1. Introduction
Modern-day healthcare is evolving at a rapid rate. Technological advances are facilitating more accessible, safe, effective, efficient and sustainable health care delivery [
1,
2]; and importantly, more patient-centred care [
3]. A key characteristic of patient-centred care is patient engagement or participation, which is defined as active involvement in one’s own health care, for example, through sharing/exchanging health information or engaging in intellectual and/or physical health care activities [
4]. Patient engagement is a core aspect of safe and quality health care endorsed by the World Health Organization’s Patients for Patient Safety movement [
5]. It is associated with improved clinical and functional outcomes, as well as increased patient safety and satisfaction with care [
6,
7]. Health information technology (HIT) has the ability to facilitate patient engagement by providing the resources and tools required for patients to play an active role in their health care [
3,
8]. While there has been wide adoption of HIT by individuals in the community (e.g., applications, programs and web-based tools for monitoring measurable health parameters or for implementing lifestyle changes), HIT adoption in hospital settings is lagging [
9]. As more research on HIT use in clinical settings emerges, showing promise for improved patient and hospital outcomes, it is imperative that healthcare professionals and organisations not only embrace HIT but contribute to its development, implementation and evaluation.
Nutrition and dietetics is a field that may benefit from HIT [
10]. Not only is nutrition an area in which patients feel comfortable participating [
11], HIT may provide solutions to long-standing problems, such as hospital malnutrition. In a landmark 1974 paper, Butterworth proposed that iatrogenic malnutrition was one of the most serious nutritional issues of the time, despite “the technological advances that have been made” [
12]. Almost five decades later, and after an explosion in HIT, malnutrition remains a significant clinical problem. It affects 20–50% of hospital patients worldwide [
13,
14] and results in poor patient outcomes, including increased risks of mortality [
15], infections [
16], pressure injury [
17], reduced mobility and falls [
18,
19]. It also contributes to increased lengths of hospital stays, readmissions and hospital costs [
15,
20], placing a large burden on healthcare systems. Malnutrition can be prevented or corrected with adequate dietary intake; however, due to a complex mix of factors, achieving optimal nutrition intake among hospitalised patients is difficult [
21]. One strategy showing promise is patient participation in their nutrition care, with preliminary research showing this is an effective strategy for improving the dietary intakes of hospitalised patients [
22,
23].
Our team previously developed a paper-based intervention that allowed hospitalised patients to participate in their nutrition care by self-monitoring their dietary intake and engaging in guided nutritional goal setting [
23]. Others have found the enactment of self-regulatory behaviours, such as setting goals and monitoring food intake, were the best predictors of participants’ nutrition status [
24]. Using an integrated knowledge translation (IKT) approach [
25], guided by the Knowledge to Action cycle [
26], our team adapted this intervention for delivery via HIT that was compatible with existing hospital systems and models of care. This intervention has the potential to improve the dietary intakes of hospitalised patients at risk of malnutrition by engaging them in their care. While previous studies have shown increased energy and protein intakes among patients participating in food intake monitoring and guided nutritional goal setting (using paper resources), none have evaluated this kind of intervention using technology as the medium for engagement.
2. Materials and Methods
2.1. Study Overview and Aims
The aim of this study was to evaluate the acceptability and feasibility of the HIT intervention NUTRI-TEC, which engages hospital patients in their nutrition care in order to improve dietary intakes. Acceptability was determined with patient satisfaction and engagement surveys, which were assessed against criteria on acceptability. Feasibility (for a larger trial) was assessed against criteria on intervention delivery and fidelity, as well as patient recruitment and retention. The accuracy of patient-recorded (vs. researcher-recorded) dietary intakes, along with an indication of the intervention’s effects on patients’ intakes, were also assessed to give further insights into the intervention. This study received ethical approval from the relevant hospital and university human research ethics committees.
Feasibility criteria included:
- (a)
≥80% of patients receive the intervention within 24 h of enrolment (delivery);
- (b)
≥80% of patients receive all intervention components as intended (fidelity);
- (c)
≥50% of patients approached for recruitment agree to participate (recruitment);
- (d)
≥75% of patients remain in the study long enough for adequate outcome data collection, i.e., three days of oral intake data (retention).
We determined that for feasibility, the intervention delivery and fidelity would have to be ≥80% to allow for intention-to-treat analyses in a larger trial. For recruitment and retention, 50% and 75% were selected, respectively, as we recognised that not all patients may want to participate in research while in hospital and that the intervention (being technology-based) may not appeal to some patients. The values selected are consistent with those developed for a similar randomised controlled feasibility study of a complex intervention to reduce frailty and falls among haemodialysis patients [
27].
Acceptability was explored quantitatively via patient satisfaction and engagement surveys. The intervention was considered acceptable if ≥80% of patients answered “satisfied” or “very satisfied” to the patient satisfaction survey questions.
Finally, patients’ daily total energy and protein intakes were monitored to give an indication of the effect of the intervention over time and to assess the accuracy of patient-recorded dietary intakes (vs. those recorded by RAs).
2.2. Setting
This study was conducted in four wards (orthopaedic, renal, respiratory and vascular/medical) at a large tertiary university hospital in southeast Queensland, Australia. The hospital used an electronic foodservice system (EFS; Delegate Food Service system, version 12.10, Delegate Technology GmbH; Vienna, Austria), which was accessed by each patient through their bedside computer. Standard practice at the hospital involved patients using the EFS to order their meals. The EFS had the functionality for patients to view nutrition information, including energy in kilojoules (kJ) and protein in grams (g), for each item on the hospital menu. Patients could choose whether to “show” or “hide” this information when ordering meals.
2.3. Sample and Recruitment
Adult patients at nutritional risk, who met all inclusion and no exclusion criteria, were eligible to participate. Inclusion criteria: (a) adults able to provide informed consent (≥18 years old, cognitively intact), (b) able to communicate in English (verbally and in writing), (c) at risk of malnutrition (aged ≥65 years and/or score of ≥2 on the Malnutrition Screening Tool (MST)), and (d) expected length of stay of ≥4 days from recruitment. Exclusion criteria: (a) previous participation in the study, (b) palliative or dying patients, (c) patients not receiving nutrition orally, and (d) patients with a history of an eating disorder.
A research assistant (RA) conducted daily participant eligibility screening on each study ward for 12 weeks. Eligible patients were given a participant information form and verbal explanation of the study and those willing to participate provided written informed consent and were enrolled. Patients remained in the study for 14 days or until death or hospital discharge, whichever occurred first. As there is no specific sample size calculation for feasibility studies [
28], a literature-informed, pragmatic approach was used to determine sample size, based on work from Hertzog (2008) and on a previous pilot study of a similar intervention [
23]. From this, it was expected that around 60–80 patients would be recruited with the available resources.
2.4. Intervention
Each patient received standard care plus the intervention, which was delivered by an RA (who was an accredited dietitian) within 24 h of recruitment. In addition to standard care, the intervention included an initial session consisting of:
- (a)
A brief nutrition history (Subjective Global Assessment (SGA)) conducted by an RA;
- (b)
A brief lesson on the importance of meeting nutritional requirements in hospital;
- (c)
Training on how to use EFS to order meals, enter food intake after each meal and view and monitor nutritional goals.
After this initial session, patients were asked to record their food intake after each meal via the EFS. This involved opening the EFS portal from their bedside computer, selecting the day and meal (e.g., Monday breakfast) and selecting the amounts consumed for each meal item, presented in quartiles (see
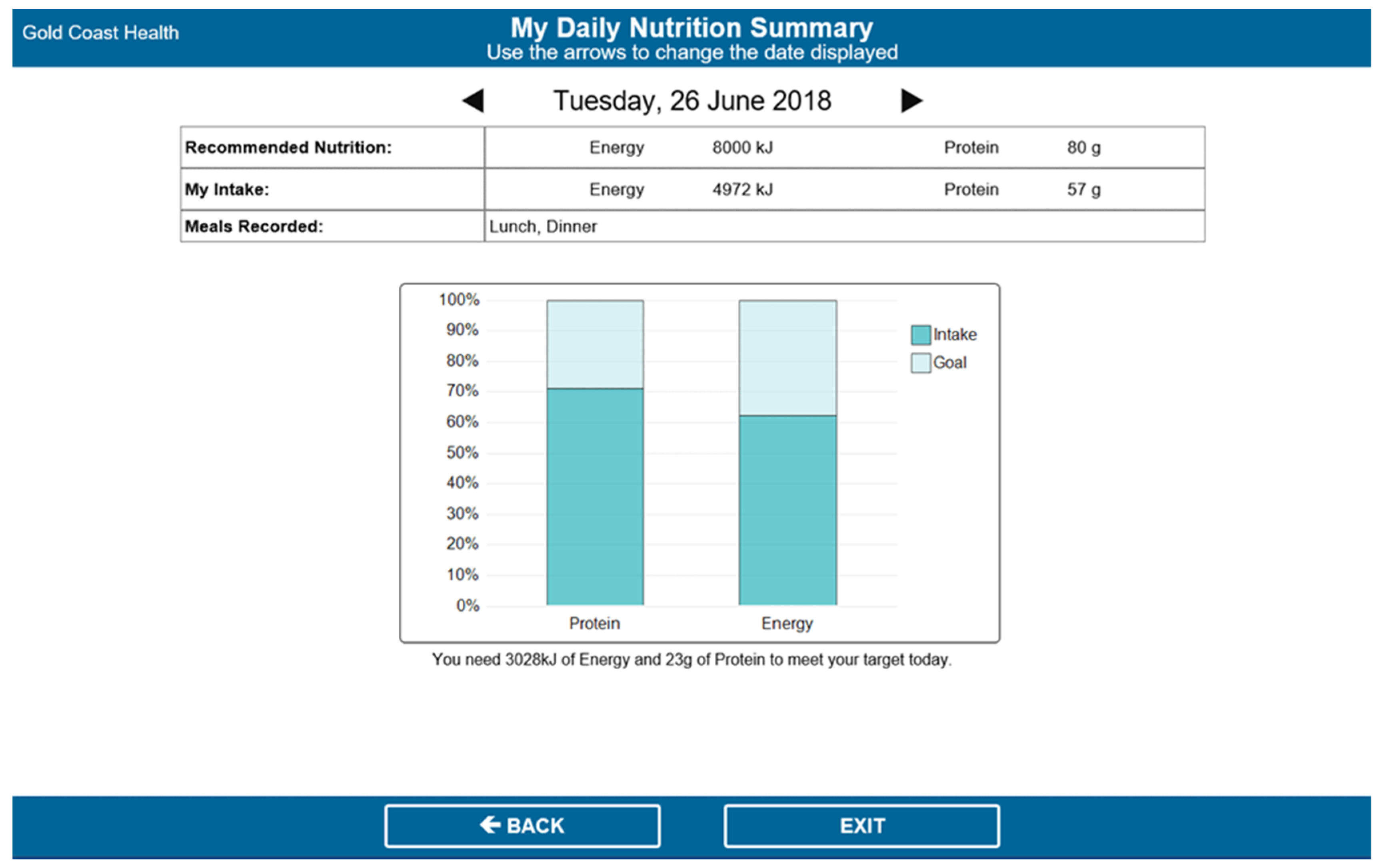
Figure 1). The EFS would only show the meal items ordered by and delivered to that patient on the intake tracking page. The EFS then calculated the patients’ energy (kJ) and protein (g) intakes for each meal and each day, and displayed this information on the nutrition goals page, alongside their individually estimated energy and protein requirements (see
Figure 2).
The intervention also included review sessions conducted by RAs (daily for the first week after enrolment and every second day for the second week after enrolment) to engage patients in guided nutritional goal setting and monitoring via the EFS. The RAs discussed patients’ goals graphs with them and showed them how to use this information to inform their menu choices for the next day. If patients met <75% of their estimated energy/protein requirements on a given day, RAs reviewed possible strategies that patients could enact to help meet their requirements. A detailed description of intervention components and development can be found in a separate publication [
29].
Clinically relevant data that were collected by the RAs (SGA score, patient preferences) were made available to ward dietitians, who prescribed nutrition support if required (e.g., oral nutrition supplements, high-energy/high-protein extras).
2.5. Data Collection
All data were collected by the same trained RAs and included the following:
Feasibility data pertaining to recruitment (number of patients recruited from those eligible), retention (number of patients remaining in the study until the end point) and intervention delivery (intervention components delivered and timing) were recorded by the RAs in standardised logs at patient recruitment, the study end point and upon intervention delivery, respectively.
Acceptability was assessed via a validated patient participation scale [
7] and patient satisfaction survey [
30], which was administered to patients by the RAs prior to discharge or on study day four, whichever came first.
Patient demographics (age, gender, admission details, diagnosis, co-morbidities) and nutrition data (weight, height, body mass index (BMI), MST and SGA scores, prescribed diet (including supplements), and dietitian input) were collected from patients’ electronic medical records and bedside charts after their recruitment. Patients’ individually estimated energy (EER) and protein requirements (EPR) were calculated by the RAs using local clinical guidelines [
31] and entered into the EFS. Data on adverse events, including pressure injuries, falls and infections were collected from electronic medical records by the RAs upon participants’ hospital discharge.
Patients’ nutrition intakes were collected via visual observation of mealtimes and recorded until the study endpoint (i.e., 14 days, hospital discharge or death, whichever occurred first). Visual plate waste was observed by the RAs at each meal and the intake of each dietary item was recorded in quartiles on a standardised food chart, which is a method that has been shown to correlate with weighed food records [
32]. Patients’ intakes were entered into a Foodworks database (version 9, Xyris Software; Brisbane, Australia) containing the nutritional information of dietary items provided by the hospital to calculate their daily energy and protein intakes. Intakes were presented as a proportion of EER and EPR for each patient. The amounts of energy and protein provided (i.e., food ordered by the patients) each day were exported from the EFS. If patients consumed <50% of their meal, the reasons for eating poorly were documented by observation (e.g., meal not delivered, patient not in bed) and/or by asking the patient (e.g., poor appetite, nutrition impacting symptoms, dislike of hospital food).
2.6. Data Analysis
Demographics, feasibility, nutrition intakes and survey data were analysed descriptively using SPSS Statistics for Windows version 23.0 (IBM Corp. 2012, Armonk, NY, USA). Continuous data were tested for normality using the Shapiro–Wilk test and presented accordingly (mean ± standard deviation for normally distributed data; median (interquartile range (IQR)) for non-normally distributed data). One-way repeated measures ANOVA tests were used to compare the proportion of EER and EPR met by patients each day over time. Patient-recorded intakes and researcher-observed intakes were compared using Pearson’s correlation to determine how accurately patients recorded their food intake. Significance was set to p ≤ 0.05.
4. Discussion
This study evaluated the acceptability and feasibility of a HIT intervention that was designed to engage hospitalised patients in their nutrition care in order to improve their dietary intakes. This work provides important insights into the use of HIT for patient engagement in acute hospital settings where patient engagement in care is encouraged (and even mandated in some countries) and where HIT has the potential to revolutionise care provision and the patient experience.
This study found that the NUTRI-TEC intervention was acceptable to patients, with most (86–94%) reporting satisfaction with its various aspects. This is consistent with a qualitative study that explored patient perceptions of NUTRI-TEC among a subset of participants from the current study [
33]. In the qualitative study, patients reported high satisfaction with NUTRI-TEC as they found it useful, valuable and easy to use, and they enjoyed gaining new knowledge and awareness regarding nutrition and participating in their nutrition care [
33]. Patient satisfaction with health care interventions is necessary, but alone, it is insufficient for success; this requires meaningful patient engagement. Patients in this study had high levels of engagement with NUTRI-TEC. Over 90% of patients reported being aware of their nutrition plan, understanding their menu options and feeling well enough to discuss nutrition with the RAs; furthermore, 80% participated in decisions about their nutrition to the extent they wanted to. This is promising, as an integrative review on patient participation in acute medical wards found that engaging hospital patients in care is difficult, and patients’ expectations and desires for participation were often not met, with some participating more or less than they wished [
34]. In our study, meaningful engagement in nutrition care (via NUTRI-TEC) involved patient education, i.e., learning about their nutrition needs and how to meet these needs in hospital; self-assessment and feedback, i.e., monitoring their nutrition intakes and requirements; behaviour change, i.e., being empowered with knowledge and control over their nutrition care, which allowed patients to make informed dietary choices. These activities align with a conceptual analysis of patient participation in care [
4]. First, meaningful information exchange between patients and dietitians (in this case, RAs) occurred via data they each entered in the EFS, which both parties used to inform nutrition care. Dietitians/RAs surrendered some power/control, allowing patients to track their dietary intakes and make decisions about their nutrition. Using the EFS’s goals page in daily review sessions to guide discussions and monitor progress allowed for mutual involvement in health care activities and allowed patients and RAs to establish and build a relationship [
4]. These concepts are of high importance for any patient engagement intervention and should be considered in both their development and delivery in practice.
The delivery of NUTRI-TEC was feasible using trained RAs, who were also dietitians. All patients received the intervention; most (82%) within 24 h and all within 29 h of recruitment. While nine patients received the intervention a few hours late due to an overlap with RA screening and recruiting, this problem would not occur in a larger trial as we would employ separate RAs for screening/recruiting, intervention delivery and data collection. Intervention fidelity was high, with >95% of patients receiving at least one component, and importantly, the intervention took only 15 min to deliver on average, which is critical for its feasibility in practice. NUTRI-TEC was designed to be used as part of routine clinical dietetic practice, which means these results are encouraging; however, as Rogers’ diffusion of innovations theory suggests, for uptake, innovations must demonstrate a relative advantage by being better than the idea/method they supersede [
35]. NUTRI-TEC has a relative advantage over traditional methods of nutrition care, such as dietary intake monitoring, which is a core part of the nutrition care process. Nutrition intake data are vital for nutrition assessment and monitoring; however, commonly used methods of patient recall and nurse-recorded food charts are known to have low accuracy and reliability and are not always able to be achieved. One study found that only 64% of patients were able to complete a diet recall interview, which was time and resource intensive [
36]. Another found that nurse-recorded food charts had low accuracy and completion, with 93% of patient intakes being unable to be determined due to missing data [
37]. NUTRI-TEC allows patients (or their family members) to record food intakes, making it easier for dietitians to access dietary data to inform nutrition care, whilst engaging patients in their care. In early usability work, nurses at the study site indicated that they would prefer to enter patients’ intakes using NUTRI-TEC than a paper food chart if they were asked to by dietitians [
38], suggesting that the intervention may also have a relative advantage for nurses. Furthermore, this study found that energy and protein intakes recorded by patients correlated strongly with those recorded by researchers, indicating that patients can accurately document their dietary intakes in the hospital using technology.
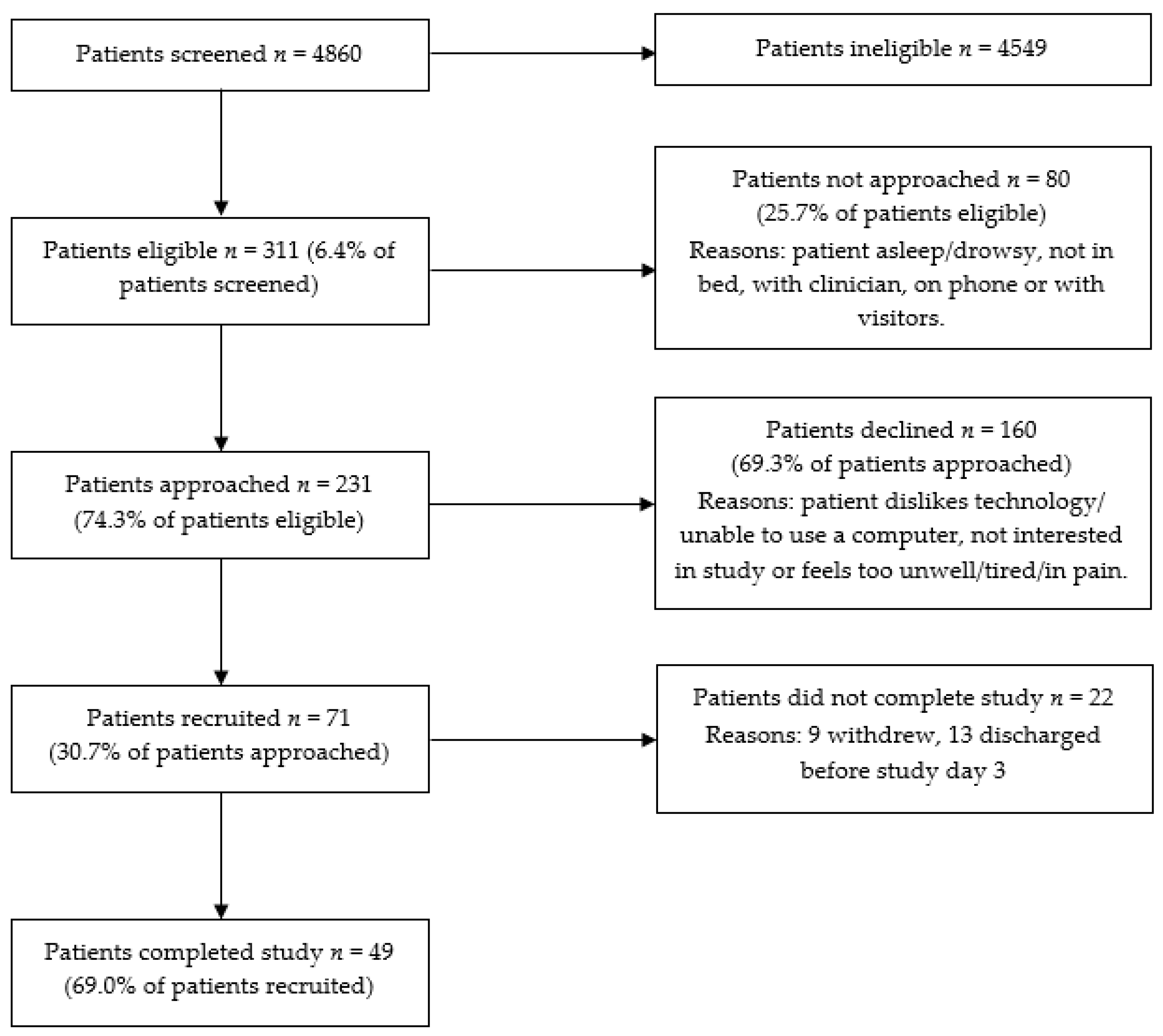
Despite promising findings on intervention delivery and fidelity, recruitment was challenging; only a third of eligible patients agreed to participate in the study. While this rate is suboptimal, it is not entirely surprising, given the intervention involved technology and participants were acutely unwell in hospital. Research shows that up to 75% of patients decline to participate in HIT studies, with a common reason being that participants perceive low benefits from technology [
39]. Recruiting hospital patients to interventional research is also challenging; one review of fall prevention interventions reported a <50% median recruitment rate among institutionalised older patients [
40]. Physical limitations, such as being tired or sick, are other frequently cited reasons for non-participation in clinical research [
41]. Reasons for declining participation in the current study were consistent with previous work and included disinterest in, dislike of or unfamiliarity with technology, as well as feeling too unwell. However, there is scope for reducing such barriers. First, at the study hospital, some patients would have been unfamiliar with the EFS, as despite hospital policy, only around a third are shown how to use it on admission. This likely reduced some patients’ willingness to participate in a study involving this technology. Given all patients who participated in usability testing found NUTRI-TEC easy to use after a brief familiarisation and said they would use it in future admissions [
11], further work could be done to improve patient orientation regarding the EFS. Second, for patients feeling too unwell to participate in NUTRI-TEC activities, there is potential for family members or hospital staff to participate on their behalf. For example, family members could enter patients’ food intakes and track their progress towards nutrition goals via the EFS, which anecdotally, some did in the current study. Allowing family members to participate on patients’ behalf could improve recruitment to a larger trial. Furthermore, family involvement in acute adult patients’ care has been shown to improve patient outcomes in some instances [
42]. In practice, hospital staff, such as nurses or dietetic assistants, could record patient intakes (as they would with dietitian-requested food charts) via the EFS to begin with, then patients could take over this task as they start to improve/feel well. This may have an added benefit of patients feeling more confident completing intake tracking via the EFS after having watched someone else do it first.
The success of HIT interventions that aim to engage hospitalised patients in their care seems to depend on five key concepts that were identified in a realist review: information sharing, self-assessment and feedback, tailored education, user-centred design, and supporting patients to use HIT [
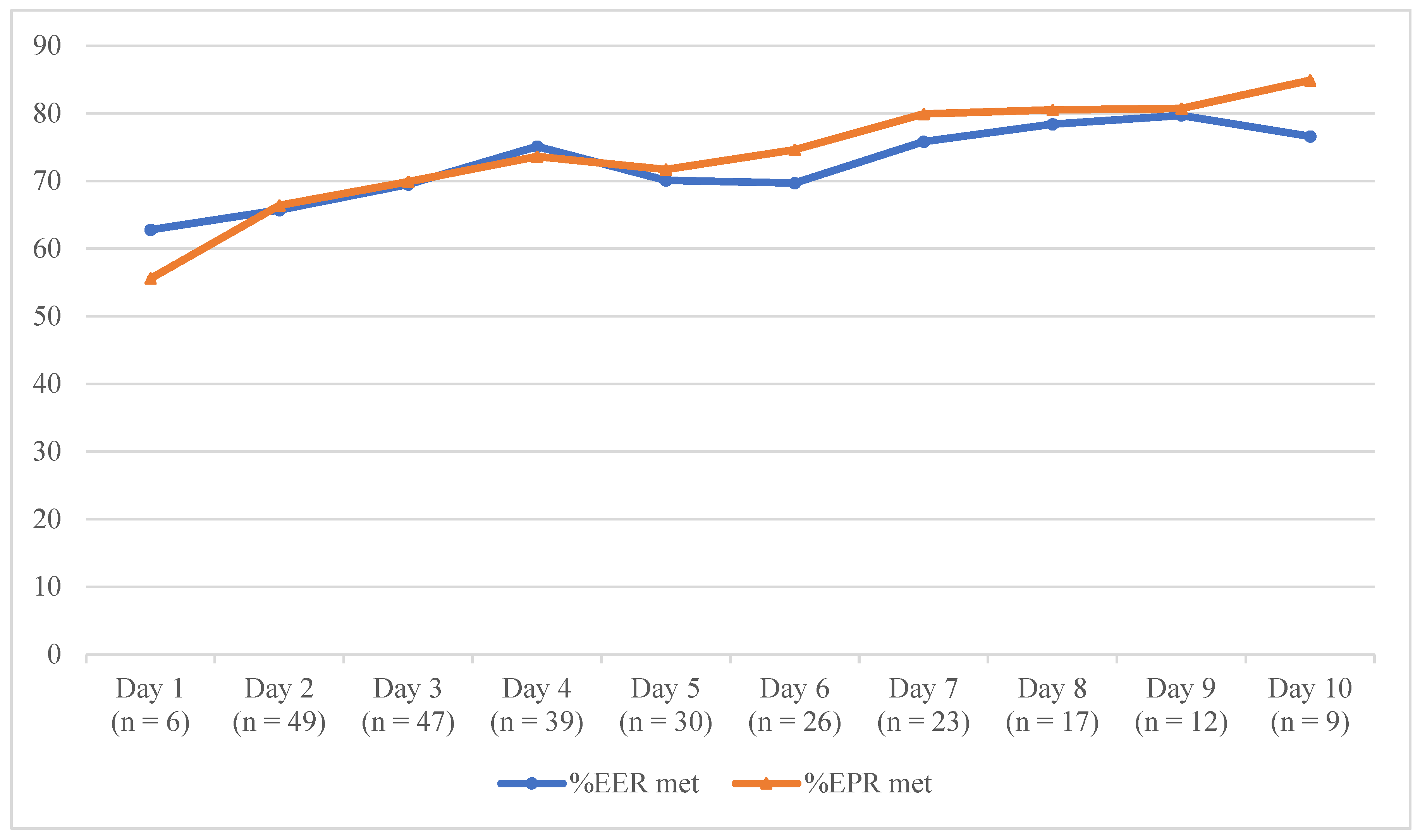
43]. NUTRI-TEC employs all these concepts by engaging patients in dietary intake tracking (self-assessment/feedback) and nutritional goal setting (tailored education, information sharing) with RAs supporting patients in using and engaging with the intervention. NUTRI-TEC was underpinned by relevant theory and research frameworks, informed by literature and local data and iteratively developed and evaluated with end-user input for optimal success. Dietary data analysis in this study found that patients’ energy and protein intakes increased steadily over the first few days after receiving the intervention, which is consistent with our previous work. An earlier, paper version of this intervention was evaluated in a pilot randomised controlled trial (RCT) using paper food charts/goal pages, and found that while control patients’ intakes remained constant, intervention patients’ energy and protein intakes increased significantly following intervention delivery to a degree that was comparable to this study [
23]. That previous pilot RCT found patients’ intakes increased from 65 to 83% of the EER being met and 65 to 91% of the EPR being met over the three days after intervention delivery, while the current study found increases in patients’ intakes from ~66% of the EER and EPR being met to ~75% of the EER and EPR being met over the same period. While this is a feasibility study, these results are promising, especially considering that research suggests patients that meet ≥75% of EER maintain their body weight during hospitalisation [
32]. While patients’ intakes in this study did not increase as much as in the pilot RCT, the findings warrant further investigation.
4.1. Implications and Recommendations
NUTRI-TEC offers an alternative approach to patient education and counselling, dietary intake monitoring and nutrition care planning, and can potentially streamline nutrition care processes for dietitians. This novel HIT program allows dietitians to enter patients’ individually estimated energy and protein requirements, which patients can view at the bedside and dietitians can use as a tool for patient education (e.g., to guide discussions on dietary adequacy or nutrition plans). This approach is likely to result in more meaningful patient engagement, as patients will receive information/education that is individually tailored to them, which is a recognised feature of successful HIT interventions that aim to engage hospital patients in care [
43]. It also allows patients to be more aware/informed of their nutrition care plan, which may improve adherence to nutrition interventions, as patients will better understand why these are needed. Using NUTRI-TEC as a patient education tool caters to different learning styles, with visual (graphical and numerical) and verbal (dietitian explanations) information available. This is important considering hospital patients may have low health literacy, particularly when it comes to accessing, appraising and understanding health information [
44]. Nutrition interventions employing HIT have unbounded potential to improve nutrition care and dietary intakes and increase consumer engagement in hospital patients. However, as this study shows, HIT interventions, such as NUTRI-TEC, may not appeal to all patients, even if they are developed in a patient-centred way. Clinicians must therefore consider patients’ needs/preferences and tailor interventions accordingly to optimise patient uptake and clinical effectiveness. For patients wanting to engage in HIT interventions, clinicians must ensure the technology is interactive and easy to use and be willing to support/assist patients in their use. Lastly, as hospitalisation reflects a very short period in a patient’s life, it may not be the best place to deliver nutrition interventions; in this study, 18% of patients were discharged early and hence did not complete the study. Alternative methods for delivering and assessing the effects of interventions are therefore warranted, i.e., for patients with shorter lengths of stay. This may involve initial intervention delivery in hospital, followed by continued patient engagement after discharge (e.g., via an app or web-based program that patients can access at home that enables information exchange with dietitians) to allow for ongoing education and dietary intake and goal monitoring, as well as longer-term outcomes assessment. This could reduce the burden on hospital clinicians (in intervention delivery) and patients (by participating in interventions at home rather than when they are acutely unwell) and could help to address the notoriously poor follow-up of patients’ nutrition in the community. While the task of designing and implementing an intervention that bridges the hospital-to-home transition may be challenging, HIT could be the answer to improving the continuity of nutrition care in this population.
4.2. Limitations
This study has some limitations. First, it was a single-site feasibility study, which means that results may not be generalisable to other hospitals; however, it does provide important insights into conducting interventional HIT research with patients in hospital settings. Second, the low consent rate resulted in the recruitment target not being met; however, by collecting a range of feasibility data, we were able to identify that recruitment methods need revising for a larger trial (the primary purpose of feasibility studies). The low consent rate may have also resulted in a selection bias; hence, some of the positive findings (e.g., patient satisfaction) may have been overestimated. The 13% drop-out rate could be an indication that the intervention was not acceptable to a proportion of patients and should be considered when interpreting findings. A major limitation of the intervention itself is that only selected hospitals in Australia have electronic foodservice systems and patient bedside computers; both of which are needed for intervention delivery and engagement. However, with the expansion of HIT in hospitals and health care institutions, it is likely that this will be more widely available in the future. There may also be alternative ways to deliver such an intervention, for example, with patients’ personal devices. Lastly, the NUTRI-TEC intervention focuses on improving patients’ dietary intakes in hospital; however, hospitalisation is a relatively short period in a person’s life. Patients would benefit from ongoing nutrition support that provides continuity through care transitions (e.g., hospital to home); hence, further work should be done to achieve this with HIT.