Lingonberries—General and Oral Effects on the Microbiome and Inflammation
Abstract
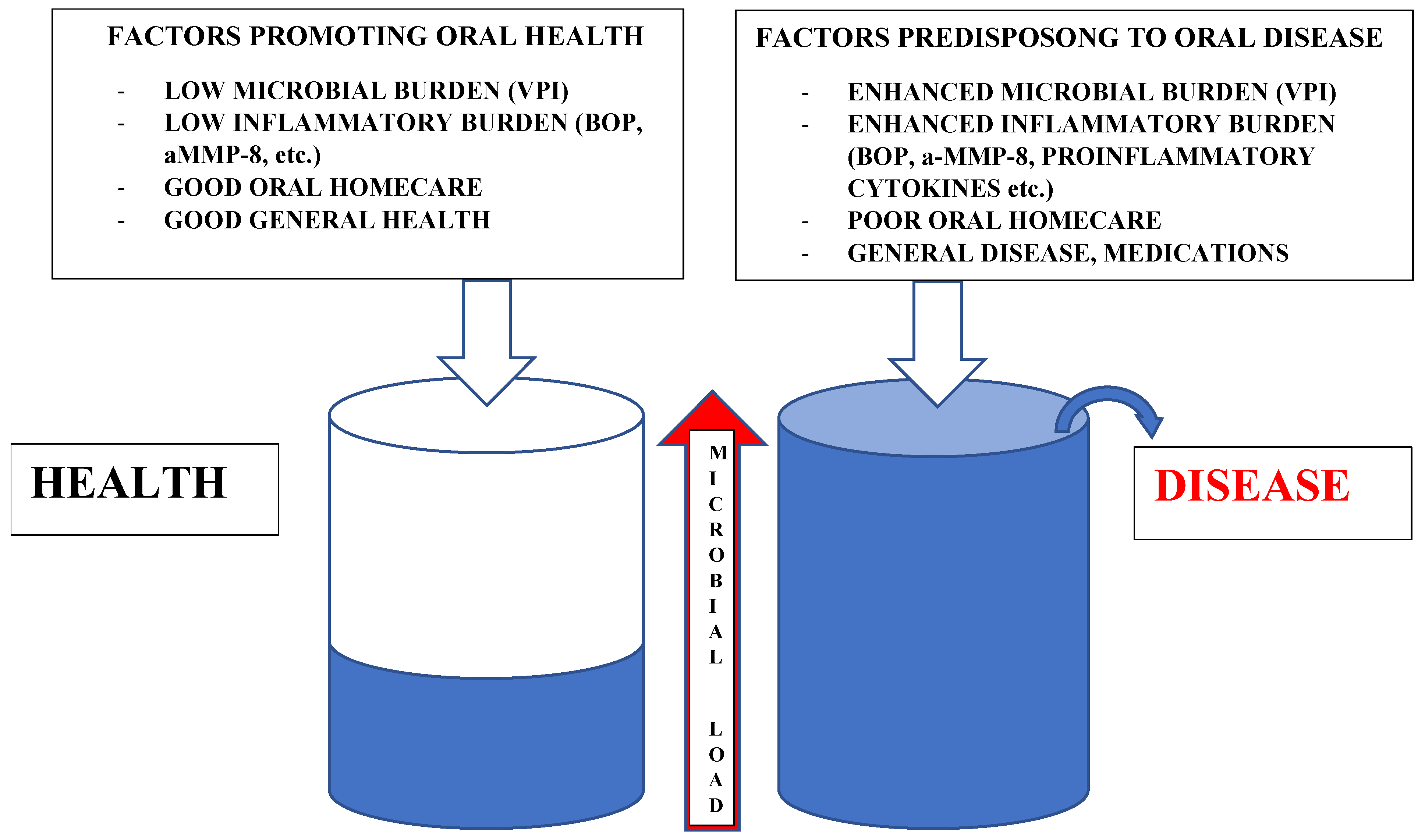
1. Symbiosis and Dysbiosis
2. Innate and Adaptive Immunity and Inflammation
3. Special Features in Microbe/Host Interactions in the Oral Environment
4. Lingonberries
5. In Vitro Anticancer Studies
6. In Vitro Antimicrobial Studies
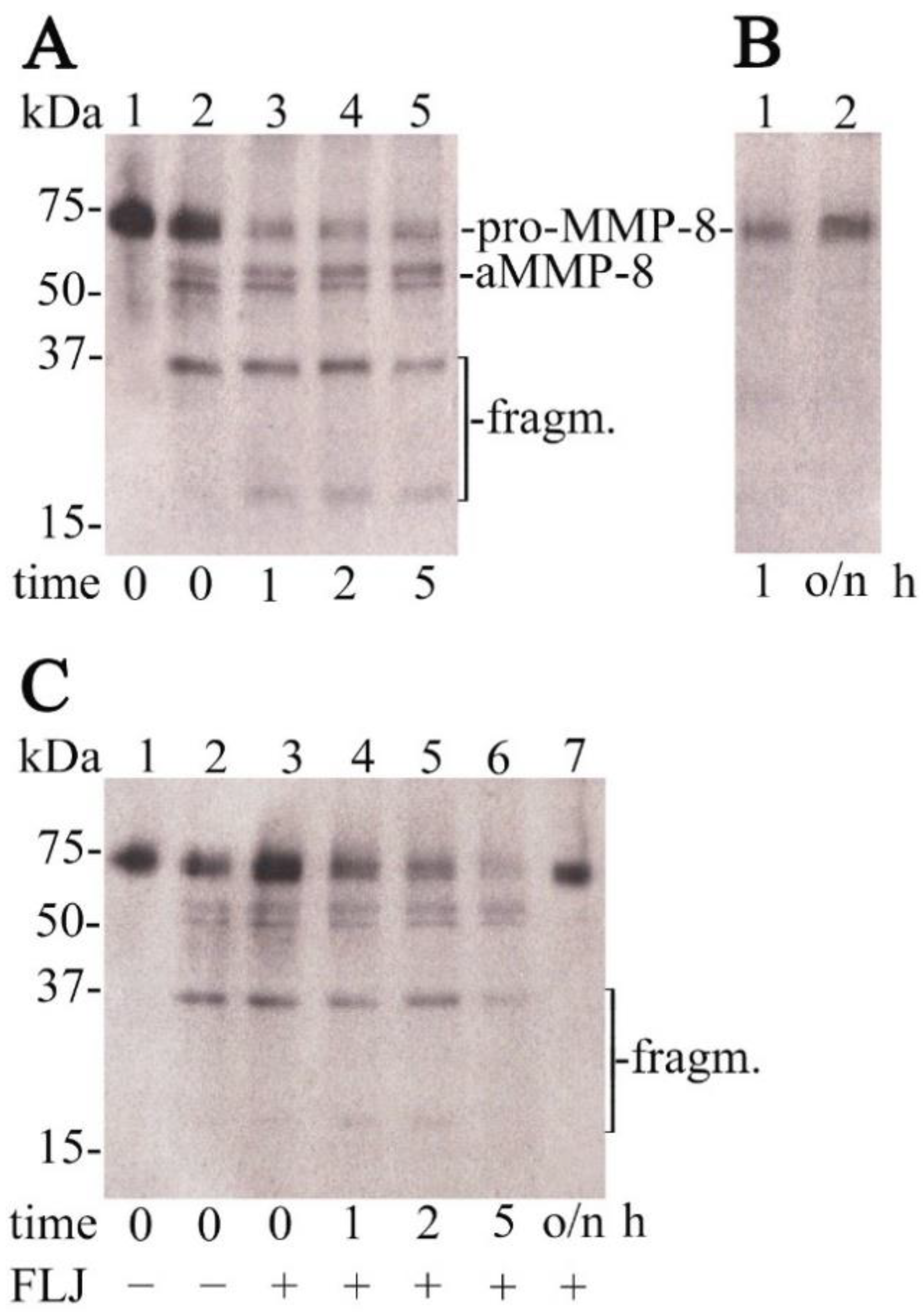
7. In Vitro Antioxidant and Anti-Inflammatory Studies
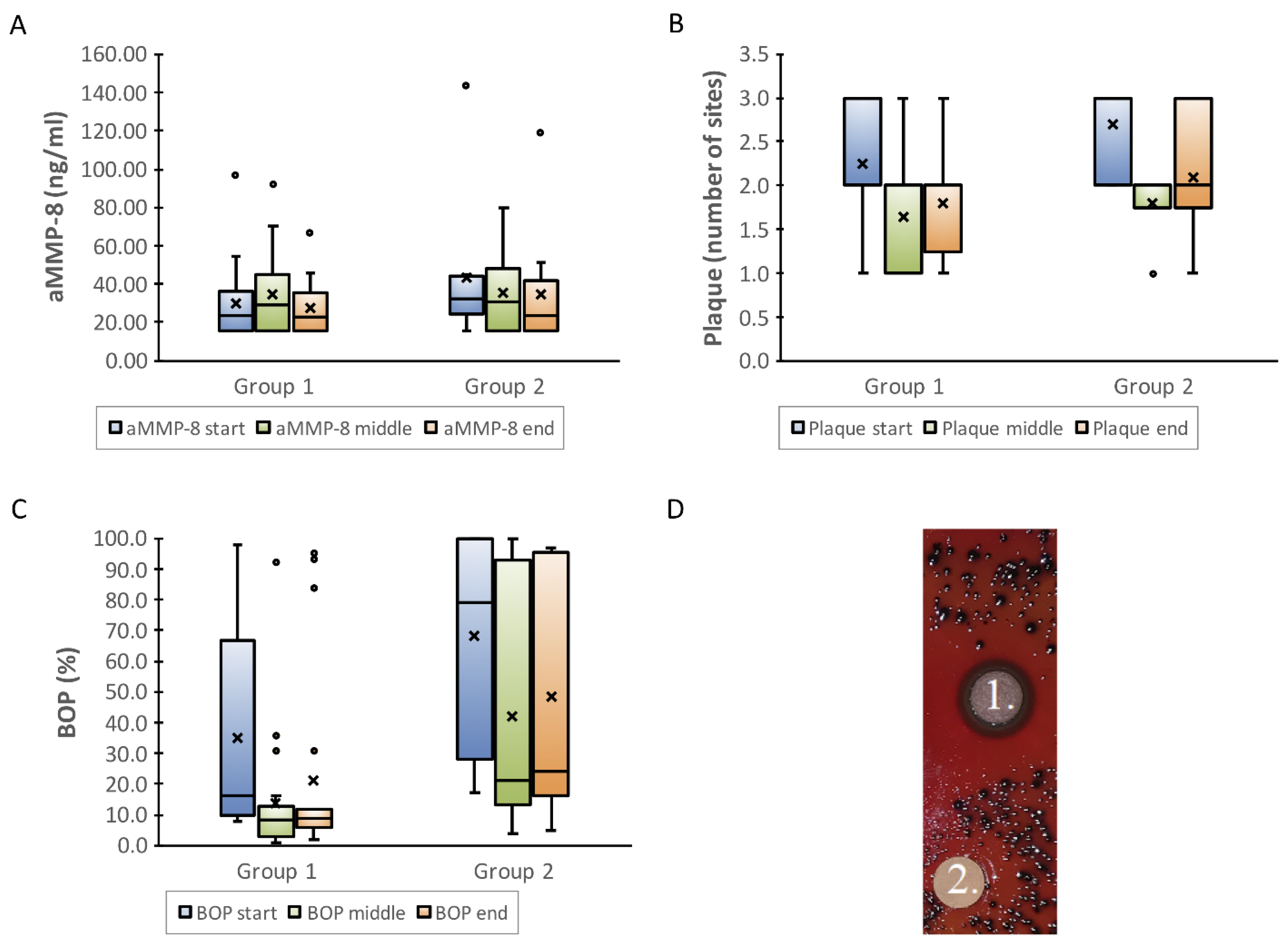
8. In Vivo Human Studies
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kilian, M.; Chapple, I.L.C.; Hannig, M.; Marsh, P.D.; Meuric, V.; Pedersen, A.M.L.; Tonetti, M.S.; Wade, W.G.; Zaura, E. The oral microbiome—An update for oral healthcare professionals. Br. Dent. J. 2016, 221, 657–666. [Google Scholar] [CrossRef]
- Dhotre, S.; Suryawanshi, N.; Selkar, S.; Nagoba, B. Viridans Group Streptococci and the Oral Ecosystem. Eur. J. Gen. Med. 2015, 13, 145–148. [Google Scholar] [CrossRef]
- Joseph, S.V.; Edirisinghe, I.; Burton-Freeman, B.M. Berries: Anti-inflammatory effects in humans. J. Agric. Food Chem. 2014, 62, 3886–3903. [Google Scholar] [CrossRef] [PubMed]
- Janeway, C.; Travers, P.; Walport, M.; Shlomchik, M. Immunobiology, The Immune System in Health and Disease 2001, 5th ed.; Garland Publishing: New York, NY, USA, 2001; pp. 1–91, 341–411. [Google Scholar]
- Pärnänen, P.; Kari, K.; Virtanen, I.; Sorsa, T.; Meurman, J. Human laminin-332 degradation by Candida proteinases. J. Oral. Pathol. Med. 2008, 37, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Pärnänen, P.; Meurman, J.; Virtanen, I. Laminin-511 and fibronectin degradation with Candida yeast. J. Oral. Pathol. Med. 2009, 38, 768–772. [Google Scholar] [CrossRef]
- Pärnänen, P.; Meurman, J.; Samaranayake, L.; Virtanen, I. Human oral keratinocyte E-cadherin degradation by Candida albicans and Candida glabrata. J. Oral. Pathol. Med. 2010, 39, 275–278. [Google Scholar] [CrossRef] [PubMed]
- Pärnänen, P.; Meurman, J.; Sorsa, T. The effects of Candida proteinases on human proMMP-9, TIMP-1 and TIMP-2. Mycoses 2010, 54, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Pärnänen, P.; Sorsa, T. Human Gingival Laminin-332 Degradation by Oral Bacteria. Laminins: Structure, Biological Activity and Role in Disease; Adams, D.C., Garcia, E.O., Eds.; Nova Science Publishers: Hauppauge, NY, USA, 2013; pp. 125–130. [Google Scholar]
- Nawaz, A.; Mäkinen, A.; Pärnänen, P.; Meurman, J. Proteolytic activity of non-albicans Candida and Candida albicans in oral cancer patients. New Microbiol. 2018, 41, 296–301. [Google Scholar]
- Lynge Pedersen, A.M.; Belstrøm, D. The role of natural salivary defences in maintaining a healthy oral microbiota. J. Dent. 2019, 80, S3–S12. [Google Scholar] [CrossRef]
- Sorsa, T.; Ingman, T.; Suomalainen, K.; Haapasalo, M.; Konttinen, Y.T.; Lindy, O.; Saari, H.; Uitto, V.-J. Identification of proteases from periodontopathogenic bacteria as activators of latent human neutrophil and fibroblast-type interstitial collagenases. Infect. Immun. 1992, 60, 4491–4495. [Google Scholar] [CrossRef]
- Nieminen, M.; Listyarifah, D.; Hagström, J.; Haglund, C.; Grenier, D.; Nordström, D.; Uitto, V.-J.; Hernandez, M.; Yucel-Lindberg, T.; Tervahartiala, T.; et al. Treponema denticola chymotrypsin-like proteinase may contribute to orodigestive carcinogenesis through immunomodulation. Br. J. Cancer 2018, 118, 428–434. [Google Scholar] [CrossRef] [PubMed]
- Pärnänen, P.; Sorsa, T.; Tervahartiala, T.; Nikula-Ijäs, P. Isolation, characterization and regulation of moonlighting proteases from Candida glabrata cell wall. Microb. Pathog. 2020, 149, 104547. [Google Scholar] [CrossRef]
- Hockensmith, K.; Dillard, K.; Sanders, B.; Harville, B.A. Identification and characterization of a chymotrypsin-like serine protease from periodontal pathogen, Tannerella forsythia. Microb. Pathog. 2016, 100, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, T.; Akaike, T.; Suga, M.; Tanase, S.; Horie, H.; Miyajima, S.; Ando, M.; Ichinose, Y.; Maeda, H. Activation of human matrix metalloproteinases by various bacterial proteinases. J. Biol. Chem. 1997, 272, 6059–6066. [Google Scholar] [CrossRef]
- Savonius, O.; Roine, I.; Alassiri, S.; Tervahartiala, T.; Helve, O.; Fernández, J.; Peltola, H.; Sorsa, T.; Pelkonen, T. The Potential Role of Matrix Metalloproteinases 8 and 9 and Myeloperoxidase in Predicting Outcomes of Bacterial Meningitis of Childhood. Mediat. Inflamm. 2019, 2019, 7436932. [Google Scholar] [CrossRef]
- Nukarinen, E.; Lindström, O.; Kuuliala, K.; Kylänpää, L.; Pettilä, V.; Puolakkainen, P.; Kuuliala, A.; Hämäläinrn, M.; Moilanen, E.; Repo, H.; et al. Association of Matrix Metalloproteinases-7, -8 and -9 and TIMP-1 with Disease Severity in Acute Pancreatitis. A Cohort Study. PLoS ONE 2016, 11, e0161480. [Google Scholar] [CrossRef] [PubMed]
- Turunen, A.; Kuuliala, K.; Kuuliala, A.; Tervahartiala, T.; Mustonen, H.; Puolakkainen, P.; Kylänpää, L.; Sorsa, T. Activated matrix metalloproteinase 8 in serum predicts severity of acute pancreatitis. Pancreatology 2021, 21, 862–869. [Google Scholar] [CrossRef]
- Lauhio, A.; Hästbacka, J.; Pettilä, V.; Tervahartiala, T.; Karlsson, S.; Varpula, T.; Varpula, M.; Ruokonen, E.; Sorsa, T.; Kolho, E. Serum MMP-8, -9 and TIMP-1 in sepsis: High serum levels of MMP-8 and TIMP-1 are associated with fatal outcome in a multicentre, prospective cohort study. Hypothetical impact of tetracyclines. Pharmacol. Res. 2011, 64, 590–594. [Google Scholar] [CrossRef] [PubMed]
- Lauhio, A.; Farkkila, E.; Pietilainen, K.H.; Astrom, P.; Winkelmann, A.; Tervahartiala, T.; Pirila, E.; Rissanen, A.; Kaprio, J.; Sorsa, T.A.; et al. Association of MMP-8 with obesity, smoking and insulin resistance. Eur. J. Clin. Investig. 2016, 46, 757–765. [Google Scholar] [CrossRef]
- Al-Majid, A.; Alassiri, S.; Rathnayake, N.; Tervahartiala, T.; Gieselmann, D.-R.; Sorsa, T. Matrix Metalloproteinase-8 as an Inflammatory and Prevention Biomarker in Periodontal and Peri-Implant Diseases. Int. J. Dent. 2018, 2018, 7891323. [Google Scholar] [CrossRef]
- Tuomainen, A.M.; Nyyssönen, K.; Laukkanen, J.A.; Tervahartiala, T.; Tuomainen, T.-P.; Salonen, J.T.; Sorsa, T.; Pussinen, P. Serum Matrix Metalloproteinase-8 Concentrations Are Associated With Cardiovascular Outcome in Men. Arter. Thromb. Vasc. Biol. 2007, 27, 2722–2728. [Google Scholar] [CrossRef]
- Kleinstein, S.E.; Nelson, K.E.; Freire, M. Inflammatory Networks Linking Oral Microbiome with Systemic Health and Disease. J. Dent. Res. 2020, 99, 1131–1139. [Google Scholar] [CrossRef]
- Shahbazi, R.; Sharifzad, F.; Bagheri, R.; Alsadi, N.; Yasavoli-Sharahi, H.; Matar, C. Anti-Inflammatory and Immunomodulatory Properties of Fermented Plant Foods. Nutrients 2021, 13, 1516. [Google Scholar] [CrossRef]
- Ek, S.; Kartimo, H.; Mattila, S.; Tolonen, A. Characterization of Phenolic Compounds from Lingonberry (Vaccinium vitis-idaea). J. Agric. Food Chem. 2006, 54, 9834–9842. [Google Scholar] [CrossRef]
- Andersen, Ø.M. Chromatographic Separation of Anthocyanins in Cowberry (Lingonberry) Vaccinium vites-idaea L. J. Food Sci. 1985, 50, 1230–1232. [Google Scholar] [CrossRef]
- Koli, R.; Erlund, I.; Jula, A.; Marniemi, J.; Mattila, P.; Alfthan, G. Bioavailability of various polyphenols from a diet containing moderate amounts of berries. J. Agric. Food Chem. 2010, 58, 3927–3932. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.M.; Nitecki, S.; Pereira-Caro, G.; McDougall, G.J.; Stewart, D.; Rowland, I.; Crozier, A.; Gill, C.I. Comparison of in vivo and in vitro digestion on polyphenol composition in lingonberries: Potential impact on colonic health. Biofactors 2014, 40, 611–623. [Google Scholar] [CrossRef]
- Kowalska, K. Lingonberry (Vaccinium vitis-idaea L.) Fruit as a Source of Bioactive Compounds with Health-Promoting Effects—A Review. Int. J. Mol. Sci. 2021, 22, 5126. [Google Scholar] [CrossRef] [PubMed]
- Hoornstra, D.; Vesterlin, J.; Parnanen, P.; Al-Samadi, A.; Zlotogorski-Hurvitz, A.; Vered, M.; Salo, T. Fermented Lingonberry Juice Inhibits Oral Tongue Squamous Cell Carcinoma Invasion In Vitro Similarly to Curcumin. In Vivo 2018, 32, 1089–1095. [Google Scholar] [CrossRef] [PubMed]
- Bomser, J.; Madhavi, D.L.; Singletary, K.; Smith, M.A. In vitro anticancer activity of fruit extracts from Vaccinium species. Planta Med. 1996, 62, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Olsson, M.E.; Gustavsson, K.-E.; Andersson, S.; Nilsson, Å.; Duan, R.-D. Inhibition of Cancer Cell Proliferation in Vitro by Fruit and Berry Extracts and Correlations with Antioxidant Levels. J. Agric. Food Chem. 2004, 52, 7264–7271. [Google Scholar] [CrossRef]
- McDougall, G.J.; Ross, H.A.; Ikeji, M.; Stewart, D. Berry Extracts Exert Different Antiproliferative Effects against Cervical and Colon Cancer Cells Grown in Vitro. J. Agric. Food Chem. 2008, 56, 3016–3023. [Google Scholar] [CrossRef] [PubMed]
- Onali, T.; Kivimäki, A.; Mauramo, M.; Salo, T.; Korpela, R. Anticancer Effects of Lingonberry and Bilberry on Digestive Tract Cancers. Antioxidants 2021, 10, 850. [Google Scholar] [CrossRef] [PubMed]
- Nohynek, L.J.; Alakomi, H.-L.; Kähkönen, M.P.; Heinonen, M.; Helander, I.M.; Oksman-Caldentey, K.-M.; Puupponen-Pimiä, R.H. Berry phenolics: Antimicrobial properties and mechanisms of action against severe human pathogens. Nutr. Cancer 2006, 54, 18–32. [Google Scholar] [CrossRef] [PubMed]
- Heinonen, M. Antioxidant activity and antimicrobial effect of berry phenolics—A Finnish perspective. Mol. Nutr. Food Res. 2007, 51, 684–691. [Google Scholar] [CrossRef] [PubMed]
- Riihinen, K.R.; Ou, Z.M.; Gödecke, T.; Lankin, D.C.; Pauli, G.F.; Wu, C.D. The antibiofilm activity of lingonberry flavonoids against oral pathogens is a case connected to residual complexity. Fitoterapia 2014, 97, 78–86. [Google Scholar] [CrossRef]
- Pärnänen, P.; Lähteenmäki, H.; Räisänen, I.; Tervahartiala, T.; Sorsa, T. Lingonberry polyphenols: Potential SARS-CoV-2 inhibitors as nutraceutical tools? Physiol. Rep. 2021, 9, e14741. [Google Scholar] [CrossRef]
- Nikolaeva-Glomb, L.; Mukova, L.; Nikolova, N.; Badjakov, I.; Dincheva, I.; Kondakova, V.; Doumanova, L.; Galabov, A.S. In vitro antiviral activity of a series of wild berry fruit extracts against representatives of Picorna-, Orthomyxo- and Paramyxoviridae. Nat. Prod. Commun. 2014, 9, 51–54. [Google Scholar] [CrossRef]
- Kylli, P.; Nohynek, L.; Puupponen-Pimiä, R.; Westerlund-Wikström, B.; Leppänen, T.; Welling, J.; Moilanen, E.; Heinonen, M. Lingonberry (Vaccinium vitis-idaea) and European Cranberry (Vaccinium microcarpon) Proanthocyanidins: Isolation, Identification, and Bioactivities. J. Agric. Food Chem. 2011, 59, 3373–3384. [Google Scholar] [CrossRef]
- Riihinen, K.; Ryynänen, A.; Toivanen, M.; Könönen, E.; Törrönen, R.; Tikkanen-Kaukanen, C. Antiaggregation potential of berry fractions against pairs of Streptococcus mutans with Fusobacterium nucleatum or Actinomyces naeslundii. Phytother. Res. 2011, 25, 81–87. [Google Scholar] [CrossRef]
- Pärnänen, P.; Nawaz, A.; Sorsa, T.; Meurman, J.; Nikula-Ijäs, P. The Effect of Fermented Lingonberry Juice on Candida glabrata Intracellular Protein Expression. Int. J. Dent. 2017, 2017, 6185395. [Google Scholar] [CrossRef] [PubMed]
- Pärnänen, P.; Nikula-Ijäs, P.; Sorsa, T. Antimicrobial and anti-inflammatory lingonberry mouthwash. a clinical pilot study in the oral cavity. Microorganisms 2019, 7, 331. [Google Scholar] [CrossRef] [PubMed]
- Puupponen-Pimiä, R.; Nohynek, L.; Meier, C.; Kähkönen, M.; Heinonen, M.; Hopia, A.; Oksman-Caldentey, K.-M. Antimicrobial properties of phenolic compounds from berries. J. Appl. Microbiol. 2001, 90, 494–507. [Google Scholar] [CrossRef]
- Puupponen-Pimiä, R.; Nohynek, L.; Hartmann-Schmidlin, S.; Kähkönen, M.; Heinonen, M.; Määttä-Riihinen, K.; Oksman-Caldentey, K.-M. Berry phenolics selectively inhibit the growth of intestinal pathogens. J. Appl. Microbiol. 2005, 98, 991–1000. [Google Scholar] [CrossRef] [PubMed]
- Puupponen-Pimiä, R.; Nohynek, L.; Alakomi, H.-L.; Oksman-Caldentey, K.-M. Bioactive berry compounds—Nnovel tools against human pathogens. Appl. Microbiol. Biotechnol. 2005, 67, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Puupponen-Pimiä, R.; Nohynek, L.; Alakomi, H.-L.; Oksman-Caldentey, K.-M. The action of berry phenolics against human intestinal pathogens. Biofactors 2005, 23, 243–251. [Google Scholar] [CrossRef]
- Ho, K.Y.; Tsai, C.C.; Huang, J.S.; Chen, C.P.; Lin, T.C.; Lin, C.C. Antimicrobial activity of tannin components from Vaccinium vitis idaea L. J. Pharm. Pharmacol. 2001, 53, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Toivanen, M.; Huttunen, S.; Duricova, J.; Soininen, P.; Laatikainen, R.; Loimaranta, V.; Haataja, S.; Finne, J.; Lapinjoki, S.; Tikkanen-Kaukanen, C. Screening of binding activity Streptococcus pneumoniae, Streptococcus agalactiae and Streptococcus suis to berries and juices. Phytother. Res. 2010, 24, S95–S101. [Google Scholar] [CrossRef] [PubMed]
- Toivanen, M.; Huttunen, S.; Lapinjoki, S.; Tikkanen-Kaukanen, C. Inhibition of adhesion of Neisseria meningitidis to human epithelial cells by berry juice polyphenolic fractions. Phytother. Res. 2011, 25, 828–832. [Google Scholar] [CrossRef]
- Kokubu, E.; Kinoshita, E.; Ishihara, K. Inhibitory effects of lingonberry extract on oral streptococci biofilm formation and bioactivity. Bull. Tokyo Dent. Coll. 2019, 60, 1–9. [Google Scholar] [CrossRef]
- Kelly, E.; Vyas, P.; Weber, J.T. Biochemical properties and neuroprotective effects of compounds in various species of berries. Molecules 2018, 23, 26. [Google Scholar] [CrossRef] [PubMed]
- Ho, K.Y.; Huang, J.S.; Tsai, C.C.; Lin, T.C.; Hsu, Y.F.; Lin, C.C. Antioxidant Activity of Tannin Components from Vaccinium vitis-idaea L. J. Pharm. Pharmacol. 1999, 51, 1075–1078. [Google Scholar] [CrossRef] [PubMed]
- Kähkönen, M.P.; Hopia, A.I.; Heinonen, M. Berry Phenolics and Their Antioxidant Activity. J. Agric. Food Chem. 2001, 49, 4076–4082. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Wang, S.Y. Oxygen radical absorbing capacity of phenolics in blueberries, cranberries, chokeberries, and lingonberries. J. Agric. Food Chem. 2003, 51, 502–509. [Google Scholar] [CrossRef]
- Viljanen, K.; Kylli, P.; Kivikari, R.; Heinonen, M. Inhibition of Protein and Lipid Oxidation in Liposomes by Berry Phenolics. J. Agric. Food Chem. 2004, 52, 7419–7424. [Google Scholar] [CrossRef]
- Määttä-Riihinen, K.R.; Kähkönen, M.P.; Törrönen, A.R.; Heinonen, M. Catechins and Procyanidins in Berries of Vaccinium Species and Their Antioxidant Activity. J. Agric. Food Chem. 2005, 53, 8485–8491. [Google Scholar] [CrossRef]
- Wu, C.-H.; Yen, G.-C. Inhibitory Effect of Naturally Occurring Flavonoids on the Formation of Advanced Glycation Endproducts. J. Agric. Food Chem. 2005, 53, 3167–3173. [Google Scholar] [CrossRef]
- Mane, C.; Loonis, M.; Juhel, C.; Dufour, C.; Malien-Aubert, C. Food Grade Lingonberry Extract: Polyphenolic Composition and In Vivo Protective Effect against Oxidative Stress. J. Agric. Food Chem. 2011, 59, 3330–3339. [Google Scholar] [CrossRef]
- Ogawa, K.; Tsuruma, K.; Tanaka, J.; Kakino, M.; Kobayashi, S.; Shimazawa, M.; Hara, H. The protective effects of bilberry and lingonberry extracts against UV light-induced retinal photoreceptor cell damage in vitro. J. Agric. Food Chem. 2013, 61, 10345–10353. [Google Scholar] [CrossRef] [PubMed]
- Kowalska, K.; Olejnik, A.; Zielińska-Wasielica, J.; Olkowicz, M. Inhibitory effects of lingonberry (Vaccinium vitis-idaea L.) fruit extract on obesity-induced inflammation in 3T3-L1 adipocytes and RAW 264.7 macrophages. J. Funct. Foods 2019, 54, 371–380. [Google Scholar] [CrossRef]
- Esposito, D.; Overall, J.; Grace, M.H.; Komarnytsky, S.; Lila, M.A. Alaskan Berry Extracts Promote Dermal Wound Repair Through Modulation of Bioenergetics and Integrin Signaling. Front. Pharmacol. 2019, 10, 1058. [Google Scholar] [CrossRef]
- Kivimäki, A.S.; Ehlers, P.I.; Siltari, A.; Turpeinen, A.M.; Vapaatalo, H.; Korpela, R. Lingonberry, cranberry and blackcurrant juices affect mRNA expressions of inflammatory and atherotrombotic markers of SHR in a long-term treatment. J. Funct. Foods 2012, 4, 496–503. [Google Scholar] [CrossRef]
- Kivimäki, A.S.; Siltari, A.; Ehlers, P.I.; Korpela, R.; Vapaatalo, H. Lingonberry juice negates the effects of a high salt diet on vascular function and low-grade inflammation. J. Funct. Foods 2014, 7, 238–245. [Google Scholar] [CrossRef]
- Eid, H.M.; Ouchfoun, M.; Brault, A.; Vallerand, D.; Musallam, L.; Arnason, J.T.; Haddad, P.S. Lingonberry (Vaccinium vitis-idaea L.) Exhibits Antidiabetic Activities in a Mouse Model of Diet-Induced Obesity. Evid. -Based Complementary Altern. Med. 2014, 2014, 645812. [Google Scholar] [CrossRef]
- Ryyti, R.; Hämäläinen, M.; Peltola, R.; Moilanen, E. Beneficial effects of lingonberry (Vaccinium vitis-idaea L.) supplementation on metabolic and inflammatory adverse effects induced by high-fat diet in a mouse model of obesity. PLoS ONE 2020, 15, e0232605. [Google Scholar] [CrossRef]
- Marungruang, N.; Kovalenko, T.; Osadchenko, I.; Voss, U.; Huang, F.; Burleigh, S.; Ushakova, G.; Skibo, G.; Nyman, M.; Prykhodko, O.; et al. Lingonberries and their two separated fractions differently alter the gut microbiota, improve metabolic functions, reduce gut inflammatory properties, and improve brain function in ApoE−/− mice fed high-fat diet. Nutr. NeuroSci. 2020, 23, 600–612. [Google Scholar] [CrossRef]
- Törrönen, R.; Kolehmainen, M.; Sarkkinen, E.; Mykkänen, H.; Niskanen, L. Postprandial glucose, insulin, and free fatty acid responses to sucrose consumed with blackcurrants and lingonberries in healthy women. Am. J. Clin. Nutr. 2012, 96, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Törrönen, R.; Kolehmainen, M.; Sarkkinen, E.; Poutanen, K.; Mykkänen, H.; Niskanen, L. Berries reduce postprandial insulin responses to wheat and rye breads in healthy women. J. Nutr. 2013, 143, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Pärnänen, P. A preparation for balancing the composition of the oral microbial flora. Europatent 2017, 2585087B1. [Google Scholar]
- Sorsa, T.; Alassiri, S.; Grigoriadis, A.; Räisänen, I.T.; Pärnänen, P.; Nwhator, S.O.; Gieselmann, D.R.; Sakellari, D. Active MMP-8 (aMMP-8) as a Grading and Staging Biomarker in the Periodontitis Classification. Diagnostics 2020, 10, 61. [Google Scholar] [CrossRef]
- Deng, K.; Pelekos, G.; Jin, L.; Tonetti, M.S. Diagnostic accuracy of a point-of-care aMMP-8 test in the discrimination of periodontal health and disease. J. Clin. Periodontol. 2021, 48, 1051–1065. [Google Scholar] [CrossRef]
- Raisanen, I.T.; Sorsa, T.; Tervahartiala, T.; Raivisto, T.; Heikkinen, A.M. Low association between bleeding on probing propensity and the salivary aMMP-8 levels in adolescents with gingivitis and stage I periodontitis. J. Periodontal. Res. 2021, 56, 289–297. [Google Scholar] [CrossRef]
- Contardo, M.S.; Diaz, N.; Lobos, O.; Padilla, C.; Giacaman, R.A. Oral colonization by Streptococcus mutans and its association with the severity of periodontal disease in adults. Rev. Clin. Periodoncia. Implantol. Rehabil. Oral. 2011, 4, 9–12. [Google Scholar] [CrossRef]
- Chen, L.-J.; Tsai, H.-T.; Chen, W.-J.; Hsieh, C.-Y.; Wang, P.-C.; Chen, C.-S.; Wang, L.; Yang, C.-C. In vitro antagonistic growth effects of Lactobacillus fermentum and Lactobacillus salivarius and their fermentatative broth on periodontal pathogens. Braz. J. Microbiol. 2012, 43, 1376–1384. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pärnänen, P.; Lähteenmäki, H.; Tervahartiala, T.; Räisänen, I.T.; Sorsa, T. Lingonberries—General and Oral Effects on the Microbiome and Inflammation. Nutrients 2021, 13, 3738. https://doi.org/10.3390/nu13113738
Pärnänen P, Lähteenmäki H, Tervahartiala T, Räisänen IT, Sorsa T. Lingonberries—General and Oral Effects on the Microbiome and Inflammation. Nutrients. 2021; 13(11):3738. https://doi.org/10.3390/nu13113738
Chicago/Turabian StylePärnänen, Pirjo, Hanna Lähteenmäki, Taina Tervahartiala, Ismo T. Räisänen, and Timo Sorsa. 2021. "Lingonberries—General and Oral Effects on the Microbiome and Inflammation" Nutrients 13, no. 11: 3738. https://doi.org/10.3390/nu13113738
APA StylePärnänen, P., Lähteenmäki, H., Tervahartiala, T., Räisänen, I. T., & Sorsa, T. (2021). Lingonberries—General and Oral Effects on the Microbiome and Inflammation. Nutrients, 13(11), 3738. https://doi.org/10.3390/nu13113738




