Feed Composition Differences Resulting from Organic and Conventional Farming Practices Affect Physiological Parameters in Wistar Rats—Results from a Factorial, Two-Generation Dietary Intervention Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Crop Production Methods
2.2. Experimental Feeds
2.3. Analysis of Rat Feeds
2.4. Experimental Animals, and Rearing/Housing Systems Used
2.5. Rat Growth Assessment
2.6. Sampling for Body Composition, Blood, and Spleen Lymphocyte Analysis
2.7. Body Composition and Physiological Parameters of Animals
2.8. Statistical Analyses
3. Results
3.1. Effect of Agronomic Practices on Feed Composition
3.2. Effect of Feed Composition on Rat Growth Parameters
3.2.1. Initial Body Weight at Weaning
3.2.2. Feed Intake
3.2.3. Total Weight Gain
3.2.4. Feed Conversion Ratio
3.3. Effect of Feed Composition on Rat Body Composition
3.4. Effect of Feed Composition on Basic Physiological Parameters in Blood
3.5. Effect of Feed Composition on Plasma Hormone Profiles
3.6. Effect of Feed Composition Differences on Immune System Parameters
3.6.1. Plasma Immunoglobulin A and G
3.6.2. Lymphocyte Proliferation
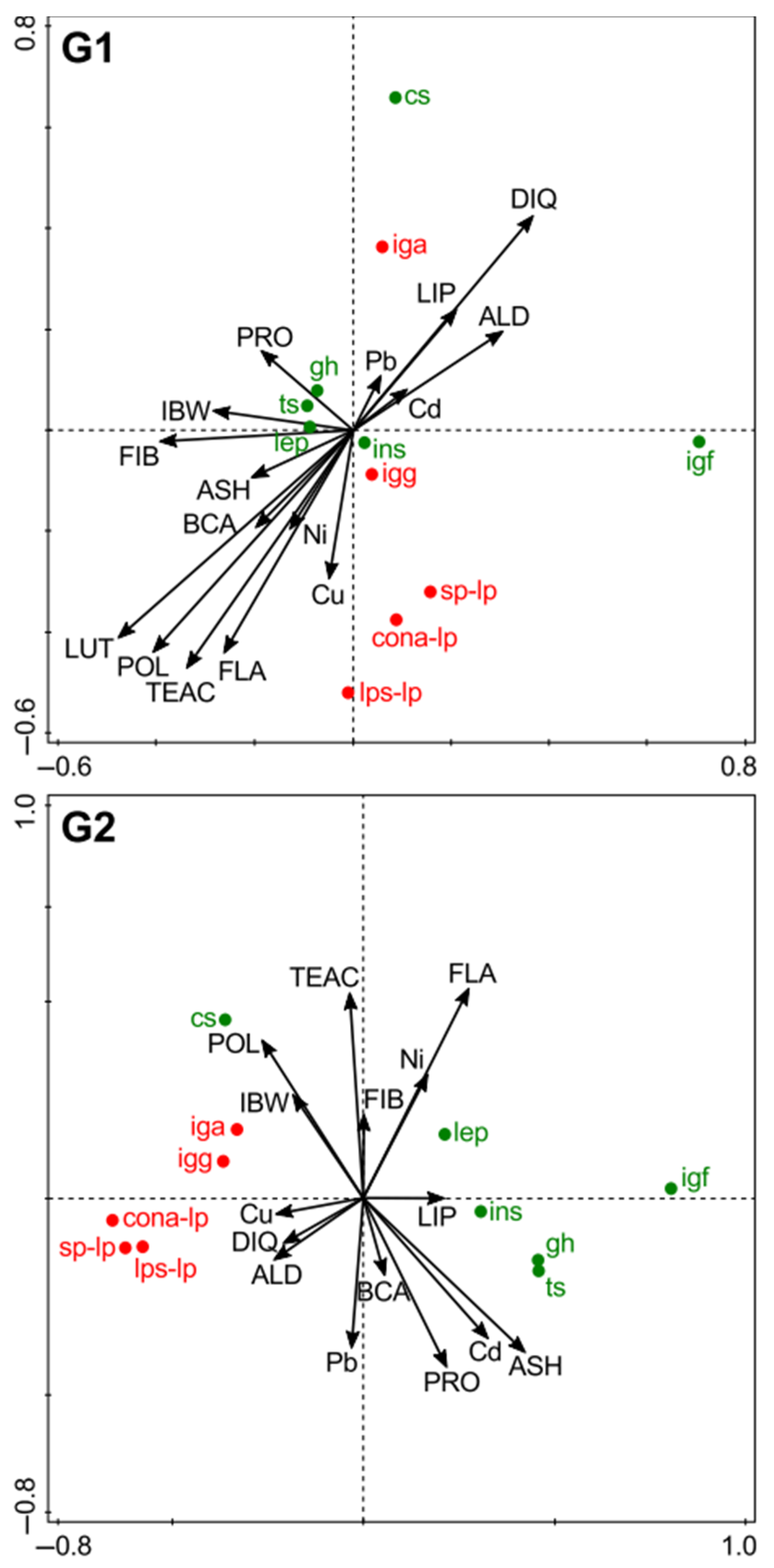
3.7. Associations Between Feed Composition, and Plasma Hormone and Immunological Parameters (RDA)
3.7.1. Plasma Hormone Profiles
3.7.2. Immunological Parameters
3.8. Correlations between Endocrine and Immunological Parameters
4. Discussion
4.1. Effect of Crop Management Practices on Feed Composition
4.2. Effects of Feed Composition Changes Resulting from Agrochemical Use on Basic Rat Physiological Parameters
4.2.1. Plasma Hormone Concentrations
4.2.2. Immunological Parameters
4.2.3. Rat Growth Parameters
4.3. Evidence for Epigenetic Changes and/or Development of ‘Adaptive’ Phenotypes
4.4. Study Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barański, M.; Średnicka-Tober, D.; Volakakis, N.; Seal, C.; Sanderson, R.; Stewart, G.B.; Benbrook, C.; Biavati, B.; Markellou, E.; Giotis, C.; et al. Higher antioxidant and lower cadmium concentrations and lower incidence of pesticide residues in organically grown crops: A systematic literature review and meta-analyses. Br. J. Nutr. 2014, 112, 794–811. [Google Scholar] [CrossRef] [PubMed]
- Średnicka-Tober, D.; Barański, M.; Seal, C.; Sanderson, R.; Benbrook, C.; Steinshamn, H.; Gromadzka-Ostrowska, J.; Rembiałkowska, E.; Skwarło-Sońta, K.; Eyre, M.; et al. Composition differences between organic and conventional meat: A systematic literature review and meta-analysis. Br. J. Nutr. 2016, 115, 994–1011. [Google Scholar] [CrossRef] [PubMed]
- Średnicka-Tober, D.; Barański, M.; Seal, C.J.; Sanderson, R.; Benbrook, C.; Steinshamn, H.; Gromadzka-Ostrowska, J.; Rembiałkowska, E.; Skwarło-Sońta, K.; Eyre, M.; et al. Higher PUFA and n-3 PUFA, conjugated linoleic acid, α-tocopherol and iron, but lower iodine and selenium concentrations in organic milk: A systematic literature review and meta- and redundancy analyses. Br. J. Nutr. 2016, 115, 1043–1060. [Google Scholar] [CrossRef]
- Sander, J.F.; Heitefuss, R. Suceptibility to Erysiphe graminis f.sp tritici and phenolic acid content of wheat as influenced by different levels of nitrogen fertilization. J. Phytopathol. 1998, 146, 495–507. [Google Scholar] [CrossRef]
- Cooper, J.; Sanderson, R.; Cakmak, I.; Ozturk, L.; Shotton, P.; Carmichael, A.; Haghighi, R.S.; Tetard-Jones, C.; Volakakis, N.; Eyre, M.; et al. Effect of organic and conventional crop rotation, fertilization, and crop protection practices on metal contents in wheat (Triticum aestivum). J. Agric. Food Chem. 2011, 59, 4715–4724. [Google Scholar] [CrossRef] [PubMed]
- Rempelos, L.; Almuayrifi, A.M.; Baranski, M.; Tetard-Jones, C.; Eyre, M.; Shotton, P.; Cakmak, I.; Ozturk, L.; Cooper, J.; Volakakis, N.; et al. Effects of agronomic management and climate on leaf phenolic profiles, disease severity and grain yield in organic and conventional wheat production systems. J. Agric. Food Chem. 2018, 66, 10369–10379. [Google Scholar] [CrossRef]
- Rempelos, L.; Almuayrifi, M.S.B.; Baranski, M.; Tetard-Jones, C.; Barkla, B.; Cakmak, I.; Ozturk, L.; Cooper, J.; Volakakis, N.; Hall, G.; et al. The effect of agronomic factors on crop health and performance of winter wheat varieties bred for the conventional and the low input farming sector. Field Crops Res. 2020, 254, 107822. [Google Scholar] [CrossRef]
- Baker, B.P.; Benbrook, C.M.; Groth, E., 3rd; Benbrook, K.L. Pesticide residues in conventional, integrated pest management (IPM)-grown and organic foods: Insights from three US data sets. Food Addit. Contam. 2002, 19, 427–446. [Google Scholar] [CrossRef]
- Wang, J.; Hasanalieva, G.; Wood, L.; Anagnostopoulos, C.; Ampadogiannis, G.; Bempelou, E.; Kiousi, M.; Markellou, E.; Iversen, P.O.; Seal, C.; et al. Effect of wheat species (Triticum aestivum vs T. spelta), farming system (organic vs conventional) and flour type (wholegrain vs white) on composition of wheat flour—Results of a retail survey in the UK and Germany—3. Pesticide residue content. Food Chem. X 2020, 100089. [Google Scholar] [CrossRef]
- Montonen, J.; Järvinen, R.; Knekt, O.; Reunanen, A. Dietary antioxidant intake and risk of type 2 diabetes. Diabetes Care 2004, 27, 362–366. [Google Scholar] [CrossRef]
- Abdali, D.; Samson, S.E.; Grover, A.K. How effective are antioxidant supplements in obesity and diabetes. Med. Princ. Pract. 2015, 24, 201–215. [Google Scholar] [CrossRef] [PubMed]
- Perez, M.C.; Knisley, B.D.; Crosby, G.E.; Zheng, S.; Barth, M. Obesity prevalence and dietary intake of antioxidants in native American adolescents. IJPHS 2016, 5, 222–227. [Google Scholar] [CrossRef]
- Stenzel, A.P.; Carvalho, R.; Jesus, P.; Bull, A.; Pereira, S.; Soboya, C.; Ramalho, A. Serum antioxidant associations with metabolic characteristics in metabolically healthy and unhealthy adolescents with severe obesity: An observational study. Nutrients 2018, 10, 150. [Google Scholar] [CrossRef] [PubMed]
- Pantavos, A.; Ruiter, R.; Feskens, E.F.; de Keyser, C.E.; Hofman, A.; Stricker, B.H.; Franco, O.H.; Kiefte-de Jong, J.C. Total dietary antioxidant capacity, individual antioxidant intake and breast cancer risk: The Rotterdam study. Int. J. Cancer 2014, 136, 2178–2186. [Google Scholar] [CrossRef]
- Egnell, M.; Fassier, P.; Lécuyer, L.; Gonzales, R.; Zelek, L.; Vasson, M.P.; Hercberg, S.; Latino-Martel, P.; Galan, P.; Druesne-Pecollo, N.; et al. Antioxidant intake from diet and supplements and risk of digestive cancers in middle-aged adults: Results from the prospective NutriNet-Santé cohort. Br. J. Nutr. 2017, 118, 541–549. [Google Scholar] [CrossRef]
- Brantsæter, A.L.; Haugen, M.; Samuelsen, S.O.; Torjusen, H.; Trogstad, L.; Alexander, J.; Magnus, P.; Meltzer, H.M. A dietary pattern characterized by high intake of vegetables, fruits, and vegetable oils is associated with reduced risk of preeclampsia in nulliparous pregnant norwegian women. J. Nutr. 2009, 139, 1162–1168. [Google Scholar] [CrossRef]
- Patel, S.; Murray, C.S.; Woodcock, A.; Simpson, A.; Custovic, A. Dietary antioxidant intake, allergic sensitisation and allergic diseases in young children. Allergy 2009, 64, 1766–1772. [Google Scholar] [CrossRef]
- Kao, Y.-H.; Hiipakka, R.A.; Liao, S. modulation of endocrine systems and food intake by green tea epigallocatechin gallate. Endocrinology 2000, 141, 980–987. [Google Scholar] [CrossRef]
- Alavanja, M.C.R.; Hoppin, J.A.; Kamel, F. Health effects of chronic pesticide exposure: Cancer and neurotoxicity. Annu. Rev. Public Health 2004, 25, 155–197. [Google Scholar] [CrossRef]
- Bassil, K.L.; Vakil, C.; Sanborn, M.; Cole, D.C.; Kaur, J.S.; Kerr, K.J. Cancer health effects of pesticides. Can. Fam. Physician 2007, 53, 1704–1711. [Google Scholar]
- Mnif, W.; Hassine, A.I.H.; Bouaziz, A.; Bartegi, A.; Thomas, O.; Roig, B. Effect of endocrine disruptor pesticides: A review. Int. J. Environ. Res. Public Health 2011, 8, 2265–2303. [Google Scholar] [CrossRef] [PubMed]
- Rocheleau, C.M.; Romitti, P.A.; Dennis, L.K. Pesticides and hypospadias: A meta-analysis. J. Pediatr. Urol. 2009, 5, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Guyton, K.Z.; Loomis, D.; Grosse, Y.; Ghissassi, F.E.; Benbrahim-Tallaa, L.; Guha, N.; Scoccianti, C.; Mattock, H.; Straif, K. Carcinogenicity of tetrachlorvinphos, parathion, malathion, diazinon, and glyphosate. Lancet Oncol. 2015, 16, 490–491. [Google Scholar] [CrossRef]
- Petrakis, D.; Vassilopoulou, L.; Mamoulakis, C.; Psycharakis, C.; Anifantaki, A.; Sifakis, S.; Docea, A.O.; Tsiaoussis, J.; Makrigiannakis, A.; Tsatsakis, A.M. Endocrine disruptors leading to obesity and related diseases. Int. J. Environ. Res. Public Health 2017, 14, 1282. [Google Scholar] [CrossRef] [PubMed]
- Chiu, Y.H.; Afeiche, M.C.; Gaskins, A.J.; Williams, P.L.; Petrozza, J.C.; Tanrikut, C.; Hauser, R.; Chavarro, J.E. Fruit and vegetable intake and their pesticide residues in relation to semen quality among men from a fertility clinic. Hum. Reprod. 2018, 30, 1342–1351. [Google Scholar] [CrossRef] [PubMed]
- Chiu, Y.H.; Williams, P.L.; Gillman, M.W.; Gaskins, A.J.; Mínguez-Alarcón, L.; Souter, I.; Toth, T.L.; Ford, J.B.; Hauser, R.; Chavarro, J.E. Association between pesticide residue intake from consumption of fruits and vegetables and pregnancy outcomes among women undergoing infertility treatment with assisted reproductive technology. JAMA Intern. Med. 2018, 178, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Mokarizadeh, A.; Faryabi, M.R.; Rezvanfar, M.A.; Abdollahi, M. A comprehensive review of pesticides and the immune dysregulation: Mechanisms, evidence and consequences. Toxicol. Mech. Methods 2015, 25, 258–278. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, D.; Ambrus, A.; Dieterle, R.; Felsot, A.; Harris, C.; Petersen, B.; Racke, K.; Wong, S.-S.; Gonzalez, R.; Tanaka, K.; et al. Pesticide residues in food—Acute dietary exposure. Pest Manag. Sci. 2004, 60, 311–339. [Google Scholar] [CrossRef]
- Winter, C.K. Chronic dietary exposure to pesticide residues in the United States. Int. J. Food Contam. 2015, 2, 11. [Google Scholar] [CrossRef]
- Markantonis, M.; van der Velde-Koerts, T.; Graven, C.; Te Biesebeek, J.D.; Zeilmaker, M.; Rietveld, A.G.; Ossendorp, B.C. Assessment of occupational and dietary exposure to pesticide residues. EFSA J. 2018, 16, e16087. [Google Scholar] [CrossRef]
- Heindel, J.J.; vom Saal, F.S. Role of nutrition and environmental endocrine disrupting chemicals during the prerinatal period on the aetiology of obesity. Mol. Cell. Endocrinol. 2009, 304, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Schug, T.T.; Janesick, A.; Blumberg, B.; Heindel, J.J. Endocrine Disrupting Chemicals and Disease Susceptibility. J. Steroid Biochem. Mol. Biol. 2011, 127, 204–215. [Google Scholar] [CrossRef] [PubMed]
- Yang, O.; Kim, H.L.; Weon, J.I.; Seo, Y.R. Endocrine-disrupting chemicals: Review of toxicological mechanisms using molecular pathway analysis. J. Cancer Prev. 2015, 20, 12–24. [Google Scholar] [CrossRef]
- Xin, F.; Susiarjo, M.; Bartolomei, M.S. Multigenerational and transgenerational effects of endocrine disrupting chemicals: A role for altered epigenetic regulation? Semin. Cell Dev. Biol. 2015, 43, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S.; Moore, M.R. Adverse Health Effects of Chronic Exposure to Low-Level Cadmium in Foodstuffs and Cigarette Smoke. Environ. Health Perspect. 2004, 112, 1099–1103. [Google Scholar] [CrossRef]
- Lugon-Moulin, N.; Ryan, L.; Donini, P.; Rossi, L. Cadmium content of phosphorus fertilizers used for tobacco production. Agron. Sustain. Dev. 2006, 26, 151–155. [Google Scholar] [CrossRef]
- Henson, M.C.; Chedrese, P.J. Endocrine disruption by cadmium, a common environmental toxicant with paradoxical effects on reproduction. Exp. Biol. Med. 2004, 229, 383–392. [Google Scholar] [CrossRef]
- Borello, P.; Giardino, A. Lead and cadmium at very low doses affect in vitro immune response of human lymphocytes. Environ. Res. 1991, 55, 165–177. [Google Scholar] [CrossRef]
- Hemdan, N.Y.; Emmrich, F.; Sack, U.; Wichmann, G.; Lehmann, J.; Adham, K.; Lehmann, I. The in vitro immune modulation by cadmium depends on the way of cell activation. Toxicology 2006, 222, 37–45. [Google Scholar] [CrossRef]
- Gerhard, I.; Waibel, S.; Daniel, V.; Runnebaum, B. Impact of heavy metals on hormonal and immunological factors in women with repeated miscarriages. Hum. Reprod. Update 1998, 4, 301–309. [Google Scholar] [CrossRef]
- Porter, W.P.; Jaeger, J.W.; Carlson, I.H. Endocrine, immune and behavioral effects of aldicarb (carbamate), atrazine (triazine) and nitrate (fertilizer) mixtures at groundwater concentrations. Toxicol. Ind. Health 1999, 15, 133–151. [Google Scholar] [CrossRef] [PubMed]
- Goel, A.; Chauhan, D.P.; Dhawan, D.K. Protective effects of Zinc in chlorpyriphos induced hapatoxicity: A biochemical and trace elemental study. Biol. Trace Elem. Res. 2000, 74, 171–183. [Google Scholar] [CrossRef]
- Hazarika, A.; Sarkar, S.N.; Hajare, S.; Kataria, M.; Malik, J.K. Influence of malathion pretreatment on the toxicity of anilofos in male rats: A biochemical interaction study. Toxicology 2003, 185, 1–8. [Google Scholar] [CrossRef]
- Kortenkamp, A. Ten years of mixing cocktails: A review of combination effects of endocrine-disrupting chemicals. Environ. Health Perspect. 2007, 115, 98–105. [Google Scholar] [CrossRef]
- Astiz, M.; de Alaniz, M.J.T.; Marra, C.A. Antioxidant defence system in rats simultaneously intoxicated with agrochemicals. Environ. Toxicol. Pharmacol. 2009, 28, 465–473. [Google Scholar] [CrossRef]
- Bradbury, K.E.; Balkwill, A.; Spencer, E.A.; Roddam, A.W.; Reeves, G.K.; Green, J.; Key, T.J.; Beral, V.; Pirie, K. Organic food consumption and the incidence of cancer in a large prospective study of women in the United Kingdom. Br. J. Cancer 2014, 110, 2321–2326. [Google Scholar] [CrossRef]
- Christensen, J.S.; Asklund, C.; Skakkebaek, N.E.; Jorgensen, N.; Andersen, H.R.; Jorgensen, T.M.; Olsen, L.H.; Hoyer, A.P.; Moesgaard, J.; Thorup, J.; et al. Association between organic dietary choice during pregnancy and hypospadias in offspring: A study of mothers of 306 boys operated on for hypospadias. J. Urol. 2013, 189, 1077–1082. [Google Scholar] [CrossRef]
- Torjusen, H.; Brantsæter, A.L.; Haugen, M.; Alexander, J.; Bakketeig, L.S.; Lieblein, G.; Stigum, H.; Næs, T.; Swartz, J.; Holmboe-Ottesen, G.; et al. Reduced risk of pre-eclampsia with organic vegetable consumption: Results from the prospective Norwegian Mother and Child Cohort Study. BMJ Open 2014, 4. [Google Scholar] [CrossRef]
- Brantsæter, A.L.; Torjusen, H.; Meltzer, H.M.; Papadopoulou, E.; Hoppin, J.A.; Alexander, J.; Lieblein, G.; Roos, G.; Holten, J.M.; Swartz, J.; et al. Organic food consumption during pregnancy and hypospadias and cryptorchidism at birth: The Norwegian Mother and Child Cohort Study (MoBa). Environ. Health Perspect. 2016, 124, 357–364. [Google Scholar] [CrossRef]
- Kesse-Guyot, E.; Baudry, J.; Assmann, K.E.; Galan, P.; Hercberg, S.; Lairon, D. Prospective association between consumption frequency of organic food and body weight change, risk of overweight or obesity: Results from the NutriNet-Santé Study. Br. J. Nutr. 2017, 117, 325–334. [Google Scholar] [CrossRef]
- Baudry, J.; Lelong, H.; Adriouch, S.; Julia, C.; Allès, B.; Hercberg, S.; Touvier, M.; Lairon, D.; Galan, P.; Kesse-Guyot, E. Association between organic food consumption and metabolic syndrome: Cross-sectional results from the NutriNet-Santé study. Eur. J. Nutr. 2018, 57, 2477–2488. [Google Scholar] [CrossRef] [PubMed]
- Baudry, J.; Assmann, K.E.; Touvier, M.; Allès, B.; Seconda, L.; Latino-Martel, P.; Ezzedine, K.; Galan, P.; Hercberg, S.; Lairon, D.; et al. Association of frequency of organic food consumption with cancer risk Findings from the NutriNet-Sante prospective cohort Study. JAMA Intern. Med. 2018, 178, 1597–1606. [Google Scholar] [CrossRef] [PubMed]
- Vigar, V.; Myers, S.; Oliver, C.; Arellano, J.; Robinson, S.; Leifert, C. A Systematic Review of Organic Versus Conventional Food Consumption: Is There a Measurable Benefit on Human Health? Nutrients 2020, 12, 7. [Google Scholar] [CrossRef] [PubMed]
- Alm, J.S.; Swartz, J.; Lilja, G.; Scheynius, A.; Pershagen, G. Atopy in children of families with an anthroposophic lifestyle. Lancet 1999, 353, 1485–1488. [Google Scholar] [CrossRef]
- Flöistrup, H.; Swartz, J.; Bergström, A.; Michaels, K.M.; Pershagen, G. Allergic disease and sensitization in Steiner school children. J. Allergy Clin. Immunol. 2006, 117, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Kesse-Guyot, E.; Peneau, S.; Mejean, C.; de Edelenyi, F.S.; Galan, P.; Hercberg, S.; Lairon, D. Profiles of organic food consumers in a large sample of French Adults: Results from the Nutrinet-Santé cohort study. PLoS ONE 2013, 8, e76998. [Google Scholar] [CrossRef]
- Baudry, J.; Méjean, C.; Allès, B.; Péneau, S.; Touvier, M.; Hercberg, S.; Lairon, D.; Galan, P.; Kesse-Guyot, E. Contribution of organic food to the diet in a large sample of French adults (the NutriNet-Santé Cohort Study). Nutrients 2015, 7, 8615–8632. [Google Scholar] [CrossRef]
- Baudry, J.; Allès, B.; Péneau, S.; Touvier, M.; Méjean, C.; Hercberg, S.; Galan, P.; Lairon, D.; Kesse-Guyot, E. Dietary intakes and diet quality according to levels of organic food consumption by French adults: Cross-sectional findings from the NutriNet-Santé Cohort Study. Public Health Nutr. 2017, 20, 638–648. [Google Scholar] [CrossRef]
- Srednicka-Tober, D.; Barański, M.; Gromadzka-Ostrowska, J.; Skwarło-Sońta, K.; Rembiałkowska, E.; Hajslova, J.; Schulzova, V.; Çakmak, I.; Öztürk, L.; Królikowski, T.; et al. Effect of crop protection and fertilization regimes used in organic and conventional production systems on feed composition and physiological parameters in rats. J. Agric. Food Chem. 2013, 61, 1017–1029. [Google Scholar] [CrossRef]
- Wang, J.; Chatzidimitriou, E.; Wood, L.; Hasanalieva, G.; Markellou, E.; Iversen, P.O.; Seal, C.; Baranski, M.; Vigar, V.; Ernst, L.; et al. Effect of wheat species (Triticum aestivum vs. T. spelta), farming system (organic vs conventional) and flour type (wholegrain vs white) on composition of wheat flour—Results of a retail survey in the UK and Germany—2. Antioxidant activity, and phenolic and mineral content. Food Chem. X 2020, 6, 100091. [Google Scholar] [CrossRef]
- Palmer, M.W.; Cooper, J.; Tétard-Jones, C.; Średnicka-Tober, D.; Barański, M.; Eyre, M.; Shotton, P.N.; Volakakis, N.; Cakmak, I.; Ozturk, L.; et al. The influence of organic and conventional fertilisation and crop protection practices, preceding crop, harvest year and weather conditions on yield and quality of potato (Solanum tuberosum) in a long-term management trial. Eur. J. Agron. 2013, 49, 83–92. [Google Scholar] [CrossRef]
- Dunn, G.A.; Bale, T.L. Maternal high-fat diet effects on third-generation female body size via the paternal lineage. Endocrinology 2011, 152, 2228–2236. [Google Scholar] [CrossRef] [PubMed]
- Parlee, S.D.; MacDougald, O.A. Maternal nutrition and risk of obesity in offspring: The Trojan horse of developmental plasticity. Biochim. Biophys. Acta Mol. Basis Dis. 2014, 1842, 495–506. [Google Scholar] [CrossRef] [PubMed]
- Alavian-Ghavanni, A.; Rüegg, J. Understanding epigenetic effects of endocrine disrupting chemicals: From mechanism to novel test methods. Basic Clin. Pharmacol. Toxicol. 2018, 122, 38–45. [Google Scholar] [CrossRef]
- Wang, S.; Moustaid-Moussa, N.; Chen, L.; Mo, H.; Shastri, A.; Su, R.; Bapat, P.; Kwun, I.; Shen, C.-L. Novel insights of dietary polyphenols and obesity. J. Nutr. Biochem. 2014, 25, 1–18. [Google Scholar] [CrossRef]
- Tinkov, A.A.; Filippini, T.; Ajsuvakova, O.P.; Aaseth, J.; Gluhcheva, Y.G.; Ivanova, J.M.; Bjørklund, G.; Skalnaya, M.G.; Gatiatulina, E.R.; Popova, E.V.; et al. The role of cadmium in obesity and diabetes. Sci. Total Environ. 2017, 601, 741–755. [Google Scholar] [CrossRef]
- Tian, Y.; Liu, Y.; Zhou, Y.; Liu, Y.; Lü, G.; Zheng, X.; Xu, D. Effect of aldicarb exposure on cellular immunity and antioxidant capacity in Kunming mice. Health 2015, 7, 830–837. [Google Scholar] [CrossRef][Green Version]
- NRC. Nutrient Requirements of Laboratory Animals, 4th ed.; The National Academies Press: Washington, DC, USA, 1995. [Google Scholar]
- AOAC. Official Methods of Analysis of the Association of Official Analytical Chemists, 15th ed.; Association of Official Analytical Chemists: Washington, DC, USA, 1990. [Google Scholar]
- Lauridsen, C.; Yong, C.; Halekoh, U.; Bügel, S.H.; Brandt, K.; Christensen, L.P.; Jørgensen, H. Rats show differences in some biomarkers of health when eating diets based on ingredients produced with three different cultivation strategies. J. Sci. Food Agric. 2008, 88, 720–732. [Google Scholar] [CrossRef]
- Harris, R.B.; Kelso, E.W.; Flatt, W.P.; Grill, H.J.; Bartness, T.J. Testosterone replacement does not normalize carcass composition in chronically decerebrate male rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 296, 1687–1694. [Google Scholar] [CrossRef]
- Kanashiro-Takeuchi, R.M.; Tziomalos, K.; Takeuchi, L.M.; Treuer, A.V.; Lamirault, G.; Dulce, R.; Hurtado, M.; Song, Y.; Block, N.L.; Rick, F.; et al. Cardioprotective effects of growth hormone-releasing hormone agonist after myocardial infarction. Proc. Natl. Acad. Sci. USA 2010, 107, 2604–2609. [Google Scholar] [CrossRef]
- Pinheiro, J.C.; Bates, D.M. Mixed-Effects Models in S and S-PLUS; Springer: New York, NY, USA, 2000. [Google Scholar]
- Crawley, M.J. The R Book; John Wiley & Sons, Ltd.: Chichester, UK, 2007. [Google Scholar]
- Ter Braak, C.J.F.; Smilauer, P. CANOCO Reference Manual and User’s Guide to Canoco for Windows: Software for Canonical Community Ordination; Version 4; Centre for Biometry: Wageningen, The Netherlands, 1998. [Google Scholar]
- Hasanaliyeva, G.; Chatzidimitrou, E.; Wang, J.; Baranski, M.; Volakakis, N.; Seal, C.; Rosa, E.A.S.; Iversen, P.O.; Vigar, V.; Barkla, B.; et al. Effects of Production Region, Production Systems and Grape Type/Variety on Nutritional Quality Parameters of Table Grapes; Results from a UK Retail Survey. Foods 2020, 9, 1874. [Google Scholar] [CrossRef] [PubMed]
- Lueck, L.; Schmidt, C.S.; Cooper, J.M.; Shotton, P.N.; Hajslova, J.; Schulzova, V.; Leifert, C. Effect of organic, low-input and conventional production systems on pesticide and growth regulator residues in wheat, potato and cabbage. In Improving Sustainability in Organic and Low Input Food Production Systems, Proceedings of the 3rd International Congress of the European Integrated Project Quality Low Input Food (QLIF), University of Hohenheim, Stuttgart, Germany, 20–23 March 2007; Niggli, U., Leifert, C., Alfoeldi, T., Lueck, L., Willer, H., Eds.; FiBL: Frick, Switzerland, 2007; pp. 96–99. Available online: https://www.researchgate.net/publication/255421475_Effect_of_organic_low-input_and_conventional_production_systems_on_pesticide_and_growth_regulator_residues_in_wheat_potato_and_cabbage (accessed on 1 January 2021).
- Wootton-Beard, P.; Ryan, L. Improving public health? The role of antioxidant-rich fruit and vegetable beverages. Food Res. Int. 2011, 44, 3135–3148. [Google Scholar] [CrossRef]
- Wang, H.J.; Liu, Z.P.; Jia, X.D.; Chen, H.; Tan, Y.J. Endocrine disruption of cadmium in rats using the OECD enhanced TG 407 test system. Biomed. Environ. Sci. 2014, 27, 950–959. [Google Scholar] [CrossRef] [PubMed]
- Abbaspour, N.; Hurrell, R.; Kelishadi, R. Review in iron and its importance for human health. J. Res. Med. Sci. 2014, 19, 164–174. [Google Scholar]
- Rose, M.S.; Crabtree, H.C.; Fletcher, K.; Wyatt, I. Biochemical effects of diquat and paraquat. Disturbance of the control of corticosteroid synthesis in rat adrenal and subsequent effects on the control of liver glycogen utilization. Biochem. J. 1974, 138, 437–443. [Google Scholar] [CrossRef]
- Marth, E.; Jelovcan, S.; Kleinhappl, B.; Gutschi, A.; Barth, S. The effect of heavy metals on the immune system at low concentrations. Int. J. Occup. Environ. Health 2001, 14, 375–386. [Google Scholar]
- McKinlay, R.; Plant, J.A.; Bell, J.N.B.; Voulvoulis, N. Endocrine disrupting pesticides: Implications for risk assessment. Environ. Int. 2008, 34, 168–183. [Google Scholar] [CrossRef]
- Grün, F.; Blumberg, B. Endocrine disrupters as obesogens. Mol. Cell Endocrinol. 2009, 304, 19–29. [Google Scholar] [CrossRef]
- Multigner, L.; Kadhel, P.; Pascal, M.; Huc-Terki, F.; Kercret, H.; Massart, C.; Janky, E.; Auger, J.; Jégou, B. Parallel assessment of male reproductive function in workers and wild rats exposed to pesticides in banana planta-tions in Guadeloupe. Environ. Health 2008, 7, 1–10. [Google Scholar] [CrossRef]
- Hughes, D.A. Dietary antioxidants and human immune function. Nutr. Bulletin 2008, 25, 35–41. [Google Scholar] [CrossRef]
- Chen, L.; Qu, G.; Sun, X.; Zhang, S.; Wang, L.; Sang, N.; Du, Y.; Liu, J.; Sijin, L. Characterization of the inter-action between cadmium and chlorpyrifos with integrative techniques in incurring synergistic hepatoxicity. PLoS ONE 2013, 8, e59553. [Google Scholar] [CrossRef]
- Langhans, W. Food components in health promotion and disease prevention. J. Agric. Food Chem. 2018, 66, 2287–2294. [Google Scholar] [CrossRef] [PubMed]
- Myers, M.G., Jr.; Leibel, R.L.; Seeley, R.J.; Schwartz, M.W. Obesity and leptin resistance: Distinguishing cause from effect. Trends Endocrinol. Metab. 2010, 21, 643–651. [Google Scholar] [CrossRef] [PubMed]
- Maurer, A.; Chen, Q.; McPherson, C.; Rainer, R.A. Changes in satiety hormones and expression of genes involved in glucose and lipid metabolism in rats weaned onto diets high in fibre or protein reflect susceptibility to increased fat mass in adulthood. J. Physiol. 2009, 587, 679–691. [Google Scholar] [CrossRef]
- Zhang, R.; Jiao, J.; Zhang, W.; Zhang, Z.; Zhang, W.; Qin, L.-Q.; Han, S.-F. Effects of cereal fiber on leptin resistance and sensitivity in C57BL/6J mice fed a high-fat/cholesterol diet. Food Nutr. Res. 2016, 60, 31690. [Google Scholar] [CrossRef]
- Nishijo, M.; Nakagawa, H.; Honda, R.; Tanebe, K.; Saito, S.; Teranishi, H.; Tawara, K. Effects of maternal exposure to cadmium on pregnancy outcome and breast milk. Occup. Environ. Med. 2002, 59, 394–397. [Google Scholar] [CrossRef]
- Rebelo, F.M.; Caldas, E.D. Arsenic, lead, mercury and cadmium: Toxicity, levels in breast milk and the risks for breastfed infants. Environ. Res. 2016, 151, 671–688. [Google Scholar] [CrossRef]
- Coutinho, A.E.; Chapman, K.E. The anti-inflammatory and immunosuppressive effects of glucocorticoids, recent developments and mechanistic insights. Mol. Cell. Endocrinol. 2011, 335, 2–13. [Google Scholar] [CrossRef]
- Blalock, J.E. Shared ligands and receptors as a molecular mechanism for communication between the immune and neuroendocrine systems. Ann. N. Y. Acad Sci. 1994, 741, 292–298. [Google Scholar] [CrossRef]
- Procaccini, C.; Pucino, V.; De Rosa, V.; Marone, G.; Matarese, G. Neuro-endocrine networks controlling immune system in health and disease. Front. Immunol. 2014, 5, 143. [Google Scholar] [CrossRef]
- Considine, R.V.; Sinha, M.K.; Heiman, M.L.; Kriauciunas, A.; Stephens, T.W.; Nyce, M.R.; Ohannesian, J.P.; Marco, C.C.; McKee, L.J.; Bauer, T.L.; et al. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N. Engl. J. Med. 1996, 334, 292–295. [Google Scholar] [CrossRef] [PubMed]
- Wallace, A.M.; McMahon, A.D.; Packard, C.J.; Kelly, A.; Shepherd, J.; Gaw, A.; Sattar, N. Plasma Leptin and the Risk of Cardiovascular Disease in the West of Scotland Coronary. Circulation 2001, 104, 3052–3056. [Google Scholar] [CrossRef] [PubMed]
- Hamer, M.; Chida, Y. Intake of fruit, vegetable, and antioxidants and risk of type 2 diabetes: Systematic review and meta-analysis. J. Hypertens. 2007, 25, 2361–2369. [Google Scholar] [CrossRef] [PubMed]
- Kadota, A.; Hozawa, A.; Okamura, T.; Kadowak, T.; Makmaura, K.; Murakami, Y.; Hayakawa, T.; Kita, Y.; Okayama, A.; Nakamura, Y.; et al. Relationship between metabolic risk factor clustering and cardiovascular Mortality stratified by high blood Glucose and obesity NIPPON DATA90, 1990–2000. Diabetes Care 2007, 30, 1533–1538. [Google Scholar] [CrossRef] [PubMed]
- Symonds, M.E.; Sebert, S.; Budge, H. The obesity epidemic: From the environment to epigenetics—Not simply a response to dietary manipulation in a thermoneutral environment. Front. Genet. 2011, 2, 24. [Google Scholar] [CrossRef]
- Ribaroff, G.A.; Wastnedge, E.; Drake, A.J.; Sharpe, R.M.; Chambers, T.J.G. Animal models of maternal high fat diet exposure and effects on metabolism in offspring: A meta-regression analysis. Obes. Rev. 2017, 18, 673–686. [Google Scholar] [CrossRef]
- Kohmura, Y.K.; Kanayama, N.; Muramatsu, K.; Tamura, N.; Yaguchi, C.; Uchida, T.; Suzuki, K.; Sugihara, K.; Aoe, S.; Sasaki, T.; et al. association between body weight at weaning and remodeling in the subcutaneous adipose tissue of obese adult mice with undernourishment in utero. Reprod. Sci. 2013, 20, 813–827. [Google Scholar] [CrossRef][Green Version]
- Doo, M.; Kim, Y. The consumption of dietary antioxidant vitamins modifies the risk of obesity among Korean men with short sleep Duration. Nutrients 2017, 9, 780. [Google Scholar] [CrossRef]
- Tang, M. Protein Intake during the First Two Years of Life and Its Association with Growth and Risk of Overweight. Int. J. Environ. Res. Public Health 2018, 8, 1742. [Google Scholar] [CrossRef]
- Sarkies, P. Molecular mechanisms of epigenetic inheritance: Possible evolutionary implications. Semin. Cell Dev. Biol. 2020, 97, 106–115. [Google Scholar] [CrossRef]
- Dupont, C.; Armant, D.R.; Brenner, C.A. Epigenetics: Definition, mechanisms and clinical perspective. Semin. Reprod. Med. 2009, 27, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Plunk, E.C.; Richards, S.M. Epigenetic modifications due to environment, ageing, nutrition, and endocrine disrupting chemicals and their effects on the endocrine system. Int. J. Endocrinol. 2020, 2020, 9251980. [Google Scholar] [CrossRef] [PubMed]
- Holliday, R. Epigenetics: A historical overview. Epigenetics 2006, 1, 76–80. [Google Scholar] [CrossRef]
- Kaufman, P.D.; Rando, O.J. Chromatin as a potential carrier of heritable information. Curr. Opin. Cell Biol. 2010, 22, 284–290. [Google Scholar] [CrossRef] [PubMed]
- Bühler, M. RNA turnover and chromatin-dependent gene silencing. Chromosoma 2009, 118, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Harvey, Z.H.; Chen, Y.; Jarosz, D.F. Protein-based inheritance: Epigenetics beyond the chromosome. Mol. Cell 2018, 69, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Milagro, F.I.; Mansego, M.L.; De Miguel, C.; Martínez, J.A. Dietary factors, epigenetic modifications and obesity outcomes: Progresses and perspectives. Mol. Asp. Med. 2013, 34, 782–812. [Google Scholar] [CrossRef]
- Amarasekera, M.; Prescott, S.L.; Palmer, D.J. Nutrition in early life, immune-programming and allergies. Asian Pac. J. Allergy Immunol. 2013, 31, 175–182. [Google Scholar]
- Yong, S.-B.; Wu, C.C.; Wang, L.; Yang, K. Influence and mechanisms of maternal and infant diets on the development of childhood asthma. Pediatrics Neonatol. 2013, 54, 5–11. [Google Scholar] [CrossRef][Green Version]
- Bishop, K.S.; Ferguson, L.R. The interaction between epigenetics, nutrition and the development of cancer. Nutrients 2015, 7, 922–948. [Google Scholar] [CrossRef]
- Topart, C.; Werner, E.; Arimondo, P.B. Wandering along the epigenetic timeline. Clin. Epigenetics 2020, 12, 97. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.-W.; Friso, S. Epigenetics: A new bridge between nutrition and health. Adv. Nutr. 2010, 1, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Tryndyak, V.P.; Ross, S.A.; Beland, F.A.; Pogribny, I.P. Down-regulation of the microRNAs miR-34a, miR-127, and miR-200b in rat liver during hepatocarcinogenesis induced by a methyl-deficient diet. Mol. Carcinog. 2009, 48, 479–487. [Google Scholar] [CrossRef] [PubMed]
- Gerhauser, C. Impact of dietary gut microbial metabolites on the epigenome. Philos. Trans. R. Soc. B Biol. Sci. 2018, 373, 20170359. [Google Scholar] [CrossRef]
- Fang, M.; Chen, D.; Yang, C.S. Dietary polyphenols may affect DNA methylation. J. Nutr. 2007, 137, 223–228. [Google Scholar] [CrossRef]
- Wang, L.-S.; Arnold, M.; Huang, Y.-W.; Sardo, C.; Seguin, C.; Martin, E.; Huang, T.H.-M.; Riedl, K.; Schwartz, S.; Frankel, W.; et al. Modulation of genetic and epigenetic biomarkers of colorectal cancer in humans by black raspberries: A phase I pilot study. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2011, 17, 598–610. [Google Scholar] [CrossRef]
- Pudenz, M.; Roth, K.; Gerhauser, C. Impact of soy isoflavones on the epigenome in cancer prevention. Nutrients 2014, 6, 4218–4272. [Google Scholar] [CrossRef]
- Lascano, S.; Lopez, M.; Arimondo, P.B. Natural products and chemical biology tools: Alternatives to target epigenetic mechanisms in cancers. Chem. Rec. 2018, 18, 1854–1876. [Google Scholar] [CrossRef]
- Donley, N. The USA lags behind other agricultural nations in banning harmful pesticides. Environ. Health 2019, 18, 1–12. [Google Scholar] [CrossRef]
- King, A.J.F. The use of animal models in diabetes research. Br. J. Pharmacol. 2012, 166, 877–894. [Google Scholar] [CrossRef]
- Buettner, R.; Schölmerich, D.; Bollheimer, L.C. High-fat diets: Modelling the metabolic disorders of human obesity in rodents. Obesity 2007, 15, 798–808. [Google Scholar] [CrossRef]
- Lutz, T.A.; Woods, S.C. Overview of animal models for obesity. Curr. Protoc. Pharmacol. 2012, 58, 5–61. [Google Scholar] [CrossRef] [PubMed]
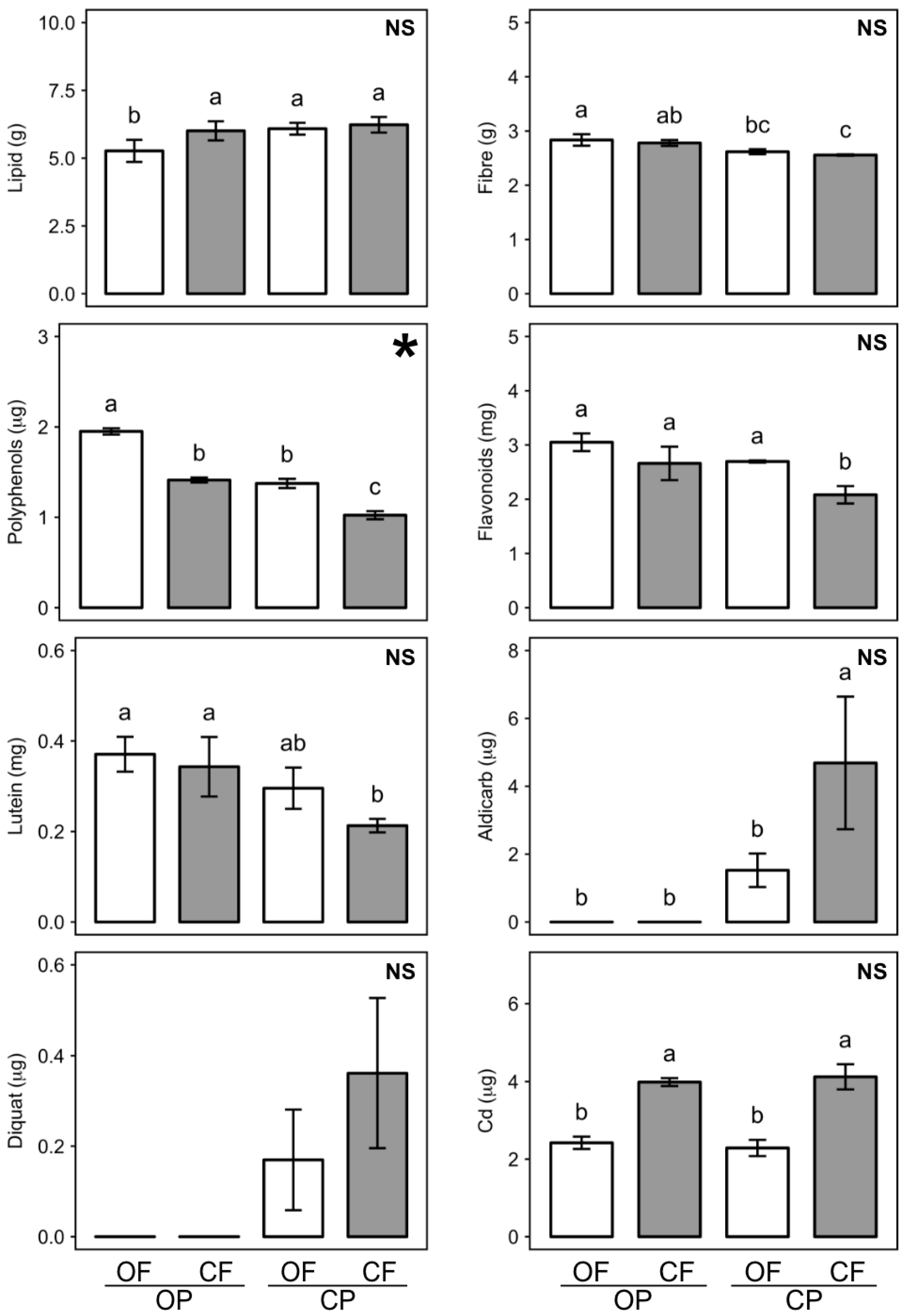
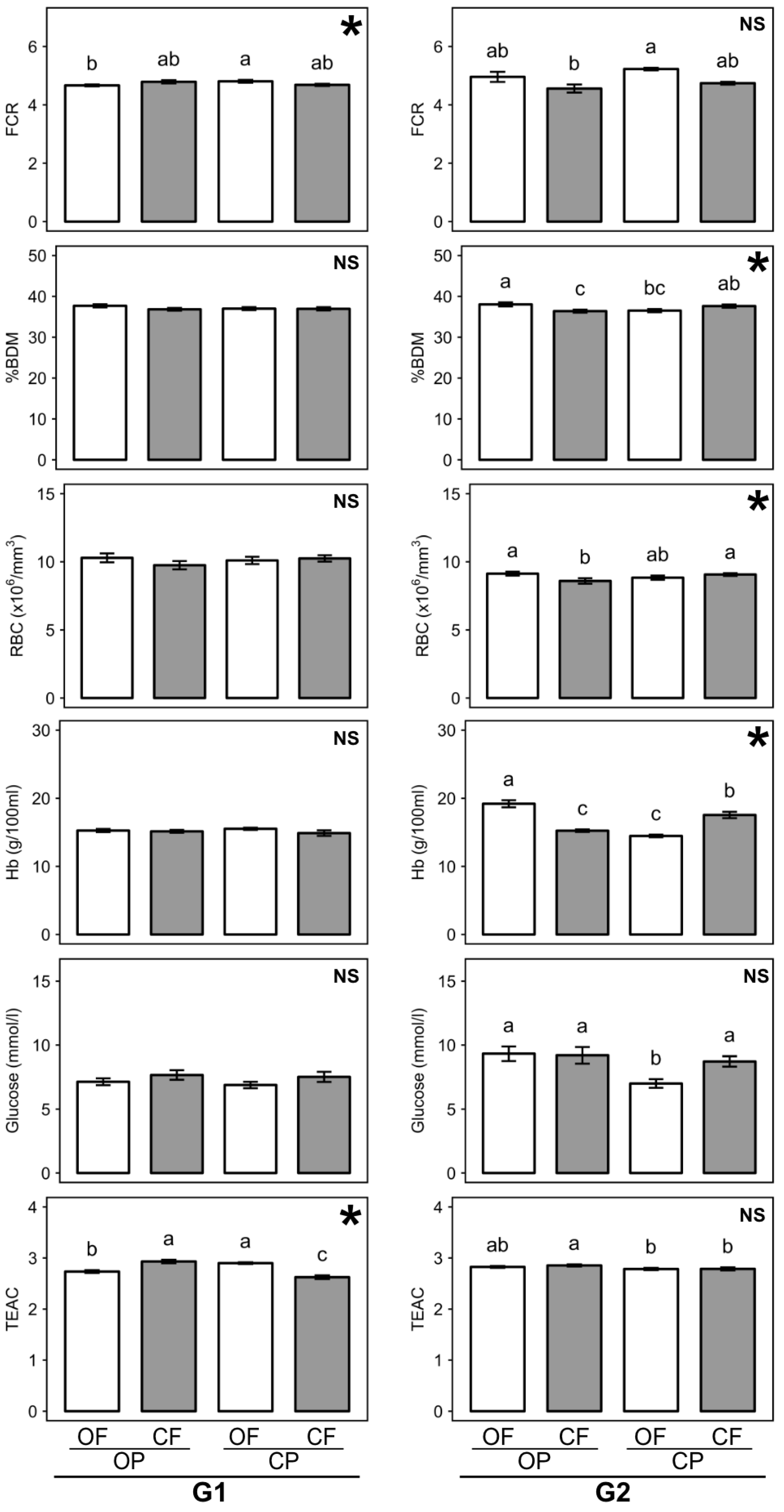
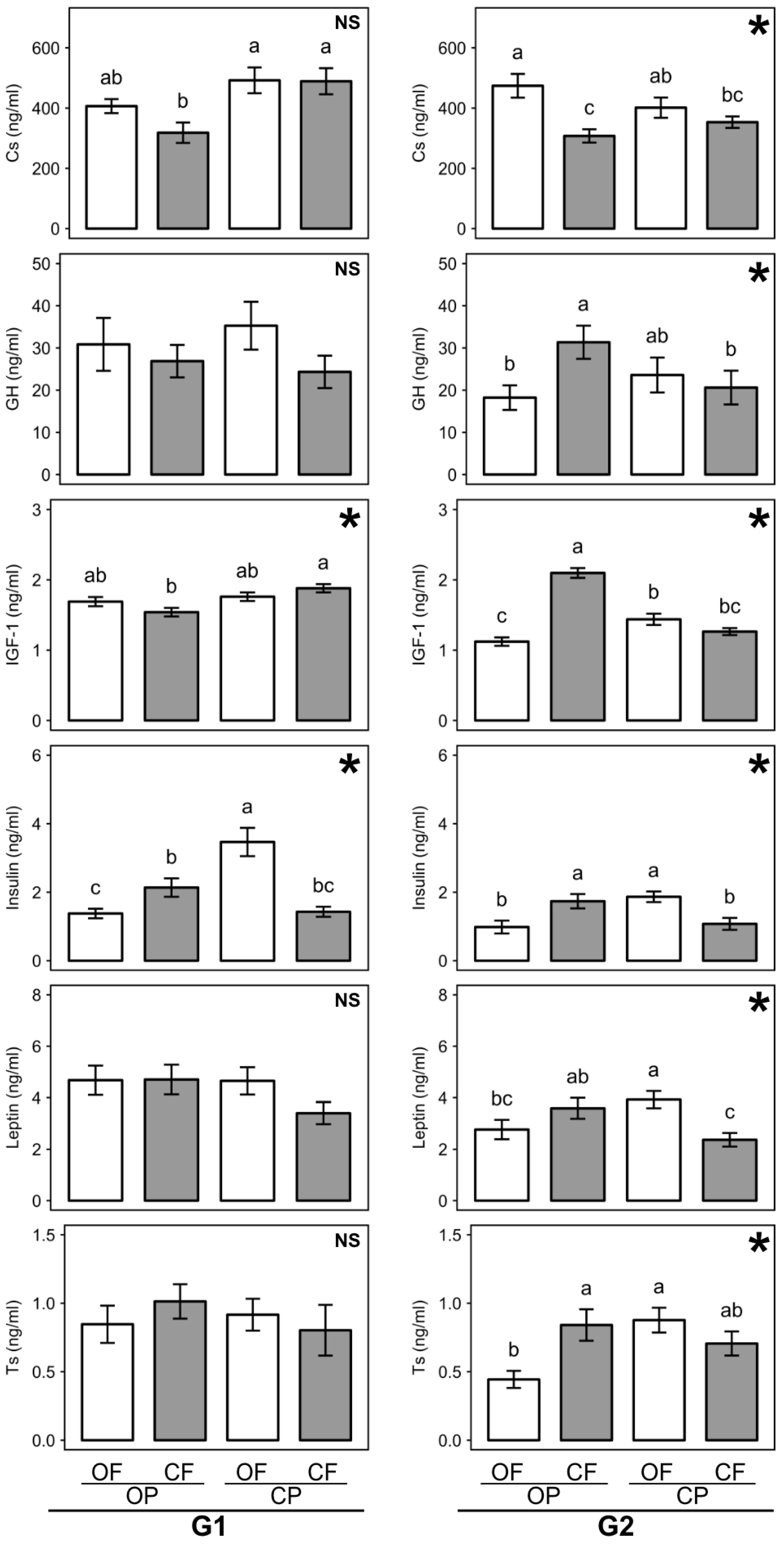
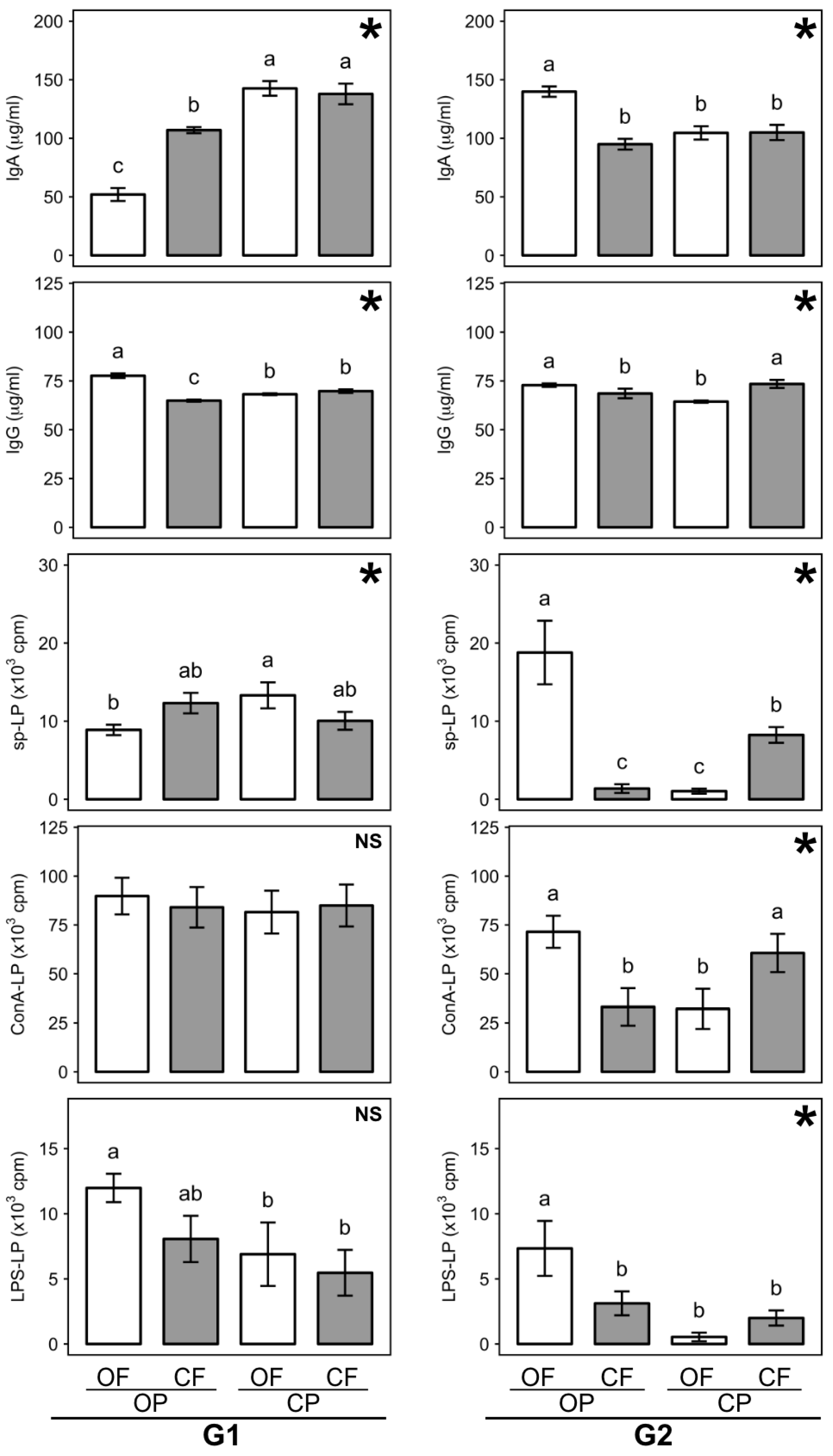
| Factor | Initial Body Weight at Weaning (g) | Feed Intake (g/day) | Total Weight Gain (g) | Feed Conversion Ratio (%) | ||||
|---|---|---|---|---|---|---|---|---|
| G1 | G2 | G1 | G2 | G1 | G2 | G1 | G2 | |
| Crop protection | ||||||||
| Organic (-pesticides) | 60 ± 1 | 95 ± 5 | 18.3 ± 0.3 | 16.9 ± 0.3 | 263 ± 3 | 221 ± 4 | 4.73 ± 0.03 | 4.75 ± 0.11 |
| Conventional (+pesticides) | 62 ± 1 | 114 ± 2 | 18.6 ± 0.2 | 17.8 ± 0.2 | 266 ± 3 | 219 ± 3 | 4.74 ± 0.03 | 4.99 ± 0.05 |
| Fertilization | ||||||||
| Organic (manure) | 61 ± 1 | 110 ± 4 | 17.9 ± 0.2 | 17.3 ± 0.2 | 256 ± 3 | 209 ± 3 | 4.73 ± 0.03 | 5.10 ± 0.09 |
| Conventional (mineral NPK) | 62 ± 1 | 99 ± 4 | 19.0 ± 0.2 | 17.4 ± 0.3 | 273 ± 3 | 230 ± 4 | 4.74 ± 0.03 | 4.65 ± 0.08 |
| ANOVA results | ||||||||
| (p-values *) | ||||||||
| Main effects | ||||||||
| Crop protection (P) | 0.200 | <0.001 | 0.158 | 0.002 | 0.360 | 0.924 | 0.703 | 0.020 |
| Fertilisation (F) | 0.644 | 0.507 | 0.050 | 0.856 | 0.050 | 0.141 | 0.985 | 0.184 |
| Interaction (PxF) | 0.984 | 0.256 | 0.080 | 0.485 | 0.680 | 0.356 | 0.006 | 0.664 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barański, M.; Średnicka-Tober, D.; Rempelos, L.; Hasanaliyeva, G.; Gromadzka-Ostrowska, J.; Skwarło-Sońta, K.; Królikowski, T.; Rembiałkowska, E.; Hajslova, J.; Schulzova, V.; et al. Feed Composition Differences Resulting from Organic and Conventional Farming Practices Affect Physiological Parameters in Wistar Rats—Results from a Factorial, Two-Generation Dietary Intervention Trial. Nutrients 2021, 13, 377. https://doi.org/10.3390/nu13020377
Barański M, Średnicka-Tober D, Rempelos L, Hasanaliyeva G, Gromadzka-Ostrowska J, Skwarło-Sońta K, Królikowski T, Rembiałkowska E, Hajslova J, Schulzova V, et al. Feed Composition Differences Resulting from Organic and Conventional Farming Practices Affect Physiological Parameters in Wistar Rats—Results from a Factorial, Two-Generation Dietary Intervention Trial. Nutrients. 2021; 13(2):377. https://doi.org/10.3390/nu13020377
Chicago/Turabian StyleBarański, Marcin, Dominika Średnicka-Tober, Leonidas Rempelos, Gultakin Hasanaliyeva, Joanna Gromadzka-Ostrowska, Krystyna Skwarło-Sońta, Tomasz Królikowski, Ewa Rembiałkowska, Jana Hajslova, Vera Schulzova, and et al. 2021. "Feed Composition Differences Resulting from Organic and Conventional Farming Practices Affect Physiological Parameters in Wistar Rats—Results from a Factorial, Two-Generation Dietary Intervention Trial" Nutrients 13, no. 2: 377. https://doi.org/10.3390/nu13020377
APA StyleBarański, M., Średnicka-Tober, D., Rempelos, L., Hasanaliyeva, G., Gromadzka-Ostrowska, J., Skwarło-Sońta, K., Królikowski, T., Rembiałkowska, E., Hajslova, J., Schulzova, V., Cakmak, I., Ozturk, L., Hallmann, E., Seal, C., Iversen, P. O., Vigar, V., & Leifert, C. (2021). Feed Composition Differences Resulting from Organic and Conventional Farming Practices Affect Physiological Parameters in Wistar Rats—Results from a Factorial, Two-Generation Dietary Intervention Trial. Nutrients, 13(2), 377. https://doi.org/10.3390/nu13020377












