Assessment of Vitamin D Metabolism in Patients with Cushing’s Disease in Response to 150,000 IU Cholecalciferol Treatment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population and Design
2.2. Socio–Demographic and Anthropometric Data Collection
2.3. Laboratory Measurements
2.4. Statistical Analysis
3. Results
3.1. Baseline Laboratory Evaluation
3.2. Laboratory Evaluation after the Intake of Cholecalciferol
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nishioka, H.; Yamada, S. Cushing’s disease. J. Clin. Med. 2019, 8, 1951. [Google Scholar] [CrossRef] [Green Version]
- Mazziotti, G.; Frara, S.; Giustina, A. Pituitary Diseases and Bone. Endocr. Rev. 2018, 39, 440–488. [Google Scholar] [CrossRef] [PubMed]
- Compston, J. Glucocorticoid-induced osteoporosis: An update. Endocrine 2018, 61, 7–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buckley, L.; Guyatt, G.; Fink, H.A.; Cannon, M.; Grossman, J.; Hansen, K.E.; Humphrey, M.B.; Lane, N.E.; Magrey, M.; Miller, M. 2017 American College of Rheumatology Guideline for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res. 2017, 69, 1095–1110. [Google Scholar] [CrossRef]
- Belaya, Z.; Rozhinskaya, L.y.; Grebennikova, T.; Kanis, J.; Pigarova, E.; Rodionova, S.; Toroptsova, N.; Nikitinskaya, O.; Skripnikova, I.; Drapkina, O.; et al. Summary of the Draft Federal Clinical Guidelines for Osteoporosis. Osteoporos. Bone Dis. 2020, 23, 4–21. [Google Scholar] [CrossRef]
- Klein, R.G.; Arnaud, S.B.; Gallagher, J.C.; Deluca, H.F.; Riggs, B.L. Intestinal calcium absorption in exogenous hypercortisolism. J. Clin. Investig. 1977, 60, 253–259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seeman, E.; Kumar, R.; Hunder, G.G.; Scott, M.; Iii, H.H.; Riggs, B.L. Production, degradation, and circulating levels of 1,25-dihydroxyvitamin D in health and in chronic glucocorticoid excess. J. Clin. Investig. 1980, 66, 664–669. [Google Scholar] [CrossRef]
- Lindgren, J.U.; Merchant, C.R.; DeLuca, H.F. Effect of 1,25-dihydroxyvitamin D3 on osteopenia induced by prednisolone in adult rats. Calcif. Tissue Int. 1982, 34, 253–257. [Google Scholar] [CrossRef]
- Chaiamnuay, S.; Chailurkit, L.O.; Narongroeknawin, P.; Asavatanabodee, P.; Laohajaroensombat, S.; Chaiamnuay, P. Current daily glucocorticoid use and serum creatinine levels are associated with lower 25(OH) vitamin D levels in thai patients with systemic lupus erythematosus. J. Clin. Rheumatol. 2013, 19, 121–125. [Google Scholar] [CrossRef]
- Kugai, N.; Koide, Y.; Yamashita, K.; Shimauchi, T.; Nagata, N.; Takatani, O. Impaired mineral metabolism in Cushing’s syndrome: Parathyroid function, vitamin D metabolites and osteopenia. Endocrinol. Jpn. 1986, 33, 345–352. [Google Scholar] [CrossRef] [Green Version]
- Hahn, T.J.; Halstead, L.R.; Baran, D.T. Effects of short term glucocorticoid administration on intestinal calcium absorption and circulating vitamin D metabolite concentrations in man. J. Clin. Endocrinol. Metab. 1981, 52, 111–115. [Google Scholar] [CrossRef]
- Findling, J.W.; Adams, N.D.; Lemann, J.; Gray, R.W.; Thomas, C.J.; Tyrrell, J.B. Vitamin D metabolites and parathyroid hormone in Cushing’s syndrome: Relationship to calcium and phosphorus homeostasis. J. Clin. Endocrinol. Metab. 1982, 54, 1039–1044. [Google Scholar] [CrossRef] [PubMed]
- Slovik, D.M.; Neer, R.M.; Ohman, J.L.; Lowell, F.C.; Clark, M.B.; Segre, G.V.; Potts, J.T., Jr. Parathyroid hormone and 25-hydroxyvitamin D levels in glucocorticoid-treated patients. Clin. Endocrinol. 1980, 12, 243–248. [Google Scholar] [CrossRef]
- Corbee, R.J.; Tryfonidou, M.A.; Meij, B.P.; Kooistra, H.S.; Hazewinkel, H.A.W. Vitamin D status before and after hypophysectomy in dogs with pituitary-dependent hypercortisolism. Domest. Anim. Endocrinol. 2012, 42, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Aloia, J.F.; Roginsky, M.; Ellis, K.; Shukla, K.; Cohn, S. Skeletal metabolism and body composition in Cushing’s syndrome. J. Clin. Endocrinol. Metab. 1974, 39, 981–985. [Google Scholar] [CrossRef]
- Van Cromphaut, S.J.; Stockmans, I.; Torrekens, S.; Van Herck, E.; Carmeliet, G.; Bouillon, R. Duodenal calcium absorption in dexamethasone-treated mice: Functional and molecular aspects. Arch. Biochem. Biophys. 2007, 460, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Akeno, N.; Matsunuma, A.; Maeda, T.; Kawane, T.; Horiuchi, N. Regulation of vitamin D-1-hydroxylase and -24-hydroxylase expression by dexamethasone in mouse kidney. J. Endocrinol. 2000, 164, 339–348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kurahashi, I.; Matsunuma, A.; Kawane, T.; Abe, M.; Horiuchi, N. Dexamethasone enhances vitamin D-24-hydroxylase expression in osteoblastic (UMR-106) and renal (LLC-PK 1) cells treated with 1a, 25-dihydroxyvitamin D3. Endocrine 2002, 17, 109–118. [Google Scholar] [CrossRef]
- Dhawan, P.; Christakos, S. Novel regulation of 25-hydroxyvitamin D3 24-hydroxylase (24(OH)ase) transcription by glucocorticoids: Cooperative effects of the glucocorticoid receptor, C/EBPb, and the vitamin D receptor in 24(OH)ase transcription. J. Cell. Biochem. 2010, 110, 1314–1323. [Google Scholar] [CrossRef] [PubMed]
- Luo, G.; Cunningham, M.; Kim, S.; Burn, T.; Lin, J.; Sinz, M.; Hamilton, G.; Rizzo, C.; Jolley, S.; Gilbert, D.; et al. CYP3A4 induction by drugs: Correlation between a pregnane X receptor reporter gene assay and CYP3A4 expression in human hepatocytes. Drug Metab. Dispos. 2002, 30, 795–804. [Google Scholar] [CrossRef] [Green Version]
- Zhou, C.; Assem, M.; Tay, J.C.; Watkins, P.B.; Blumberg, B.; Schuetz, E.G.; Thummel, K.E. Steroid and xenobiotic receptor and vitamin D receptor crosstalk mediates CYP24 expression and drug-induced osteomalacia. J. Clin. Investig. 2006, 116, 1703–1712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, P.; Xue, Y.; Li, H.; Liu, Y. Dysregulation of vitamin D metabolism in the brain and myocardium of rats following prolonged exposure to dexamethasone. Psychopharmacology 2014, 231, 3445–3451. [Google Scholar] [CrossRef] [PubMed]
- Zayny, A.; Almokhtar, M.; Wikvall, K.; Ljunggren, Ö.; Ubhayasekera, K.; Bergquist, J.; Kibar, P.; Norlin, M. Effects of glucocorticoids on vitamin D3-metabolizing 24-hydroxylase (CYP24A1) in Saos-2 cells and primary human osteoblasts. Mol. Cell. Endocrinol. 2019, 496, 110525. [Google Scholar] [CrossRef] [PubMed]
- Davidson, Z.E.; Walker, K.Z.; Truby, H. Do glucocorticosteroids alter vitamin D status? A systematic review with meta-analyses of observational studies. J. Clin. Endocrinol. Metab. 2014, 97, 738–744. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huybers, S.; Naber, T.H.J.; Bindels, R.J.M.; Hoenderop, J.G.J. Prednisolone-induced Ca2+ malabsorption is caused by diminished expression of the epithelial Ca2+ channel TRPV6. Am. J. Physiol.-Gastrointest. Liver Physiol. 2007, 292, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, P.; Bianchetti, M.G.; Sansonnens, A.; Frey, F.J. Modulation of renal calcium handling by 11β-hydroxysteroid dehydrogenase type 2. J. Am. Soc. Nephrol. 2002, 13, 2540–2546. [Google Scholar] [CrossRef] [Green Version]
- Faggiano, A.; Pivonello, R.; Melis, D.; Filippella, M.; Di Somma, C.; Petretta, M.; Lombardi, G.; Colao, A. Nephrolithiasis in Cushing’s disease: Prevalence, etiopathogenesis, and modification after disease cure. J. Clin. Endocrinol. Metab. 2003, 88, 2076–2080. [Google Scholar] [CrossRef] [PubMed]
- Ramsey, I.K.; Tebb, A.; Harris, E.; Evans, H.; Herrtage, M.E. Hyperparathyroidism in dogs with hyperadrenocorticism. J. Small Anim. Pract. 2005, 46, 531–536. [Google Scholar] [CrossRef]
- Freiberg, J.M.; Kinsella, J.; Sacktor, B. Glucocorticoids increase the Na+-H+ exchange and decrease the Na+ gradient-dependent phosphate-uptake systems in renal brush border membrane vesicles. Proc. Natl. Acad. Sci. USA 1982, 79, 4932–4936. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sempos, C.T.; Heijboer, A.C.; Bikle, D.D.; Bollerslev, J.; Bouillon, R.; Brannon, P.M.; DeLuca, H.F.; Jones, G.; Munns, C.F.; Bilezikian, J.P.; et al. Vitamin D assays and the definition of hypovitaminosis D: Results from the First International Conference on Controversies in Vitamin D. Br. J. Clin. Pharmacol. 2018, 84, 2194–2207. [Google Scholar] [CrossRef]
- Melnichenko, G.A.; Dedov, I.I.; Belaya, Z.E.; Rozhinskaya, L.Y.; Vagapova, G.R.; Volkova, N.I.; Grigor’ev, A.Y.; Grineva, E.N.; Marova, E.I.; Mkrtumayn, A.M.; et al. Cushing’s disease: The clinical features, diagnostics, differential diagnostics, and methods of treatment. Probl. Endocrinol. 2015, 61, 55–77. [Google Scholar] [CrossRef] [Green Version]
- Machado, M.C.; De Sa, S.V.; Domenice, S.; Fragoso, M.C.B.V.; Puglia, P.; Pereira, M.A.A.; De Mendonça, B.B.; Salgado, L.R. The role of desmopressin in bilateral and simultaneous inferior petrosal sinus sampling for differential diagnosis of ACTH-dependent Cushing’s syndrome. Clin. Endocrinol. 2007, 66, 136–142. [Google Scholar] [CrossRef]
- Findling, J.W.; Kehoe, M.E.; Raff, H. Identification of patients with Cushing’s disease with negative pituitary adrenocorticotropin gradients during inferior petrosal sinus sampling: Prolactin as an index of pituitary venous effluent. J. Clin. Endocrinol. Metab. 2004, 89, 6005–6009. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pigarova, E.A.; Rozhinskaya, L.Y.; Belaya, J.E.; Dzeranova, L.K.; Karonova, T.L.; Ilyin, A.V.; Melnichenko, G.A.; Dedov, I.I. Russian Association of Endocrinologists recommendations for diagnosis, treatment and prevention of vitamin D deficiency in adults. Probl. Endocrinol. 2016, 62, 60–84. [Google Scholar] [CrossRef]
- Petrushkina, A.A.; Pigarova, E.A.; Tarasova, T.S.; Rozhinskaya, L.Y. Efficacy and safety of high-dose oral vitamin D supplementation: A pilot study. Osteoporos. Int. 2016, 27, 512–513. [Google Scholar] [CrossRef]
- Pivonello, R.; De Martino, M.C.; De Leo, M.; Lombardi, G.; Colao, A. Cushing’s Syndrome. Endocrinol. Metab. Clin. N. Am. 2008, 37, 135–149. [Google Scholar] [CrossRef]
- Belaya, Z.E.; Iljin, A.V.; Melnichenko, G.A.; Rozhinskaya, L.Y.; Dragunova, N.V.; Dzeranova, L.K.; Butrova, S.A.; Troshina, E.A.; Dedov, I.I. Diagnostic performance of late-night salivary cortisol measured by automated electrochemiluminescence immunoassay in obese and overweight patients referred to exclude Cushing’s syndrome. Endocrine 2012, 41, 494–500. [Google Scholar] [CrossRef]
- Povaliaeva, A.; Pigarova, E.; Zhukov, A.; Bogdanov, V.; Dzeranova, L.; Mel’nikova, O.; Pekareva, E.; Malysheva, N.; Ioutsi, V.; Nikankina, L.; et al. Evaluation of vitamin D metabolism in patients with type 1 diabetes mellitus in the setting of cholecalciferol treatment. Nutrients 2020, 12, 3873. [Google Scholar] [CrossRef] [PubMed]
- Thode, J.; Juul-Jørgensen, B.; Bhatia, H.M.; Kjaerulf-Nielsen, M.; Bartels, P.D.; Fogh-Andersen, N.; Siggaard-Andersen, O. Comparison of serum total calcium, albumin-corrected total calcium, and ionized calcium in 1213 patients with suspected calcium disorders. Scand. J. Clin. Lab. Investig. 1989, 49, 217–223. [Google Scholar] [CrossRef]
- Bikle, D.D.; Siiteri, P.K.; Ryzen, E.; Haddad, J.G.; Gee, E. Serum protein binding of 1, 25-Dihydroxyvitamin D: A reevaluation by direct measurement of free metabolite levels. J. Clin. Endocrinol. Metab. 1985, 61, 969–975. [Google Scholar] [CrossRef]
- Bikle, D.D.; Gee, E.; Halloran, B.; Kowalski, M.A.N.N.; Ryzen, E.; Haddad, J.G. Assessment of the Free Fraction of 25-Hydroxyvitamin D in Serum and Its Regulation by Albumin and the Vitamin D-Binding Protein. J. Clin. Endocrinol. Metab. 1986, 63, 954–959. [Google Scholar] [CrossRef]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dirks, N.F.; Martens, F.; Vanderschueren, D.; Billen, J.; Pauwels, S.; Ackermans, M.T.; Endert, E.; den Heijer, M.; Blankenstein, M.A.; Heijboer, A.C. Determination of human reference values for serum total 1,25-dihydroxyvitamin D using an extensively validated 2D ID-UPLC–MS/MS method. J. Steroid Biochem. Mol. Biol. 2016, 164, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.C.Y.; Nicholls, H.; Piec, I.; Washbourne, C.J.; Dutton, J.J.; Jackson, S.; Greeves, J.; Fraser, W.D. Reference intervals for serum 24,25-dihydroxyvitamin D and the ratio with 25-hydroxyvitamin D established using a newly developed LC–MS/MS method. J. Nutr. Biochem. 2017, 46, 21–29. [Google Scholar] [CrossRef] [Green Version]
- Máčová, L.; Bičíková, M. Vitamin D: Current challenges between the laboratory and clinical practice. Nutrients 2021, 13, 1758. [Google Scholar] [CrossRef] [PubMed]
- Ginsberg, C.; Hoofnagle, A.N.; Katz, R.; Hughes-Austin, J.; Miller, L.M.; Becker, J.O.; Kritchevsky, S.B.; Shlipak, M.G.; Sarnak, M.J.; Ix, J.H. The Vitamin D Metabolite Ratio Is Associated With Changes in Bone Density and Fracture Risk in Older Adults. J. Bone Miner. Res. 2021, 1–8. [Google Scholar] [CrossRef]
- Cavalier, E.; Huyghebaert, L.; Rousselle, O.; Bekaert, A.C.; Kovacs, S.; Vranken, L.; Peeters, S.; Le Goff, C.; Ladang, A. Simultaneous measurement of 25(OH)-vitamin D and 24,25(OH)2-vitamin D to define cut-offs for CYP24A1 mutation and vitamin D deficiency in a population of 1200 young subjects. Clin. Chem. Lab. Med. 2020, 58, 197–201. [Google Scholar] [CrossRef]
- Rondeau, P.; Bourdon, E. The glycation of albumin: Structural and functional impacts. Biochimie 2011, 93, 645–658. [Google Scholar] [CrossRef]
- Soudahome, A.G.; Catan, A.; Giraud, P.; Kouao, S.A.; Guerin-Dubourg, A.; Debussche, X.; Le Moullec, N.; Bourdon, E.; Bravo, S.B.; Paradela-Dobarro, B.; et al. Glycation of human serum albumin impairs binding to the glucagon-like peptide-1 analogue liraglutide. J. Biol. Chem. 2018, 293, 4778–4791. [Google Scholar] [CrossRef] [Green Version]
- McLeod, J.F.; Kowalski, M.A.; Haddad, J.G. Interactions among serum vitamin D binding protein, monomeric actin, profilin, and profilactin. J. Biol. Chem. 1989, 264, 1260–1267. [Google Scholar] [CrossRef]
- Gupta, Y.; Gupta, A. Glucocorticoid-induced myopathy: Pathophysiology, diagnosis, and treatment. Indian J. Endocrinol. Metab. 2013, 17, 913. [Google Scholar] [CrossRef] [PubMed]
- Smets, P.; Meyer, E.; Maddens, B.; Daminet, S. Cushing’s syndrome, glucocorticoids and the kidney. Gen. Comp. Endocrinol. 2010, 169, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Arnaud, J.; Constans, J. Affinity differences for vitamin D metabolites associated with the genetic isoforms of the human serum carrier protein (DBP). Hum. Genet. 1993, 92, 183–188. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Study Group (n = 30) | Control Group (n = 30) | p |
|---|---|---|---|
| Age, years | 39.1 [31.2; 48.2] | 33.4 [26.5; 42.5] | 0.12 |
| Sex (female/male, n) | 26/4 | 19/11 | 0.07 |
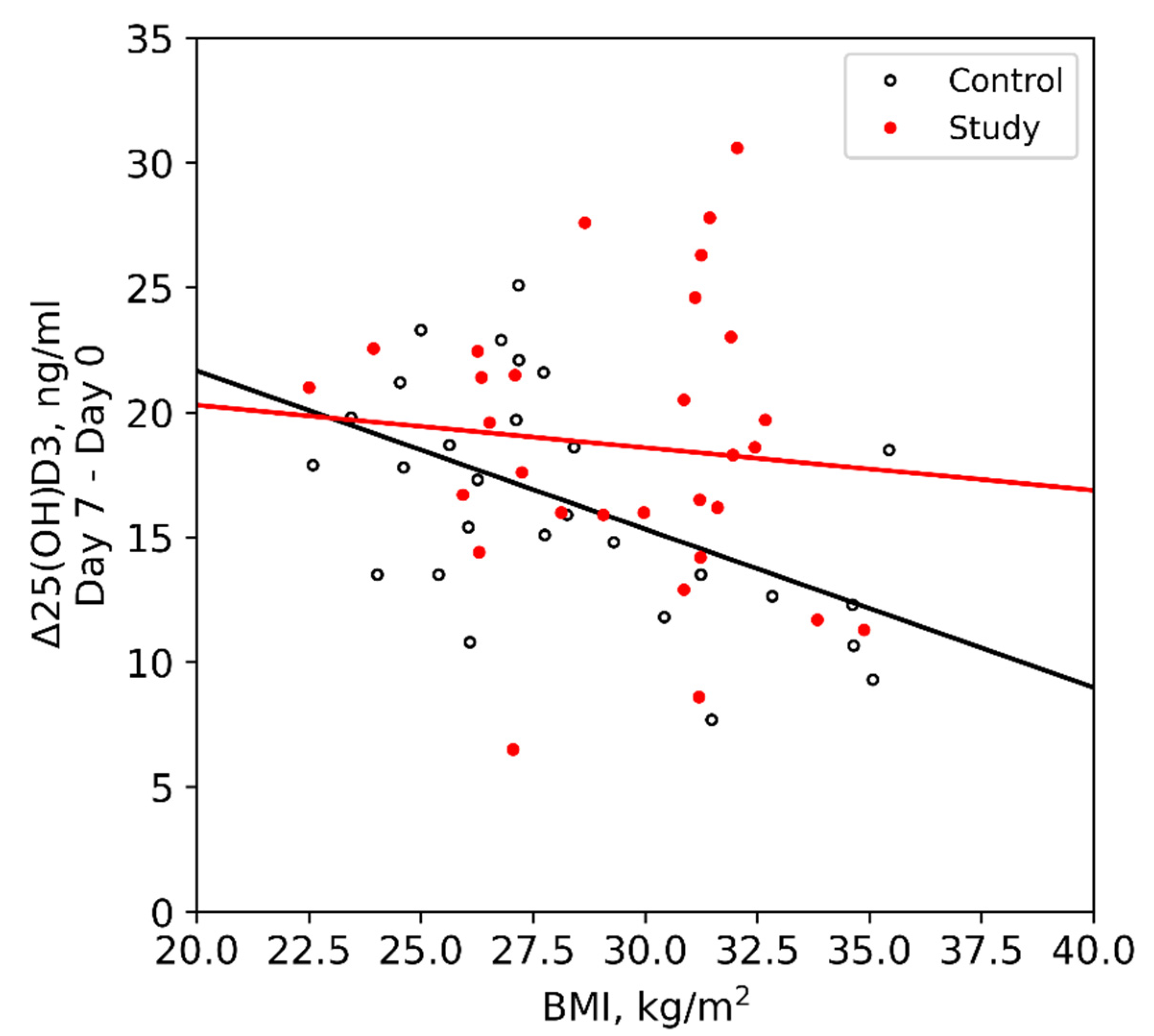
| BMI, kg/m2 | 30.9 [27.1; 31.6] | 27.2 [25.4; 30.4] | 0.07 |
| 25(OH)D total, ng/mL | 13.1 [9.6; 17.9] | 21.7 [14.4; 28.0] | 0.002 |
| Parameter | Value |
|---|---|
| 24-h UFC, nmoL/24 h | 1227 [813; 2970] |
| Morning ACTH, pg/mL | 87 [60; 125] |
| Diabetes mellitus, n (%) | 15 (50%) |
| HbA1c, % | 7.8 [7.0; 8.4] |
| History of low energy fracture, n (%) | 7 (23%) |
| Parameter | Study Group (n = 30) | Control Group (n = 30) | p |
|---|---|---|---|
| Current smokers, n (%) | 6 (20%) | 13 (43%) | 0.09 |
| Total smokers, n (%) | 10 (33%) | 18 (60%) | 0.07 |
| Alcohol units, per week | 0 [0; 0] | 1 [0; 2] | 0.007 |
| Exercises lasting more than 30 min, per week | 5 [2; 7] | 3 [2; 3] | 0.09 |
| Dairy products consumption, servings per day | 1 [1; 1] | 1 [1; 1] | 1.0 |
| Meat dishes consumption, portions per week | 5 [4; 7] | 5 [3; 7] | 0.64 |
| Coffee consumption, cups per week | 6 [2; 8] | 7 [1; 10] | 0.4 |
| Soft drinks, mL per week | 0 [0; 0] | 0 [0; 0] | 0.76 |
| Travelers to the south, n (%) | 3 (10%) | 4 (13%) | 1.0 |
| Daytime walks in the sunny weather, n | 7 [0; 20] | 4 [1; 11] | 0.49 |
| Solarium usage, n (%) | 0 | 1 (3%) | 1.0 |
| Laboratory Parameter | Group | Day 0 | Day 1 | Day 3 | Day 7 | p (Friedman ANOVA) | p (Day 0–1) | p (Day 1–3) | p (Day 3–7) | Reference Range |
|---|---|---|---|---|---|---|---|---|---|---|
| Total calcium, mmol/L | Study | 2.39 [2.25; 2.44] | 2.37 [2.31; 2.47] | 2.35 [2.27; 2.46] | 2.39 [2.27; 2.51] | 0.89 | - | - | - | 2.15–2.55 |
| Control | 2.37 [2.31; 2.43] | 2.41 [2.36; 2.46] | 2.41 [2.37; 2.46] | 2.37 [2.34; 2.48] | 0.03 | 0.01 | 0.47 | 0.28 | ||
| Albumin-adjusted calcium, mmol/L | Study | 2.28 [2.21; 2.36] | 2.31 [2.23; 2.38] | 2.30 [2.22; 2.36] | 2.31 [2.23; 2.37] | 0.92 | - | - | - | 2.15–2.55 |
| Control | 2.25 [2.21; 2.31] | 2.29 [2.23; 2.33] | 2.30 [2.26; 2.35] | 2.27 [2.24; 2.34] | 0.005 | 0.01 | 0.24 | 0.29 | ||
| Phosphorus, mmol/L | Study | 1.04 [0.98; 1.13] | 1.15 [1.02; 1.19] | 1.18 [1.04; 1.23] | 1.07 [0.97; 1.19] | 0.003 | 0.006 | 0.58 | 0.03 | 0.74–1.52 |
| Control | 1.10 [1.00; 1.22] | 1.15 [1.01; 1.26] | 1.19 [1.07; 1.27] | 1.15 [1.09; 1.31] | 0.06 | - | - | - | ||
| PTH, pg/mL | Study | 38.9 [33.8; 55.2] | 39.5 [29.7; 52.3] | 40.1 [31.7; 52.8] | 40.1 [30.5; 53.6] | 0.6 | - | - | - | 15–65 |
| Control | 38.6 [31.0; 50.3] | 37.0 [28.9; 51.4] | 35.8 [28.9; 45.3] | 34.3 [25.3; 47.7] | 0.03 | 0.74 | 0.02 | 0.93 | ||
| Creatinine, μmol/L | Study | 67.6 [62.4; 70.1] * | 68.9 [63.4; 72.8] * | 68.9 [62.1; 72.9] | 69.0 [63.3; 72.5] | 0.3 | - | - | - | 63–110 (male) 50–98 (female) |
| Control | 70.3 [67.4; 78.0] | 73.5 [66.9; 79.8] | 70.3 [67.1; 79.4] | 72.2 [64.0; 83.9] | 0.02 | 0.002 | 0.20 | 0.21 | ||
| Albumin, g/L | Study | 44 [42; 46] * | 44 [41; 45] * | 44 [40; 47] * | 44 [41; 47] | 0.3 | - | - | - | 35–50 |
| Control | 46 [44; 47] | 46 [44; 48] | 46 [44; 47] | 46 [44; 47] | 0.48 | - | - | - | ||
| Magnesium, mmol/L | Study | 0.87 [0.81; 0.92] * | 0.86 [0.79; 0.94] * | 0.84 [0.78; 0.91] * | 0.87 [0.81; 0.93] * | 0.27 | - | - | - | 0.7–1.05 |
| Control | 0.82 [0.76; 0.85] | 0.79 [0.77; 0.84] | 0.79 [0.76; 0.82] | 0.79 [0.75; 0.84] | 0.67 | - | - | - | ||
| Urine calcium-creatinine ratio, mmol/mmol | Study | 0.36 [0.16; 0.49] | 0.49 [0.28; 0.63] * | 0.37 [0.22; 0.59] | 0.49 [0.23; 0.80] | 0.05 | - | - | - | 0.1–0.8 |
| Control | 0.30 [0.13; 0.42] | 0.26 [0.21; 0.41] | 0.29 [0.21; 0.42] | 0.35 [0.20; 0.50] | 0.88 | - | - | - | ||
| Urine phosphorus-creatinine ratio, mmol/mmol | Study | 2.6 [2.0; 3.1] * | 2.6 [1.7; 3.1] * | 2.6 [2.0; 3.6] * | 3.4 [2.4; 4.1] * | 0.001 | 0.51 | 0.66 | 0.02 | 1.4–3.5 |
| Control | 1.8 [1.4; 2.7] | 1.7 [0.9; 2.4] | 1.6 [1.4; 2.3] | 1.7 [1.2; 2.3] | 0.09 | - | - | - |
| Laboratory Parameter | Group | Day 0 | Day 1 | Day 3 | Day 7 | p (Friedman ANOVA) | p (Day 0–1) | p (Day 1–3) | p (Day 3–7) | Reference Range |
|---|---|---|---|---|---|---|---|---|---|---|
| Free 25(OH)D, pg/mL | Study | 4.9 [4.0; 6.1] * | 10.7 [8.4; 12.5] | 12.9 [11.0; 14.3] | 11.4 [10.0; 12.5] | <0.001 | <0.001 | <0.001 | <0.001 | 2.4–35 1 |
| Control | 6.4 [4.1; 7.7] | 12.1 [9.5; 15.0] | 14.0 [10.3; 18.1] | 12.9 [9.4; 15.4] | <0.001 | <0.001 | <0.001 | <0.001 | ||
| DBP, mg/L | Study | 270 [227; 298] | 277 [247; 328] | 276 [236; 301] * | 252 [206; 281] | 0.16 | - | - | - | 176–623 1 |
| Control | 247 [212; 281] | 258 [237; 300] | 236 [204; 274] | 245 [220; 277] | 0.31 | - | - | - | ||
| 25(OH)D3, ng/mL | Study | 17.9 [13.0; 24.5] | 34.5 [27.6; 38.8] | 37.6 [33.2; 45.5] | 35.4 [32.1; 42.7] | <0.001 | <0.001 | <0.001 | 0.01 | ≥30 2 |
| Control | 19.5 [12.5; 25.7] | 31.0 [28.1; 35.0] | 33.9 [30.2; 43.1] | 34.3 [30.9; 42.9] | <0.001 | <0.001 | <0.001 | 0.65 | ||
| 3-epi-25(OH)D3, ng/mL | Study | 0.8 [0.6; 1.1] * | 3.0 [2.4; 3.5] | 4.2 [3.6; 5.1] | 3.1 [2.7; 3.8] | <0.001 | <0.001 | <0.001 | <0.001 | not available |
| Control | 1.4 [0.9; 1.7] | 2.7 [2.1; 3.5] | 3.9 [3.3; 4.9] | 3.6 [3.0; 4.6] | <0.001 | <0.001 | <0.001 | 0.003 | ||
| 1,25(OH)2D3, pg/mL | Study | 41 [35; 50] | 48 [37; 53] | 47 [42; 56] | 42 [39; 52] | <0.001 | <0.001 | 0.22 | 0.02 | 25–66 3 |
| Control | 42 [34; 48] | 48 [41; 55] | 46 [39; 54] | 43 [35; 47] | 0.09 | - | - | - | ||
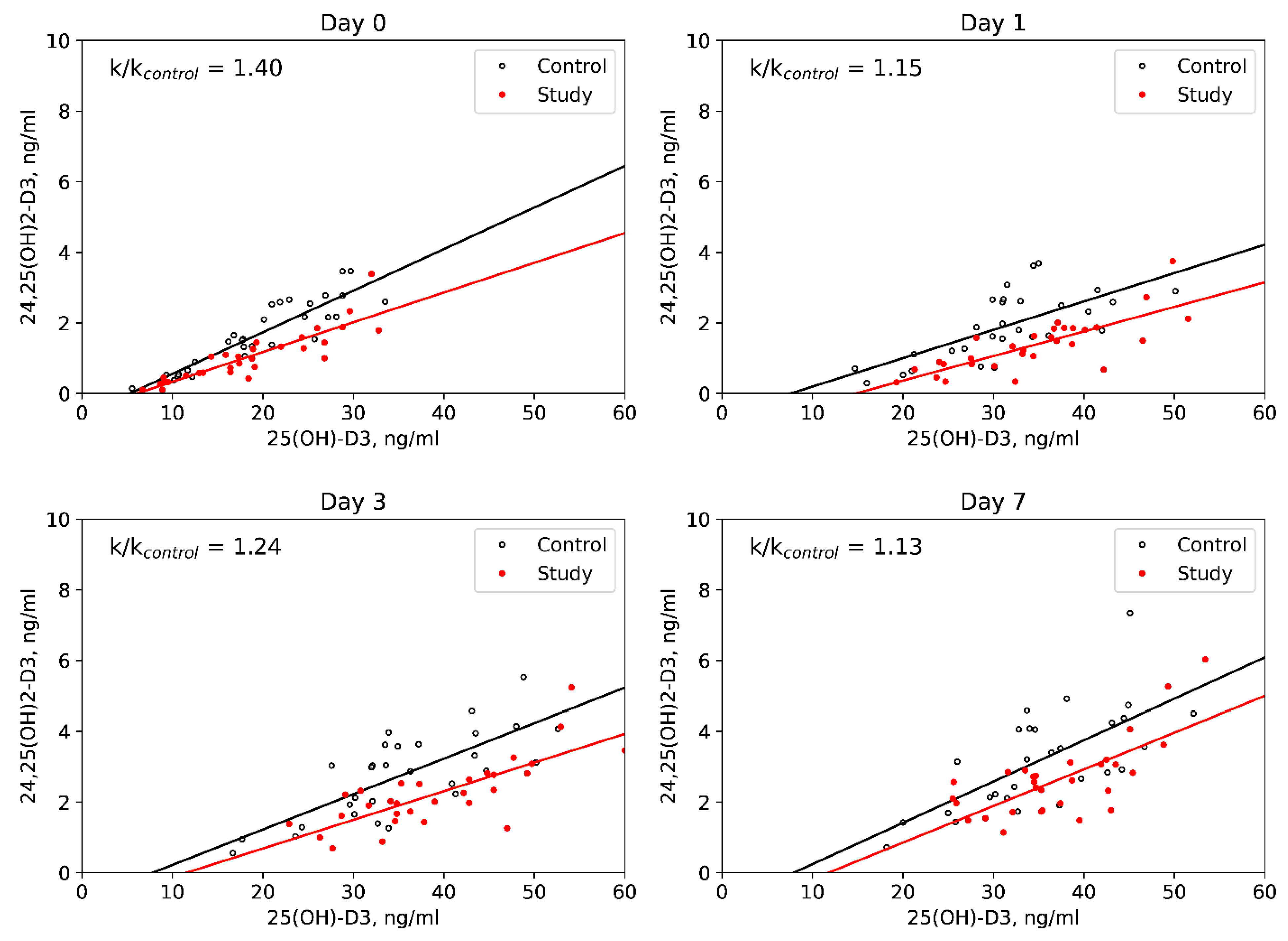
| 24,25(OH)2D3, ng/mL | Study | 1.0 [0.5; 1.4] * | 1.4 [0.8; 1.8] * | 2.1 [1.6; 2.8] | 2.6 [1.8; 3.1] | <0.001 | <0.001 | <0.001 | <0.001 | 0.5–5.6 3 |
| Control | 1.5 [0.9; 2.6] | 1.8 [1.2; 2.6] | 3.0 [1.9; 3.6] | 3.2 [2.1; 4.2] | <0.001 | <0.001 | <0.001 | <0.001 | ||
| 25(OH)D3/24,25(OH)2D3 | Study | 19.5 [15.3; 26.8] * | 26.8 [21.2; 32.5] * | 17.4 [15.9; 21.0] * | 13.9 [12.3; 18.8] * | <0.001 | <0.001 | <0.001 | <0.001 | 7–23 3 |
| Control | 12.7 [9.9; 17.0] | 18.2 [14.2; 21.3] | 13.1 [10.6; 18.2] | 12.3 [9.0; 14.9] | <0.001 | <0.001 | <0.001 | <0.001 | ||
| 25(OH)D3/1,25(OH)2D3 | Study | 425 [373; 555] | 716 [588; 917] | 783 [628; 1034] | 814 [666; 1044] | <0.001 | <0.001 | 0.08 | 0.86 | not available |
| Control | 501 [356; 641] | 689 [548; 789] | 757 [602; 972] | 851 [727; 1028] | <0.001 | <0.001 | 0.03 | 0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Povaliaeva, A.; Bogdanov, V.; Pigarova, E.; Zhukov, A.; Dzeranova, L.; Belaya, Z.; Rozhinskaya, L.; Mel’nichenko, G.; Mokrysheva, N. Assessment of Vitamin D Metabolism in Patients with Cushing’s Disease in Response to 150,000 IU Cholecalciferol Treatment. Nutrients 2021, 13, 4329. https://doi.org/10.3390/nu13124329
Povaliaeva A, Bogdanov V, Pigarova E, Zhukov A, Dzeranova L, Belaya Z, Rozhinskaya L, Mel’nichenko G, Mokrysheva N. Assessment of Vitamin D Metabolism in Patients with Cushing’s Disease in Response to 150,000 IU Cholecalciferol Treatment. Nutrients. 2021; 13(12):4329. https://doi.org/10.3390/nu13124329
Chicago/Turabian StylePovaliaeva, Alexandra, Viktor Bogdanov, Ekaterina Pigarova, Artem Zhukov, Larisa Dzeranova, Zhanna Belaya, Liudmila Rozhinskaya, Galina Mel’nichenko, and Natalia Mokrysheva. 2021. "Assessment of Vitamin D Metabolism in Patients with Cushing’s Disease in Response to 150,000 IU Cholecalciferol Treatment" Nutrients 13, no. 12: 4329. https://doi.org/10.3390/nu13124329
APA StylePovaliaeva, A., Bogdanov, V., Pigarova, E., Zhukov, A., Dzeranova, L., Belaya, Z., Rozhinskaya, L., Mel’nichenko, G., & Mokrysheva, N. (2021). Assessment of Vitamin D Metabolism in Patients with Cushing’s Disease in Response to 150,000 IU Cholecalciferol Treatment. Nutrients, 13(12), 4329. https://doi.org/10.3390/nu13124329






