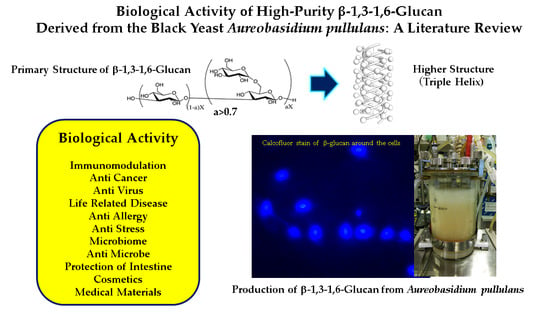
Biological Activity of High-Purity β-1,3-1,6-Glucan Derived from the Black Yeast Aureobasidium pullulans: A Literature Review
Abstract
1. Introduction
- Kingdom: Fungi
- Phylum: Ascomycota
- Class: Dothideomycetes
- Subclass: Dothideomycetidae
- Order: Dothideales
- Family: Dothioraceae
- Genus: Aureobasidium
- Species: A. pullulans
2. Results
2.1. Production of APβG via Fermentation
2.1.1. Fermentation
2.1.2. β-Glucan Recovery and Purification
2.2. Structure and Bioactivity of APβG
2.3. Safety
2.4. Physiological Function of β-1,3-1,6-glucan
2.4.1. Intestinal Immunostimulatory Effect
2.4.2. Splenic Immunomodulation
2.4.3. Antitumor and Antimetastatic Activities
2.4.4. Antimicrobial Activities
2.4.5. Effect of Alleviating Influenza Symptoms
2.4.6. Effects on Improving Lifestyle-Related Diseases, Including Obesity
2.4.7. Postprandial Blood Glucose Reduction
2.4.8. Anti-Type I Allergic and Anti-Inflammatory Effects
2.4.9. Anti-stress and Immunomodulatory Effects
2.4.10. Protective Effect on the Intestinal and Gastrointestinal Mucosa
2.4.11. Effects on the Microbiome
2.4.12. Application to the Skin
2.4.13. Osteoporosis Improvement
2.4.14. Use as a Material Inclusion Agent
3. Discussion
3.1. Physiological Functions and Polysaccharides
3.2. Recognition Mechanism of β-Glucans
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ohno, N. Immunopharmacological Activity of Polysaccharides. Fragr. J. 1995, 5, 70–75. [Google Scholar] [CrossRef]
- Yadomae, T. Structure and Biological Activity of Fungus β-1,3-Glucans. J. Pharm. Soc. Jpn. 2000, 120, 413–431. [Google Scholar] [CrossRef]
- Ohno, N. Modulation of Host Defense Systems by Beta-Glucans. Jpn. J. Bacteriol. 2000, 55, 527–537. [Google Scholar] [CrossRef]
- Ohno, N. Structural Diversity and Physiological Functions of β-Glucans. Int. J. Med. Mushrooms 2005, 7, 167–174. [Google Scholar] [CrossRef]
- Suzuki, T.; Nishikawa, K.; Nakamura, S.; Suzuki, T. [Review: Prize-Awarded Article] Research and Development of β-1,3–1,6-Glucan From Black Yeast for a Functional Food Ingredient. Bull. Appl. Glycosci. 2012, 2, 51–60. [Google Scholar] [CrossRef]
- Miyamoto, J.; Watanabe, K.; Taira, S.; Kasubuchi, M.; Li, X.; Irie, J.; Itoh, H.; Kimura, I. Barley β-Glucan Improves Metabolic Condition via Short-Chain Fatty Acids Produced by Gut Microbial Fermentation in High Fat Diet Fed Mice. PLoS ONE 2018, 13, e0196579. [Google Scholar] [CrossRef]
- Sobieralski, K.; Siwulski, M.; Lisiecka, J.; Jedryczka, M.; Sas-Golak, I.; Fruzynska-Józwiak, D. Fungi-Derived β-Glucans as a Component of Fungal Food. Acta Sci. Pol. Hortorum Cultus 2012, 11, 111–128. [Google Scholar]
- Bzducha, A.-W.; Pobiega, K.; Błażejak, S.; Kieliszek, M. The Scale-Up Cultivation of Candida utilis in Waste Potato Juice Water With Glycerol Affects Biomass and β (1, 3)/(1, 6)–Glucan Characteristic and Yield. Appl. Microbiol. Biotechnol. 2018, 102, 9131–9145. [Google Scholar] [CrossRef]
- Bzducha, A.-W.; Koczoń, P.; Błażejak, S.; Kozera, J.; Kieliszek, M. Valorization of Deproteinated Potato Juice Water Into β-Glucan Preparation of C. utilis Origin: Comparative Study of Preparations Obtained by Two Isolation Methods. Waste Biomass Valori. 2020, 11, 3257–3271. [Google Scholar] [CrossRef]
- Gantner, B.N.; Simmons, R.M.; Canavera, S.J.; Akira, S.; Underhill, D.M. Collaborative Induction of Inflammatory Responses by dectin-1 and Toll-Like receptor 2. J. Exp. Med. 2003, 197, 1107–1117. [Google Scholar] [CrossRef]
- Nagano, Y.; Elborn, J.S.; Millar, B.C.; Goldsmith, C.E.; Rendall, J.; Moore, J.E. Development of a Novel PCR Assay for the Identification of the Black Yeast, Exophiala (Wangiella) Dermatitidis from Adult Patients with Cystic Fibrosis (CF). J. Cyst. Fibros. 2008, 7, 576–580. [Google Scholar] [CrossRef] [PubMed]
- Gostinčar, C.; Ohm, R.A.; Kogej, T.; Sonjak, S.; Turk, M.; Zajc, J.; Zalar, P.; Grube, M.; Sun, H.; Han, J.; et al. Genome Sequencing of Four Aureobasidium pullulans Varieties: Biotechnological Potential, Stress Tolerance, and Description of New species. BMC Genom. 2014, 15, 549–576. [Google Scholar] [CrossRef] [PubMed]
- Arkadjeva, G.F. The Biological Activity of Polysaccharides Isolated From Rhodotorula glutinis and Aureobasidium pullulans. Antonie Leeuwenhoek 1969, 35, E9–E10. [Google Scholar]
- Han, Y.W.; Cheeke, P.R.; Anderson, A.W.; Lekprayoon, C. Growth of Aureobasidium pullulans on Straw Hydrolysate. Appl. Environ. Microbiol. 1976, 32, 799–802. [Google Scholar] [CrossRef] [PubMed]
- Anastassiadis, S.; Morgunov, I.G. Gluconic Acid Production. Recent Pat. Biotechnol. 2007, 1, 167–180. [Google Scholar] [CrossRef] [PubMed]
- Bharti, S.K.; Krishnan, S.; Kumar, A.; Rajak, K.K.; Murari, K.; Bharti, B.K.; Gupta, A.K. Antidiabetic Activity and Molecular Docking of Fructooligosaccharides Produced by Aureobasidium pullulans in poloxamer-407-induced T2DM Rats. Food Chem. 2013, 136, 813–821. [Google Scholar] [CrossRef]
- Tamano, K.; Nakasha, K.; Iwamoto, M.; Numata, M.; Suzuki, T.; Uyama, H.; Fukuhara, G. Chiroptical Properties of Reporter-Modified or Reporter-Complexed Highly 1,6-Glucose-Branched β-1,3-Glucan. Polym. J. 2019, 51, 1063–1071. [Google Scholar] [CrossRef]
- Suzuki, T.; Nakamura, S.; Nishikawa, K.; Nakayama, S.; Suzuki, T. Production of Highly Purified β-Glucan From Aureobasidium pullulans and Its Characteristics. Food Funct. 2006, 2, 45–50. [Google Scholar]
- Hamada, N.; Tsujisaka, Y. The Structure of the Carbohydrate Moiety of an Acidic Polysaccharide Produced by Aureobasidium sp. K-1. Agric. Biol. Chem. 1983, 47, 1167–1172. [Google Scholar] [CrossRef]
- Tada, R.; Tanioka, A.; Iwasawa, H.; Hatashima, K.; Shoji, Y.; Ishibashi, K.I.; Adachi, Y.; Yamazaki, M.; Tsubaki, K.; Ohno, N. Structural Characterisation and Biological Activities of a Unique Type Beta-D-Glucan Obtained From Aureobasidium pullulans. Glycoconj. J. 2008, 25, 851–861. [Google Scholar] [CrossRef]
- Hirabayashi, K.; Kondo, N.; Hayashi, S. Characterization and Enzymatic Hydrolysis of Hydrothermally Treated β-1,3–1,6-Glucan From Aureobasidium pullulans. World J. Microbiol. Biotechnol. 2016, 32, 206. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, N.; Shoubayashi, Y.; Kondo, N.; Fukudome, K. Hydrothermal Processing of β-Glucan From Aureobasidium pullulans Produces a Low Molecular Weight Reagent That Regulates Inflammatory Responses Induced by TLR Ligands. Biochem. Biophys. Res. Commun. 2019, 511, 318–322. [Google Scholar] [CrossRef] [PubMed]
- Survey and Research on the Review of the Safety of Existing Additives. In Proceedings of the Pharmaceutical Affairs and Food Sanitation Council Additives Sub-Committee, Tokyo, Japan, 24 June 2004; Japan Food Chemical Research Foundation: Osaka, Japan, 2004.
- Amakawa, M.; Nakano, T.; Kusaka, M.; Ichihara, N.; Kameko, F.; Kato, R. Evaluation of the Clinical Laboratory Data Depends on Long Oral Taking of Aureobasidium Cultured Solution. Jpn. J. PharmSci 2016, 73, 477–487. [Google Scholar]
- Suzuki, T.; Hosono, A.; Hachimura, S.; Suzuki, T.; Kaminogawa, S. Modulation of Cytokine and Immunoglobulin A Release by Beta-(1,3–1,6)–Glucan From Aureobasidium pullulans strain 1A1. In Animal Cell Technology: Basic & Applied Aspects; Iijima, S., Nishijima, K., Eds.; Springer: Dordrecht, The Netherlands, 2004; Volume 14, pp. 369–375. [Google Scholar]
- Kimura, Y.; Sumiyoshi, M.; Suzuki, T.; Sakanaka, M. Antitumor and Antimetastatic Activity of a Novel Water-Soluble Low Molecular Weight β-1,3-D-glucan (branch β-1,6) isolated from AP 1A1 strain black yeast. Anticancer Res. 2006, 26, 4131–4142. [Google Scholar]
- Kim, H.D.; Cho, H.R.; Moon, S.B.; Shin, H.D.; Yang, K.J.; Park, B.R.; Jang, H.J.; Kim, L.S.; Lee, H.S.; Ku, S.K. Effects of Beta-Glucan From Aureobasidium pullulans on Acute Inflammation in Mice. Arch. Pharm. Res. 2007, 30, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Sumiyoshi, M.; Suzuki, T.; Suzuki, T.; Sakanaka, M. Inhibitory Effects of Water-Soluble Low-Molecular-Weight Beta-(1,3–1,6) d-Glucan Purified From Aureobasidium pullulans GM-NH-1A1 Strain on Food Allergic Reactions in Mice. Int. Immunopharmacol. 2007, 7, 963–972. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Sumiyoshi, M.; Suzuki, T.; Suzuki, T.; Sakanaka, M. Effects of Water-Soluble Low-Molecular-Weight β-1,3-D-Glucan(Branch β-1,6) Isolated From Aureobasidium pullulans 1A1 Strain Black Yeast on Restraint Stress in Mice. J. Pharm. Pharmacol. 2007, 59, 1137–1144. [Google Scholar] [CrossRef]
- Shin, H.D.; Yang, K.J.; Park, B.R.; Son, C.W.; Jang, H.J.; Ku, S.K. Antiosteoporotic Effect of Polycan, Beta-Glucan From Aureobasidium, in Ovariectomized Osteoporotic Mice. Nutrition 2007, 23, 853–860. [Google Scholar] [CrossRef]
- Ikewaki, N.; Fujii, N.; Onaka, T.; Ikewaki, S.; Inoko, H. Immunological Actions of Sophy Beta-Glucan (Beta-1,3–1,6 Glucan), Currently Available Commercially as a Health Food Supplement. Microbiol. Immunol. 2007, 51, 861–873. [Google Scholar] [CrossRef]
- Tada, R.; Yoshikawa, M.; Kuge, T.; Tanioka, A.; Ishibashi, K.I.; Adachi, Y.; Tsubaki, K.; Ohno, N. A Highly Branched 1,3-beta-D-glucan Extracted From Aureobasidium pullulans Induces Cytokine Production in DBA/2 Mouse-Derived Splenocytes. Int. Immunopharmacol. 2009, 9, 1431–1436. [Google Scholar] [CrossRef]
- Tada, R.; Tanioka, A.; Ishibashi, K.I.; Adachi, Y.; Tsubaki, K.; Ohno, N. Involvement of Branched Units at position 6 in the Reactivity of a Unique Variety of Beta-D-Glucan From Aureobasidium pullulans to Antibodies in Human Sera. Biosci. Biotechnol. Biochem. 2009, 73, 908–911. [Google Scholar] [CrossRef]
- Sumiyoshi, M.; Suzuki, T.; Kimura, Y. Protective Effects of Water-Soluble Low-Molecular-Weight β-(1,3–1,6)D-Glucan Purified From Aureobasidium pullulans GM-NH-1A1 Against UFT Toxicity in Mice. J. Pharm. Pharmacol. 2009, 61, 795–800. [Google Scholar] [CrossRef]
- Kimura, Y.; Sumiyoshi, M.; Suzuki, T.; Suzuki, T.; Sakanaka, M. Effects of Water-Soluble Low-Molecular Weight b-1,3-D-glucan (Branch b-1,6) Isolated From Aureobasidium pullulans 1A1 Strain (Black Yeast) on Blood Glucose and Insulin Concentrations in Oral Glucose Tolerance Test of Mice. J. Pharm. Pharmacol. 2009, 61, 115–119. [Google Scholar] [CrossRef]
- Yoon, H.S.; Kim, J.W.; Cho, H.R.; Moon, S.B.; Shin, H.D.; Yang, K.J.; Lee, H.S.; Kwon, Y.S.; Ku, S.K. Immunomodulatory Effects of Aureobasidium pullulans SM-2001 Exopolymers on the Cyclophosphamide-Treated Mice. J. Microbiol. Biotechnol. 2010, 20, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Tada, R.; Yoshikawa, M.; Ikeda, F.; Adachi, Y.; Kato, Y.; Kuge, T.; Tanioka, A.; Ishibashi, K.I.; Tsubaki, K.; Ohno, N. Induction of IFN-γ by a Highly Branched 1,3-β-d-glucan From Aureobasidium pullulans in Mouse-Derived Splenocytes via dectin-1-independent Pathways. Biochem. Biophys. Res. Commun. 2011, 404, 1105–1110. [Google Scholar] [CrossRef] [PubMed]
- Tada, R.; Yoshikawa, M.; Kuge, T.; Tanioka, A.; Ishibashi, K.I.; Adachi, Y.; Tsubaki, K.; Ohno, N. Granulocyte Macrophage Colony-Stimulating Factor Is Required for Cytokine Induction by a Highly 6-Branched 1,3-β-D-glucan From Aureobasidium pullulans in Mouse-Derived Splenocytes. Immunopharmacol. Immunotoxicol. 2011, 33, 302–308. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Tanaka, Y.; Suzuki, T.; Mizushima, T. Protective Effect β-(1, 3→ 1, 6)-D-Glucan Against Irritant-Induced Gastric Lesions. Br. J. Nutr. 2011, 106, 475–485. [Google Scholar] [CrossRef] [PubMed]
- Muramatsu, D.; Iwai, A.; Aoki, S.; Uchiyama, H.; Kawata, K.; Nakayama, Y.; Nikawa, Y.; Kusano, K.; Okabe, M.; Miyazaki, T. β-Glucan Derived From Aureobasidium pullulans Is Effective for the Prevention of Influenza in Mice. PLoS ONE 2012, 7, e41399. [Google Scholar] [CrossRef]
- Ku, S.K.; Kim, J.W.; Cho, H.R.; Kim, K.Y.; Min, Y.H.; Park, J.H.; Kim, J.S.; Park, J.H.; Seo, B.I.; Roh, S.S. Effect of β-Glucan Originated From Aureobasidium pullulans on Asthma Induced by Ovalbumin in Mouse. Arch. Pharm. Res. 2012, 35, 1073–1081. [Google Scholar] [CrossRef]
- Uchiyama, H.; Iwai, A.; Asada, Y.; Muramatsu, D.; Aoki, S.; Kawata, K.; Kusano, K.; Nagashima, K.; Yasokawa, D.; Okabe, M.; et al. A Small Scale Study on the Effects of Oral Administration of the β-Glucan Produced by Aureobasidium pullulans on Milk Quality and Cytokine Expressions of Holstein Cows, and on Bacterial Flora in the Intestines of Japanese Black Calves. BMC Res. Notes 2012, 5, 189. [Google Scholar] [CrossRef]
- Kim, J.W.; Cho, H.R.; Ku, S.K. Efficacy Test of Polycan, a Beta-Glucan Originated From Aureobasidium pullulans SM-2001, on Anterior Cruciate Ligament Transection and Partial Medial Meniscectomy-Induced-Osteoarthritis Rats. J. Microbiol. Biotechnol. 2012, 22, 274–282. [Google Scholar] [CrossRef] [PubMed]
- Sato, H.; Kobayashi, Y.; Hattori, A.; Suzuki, T.; Shigekawa, M.; Jippo, T. Inhibitory Effects of Water-Soluble Low-Molecular-Weight β-(1,3–1,6) D-Glucan Isolated From Aureobasidium pullulans 1A1 Strain Black Yeast on Mast Cell Degranulation and Passive Cutaneous Anaphylaxis. Biosci. Biotechnol. Biochem. 2012, 76, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Tanioka, A.; Hayama, K.; Mitsuya, M.; Tansho, S.; Ono, Y.; Tsubaki, K.; Abe, S. Effect of Oral Administration of β-D-Glucan From Aureobasidium pullulans ADK-34 on Candida and MRSA Infections in Immunosuppressed Mice. Med. Mycol. J. 2012, 53, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Tanioka, A.; Tanabe, K.; Hosono, A.; Kawakami, H.; Kaminogawa, S.; Tsubaki, K.; Hachimura, S. Enhancement of Intestinal Immune Function in Mice by β-D-Glucan From Aureobasidium pullulans ADK-34. Scand. J. Immunol. 2013, 78, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Tamegai, H.; Takada, Y.; Okabe, M.; Asada, Y.; Kusano, K.; Katagiri, Y.U.; Nagahara, Y. Aureobasidium pullulans Culture Supernatant Significantly Stimulates R-848-activated Phagocytosis of PMA-Induced THP-1 Macrophages. Immunopharmacol. Immunotoxicol. 2013, 35, 455–461. [Google Scholar] [CrossRef]
- Iwai, A.; Shiozaki, T.; Miyazaki, T. Relevance of Signaling Molecules for Apoptosis Induction on Influenza A Virus Replication. Biochem. Biophys. Res. Commun. 2013, 441, 531–537. [Google Scholar] [CrossRef]
- Kim, K.H.; Park, S.J.; Lee, J.E.; Lee, Y.J.; Song, C.H.; Choi, S.H.; Ku, S.K.; Kang, S.J. Anti-Skin-Aging Benefits of Exopolymers From Aureobasidium pullulans SM2001. J. Cosmet. Sci. 2014, 65, 285–298. [Google Scholar]
- Muramatsu, D.; Kawata, K.; Aoki, S.; Uchiyama, H.; Okabe, M.; Miyazaki, T.; Kida, H.; Iwai, A. Stimulation With the Aureobasidium pullulans-Produced β-Glucan Effectively Induces Interferon Stimulated Genes in Macrophage-Like Cell Lines. Sci. Rep. 2014, 4, 4777. [Google Scholar] [CrossRef]
- Aoki, S.; Iwai, A.; Kawata, K.; Muramatsu, D.; Uchiyama, H.; Okabe, M.; Ikesue, M.; Maeda, N.; Uede, T. Oral Administration of the β-Glucan Produced by Aureobasidium pullulans Ameliorates Development of Atherosclerosis in Apolipoprotein E Deficient Mice. J. Funct. Foods 2015, 18, 22–27. [Google Scholar] [CrossRef]
- Oboshi, W.; Amakawa, M.; Kato, R. Effects of β-Glucan and Lactic Acid Bacteria on Gut Immune System. Jpn. J. Med. Technol. 2014, 63, 673–679. [Google Scholar]
- Ganesh, J.S.; Rao, Y.Y.; Ravikumar, R.; Jayakrishnan, G.A.; Iwasaki, M.; Preethy, S.; Abraham, S.J. Beneficial Effects of Black Yeast Derived 1–3, 1–6 Beta Glucan-Nichi Glucan in a Dyslipidemic Individual of Indian Origin—A Case Report. J. Diet. Suppl. 2014, 11, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Park, S.J.; Lee, Y.J.; Lee, J.E.; Song, C.H.; Choi, S.H.; Ku, S.K.; Kang, S.J. Inhibition of UVB-Induced Skin Damage by Exopolymers From Aureobasidium pullulans SM-2001 in Hairless Mice. Basic Clin. Pharmacol. Toxicol. 2015, 116, 73–86. [Google Scholar] [CrossRef] [PubMed]
- Kawata, K.; Iwai, A.; Muramatsu, D.; Aoki, S.; Uchiyama, H.; Okabe, M.; Hayakawa, S.; Takaoka, A.; Miyazaki, T. Stimulation of Macrophages With the β-Glucan Produced by Aureobasidium pullulans Promotes the Secretion of Tumor Necrosis Factor-Related Apoptosis Inducing Ligand (TRAIL). PLoS ONE 2015, 10, e0124809. [Google Scholar] [CrossRef] [PubMed]
- Lai, L.P.; Lee, M.T.; Chen, C.S.; Yu, B.; Lee, T.T. Effects of Co-Fermented Pleurotus eryngii Stalk Residues and Soybean Hulls by Aureobasidium pullulans on Performance and Intestinal Morphology in Broiler Chickens. Poult. Sci. 2015, 94, 2959–2969. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Aizawa, Y.; Hiramoto, K.; Kasahara, E.; Tsuruta, D.; Suzuki, T.; Ikeda, A.; Azuma, H.; Nagasaki, T. Anti-Inflammatory Effect of Water-Soluble Complex of 1′-Acetoxychavicol Acetate With Highly Branched β-1,3-Glucan on Contact Dermatitis. Biomed. Pharmacother. 2015, 69, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Aoki, S.; Iwai, A.; Kawata, K.; Muramatsu, D.; Uchiyama, H.; Okabe, M.; Ikesue, M.; Maeda, N.; Uede, T. Oral Administration of the Aureobasidium pullulans-Derived β-Glucan Effectively Prevents the Development of High Fat Diet-Induced Fatty Liver in Mice. Sci. Rep. 2015, 5, 10457. [Google Scholar] [CrossRef]
- Jippo, T.; Suzuki, T.; Sato, H.; Kobayashi, Y.; Shigekawa, M. Water Soluble Low Molecular Weight β -(1, 3 1, 6)D-Glucan Inhibit Cedar Pollinosis. Funct. Foods Health Dis. 2015, 5, 80–88. [Google Scholar] [CrossRef]
- Iinuma, K. Case Report: A Patient Was Completely Recovered From stage 3 of Colorectal Cancer and 2 Sites of Liver Metastatic Cancer by Anti-Cancer Agents and β-Glucan EX After Resection of Colorectal Cancer. New Food Indust. 2016, 58, 29–34. [Google Scholar]
- Yamamoto, K.; Yamauchi, Y.; Kusano, K.; Iinuma, K. Healing of Third-Degree Moderate-Temperature Burn Promoted Containing β -Glucan: A Case Report. Jpn. J. PharmSci 2017, 74, 697–703. [Google Scholar]
- Ikeda, A.; Akiyama, M.; Sugikawa, K.; Koumoto, K.; Kagoshima, Y.; Li, J.; Suzuki, T.; Nagasaki, T. Formation of β-(1,3–1,6)-D-Glucan-Complexed Fullerene and Its Photodynamic Activity Towards Macrophages. Org. Biomol. Chem. 2017, 15, 1990–1997. [Google Scholar] [CrossRef]
- Brown, G.D.; Herre, J.; Williams, D.L.; Willment, J.A.; Marshall, A.S.J.; Gordon, S. Dectin-1 Mediates the Biological Effects of β-Glucans. J. Exp. Med. 2003, 197, 1119–1124. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.S.; Jeong, H.S.; Kim, I.Y.; Jung, G.W.; Ku, B.H.; Park, D.C.; Moon, S.B.; Cho, H.R.; Bashir, K.M.I.; Ku, S.K.; et al. Anti-Osteoporotic Effects of Mixed Compositions of Extracellular Polymers Isolated From Aureobasidium pullulans and Textoria Morbifera in Ovariectomized Mice. BMC Complement. Altern. Med. 2018, 18, 295. [Google Scholar] [CrossRef] [PubMed]
- Fujikura, D.; Muramatsu, D.; Toyomane, K.; Chiba, S.; Daito, T.; Iwai, A.; Kouwaki, T.; Okamoto, M.; Higashi, H.; Kida, H.; et al. Aureobasidium pullulans-Cultured Fluid Induces IL-18 Production, Leading to Th1-Polarization During Influenza A Virus Infection. J. Biochem. 2018, 163, 31–38. [Google Scholar] [CrossRef]
- Lim, J.M.; Lee, Y.J.; Cho, H.R.; Park, D.C.; Jung, G.W.; Ku, S.K.; Choi, J.S. Extracellular Polysaccharides Purified from Aureobasidium Pullulans SM‑2001 (Polycan) Inhibit Dexamethasone‑Induced Muscle Atrophy in Mice. Int. J. Mol. Med. 2018, 41, 1245–1264. [Google Scholar] [CrossRef] [PubMed]
- Hino, S.; Funada, R.; Sugikawa, K.; Koumoto, K.; Suzuki, T.; Nagasaki, T.; Ikeda, A. Turn-On Fluorescence and Photodynamic Activity of β-(1,3–1,6)-D-Glucan-Complexed Porphyrin Derivatives Inside HeLa Cells. Photochem. Photobiol. Sci. 2019, 18, 2854–2858. [Google Scholar] [CrossRef]
- Tsuji, N.M.; Kosaka, A. Oral Tolerance: Intestinal Homeostasis and Antigen-Specific Regulatory T Cells. Trends Immunol. 2008, 29, 532–540. [Google Scholar] [CrossRef]
- Tang, C.; Kakuta, S.; Shimizu, K.; Kadoki, M.; Kamiya, T.; Shimazu, T.; Kubo, S.; Saijo, S.; Ishigame, H.; Nakae, S.; et al. Suppression of IL-17F, but not of IL-17A, provides protection against colitis by inducing Treg cells through modification of the intestinal microbiota. Nat. Immunol 2018, 19, 755–765. [Google Scholar] [CrossRef]
- Ross, G.D.; Cain, J.A.; Lachmann, P.J. Membrane Complement Receptor Type Three (CR3) Has Lectin-Like Properties Analogous to Bovine Conglutinin as Functions as a Receptor for Zymosan and Rabbit Erythrocytes as Well as a Receptor for iC3b. J. Immunol. 1985, 134, 3307–3315. [Google Scholar]
- Vetvicka, V.; Thornton, B.P.; Ross, G.D. Soluble Beta-Glucan Polysaccharide Binding to the Lectin Site of Neutrophil or Natural Killer Cell Complement Receptor type 3 (CD11b/CD18) Generates a Primed State of the Receptor Capable of Mediating Cytotoxicity of iC3b- Opsonized Target Cells. J. Clin. Investig. 1996, 98, 50–61. [Google Scholar] [CrossRef]
- Brown, G.D.; Gordon, S. Immune Recognition. A New Receptor for Beta-Glucans. Nature 2001, 413, 36–37. [Google Scholar] [CrossRef]
- Ohno, N. Analysis of Glycans; Polysaccharide Functional Properties. Compr. Glycosci. 2007, 2, 559–577. [Google Scholar]
- Hardy, G. Nutraceuticals and Functional Foods: Introduction and Meaning. Nutrition 2000, 16, 688–689. [Google Scholar] [CrossRef]
- Roberfroid, M.B. Global View on Functional Foods: European Perspectives. Br. J. Nutr. 2002, 88 (Suppl. 2), S133–S138. [Google Scholar] [CrossRef] [PubMed]
| Type of β-glucan Structure | Natural Source and Trivial Name of β-glucan |
|---|---|
| 1,3-β-glucan (linear unbranched, homogeneous) | bacterium Alcaligenes faecalis, curdlan algae Euglena gracilis, paramylon fungus Poria cocos, pachyman grape Vitis vinifera, callose |
| 1,3-1,6-β-glucan (linear with 1,6-linked β-glucosyl side branches) | algae Laminaria sp. laminarin (unbranched) algae Eisenia bicyclis, laminarin (some branched) fungus Claviceps purpuria, cell wall glucan fungus Sclerotinia sclerotiorum, cell wall or extracelluar glucan fungus (black yeast) Aureobasidium pullulas, extracellular glucan fungus/mushroom schizophyllum commune,extracelluar or cell wall glucan mushroom Grifola frondosa, cell wall glucan mushroom Lentinula edodes, cell wall glucan |
| 1,3-1,6-β-glucan (branch on branch structure) | yeast Saccharomyces cerevisiae, cell wall glucan yeast Candida albicans, cell wall glucan yeast Candia utilis, cell wall glucan |
| 1,3-1,4-β-glucan (linear) | cereal β-glucan, such as barley, oat, wheat, and rye Iceland moss Centraria islandia, lichenin |
| 1,3-1,4-β-glucan (linear with 1,4-lincked β-glucosyl side branches) | oyster mushroom Pleurotus ostreatus, cell wall glucan |
| No. | Author, Year, Reference 1 | Item of Chracterization or Healthy Function | Outline of Functionality and Efficacy |
|---|---|---|---|
| 1 | Arkadjeva 1969 [13] | Single cell protein | The first information on bioactive polysaccharide from A. pullulans. |
| 2 | Han et al., 1976 [14] | Intestinal immunity | Production of single-cell protein from cellulosic wastes using A. pullulans. A. pullulans cells were not toxic, and the values of their feed intake, weight gain, and protein efficiency ratio were superior to those of other cells. |
| 3 | T. Suzuki et al., 2004 [25] | Intestinal immunity | Intestinal immunostimulatory and modulatory effects. Cellular-level in vitro experiments using mouse lymphocytes from Peyer’s patch showed that immunoglobulin A was produced at APβG concentrations ranging from 0 to 200 μg/mL in a dose-dependent manner. |
| 4 | Kimura et al., 2006 [26] | Antitumor | Antitumor and antimetastatic actions in mice. The antitumor and antimetastatic actions of APβG may be involve in the enhancement of intestinal immune functions through the increases in NK- and IFN-gamma-positive cell numbers. |
| 5 | Kim et al., 2007 [27] | Reduction of inflamamatory | Reduction of the acute inflammatory responses induced by xylene application in mice. Xylene-induced acute inflammatory changes were significantly and dose-dependently decreased by beta-glucan treatment (up to 250 mg/kg). |
| 6 | Kimura et al., 2007 [28] | Anti food allergy | The anti-food allergic action of beta-glucan may be caused by the induction of IFN-g production in the small intestine and splenocytes. APβG diets (0.5–1%) significantly inhibited not only the OVA-specific IgE elevation but also reduced the production of IFN-g and the number of CD8- and IFN-gamma-positive cells from the splenocytes and in the small intestine, respectively. |
| 7 | Kimura et al., 2007 [29] | Anti stress | Inhibitory actions of APβG (100 mg/kg) on the increase in corticosterone level and reduction of NK activity induced by restraint stress. These effects may be associated with the abrogation of interleukin-6 (IL-6) and IL-12. |
| 8 | Shin et al., 2007 [30] | Reduction of osteoporosis | APβG exhibited favorable effects on ovariectomy-induced osteoporosis. It significantly and dose-dependently suppressed the decrease in bone weight, bone mineral content, failure load, bone mineral density, and serum calcium and phosphorus levels and the increase in serum osteocalcin levels. |
| 9 | Ikewaki et al., 2007 [31] | Immunomodulatory mechanism | APβG may have unique immune regulatory or enhancing properties. It stimulated the production of interleukin-8 (IL-8) or soluble Fas (sFas); however, it did not stimulate that of IL-1beta, IL-2, IL-6, IL-12 (p70 + 40), IFN-g, TNF-a, or soluble Fas ligand (sFasL), in either cultured PBMCs or cells of the human monocyte-like cell line U937. |
| 10 | Tada et al., 2008 [20] | Structure information | The primary structure of APβG and its biological activities were determined and evaluated, respectively using NMR spectroscopy. The structure comprises a mixture of a 1-3-beta-d-glucan backbone with single 1-6-beta-d-glucopyranosyl side-branching units in every two residues (major structure) and a 1-3-beta-d-glucan backbone with single 1-6-beta-d-glucopyranosyl side-branching units in every three residues (minor structure). |
| 11 | Tada et al., 2009 [32] | Immunomodulation mechanism | Immunomodulatory effect of APβG on DBA/2 mouse-derived splenocytes in vitro. APβG strongly induced the production of various cytokines, especially Th1 cytokines (e.g., IFN-g and IL-12p70) and Th17 cytokines (e.g., IL-17A), but did not induce the production of IL-4, IL-10, and TNF-a on splenocytes in vitro. |
| 12 | Tada et al., 2009 [33] | Immunomodulatory mechanism | This is the first study in which the branched chains at position 6 of beta-D-glucan strongly contribute to its recognition by antibodies in human sera. APβG reacted to IgG antibodies in human sera and the IgGs recognize branched chains at position 6. |
| 13 | Kimura et al., 2009 [34] | Protection of intestine | Protective effects of APβG (50 and 100 mg/kg twice daily) against the toxicity of UFT (combination of tegafur (1-(2-tetrahydrofuryl)-5-fluorouracil) and uracil) in mice bearing colon 26 tumors. Histological analysis showed that the damage found in the villi of the small-intestine by UFT was inhibited by the orally administered beta-glucan. |
| 14 | Sumiyoshi et al., 2009 [35] | Reduction and control of blood glucose | Reduction and control of blood glucose level in mice. In the 100 mg/kg and 200 mg/kg APβG dose groups, the increase in blood glucose from 15 to 30 min after glucose administration was minimal, after which the blood glucose level decreased, and significantly decreased 60 min after administration. |
| 15 | Yoon et al., 2010 [36] | Immunomodulatory mechanism | Immunomodulatory effects of exopolymers of A. pullulans containing APβG, which were orally administered at 10 mL/kg, were evaluated on cyclophosphamide (CPA)-treated mice. APβG can be effectively used to prevent an immunosuppress mediated (at least partially) and the recruitment of T cells and TNF-a-positive cells or enhancement of their activity. |
| 16 | Tada et al., 2011 [37] | Immunomodulatory mechanism | Immunomodulatory production of various cytokines in DBA/2 mouse-derived splenocytes in vitro was found via dectin-1-independent pathways. The production of IFN-γ in DBA/2 mouse-derived splenocytes by APβG was not inhibited following a treatment with an anti-dectin-1 neutralizing antibody. |
| 17 | Tada et al., 2011 [38] | Immunomodulatory mechanism | The induction of cytokines by APβG was dependent on the existence of a granulocyte macrophage colony-stimulating factor (GM-CSF). GM-CSF is indispensable for the induction of cytokines by APβG in mouse-derived splenocytes, similar to a typical 1,3-β-d-glucan from Sparassis crispa (SCG). |
| 18 | Tanaka et al., 2011 [39] | Reduction of ulcer | Oral administration of APβG (>100 mg/kg) ameliorated the gastric lesions induced by ethanol (EtOH) or HCl in mice. The administration of APβG also suppressed EtOH-induced inflammatory responses through the protection of the gastric mucosa from the formation of irritant-induced lesions by increasing the levels of defensive factors, such as HSP70 and mucin. |
| 19 | Muramatsu et al., 2012 [40] | Anti virus | Oral administration of A. pullulans-cultured fluid enriched with APβG exhibits efficacy in protecting mice infected with a lethal titer of the A/Puerto Rico/8/34 (PR8; H1N1) strain of the influenza virus. |
| 20 | Ku et al., 2012 [41] | Anti allergy | The effect of APβG, orally administered at 125 mg/kg, on ovalbumin (OVA)-induced allergic asthma was found in OVA-inducing asthmatic mice. The increase in body weight after OVA aerosol challenge, lung weight, total leukocytes and eosinophils in peripheral blood, total cell numbers, and neutrophil and eosinophils in BALF were detected in the OVA control compared to sham control (non-OVA). |
| 21 | Uchiyama et al., 2012 [42] | Microbiome | The effects of oral administration on bacterial flora in the intestines of domestic animals, using Holstein cows and newborn Japanese Black calves, were observed. The expressions of TNF-α and interleukin (IL)-6 in all cows became slightly lower and the bacterial flora were tendentiously changed. |
| 22 | Kim et al., 2012 [43] | Reduction of osteoarthritis | Osteoarthritis (OA) was effectively induced by anterior cruciate ligament transection and partial medial meniscectomy (ACLT&PMM) by APβG (42.5mg/kg). |
| 23 | Sato et al., 2012 [44] | Anti allergy | Effective therapeutic treatment of allergic diseases, inhibition of mast cell degranulation, and passive cutaneous anaphylaxis (PCA) were shown. APβG (100 to 150 mg/kg) dose-dependently inhibited the degranulation of both rat basophilic leukemia (RBL-2H3) and cultured mast cells (CMCs) activated by calcium ionophore A23187 or IgE. |
| 24 | Tanioka et al., 2012 [45] | Anti microorganism | Positive effect of oral administration of APβG on Candida albicans or methicillin-resistant Staphylococcus aureus (MRSA) infection in immunosuppressed mice fed 2.5% APβG diet for 14–30 days. |
| 25 | Tanioka et al., 2013 [46] | Immunomodulatory mechanism | APβG had effects on intestinal immune systems by Peyer’s patch (PP) cells and interleukin (IL)-5, IL-6, and IgA production in culture media. The production of IL-6 and IgA by PP cells and that of IL-6 by PP dendritic cells (PPDCs) in APβG-fed and cyclophosphamide (CY)-treated mice also increased. |
| 26 | Tamegai et al., 2013 [47] | Immunomodulatory mechanism | Activation of several distinct innate immune receptor signaling pathways enhances the immune response induced by R-848, indicating non-influenza antiviral efficacy. The expression of TNF-a and IL-12p40 was significantly enhanced when co-stimulated with culture supernatants of R-848 and APβG compared with the culture supernatant of R-848 alone. |
| 27 | Iwai et al., 2013 [48] | Anti virus | Antiviral effects of the expression of interferon-inducible genes, through the induction of interferon, and the enhancement of the transcriptional activity of STAT1 were observed. |
| 28 | Kim et al., 2014 [49] | Anti oxidant | Several antioxidants may serve as a functional ingredient in cosmetic products by reducing hyaluronidase, elastase, collagenase, and MMP-1 activities and inhibiting melanin production and tyrosinase activities. |
| 29 | Muramatsu et al., 2014 [50] | Immunomodulatory mechanism | Stimulation with APβG effectively induces the interferon (IFN) stimulated genes (ISGs) in macrophage-like cell lines through the induction of IFN and the enhancement of STAT1-mediated transcriptional activation. |
| 30 | Ganesh et al., 2014 [51] | Reduction of triglyceride and cholesterol, in human | In a clinical study, one male was orally administered 1.5 mg of APβG for two months, and his triglyceride, VLDL, and HDL cholesterol levels decreased from 523 mg/dL to 175 mg/dL, 104.6 mg/dL to 35 mg/dL, and 27 mg/dL to 38 mg/dL, respectively. |
| 31 | Oboshi et al., 2014 [52] | Immunomodulatory mechanism | When AGβP-containing foods were orally administered to mice (BALB/c six-week old females), an increase in the titer of antibodies in the blood and the phagocytic capacity of blood macrophages were observed. |
| 32 | Aoki et al., 2015 [53] | Reduction of triglyceride and cholesterol | Oral administration of APβG is effective in preventing the development of high-fat diet (HFD)-induced fatty liver in mice. After 16 weeks of oral administration of APβG, serological analysis showed that HFD-induced high blood cholesterol and triglyceride levels were reduced by the oral administration of APβG. HFD induced-fatty liver was also significantly reduced. |
| 33 | Kim et al., 2015 [54] | Reduction osteoporosis | The effects of purified exopolymers from APβG were evaluated in UVB-induced hairless mice. E-AP-SM2001 consists of 1.7% β-1,3/1,6-glucan, fibrous polysaccharides, and other organic materials, such as amino acids and mono- and di-unsaturated fatty acids (linoleic and linolenic acids), and shows anti-osteoporotic and immunomodulatory effects through antioxidant and anti-inflammatory mechanisms. |
| 34 | Kawata et al., 2015 [55] | Anti-tumor | Mechanism of anti-tumor activities in mice have been demonstrated. Stimulation with APβG induces TRAIL expression in mouse and human macrophage-like cell lines. TRAIL is known to be a cytokine that specifically induces apoptosis in transformed cells, but not in untransformed cells. |
| 35 | Lai et al., 2015 [56] | Improvement of intestine morphology | The effects of co-fermented Pleurotus eryngii stalk residues (PESR) and soybean hulls with A. pullulans on performance and intestinal morphology of broiler chickens significantly increased the ratio of lactic acid bacteria to Clostridium perfringens in ceca, ileum villus height, and jejunum villus height/crypt depth ratio of 35-day old birds. |
| 36 | Li et al., 2015 [57] | Anti inflammatory effect | Anti-inflammatory effect of water solubilized 1′-Acetoxychavicol acetate (ACA) on contact dermatitis by complexation with β-1,3-glucan isolated form A. pullulans black yeast was reported. |
| 37 | Aoki et al., 2015 [58] | Reduction of atherosclerosis | The effect of oral administration of APβG on high-fat diet (HFD)-induced atherosclerosis was evaluated using apolipoprotein E deficient mice, a widely used mouse model for atherosclerosis. HFD-induced atherosclerosis was significantly reduced in the APβG-treated mice. |
| 38 | Jippo et al., 2015 [59] | Anti allergy in human | A clinical study, comprising a randomized, single blind, placebo-controlled, and parallel group, was performed in 65 subjects (aged 22 to 62). All subjects consumed one bottle of placebo or beta glucan (150 mg) daily and their allergic symptoms were recorded in a diary. APβG group had a significantly lower prevalence of sneezing, nose blowing, tears, and hindrance to the activities of daily living. |
| 39 | Iinuma et al., 2016 [60] | Anti colongenomic cancer | APβG was orally administered in combination with anticancer drugs, such as Avastin, Elplat, Levofolinate and fluorouracil, to treat for stage III colon cancer patients, on which two sites of cologenomic cancer were transferred, and remarkable cancer elimination was reported in the all cases. |
| 40 | Hirabayashi et al., 2016 [21] | Structural characterization of by hydrothermal treatment | The chemical structure of hydrothermally treated APβG was characterized using techniques such as gas chromatography/mass spectrometry (GC/MS) and nuclear magnetic resonance (NMR). It became water soluble, with an average molecular weight of 128,000 and was completely hydrolyzed to glucose by enzymatic reaction. Gentiobiose and glucose were released as products during the reaction with the maximum yield approximately 70% (w/w) of gentiobiose. |
| 41 | Yamamoto et al., 2017 [61] | Wound healimg | Muscle transplantation was avoided, and rapid healing was observed when there was external application of APβG in the case of third degree deep low temperature burn. |
| 42 | Ikeda et al., 2017 [62] | Inclusion of [63] Fullerene | [63] Fullerene was dissolved in water via complexation with β-1,3-glucan using a mechanochemical highspeed vibration milling apparatus. The photodynamic activity of APβG-complexed C70 was highly dependent on the expression level of dectin-1 on the cell surfaces of macrophages. The photodynamic activity increased owing to the synergistic effect between β-1,3-glucan-complexed 1′-acetoxychavicol acetate and the C70 complex. |
| 43 | Cho et al., 2018 [64] | Anti osteoporotic | The synergistic anti-osteoporotic potential of mixtures containing different proportions of APβG and TM compared with that of single formulations of each herbal extract using bilateral ovariectomized (OVX) mice. The EAP:TM (3:1) formulation synergistically enhanced the anti-osteoporotic potential of individual EAP or TM formulations, possibly owing to the enhanced variety of active ingredients. |
| 44 | Fujikura et al., 2018 [65] | Anti virus | APβG exhibits adjuvant activity and renders mice to be resistant to influenza A virus infection. Intraperitoneal administration of APβG increased the serum level of IL-18 and the number of splenic IFN-γ producing CD4+ cells during influenza A virus infection. The adjuvant effect of APβG was distinct from that of alum. In addition, AP-CF injection barely increased the number of peritoneal neutrophils and inflammatory macrophages. |
| 45 | Lim et al., 2018 [66] | Muscle preserving effect | The beneficial skeletal muscle‑preserving effects of extracellular polysaccharides from APβG on dexamethasone (DEXA)‑induced catabolic muscle atrophy in mice. APβG at 400 mg/kg exhibited favorable muscle protective effects against DEXA‑induced catabolic muscle atrophy; the effects are comparable with those of oxymetholone (50 mg/kg). |
| 46 | Hayashi et al., 2019 [22] | Anti inflammatory effect | Inflammatory immunostimulatory action of nitrogen oxide (NO) synthesis and TNF a production in RAW264.7 cells induced by Lipopolysaccharide (LPS) were suppressed by APβG with lower molecular weight less than 10,000. |
| 47 | Hino et al., 2019 [67] | Inclusion of porphyrin | Fluorescence intensities of water-soluble APβG-complexed porphyrin derivatives were very weak owing to self-quenching. However, APβG-complexed tetra (aminophenyl) porphyrin exhibited ‘off-state’ to ‘on-state’ fluorescence switching activity via intracellular uptake. Furthermore, the internalized complex showed a high level of photodynamic activity toward HeLa cells under photoirradiation at long wavelengths. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suzuki, T.; Kusano, K.; Kondo, N.; Nishikawa, K.; Kuge, T.; Ohno, N. Biological Activity of High-Purity β-1,3-1,6-Glucan Derived from the Black Yeast Aureobasidium pullulans: A Literature Review. Nutrients 2021, 13, 242. https://doi.org/10.3390/nu13010242
Suzuki T, Kusano K, Kondo N, Nishikawa K, Kuge T, Ohno N. Biological Activity of High-Purity β-1,3-1,6-Glucan Derived from the Black Yeast Aureobasidium pullulans: A Literature Review. Nutrients. 2021; 13(1):242. https://doi.org/10.3390/nu13010242
Chicago/Turabian StyleSuzuki, Toshio, Kisato Kusano, Nobuhiro Kondo, Kouji Nishikawa, Takao Kuge, and Naohito Ohno. 2021. "Biological Activity of High-Purity β-1,3-1,6-Glucan Derived from the Black Yeast Aureobasidium pullulans: A Literature Review" Nutrients 13, no. 1: 242. https://doi.org/10.3390/nu13010242
APA StyleSuzuki, T., Kusano, K., Kondo, N., Nishikawa, K., Kuge, T., & Ohno, N. (2021). Biological Activity of High-Purity β-1,3-1,6-Glucan Derived from the Black Yeast Aureobasidium pullulans: A Literature Review. Nutrients, 13(1), 242. https://doi.org/10.3390/nu13010242