The Edible Brown Seaweed Sargassum horneri (Turner) C. Agardh Ameliorates High-Fat Diet-Induced Obesity, Diabetes, and Hepatic Steatosis in Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Powder and Extracts of S. horneri
2.2. Animal Experiments and Dietary Treatment
2.3. Serum Biochemical Analyses
2.4. Liver Lipid Analyses
2.5. Histological Analyses
2.6. Glucose Tolerance Test
2.7. Fecal Analyses
2.8. Lipase Assay
2.9. Statistical Analysis
3. Results
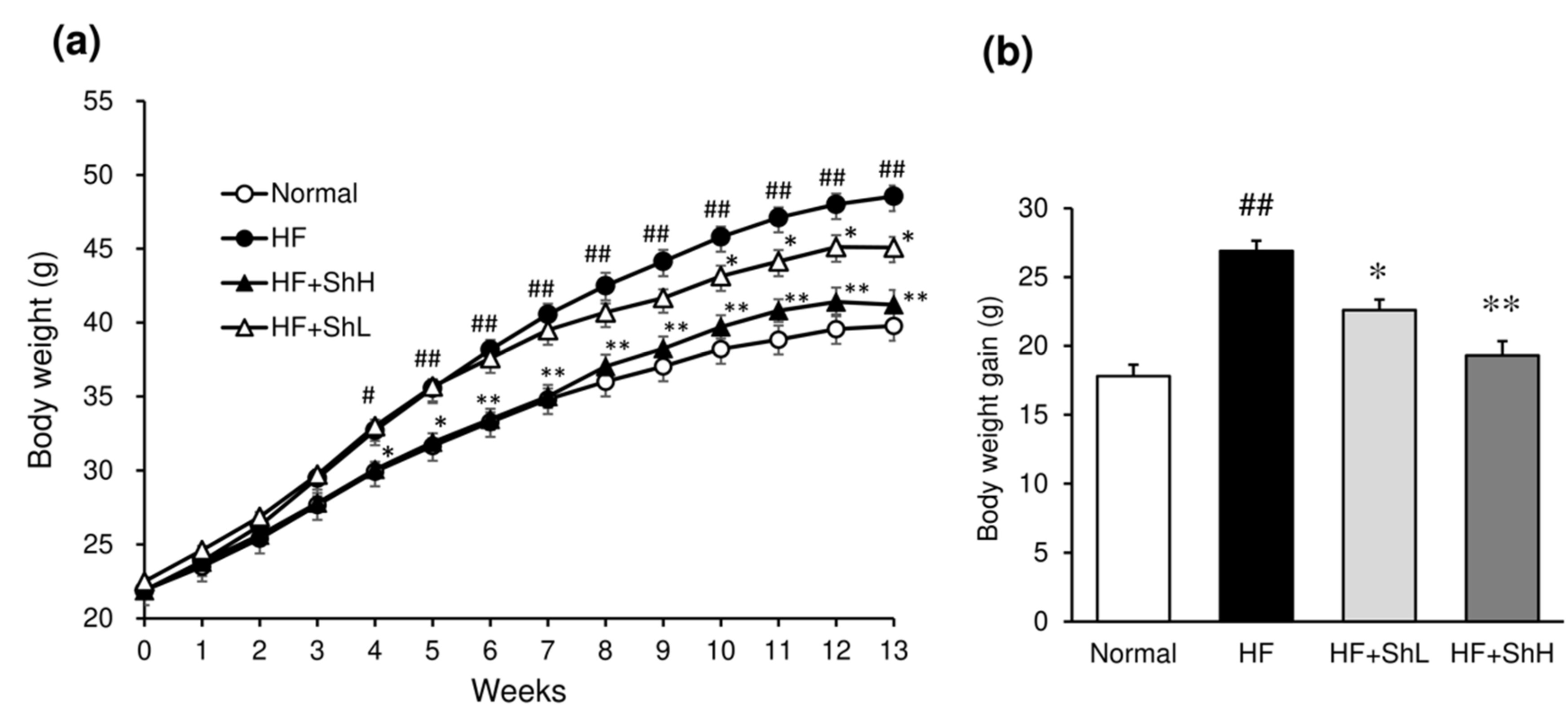
3.1. Effects of S. horneri on Food Intake and Body Weight
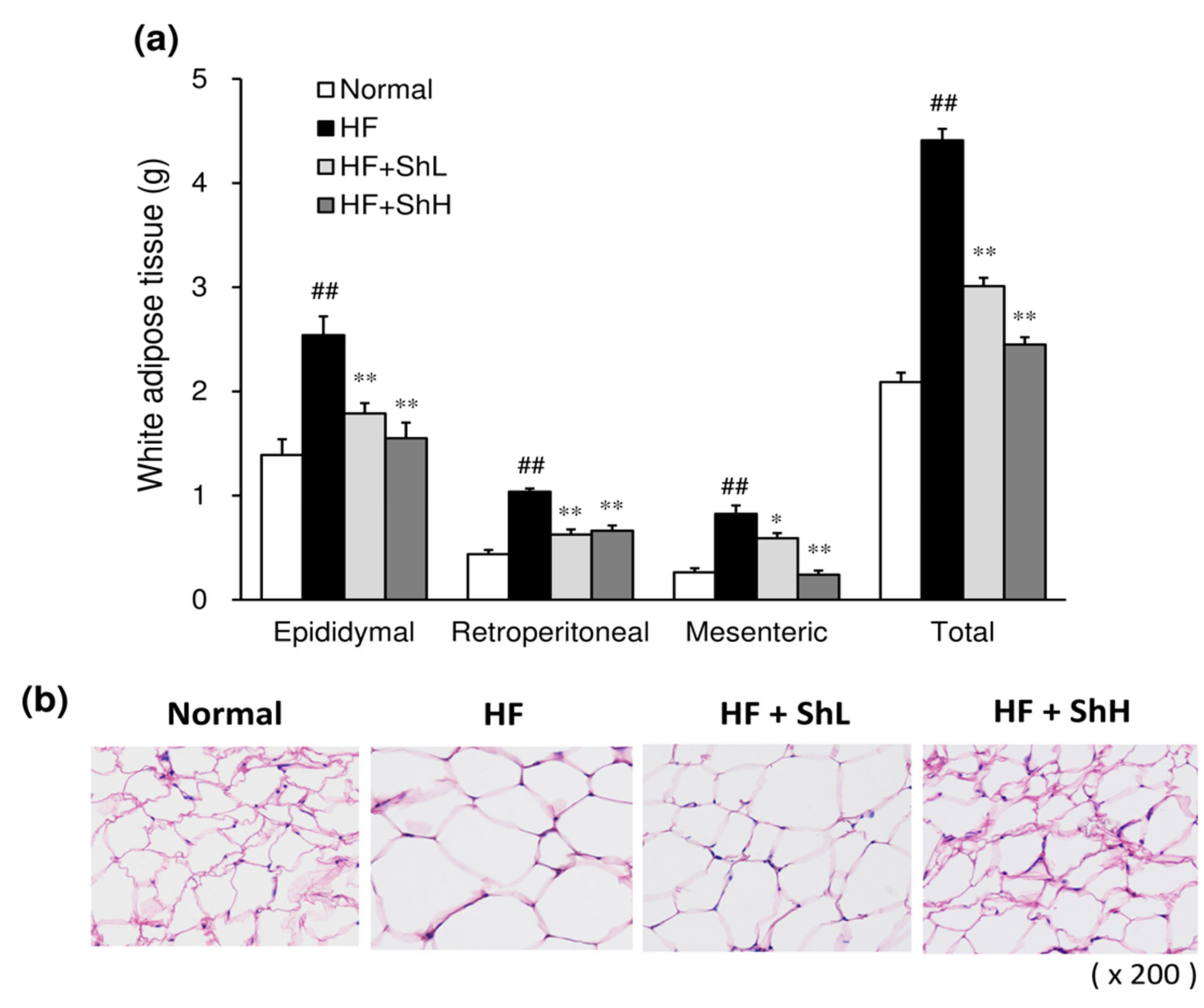
3.2. Effects of S. horneri on the Mass and Morphology of White Adipose Tissue
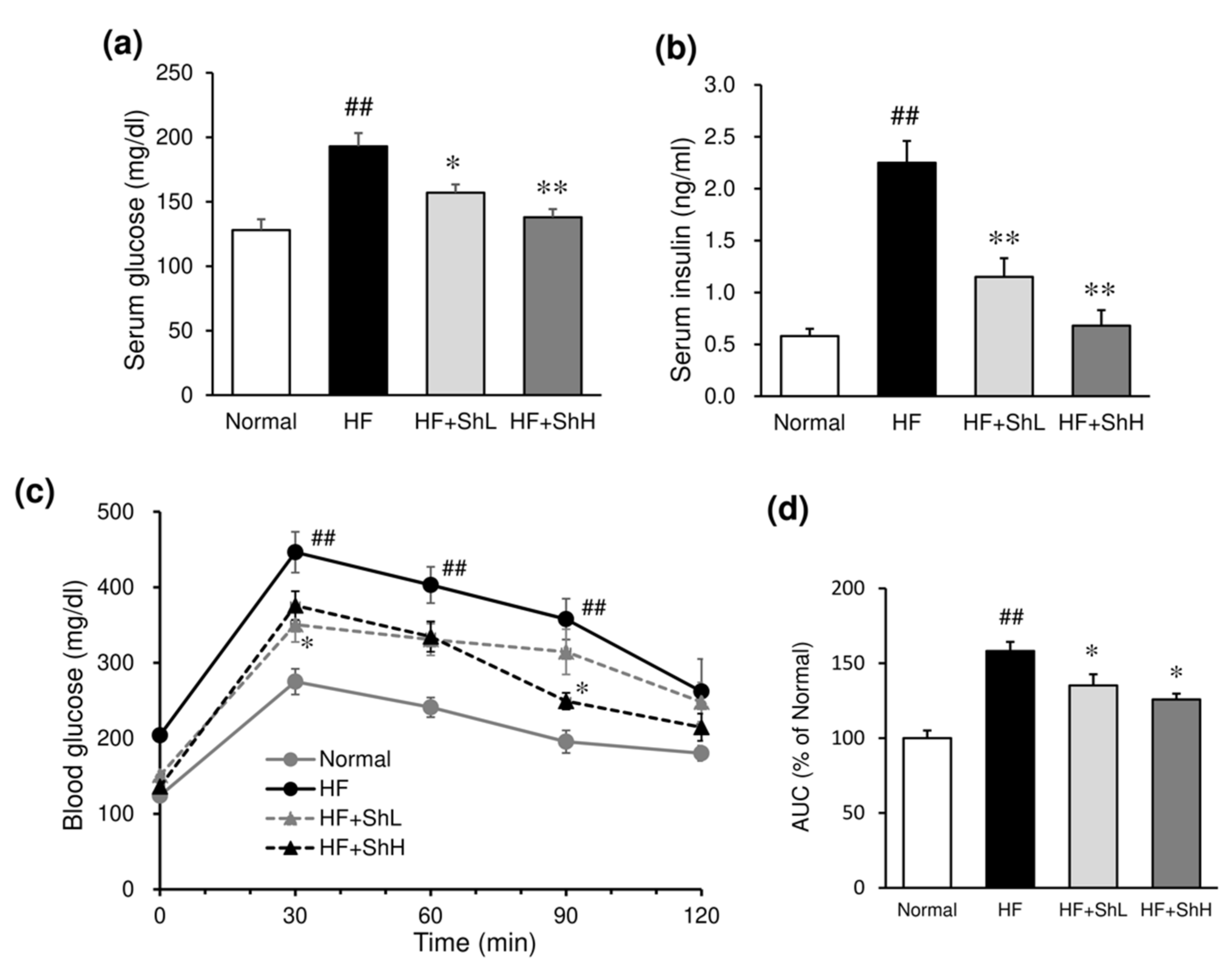
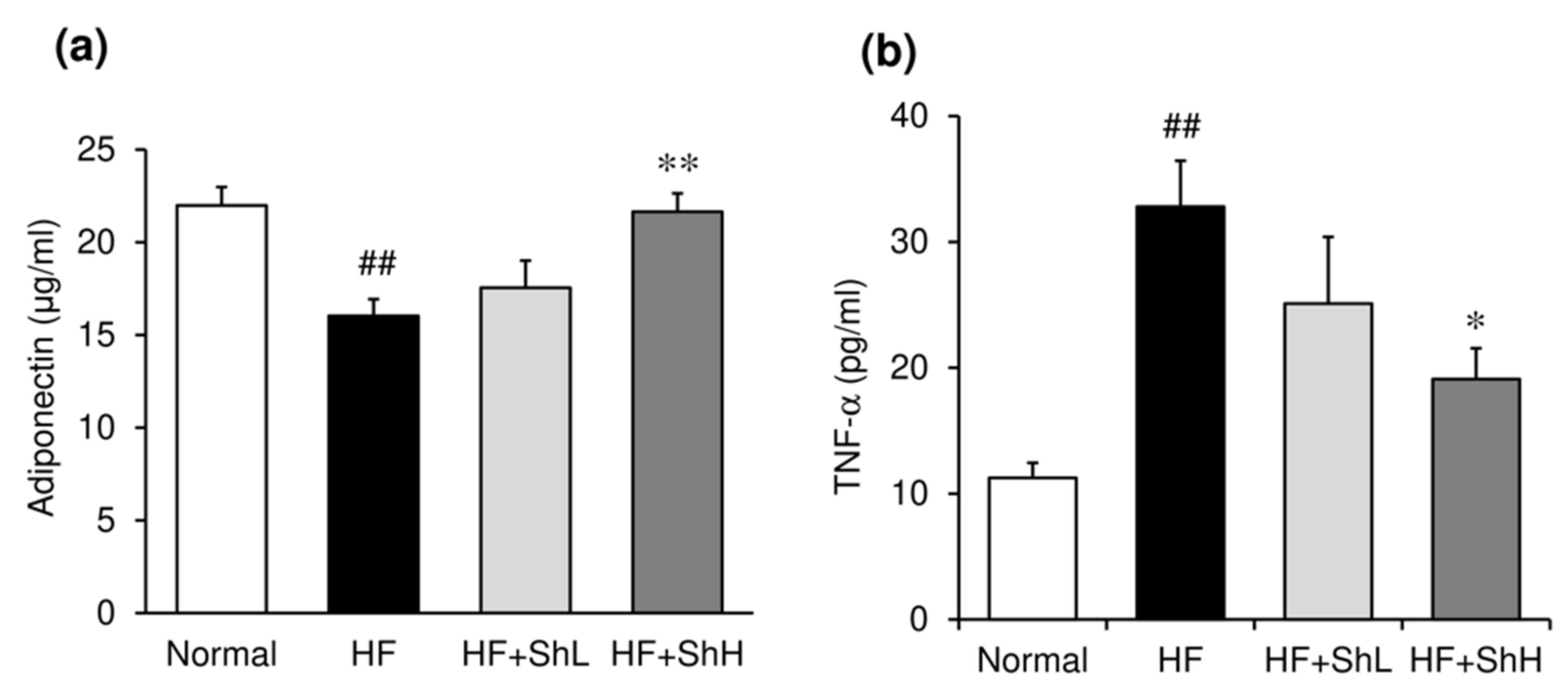
3.3. Effects of S. horneri on Serum Levels of Glucose, Insulin and Adipokines, and Insulin Resistance
3.4. Effects of S. horneri on Serum Lipid Levels
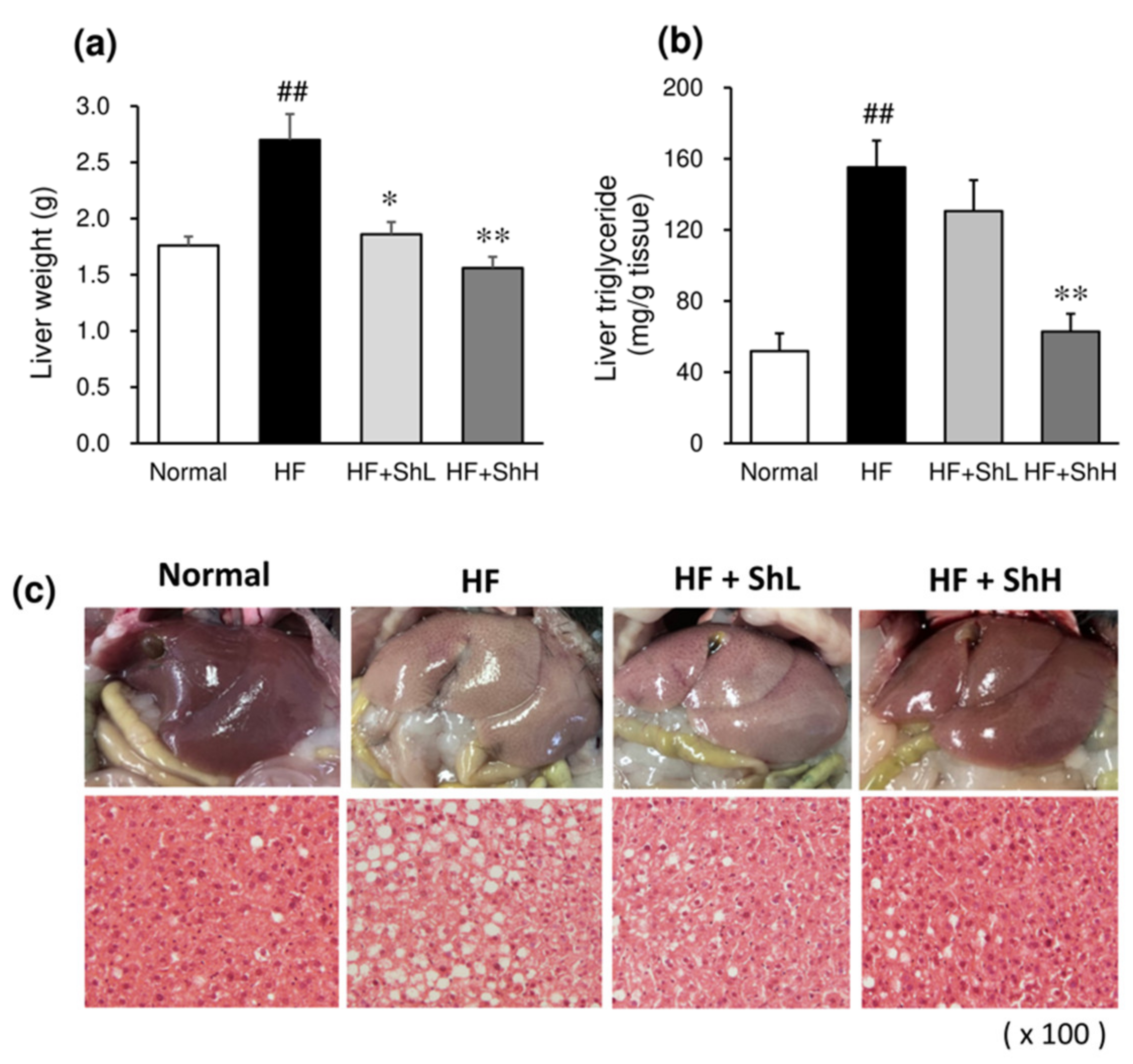
3.5. Effects of S. horneri on Hepatic Steatosis
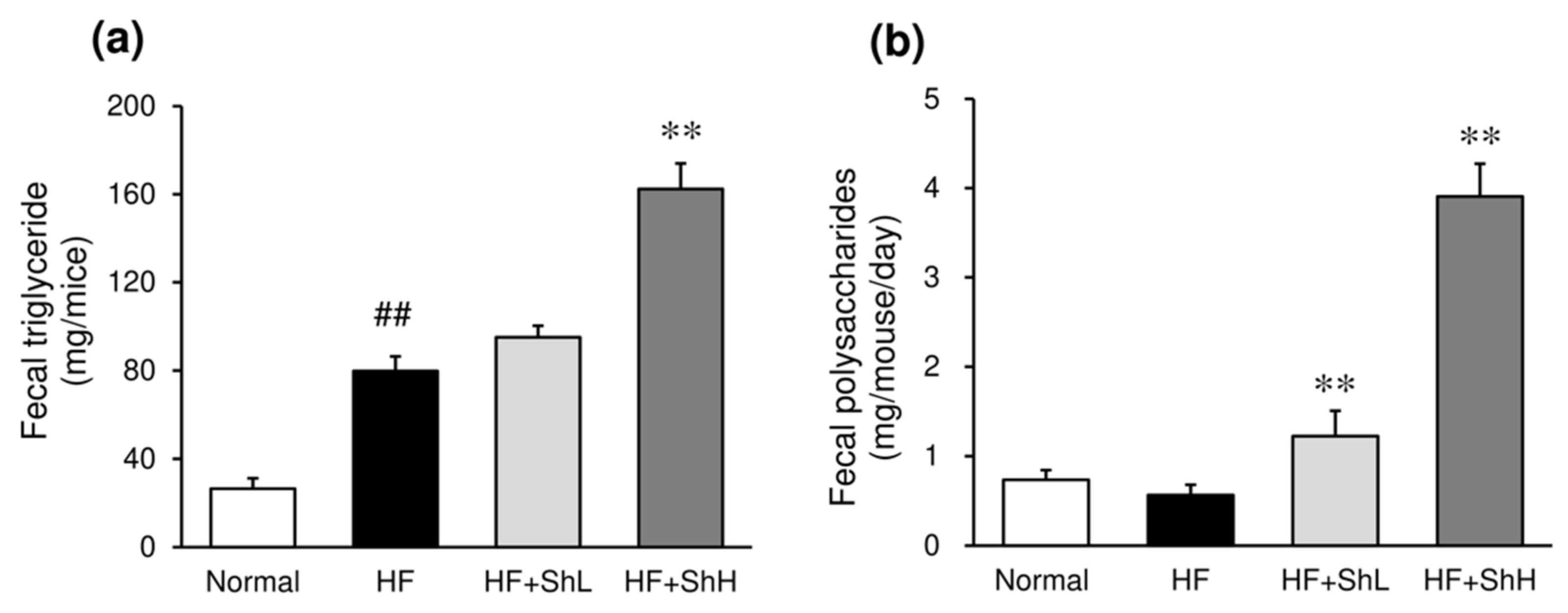
3.6. Effects of S. horneri on the Fecal Content of Triglycerides and Polysaccharides
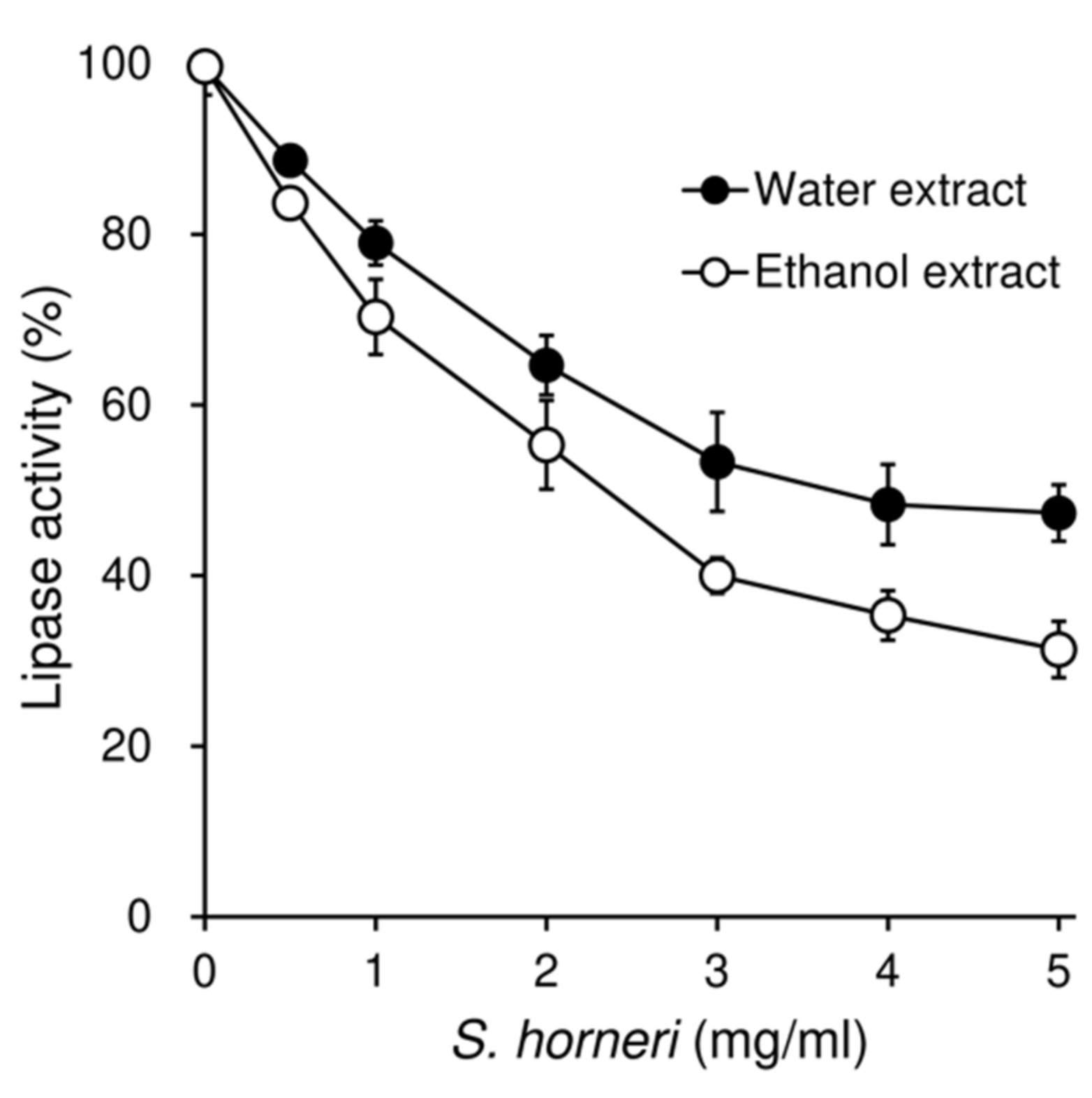
3.7. Effects of S. horneri on the Pancreatic Lipase Activity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Brown, E.S.; Allsopp, P.J.; Magee, P.J.; Gill, C.I.; Nitecki, S.; Strain, C.R.; McSorley, E.M. Seaweed and human health. Nutr. Rev. 2014, 72, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, S.M.; Pereira, O.R.; Seca, A.M.; Pinto, D.C.; Silva, A.M. Seaweeds as preventive agents for cardiovascular diseases from nutrients to functional foods. Mar. Drugs 2015, 13, 6838–6865. [Google Scholar]
- Cherry, P.; O’Hara, C.; Magee, P.J.; McSorley, E.M.; Allsopp, P.J. Risks and benefits of consuming edible seaweeds. Nutr. Rev. 2019, 77, 307–329. [Google Scholar] [CrossRef]
- Murai, U.; Yamagishi, K.; Sata, M.; Kokubo, Y.; Saito, I.; Yatsuya, H.; Ishihara, J.; Inoue, M.; Sawada, N.; Iso, H.; et al. JPHC Study Group. Seaweed intake and risk of cardiovascular disease: The Japan Public Health Center-based Prospective (JPHC) Study. Am. J. Clin. Nutr. 2019, 110, 1449–1455. [Google Scholar] [CrossRef]
- Kishida, R.; Yamagishi, K.; Muraki, I.; Sata, M.; Tamakoshi, A.; Iso, H. JACC Study Group. Frequency of seaweed intake and its association with cardiovascular disease mortality: The JACC Study. J. Atheroscler. Thromb. 2020, 27, 1340–1347. [Google Scholar] [CrossRef]
- Sho, H. History and characteristics of Okinawan longevity food. Asia Pac. J. Clin. Nutr. 2001, 10, 159–164. [Google Scholar] [CrossRef]
- Blüher, M. Obesity: Global epidemiology and pathogenesis. Nat. Rev. Endocrinol. 2019, 15, 288–298. [Google Scholar] [CrossRef]
- Van Gaal, L.; Mertens, I.L.; De Block, C.E. Mechanisms linking obesity with cardiovascular disease. Nature 2006, 444, 875–880. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, E.J.; LeRoith, D. Obesity and diabetes: The increased risk of cancer and cancer-related mortality. Physiol. Rev. 2015, 95, 727–948. [Google Scholar] [CrossRef]
- Zhang, W.L.; Zhu, L.; Jiang, J.G. Active ingredients from natural botanicals in the treatment of obesity. Obes. Rev. 2014, 15, 957–967. [Google Scholar] [CrossRef] [PubMed]
- Sun, N.N.; Wu., T.Y.; Chau, C.F. Natural dietary and herbal products in anti-obesity treatment. Molecules 2016, 21, E1351. [Google Scholar] [CrossRef]
- Montero, L.; Del. Pilar Sánchez-Camargo, A.; Ibáñez, E.; Gilbert-López, B. Phenolic compounds from edible algae Bioactivity and health benefits. Curr. Med. Chem. 2018, 25, 4808–4826. [Google Scholar]
- Rodríguez-Pérez, C.; Segura-Carretero, A.; Del. Mar. Contreras, M. Phenolic compounds as natural and multifunctional anti-obesity agents: A review. Crit. Rev. Food. Sci. Nutr. 2019, 59, 1212–1229. [Google Scholar]
- Komatsu, T.; Fukuda, M.; Mikami, A.; Mizuno, S.; Kantachumpoo, A.; Tanoue, H.; Kawamiya, M. Possible change in distribution of seaweed, Sargassum horneri, in northeast Asia under A2 scenario of global warming and consequent effect on some fish. Mar. Pollut. Bull. 2014, 85, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Liu, H.B. Research progress in chemical components and biological activity of Sargassum. Chin. J. Mar. Drugs. 2012, 31, 41–51. [Google Scholar]
- Silchenko, A.S.; Rasin, A.B.; Kusaykin, M.I.; Kalinovsky, A.I.; Miansong, Z.; Changheng, L.; Malyarenko, O.; Zueva, A.O.; Zvyagintseva, T.N.; Ermakova, S.P. Structure, enzymatic transformation, anticancer activity of fucoidan and sulphated fucooligosaccharides from Sargassum horneri. Carbohydr. Polym 2017, 175, 654–660. [Google Scholar] [CrossRef] [PubMed]
- Herath, K.H.I.N.M.; Cho, J.; Kim, A.; Kim, H.S.; Han, E.J.; Kim, H.J.; Kim, M.S.; Ahn, G.; Jeon, Y.J.; Jee, Y. Differential modulation of immune response and cytokine profiles of Sargassum horneri ethanol extract in murine spleen with or without Concanavalin A stimulation. Biomed. Pharmacother. 2019, 110, 930–942. [Google Scholar] [CrossRef] [PubMed]
- Kwak, J.Y. Fucoidan as a marine anticancer agent in preclinical development. Mar. Drugs 2014, 12, 851–870. [Google Scholar] [CrossRef] [PubMed]
- Luthuli, S.; Wu, S.; Cheng, Y.; Zheng, X.; Wu, M.; Tong, H. Therapeutic effects of fucoidan: A review on recent studies. Mar. Drugs 2019, 17, E487. [Google Scholar] [CrossRef] [PubMed]
- Martău, G.A.; Mihai, M.; Vodnar, D.C. The use of chitosan, alginate, and pectin in the biomedical and food sector-biocompatibility, bioadhesiveness, and biodegradability. Polymers 2019, 11, E1837. [Google Scholar] [CrossRef]
- Anderson, J.W.; Baird, P.; Davis, R.H.; Ferreri, S.; Knudtson, M.; Koraym, A.; Waters, V.; Williams, C.L. Health benefits of dietary fiber. Nutr. Rev. 2009, 67, 188–205. [Google Scholar] [CrossRef]
- Wan-Loy, C.; Siew-Moi, P. Marine algae as a potential source for anti-obesity agents. Mar. Drugs 2016, 14, E222. [Google Scholar] [CrossRef]
- Peters, H.P.; Koppert, R.J.; Boers, H.M.; Ström, A.; Melnikov, S.M.; Haddeman, E.; Schuring, E.A.; Mela, D.J.; Wiseman, S.A. Dose-dependent suppression of hunger by a specific alginate in a low-viscosity drink formulation. Obesity 2011, 19, 1171–1176. [Google Scholar] [CrossRef]
- Georg Jensen, M.; Kristensen, M.; Astrup, A. Effect of alginate supplementation on weight loss in obese subjects completing a 12-wk energy-restricted diet: A randomized controlled trial. Am. J. Clin. Nutr. 2012, 96, 5–13. [Google Scholar] [CrossRef]
- Peng, J.; Yuan, J.P.; Wu, C.F.; Wang, J.H. Fucoxanthin, a marine carotenoid present in brown seaweeds and diatoms: Metabolism and bioactivities relevant to human health. Mar. Drugs 2011, 9, 1806–1828. [Google Scholar] [CrossRef] [PubMed]
- Murakami, S.; Yamagishi, I.; Sato, M.; Tomisawa, K.; Nara, Y.; Yamori, Y. ACAT inhibitor HL-004 accelerates the regression of hypercholesterolemia in stroke-prone spontaneously hypertensive rats (SHRSP): Stimulation of bile acid production by HL-004. Atherosclerosis 1997, 133, 97–104. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Bendicho, S.; Trigueros, M.C.; Hernández, T.; Martin, O. Validation and comparison of analytical methods based on the release of p-nitrophenol to determine lipase activity in milk. J. Dairy Sci. 2001, 84, 1590–1596. [Google Scholar] [CrossRef]
- Kim, M.J.; Jeon, J.; Lee, J.S. Fucoidan prevents high-fat diet-induced obesity in animals by suppression of fat accumulation. Phytother. Res. 2014, 28, 137–143. [Google Scholar] [CrossRef]
- Park, M.K.; Jung, U.; Roh, C. Fucoidan from marine brown algae inhibits lipid accumulation. Mar. Drugs 2011, 9, 1359–1367. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Corona, D.M.; Martínez-Abundis, E.; González-Ortiz, M. Effect of fucoidan administration on insulin secretion and insulin resistance in overweight or obese adults. J. Med. Food 2014, 17, 830–831. [Google Scholar] [CrossRef]
- Wilcox, M.D.; Brownlee, I.A.; Richardson, J.C.; Dettmar, P.W.; Pearson, J.P. The modulation of pancreatic lipase activity by alginates. Food Chem. 2014, 146, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Georg Jensen, M.; Pedersen, C.; Kristensen, M.; Frost, G.; Frost, G.; Astrup, A. Review: Efficacy of alginate supplementation in relation to appetite regulation and metabolic risk factors: Evidence from animal and human studies. Obes. Rev. 2013, 14, 129–144. [Google Scholar] [CrossRef] [PubMed]
- Galic, S.; Oakhill, J.S.; Steinberg, G.R. Adipose tissue as an endocrine organ. Mol. Cell. Endocrinol. 2010, 316, 129–139. [Google Scholar] [CrossRef]
- Ouchi, N.; Parker, J.L.; Lugus, J.J.; Walsh, K. Adipokines in inflammation and metabolic disease. Nat. Rev. Immunol. 2011, 11, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Lihn, A.S.; Pedersen, S.B.; Richelsen, B. Adiponectin: Action, regulation and association to insulin sensitivity. Obes. Rev. 2005, 6, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Hirai, S.; Yamaguchi, Y.; Goto, T.; Takahashi, N.; Tani, F.; Mutoh, C.; Sakurai, T.; Murakami, S.; Yu, R.; et al. Taurine improves obesity-induced inflammatory responses and modulates the unbalanced phenotype of adipose tissue macrophages. Mol. Nutr. Food Res. 2013, 57, 2155–2165. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.E.; Jung, Y.C.; Jung, I.; Lee, H.W.; Youn, H.Y.; Lee, J.S. Anti-inflammatory effects of ethanolic extract from Sargassum horneri (Turner) C. Agardh on lipopolysaccharide-stimulated macrophage activation via NF-κB pathway regulation. Immunol. Invest. 2015, 44, 137–146. [Google Scholar] [CrossRef]
- Park, J.; Cha, J.D.; Choi, K.M.; Lee, K.Y.; Han, K.M.; Jang, Y.S. Fucoidan inhibits LPS-induced inflammation in vitro and during the acute response in vivo. Int. Immunopharmacol. 2017, 43, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Kawauchi, S.; Horibe, S.; Sasaki, N.; Tanahashi, T.; Mizuno, S.; Hamaguchi, T.; Rikitake, Y. Inhibitory effects of sodium alginate on hepatic steatosis in mice induced by a methionine- and choline-deficient diet. Mar. Drugs 2019, 17, 104. [Google Scholar] [CrossRef]
- Tan, C.P.; Hou, Y.H. First evidence for the anti-inflammatory activity of fucoxanthin in high-fat-diet-induced obesity in mice and the antioxidant functions in PC12 cells. Inflammation 2014, 37, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Tilg, H.; Moschen, A.R. Evolution of inflammation in nonalcoholic fatty liver disease: The multiple parallel hits hypothesis. Hepatology 2010, 52, 1836–1846. [Google Scholar] [CrossRef]
- de la Garza, A.L.; Milagro, F.I.; Boque, N.; Campión, J.; Martínez, J.A. Natural inhibitors of pancreatic lipase as new players in obesity treatment. Planta Med. 2011, 77, 773–785. [Google Scholar] [CrossRef] [PubMed]
- Chater, P.I.; Wilcox, M.; Cherry, P.; Herford, A.; Mustar, S.; Wheater, H.; Brownlee, I.; Seal, C.; Pearson, J. Inhibitory activity of extracts of Hebridean brown seaweeds on lipase activity. J. Appl. Phycol. 2016, 28, 1303–1313. [Google Scholar] [CrossRef] [PubMed]
- Kasahara, F.; Kato, T.; Idota, Y.; Takahashi, H.; Kakinuma, C.; Yano, K.; Arakawa, H.; Hara, K.; Miyajima, C.; Ogihara, T. Reduction effect of calcium alginate on blood triglyceride levels causing the inhibition of hepatic and total body accumulation of fat in rats. Biol. Pharm. Bull. 2019, 42, 365–372. [Google Scholar] [CrossRef]
- Sandberg, A.S.; Andersson, H.; Bosaeus, I.; Carlsson, N.G.; Hasselblad, K.; Härröd, M. Alginate, small bowel sterol excretion, and absorption of nutrients in ileostomy subjects. Am. J. Clin. Nutr. 1994, 60, 751–756. [Google Scholar] [CrossRef]
- Chater, P.I.; Wilcox, M.D.; Houghton, D.; Pearson, J.P. The role of seaweed bioactives in the control of digestion: Implications for obesity treatments. Food Funct. 2015, 6, 3420–3427. [Google Scholar] [CrossRef]
- Mackie, A.R.; Macierzanka, A.; Aarak, K.; Rigby, N.M.; Parker, R.; Channell, G.A.; Harding, S.E.; Bajka, B.H. Sodium alginate decreases the permeability of intestinal mucus. Food Hydrocoll. 2016, 52, 749–755. [Google Scholar] [CrossRef]
- Lowe, M.E. Pancreatic triglyceride lipase and colipase: Insights into dietary fat digestion. Gastroenterology 1994, 107, 1524–1536. [Google Scholar] [CrossRef]
- Heck, A.M.; Yanovski, J.A.; Calis, K.A. Orlistat, a new lipase inhibitor for the management of obesity. Pharmacotherapy 2000, 20, 270–279. [Google Scholar] [CrossRef] [PubMed]
- Terasaki, M.; Hirose, A.; Narayan, B.; Baba, Y.; Kawagoe, C.; Yasui, H.; Saga, N.; Hosokawa, M.; Miyashita, K. Evaluation of recoverable functional lipid components of several brown seaweeds (Phaeophyta) from Japan with special reference to fucoxanthin and fucosterol contents (1). J. Phycol. 2009, 45, 974–980. [Google Scholar] [CrossRef] [PubMed]
- Gammone, M.A.; D’Orazio, N. Anti-obesity activity of the marine carotenoid fucoxanthin. Mar. Drugs 2015, 13, 2196–2214. [Google Scholar] [CrossRef]
- Abidov, M.; Ramazanov, Z.; Seifulla, R.; Grachev, S. The effects of Xanthigen in the weight management of obese premenopausal women with non-alcoholic fatty liver disease and normal liver fat. Diabetes Obes. Metab. 2010, 12, 72–81. [Google Scholar] [CrossRef]
- Matsumoto, M.; Hosokawa, M.; Matsukawa, N.; Hagio, M.; Shinoki, A.; Nishimukai, M.; Miyashita, K.; Yajima, T.; Hara, H. Suppressive Effects of the marine carotenoids, fucoxanthin and fucoxanthinol on triglyceride absorption in lymph duct-cannulated rats. Eur J. Nutr. 2010, 49, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Hosokawa, M.; Sashima, T.; Funayama, K.; Miyashita, K. Fucoxanthin from edible seaweed, Undaria pinnatifida, shows antiobesity effect through UCP1 expression in white adipose tissues. Biochem. Biophys. Res. Commun. 2005, 332, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Woo, M.N.; Jeon, S.M.; Shin, Y.C.; Lee, M.K.; Kang, M.A.; Choi, M.S. Anti-obese property of fucoxanthin is partly mediated by altering lipid-regulating enzymes and uncoupling proteins of visceral adipose tissue in mice. Mol. Nutr. Food Res. 2009, 53, 1603–1611. [Google Scholar] [CrossRef] [PubMed]
| Ingredients (g/100 g) | Normal Diet | High-Fat (HF) | HF + ShL (2%) | HF + ShH (6%) |
|---|---|---|---|---|
| Cornstarch | 46.57 | |||
| α-Cornstarch | 15.5 | 16.0 | 15.0 | 13.0 |
| Maltodextrin | 6.0 | 6.0 | 6.0 | |
| Sucrose | 10.0 | 5.5 | 5.5 | 5.5 |
| Casein | 14.0 | 25.6 | 25.6 | 25.6 |
| Cellulose | 5.0 | 6.6 | 5.6 | 3.6 |
| Soybean oil | 4.0 | 2.0 | 2.0 | 2.0 |
| Lard | 33.0 | 33.0 | 33.0 | |
| Mineral mix | 3.5 | 3.5 | 3.5 | 3.5 |
| Vitamin mix | 1.0 | 1.0 | 1.0 | 1.0 |
| L-Cysteine | 0.18 | 0.36 | 0.36 | 0.36 |
| Cholin bitartrate | 0.25 | 0.25 | 0.25 | 0.25 |
| Calcium carbonate | 0.18 | 0.18 | 0.18 | |
| S. horneri powder | 2.0 | 6.0 |
| Normal | HF | HF + ShL | HF + ShH | |
|---|---|---|---|---|
| Total cholesterol (mg/dL) | 150 ± 9 | 217 ± 6 ## | 215 ± 7 | 189 ± 7 * |
| HDL cholesterol (mg/dL) | 86 ± 2 | 88 ± 2 | 85 ± 2 | 87 ± 2 |
| Non-HDL cholesterol (mg/dL) | 64 ± 7 | 128 ± 7 ## | 124 ± 6 | 99 ± 5 ** |
| Triglyceride (mg/dL) | 69 ± 8 | 48 ± 6 | 33 ± 3 | 45 ± 4 |
| Normal | HF | HF + ShL | HF + ShH | |
|---|---|---|---|---|
| ALT (IU/L) | 32.7 ± 6.7 | 126.7 ± 23.5 ## | 102.4 ± 17.2 | 40.1 ± 4.9 ** |
| AST (IU/L) | 107.4 ± 10.6 | 155.0 ± 17.4 ## | 120.3 ± 10.6 | 103.0 ± 7.8 * |
| ALP (IU/L) | 223.7 ± 12.3 | 287.3 ± 27.1 ## | 255.8 ± 18.6 | 195.6 ± 8.8 ** |
| LAP (IU/L) | 33.8 ± 1.4 | 43.6 ± 3.7 # | 40.6 ± 2.7 | 32.1 ± 0.93 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Murakami, S.; Hirazawa, C.; Ohya, T.; Yoshikawa, R.; Mizutani, T.; Ma, N.; Moriyama, M.; Ito, T.; Matsuzaki, C. The Edible Brown Seaweed Sargassum horneri (Turner) C. Agardh Ameliorates High-Fat Diet-Induced Obesity, Diabetes, and Hepatic Steatosis in Mice. Nutrients 2021, 13, 551. https://doi.org/10.3390/nu13020551
Murakami S, Hirazawa C, Ohya T, Yoshikawa R, Mizutani T, Ma N, Moriyama M, Ito T, Matsuzaki C. The Edible Brown Seaweed Sargassum horneri (Turner) C. Agardh Ameliorates High-Fat Diet-Induced Obesity, Diabetes, and Hepatic Steatosis in Mice. Nutrients. 2021; 13(2):551. https://doi.org/10.3390/nu13020551
Chicago/Turabian StyleMurakami, Shigeru, Chihiro Hirazawa, Takuma Ohya, Rina Yoshikawa, Toshiki Mizutani, Ning Ma, Mitsuru Moriyama, Takashi Ito, and Chiaki Matsuzaki. 2021. "The Edible Brown Seaweed Sargassum horneri (Turner) C. Agardh Ameliorates High-Fat Diet-Induced Obesity, Diabetes, and Hepatic Steatosis in Mice" Nutrients 13, no. 2: 551. https://doi.org/10.3390/nu13020551
APA StyleMurakami, S., Hirazawa, C., Ohya, T., Yoshikawa, R., Mizutani, T., Ma, N., Moriyama, M., Ito, T., & Matsuzaki, C. (2021). The Edible Brown Seaweed Sargassum horneri (Turner) C. Agardh Ameliorates High-Fat Diet-Induced Obesity, Diabetes, and Hepatic Steatosis in Mice. Nutrients, 13(2), 551. https://doi.org/10.3390/nu13020551








