The Effect of Omega-3 Fatty Acid Supplementation on Serum Adipocytokines, Lipid Profile and Biochemical Markers of Inflammation in Recreational Runners
Abstract
1. Introduction
2. Material and Methods
2.1. Subjects
2.2. Supplementation Procedure
2.3. Biochemical Analyses
2.4. Statistical Analysis
3. Results
3.1. Somatic and Physiological Characteristics of the Subjects Prior to the Intervention Protocols
3.2. Impact of ω-3 PUFA and Vitamin E and D on the Omega-3 Index
3.3. Effect of ω-3 PUFA and Vitamin E and D on Cytokines
3.4. Effect of ω-3 PUFA and Vitamin E and D on Antioxidant Status and Lipid Profile
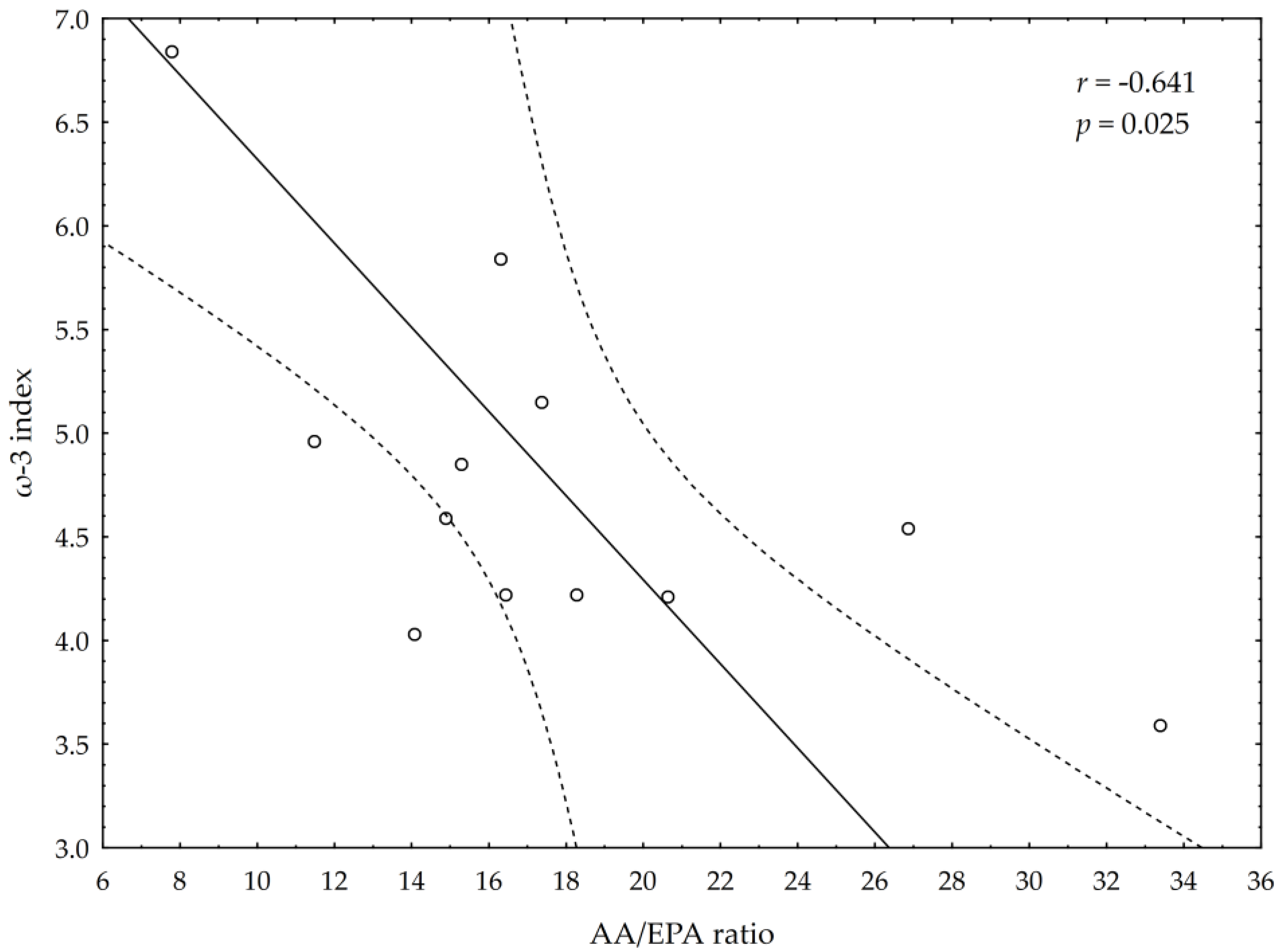
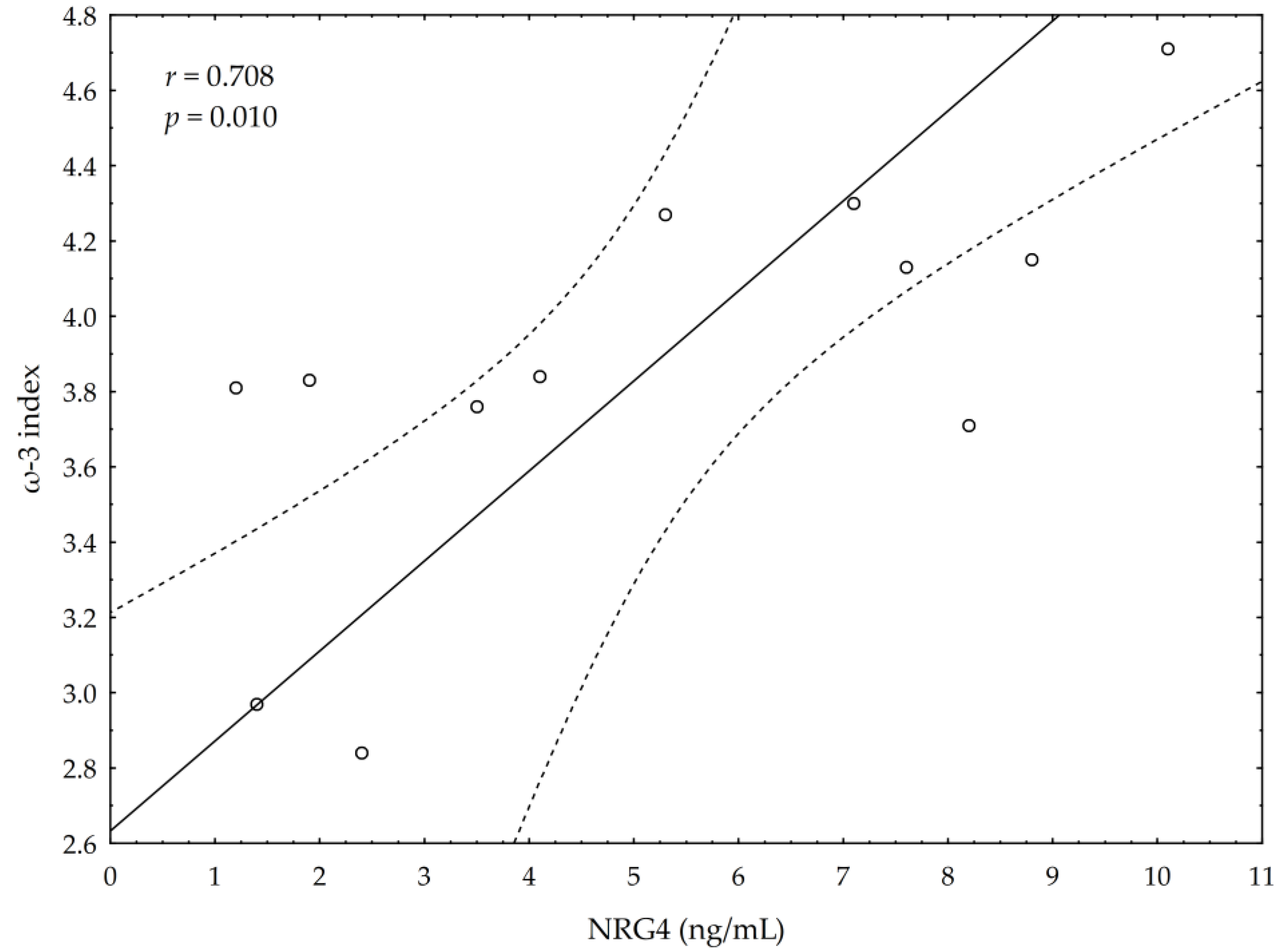
3.5. Correlations between Omega-3 Index, AA/EPA Ratio and Serum Variables of Adipocytes and Inflammation Markers
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Von Schacky, C.; Harris, W.S. Cardiovascular benefits of omega-3 fatty acids. Cardiovasc. Res. 2007, 73, 310–315. [Google Scholar] [CrossRef]
- Gutiérrez, S.; Svahn, S.L.; Johansson, M.E. Effects of Omega-3 fatty acids on immune cells. Int. J. Mol. Sci. 2019, 20, 5028. [Google Scholar] [CrossRef]
- Innes, J.K.; Calder, P.C. Marine Omega-3 (N-3) Fatty Acids for Cardiovascular Health: An Update for 2020. Int. J. Mol. Sci. 2020, 21, 1362. [Google Scholar] [CrossRef]
- Couet, C.; Delarue, J.; Rotz, P.; Antoine, J.M.; Lamisse, F. Effect of dietary fish oil on body fat mass and basal fat oxidation in healthy adults. Int. J. Obes. Relat. Metab. Disord. 1997, 21, 637–643. [Google Scholar] [CrossRef]
- Mori, T.A. Effect of fish oil-derived omega-3 fatty acids on lipid oxidation. Redox. Rep. 2004, 9, 193–197. [Google Scholar] [CrossRef]
- Mori, T.A.; Beilin, L.J. Omega-3 fatty acids and inflammation. Curr. Atheroscler. Rep. 2004, 6, 461–467. [Google Scholar] [CrossRef]
- Siscovick, D.S.; Raghunathan, T.E.; King, I. Dietary intake and cell membrane levels of long-chain n-3 polyunsaturated fatty acids and the risk of primary cardiac arrest. JAMA 1995, 274, 1363–1367. [Google Scholar] [CrossRef] [PubMed]
- Lemaitre, R.N.; King, I.B.; Mozaffarian, D.; Kuller, L.H.; Tracy, R.P.; Siscovick, D.S. N-3 polyunsaturated fatty acids, fatal ischemic heart disease, and nonfatal myocardial infarction in older adults: The Cardiovascular Health Study. Am. J. Clin. Nutr. 2003, 77, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; von Schacky, C. The Omega-3 Index: A new risk factor for death from coronary heart disease? Prev. Med. 2004, 39, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Gray, B.; Steyen, F.; Davies, P.S.W.; Vitetta, L. Omega-3 fatty acids: A review of the effects on adiponectin and leptin and potential implications for obesity management. Eur. J. Clin. Nutr. 2013, 67, 1234–1242. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. An increase in the omega-6/omega-3 fatty acid ratio increases the risk for obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef] [PubMed]
- Stojanović, S.; Fiedler, J.; Bauersachs, J.; Thum, T.; Sedding, D.G. Senescence-induced inflammation: An important player and key therapeutic target in atherosclerosis. Eur. Heart J. 2020, 41, 2983–2996. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. N-3 polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am. J. Clin. Nutr. 2006, 83, 1505–1519. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Omega-3 Fatty Acids and Inflammatory Processes. Nutrients 2010, 2, 355–374. [Google Scholar] [CrossRef]
- Calder, P.C. Metabolic benefits of marine n-3 fatty acids demonstrated in nonhuman primates. J. Nutr. 2014, 144, 1–2. [Google Scholar] [CrossRef]
- Cholewski, M.; Tomczykowa, M.; Tomczyk, M. A comprehensive review of chemistry, sources and bioavailability of omega-3 fatty acids. Nutrients 2018, 10, 1662. [Google Scholar] [CrossRef]
- Cao, J.; Schwichtenberg, K.A.; Hanson, N.Q.; Tsai, M.Y. Incorporation and clearance of Omega-3 fatty acids in erythrocyte membranes and plasma phospholipids. Clin. Chem. 2006, 52, 2265–2272. [Google Scholar] [CrossRef]
- Abbott, S.K.; Else, P.L.; Atkins, T.A.; Hulbert, A.J. Fatty acid composition of membrane bilayers: Importance of diet polyunsaturated fat balance. Biochim. Biophys. Acta 2012, 1818, 1309–1317. [Google Scholar] [CrossRef]
- Shiroma, E.J.; Lee, I.M. Physical activity and cardiovascular health: Lessons learned from epidemiological studies across age, gender, and race/ethnicity. Circulation 2010, 122, 743–752. [Google Scholar] [CrossRef]
- Lee, D.C.; Brellenthin, A.G.; Thompson, P.D.; Lee, I.M.; Lavie, C.J. Running as a key lifestyle medicine for longevity. Prog. Cardiovasc. Dis. 2017, 60, 45–55. [Google Scholar] [CrossRef]
- Scott, J.M.; Esch, B.T.; Shave, R.; Warburton, D.E.; Gaze, D.; George, K. Cardiovascular consequences of completing a 160-km ultramarathon. Med. Sci. Sports Exerc. 2009, 41, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Sharlene, M.; Thompson, P.D. Cardiac Risks Associated With Marathon Running. Sports Health 2010, 2, 301–306. [Google Scholar]
- Merghani, A.; Malhotra, A.; Sharma, S. The U-shaped relationship between exercise and cardiac morbidity. Trends Cardiovasc. Med. 2016, 26, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Dawson, E.; George, K.; Shave, R.; Whyte, G.; Ball, D. Does the human heart fatigue subsequent to prolonged exercise? Sports Med. 2003, 33, 365–380. [Google Scholar] [CrossRef]
- Kłapcińska, B.; Waśkiewicz, Z.; Chrapusta, S.J.; Sadowska-Krępa, E.; Czuba, M.; Langfort, J. Metabolic responses to a 48-h ultra-marathon run in middle-aged male amateur runners. Eur. J. Appl. Physiol. 2013, 113, 2781–2793. [Google Scholar] [CrossRef]
- Albert, C.M.; Campos, H.; Stampfer, M.J. Blood levels of long-chain n-3 fatty acids and the risk of sudden death. N. Engl. J. Med. 2002, 346, 1113–1118. [Google Scholar] [CrossRef]
- McAnulty, S.R.; Nieman, D.C.; Fox-Rabinovich, M.; Duran, V.; McAnulty, L.S.; Henson, D.A.; Jin, F.; Landram, M.J. Effect of n-3 fatty acids and antioxidants on oxidative stress after exercise. Med. Sci. Sports Exerc. 2010, 42, 1704–1711. [Google Scholar] [CrossRef]
- Wilson, P.B.; Madrigal, L.A. Associations between whole blood and dietary omega-3 polyunsaturated fatty acid levels in collegiate athletes. Int. J. Sport Nutr. Exerc. Metab. 2016, 26, 497–505. [Google Scholar] [CrossRef]
- Davinelli, S.; Corbi, G.; Righetti, S.; Casiraghi, E.; Chiappero, F.; Martegani, S.; Pina, R.; De Vivo, I.; Simopoulos, A.P.; Scapagnini, G. Relationship between distance run per week, omega-3 index, and arachidonic acid (AA)/ Eicosapentaenoic acid (EPA) ratio: An observational retrospective study in non-elite runners. Front. Physiol. 2019, 10, 487. [Google Scholar] [CrossRef]
- Avramovic, N.; Dragutinovic, V.; Krstic, D.; Colovic, M.; Trbovic, A.; De Luka, S.; Milovanovic, I.; Popovic, T. The effects of omega 3 fatty acid supplementation on brain tissue oxidative status in aged wistar rats. Hippokratia 2012, 16, 241–245. [Google Scholar]
- Ramprasath, V.R.; Eyal, I.; Zchut, S.; Jones, P.J. Enhanced increase of omega-3 index in healthy individuals with response to 4-week n-3 fatty acid supplementation from krill oil versus fish oil. Lipids Health Dis. 2013, 12, 178. [Google Scholar] [CrossRef] [PubMed]
- Mickleborough, T.D. Omega-3 polyunsaturated Fatty acids in physical performance optimization. Int. J. Sport Nutr. Exerc. Metab. 2013, 23, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Chacińska, M.; Zabielski, P.; Książek, M.; Szałaj, P.; Jarząbek, K.; Kojta, I.; Chabowski, A.; Błachnio-Zabielska, A.U. The impact of OMEGA-3 fatty acids supplementation on insulin resistance and content of adipocytokines and biollogically active lipids in adipose tissue of high-fat diet fed rats. Nutrients 2019, 11, 835. [Google Scholar] [CrossRef] [PubMed]
- Chandran, M.; Philips, S.A.; Ciaraldi, T.; Henry, R.R. Adiponectin: More than just another fat cell hormone? Diabetes Care 2003, 26, 2442–2450. [Google Scholar] [CrossRef] [PubMed]
- Diez, J.J.; Iglesias, P. The role of the novel adipocyte-derived hormone adiponectin in human disease. Eur. J. Endocrinol. 2003, 148, 293–300. [Google Scholar] [CrossRef]
- Fasshauer, M.; Blüher, M. Adipokines in health and disease. Trends Pharmacol. Sci. 2015, 36, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Ouchi, N.; Parker, J.L.; Lugus, J.J.; Walsh, K. Adipokines in inflammation and metabolic disease. Nat. Rev. Immunol. 2011, 11, 85–97. [Google Scholar] [CrossRef]
- Fève, B.; Bastard, C.; Fellahi, S.; Bastard, J.P.; Capeau, J. New adipokines. Ann. Endocrinol. 2016, 77, 49–56. [Google Scholar] [CrossRef]
- Pérez-Matute, P.; Pérez-Echarri, N.; Marti, A.; Martinez, J.A.; Fernández-Otero, M.P.; Stanhope, K.L.; Havel, P.J.; Moreno-Aliaga, M.J. Eicosapentaenoic fatty acid increases leptin secretion from primary cultured rat adipocytes; role of glucose metabolism. Am. J. Regul. Integr. Comp. Physiol. 2005, 288, 1682–1688. [Google Scholar] [CrossRef]
- Staiger, H.; Tschritter, O.; Machann, J.; Thamer, C.; Fritsche, A.; Maerker, E.; Schick, F.; Häring, H.U.; Stumvoll, M. Relationship of serum adiponectin and leptin concentrations with fat distribution in humans. Obes. Res. 2003, 11, 368–372. [Google Scholar] [CrossRef]
- Ren, J. Leptin and hyperleptinemia—From friend to foe for cardiovascular function. J. Endocrinol. 2004, 181, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Loffreda, S.; Yang, S.Q.; Lin, H.Z.; Karp, C.L.; Brengman, M.L.; Wang, D.J.; Klein, A.S.; Bulkley, G.B.; Bao, C.; Noble, P.W.; et al. Leptin regulates proinflammatory immune responses. FASEB J. 1998, 12, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.-X.; Zhao, X.-Y.; Meng, Z.-X.; Kern, M.; Dietrich, A.; Chen, Z.; Cozacov, Z.; Zhou, D.; Okunade, A.L.; Su, X.; et al. The brown fat-enriched secreted factor Nrg4 preserves metabolic homeostasis through attenuation of hepatic lipogenesis. Nat. Med. 2014, 20, 1436–1443. [Google Scholar] [CrossRef] [PubMed]
- Bartelet, A.; Heeren, J. Adipose tissue browning and metabolic health. Nat. Rev. Endocrinol. 2014, 10, 24–36. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.E.; Kim, J.M.; Choung, S.; Joung, K.H.; Lee, J.-H.; Kim, H.J.; Ku, B.J. Comparison of serum Neuregulin 4 (Nrg4) levels in adults with newly diagnosed type 2 diabetes mellitus and controls without diabetes. Diabetes Res. Clin. Pr. 2016, 117, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Blüher, M. Neuregulin 4: A “hotline” between brown fat and liver. Obesity 2019, 27, 1555–1557. [Google Scholar] [CrossRef]
- Gumá, A.; Diaz-Sáez, F.; Camps, M.; Zorzano, A. Neuregulin, an effector on mitochondrial metabolism that preserves insulin sensitivity. Front. Physiol. 2020, 11, 696. [Google Scholar] [CrossRef]
- Cantó, C.; Pich, S.; Paz, J.C.; Sanches, R.; Martínez, V.; Orpinell, M.; Palacín, M.; Zorzano, A.; Gumà, A. Neuregulin increases mitochondrial oxidative capacity and insulin sensitivity in skeletall muscle cells. Diabetes Metab. Res. Rev. 2007, 56, 2185–2193. [Google Scholar]
- Jiang, J.; Lin, M.; Xu, Y.; Shao, J.; Li, X.; Zhang, H.; Yang, S. Circulating neuregulin 4 levels are inversely associated with subclinical cardiovascular disease in obese adults. Sci. Rep. 2016, 6, 36710. [Google Scholar] [CrossRef]
- Pfeifer, A. NRG4: An endocrine link between brown adipose tissue and liver. Cell Metab. 2015, 21, 13–14. [Google Scholar] [CrossRef]
- Comas, F.; Martinez, C.; Sabater, M.; Ortega, F.; Latorre, J.; Diaz-Sáez, F.; Aragonés, J.; Camps, M.; Gumá, A.; Ricart, W.; et al. Neurogulin 4 is a novel marker of beige adipocyte precursor cell in human adipose tissue. Front. Physiol. 2019, 10, 39. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Wang, G.X.; Ma, S.L.; Jung, D.Y.; Ha, H.; Altamimi, T.; Zhao, X.Y.; Guo, L.; Zhang, P.; Hu, C.R.; et al. Nrg4 promotes fuel oxidation and a healthy adipokine profile to ameliorate diet-induced metabolic disorders. Mol. Metab. 2017, 6, 863–872. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S. The Omega-3 index and cardiovascular disease: A case for omega-3 index as a new risk factor. A new risk factor for death from coronary heart disease? Pharmacol. Res. 2007, 55, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Gabrielli, L.; Sitges, M.; Chiong, M.; Jalil, J.; Ocaranza, M.; Llevaneras, S.; Herrera, S.; Fernandez, R.; Saavedra, R.; Yañez, F.; et al. Potential adverse cardiac remodeling in highly trained athletes: Still unknown clinical significance. Eur. J. Sport Sci. 2018, 18, 1288–1297. [Google Scholar] [CrossRef]
- Flock, M.R.; Skulas-Ray, A.C.; Harris, W.S.; Etherton, T.D.; Fleming, J.A.; Kris-Etherton, P.M. Determinants of erythrocyte omega-3 fatty acid content in response to fish oil supplementation: A dose-response randomized controlled trial. J. Am. Heart Assoc. 2013, 2, e000513. [Google Scholar] [CrossRef]
- Santos, V.C.; Levada-Pires, A.C.; Pithon-Curi, S.R.; Curi, R.; Cury-Boaventura, M.F. Effects of DHA-rich fish oil supplementation on lymphocyte function before and after a marathon race. Int. J. Sport Nutr. Exerc. Metab. 2013, 23, 161–169. [Google Scholar] [CrossRef]
- Von Schacky, C.; Kemper, M.; Haslbauer, R.; Halle, M. Low Omega-3 Index in 106 German elite winter endurance athletes: A pilot study. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 559–564. [Google Scholar] [CrossRef]
- Beutler, E.; Duron, O.; Kelly, B.M. Improved method for the determination of blood glutathione. J. Lab. Clin. Med. 1963, 61, 882–888. [Google Scholar]
- Sadowska-Krępa, E.; Kłapcińska, B.; Pokora, I.; Domaszewski, P.; Kempa, K.; Podgórski, T. Effects of six-week Ginkgo biloba supplementation on aerobic performance, blood pro/antioxidant balance, and serum brain-derived neurotrophic factor in physically active men. Nutrients 2017, 9, 803. [Google Scholar] [CrossRef]
- Buege, J.A.; Aust, S.D. Microsomal lipid peroxidation. Methods Enzymol. 1978, 52, 302–310. [Google Scholar] [CrossRef]
- Institute of Medicine (US) Panel on Dietary Antioxidants and Related Compounds. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids; National Academies Press (US): Washington, DC, USA, 2000. [Google Scholar]
- Drobnic, F.; Rueda, F.; Pons, V.; Banquells, M.; Cordobilla, B.; Domingo, J.C. Erythrocyte Omega-3 Fatty Acid Content in Elite Athletes in Response to Omega-3 Supplementation: A Dose-Response Pilot Study. J. Lipids 2017, 2017, 1472719. [Google Scholar] [CrossRef] [PubMed]
- Żebrowska, A.; Mizia-Stec, K.; Mizia, M.; Gąsior, Z.; Poprzęcki, S. Omega-3 fatty acids supplementation improves endothelial function and maximal oxygen uptake in endurance-trained athletes. Eur. J. Sport Sci. 2015, 15, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Burlingame, B.; Nishida, C.; Uauy, R.; Weisell, R. Fats and Fatty Acids in Human Nutrition. Ann. Nutr. Metab. 2009, 55, 5–7. [Google Scholar] [CrossRef] [PubMed]
- Psota, T.L.; Gebauer, S.K.; Kris-Etherton, P. Dietary Omega-3 Fatty Acid Intake and Cardiovascular Risk. Am. J. Cardiol. 2006, 98, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Villegas García, J.A.; Martínez Rocamora, M.T.; López Román, F.J.; Martínez Gonzálvez, A.B.; Luque Rubia, A.J. Ácidos grasos omega 3 en las lesiones deportivas. ¿Una posible ayuda terapéutica? Arch. Med. Deportea 2004, 104, 529–532. [Google Scholar]
- Bloomer, R.J.; Larson, D.E.; Fisher-Wellman, K.H.; Galpin, A.J.; Schilling, B.K. Lipids in health and disease. BioMed Cent. 2009, 8, 36. [Google Scholar] [CrossRef]
- Niki, E. Role of vitamin E as a lipid-soluble peroxyl radical scavenger: In vitro and in vivo evidence. Free Radic. Biol. Med. 2014, 66, 3–12. [Google Scholar] [CrossRef]
- Valk, E.E.; Hornstra, G. Relationship between vitamin E requirement and polyunsaturated fatty acid intake in man: A review. Int. J. Vitam. Nutr. Res. 2000, 70, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Raederstorff, D.; Wyss, A.; Calder, P.C.; Weber, P.; Eggersdorfer, M. Vitamin E function and requirements in relation to PUFA. Br. J. Nutr. 2015, 114, 1113–1122. [Google Scholar] [CrossRef]
- Ji, L.L.; Gomez-Cabrera, M.C.; Vina, J. Exercise and hormesis: Activation of cellular antioxidant signaling pathway. Ann. N. Y. Acad. Sci. 2006, 1067, 425–435. [Google Scholar] [CrossRef]
- Gomez-Cabrera, M.C.; Domenech, E.; Vina, J. Moderate exercise is an antioxidant: Upregulation of antioxidant genes by training. Free Radic. Biol. Med. 2008, 44, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Fisher-Wellman, K.; Bloomer, R.J. Acute exercise and oxidative stress: A 30 year history. Dyn. Med. 2009, 8, 1. [Google Scholar] [CrossRef] [PubMed]
- Tutino, V.; De Nunzio, V.; Caruso, M.G.; Veronese, N.; Lorusso, D.; Di Masi, M.; Benedetto, M.L.; Notarnicola, M. Elevated AA/EPA Ratio Represents an Inflammatory Biomarker in Tumor Tissue of Metastatic Colorectal Cancer Patients. Int. J. Mol. Sci. 2019, 20, 2050. [Google Scholar] [CrossRef] [PubMed]
- Whelan, J. Antagonistic effects of dietary arachidonic acid and n-3 polyunsaturated fatty acids. J. Nutr. 1996, 126, 1086S–1091S. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, A.M.; Montorfano, G.; Negroni, M.; Adorni, L.; Berselli, P.; Corsetto, P.; Wahle, K.W.J.; Berra, B. A rapid method for determining arachidonic:eicosapentaenoic acid ratios in whole blood lipids: Correlation with erythrocyte membrane ratios and validation in a large Italian population of various ages and pathologies. Lipids Health Dis. 2010, 9, 7. [Google Scholar] [CrossRef] [PubMed]
- Becic, T.; Studenik, C. Effects of omega-3 supplementation on adipocytokines in prediabetes and type 2 diabetes mellitus: Systemic review and meta-analysis of randomized controlled trials. Diabetes Metab. J. 2018, 42, 101–116. [Google Scholar] [CrossRef]
- Kojta, I.; Chacińska, M.; Błachnio-Zabieska, A. Obesity, bioactive lipids, and adipose tissue inflammation in insulin resistance. Nutrients 2020, 12, 1305. [Google Scholar] [CrossRef]
- Yamauchi, T.; Kamon, J.; Waki, H.; Terauchi, Y.; Kubota, N.; Hara, K.; Mori, Y.; Ide, T.; Murakami, K.; Tsuboyama-Kasaoka, N.; et al. The fat-derived hormone adiponectin reverses insulin resistance associated with lipoatrophy and obesity. Nat. Med. 2001, 7, 941–946. [Google Scholar] [CrossRef]
- Kany, S.; Vollrath, J.T.; Relja, B. Cytokines in Inflammatory Disease. Int. J. Mol. Sci. 2019, 20, 6008. [Google Scholar] [CrossRef]
- Zhang, Y.; Leung, D.Y.M.; Richers, B.N.; Liu, Y.; Remigio, L.K.; Riches, D.; Goleva, E. Vitamin D Inhibits Monocyte/Macrophage Proinflammatory Cytokine Production by Targeting MAPK Phosphatase-1. J. Immun. 2012, 188, 2127–2135. [Google Scholar] [CrossRef]
- Devaraj, S.; Leonard, S.; Traber, M.G.; Jialal, I. Gamma-tocopherol supplementation alone and in combination with alpha-tocopherol alters biomarkers of oxidative stress and inflammation in subjects with metabolic syndrome. Free Radic. Biol. Med. 2008, 44, 1203–1208. [Google Scholar] [CrossRef] [PubMed]
- Hariri, M.; Ghiasvand, R.; Shiranian, A.; Askari, G.; Iraj, B.; Salehi-Abargouei, A. Does omega-3 fatty acids supplementation affect circulating leptin levels? A systematic review and meta-analysis on randomized controlled clinical trials. Clin. Endocrinol. 2015, 82, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S. The omega-3 index as a risk factor for coronary heart disease. Am. J. Clin. Nutr. 2008, 87, 1997–2002. [Google Scholar] [CrossRef] [PubMed]
- Arca, M.; Montali, A.; Valiante, S.; Campagna, F.; Pigna, G.; Paoletti, V.; Antonini, R.; Barillà, F.; Tanzilli, G.; Vestri, A.; et al. Usefulness of atherogenic dyslipidemia for predicting cardiovascular risk in patients with angiographically defined coronary artery disease. Am. J. Cardiol. 2007, 100, 1511–1516. [Google Scholar] [CrossRef] [PubMed]
- Quesada-López, T.; Cereijo, R.; Turatsinze, J.V.; Planavila, A.; Cairó, M.; Gavaldà-Navarro, A.; Peyrou, M.; Moure, R.; Iglesias, R.; Giralt, M.; et al. The lipid sensor GPR120 promotes brown fat activation and FGF21 release from adipocytes. Nat. Commun. 2016, 7, 13479. [Google Scholar] [CrossRef]
- Villarroya, J.; Cereijo, R.; Giralt, M.; Villarroya, F. Secretory proteome of brown adipocytes in response to cAMP-mediated thermogenic activation. Front. Physiol. 2019, 10, 67. [Google Scholar] [CrossRef]
- Zhang, L.; Fu, Y.; Zhou, N.; Cheng, X.; Chen, C. Circulating neuregulin 4 concentrations in patients with newly diagnosed type 2 diabetes: A cross-sectional study. Endocrine 2017, 57, 535–538. [Google Scholar] [CrossRef]
- Yates, C.M.; Calder, P.C.; Ed Rainger, G. Pharmacology and therapeutics of omega-3 polyunaaturated fatty acids in chronic inflammatory disease. Pharmacol. Ther. 2014, 141, 272–282. [Google Scholar] [CrossRef]


| Variables | (ω-3 PUFA n = 12) | Placebo (n = 12) |
|---|---|---|
| Age (years) | 33.3 ± 6.8 | 35.0 ± 5.8 |
| Body mass (kg) | 74.3 ± 9.7 | 74.4 ± 7.6 |
| Body height (cm) | 177.2 ± 5.2 | 178.1 ± 6.8 |
| BMI (kg/m2) | 23.5 ± 1.9 | 23.5 ± 2.0 |
| FAT (%) | 13.6 ± 3.3 | 13.5 ± 4.1 |
| MM (kg) | 36.8 ± 5.3 | 36.9 ± 3.5 |
| TBW (L) | 47.2 ± 6.4 | 47.5 ± 5.4 |
| VO2max (mL/kg/min) | 55.0 ± 9.2 | 58.1 ± 7.4 |
| HR max (b/min) | 182.0 ± 10.0 | 186.0 ± 10.0 |
| Training volume (min/week) | 360.0 ± 55.0 | 380.0 ± 60.0 |
| Variables | ω-3 PUFA (n = 12) | Placebo (n = 12) |
|---|---|---|
| Energy [kcal/kg/day] | 29.6 ± 3.0 | 28.0 ± 2.0 |
| Fat [%] | 31.7 ± 9.6 | 30.8 ± 8.3 |
| Carbohydrate [%] | 46.1 ± 6.6 | 46.7 ± 8.5 |
| Protein [%] | 22.8 ± 5.4 | 22.4 ± 3.3 |
| Vitamin C [mg/day] | 171.2 ± 73.0 | 176.4 ± 122.0 |
| Vitamin E [mg/day] | 11.8 ± 3.6 | 11.2 ± 4.7 |
| Vitamin D [µg/day] | 7.9 ± 5.0 | 7.8 ± 7.3 |
| Cholesterol [mg/day] | 436.0 ± 228.6 | 423.1 ± 159.0 |
| SFA [g/day] | 37.4 ± 15.2 | 33.5 ± 9.6 |
| MUFA [g/day] | 29.6 ± 10.1 | 26.0 ± 8.7 |
| Variables | ω-3 PUFA (n = 12) | Placebo (n = 12) | p | ||
|---|---|---|---|---|---|
| Pre-Suppl | Post-Suppl | Pre-Placebo | Post-Placebo | Post-Suppl vs. Post-Placebo | |
| Omega-3 Index [%] | 3.9 ± 0.5 | 4.8 ± 0.8 *** | 3.5 ± 0.3 | 3.9 ± 0.4 | 0.01 |
| ω-3 FA [%] | 3.9 ± 0.5 | 4.8 ± 0.9 *** | 3.5 ± 0.4 | 4.1 ± 0.5 | 0.05 |
| Alpha-Linolenic [%] | 0.1 ± 0.1 | 0.1 ± 0.1 | 0.1 ± 0.0 | 0.2 ± 0.0 | ns |
| EPA [%] | 0.4 ± 0.2 | 0.8 ± 0.3 *** | 0.5 ± 0.1 | 0.7 ± 0.1 | ns |
| DPA [%] | 0.9 ± 0.2 | 1.0 ± 0.1 | 0.9 ± 0.2 | 1.0 ± 0.2 | ns |
| DHA [%] | 2.5 ± 0.6 | 2.9 ± 0.6 ** | 2.0 ± 0.3 | 2.1 ± 0.4 | 0.05 |
| AA/EPA | 17.7 ± 6.5 | 8.1 ± 2.4 *** | 14.2 ± 3.4 | 12.0 ± 3.7 | 0.05 |
| ω-6 FA [%] | 28.7 ± 2.8 | 27.4 ± 2.4 | 28.8 ± 3.1 | 28.9 ± 3.5 | ns |
| AA [%] | 6.6 ± 1.1 | 6.1 ± 1.2 | 7.3 ± 1.0 | 7.5 ± 1.0 | 0.05 |
| SFA [%] | 45.6 ± 1.8 | 47.3 ± 1.3 | 45.0 ± 1.9 | 45.6 ± 2.0 | ns |
| MUFA [%] | 20.8 ± 1.9 | 20.1 ± 1.7 | 22.7 ± 2.5 | 21.0 ± 2.1 | ns |
| SFA/MUFA | 2.2 ± 0.2 | 2.4 ± 0.2 | 2.0 ± 0.2 | 2.2 ± 0.2 | ns |
| TRANS index [%] | 0.2 ± 0.1 | 0.3 ± 0.1 | 0.3 ± 0.1 | 0.3 ± 0.1 | ns |
| Variables | ω-3 PUFA (n = 12) | Placebo (n = 12) | p | ||
|---|---|---|---|---|---|
| Pre-Suppl | Post-Suppl | Pre-Placebo | Post-Placebo | Post-Suppl vs. Post-Placebo | |
| Adiponectin [µg/mL] | 23.8 ± 8.5 | 31.4 ± 7.7 ** | 28.8 ± 8.5 | 30.1 ± 13.5 | ns |
| Leptin [ng/mL] | 3.3 ± 1.9 | 2.7 ± 1.0 ** | 2.6 ± 0.4 | 2.7 ± 0.3 | ns |
| NRG-4 [ng/mL] | 3.6 ± 2.1 | 5.4 ± 2.1 * | 2.9 ± 2.0 | 3.2 ± 1.6 | ns |
| TNFα [pg/mL] | 9.7 ± 2.7 | 5.6 ± 2.6 * | 13.7 ± 7.4 | 12.5 ± 2.4 | 0.05 |
| IL-6 [pg/mL] | 1.4 ± 1.3 | 1.9± 1.8 | 1.5 ± 1.3 | 2.2 ± 2.0 | ns |
| TCh [mg/dL] | 197.0 ± 31.4 | 210.0 ± 34.7 | 184.7 ± 15.4 | 187.3 ± 11.7 | ns |
| HDL-C [mg/dL] | 57.6 ± 12.3 | 68.9 ± 13.0 * | 60.5 ± 5.6 | 68.2 ± 11.8 | ns |
| LDL-C [mg/dL] | 114.2 ± 27.1 | 106.8 ± 30.3 | 104.0 ± 21.7 | 98.4 ± 15.2 | ns |
| TG [mg/dL] | 118.0 ± 27.1 | 103.0 ± 25.4 | 113.5 ± 25.1 | 87.7 ± 16.3 | ns |
| BG [mg/dL] | 80.3 ± 11.0 | 85.7 ± 12.0 | 90.2 ± 10.4 | 92.0 ± 15.1 | ns |
| Variable | F | p | ɳ2 | α |
|---|---|---|---|---|
| Adiponectin [µg/mL] | 22.7 | 0.000 | 0.51 | 1.0 |
| Leptin [ng/mL] | 2.3 | 0.14 | 0.09 | 0.3 |
| NRG4 [ng/mL] | 2.2 | 0.17 | 0.26 | 0.3 |
| TNF α [pg/mL] | 4.7 | 0.05 | 1.18 | 0.6 |
| IL-6 [pg/mL] | 0.4 | 0.59 | 0.02 | 0.2 |
| Cholesterol [mg/dl] | 0.7 | 0.42 | 0.03 | 0.1 |
| HDL [mg/dL] | 11.1 | 0.003 | 0.34 | 0.9 |
| LDL [mg/dL] | 0.4 | 0.53 | 0.02 | 0.1 |
| TG [mg/dL] | 1.2 | 0.31 | 0.05 | 0.3 |
| MDA [µmoL/L] | 46.1 | 0.000 | 0.68 | 1.0 |
| SOD [U/gHb] | 13.0 | 0.002 | 0.37 | 0.9 |
| GPx [U/gHb] | 8.9 | 0.007 | 0.29 | 0.8 |
| CAT [U/gHb] | 0.9 | 0.35 | 0.04 | 0.2 |
| GSH [µg/gHb] | 11.4 | 0.002 | 0.34 | 0.9 |
| α-Tocopherol [µmoL/L] | 13.4 | 0.004 | 0.55 | 0.9 |
| Variables | ω-3 PUFA (n = 12) | Placebo (n = 12) | p | ||
|---|---|---|---|---|---|
| Pre-Suppl | Post-Suppl | Pre-Placebo | Post-Placebo | Post-Suppl vs. Post-Placebo | |
| MDA [µmol/L] | 5.0 ± 1.1 | 5.6 ± 1.3 * | 5.3 ± 0.8 | 6.3 ± 1.3 | ns |
| SOD [U/gHb] | 1430 ± 220 | 1535 ± 259 ** | 1330 ± 115 | 1620 ± 407 | 0.05 |
| GPx [U/gHb] | 45.1 ± 9.4 | 57.4 ± 7.6 ** | 49.4 ± 11.4 | 59.5 ± 15.4 | ns |
| CAT [U/gHb] | 179.6 ± 23.4 | 185.6 ± 34.1 | 212.5 ± 26.0 | 207.4 ± 31.6 | ns |
| GSH [µg/gHb] | 2.7 ± 0.3 | 2.5 ± 0.3 | 2.8 ± 0.4 | 2.6 ± 0.2 | ns |
| α-Tocopherol [µmol/L] | 6.5 ± 2.0 | 8.7 ± 3.8 * | 4.8 ± 1.7 | 4.9 ± 2.5 | 0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Żebrowska, A.; Hall, B.; Stolecka-Warzecha, A.; Stanula, A.; Sadowska-Krępa, E. The Effect of Omega-3 Fatty Acid Supplementation on Serum Adipocytokines, Lipid Profile and Biochemical Markers of Inflammation in Recreational Runners. Nutrients 2021, 13, 456. https://doi.org/10.3390/nu13020456
Żebrowska A, Hall B, Stolecka-Warzecha A, Stanula A, Sadowska-Krępa E. The Effect of Omega-3 Fatty Acid Supplementation on Serum Adipocytokines, Lipid Profile and Biochemical Markers of Inflammation in Recreational Runners. Nutrients. 2021; 13(2):456. https://doi.org/10.3390/nu13020456
Chicago/Turabian StyleŻebrowska, Aleksandra, Barbara Hall, Anna Stolecka-Warzecha, Arkadiusz Stanula, and Ewa Sadowska-Krępa. 2021. "The Effect of Omega-3 Fatty Acid Supplementation on Serum Adipocytokines, Lipid Profile and Biochemical Markers of Inflammation in Recreational Runners" Nutrients 13, no. 2: 456. https://doi.org/10.3390/nu13020456
APA StyleŻebrowska, A., Hall, B., Stolecka-Warzecha, A., Stanula, A., & Sadowska-Krępa, E. (2021). The Effect of Omega-3 Fatty Acid Supplementation on Serum Adipocytokines, Lipid Profile and Biochemical Markers of Inflammation in Recreational Runners. Nutrients, 13(2), 456. https://doi.org/10.3390/nu13020456









