Effects of a Protein-Rich, Low-Glycaemic Meal Replacement on Changes in Dietary Intake and Body Weight Following a Weight-Management Intervention—The ACOORH Trial
Abstract
1. Introduction
2. Materials and Methods
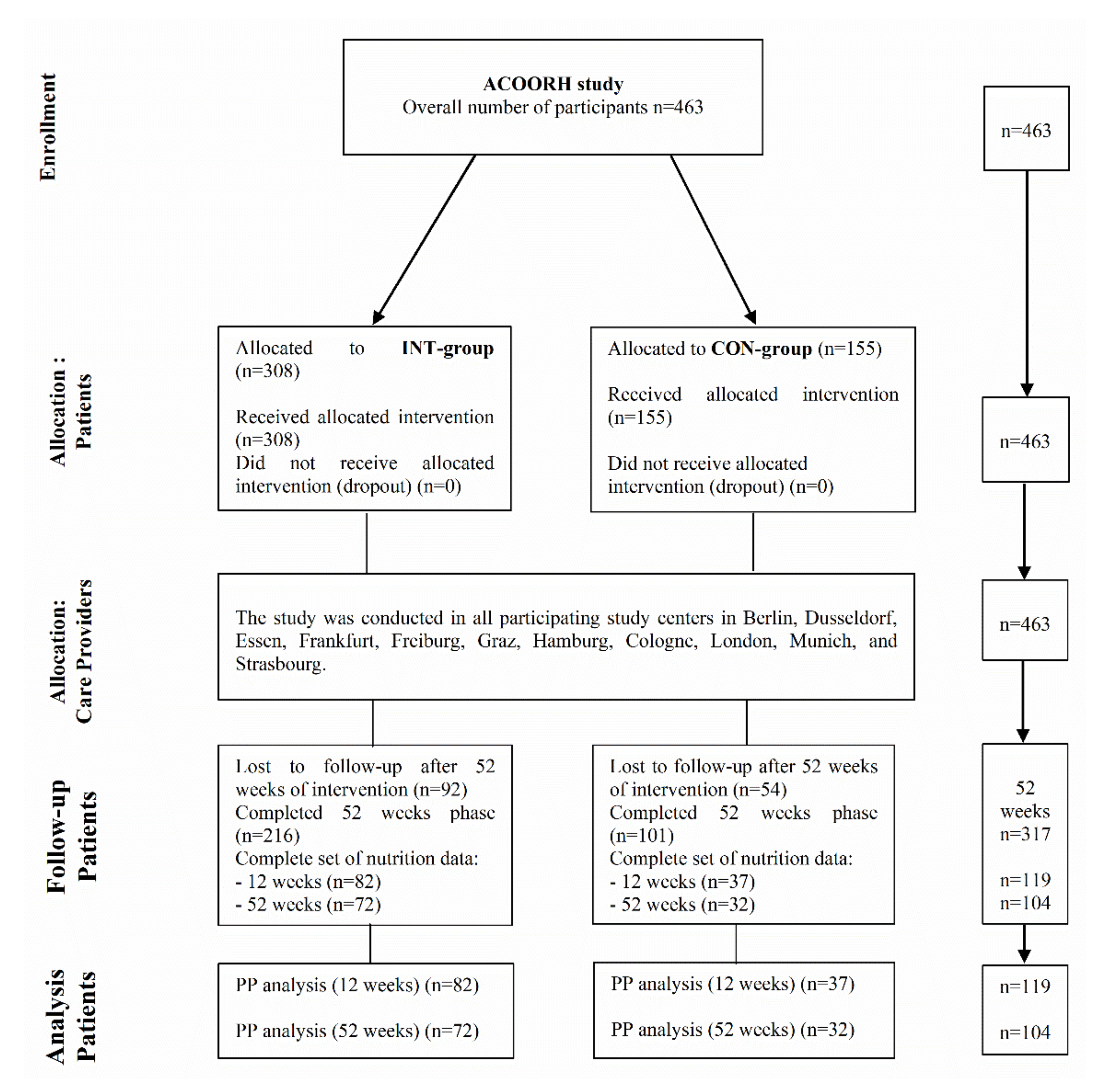
2.1. Study Design and Participants
2.2. Intervention and Monitoring
2.3. Statistical Analyses
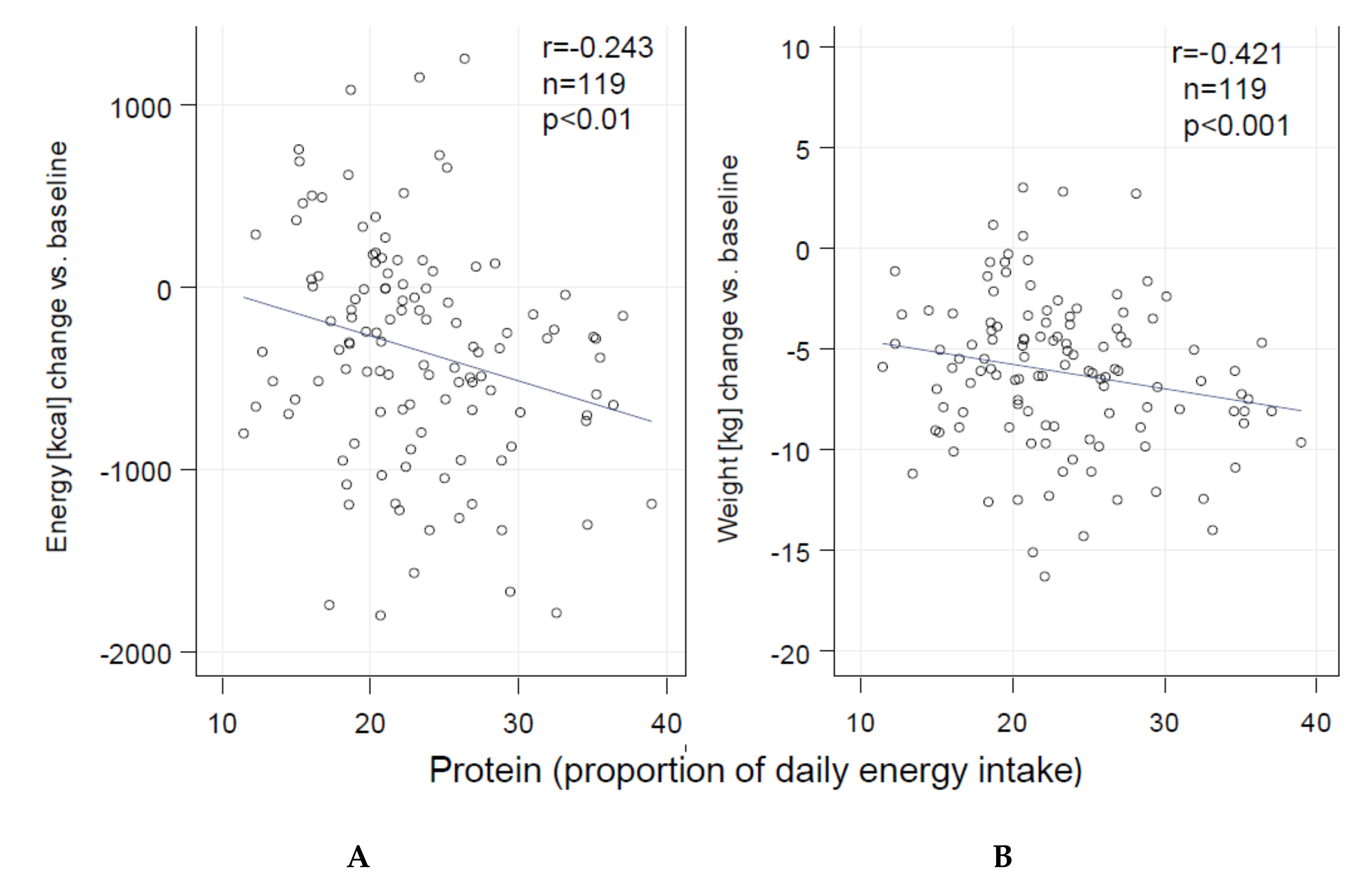
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kempf, K.; Altpeter, B.; Berger, J.; Reuß, O.; Fuchs, M.; Schneider, M.; Gärtner, B.; Niedermeier, K.; Martin, S. Efficacy of the Telemedical Lifestyle intervention Program TeLiPro in Advanced Stages of Type 2 Diabetes: A Randomized Controlled Trial. Diabetes Care 2017, 40, 863–871. [Google Scholar] [CrossRef] [PubMed]
- Leslie, W.S.; Taylor, R.; Harris, L.; Lean, M.E.J. Weight losses with low-energy formula diets in obese patients with and without type 2 diabetes: Systematic review and meta-analysis. Int. J. Obes. 2017, 41, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Bowen, J.; Brindal, E.; James-Martin, G.; Noakes, M. Randomized Trial of a High Protein, Partial Meal Replacement Program with or without Alternate Day Fasting: Similar Effects on Weight Loss, Retention Status, Nutritional, Metabolic, and Behavioral Outcomes. Nutrients 2018, 10, 1145. [Google Scholar] [CrossRef]
- Kempf, K.; Röhling, M.; Niedermeier, K.; Gärtner, B.; Martin, S. Individualized Meal Replacement Therapy Improves Clinically Relevant Long-Term Glycemic Control in Poorly Controlled Type 2 Diabetes Patients. Nutrients 2018, 10, 1022. [Google Scholar] [CrossRef] [PubMed]
- Lean, M.E.; Leslie, W.S.; Barnes, A.C.; Brosnahan, N.; Thom, G.; McCombie, L.; Peters, C.; Zhyzhneuskaya, S.; Al-Mrabeh, A.; Hollingsworth, K.G.; et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): An open-label, cluster-randomised trial. Lancet 2018, 391, 541–551. [Google Scholar] [CrossRef]
- Halle, M.; ACOORH Study Group; Röhling, M.; Banzer, W.; Braumann, K.M.; Kempf, K.; McCarthy, D.; Schaller, N.; Predel, H.G.; Scholze, J.; et al. Meal replacement by formula diet reduces weight more than a lifestyle intervention alone in patients with overweight or obesity and accompanied cardiovascular risk factors—The ACOORH trial. Eur. J. Clin. Nutr. 2020, 1–9. [Google Scholar] [CrossRef]
- Röhling, M.; Kempf, K.; Banzer, W.; Berg, A.; Braumann, K.-M.; Tan, S.; Halle, M.; McCarthy, H.D.; Pinget, M.; Predel, H.-G.; et al. Prediabetes Conversion to Normoglycemia Is Superior Adding a Low-Carbohydrate and Energy Deficit Formula Diet to Lifestyle Intervention—A 12-Month Subanalysis of the ACOORH Trial. Nutrients 2020, 12, 2022. [Google Scholar] [CrossRef]
- American_Diabetes_Association. 4. Lifestyle Management: Standards of Medical Care in Diabetes-2018. Diabetes Care 2018, 41, S38–S50. [Google Scholar] [CrossRef]
- Wirth, A.; Wabitsch, M.; Hauner, H. The Prevention and Treatment of Obesity. Dtsch. Aerzteblatt Online 2014, 111, 705–713. [Google Scholar] [CrossRef]
- Leader, N.J.; Ryan, L.; Molyneaux, L.; Yue, D.K. How best to use partial meal replacement in managing overweight or obese patients with poorly controlled type 2 diabetes. Obesity 2013, 21, 251–253. [Google Scholar] [CrossRef]
- Astbury, N.M.; Piernas, C.; Hartmann-Boyce, J.; Lapworth, S.; Aveyard, P.; Jebb, S. A systematic review and meta-analysis of the effectiveness of meal replacements for weight loss. Obes. Rev. 2019, 20, 569–587. [Google Scholar] [CrossRef] [PubMed]
- Steven, S.; Taylor, R. Restoring normoglycaemia by use of a very low calorie diet in long- and short-duration Type 2 diabetes. Diabet. Med. 2015, 32, 1149–1155. [Google Scholar] [CrossRef] [PubMed]
- Röhling, M.; Martin, K.; Ellinger, S.; Schreiber, M.; Martin, M.S.; Kempf, K. Weight Reduction by the Low-Insulin-Method—A Randomized Controlled Trial. Nutrients 2020, 12, 3004. [Google Scholar] [CrossRef] [PubMed]
- Kempf, K.; Schloot, N.C.; Gärtner, B.; Keil, R.; Schadewaldt, P.; Martin, S. Meal replacement reduces insulin requirement, HbA1c and weight long-term in type 2 diabetes patients with >100 U insulin per day. J. Hum. Nutr. Diet. 2014, 27, 21–27. [Google Scholar] [CrossRef]
- Hartmann-Boyce, J.; Johns, D.J.; Jebb, S.A.; Summerbell, C.; Aveyard, P. Behavioural Weight Management Review Group Behavioural weight management programmes for adults assessed by trials conducted in everyday contexts: Systematic review and meta-analysis. Obes. Rev. 2014, 15, 920–932. [Google Scholar] [CrossRef]
- Lean, M.E.J.; Leslie, W.S.; Barnes, A.C.; Brosnahan, N.; Thom, G.; McCombie, L.; Peters, C.; Zhyzhneuskaya, S.; Al-Mrabeh, A.; Hollingsworth, K.G.; et al. Durability of a primary care-led weight-management intervention for remission of type 2 diabetes: 2-year results of the DiRECT open-label, cluster-randomised trial. Lancet Diabetes Endocrinol. 2019, 7, 344–355. [Google Scholar] [CrossRef]
- Yumuk, V.; Tsigos, C.; Fried, M.P.; Schindler, K.; Busetto, L.; Micic, D.; Toplak, H. European Guidelines for Obesity Management in Adults. Obes. Facts 2015, 8, 402–424. [Google Scholar] [CrossRef]
- Arciero, P.J.; Edmonds, R.; He, F.; Ward, E.G.; Gumpricht, E.; Mohr, A.; Ormsbee, M.J.; Astrup, A. Protein-Pacing Caloric-Restriction Enhances Body Composition Similarly in Obese Men and Women during Weight Loss and Sustains Efficacy during Long-Term Weight Maintenance. Nutrients 2016, 8, 476. [Google Scholar] [CrossRef]
- Astrup, A.; Raben, A.; Geiker, N.R.W. The role of higher protein diets in weight control and obesity-related comorbidities. Int. J. Obes. 2015, 39, 721–726. [Google Scholar] [CrossRef]
- Larsen, T.; Dalskov, S.-M.; Van-Baak, M.; Jebb, S.; Susan, A.; Papadaki, A.; Pfeiffer, A.F.; Martinez, J.; José, A.; Handjieva-Darlenska, T.; et al. Diets with High or Low Protein Content and Glycemic Index for Weight-Loss Maintenance. N. Engl. J. Med. 2010, 363, 2102–2113. [Google Scholar] [CrossRef]
- Rosenbaum, N. Ernährungssoftware im Vergleich. Ernährungs Umsch. 2006, 53, 150–151. [Google Scholar]
- Frey, I.; Berg, A.; Grathwohl, D.; Keul, J. Freiburg Questionnaire of physical activity--development, evaluation and application. Soz. Praventivmed. 1999, 44, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Koohkan, S.; McCarthy, D.; Berg, A. The effect of a soy-yoghurt-honey product on excess weight and related Page health risk factors—A review. J. Nutr. Health Food Sci. 2017, 5, 1–10. [Google Scholar]
- Oliveira, C.L.P.; Boulé, N.G.; Sharma, A.M.; Elliott, S.A.; Siervo, M.; Ghosh, S.; Berg, A.; Prado, C.M. Examining the effects of a high-protein total diet replacement on energy metabolism, metabolic blood markers, and appetite sensations in healthy adults: Protocol for two complementary, randomized, controlled, crossover trials. Trials 2019, 20, 787–810. [Google Scholar] [CrossRef] [PubMed]
- Bauer, J.; Biolo, G.; Cederholm, T.; Cesari, M.; Cruz-Jentoft, A.J.; Morley, J.E.; Phillips, S.; Sieber, C.; Stehle, P.; Teta, D.; et al. Evidence-Based Recommendations for Optimal Dietary Protein Intake in Older People: A Position Paper From the PROT-AGE Study Group. J. Am. Med. Dir. Assoc. 2013, 14, 542–559. [Google Scholar] [CrossRef]
- Phillips, S.M.; Chevalier, S.; Leidy, H.J. Protein “requirements” beyond the RDA: Implications for optimizing health. Appl. Physiol. Nutr. Metab. 2016, 41, 565–572. [Google Scholar] [CrossRef]
- Miller, G.D. Improved nutrient intake in older obese adults undergoing a structured diet and exercise intentional weight loss program. J. Nutr. Health Aging 2010, 14, 461–466. [Google Scholar] [CrossRef]
- Ashley, J.M.; Herzog, H.; Clodfelter, S.; Bovee, V.; Schrage, J.; Pritsos, C.A. Nutrient adequacy during weight loss interventions: A randomized study in women comparing the dietary intake in a meal replacement group with a traditional food group. Nutr. J. 2007, 6, 12. [Google Scholar] [CrossRef]
- Bopp, M.J.; Houston, D.K.; Lenchik, L.; Easter, L.; Kritchevsky, S.B.; Nicklas, B.J. Lean Mass Loss Is Associated with Low Protein Intake during Dietary-Induced Weight Loss in Postmenopausal Women. J. Am. Diet. Assoc. 2008, 108, 1216–1220. [Google Scholar] [CrossRef]
- Campos-Nonato, I.; Hernandez, L.; Barquera, S. Effect of a High-Protein Diet versus Standard-Protein Diet on Weight Loss and Biomarkers of Metabolic Syndrome: A Randomized Clinical Trial. Obes. Facts 2017, 10, 238–251. [Google Scholar] [CrossRef]
- Gregorio, L.; Brindisi, J.; Kleppinger, A.; Sullivan, R.; Mangano, K.M.; Bihuniak, J.D.; Kenny, A.M.; Kerstetter, J.E.; Insogn, K.L. Adequate dietary protein is associated with better physical performance among post-menopausal women 60–90 years. J. Nutr. Heal. Aging 2014, 18, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Merra, G.; Miranda, R.; Barrucco, S.; Gualtieri, P.; Mazza, M.; Moriconi, E.; Marchetti, M.; Chang, T.F.M.; De Lorenzo, A.; Di Renzo, L. Very-low-calorie ketogenic diet with aminoacid supplement versus very low restricted-calorie diet for preserving muscle mass during weight loss: A pilot double-blind study. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 2613–2621. [Google Scholar] [PubMed]
- Mettler, S.; Mitchell, N.; Tipton, K.D. Increased Protein Intake Reduces Lean Body Mass Loss during Weight Loss in Athletes. Med. Sci. Sports Exerc. 2010, 42, 326–337. [Google Scholar] [CrossRef] [PubMed]
- Brehm, B.J.; D’Alessio, D.A. Benefits of high-protein weight loss diets: Enough evidence for practice? Curr. Opin. Endocrinol. Diabetes Obes. 2008, 15, 416–421. [Google Scholar] [CrossRef] [PubMed]
- Huecker, M.R.; Sarav, M.; Pearlman, M.; Laster, J. Protein Supplementation in Sport: Source, Timing, and Intended Benefits. Curr. Nutr. Rep. 2019, 8, 382–396. [Google Scholar] [CrossRef]
- Rizzoli, R.; Stevenson, J.C.; Bauer, J.M.; Van Loon, L.J.; Walrand, S.; Kanis, J.A.; Cooper, C.; Brandi, M.-L.; Diez-Perez, A.; Reginster, J.-Y. The role of dietary protein and vitamin D in maintaining musculoskeletal health in postmenopausal women: A consensus statement from the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO). Maturitas 2014, 79, 122–132. [Google Scholar] [CrossRef]
- Seimon, R.V.; Wild-Taylor, A.L.; Keating, S.E.; McClintock, S.; Harper, C.; Gibson, A.A.; Johnson, N.A.; Fernando, H.A.; Markovic, T.P.; Franklin, J.; et al. Effect of Weight Loss via Severe vs Moderate Energy Restriction on Lean Mass and Body Composition Among Postmenopausal Women with Obesity: The TEMPO Diet Randomized Clinical Trial. JAMA Netw. Open 2019, 2, e1913733. [Google Scholar] [CrossRef]
- McVay, M.; Jeffreys, A.S.; King, H.A.; Olsen, M.K.; Voils, C.I.; Yancy, W.S. The relationship between pretreatment dietary composition and weight loss during a randomised trial of different diet approaches. J. Hum. Nutr. Diet. 2013, 28, 16–23. [Google Scholar] [CrossRef]
- Patel, K.V.; Bahnson, J.; Gaussoin, S.; Johnson, K.C.; Pi-Sunyer, X.; White, U.; Olson, K.L.; Bertoni, A.G.; Kitzman, D.W.; Berry, J.D.; et al. Abstract 15067: Association of Baseline and Longitudinal Changes in Body Composition Measures With Risk of Heart Failure and Myocardial Infarction in Type 2 Diabetes Mellitus—Findings From the Look Ahead Trial. Circulation 2020, 142, 2420–2430. [Google Scholar] [CrossRef]
- Hartmann, C.; Keller, C.; Siegrist, M. Compensatory beliefs, nutrition knowledge and eating styles of users and non-users of meal replacement products. Appetite 2016, 105, 775–781. [Google Scholar] [CrossRef]
- Larson-Meyer, D.E.; Willis, K.S.; Willis, L.M.; Austin, K.J.; Hart, A.M.; Breton, A.B.; Alexander, B.M. Effect of honey versus sucrose on appetite, appetite-regulating hormones, and postmeal thermogenesis. J. Am. Coll. Nutr. 2010, 29, 482–493. [Google Scholar] [CrossRef] [PubMed]
- König, D.; Muser, K.; Berg, A.; Deibert, P. Fuel selection and appetite-regulating hormones after intake of a soy protein-based meal replacement. Nutrients 2012, 28, 35–39. [Google Scholar] [CrossRef] [PubMed]
- Berg, A.; Schaffner, D.; Pohlmann, Y.; Baumstark, M.W.; Deibert, P.; König, D.; Gollhofer, A. A soy-based supplement alters energy metabolism but not the exercise-induced stress response. Exerc. Immunol. Rev. 2012, 18, 128–141. [Google Scholar] [PubMed]
- Erejuwa, O.O.; Sulaiman, S.A.; Ab Wahab, M.S. Oligosaccharides Might Contribute to the Antidiabetic Effect of Honey: A Review of the Literature. Molecules 2011, 17, 248–266. [Google Scholar] [CrossRef]
- Oliveira, C.L.P.; Boulé, N.G.; Berg, A.; Sharma, A.M.; Elliott, S.A.; Siervo, M.; Ghosh, S.; Prado, C.M. Consumption of a High-Protein Meal Replacement Leads to Higher Fat Oxidation, Suppression of Hunger, and Improved Metabolic Profile after an Exercise Session. Nutrients 2021, 13, 155. [Google Scholar] [CrossRef]
- Malinowska, A.M.; Mlodzik-Czyzewska, M.A.; Chmurzynska, A. Dietary patterns associated with obesity and overweight: When should misreporters be included in analysis? Nutrients 2020, 70, 110605. [Google Scholar] [CrossRef]
- Westerterp, K.R.; Goris, A.H. Validity of the assessment of dietary intake: Problems of misreporting. Curr. Opin. Clin. Nutr. Metab. Care 2002, 5, 489–493. [Google Scholar] [CrossRef]
| Anthropometrical and Clinical Parameters | INT-Group (n = 82) [Men (n = 32) Women (n = 50)] | CON-Group (n = 37) [Men (n = 13) Women (n = 24)] |
|---|---|---|
| Sex (%) | 39.0 | 35.1 |
| Age (years) | 52 ± 8 | 52 ± 8 |
| Weight (kg) | 92.8 ± 10.1 | 92.2 ± 10.4 |
| BMI (kg/m2) | 31.6 ± 2.4 | 30.8 ± 2.4 |
| WC (cm) | 107 ± 7 | 106 ± 8 |
| WHR | 0.97 ± 0.06 | 0.96 ± 0.06 |
| FM (kg) | 36.0 ± 6.0 | 36.3 ± 6.1 |
| FFM (kg) | 57.0 ± 5.5 | 55.7 ± 6.6 |
| HbA1c (%) | 5.49 ± 0.33 | 5.45 ± 0.29 |
| FBG (mg/dl) | 92 ± 10 | 92 ± 11 |
| SBP (mmHg) | 135 ± 15 | 134 ± 17 |
| DBP (mmHg) | 92 ± 11 | 91 ± 8 |
| Total cholesterol (mg/dl) | 213 ± 36 | 218 ± 51 |
| HDL-C (mg/dl) | 53 ± 14 | 55 ± 10 |
| LDL-C (mg/dl) | 137 ± 31 | 136 ± 43 |
| Triglycerides (mg/dl) | 140 ± 73 | 164 ± 73 |
| Complete Case Analysis | Baseline | 12 Weeks | p | 52 Weeks | p | |||
|---|---|---|---|---|---|---|---|---|
| INT | CON | INT | CON | (INT vs. CON) | INT | CON | (INT vs. CON) | |
| Energy (kcal) | 2129 ± 580 | 2208 ± 587 | −372 [−556; −174] *** | −288 [−625; 55] * | 0.112 | −217 [−440; 8.80] * | −493 [−809; −154] *** | 0.389 |
| Protein (g) | 91 ± 21 | 92 ± 24 | 9.10 [1.05; 18.6] * | 1.40 [−13.8; 14.9] | 0.680 | 5.72 [−2.35; 13.2] | −16.4 [−26.0; −2.31] * | 0.075 |
| Protein (proportion of daily energy intake (%)) | 18.3 ± 5.5 | 17.4 ± 3.2 | 6.37 [4.69; 8.04] *** | 2.48 [0.73; 4.23] ** | <0.001 | 2.86 [1.40; 4.32] *** | 1.45 [0.04; 2.86] | 0.052 |
| Fat (g) | 90.1 ± 30.2 | 94.2 ± 29.2 | −20.7 [−30.9; −10.1] *** | −9.72 [−28.3; 8.64] | 0.051 | −13.5 [−23.6; −3.02] ** | −24.3 [−38.7; −6.91] ** | 0.509 |
| Fat (proportion of daily energy intake (%)) | 40.0 ± 6.5 | 39.4 ± 6.1 | −3.08 [−4.82; −1.34] *** | 1.08 [−1.23; 3.39] | 0.006 | −2.11 [−3.87; −0.36] * | −0.65 [−3.21; 1.91] | 0.424 |
| Carbohydrates (g) | 198 ± 71 | 215 ± 69 | −52.0 [−67.2; −36.8] *** | −45.2 [−67.6; −22.7] *** | 0.046 | −28.2 [−46.3; −10.2] ** | −54.6 [−78.7; −30.5] *** | 0.416 |
| Carbohydrates (proportion of daily energy intake (%)) | 38.9 ± 7.5 | 40.4 ± 7.4 | −3.22 [−4.82; −1.63] *** | −3.27 [−5.52; −1.02] ** | 0.008 | −1.24 [−3.06; 0.57] | −1.27 [−4.16; 1.63] | 0.362 |
| Glucose (g) | 10.6 ± 6.6 | 12.8 ± 8.3 | −3.23 [−4.54; −1.93] *** | −2.34 [−5.37; 0.68] | <0.001 | −1.29 [−3.00; 0.42] * | −3.10 [−6.57; 0.37] * | 0.981 |
| Fructose (g) | 13.0 ± 8.3 | 17.1 ± 12.8 | −3.46 [−5.21; −1.71] *** | −3.51 [−8.15; 1.12] | <0.001 | −0.92 [−3.21; 1.38] | −4.41 [−9.54; 0.72] | 0.969 |
| Sucrose (g) | 36.2 ± 21.2 | 39.8 ± 21.0 | −13.7 [−18.8; −8.60] *** | −11.9 [−19.5; −4.25] ** | 0.144 | −9.58 [−15.2; −4.01] ** | −15.2 [−22.5; −7.87] *** | 0.814 |
| Alcohol (g) | 16.3 ± 17.1 | 15.3 ± 18.7 | −2.38 [−6.74; 2.32] | −1.21 [−9.89; 7.11] | 0.614 | 0.07 [−5.92; 6.02] | −0.82 [−8.91; 7.94] | 0.663 |
| Alcohol (proportion of daily energy intake (%)) | 4.62 ± 6.03 | 4.56 ± 5.75 | −0.24 [−1.13; 0.65] | −0.11 [−1.60; 1.38] | 0.884 | 0.29 [−0.98; 1.56] | 0.28 [−1.24; 1.79] | 0.861 |
| Dietary fibre (g) | 17.4 ± 7.4 | 20.2 ± 7.2 | −2.37 [−3.69; −1.04] *** | −0.68 [−2.84; 1.47] | 0.008 | 0.24 [−1.62; 2.10] | −2.60 [−4.77; −0.43] * | 0.171 |
| Weight (kg) | 92.8 ± 10.1 | 92.2 ± 10.4 | −7.23 [−8.37; −6.13] *** | −4.84 [−6.35; −3.22] *** | 0.008 | −5.27 [−6.82; −3.81] *** | −4.45 [−7.01; −1.91] * | 0.469 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Röhling, M.; Stensitzky, A.; Oliveira, C.L.P.; Beck, A.; Braumann, K.M.; Halle, M.; Führer-Sakel, D.; Kempf, K.; McCarthy, D.; Predel, H.G.; et al. Effects of a Protein-Rich, Low-Glycaemic Meal Replacement on Changes in Dietary Intake and Body Weight Following a Weight-Management Intervention—The ACOORH Trial. Nutrients 2021, 13, 376. https://doi.org/10.3390/nu13020376
Röhling M, Stensitzky A, Oliveira CLP, Beck A, Braumann KM, Halle M, Führer-Sakel D, Kempf K, McCarthy D, Predel HG, et al. Effects of a Protein-Rich, Low-Glycaemic Meal Replacement on Changes in Dietary Intake and Body Weight Following a Weight-Management Intervention—The ACOORH Trial. Nutrients. 2021; 13(2):376. https://doi.org/10.3390/nu13020376
Chicago/Turabian StyleRöhling, Martin, Andrea Stensitzky, Camila L. P. Oliveira, Andrea Beck, Klaus Michael Braumann, Martin Halle, Dagmar Führer-Sakel, Kerstin Kempf, David McCarthy, Hans Georg Predel, and et al. 2021. "Effects of a Protein-Rich, Low-Glycaemic Meal Replacement on Changes in Dietary Intake and Body Weight Following a Weight-Management Intervention—The ACOORH Trial" Nutrients 13, no. 2: 376. https://doi.org/10.3390/nu13020376
APA StyleRöhling, M., Stensitzky, A., Oliveira, C. L. P., Beck, A., Braumann, K. M., Halle, M., Führer-Sakel, D., Kempf, K., McCarthy, D., Predel, H. G., Schenkenberger, I., Toplak, H., & Berg, A. (2021). Effects of a Protein-Rich, Low-Glycaemic Meal Replacement on Changes in Dietary Intake and Body Weight Following a Weight-Management Intervention—The ACOORH Trial. Nutrients, 13(2), 376. https://doi.org/10.3390/nu13020376







