggraber, S.; Pereira, D.I.; Dainty, J.R.; Mushtaq, S. A 1-h time interval between a meal containing iron and consumption of tea attenuates the inhibitory effects on iron absorption: A controlled trial in a cohort of healthy UK women using a stable iron isotope. Am. J. Clin. Nutr. 2017, 106, 1413–1421. [Google Scholar] [CrossRef]
- Sentilhes, L.; Maillard, F.; Brun, S.; Madar, H.; Merlot, N.; Goffinet, F.; Deneux-Tharaux, C. Risk factors for chronic posttraumatic stress disorder development one year after vaginal delivery: A prospective, observational study. Sci. Rep. 2017, 7, 8724–8732. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, J.; Bell, S.J.; Guan, Y.; Yu, Y.-H. Folic Acid Supplementation and Pregnancy: More than Just Neural Tube Defect Prevention. Rev. Obstet. Gynecol. 2011, 4, 52–59. [Google Scholar] [PubMed]
- Argyridis, S. Folic acid in pregnancy. Obstet. Gynaecol. Reprod. Med. 2019, 29, 118–120. [Google Scholar] [CrossRef]
- Winkels, R.M.; Brouwer, I.; Siebelink, E.; Katan, M.B.; Verhoef, P. Bioavailability of food folates is 80% of that of folic acid. Am. J. Clin. Nutr. 2007, 85, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Öhrvik, V.E.; Witthöft, C.M. Human Folate Bioavailability. Nutrients 2011, 3, 475–490. [Google Scholar] [CrossRef]
- Wilson, R.D.; Davies, G.; Désilets, V.; Reid, G.J.; Summers, A.; Wyatt, P.; Young, D. The use of folic acid for the prevention of neural tube defects and other congenital anomalies. J. Obstet. Gynaecol. Can. 2003, 25, 959–973. [Google Scholar]
- Patel, K.R.; Sobczyńska-Malefora, A. The adverse effects of an excessive folic acid intake. Eur. J. Clin. Nutr. 2016, 71, 159–163. [Google Scholar] [CrossRef]
- Allen, L. Pregnancy: Nutrient Requirements. In Encyclopedia of Human Nutrition; Elsevier BV: Amsterdam, The Netherlands, 2013; pp. 61–67. [Google Scholar]
- De-Regil, L.M.; Peña-Rosas, J.P.; Fernández-Gaxiola, A.C.; Rayco-Solon, P. Effects and safety of periconceptional oral folate supplementation for preventing birth defects. Cochrane Database Syst. Rev. 2015, 12, CD007950. [Google Scholar] [CrossRef]
- Peña-Rosas, J.P.; De-Regil, L.M.; Gomez Malave, H.; Flores-Urrutia, M.C.; Dowswell, T. Intermittent oral iron supplementation during pregnancy. Cochrane Database Syst. Rev. 2015, 10, CD009997. [Google Scholar] [CrossRef] [PubMed]
- Zhou, K.; West, H.M.; Zhang, J.; Xu, L.; Li, W. Interventions for leg cramps in pregnancy. Cochrane Database Syst. Rev. 2015, 8, 010655. [Google Scholar] [CrossRef] [PubMed]
- Hammar, M.; Larsson, L.; Tegler, L. Calcium treatment of leg cramps in pregnancy: Effect on clinical symptoms and total serum and ionized serum calcium concentrations. Acta Obstet. Gynecol. Scand. 1981, 60, 345–347. [Google Scholar] [CrossRef] [PubMed]
- Hammar, M.; Berg, G.; Solheim, F.; Larsson, L. Calcium and magnesium status in pregnant women. A comparison between treatment with calcium and vitamin C in pregnant women with leg cramps. Int. J. Vitam. Nutr. Res. 1987, 57, 179–183. [Google Scholar] [PubMed]
- Sohrabvand, F.; Shariat, M.; Haghollahi, F. Vitamin B supplementation for leg cramps during pregnancy. Int. J. Gynecol. Obstet. 2006, 95, 48–49. [Google Scholar] [CrossRef]
- O’Brien, E.C.; Kilbane, M.T.; McKenna, M.J.; Segurado, R.; Geraghty, A.A.; McAuliffe, F.M. Calcium intake in winter pregnancy attenuates impact of vitamin D inadequacy on urine NTX, a marker of bone resorption. Eur. J. Nutr. 2017, 57, 1015–1023. [Google Scholar] [CrossRef] [PubMed]
- Hofmeyr, G.J.; Lawrie, T.; Atallah, Á.N.; Torloni, M.R. Calcium supplementation during pregnancy for preventing hypertensive disorders and related problems. Cochrane Database Syst. Rev. 2018, 10, 001059. [Google Scholar] [CrossRef] [PubMed]
- Buppasiri, P.; Lumbiganon, P.; Thinkhamrop, J.; Ngamjarus, C.; Laopaiboon, M.; Medley, N. Calcium supplementation (other than for preventing or treating hypertension) for improving pregnancy and infant outcomes. Cochrane Database Syst. Rev. 2015, 2, CD007079. [Google Scholar] [CrossRef]
- Spätling, L.; Disch, G.; Classen, H.G. Magnesium in pregnant women and the newborn. Magnes. Res. 1989, 2, 271–280. [Google Scholar] [PubMed]
- Makrides, M.; Crowther, C. Magnesium supplementation in pregnancy. Cochrane Database Syst. Rev. 2001, 4, CD000937. [Google Scholar] [CrossRef]
- Spätling, L.; Kunz, P.A.; Huch, R.; Huch, A. Magnesium and calcium excretion during pregnancy. Mag. Bull. 1985, 3, 91–93. [Google Scholar]
- Spätling, L.; Classen, H.G.; Kisters, K.; Liebscher, U.; Rylander, R.; Vierling, W.; Von Ehrlich, B.; Vormann, J. Supplementation of Magnesium in Pregnancy. J. Pregnancy Child Health 2017, 4, 1–6. [Google Scholar] [CrossRef]
- Morton, A. Hypomagnesaemia and pregnancy. Obstet. Med. 2018, 11, 67–72. [Google Scholar] [CrossRef]
- Gröber, U.; Schmidt, J.; Kisters, K. Magnesium in Prevention and Therapy. Nutrients 2015, 7, 8199–8226. [Google Scholar] [CrossRef]
- Dahle, L.O.; Berg, G.; Hammar, M.; Hurtig, M.; Larsson, L. The effect of oral magnesium substitution on pregnancy-induced leg cramps. Am. J. Obstet. Gynecol. 1995, 173, 175–180. [Google Scholar] [CrossRef]
- Nygaard, I.H.; Valbø, A.; Pethick, S.V.; Bøhmer, T. Does oral magnesium substitution relieve pregnancy-induced leg cramps? Eur. J. Obstet. Gynecol. Reprod. Biol. 2008, 141, 23–26. [Google Scholar] [CrossRef] [PubMed]
- Supakatisant, C.; Phupong, V. Oral magnesium for relief in pregnancy–induced leg cramps: A randomized controlled trial. Matern. Child Nutr. 2015, 11, 139–145. [Google Scholar] [CrossRef]
- Zimmermann, M.B. The Effects of Iodine Deficiency in Pregnancy and Infancy. Paediatr. Périnat. Epidemiol. 2012, 26, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Shah, D.; Sachdev, H. Zinc Deficiency in Pregnancy and Fetal Outcome. Nutr. Rev. 2006, 64, 15–30. [Google Scholar] [CrossRef] [PubMed]
- Maxfield, L.; Crane, J.S. Zinc Deficiency; StatPearls Publishing: Treasure Island, FL, USA, 2020; Available online: https://www.ncbi.nlm.nih.gov/books/NBK493231 (accessed on 27 December 2020).
- King, J.C. Determinants of maternal zinc status during pregnancy. Am. J. Clin. Nutr. 2000, 71, S1334–S1343. [Google Scholar] [CrossRef] [PubMed]
- Ota, E.; Mori, R.; Middleton, P.; Tobe-Gai, R.; Mahomed, K.; Miyazaki, C.; Bhutta, Z. Zinc supplementation for improving pregnancy and infant outcome. Cochrane Database Syst. Rev. 2015, 2015, CD000230. [Google Scholar] [CrossRef]
- Whittaker, P. Iron and zinc interactions in humans. Am. J. Clin. Nutr. 1998, 68, 442S–446S. [Google Scholar] [CrossRef] [PubMed]
- Paulson, S.; DeLuca, H. Review article: Vitamin D metabolism during pregnancy. Bone 1986, 7, 331–336. [Google Scholar] [CrossRef]
- Beaufrère, B.; Bresson, J.L.; Briend, A.; Farriaux, J.P.; Ghisolfi, J.; Navarro, J.; Rey, J.; Ricour, C.; Rieu, D.; Vidailhet, M.; et al. La supplémentation en vitamine D durant la grossesse: Une nécessité. Arch. Pediatr. 1995, 2, 373–376. [Google Scholar]
- Hellouin de Menibos, C.; Mallet, E.; Henocq, A.; Lemeur, H.; L’Hostis, C. Hypocalcémie néonatale. Résultats de la supplémentation de la mère en vitamine D. Etude portant chez 13377 nouveau–nés. Bull. Acad. Natl. Med. 1990, 174, 1051–1060. [Google Scholar]
- Palacios, C.; Kostiuk, L.K.; Peña-Rosas, J.P. Vitamin D supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2019, 7, 008873. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Antenatal Care Recommendations for a Positive Pregnancy Experience. Nutritional Interventions Update: Vitamin D Supplements During Pregnancy; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Holick, M.F. A Call to Action: Pregnant Women In-Deed Require Vitamin D Supplementation for Better Health Outcomes. J. Clin. Endocrinol. Metab. 2018, 104, 13–15. [Google Scholar] [CrossRef] [PubMed]
- EFSA NDA Panel (EFSA Panel on Dietetic Products, Nutrition and Allergies). Scientific Opinion on dietary Reference Values for vitamin A. EFSA J. 2015, 13, 4028. [Google Scholar] [CrossRef]
- McCauley, M.; Broek, N.V.D.; Dou, L.; Othman, M. Vitamin A supplementation during pregnancy for maternal and newborn outcomes. Cochrane Database Syst. Rev. 2015, 10, CD008666. [Google Scholar] [CrossRef] [PubMed]
- Salam, R.; Zuberi, N.F.; Bhutta, Z.A. Pyridoxine (vitamin B6) supplementation during pregnancy or labour for maternal and neonatal outcomes. Cochrane Database Syst. Rev. 2015, 6, CD000179. [Google Scholar] [CrossRef] [PubMed]
- Rumbold, A.; Ota, E.; Hori, H.; Miyazaki, C.; Crowther, C. Vitamin E supplementation in pregnancy. Cochrane Database Syst. Rev. 2015, 9, CD004069. [Google Scholar] [CrossRef]
- Rumbold, A.; Crowther, C. Vitamin C supplementation in pregnancy. Cochrane Database Syst. Rev. 2005, 9, CD004072. [Google Scholar] [CrossRef]
- Medeiros, D.M. Dietary Reference Intakes: The Essential Guide to Nutrient Requirements. Am. J. Clin. Nutr. 2007, 85, 924. [Google Scholar] [CrossRef]
- Guesnet, P.; Alessandri, J.M. Acides gras polyinsaturés du lait et développement du système nerveux central du nouveau–né. Cah. Nutr. Diet. 1995, 30, 109–111. [Google Scholar] [CrossRef]
- Kadawathagedara, M.; Kersuzan, C.; Wagner, S.; Tichit, C.; Gojard, S.; Charles, M.A.; Lioret, S.; De Lauzon-Guillain, B. Adéquation des consommations alimentaires des femmes enceintes de l’étude ELFE aux recommandations du Programme national nutrition santé. Cah. Nutr. Diététique 2017, 52, 78–88. [Google Scholar] [CrossRef][Green Version]
- Coletta, J.M.; Bell, S.J.; Roman, A.S. Omega–3 Fatty acids and pregnancy. Rev. Obstet. Gynecol. 2010, 3, 163–171. [Google Scholar] [PubMed]
- Institute of Medicine. Nutrition During Lactation; National Academies Press: Washington, DC, USA, 1991. [Google Scholar]
- Gartner, L.M. Prevention of Rickets and Vitamin D Deficiency: New Guidelines for Vitamin D Intake. Pediatrics 2003, 111, 908–910. [Google Scholar] [CrossRef]
- Butte, N.F.; Calloway, D.H. Evaluation of lactational performance of Navajo women. Am. J. Clin. Nutr. 1981, 34, 2210–2215. [Google Scholar] [CrossRef] [PubMed]
- Hollis, B.; Lambert, P.W.; Horst, R.L. Factors affecting the antirachitic sterol content of native milk. In Perinatal Calcium and Phosphorous Metabolism; Holick, M.F., Gray, T.K., Anast, C.S., Eds.; Elsevier Science Publishers BV: Amsterdam, The Netherlands, 1983; 157p. [Google Scholar]
- Kries, R.V.; Shearer, M.; McCarthy, P.T.; Haug, M.; Harzer, G.; Göbel, U. Vitamin K1 Content of Maternal Milk: Influence of the Stage of Lactation, Lipid Composition, and Vitamin K1 Supplements Given to the Mother. Pediatr. Res. 1987, 22, 513–517. [Google Scholar] [CrossRef]
- Prentice, A. Calcium Supplementation during Breast-Feeding. New Engl. J. Med. 1997, 337, 558–559. [Google Scholar] [CrossRef] [PubMed]
- Hollis, B.W.; Wagner, C.L.; Howard, C.R.; Ebeling, M.; Shary, J.R.; Smith, P.G.; Taylor, S.N.; Morella, K.; Lawrence, R.A.; Hulsey, T.C. Maternal Versus Infant Vitamin D Supplementation During Lactation: A Randomized Controlled Trial. Pediatrics 2015, 136, 625–634. [Google Scholar] [CrossRef]
- Innis, S.M.; Friesen, R.W. Essential n−3 fatty acids in pregnant women and early visual acuity maturation in term infants. Am. J. Clin. Nutr. 2008, 87, 548–557. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, D.R.; Theuer, R.C.; Castañeda, Y.S.; Wheaton, D.H.; Bosworth, R.G.; O’Connor, A.R.; Morale, S.E.; Wiedemann, L.E.; Birch, E.E. Maturation of Visual Acuity Is Accelerated in Breast-Fed Term Infants Fed Baby Food Containing DHA-Enriched Egg Yolk. J. Nutr. 2004, 134, 2307–2313. [Google Scholar] [CrossRef]
- Guesnet, P.; Pugo-Gunsam, P.; Maurage, C.; Pinault, M.; Giraudeau, B.; Alessandri, J.-M.; Durand, G.; Antoine, J.-M.; Couet, C. Blood lipid concentrations of docosahexaenoic and arachidonic acids at birth determine their relative postnatal changes in term infants fed breast milk or formula. Am. J. Clin. Nutr. 1999, 70, 292–298. [Google Scholar] [CrossRef]
- World Health Organization. The Micronutrient Initiative. In Safe Vitamin A Dosage during Pregnancy and Lactation Recommendations and Report of a Consultation; World Health Organization: Geneva, Switzerland, 1998. [Google Scholar]
- WHO; UNICEF; UNU. Iron Deficiency Anaemia Assessment, Prevention, and Control: A Guide for Programme Managers; World Health Organization: Geneva, Switzerland, 2001. [Google Scholar]
- Stoltzfus, R.J.; Dreyfuss, M.L. Guidelines for the Use of Iron Supplements to Prevent and Treat Iron Deficiency Anemia; ILSI Press: Washington, DC, USA, 1998; ISBN 1-57881-020-5. [Google Scholar]
- Tang, A.M.; Chung, M.; Dong, K.; Terrin, N.; Edmonds, A.; Assefa, N.; Chetty, T.; Ramlal, R.; Christian, P.; West, K.; et al. Determining a Global Mid-Upper Arm Circumference Cutoff to Assess Malnutrition in Pregnant Women; FHI 360/Food and Nutrition Technical Assistance III Project (FANTA): Washington, DC, USA, 2016. [Google Scholar]
- World Health Organization. Food and Agriculture Organization of the United Nations. Vitamin and Mineral Requirements in Human Nutrition, 2nd ed.; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Allen, L.H. MMN in pregnancy and lactation: An overview. Am. J. Clin. Nutr. 2005, 81, S1206–S1212. [Google Scholar] [CrossRef] [PubMed]
- Bhutta, Z.A.; Ahmed, T.; Black, R.E.; Cousens, S.; Dewey, K.; Giugliani, E.; Haider, B.A.; Kirkwood, B.; Morris, S.S.; Sachdev, H.P.S.; et al. Maternal and Child Undernutrition Study. Lancet 2008, 371, 417–440. [Google Scholar] [CrossRef]
- WHO. Nutritional Anaemias: Tools for Effective Prevention and Control; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- United Nations Children’s Fund (UNICEF); World Health Organization (WHO). UNICEF—WHO Low Birthweight Estimates: Levels and Trends 2000–2015; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
| Nutrient | RDA, Adult Non-Pregnant Women | EAR, Pregnancy | RDA, Pregnancy | UL, Pregnancy | Justifications |
|---|---|---|---|---|---|
| Vitamin A (μg/day) | 700 | 550 | 770 | 3000 | Regulation of genome expression and in cell differentiation |
| Vitamin D (μg/day) | 15 | 10 | 15 | 100 | Mineralization of the fetal skeleton and decreased risk of hypocalcemia accidents and symptomatic osteomalacia |
| Vitamin E (mg/day) | 15 | 12 | 15 | 1000 | – |
| Vitamin K (μg/day) | 90 | – | 90 | none | – |
| Vitamin B1 (mg/day) | 1.1 | 1.2 | 1.4 | none | – |
| Vitamin B2 (mg/day) | 1.1 | 1.2 | 1.4 | none | – |
| Vitamin B3 (mg/day) | 14 | 14 | 18 | 35 | – |
| Vitamin B6 (mg/day) | 1.3 | 1.6 | 1.9 | 100 | Relieve nausea in early pregnancy |
| Vitamin B9 (μg/day) | 400 | 520 | 600 | 1000 | Decreases the risk of spina bifida and other neural tube defects |
| Vitamin B12 (mg/day) | 2.4 | 2.2 | 2.6 | none | – |
| Vitamin C (mg/day) | 75 | 70 | 85 | 2000 | – |
| Calcium (mg/day) | 1000 | 800 | 1000 | 2500 | Mineralization of the fetal skeleton Prevents pre-eclampsia |
| Iodine (μg/day) | 150 | 160 | 220 | 1100 | Maintenance of thyroid homeostasis |
| Iron (mg/day) | 18 | 22 | 27 | 45 | Decreases the risk of having a low birth weight or a premature baby |
| Magnesium (mg/day) | 320 | 290 | 350 | 350 | Involvement in the occurrence of hypertensive disorders, gestational diabetes mellitus, preterm labor, or intrauterine growth retardation |
| Phosphorus (mg/day) | 700 | 580 | 700 | 3500 | – |
| Selenium (μg/day) | 55 | 49 | 60 | 400 | – |
| Zinc (mg/day) | 8 | 9.5 | 11 | 40 | Involvement in cell division, protein synthesis and growth, nucleic acid metabolism |
| Recommendations | Justifications for Breastfeeding | |
|---|---|---|
| Calcium | 1000 mg/day | Maintenance and production of breast milk |
| Magnesium | 390 mg/day | Muscle relaxant Prevention of constipation |
| Zinc | 19 mg/day | Participation in postpartum healing |
| Vitamin C | 130 mg/day | Stimulation of immune functions |
| Vitamin D | 10 µg/day = 400 IU a/day | Important contribution to obtain good quality milk |
| Vitamin A | 10,000 IU/day or max 25,000 IU/week or unique intake 200,000 IU | Only in deficient populations, as soon as possible after childbirth, but not more than 8 weeks afterwards |
| Iron | 60 mg/day | Prevention of maternal anemia For 3 months after postpartum |
| Vitamin B9 | 400 µg/day | |
| Omega–3 | 100 mg/day of DHA b during the 1st year of the newborn’s life | Newborn brain development |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jouanne, M.; Oddoux, S.; Noël, A.; Voisin-Chiret, A.S. Nutrient Requirements during Pregnancy and Lactation. Nutrients 2021, 13, 692. https://doi.org/10.3390/nu13020692
Jouanne M, Oddoux S, Noël A, Voisin-Chiret AS. Nutrient Requirements during Pregnancy and Lactation. Nutrients. 2021; 13(2):692. https://doi.org/10.3390/nu13020692
Chicago/Turabian StyleJouanne, Marie, Sarah Oddoux, Antoine Noël, and Anne Sophie Voisin-Chiret. 2021. "Nutrient Requirements during Pregnancy and Lactation" Nutrients 13, no. 2: 692. https://doi.org/10.3390/nu13020692
APA StyleJouanne, M., Oddoux, S., Noël, A., & Voisin-Chiret, A. S. (2021). Nutrient Requirements during Pregnancy and Lactation. Nutrients, 13(2), 692. https://doi.org/10.3390/nu13020692