Current Evidence to Propose Different Food Supplements for Weight Loss: A Comprehensive Review
Abstract
1. Introduction
2. Methods
- -
- High if based on one or more updated, high-quality systematic reviews based on at least two high quality primary studies with consistent results;
- -
- Moderate, if based on one or more updated systematic reviews of high or moderate quality based on at least one high-quality primary study or two primary studies of moderate quality with consistent results;
- -
- Low, if based on a limited number of clinical studies or one or more systematic reviews of variable quality based on primary studies of moderate quality with inconsistent results.
3. Results
3.1. Reduced Nutrients Absorption as Purported Mechanisms of Action
3.1.1. Green Tea
3.1.2. Ginseng
3.1.3. White Kidney Bean
3.1.4. Chitosan
3.1.5. β-Glucans
3.1.6. Psyllium
3.1.7. Glucomannan
3.1.8. Guar Gum
3.1.9. Agar
3.1.10. Inulin and Inulin Type Fructans
3.2. Reduced Appetite as Purported Mechanisms of Action
3.2.1. Caralluma
3.2.2. Spirulina
3.2.3. Whey Protein
3.2.4. Coffee, Caffeine and Chlorogenic Acids
3.2.5. Bitter Orange
3.2.6. Guarana
3.3. Increased Energy Expenditure as Purported Mechanisms of Action
3.3.1. Capsaicin, Capsaicinoids and Capsinoids
3.3.2. Curcumin
3.3.3. L-Carnitine
3.4. Improved Fat Metabolism as Purported Mechanisms of Action
3.4.1. Pyruvate
3.4.2. Diacylglycerol
3.4.3. Licorice
3.4.4. Garcinia Cambogia
3.4.5. Resveratrol
3.4.6. Conjugated Linoleic Acid
3.4.7. Aloe Vera
3.4.8. Flaxseed
3.4.9. Grapefruit
3.5. Carbohydrate Metabolism Improvement as Purported Mechanisms of Action
3.5.1. Mangosteen
3.5.2. Chromium
3.5.3. Lipoic Acid
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Obesity and Overweight Factsheet. Available online: http://www.who.int/mediacentre/factsheets/fs311/en/ (accessed on 8 April 2017).
- Watanabe, M.; Risi, R.; De Giorgi, F.; Tuccinardi, D.; Mariani, S.; Basciani, S.; Lubrano, C.; Lenzi, A.; Gnessi, L. Obesity treatment within the Italian national healthcare system tertiary care centers: What can we learn? Eat. Weight Disord. 2020. [Google Scholar] [CrossRef] [PubMed]
- Mariani, S.; Fiore, D.; Varone, L.; Basciani, S.; Persichetti, A.; Watanabe, M.; Saponara, M.; Spera, G.; Moretti, C.; Gnessi, L. Obstructive sleep apnea and bone mineral density in obese patients. Diabetes Metab. Syndr. Obes. Targets Ther. 2012, 5, 395–401. [Google Scholar] [CrossRef]
- Lonardo, A.; Mantovani, A.; Lugari, S.; Targher, G. Epidemiology and pathophysiology of the association between NAFLD and metabolically healthy or metabolically unhealthy obesity. Ann. Hepatol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, A.K.; Dubey, P.; Cistola, D.P.; Reddy, S.Y. Association Between Obesity and Cardiovascular Outcomes: Updated Evidence from Meta-analysis Studies. Curr. Cardiol. Rep. 2020, 22, 25. [Google Scholar] [CrossRef] [PubMed]
- Okamura, T.; Hashimoto, Y.; Hamaguchi, M.; Obora, A.; Kojima, T.; Fukui, M. Ectopic fat obesity presents the greatest risk for incident type 2 diabetes: A population-based longitudinal study. Int. J. Obes. 2019, 43, 139–148. [Google Scholar] [CrossRef]
- Palermo, A.; Tuccinardi, D.; Defeudis, G.; Watanabe, M.; D’Onofrio, L.; Lauria, A.P.; Napoli, N.; Pozzilli, P.; Manfrini, S. BMI and BMD: The potential interplay between obesity and bone fragility. Int. J. Environ. Res. Public Health 2016, 13, 544. [Google Scholar] [CrossRef]
- Arboleda, S.; Vargas, M.; Losada, S.; Pinto, A. Review of obesity and periodontitis: An epidemiological view. Br. Dent. J. 2019, 227, 235–239. [Google Scholar] [CrossRef]
- Basciani, S.; Camajani, E.; Contini, S.; Persichetti, A.; Risi, R.; Bertoldi, L.; Strigari, L.; Prossomariti, G.; Watanabe, M.; Mariani, S.; et al. Very-Low-Calorie Ketogenic Diets with Whey, Vegetable or Animal Protein in Patients with Obesity: A Randomized Pilot Study. J. Clin. Endocrinol. Metab. 2020. [Google Scholar] [CrossRef]
- Basciani, S.; Costantini, D.; Contini, S.; Persichetti, A.; Watanabe, M.; Mariani, S.; Lubrano, C.; Spera, G.; Lenzi, A.; Gnessi, L. Safety and efficacy of a multiphase dietetic protocol with meal replacements including a step with very low calorie diet. Endocrine 2015, 48, 863–870. [Google Scholar] [CrossRef]
- Farr, O.M.; Tuccinardi, D.; Upadhyay, J.; Oussaada, S.M.; Mantzoros, C.S. Walnut consumption increases activation of the insula to highly desirable food cues: A randomized, double-blind, placebo-controlled, cross-over fMRI study. Diabetes Obes. Metab. 2018, 20, 173–177. [Google Scholar] [CrossRef]
- Filippou, C.D.; Tsioufis, C.P.; Thomopoulos, C.G.; Mihas, C.C.; Dimitriadis, K.S.; Sotiropoulou, L.I.; Chrysochoou, C.A.; Nihoyannopoulos, P.I.; Tousoulis, D.M. Dietary Approaches to Stop Hypertension (DASH) Diet and Blood Pressure Reduction in Adults with and without Hypertension: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Adv. Nutr. 2020. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Tozzi, R.; Risi, R.; Tuccinardi, D.; Mariani, S.; Basciani, S.; Spera, G.; Lubrano, C.; Gnessi, L. Beneficial effects of the ketogenic diet on nonalcoholic fatty liver disease: A comprehensive review of the literature. Obes. Rev. 2020. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Risi, R.; Camajani, E.; Contini, S.; Persichetti, A.; Tuccinardi, D.; Ernesti, I.; Mariani, S.; Lubrano, C.; Genco, A.; et al. Baseline HOMA IR and Circulating FGF21 Levels Predict NAFLD Improvement in Patients Undergoing a Low Carbohydrate Dietary Intervention for Weight Loss: A Prospective Observational Pilot Study. Nutrients 2020, 12, 2141. [Google Scholar] [CrossRef] [PubMed]
- Tuccinardi, D.; Farr, O.M.; Upadhyay, J.; Oussaada, S.M.; Klapa, M.I.; Candela, M.; Rampelli, S.; Lehoux, S.; Lazaro, I.; Sala-Vila, A.; et al. Mechanisms underlying the cardiometabolic protective effect of walnut consumption in obese people: A cross-over, randomized, double-blind, controlled inpatient physiology study. Diabetes Obes. Metab. 2019, 21, 2086–2095. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, G.; Apovian, C.M. Current pharmacotherapy for obesity. Nat. Rev. Endocrinol. 2018, 14, 12–24. [Google Scholar] [CrossRef] [PubMed]
- Tuccinardi, D.; Farr, O.M.; Upadhyay, J.; Oussaada, S.M.; Mathew, H.; Paschou, S.A.; Perakakis, N.; Koniaris, A.; Kelesidis, T.; Mantzoros, C.S. Lorcaserin treatment decreases body weight and reduces cardiometabolic risk factors in obese adults: A six-month, randomized, placebo-controlled, double-blind clinical trial. Diabetes Obes. Metab. 2019, 21, 1487–1492. [Google Scholar] [CrossRef] [PubMed]
- Wiklund, P. The role of physical activity and exercise in obesity and weight management: Time for critical appraisal. J. Sport Health Sci. 2016, 5, 151–154. [Google Scholar] [CrossRef] [PubMed]
- Castelnuovo, G.; Pietrabissa, G.; Manzoni, G.M.; Cattivelli, R.; Rossi, A.; Novelli, M.; Varallo, G.; Molinari, E. Cognitive behavioral therapy to aid weight loss in obese patients: Current perspectives. Psychol. Res. Behav. Manag. 2017, 10, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Bruci, A.; Tuccinardi, D.; Tozzi, R.; Balena, A.; Santucci, S.; Frontani, R.; Mariani, S.; Basciani, S.; Spera, G.; Gnessi, L.; et al. Very Low-Calorie Ketogenic Diet: A Safe and Effective Tool for Weight Loss in Patients With Obesity and Mild Kidney Failure. Nutrients 2020, 12, 333. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Tuccinardi, D.; Ernesti, I.; Basciani, S.; Mariani, S.; Genco, A.; Manfrini, S.; Lubrano, C.; Gnessi, L. Scientific evidence underlying contraindications to the ketogenic diet: An update. Obes. Rev. 2020. [Google Scholar] [CrossRef]
- FDA Requests the Withdrawal of the Weight-Loss Drug Belviq, Belviq XR (Lorcaserin) from the Market. Available online: https://www.fda.gov/drugs/drug-safety-and-availability/fda-requests-withdrawal-weight-loss-drug-belviq-belviq-xr-lorcaserin-market (accessed on 17 June 2020).
- Brancheau, D.; Patel, B.; Zughaib, M. Do cinnamon supplements cause acute hepatitis? Am. J. Case Rep. 2015, 16, 250–254. [Google Scholar] [CrossRef] [PubMed]
- Brozek, J.L.; Akl, E.A.; Alonso-Coello, P.; Lang, D.; Jaeschke, R.; Williams, J.W.; Phillips, B.; Lelgemann, M.; Lethaby, A.; Bousquet, J.; et al. Grading quality of evidence and strength of recommendations in clinical practice guidelines. Part 1 of 3. An overview of the GRADE approach and grading quality of evidence about interventions. Allergy 2009, 64, 669–677. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Hong, J. Prevention of chronic diseases by tea: Possible mechanisms and human relevance. Annu. Rev. Nutr. 2013, 33, 161–181. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, W.; Hou, L.; Wu, H.; Wu, Y.; Xu, R.; Xiao, Y.; Wang, X. Does tea extract supplementation benefit metabolic syndrome and obesity? A systematic review and meta-analysis. Clin. Nutr. 2020, 39, 1049–1058. [Google Scholar] [CrossRef]
- Chen, I.J.; Liu, C.Y.; Chiu, J.P.; Hsu, C.H. Therapeutic effect of high-dose green tea extract on weight reduction: A randomized, double-blind, placebo-controlled clinical trial. Clin. Nutr. 2016, 35, 592–599. [Google Scholar] [CrossRef]
- Grove, K.A.; Sae-tan, S.; Kennett, M.J.; Lambert, J.D. (-)-Epigallocatechin-3-gallate inhibits pancreatic lipase and reduces body weight gain in high fat-fed obese mice. Obesity 2012, 20, 2311–2313. [Google Scholar] [CrossRef]
- Koo, S.I.; Noh, S.K. Green tea as inhibitor of the intestinal absorption of lipids: Potential mechanism for its lipid-lowering effect. J. Nutr. Biochem. 2007, 18, 179–183. [Google Scholar] [CrossRef]
- Unno, T.; Osada, C.; Motoo, Y.; Suzuki, Y.; Kobayashi, M.; Nozawa, A. Dietary tea catechins increase fecal energy in rats. J. Nutr. Sci. Vitaminol. 2009, 55, 447–451. [Google Scholar] [CrossRef]
- Yang, X.; Kong, F. Effects of tea polyphenols and different teas on pancreatic α-amylase activity in vitro. LWT Food Sci. Technol. 2016, 66, 232–238. [Google Scholar] [CrossRef]
- Glisan, S.L.; Grove, K.A.; Yennawar, N.H.; Lambert, J.D. Inhibition of pancreatic lipase by black tea theaflavins: Comparative enzymology and in silico modeling studies. Food Chem. 2017, 216, 296–300. [Google Scholar] [CrossRef]
- Henning, S.M.; Yang, J.; Hsu, M.; Lee, R.P.; Grojean, E.M.; Ly, A.; Tseng, C.H.; Heber, D.; Li, Z. Decaffeinated green and black tea polyphenols decrease weight gain and alter microbiome populations and function in diet-induced obese mice. Eur. J. Nutr. 2018, 57, 2759–2769. [Google Scholar] [CrossRef] [PubMed]
- Korpela, K.; Flint, H.J.; Johnstone, A.M.; Lappi, J.; Poutanen, K.; Dewulf, E.; Delzenne, N.; de Vos, W.M.; Salonen, A. Gut microbiota signatures predict host and microbiota responses to dietary interventions in obese individuals. PLoS ONE 2014, 9, e90702. [Google Scholar] [CrossRef] [PubMed]
- Tilg, H.; Moschen, A.R. Microbiota and diabetes: An evolving relationship. Gut 2014, 63, 1513–1521. [Google Scholar] [CrossRef] [PubMed]
- Balaji, M.; Ganjayi, M.S.; Hanuma Kumar, G.E.; Parim, B.N.; Mopuri, R.; Dasari, S. A review on possible therapeutic targets to contain obesity: The role of phytochemicals. Obes. Res. Clin. Pract. 2016, 10, 363–380. [Google Scholar] [CrossRef]
- Bradlow, H.L. Obesity and the gut microbiome: Pathophysiological aspects. Horm. Mol. Biol. Clin. Investig. 2014, 17, 53–61. [Google Scholar] [CrossRef]
- Mahmood, N. A review of α-amylase inhibitors on weight loss and glycemic control in pathological state such as obesity and diabetes. Comp. Clin. Pathol. 2016, 25, 1253–1264. [Google Scholar] [CrossRef]
- Unno, T.; Sakuma, M.; Mitsuhashi, S. Effect of dietary supplementation of (-)-epigallocatechin gallate on gut microbiota and biomarkers of colonic fermentation in rats. J. Nutr. Sci. Vitaminol. 2014, 60, 213–219. [Google Scholar] [CrossRef]
- Yang, C.S.; Zhang, J.; Zhang, L.; Huang, J.; Wang, Y. Mechanisms of body weight reduction and metabolic syndrome alleviation by tea. Mol. Nutr. Food Res. 2016, 60, 160–174. [Google Scholar] [CrossRef]
- Yang, C.S.; Wang, H.; Sheridan, Z.P. Studies on prevention of obesity, metabolic syndrome, diabetes, cardiovascular diseases and cancer by tea. J. Food Drug Anal. 2018, 26, 1–13. [Google Scholar] [CrossRef]
- Espin, J.C.; Gonzalez-Sarrias, A.; Tomas-Barberan, F.A. The gut microbiota: A key factor in the therapeutic effects of (poly)phenols. Biochem. Pharmacol. 2017, 139, 82–93. [Google Scholar] [CrossRef]
- Rothenberg, D.O.; Zhou, C.; Zhang, L. A Review on the Weight-Loss Effects of Oxidized Tea Polyphenols. Molecules 2018, 23, 1176. [Google Scholar] [CrossRef] [PubMed]
- Vazquez Cisneros, L.C.; Lopez-Uriarte, P.; Lopez-Espinoza, A.; Navarro Meza, M.; Espinoza-Gallardo, A.C.; Guzman Aburto, M.B. Effects of green tea and its epigallocatechin (EGCG) content on body weight and fat mass in humans: A systematic review. Nutr. Hosp. 2017, 34, 731–737. [Google Scholar] [CrossRef]
- Hursel, R.; Viechtbauer, W.; Westerterp-Plantenga, M.S. The effects of green tea on weight loss and weight maintenance: A meta-analysis. Int. J. Obes. 2009, 33, 956–961. [Google Scholar] [CrossRef] [PubMed]
- Van Baak, M.A.; Mariman, E.C.M. Dietary Strategies for Weight Loss Maintenance. Nutrients 2019, 11, 1916. [Google Scholar] [CrossRef] [PubMed]
- Mielgo-Ayuso, J.; Barrenechea, L.; Alcorta, P.; Larrarte, E.; Margareto, J.; Labayen, I. Effects of dietary supplementation with epigallocatechin-3-gallate on weight loss, energy homeostasis, cardiometabolic risk factors and liver function in obese women: Randomised, double-blind, placebo-controlled clinical trial. Br. J. Nutr. 2014, 111, 1263–1271. [Google Scholar] [CrossRef] [PubMed]
- Popovich, D.; Yeo, C.R.; Zhang, W. Ginsenosides derived from Asian (Panax ginseng), American ginseng (Panax quinquefolius) and potential cytoactivity. Int. J. Biomed. Pharm. Sci. 2012, 6, 6. [Google Scholar]
- Han, L.K.; Zheng, Y.N.; Yoshikawa, M.; Okuda, H.; Kimura, Y. Anti-obesity effects of chikusetsusaponins isolated from Panax japonicus rhizomes. BMC Complement. Altern. Med. 2005, 5, 9. [Google Scholar] [CrossRef]
- Liu, W.; Zheng, Y.; Han, L.; Wang, H.; Saito, M.; Ling, M.; Kimura, Y.; Feng, Y. Saponins (Ginsenosides) from stems and leaves of Panax quinquefolium prevented high-fat diet-induced obesity in mice. Phytomedicine 2008, 15, 1140–1145. [Google Scholar] [CrossRef]
- Lee, Y.S.; Cha, B.Y.; Yamaguchi, K.; Choi, S.S.; Yonezawa, T.; Teruya, T.; Nagai, K.; Woo, J.T. Effects of Korean white ginseng extracts on obesity in high-fat diet-induced obese mice. Cytotechnology 2010, 62, 367–376. [Google Scholar] [CrossRef][Green Version]
- Kwon, D.H.; Bose, S.; Song, M.Y.; Lee, M.J.; Lim, C.Y.; Kwon, B.S.; Kim, H.J. Efficacy of Korean Red Ginseng by Single Nucleotide Polymorphism in Obese Women: Randomized, Double-blind, Placebo-controlled Trial. J. Ginseng. Res. 2012, 36, 176–189. [Google Scholar] [CrossRef]
- Sotaniemi, E.A.; Haapakoski, E.; Rautio, A. Ginseng therapy in non-insulin-dependent diabetic patients. Diabetes Care 1995, 18, 1373–1375. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.H.; Ahn, S.C.; Lee, S.Y.; Jeong, D.W.; Choi, E.J.; Kim, Y.J.; Lee, J.G.; Lee, Y.H.; Shin, B.C. Effect of Korean red ginseng on insulin sensitivity in non-diabetic healthy overweight and obese adults. Asia Pac. J. Clin. Nutr. 2013, 22, 365–371. [Google Scholar] [CrossRef]
- Reeds, D.N.; Patterson, B.W.; Okunade, A.; Holloszy, J.O.; Polonsky, K.S.; Klein, S. Ginseng and ginsenoside Re do not improve beta-cell function or insulin sensitivity in overweight and obese subjects with impaired glucose tolerance or diabetes. Diabetes Care 2011, 34, 1071–1076. [Google Scholar] [CrossRef]
- Kumar, S.; Verma, A.K.; Das, M.; Jain, S.K.; Dwivedi, P.D. Clinical complications of kidney bean (Phaseolus vulgaris L.) consumption. Nutrition 2013, 29, 821–827. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Simpson, B.K.; Sun, H.; Ngadi, M.O.; Ma, Y.; Huang, T. Phaseolus vulgaris lectins: A systematic review of characteristics and health implications. Crit. Rev. Food Sci. Nutr. 2018, 58, 70–83. [Google Scholar] [CrossRef] [PubMed]
- Nciri, N.; Cho, N.; El Mhamdi, F.; Ben Ismail, H.; Ben Mansour, A.; Sassi, F.H.; Ben Aissa-Fennira, F. Toxicity Assessment of Common Beans (Phaseolus vulgaris L.) Widely Consumed by Tunisian Population. J. Med. Food 2015, 18, 1049–1064. [Google Scholar] [CrossRef] [PubMed]
- Layer, P.; Carlson, G.L.; DiMagno, E.P. Partially purified white bean amylase inhibitor reduces starch digestion in vitro and inactivates intraduodenal amylase in humans. Gastroenterology 1985, 88, 1895–1902. [Google Scholar] [CrossRef]
- Barrett, M.L.; Udani, J.K. A proprietary alpha-amylase inhibitor from white bean (Phaseolus vulgaris): A review of clinical studies on weight loss and glycemic control. Nutr. J. 2011, 10, 24. [Google Scholar] [CrossRef]
- Ganesan, K.; Xu, B. Polyphenol-Rich Dry Common Beans (Phaseolus vulgaris L.) and Their Health Benefits. Int. J. Mol. Sci. 2017, 18, 2331. [Google Scholar] [CrossRef]
- Obiro, W.C.; Zhang, T.; Jiang, B. The nutraceutical role of the Phaseolus vulgaris alpha-amylase inhibitor. Br. J. Nutr. 2008, 100, 1–12. [Google Scholar] [CrossRef]
- Oseguera-Toledo, M.E.; de Mejia, E.G.; Dia, V.P.; Amaya-Llano, S.L. Common bean (Phaseolus vulgaris L.) hydrolysates inhibit inflammation in LPS-induced macrophages through suppression of NF-kappaB pathways. Food Chem. 2011, 127, 1175–1185. [Google Scholar] [CrossRef] [PubMed]
- Santimone, M.; Koukiekolo, R.; Moreau, Y.; Le Berre, V.; Rouge, P.; Marchis-Mouren, G.; Desseaux, V. Porcine pancreatic alpha-amylase inhibition by the kidney bean (Phaseolus vulgaris) inhibitor (alpha-AI1) and structural changes in the alpha-amylase inhibitor complex. Biochim. Biophys. Acta 2004, 1696, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Udani, J.; Tan, O.; Molina, J. Systematic Review and Meta-Analysis of a Proprietary Alpha-Amylase Inhibitor from White Bean (Phaseolus vulgaris L.) on Weight and Fat Loss in Humans. Foods 2018, 7, 63. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Chen, L.; Yang, H.; Gu, J.; Wang, J.; Ren, F. Regular intake of white kidney beans extract (Phaseolus vulgaris L.) induces weight loss compared to placebo in obese human subjects. Food Sci. Nutr. 2020, 8, 1315–1324. [Google Scholar] [CrossRef] [PubMed]
- Onakpoya, I.; Aldaas, S.; Terry, R.; Ernst, E. The efficacy of Phaseolus vulgaris as a weight-loss supplement: A systematic review and meta-analysis of randomised clinical trials. Br. J. Nutr. 2011, 106, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on the substantiation of health claims related to chitosan and reduction in body weight (ID 679, 1499), maintenance of normal blood LDL-cholesterol concentrations (ID 4663), reduction of intestinal transit time (ID 4664) and reduction of inflammation (ID 1985) pursuant to Article 13(1) of Regulation (EC) No 1924/2006. EFSA J. 2011, 9, 2214. [Google Scholar] [CrossRef]
- Ylitalo, R.; Lehtinen, S.; Wuolijoki, E.; Ylitalo, P.; Lehtimaki, T. Cholesterol-lowering properties and safety of chitosan. Arzneimittelforschung 2002, 52, 1–7. [Google Scholar] [CrossRef]
- Chiang, M.T.; Yao, H.T.; Chen, H.C. Effect of dietary chitosans with different viscosity on plasma lipids and lipid peroxidation in rats fed on a diet enriched with cholesterol. Biosci. Biotechnol. Biochem. 2000, 64, 965–971. [Google Scholar] [CrossRef]
- Mesa Ospina, N.; Ospina Alvarez, S.P.; Escobar Sierra, D.M.; Rojas Vahos, D.F.; Zapata Ocampo, P.A.; Ossa Orozco, C.P. Isolation of chitosan from Ganoderma lucidum mushroom for biomedical applications. J. Mater. Sci. Mater. Med. 2015, 26, 135. [Google Scholar] [CrossRef]
- Walsh, A.M.; Sweeney, T.; Bahar, B.; O’Doherty, J.V. Multi-functional roles of chitosan as a potential protective agent against obesity. PLoS ONE 2013, 8, e53828. [Google Scholar] [CrossRef] [PubMed]
- Moraru, C.; Mincea, M.M.; Frandes, M.; Timar, B.; Ostafe, V. A Meta-Analysis on Randomised Controlled Clinical Trials Evaluating the Effect of the Dietary Supplement Chitosan on Weight Loss, Lipid Parameters and Blood Pressure. Medicina 2018, 54, 109. [Google Scholar] [CrossRef] [PubMed]
- Charalampopoulos, D.; Wang, R.; Pandiella, S.S.; Webb, C. Application of cereals and cereal components in functional foods: A review. Int. J. Food Microbiol. 2002, 79, 131–141. [Google Scholar] [CrossRef]
- Jenkins, A.L.; Jenkins, D.J.; Zdravkovic, U.; Wursch, P.; Vuksan, V. Depression of the glycemic index by high levels of beta-glucan fiber in two functional foods tested in type 2 diabetes. Eur. J. Clin. Nutr. 2002, 56, 622–628. [Google Scholar] [CrossRef] [PubMed]
- Tapola, N.; Karvonen, H.; Niskanen, L.; Mikola, M.; Sarkkinen, E. Glycemic responses of oat bran products in type 2 diabetic patients. Nutr. Metab. Cardiovasc. Dis. 2005, 15, 255–261. [Google Scholar] [CrossRef]
- Granfeldt, Y.; Nyberg, L.; Bjorck, I. Muesli with 4 g oat beta-glucans lowers glucose and insulin responses after a bread meal in healthy subjects. Eur. J. Clin. Nutr. 2008, 62, 600–607. [Google Scholar] [CrossRef]
- Cugnet-Anceau, C.; Nazare, J.A.; Biorklund, M.; Le Coquil, E.; Sassolas, A.; Sothier, M.; Holm, J.; Landin-Olsson, M.; Onning, G.; Laville, M.; et al. A controlled study of consumption of beta-glucan-enriched soups for 2 months by type 2 diabetic free-living subjects. Br. J. Nutr. 2010, 103, 422–428. [Google Scholar] [CrossRef]
- Makelainen, H.; Anttila, H.; Sihvonen, J.; Hietanen, R.M.; Tahvonen, R.; Salminen, E.; Mikola, M.; Sontag-Strohm, T. The effect of beta-glucan on the glycemic and insulin index. Eur. J. Clin. Nutr. 2007, 61, 779–785. [Google Scholar] [CrossRef]
- Huang, X.F.; Yu, Y.; Beck, E.J.; South, T.; Li, Y.; Batterham, M.J.; Tapsell, L.C.; Chen, J. Diet high in oat beta-glucan activates the gut-hypothalamic (PYY(3)(-)(3)(6)-NPY) axis and increases satiety in diet-induced obesity in mice. Mol. Nutr. Food Res. 2011, 55, 1118–1121. [Google Scholar] [CrossRef]
- Beck, E.J.; Tapsell, L.C.; Batterham, M.J.; Tosh, S.M.; Huang, X.F. Increases in peptide Y-Y levels following oat beta-glucan ingestion are dose-dependent in overweight adults. Nutr. Res. 2009, 29, 705–709. [Google Scholar] [CrossRef]
- Beck, E.J.; Tapsell, L.C.; Batterham, M.J.; Tosh, S.M.; Huang, X.F. Oat beta-glucan supplementation does not enhance the effectiveness of an energy-restricted diet in overweight women. Br. J. Nutr. 2010, 103, 1212–1222. [Google Scholar] [CrossRef] [PubMed]
- Abutair, A.S.; Naser, I.A.; Hamed, A.T. Soluble fibers from psyllium improve glycemic response and body weight among diabetes type 2 patients (randomized control trial). Nutr. J. 2016, 15, 86. [Google Scholar] [CrossRef] [PubMed]
- Akbarian, S.A.; Asgary, S.; Feizi, A.; Iraj, B.; Askari, G. Comparative Study on the Effect of Plantago psyllium and Ocimum basilicum Seeds on Anthropometric Measures in Nonalcoholic Fatty Liver Patients. Int. J. Prev. Med. 2016, 7, 114. [Google Scholar] [CrossRef] [PubMed]
- Dikeman, C.L.; Fahey, G.C. Viscosity as related to dietary fiber: A review. Crit. Rev. Food Sci. Nutr. 2006, 46, 649–663. [Google Scholar] [CrossRef] [PubMed]
- Riccioni, G.; Sblendorio, V.; Gemello, E.; Di Bello, B.; Scotti, L.; Cusenza, S.; D’Orazio, N. Dietary fibers and cardiometabolic diseases. Int. J. Mol. Sci. 2012, 13, 1524–1540. [Google Scholar] [CrossRef] [PubMed]
- Jane, M.; McKay, J.; Pal, S. Effects of daily consumption of psyllium, oat bran and polyGlycopleX on obesity-related disease risk factors: A critical review. Nutrition 2019, 57, 84–91. [Google Scholar] [CrossRef]
- Wharton, S.; Bonder, R.; Jeffery, A.; Christensen, R.A.G. The safety and effectiveness of commonly-marketed natural supplements for weight loss in populations with obesity: A critical review of the literature from 2006 to 2016. Crit. Rev. Food Sci. Nutr. 2020, 60, 1614–1630. [Google Scholar] [CrossRef]
- Xiao, Z.; Chen, H.; Zhang, Y.; Deng, H.; Wang, K.; Bhagavathula, A.S.; Almuhairi, S.J.; Ryan, P.M.; Rahmani, J.; Dang, M.; et al. The effect of psyllium consumption on weight, body mass index, lipid profile, and glucose metabolism in diabetic patients: A systematic review and dose-response meta-analysis of randomized controlled trials. Phytother. Res. 2020, 34, 1237–1247. [Google Scholar] [CrossRef]
- Darooghegi Mofrad, M.; Mozaffari, H.; Mousavi, S.M.; Sheikhi, A.; Milajerdi, A. The effects of psyllium supplementation on body weight, body mass index and waist circumference in adults: A systematic review and dose-response meta-analysis of randomized controlled trials. Crit. Rev. Food Sci. Nutr. 2020, 60, 859–872. [Google Scholar] [CrossRef]
- Alonso-Sande, M.; Teijeiro-Osorio, D.; Remunan-Lopez, C.; Alonso, M.J. Glucomannan, a promising polysaccharide for biopharmaceutical purposes. Eur. J. Pharm. Biopharm. 2009, 72, 453–462. [Google Scholar] [CrossRef]
- Keithley, J.K.; Swanson, B.; Mikolaitis, S.L.; DeMeo, M.; Zeller, J.M.; Fogg, L.; Adamji, J. Safety and efficacy of glucomannan for weight loss in overweight and moderately obese adults. J. Obes. 2013, 2013, 610908. [Google Scholar] [CrossRef] [PubMed]
- Scalfi, L.; Coltorti, A.; D’Arrigo, E.; Carandente, V.; Mazzacano, C.; Di Palo, M.; Contaldo, F. Effect of dietary fibre on postprandial thermogenesis. Int. J. Obes. 1987, 11 (Suppl. 1), 95–99. [Google Scholar]
- Keithley, J.; Swanson, B. Glucomannan and obesity: A critical review. Altern. Ther. Health Med. 2005, 11, 30–34. [Google Scholar] [PubMed]
- Sood, N.; Baker, W.L.; Coleman, C.I. Effect of glucomannan on plasma lipid and glucose concentrations, body weight, and blood pressure: Systematic review and meta-analysis. Am. J. Clin. Nutr. 2008, 88, 1167–1175. [Google Scholar] [CrossRef]
- Zalewski, B.M.; Szajewska, H. No Effect of Glucomannan on Body Weight Reduction in Children and Adolescents with Overweight and Obesity: A Randomized Controlled Trial. J. Pediatr. 2019, 211, 85–91.e1. [Google Scholar] [CrossRef] [PubMed]
- Maia-Landim, A.; Ramirez, J.M.; Lancho, C.; Poblador, M.S.; Lancho, J.L. Long-term effects of Garcinia cambogia/Glucomannan on weight loss in people with obesity, PLIN4, FTO and Trp64Arg polymorphisms. BMC Complement. Altern. Med. 2018, 18, 26. [Google Scholar] [CrossRef] [PubMed]
- Onakpoya, I.; Posadzki, P.; Ernst, E. The efficacy of glucomannan supplementation in overweight and obesity: A systematic review and meta-analysis of randomized clinical trials. J. Am. Coll. Nutr. 2014, 33, 70–78. [Google Scholar] [CrossRef]
- Thombare, N.; Jha, U.; Mishra, S.; Siddiqui, M.Z. Guar gum as a promising starting material for diverse applications: A review. Int. J. Biol. Macromol. 2016, 88, 361–372. [Google Scholar] [CrossRef]
- Mudgil, D.; Barak, S.; Khatkar, B.S. Guar gum: Processing, properties and food applications-A Review. J. Food Sci. Technol. 2014, 51, 409–418. [Google Scholar] [CrossRef]
- Pittler, M.H.; Ernst, E. Guar gum for body weight reduction: Meta-analysis of randomized trials. Am. J. Med. 2001, 110, 724–730. [Google Scholar] [CrossRef]
- Uusitupa, M.; Tuomilehto, J.; Karttunen, P.; Wolf, E. Long term effects of guar gum on metabolic control, serum cholesterol and blood pressure levels in type 2 (non-insulin-dependent) diabetic patients with high blood pressure. Ann. Clin. Res. 1984, 16 (Suppl. 43), 126–131. [Google Scholar]
- Rao, T.P. Role of guar fiber in appetite control. Physiol. Behav. 2016, 164, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Butt, M.S.; Shahzadi, N.; Sharif, M.K.; Nasir, M. Guar gum: A miracle therapy for hypercholesterolemia, hyperglycemia and obesity. Crit. Rev. Food Sci. Nutr. 2007, 47, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Medina-Esquivel, R.; Freile-Pelegrin, Y.; Quintana-Owen, P.; Yáñez-Limón, J.M.; Alvarado-Gil, J.J. Measurement of the Sol–Gel Transition Temperature in Agar. Int. J. Thermophys. 2008, 29, 2036. [Google Scholar] [CrossRef]
- Sanaka, M.; Yamamoto, T.; Anjiki, H.; Nagasawa, K.; Kuyama, Y. Effects of agar and pectin on gastric emptying and post-prandial glycaemic profiles in healthy human volunteers. Clin. Exp. Pharmacol. Physiol. 2007, 34, 1151–1155. [Google Scholar] [CrossRef] [PubMed]
- Clegg, M.E.; Shafat, A. The effect of agar jelly on energy expenditure, appetite, gastric emptying and glycaemic response. Eur. J. Nutr. 2014, 53, 533–539. [Google Scholar] [CrossRef]
- Maeda, H.; Yamamoto, R.; Hirao, K.; Tochikubo, O. Effects of agar (kanten) diet on obese patients with impaired glucose tolerance and type 2 diabetes. Diabetes Obes. Metab. 2005, 7, 40–46. [Google Scholar] [CrossRef]
- Thompson, S.V.; Hannon, B.A.; An, R.; Holscher, H.D. Effects of isolated soluble fiber supplementation on body weight, glycemia, and insulinemia in adults with overweight and obesity: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2017, 106, 1514–1528. [Google Scholar] [CrossRef]
- Roberfroid, M.B. Inulin-type fructans: Functional food ingredients. J. Nutr. 2007, 137, 2493S–2502S. [Google Scholar] [CrossRef]
- Wilson, B.; Whelan, K. Prebiotic inulin-type fructans and galacto-oligosaccharides: Definition, specificity, function, and application in gastrointestinal disorders. J. Gastroenterol. Hepatol. 2017, 32 (Suppl. 1), 64–68. [Google Scholar] [CrossRef]
- Shoaib, M.; Shehzad, A.; Omar, M.; Rakha, A.; Raza, H.; Sharif, H.R.; Shakeel, A.; Ansari, A.; Niazi, S. Inulin: Properties, health benefits and food applications. Carbohydr. Polym. 2016, 147, 444–454. [Google Scholar] [CrossRef] [PubMed]
- Delzenne, N.M.; Cani, P.D.; Daubioul, C.; Neyrinck, A.M. Impact of inulin and oligofructose on gastrointestinal peptides. Br. J. Nutr. 2005, 93 (Suppl. 1), S157–S161. [Google Scholar] [CrossRef]
- Parnell, J.A.; Reimer, R.A. Weight loss during oligofructose supplementation is associated with decreased ghrelin and increased peptide YY in overweight and obese adults. Am. J. Clin. Nutr. 2009, 89, 1751–1759. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, S.; Chouinard-Castonguay, S.; Gagnon, C.; Rudkowska, I. Prebiotics in the management of components of the metabolic syndrome. Maturitas 2017, 104, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Liber, A.; Szajewska, H. Effects of inulin-type fructans on appetite, energy intake, and body weight in children and adults: Systematic review of randomized controlled trials. Ann. Nutr. Metab. 2013, 63, 42–54. [Google Scholar] [CrossRef]
- Hess, A.L.; Benitez-Paez, A.; Blaedel, T.; Larsen, L.H.; Iglesias, J.R.; Madera, C.; Sanz, Y.; Larsen, T.M.; MyNewGut, C. The effect of inulin and resistant maltodextrin on weight loss during energy restriction: A randomised, placebo-controlled, double-blinded intervention. Eur. J. Nutr. 2019. [Google Scholar] [CrossRef]
- Vaghef-Mehrabany, E.; Ranjbar, F.; Asghari-Jafarabadi, M.; Hosseinpour-Arjmand, S.; Ebrahimi-Mameghani, M. Calorie restriction in combination with prebiotic supplementation in obese women with depression: Effects on metabolic and clinical response. Nutr. Neurosci. 2019, 1–15. [Google Scholar] [CrossRef]
- Guess, N.D.; Dornhorst, A.; Oliver, N.; Bell, J.D.; Thomas, E.L.; Frost, G.S. A randomized controlled trial: The effect of inulin on weight management and ectopic fat in subjects with prediabetes. Nutr. Metab. 2015, 12, 36. [Google Scholar] [CrossRef]
- Devi, S.G.; Dhamotharan, R. Caralluma fimbriata- An Important Medicinal Plant: A Review of Its Traditional Uses, Phytochemistry and Pharmacological Properties. Int. J. PharmTech Res. 2016, 9, 223–230. [Google Scholar]
- Dutt, H.C.; Singh, S.; Avula, B.; Khan, I.A.; Bedi, Y.S. Pharmacological review of Caralluma R.Br. with special reference to appetite suppression and anti-obesity. J. Med. Food 2012, 15, 108–119. [Google Scholar] [CrossRef]
- Kamalakkannan, S.; Rajendran, R.; Venkatesh, R.V.; Clayton, P.; Akbarsha, M.A. Antiobesogenic and Antiatherosclerotic Properties of Caralluma fimbriata Extract. J. Nutr. Metab. 2010, 2010, 285301. [Google Scholar] [CrossRef] [PubMed]
- Astell, K.J.; Mathai, M.L.; McAinch, A.J.; Stathis, C.G.; Su, X.Q. A pilot study investigating the effect of Caralluma fimbriata extract on the risk factors of metabolic syndrome in overweight and obese subjects: A randomised controlled clinical trial. Complement. Ther. Med. 2013, 21, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Kuriyan, R.; Raj, T.; Srinivas, S.K.; Vaz, M.; Rajendran, R.; Kurpad, A.V. Effect of Caralluma fimbriata extract on appetite, food intake and anthropometry in adult Indian men and women. Appetite 2007, 48, 338–344. [Google Scholar] [CrossRef]
- Arora, E.; Khajuria, V.; Tandon, V.R.; Sharma, A.; Mahajan, A.; Gillani, Z.H.; Choudhary, N. To evaluate efficacy and safety of Caralluma fimbriata in overweight and obese patients: A randomized, single blinded, placebo control trial. Perspect. Clin. Res. 2015, 6, 39–44. [Google Scholar] [CrossRef]
- Yousefi, R.; Saidpour, A.; Mottaghi, A. The effects of Spirulina supplementation on metabolic syndrome components, its liver manifestation and related inflammatory markers: A systematic review. Complement. Ther. Med. 2019, 42, 137–144. [Google Scholar] [CrossRef]
- Zhao, B.; Cui, Y.; Fan, X.; Qi, P.; Liu, C.; Zhou, X.; Zhang, X. Anti-obesity effects of Spirulina platensis protein hydrolysate by modulating brain-liver axis in high-fat diet fed mice. PLoS ONE 2019, 14, e0218543. [Google Scholar] [CrossRef] [PubMed]
- Seo, Y.J.; Kim, K.J.; Choi, J.; Koh, E.J.; Lee, B.Y. Spirulina maxima Extract Reduces Obesity through Suppression of Adipogenesis and Activation of Browning in 3T3-L1 Cells and High-Fat Diet-Induced Obese Mice. Nutrients 2018, 10, 712. [Google Scholar] [CrossRef]
- Zeinalian, R.; Farhangi, M.A.; Shariat, A.; Saghafi-Asl, M. The effects of Spirulina Platensis on anthropometric indices, appetite, lipid profile and serum vascular endothelial growth factor (VEGF) in obese individuals: A randomized double blinded placebo controlled trial. BMC Complement. Altern. Med. 2017, 17, 225. [Google Scholar] [CrossRef]
- Moradi, S.; Ziaei, R.; Foshati, S.; Mohammadi, H.; Nachvak, S.M.; Rouhani, M.H. Effects of Spirulina supplementation on obesity: A systematic review and meta-analysis of randomized clinical trials. Complement. Ther. Med. 2019, 47, 102211. [Google Scholar] [CrossRef]
- Hernandez-Lepe, M.A.; Wall-Medrano, A.; Lopez-Diaz, J.A.; Juarez-Oropeza, M.A.; Hernandez-Torres, R.P.; Ramos-Jimenez, A. Hypolipidemic Effect of Arthrospira (Spirulina) maxima Supplementation and a Systematic Physical Exercise Program in Overweight and Obese Men: A Double-Blind, Randomized, and Crossover Controlled Trial. Mar. Drugs 2019, 17, 270. [Google Scholar] [CrossRef]
- Hall, W.L.; Millward, D.J.; Long, S.J.; Morgan, L.M. Casein and whey exert different effects on plasma amino acid profiles, gastrointestinal hormone secretion and appetite. Br. J. Nutr. 2003, 89, 239–248. [Google Scholar] [CrossRef]
- Miller, P.E.; Alexander, D.D.; Perez, V. Effects of whey protein and resistance exercise on body composition: A meta-analysis of randomized controlled trials. J. Am. Coll. Nutr. 2014, 33, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Castro, L.H.A.; de Araújo, F.H.S.; Olimpio, M.Y.M.; Primo, R.B.d.; Pereira, T.T.; Lopes, L.A.F.; Trindade, E.B.S.d.; Fernandes, R.; Oesterreich, S.A. Comparative Meta-Analysis of the Effect of Concentrated, Hydrolyzed, and Isolated Whey Protein Supplementation on Body Composition of Physical Activity Practitioners. Nutrients 2019, 11, 2047. [Google Scholar] [CrossRef]
- Nehlig, A.; Daval, J.L.; Debry, G. Caffeine and the central nervous system: Mechanisms of action, biochemical, metabolic and psychostimulant effects. Brain Res. Rev. 1992, 17, 139–170. [Google Scholar] [CrossRef]
- Velickovic, K.; Wayne, D.; Leija, H.A.L.; Bloor, I.; Morris, D.E.; Law, J.; Budge, H.; Sacks, H.; Symonds, M.E.; Sottile, V. Caffeine exposure induces browning features in adipose tissue in vitro and in vivo. Sci. Rep. 2019, 9, 9104. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, K.; Yoshida, T.; Kamanaru, K.; Hiraoka, N.; Kondo, M. Caffeine activates brown adipose tissue thermogenesis and metabolic rate in mice. J. Nutr. Sci. Vitaminol. 1990, 36, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Naveed, M.; Hejazi, V.; Abbas, M.; Kamboh, A.A.; Khan, G.J.; Shumzaid, M.; Ahmad, F.; Babazadeh, D.; FangFang, X.; Modarresi-Ghazani, F.; et al. Chlorogenic acid (CGA): A pharmacological review and call for further research. Biomed. Pharmacother. 2018, 97, 67–74. [Google Scholar] [CrossRef]
- Stefanello, N.; Spanevello, R.M.; Passamonti, S.; Porciuncula, L.; Bonan, C.D.; Olabiyi, A.A.; Teixeira da Rocha, J.B.; Assmann, C.E.; Morsch, V.M.; Schetinger, M.R.C. Coffee, caffeine, chlorogenic acid, and the purinergic system. Food Chem. Toxicol. 2019, 123, 298–313. [Google Scholar] [CrossRef]
- Tabrizi, R.; Saneei, P.; Lankarani, K.B.; Akbari, M.; Kolahdooz, F.; Esmaillzadeh, A.; Nadi-Ravandi, S.; Mazoochi, M.; Asemi, Z. The effects of caffeine intake on weight loss: A systematic review and dos-response meta-analysis of randomized controlled trials. Crit. Rev. Food Sci. Nutr. 2019, 59, 2688–2696. [Google Scholar] [CrossRef]
- Thom, E. The effect of chlorogenic acid enriched coffee on glucose absorption in healthy volunteers and its effect on body mass when used long-term in overweight and obese people. J. Int. Med. Res. 2007, 35, 900–908. [Google Scholar] [CrossRef]
- Davoodi, S.H.; Hajimiresmaiel, S.J.; Ajami, M.; Mohseni-Bandpei, A.; Ayatollahi, S.A.; Dowlatshahi, K.; Javedan, G.; Pazoki-Toroudi, H. Caffeine treatment prevented from weight regain after calorie shifting diet induced weight loss. Iran. J. Pharm. Res. 2014, 13, 707–718. [Google Scholar] [PubMed]
- Stohs, S.J. Safety, Efficacy, and Mechanistic Studies Regarding Citrus aurantium (Bitter Orange) Extract and p-Synephrine. Phytother. Res. 2017, 31, 1463–1474. [Google Scholar] [CrossRef]
- Gutierrez-Hellin, J.; Del Coso, J. Effects of p-Synephrine and Caffeine Ingestion on Substrate Oxidation during Exercise. Med. Sci. Sports Exerc. 2018, 50, 1899–1906. [Google Scholar] [CrossRef]
- Stohs, S.J.; Shara, M.; Ray, S.D. p-Synephrine, ephedrine, p-octopamine and m-synephrine: Comparative mechanistic, physiological and pharmacological properties. Phytother. Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Cardile, V.; Graziano, A.C.; Venditti, A. Clinical evaluation of Moro (Citrus sinensis (L.) Osbeck) orange juice supplementation for the weight management. Nat. Prod. Res. 2015, 29, 2256–2260. [Google Scholar] [CrossRef]
- Cho, Y.G.; Jung, J.H.; Kang, J.H.; Kwon, J.S.; Yu, S.P.; Baik, T.G. Effect of a herbal extract powder (YY-312) from Imperata cylindrica Beauvois, Citrus unshiu Markovich, and Evodia officinalis Dode on body fat mass in overweight adults: A 12-week, randomized, double-blind, placebo-controlled, parallel-group clinical trial. BMC Complement. Altern. Med. 2017, 17, 375. [Google Scholar] [CrossRef] [PubMed]
- Stohs, S.J.; Preuss, H.G.; Shara, M. A review of the human clinical studies involving Citrus aurantium (bitter orange) extract and its primary protoalkaloid p-synephrine. Int. J. Med. Sci. 2012, 9, 527–538. [Google Scholar] [CrossRef]
- Stohs, S.J.; Preuss, H.G.; Shara, M. The safety of Citrus aurantium (bitter orange) and its primary protoalkaloid p-synephrine. Phytother. Res. 2011, 25, 1421–1428. [Google Scholar] [CrossRef]
- Tung, Y.C.; Chang, W.T.; Li, S.; Wu, J.C.; Badmeav, V.; Ho, C.T.; Pan, M.H. Citrus peel extracts attenuated obesity and modulated gut microbiota in mice with high-fat diet-induced obesity. Food Funct. 2018, 9, 3363–3373. [Google Scholar] [CrossRef]
- Park, J.; Kim, H.L.; Jung, Y.; Ahn, K.S.; Kwak, H.J.; Um, J.Y. Bitter Orange (Citrus aurantium Linne) Improves Obesity by Regulating Adipogenesis and Thermogenesis through AMPK Activation. Nutrients 2019, 11, 988. [Google Scholar] [CrossRef]
- Lima, N.D.S.; Numata, E.P.; Mesquita, L.M.S.; Dias, P.H.; Vilegas, W.; Gambero, A.; Ribeiro, M.L. Modulatory Effects of Guarana (Paullinia cupana) on Adipogenesis. Nutrients 2017, 9, 635. [Google Scholar] [CrossRef] [PubMed]
- Bortolin, R.C.; Vargas, A.R.; de Miranda Ramos, V.; Gasparotto, J.; Chaves, P.R.; Schnorr, C.E.; da Boit Martinello, K.; Silveira, A.K.; Gomes, H.M.; Rabelo, T.K.; et al. Guarana supplementation attenuated obesity, insulin resistance, and adipokines dysregulation induced by a standardized human Western diet via brown adipose tissue activation. Phytother. Res. 2019, 33, 1394–1403. [Google Scholar] [CrossRef] [PubMed]
- Berube-Parent, S.; Pelletier, C.; Dore, J.; Tremblay, A. Effects of encapsulated green tea and Guarana extracts containing a mixture of epigallocatechin-3-gallate and caffeine on 24 h energy expenditure and fat oxidation in men. Br. J. Nutr. 2005, 94, 432–436. [Google Scholar] [CrossRef] [PubMed]
- Boozer, C.N.; Nasser, J.A.; Heymsfield, S.B.; Wang, V.; Chen, G.; Solomon, J.L. An herbal supplement containing Ma Huang-Guarana for weight loss: A randomized, double-blind trial. Int. J. Obes. Relat. Metab. Disord. 2001, 25, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Krewer Cda, C.; Ribeiro, E.E.; Ribeiro, E.A.; Moresco, R.N.; da Rocha, M.I.; Montagner, G.F.; Machado, M.M.; Viegas, K.; Brito, E.; da Cruz, I.B. Habitual intake of guarana and metabolic morbidities: An epidemiological study of an elderly Amazonian population. Phytother. Res. 2011, 25, 1367–1374. [Google Scholar] [CrossRef]
- Andersen, T.; Fogh, J. Weight loss and delayed gastric emptying following a South American herbal preparation in overweight patients. J. Hum. Nutr. Diet. 2001, 14, 243–250. [Google Scholar] [CrossRef]
- Restani, P.; Di Lorenzo, C.; Garcia-Alvarez, A.; Badea, M.; Ceschi, A.; Egan, B.; Dima, L.; Lude, S.; Maggi, F.M.; Marculescu, A.; et al. Adverse Effects of Plant Food Supplements Self-Reported by Consumers in the PlantLIBRA Survey Involving Six European Countries. PLoS ONE 2016, 11, e0150089. [Google Scholar] [CrossRef]
- Fisher, F.M.; Maratos-Flier, E. Understanding the Physiology of FGF21. Annu. Rev. Physiol. 2016, 78, 223–241. [Google Scholar] [CrossRef]
- Watanabe, M.; Singhal, G.; Fisher, F.M.; Beck, T.C.; Morgan, D.A.; Socciarelli, F.; Mather, M.L.; Risi, R.; Bourke, J.; Rahmouni, K.; et al. Liver-derived FGF21 is essential for full adaptation to ketogenic diet but does not regulate glucose homeostasis. Endocrine 2019. [Google Scholar] [CrossRef]
- Rollyson, W.D.; Stover, C.A.; Brown, K.C.; Perry, H.E.; Stevenson, C.D.; McNees, C.A.; Ball, J.G.; Valentovic, M.A.; Dasgupta, P. Bioavailability of capsaicin and its implications for drug delivery. J. Control. Release 2014, 196, 96–105. [Google Scholar] [CrossRef]
- Groninger, H.; Schisler, R.E. Topical capsaicin for neuropathic pain #255. J. Palliat. Med. 2012, 15, 946–947. [Google Scholar] [CrossRef] [PubMed]
- Iida, T.; Moriyama, T.; Kobata, K.; Morita, A.; Murayama, N.; Hashizume, S.; Fushiki, T.; Yazawa, S.; Watanabe, T.; Tominaga, M. TRPV1 activation and induction of nociceptive response by a non-pungent capsaicin-like compound, capsiate. Neuropharmacology 2003, 44, 958–967. [Google Scholar] [CrossRef]
- Panchal, S.K.; Bliss, E.; Brown, L. Capsaicin in Metabolic Syndrome. Nutrients 2018, 10, 630. [Google Scholar] [CrossRef] [PubMed]
- Smeets, A.J.; Westerterp-Plantenga, M.S. The acute effects of a lunch containing capsaicin on energy and substrate utilisation, hormones, and satiety. Eur. J. Nutr. 2009, 48, 229–234. [Google Scholar] [CrossRef]
- Kang, J.H.; Goto, T.; Han, I.S.; Kawada, T.; Kim, Y.M.; Yu, R. Dietary capsaicin reduces obesity-induced insulin resistance and hepatic steatosis in obese mice fed a high-fat diet. Obesity 2010, 18, 780–787. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Ma, X.; Zhang, L.; Sun, H.; Liu, X. Capsaicin Reduces Blood Glucose by Increasing Insulin Levels and Glycogen Content Better than Capsiate in Streptozotocin-Induced Diabetic Rats. J. Agric. Food Chem. 2017, 65, 2323–2330. [Google Scholar] [CrossRef]
- Baskaran, P.; Krishnan, V.; Fettel, K.; Gao, P.; Zhu, Z.; Ren, J.; Thyagarajan, B. TRPV1 activation counters diet-induced obesity through sirtuin-1 activation and PRDM-16 deacetylation in brown adipose tissue. Int. J. Obes. 2017, 41, 739–749. [Google Scholar] [CrossRef] [PubMed]
- Kawabata, F.; Inoue, N.; Masamoto, Y.; Matsumura, S.; Kimura, W.; Kadowaki, M.; Higashi, T.; Tominaga, M.; Inoue, K.; Fushiki, T. Non-pungent capsaicin analogs (capsinoids) increase metabolic rate and enhance thermogenesis via gastrointestinal TRPV1 in mice. Biosci. Biotechnol. Biochem. 2009, 73, 2690–2697. [Google Scholar] [CrossRef]
- Baskaran, P.; Krishnan, V.; Ren, J.; Thyagarajan, B. Capsaicin induces browning of white adipose tissue and counters obesity by activating TRPV1 channel-dependent mechanisms. Br. J. Pharmacol. 2016, 173, 2369–2389. [Google Scholar] [CrossRef]
- Whiting, S.; Derbyshire, E.; Tiwari, B.K. Capsaicinoids and capsinoids. A potential role for weight management? A systematic review of the evidence. Appetite 2012, 59, 341–348. [Google Scholar] [CrossRef]
- Whiting, S.; Derbyshire, E.J.; Tiwari, B. Could capsaicinoids help to support weight management? A systematic review and meta-analysis of energy intake data. Appetite 2014, 73, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Zsiboras, C.; Matics, R.; Hegyi, P.; Balasko, M.; Petervari, E.; Szabo, I.; Sarlos, P.; Miko, A.; Tenk, J.; Rostas, I.; et al. Capsaicin and capsiate could be appropriate agents for treatment of obesity: A meta-analysis of human studies. Crit. Rev. Food Sci. Nutr. 2018, 58, 1419–1427. [Google Scholar] [CrossRef] [PubMed]
- Inoue, N.; Matsunaga, Y.; Satoh, H.; Takahashi, M. Enhanced energy expenditure and fat oxidation in humans with high BMI scores by the ingestion of novel and non-pungent capsaicin analogues (capsinoids). Biosci. Biotechnol. Biochem. 2007, 71, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Oyagbemi, A.A.; Saba, A.B.; Ibraheem, A.O. Curcumin: From food spice to cancer prevention. Asian Pac. J. Cancer Prev. 2009, 10, 963–967. [Google Scholar] [PubMed]
- Lao, C.D.; Ruffin, M.T.t.; Normolle, D.; Heath, D.D.; Murray, S.I.; Bailey, J.M.; Boggs, M.E.; Crowell, J.; Rock, C.L.; Brenner, D.E. Dose escalation of a curcuminoid formulation. BMC Complement. Altern. Med. 2006, 6, 10. [Google Scholar] [CrossRef] [PubMed]
- Di Pierro, F.; Bressan, A.; Ranaldi, D.; Rapacioli, G.; Giacomelli, L.; Bertuccioli, A. Potential role of bioavailable curcumin in weight loss and omental adipose tissue decrease: Preliminary data of a randomized, controlled trial in overweight people with metabolic syndrome. Preliminary study. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 4195–4202. [Google Scholar] [PubMed]
- Rahmani, S.; Asgary, S.; Askari, G.; Keshvari, M.; Hatamipour, M.; Feizi, A.; Sahebkar, A. Treatment of Non-alcoholic Fatty Liver Disease with Curcumin: A Randomized Placebo-controlled Trial. Phytother. Res. 2016, 30, 1540–1548. [Google Scholar] [CrossRef]
- Akbari, M.; Lankarani, K.B.; Tabrizi, R.; Ghayour-Mobarhan, M.; Peymani, P.; Ferns, G.; Ghaderi, A.; Asemi, Z. The Effects of Curcumin on Weight Loss Among Patients with Metabolic Syndrome and Related Disorders: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Front. Pharmacol. 2019, 10, 649. [Google Scholar] [CrossRef]
- Mohammadi, A.; Sahebkar, A.; Iranshahi, M.; Amini, M.; Khojasteh, R.; Ghayour-Mobarhan, M.; Ferns, G.A. Effects of supplementation with curcuminoids on dyslipidemia in obese patients: A randomized crossover trial. Phytother. Res. 2013, 27, 374–379. [Google Scholar] [CrossRef]
- Ghazimoradi, M.; Saberi-Karimian, M.; Mohammadi, F.; Sahebkar, A.; Tavallaie, S.; Safarian, H.; Ferns, G.A.; Ghayour-Mobarhan, M.; Moohebati, M.; Esmaeili, H.; et al. The Effects of Curcumin and Curcumin-Phospholipid Complex on the Serum Pro-oxidant-Antioxidant Balance in Subjects with Metabolic Syndrome. Phytother. Res. 2017, 31, 1715–1721. [Google Scholar] [CrossRef]
- Jamwal, R. Bioavailable curcumin formulations: A review of pharmacokinetic studies in healthy volunteers. J. Integr. Med. 2018, 16, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.A.; Euden, S.A.; Platton, S.L.; Cooke, D.N.; Shafayat, A.; Hewitt, H.R.; Marczylo, T.H.; Morgan, B.; Hemingway, D.; Plummer, S.M.; et al. Phase I clinical trial of oral curcumin: Biomarkers of systemic activity and compliance. Clin. Cancer Res. 2004, 10, 6847–6854. [Google Scholar] [CrossRef] [PubMed]
- National Research Council. Recommended Dietary Allowances, 10th ed.; National Academies Press: Washington, DC, USA, 1989. [Google Scholar]
- Xu, Y.; Jiang, W.; Chen, G.; Zhu, W.; Ding, W.; Ge, Z.; Tan, Y.; Ma, T.; Cui, G. L-carnitine treatment of insulin resistance: A systematic review and meta-analysis. Adv. Clin. Exp. Med. 2017, 26, 333–338. [Google Scholar] [CrossRef]
- Brandsch, C.; Eder, K. Effect of L-carnitine on weight loss and body composition of rats fed a hypocaloric diet. Ann. Nutr. Metab. 2002, 46, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Stephens, F.B.; Wall, B.T.; Marimuthu, K.; Shannon, C.E.; Constantin-Teodosiu, D.; Macdonald, I.A.; Greenhaff, P.L. Skeletal muscle carnitine loading increases energy expenditure, modulates fuel metabolism gene networks and prevents body fat accumulation in humans. J. Physiol. 2013, 591, 4655–4666. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.S.; Lee, H.J.; Lee, H.S.; Kim, Y. L-carnitine stimulates lipolysis via induction of the lipolytic gene expression and suppression of the adipogenic gene expression in 3T3-L1 adipocytes. J. Med. Food 2006, 9, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Obici, S.; Feng, Z.; Arduini, A.; Conti, R.; Rossetti, L. Inhibition of hypothalamic carnitine palmitoyltransferase-1 decreases food intake and glucose production. Nat. Med. 2003, 9, 756–761. [Google Scholar] [CrossRef] [PubMed]
- Karimi, M.; Rafraf, M.; Rashidi, M.; Jafari, A. Effect of L-carnitine Supplementation with or Without Moderate Aerobic Training on Serum Lipid Profile and Body Fat Percentage in Obese Women. Iran. J. Endocrinol. Metab. 2013, 14, 445–454. [Google Scholar]
- Rafraf, M.; Karimi, M.; Jafari, A. Effect of L-carnitine supplementation in comparison with moderate aerobic training on serum inflammatory parameters in healthy obese women. J. Sports Med. Phys. Fitness 2015, 55, 1363–1370. [Google Scholar]
- Pooyandjoo, M.; Nouhi, M.; Shab-Bidar, S.; Djafarian, K.; Olyaeemanesh, A. The effect of (L-)carnitine on weight loss in adults: A systematic review and meta-analysis of randomized controlled trials. Obes. Rev. 2016, 17, 970–976. [Google Scholar] [CrossRef]
- Askarpour, M.; Hadi, A.; Miraghajani, M.; Symonds, M.E.; Sheikhi, A.; Ghaedi, E. Beneficial effects of l-carnitine supplementation for weight management in overweight and obese adults: An updated systematic review and dose-response meta-analysis of randomized controlled trials. Pharmacol. Res. 2020, 151, 104554. [Google Scholar] [CrossRef] [PubMed]
- Ivy, J.L. Effect of pyruvate and dihydroxyacetone on metabolism and aerobic endurance capacity. Med. Sci. Sports Exerc. 1998, 30, 837–843. [Google Scholar] [CrossRef] [PubMed]
- Ivy, J.L.; Cortez, M.Y.; Chandler, R.M.; Byrne, H.K.; Miller, R.H. Effects of pyruvate on the metabolism and insulin resistance of obese Zucker rats. Am. J. Clin. Nutr. 1994, 59, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Stanko, R.T.; Adibi, S.A. Inhibition of lipid accumulation and enhancement of energy expenditure by the addition of pyruvate and dihydroxyacetone to a rat diet. Metabolism 1986, 35, 182–186. [Google Scholar] [CrossRef]
- Onakpoya, I.; Hunt, K.; Wider, B.; Ernst, E. Pyruvate supplementation for weight loss: A systematic review and meta-analysis of randomized clinical trials. Crit. Rev. Food Sci. Nutr. 2014, 54, 17–23. [Google Scholar] [CrossRef]
- Flickinger, B.D.; Matsuo, N. Nutritional characteristics of DAG oil. Lipids 2003, 38, 129–132. [Google Scholar] [CrossRef]
- Yasunaga, K.; Glinsmann, W.H.; Seo, Y.; Katsuragi, Y.; Kobayashi, S.; Flickinger, B.; Kennepohl, E.; Yasukawa, T.; Borzelleca, J.F. Safety aspects regarding the consumption of high-dose dietary diacylglycerol oil in men and women in a double-blind controlled trial in comparison with consumption of a triacylglycerol control oil. Food Chem. Toxicol. 2004, 42, 1419–1429. [Google Scholar] [CrossRef]
- Taguchi, H.; Nagao, T.; Watanabe, H.; Onizawa, K.; Matsuo, N.; Tokimitsu, I.; Itakura, H. Energy value and digestibility of dietary oil containing mainly 1,3-diacylglycerol are similar to those of triacylglycerol. Lipids 2001, 36, 379–382. [Google Scholar] [CrossRef]
- Murase, T.; Mizuno, T.; Omachi, T.; Onizawa, K.; Komine, Y.; Kondo, H.; Hase, T.; Tokimitsu, I. Dietary diacylglycerol suppresses high fat and high sucrose diet-induced body fat accumulation in C57BL/6J mice. J. Lipid Res. 2001, 42, 372–378. [Google Scholar]
- Saito, S.; Fukuhara, I.; Osaki, N.; Nakamura, H.; Katsuragi, Y. Consumption of alpha-Linolenic Acid-enriched Diacylglycerol Reduces Visceral Fat Area in Overweight and Obese Subjects: A Randomized, Double-blind Controlled, Parallel-group Designed Trial. J. Oleo Sci. 2016, 65, 603–611. [Google Scholar] [CrossRef]
- Xu, T.; Li, X.; Zhang, Z.; Ma, X.; Li, D. Effect of diacylglycerol on body weight: A meta-analysis. Asia Pac. J. Clin. Nutr. 2008, 17, 415–421. [Google Scholar] [PubMed]
- Rudkowska, I.; Roynette, C.E.; Demonty, I.; Vanstone, C.A.; Jew, S.; Jones, P.J. Diacylglycerol: Efficacy and mechanism of action of an anti-obesity agent. Obes. Res. 2005, 13, 1864–1876. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Xu, T.; Takase, H.; Tokimitsu, I.; Zhang, P.; Wang, Q.; Yu, X.; Zhang, A. Diacylglycerol-induced improvement of whole-body insulin sensitivity in type 2 diabetes mellitus: A long-term randomized, double-blind controlled study. Clin. Nutr. 2008, 27, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Bessesen, D.H.; Vensor, S.H.; Jackman, M.R. Trafficking of dietary oleic, linolenic, and stearic acids in fasted or fed lean rats. Am. J. Physiol. Endocrinol. Metab. 2000, 278, E1124–E1132. [Google Scholar] [CrossRef]
- Leyton, J.; Drury, P.J.; Crawford, M.A. Differential oxidation of saturated and unsaturated fatty acids in vivo in the rat. Br. J. Nutr. 1987, 57, 383–393. [Google Scholar] [CrossRef]
- DeLany, J.P.; Windhauser, M.M.; Champagne, C.M.; Bray, G.A. Differential oxidation of individual dietary fatty acids in humans. Am. J. Clin. Nutr. 2000, 72, 905–911. [Google Scholar] [CrossRef]
- Ando, Y.; Saito, S.; Miura, H.; Osaki, N.; Katsuragi, Y. Consumption of alpha-linolenic acid-enriched diacylglycerol induces increase in dietary fat oxidation compared with alpha-linolenic acid-enriched triacylglycerol: A randomized, double-blind trial. Nutr. Res. 2017, 48, 85–92. [Google Scholar] [CrossRef]
- Saito, S.; Mori, A.; Osaki, N.; Katsuragi, Y. Diacylglycerol Enhances the Effects of Alpha-Linolenic Acid Against Visceral Fat: A Double-Blind Randomized Controlled Trial. Obesity 2017, 25, 1667–1675. [Google Scholar] [CrossRef]
- Pinder, R.M.; Brogden, R.N.; Sawyer, P.R.; Speight, T.M.; Spencer, R.; Avery, G.S. Carbenoxolone: A review of its pharmacological properties and therapeutic efficacy in peptic ulcer disease. Drugs 1976, 11, 245–307. [Google Scholar] [CrossRef]
- Aoki, F.; Nakagawa, K.; Kitano, M.; Ikematsu, H.; Nakamura, K.; Yokota, S.; Tominaga, Y.; Arai, N.; Mae, T. Clinical safety of licorice flavonoid oil (LFO) and pharmacokinetics of glabridin in healthy humans. J. Am. Coll. Nutr. 2007, 26, 209–218. [Google Scholar] [CrossRef]
- Hajiaghamohammadi, A.A.; Ziaee, A.; Samimi, R. The efficacy of licorice root extract in decreasing transaminase activities in non-alcoholic fatty liver disease: A randomized controlled clinical trial. Phytother. Res. 2012, 26, 1381–1384. [Google Scholar] [CrossRef] [PubMed]
- Mirtaheri, E.; Namazi, N.; Alizadeh, M.; Sargheini, N.; Karimi, S. Effects of dried licorice extract with low-calorie diet on lipid profile and atherogenic indices in overweight and obese subjects: A randomized controlled clinical trial. Eur. J. Integr. Med. 2015, 7, 287–293. [Google Scholar] [CrossRef]
- Leskinen, M.H.; Hautaniemi, E.J.; Tahvanainen, A.M.; Koskela, J.K.; Paallysaho, M.; Tikkakoski, A.J.; Kahonen, M.; Koobi, T.; Niemela, O.; Mustonen, J.; et al. Daily liquorice consumption for two weeks increases augmentation index and central systolic and diastolic blood pressure. PLoS ONE 2014, 9, e105607. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.; Lee, H.; Jang, J.; Kim, S.; Ha, T. Anti-obesity effects of glabridin-rich supercritical carbon dioxide extract of licorice in high-fat-fed obese mice. Food Chem. Toxicol. 2013, 51, 439–445. [Google Scholar] [CrossRef]
- Lee, J.W.; Choe, S.S.; Jang, H.; Kim, J.; Jeong, H.W.; Jo, H.; Jeong, K.H.; Tadi, S.; Park, M.G.; Kwak, T.H.; et al. AMPK activation with glabridin ameliorates adiposity and lipid dysregulation in obesity. J. Lipid Res. 2012, 53, 1277–1286. [Google Scholar] [CrossRef]
- Luis, A.; Domingues, F.; Pereira, L. Metabolic changes after licorice consumption: A systematic review with meta-analysis and trial sequential analysis of clinical trials. Phytomedicine 2018, 39, 17–24. [Google Scholar] [CrossRef]
- Alizadeh, M.; Namazi, N.; Mirtaheri, E.; Sargheini, N.; Kheirouri, S. Changes of Insulin Resistance and Adipokines Following Supplementation with Glycyrrhiza Glabra L. Extract in Combination with a Low-Calorie Diet in Overweight and Obese Subjects: A Randomized Double Blind Clinical Trial. Adv. Pharm. Bull. 2018, 8, 123–130. [Google Scholar] [CrossRef]
- Lopez, H.L.; Ziegenfuss, T.N.; Hofheins, J.E.; Habowski, S.M.; Arent, S.M.; Weir, J.P.; Ferrando, A.A. Eight weeks of supplementation with a multi-ingredient weight loss product enhances body composition, reduces hip and waist girth, and increases energy levels in overweight men and women. J. Int. Soc. Sports Nutr. 2013, 10, 22. [Google Scholar] [CrossRef]
- Watson, J.A.; Lowenstein, J.M. Citrate and the conversion of carbohydrate into fat. Fatty acid synthesis by a combination of cytoplasm and mitochondria. J. Biol. Chem. 1970, 245, 5993–6002. [Google Scholar]
- Liu, G.; Han, N.; Han, J.; Chen, D.; Kang, J.; Ma, H. Garcinia Cambogia Extracts Prevented Fat Accumulation via Adiponectin-AMPK Signaling Pathway in Developing Obesity Rats. Food Sci. Technol. Res. 2015, 21, 835–845. [Google Scholar] [CrossRef]
- Haber, S.L.; Awwad, O.; Phillips, A.; Park, A.E.; Pham, T.M. Garcinia cambogia for weight loss. Am. J. Health Syst. Pharm. 2018, 75, 17–22. [Google Scholar] [CrossRef]
- Mattes, R.D.; Bormann, L. Effects of (−)-hydroxycitric acid on appetitive variables. Phys. Behav. 2000, 71, 87–94. [Google Scholar] [CrossRef]
- Heymsfield, S.B.; Allison, D.B.; Vasselli, J.R.; Pietrobelli, A.; Greenfield, D.; Nunez, C. Garcinia cambogia (hydroxycitric acid) as a potential antiobesity agent: A randomized controlled trial. JAMA 1998, 280, 1596–1600. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, E.M.; Westerterp-Plantenga, M.S.; Saris, W.H. The effects of 2-week ingestion of (—)-hydroxycitrate and (—)-hydroxycitrate combined with medium-chain triglycerides on satiety, fat oxidation, energy expenditure and body weight. Int. J. Obes. Relat. Metab. Disord. 2001, 25, 1087–1094. [Google Scholar] [CrossRef] [PubMed]
- Hayamizu, K.; Ishii, Y.; Kaneko, I.; Shen, M.; Okuhara, Y.; Shigematsu, N.; Tomi, H.; Furuse, M.; Yoshino, G.; Shimasaki, H. Effects of Garcinia cambogia (Hydroxycitric Acid) on visceral fat accumulation: A double-blind, randomized, placebo-controlled trial. Curr. Ther. Res. Clin. Exp. 2003, 64, 551–567. [Google Scholar] [CrossRef] [PubMed]
- Preuss, H.G.; Bagchi, D.; Bagchi, M.; Rao, C.V.; Dey, D.K.; Satyanarayana, S. Effects of a natural extract of (-)-hydroxycitric acid (HCA-SX) and a combination of HCA-SX plus niacin-bound chromium and Gymnema sylvestre extract on weight loss. Diabetes Obes. Metab. 2004, 6, 171–180. [Google Scholar] [CrossRef]
- Kim, J.E.; Jeon, S.M.; Park, K.H.; Lee, W.S.; Jeong, T.S.; McGregor, R.A.; Choi, M.S. Does Glycine max leaves or Garcinia Cambogia promote weight-loss or lower plasma cholesterol in overweight individuals: A randomized control trial. Nutr. J. 2011, 10, 94. [Google Scholar] [CrossRef] [PubMed]
- Vasques, C.A.; Schneider, R.; Klein-Junior, L.C.; Falavigna, A.; Piazza, I.; Rossetto, S. Hypolipemic effect of Garcinia cambogia in obese women. Phytother. Res. 2014, 28, 887–891. [Google Scholar] [CrossRef]
- Food and Drug Administration. Warning on Hydroxycut Products. Available online: www.fda.gov/ForConsumers/ConsumerUpdates/ucm152152.htm (accessed on 17 June 2020).
- Guven, B.; Sag, E.; Karaguzel, G.; Cakir, M. Acute liver failure associated with metabolic diseases: A 10-year single-center experience. Pediatr. Int. 2020. [Google Scholar] [CrossRef]
- Melendez-Rosado, J.; Snipelisky, D.; Matcha, G.; Stancampiano, F. Acute hepatitis induced by pure Garcinia cambogia. J. Clin. Gastroenterol. 2015, 49, 449–450. [Google Scholar] [CrossRef]
- Lunsford, K.E.; Bodzin, A.S.; Reino, D.C.; Wang, H.L.; Busuttil, R.W. Dangerous dietary supplements: Garcinia cambogia-associated hepatic failure requiring transplantation. World J. Gastroenterol. 2016, 22, 10071–10076. [Google Scholar] [CrossRef] [PubMed]
- Yousaf, M.N.; Chaudhary, F.S.; Hodanazari, S.M.; Sittambalam, C.D. Hepatotoxicity associated with Garcinia cambogia: A case report. World J. Hepatol. 2019, 11, 735–742. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Akagi, E.; Njie, A.; Goyal, S.; Arsene, C.; Krishnamoorthy, G.; Ehrinpreis, M. Acute Hepatitis due to Garcinia Cambogia Extract, an Herbal Weight Loss Supplement. Case Rep. Gastrointest. Med. 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Licata, A.; Minissale, M.G. Weight-loss supplementation and acute liver failure: The case of Garcinia Cambogia. Intern. Emerg. Med. 2018, 13, 833–835. [Google Scholar] [CrossRef] [PubMed]
- Timmers, S.; Konings, E.; Bilet, L.; Houtkooper, R.H.; van de Weijer, T.; Goossens, G.H.; Hoeks, J.; van der Krieken, S.; Ryu, D.; Kersten, S.; et al. Calorie restriction-like effects of 30 days of resveratrol supplementation on energy metabolism and metabolic profile in obese humans. Cell Metab. 2011, 14, 612–622. [Google Scholar] [CrossRef]
- Salehi, B.; Mishra, A.P.; Nigam, M.; Sener, B.; Kilic, M.; Sharifi-Rad, M.; Fokou, P.V.T.; Martins, N.; Sharifi-Rad, J. Resveratrol: A Double-Edged Sword in Health Benefits. Biomedicines 2018, 6, 91. [Google Scholar] [CrossRef]
- Pearson, K.J.; Baur, J.A.; Lewis, K.N.; Peshkin, L.; Price, N.L.; Labinskyy, N.; Swindell, W.R.; Kamara, D.; Minor, R.K.; Perez, E.; et al. Resveratrol delays age-related deterioration and mimics transcriptional aspects of dietary restriction without extending life span. Cell Metab. 2008, 8, 157–168. [Google Scholar] [CrossRef]
- Lagouge, M.; Argmann, C.; Gerhart-Hines, Z.; Meziane, H.; Lerin, C.; Daussin, F.; Messadeq, N.; Milne, J.; Lambert, P.; Elliott, P.; et al. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1alpha. Cell 2006, 127, 1109–1122. [Google Scholar] [CrossRef]
- Baile, C.A.; Yang, J.Y.; Rayalam, S.; Hartzell, D.L.; Lai, C.Y.; Andersen, C.; Della-Fera, M.A. Effect of resveratrol on fat mobilization. Ann. N. Y. Acad. Sci. 2011, 1215, 40–47. [Google Scholar] [CrossRef]
- Poulsen, M.M.; Vestergaard, P.F.; Clasen, B.F.; Radko, Y.; Christensen, L.P.; Stodkilde-Jorgensen, H.; Moller, N.; Jessen, N.; Pedersen, S.B.; Jorgensen, J.O. High-dose resveratrol supplementation in obese men: An investigator-initiated, randomized, placebo-controlled clinical trial of substrate metabolism, insulin sensitivity, and body composition. Diabetes 2013, 62, 1186–1195. [Google Scholar] [CrossRef]
- Yoshino, J.; Conte, C.; Fontana, L.; Mittendorfer, B.; Imai, S.; Schechtman, K.B.; Gu, C.; Kunz, I.; Rossi Fanelli, F.; Patterson, B.W.; et al. Resveratrol supplementation does not improve metabolic function in nonobese women with normal glucose tolerance. Cell Metab. 2012, 16, 658–664. [Google Scholar] [CrossRef] [PubMed]
- Steck, S.E.; Chalecki, A.M.; Miller, P.; Conway, J.; Austin, G.L.; Hardin, J.W.; Albright, C.D.; Thuillier, P. Conjugated linoleic acid supplementation for twelve weeks increases lean body mass in obese humans. J. Nutr. 2007, 137, 1188–1193. [Google Scholar] [CrossRef] [PubMed]
- French, P.; Stanton, C.; Lawless, F.; O’Riordan, E.G.; Monahan, F.J.; Caffrey, P.J.; Moloney, A.P. Fatty acid composition, including conjugated linoleic acid, of intramuscular fat from steers offered grazed grass, grass silage, or concentrate-based diets. J. Anim. Sci. 2000, 78, 2849–2855. [Google Scholar] [CrossRef] [PubMed]
- Kelly, M.L.; Berry, J.R.; Dwyer, D.A.; Griinari, J.M.; Chouinard, P.Y.; Van Amburgh, M.E.; Bauman, D.E. Dietary fatty acid sources affect conjugated linoleic acid concentrations in milk from lactating dairy cows. J. Nutr. 1998, 128, 881–885. [Google Scholar] [CrossRef] [PubMed]
- Daley, C.A.; Abbott, A.; Doyle, P.S.; Nader, G.A.; Larson, S. A review of fatty acid profiles and antioxidant content in grass-fed and grain-fed beef. Nutr. J. 2010, 9, 10. [Google Scholar] [CrossRef] [PubMed]
- Scollan, N.D.; Price, E.M.; Morgan, S.A.; Huws, S.A.; Shingfield, K.J. Can we improve the nutritional quality of meat? Proc. Nutr. Soc. 2017, 76, 603–618. [Google Scholar] [CrossRef] [PubMed]
- Duckett, S.K.; Wagner, D.G.; Yates, L.D.; Dolezal, H.G.; May, S.G. Effects of time on feed on beef nutrient composition. J. Anim. Sci. 1993, 71, 2079–2088. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kim, Y.; Kim, Y.J.; Park, Y. Conjugated Linoleic Acid: Potential Health Benefits as a Functional Food Ingredient. Annu. Rev. Food Sci. Technol. 2016, 7, 221–244. [Google Scholar] [CrossRef]
- Hassan Eftekhari, M.; Aliasghari, F.; Babaei-Beigi, M.A.; Hasanzadeh, J. Effect of conjugated linoleic acid and omega-3 fatty acid supplementation on inflammatory and oxidative stress markers in atherosclerotic patients. ARYA Atheroscler. 2013, 9, 311–318. [Google Scholar]
- Mozaffarian, D.; Katan, M.B.; Ascherio, A.; Stampfer, M.J.; Willett, W.C. Trans fatty acids and cardiovascular disease. N. Engl. J. Med. 2006, 354, 1601–1613. [Google Scholar] [CrossRef]
- Jakobsen, M.U.; Overvad, K.; Dyerberg, J.; Heitmann, B.L. Intake of ruminant trans fatty acids and risk of coronary heart disease. Int. J. Epidemiol. 2008, 37, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Blankson, H.; Stakkestad, J.A.; Fagertun, H.; Thom, E.; Wadstein, J.; Gudmundsen, O. Conjugated linoleic acid reduces body fat mass in overweight and obese humans. J. Nutr. 2000, 130, 2943–2948. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Lim, H.R.; Lee, H.; Lee, H.; Kang, W.; Kim, E. The effects of conjugated linoleic acid (CLA) on metabolic syndrome patients: A systematic review and meta-analysis. J. Funct. Foods 2016, 25, 588–598. [Google Scholar] [CrossRef]
- Den Hartigh, L.J.; Han, C.Y.; Wang, S.; Omer, M.; Chait, A. 10E,12Z-conjugated linoleic acid impairs adipocyte triglyceride storage by enhancing fatty acid oxidation, lipolysis, and mitochondrial reactive oxygen species. J. Lipid Res. 2013, 54, 2964–2978. [Google Scholar] [CrossRef]
- Shen, W.; Chuang, C.C.; Martinez, K.; Reid, T.; Brown, J.M.; Xi, L.; Hixson, L.; Hopkins, R.; Starnes, J.; McIntosh, M. Conjugated linoleic acid reduces adiposity and increases markers of browning and inflammation in white adipose tissue of mice. J. Lipid Res. 2013, 54, 909–922. [Google Scholar] [CrossRef]
- Shen, W.; Baldwin, J.; Collins, B.; Hixson, L.; Lee, K.T.; Herberg, T.; Starnes, J.; Cooney, P.; Chuang, C.C.; Hopkins, R.; et al. Low level of trans-10, cis-12 conjugated linoleic acid decreases adiposity and increases browning independent of inflammatory signaling in overweight Sv129 mice. J. Nutr. Biochem. 2015, 26, 616–625. [Google Scholar] [CrossRef]
- Marques, T.M.; Wall, R.; O’Sullivan, O.; Fitzgerald, G.F.; Shanahan, F.; Quigley, E.M.; Cotter, P.D.; Cryan, J.F.; Dinan, T.G.; Ross, R.P.; et al. Dietary trans-10, cis-12-conjugated linoleic acid alters fatty acid metabolism and microbiota composition in mice. Br. J. Nutr. 2015, 113, 728–738. [Google Scholar] [CrossRef]
- den Hartigh, L.J.; Gao, Z.; Goodspeed, L.; Wang, S.; Das, A.K.; Burant, C.F.; Chait, A.; Blaser, M.J. Obese Mice Losing Weight Due to trans-10,cis-12 Conjugated Linoleic Acid Supplementation or Food Restriction Harbor Distinct Gut Microbiota. J. Nutr. 2018, 148, 562–572. [Google Scholar] [CrossRef]
- Namazi, N.; Irandoost, P.; Larijani, B.; Azadbakht, L. The effects of supplementation with conjugated linoleic acid on anthropometric indices and body composition in overweight and obese subjects: A systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2019, 59, 2720–2733. [Google Scholar] [CrossRef]
- Den Hartigh, L.J. Conjugated Linoleic Acid Effects on Cancer, Obesity, and Atherosclerosis: A Review of Pre-Clinical and Human Trials with Current Perspectives. Nutrients 2019, 11, 370. [Google Scholar] [CrossRef]
- Pothuraju, R.; Sharma, R.K.; Onteru, S.K.; Singh, S.; Hussain, S.A. Hypoglycemic and Hypolipidemic Effects of Aloe vera Extract Preparations: A Review. Phytother. Res. 2016, 30, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Shakib, Z.; Shahraki, N.; Razavi, B.M.; Hosseinzadeh, H. Aloe vera as an herbal medicine in the treatment of metabolic syndrome: A review. Phytother. Res. 2019, 33, 2649–2660. [Google Scholar] [CrossRef] [PubMed]
- Shin, E.; Shin, S.; Kong, H.; Lee, S.; Do, S.G.; Jo, T.H.; Park, Y.I.; Lee, C.K.; Hwang, I.K.; Kim, K. Dietary Aloe Reduces Adipogenesis via the Activation of AMPK and Suppresses Obesity-related Inflammation in Obese Mice. Immune Netw. 2011, 11, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Pothuraju, R.; Sharma, R.K.; Kavadi, P.K.; Chagalamarri, J.; Jangra, S.; Bhakri, G.; De, S. Anti-obesity effect of milk fermented by Lactobacillus plantarum NCDC 625 alone and in combination with herbs on high fat diet fed C57BL/6J mice. Benef. Microbes 2016, 7, 375–385. [Google Scholar] [CrossRef]
- Jain, A.; Gupta, Y.; Jain, S.K. Perspectives of biodegradable natural polysaccharides for site-specific drug delivery to the colon. J. Pharm. Pharm. Sci. 2007, 10, 86–128. [Google Scholar]
- Choi, H.C.; Kim, S.J.; Son, K.Y.; Oh, B.J.; Cho, B.L. Metabolic effects of aloe vera gel complex in obese prediabetes and early non-treated diabetic patients: Randomized controlled trial. Nutrition 2013, 29, 1110–1114. [Google Scholar] [CrossRef]
- Cunnane, S.C.; Ganguli, S.; Menard, C.; Liede, A.C.; Hamadeh, M.J.; Chen, Z.Y.; Wolever, T.M.; Jenkins, D.J. High alpha-linolenic acid flaxseed (Linum usitatissimum): Some nutritional properties in humans. Br. J. Nutr. 1993, 69, 443–453. [Google Scholar] [CrossRef]
- Pan, A.; Yu, D.; Demark-Wahnefried, W.; Franco, O.H.; Lin, X. Meta-analysis of the effects of flaxseed interventions on blood lipids. Am. J. Clin. Nutr. 2009, 90, 288–297. [Google Scholar] [CrossRef]
- Goyal, A.; Sharma, V.; Upadhyay, N.; Gill, S.; Sihag, M. Flax and flaxseed oil: An ancient medicine & modern functional food. J. Food Sci. Technol. 2014, 51, 1633–1653. [Google Scholar] [CrossRef]
- Ursoniu, S.; Sahebkar, A.; Andrica, F.; Serban, C.; Banach, M.; Lipid and Blood Pressure Meta-analysis Collaboration. Effects of flaxseed supplements on blood pressure: A systematic review and meta-analysis of controlled clinical trial. Clin. Nutr. 2016, 35, 615–625. [Google Scholar] [CrossRef]
- Hadi, A.; Askarpour, M.; Salamat, S.; Ghaedi, E.; Symonds, M.E.; Miraghajani, M. Effect of flaxseed supplementation on lipid profile: An updated systematic review and dose-response meta-analysis of sixty-two randomized controlled trials. Pharmacol. Res. 2020, 152, 104622. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi-Sartang, M.; Sohrabi, Z.; Barati-Boldaji, R.; Raeisi-Dehkordi, H.; Mazloom, Z. Flaxseed supplementation on glucose control and insulin sensitivity: A systematic review and meta-analysis of 25 randomized, placebo-controlled trials. Nutr. Rev. 2018, 76, 125–139. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi-Sartang, M.; Mazloom, Z.; Raeisi-Dehkordi, H.; Barati-Boldaji, R.; Bellissimo, N.; Totosy de Zepetnek, J.O. The effect of flaxseed supplementation on body weight and body composition: A systematic review and meta-analysis of 45 randomized placebo-controlled trials. Obes. Rev. 2017, 18, 1096–1107. [Google Scholar] [CrossRef] [PubMed]
- Fukumitsu, S.; Aida, K.; Ueno, N.; Ozawa, S.; Takahashi, Y.; Kobori, M. Flaxseed lignan attenuates high-fat diet-induced fat accumulation and induces adiponectin expression in mice. Br. J. Nutr. 2008, 100, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, M.; Jensen, M.G. Dietary fibres in the regulation of appetite and food intake. Importance of viscosity. Appetite 2011, 56, 65–70. [Google Scholar] [CrossRef]
- McCullough, R.S.; Edel, A.L.; Bassett, C.M.; Lavallee, R.K.; Dibrov, E.; Blackwood, D.P.; Ander, B.P.; Pierce, G.N. The alpha linolenic acid content of flaxseed is associated with an induction of adipose leptin expression. Lipids 2011, 46, 1043–1052. [Google Scholar] [CrossRef]
- Luo, J.; Qi, J.; Wang, W.; Luo, Z.; Liu, L.; Zhang, G.; Zhou, Q.; Liu, J.; Peng, X. Antiobesity Effect of Flaxseed Polysaccharide via Inducing Satiety due to Leptin Resistance Removal and Promoting Lipid Metabolism through the AMP-Activated Protein Kinase (AMPK) Signaling Pathway. J. Agric. Food Chem. 2019, 67, 7040–7049. [Google Scholar] [CrossRef]
- Buckley, J.D.; Howe, P.R. Long-chain omega-3 polyunsaturated fatty acids may be beneficial for reducing obesity-a review. Nutrients 2010, 2, 1212–1230. [Google Scholar] [CrossRef]
- Onakpoya, I.; O’Sullivan, J.; Heneghan, C.; Thompson, M. The effect of grapefruits (Citrus paradisi) on body weight and cardiovascular risk factors: A systematic review and meta-analysis of randomized clinical trials. Crit. Rev. Food Sci. Nutr. 2017, 57, 602–612. [Google Scholar] [CrossRef]
- Dow, C.A.; Going, S.B.; Chow, H.H.; Patil, B.S.; Thomson, C.A. The effects of daily consumption of grapefruit on body weight, lipids, and blood pressure in healthy, overweight adults. Metabolism 2012, 61, 1026–1035. [Google Scholar] [CrossRef]
- Zunino, S. Type 2 diabetes and glycemic response to grapes or grape products. J. Nutr. 2009, 139, 1794S–1800S. [Google Scholar] [CrossRef] [PubMed]
- Pu, P.; Gao, D.M.; Mohamed, S.; Chen, J.; Zhang, J.; Zhou, X.Y.; Zhou, N.J.; Xie, J.; Jiang, H. Naringin ameliorates metabolic syndrome by activating AMP-activated protein kinase in mice fed a high-fat diet. Arch. Biochem. Biophys. 2012, 518, 61–70. [Google Scholar] [CrossRef]
- Gupta, R.; Guptha, S. Strategies for initial management of hypertension. Indian J. Med. Res. 2010, 132, 531–542. [Google Scholar] [PubMed]
- Dallas, C.; Gerbi, A.; Tenca, G.; Juchaux, F.; Bernard, F.X. Lipolytic effect of a polyphenolic citrus dry extract of red orange, grapefruit, orange (SINETROL) in human body fat adipocytes. Mechanism of action by inhibition of cAMP-phosphodiesterase (PDE). Phytomedicine 2008, 15, 783–792. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.W.; Kim, Y.O.; Andrade, J.E.; Burgess, J.R.; Kim, Y.C. Dietary naringenin increases hepatic peroxisome proliferators-activated receptor alpha protein expression and decreases plasma triglyceride and adiposity in rats. Eur. J. Nutr. 2011, 50, 81–88. [Google Scholar] [CrossRef]
- Suttirak, W.; Manurakchinakorn, S. In vitro antioxidant properties of mangosteen peel extract. J. Food Sci. Technol. 2014, 51, 3546–3558. [Google Scholar] [CrossRef]
- Chae, H.S.K.E.Y.H.L.; Kim, N.R.; Chin, Y.W. Xanthones with pancreatic lipase inhibitory activity from the pericarps of Garcinia mangostana L. (Guttiferae). Eur. J. Lipid Sci. Technol. 2016, 118, 1416–1421. [Google Scholar] [CrossRef]
- Quan, X.; Wang, Y.; Ma, X.; Liang, Y.; Tian, W.; Ma, Q.; Jiang, H.; Zhao, Y. alpha-Mangostin induces apoptosis and suppresses differentiation of 3T3-L1 cells via inhibiting fatty acid synthase. PLoS ONE 2012, 7, e33376. [Google Scholar] [CrossRef]
- Jariyapongskul, A.; Areebambud, C.; Suksamrarn, S.; Mekseepralard, C. Alpha-mangostin attenuation of hyperglycemia-induced ocular hypoperfusion and blood retinal barrier leakage in the early stage of type 2 diabetes rats. Biomed. Res. Int. 2015, 2015, 785826. [Google Scholar] [CrossRef]
- Ryu, H.W.; Cho, J.K.; Curtis-Long, M.J.; Yuk, H.J.; Kim, Y.S.; Jung, S.; Kim, Y.S.; Lee, B.W.; Park, K.H. alpha-Glucosidase inhibition and antihyperglycemic activity of prenylated xanthones from Garcinia mangostana. Phytochemistry 2011, 72, 2148–2154. [Google Scholar] [CrossRef]
- Taher, M.; Tg Zakaria, T.M.; Susanti, D.; Zakaria, Z.A. Hypoglycaemic activity of ethanolic extract of Garcinia mangostana Linn. in normoglycaemic and streptozotocin-induced diabetic rats. BMC Complement. Altern. Med. 2016, 16, 135. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.H.; Bae, J.K.; Chae, H.S.; Kim, Y.M.; Sreymom, Y.; Han, L.; Jang, H.Y.; Chin, Y.W. alpha-Mangostin Regulates Hepatic Steatosis and Obesity through SirT1-AMPK and PPARgamma Pathways in High-Fat Diet-Induced Obese Mice. J. Agric. Food Chem. 2015, 63, 8399–8406. [Google Scholar] [CrossRef] [PubMed]
- Chae, H.S.; Kim, Y.M.; Bae, J.K.; Sorchhann, S.; Yim, S.; Han, L.; Paik, J.H.; Choi, Y.H.; Chin, Y.W. Mangosteen Extract Attenuates the Metabolic Disorders of High-Fat-Fed Mice by Activating AMPK. J. Med. Food 2016, 19, 148–154. [Google Scholar] [CrossRef]
- Xie, Z.; Sintara, M.; Chang, T.; Ou, B. Daily consumption of a mangosteen-based drink improves in vivo antioxidant and anti-inflammatory biomarkers in healthy adults: A randomized, double-blind, placebo-controlled clinical trial. Food Sci. Nutr. 2015, 3, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Udani, J.K.; Singh, B.B.; Barrett, M.L.; Singh, V.J. Evaluation of Mangosteen juice blend on biomarkers of inflammation in obese subjects: A pilot, dose finding study. Nutr. J. 2009, 8, 48. [Google Scholar] [CrossRef]
- Stern, J.S.; Peerson, J.; Mishra, A.T.; Sadasiva Rao, M.V.; Rajeswari, K.P. Efficacy and tolerability of a novel herbal formulation for weight management. Obesity 2013, 21, 921–927. [Google Scholar] [CrossRef]
- Kudiganti, V.; Kodur, R.R.; Kodur, S.R.; Halemane, M.; Deep, D.K. Efficacy and tolerability of Meratrim for weight management: A randomized, double-blind, placebo-controlled study in healthy overweight human subjects. Lipids Health Dis. 2016, 15, 136. [Google Scholar] [CrossRef][Green Version]
- Watanabe, M.; Gangitano, E.; Francomano, D.; Addessi, E.; Toscano, R.; Costantini, D.; Tuccinardi, D.; Mariani, S.; Basciani, S.; Spera, G.; et al. Mangosteen extract shows a potent insulin sensitizing effect in obese female patients: A prospective randomized controlled pilot study. Nutrients 2018, 10, 586. [Google Scholar] [CrossRef]
- Havel, P.J. A scientific review: The role of chromium in insulin resistance. Diabetes Educ. 2004, 30 (Suppl. 3), 1–14. [Google Scholar]
- Wang, Z.Q.; Qin, J.; Martin, J.; Zhang, X.H.; Sereda, O.; Anderson, R.A.; Pinsonat, P.; Cefalu, W.T. Phenotype of subjects with type 2 diabetes mellitus may determine clinical response to chromium supplementation. Metabolism 2007, 56, 1652–1655. [Google Scholar] [CrossRef]
- Tsang, C.; Taghizadeh, M.; Aghabagheri, E.; Asemi, Z.; Jafarnejad, S. A meta-analysis of the effect of chromium supplementation on anthropometric indices of subjects with overweight or obesity. Clin. Obes. 2019, 9, e12313. [Google Scholar] [CrossRef] [PubMed]
- Onakpoya, I.; Posadzki, P.; Ernst, E. Chromium supplementation in overweight and obesity: A systematic review and meta-analysis of randomized clinical trials. Obes. Rev. 2013, 14, 496–507. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Cotillard, A.; Vatier, C.; Bastard, J.P.; Fellahi, S.; Stevant, M.; Allatif, O.; Langlois, C.; Bieuvelet, S.; Brochot, A.; et al. A Dietary Supplement Containing Cinnamon, Chromium and Carnosine Decreases Fasting Plasma Glucose and Increases Lean Mass in Overweight or Obese Pre-Diabetic Subjects: A Randomized, Placebo-Controlled Trial. PLoS ONE 2015, 10, e0138646. [Google Scholar] [CrossRef] [PubMed]
- Whitfield, P.; Parry-Strong, A.; Walsh, E.; Weatherall, M.; Krebs, J.D. The effect of a cinnamon-, chromium- and magnesium-formulated honey on glycaemic control, weight loss and lipid parameters in type 2 diabetes: An open-label cross-over randomised controlled trial. Eur. J. Nutr. 2016, 55, 1123–1131. [Google Scholar] [CrossRef]
- Cerulli, J.; Grabe, D.W.; Gauthier, I.; Malone, M.; McGoldrick, M.D. Chromium picolinate toxicity. Ann. Pharmacother 1998, 32, 428–431. [Google Scholar] [CrossRef]
- Grant, K.E.; Chandler, R.M.; Castle, A.L.; Ivy, J.L. Chromium and exercise training: Effect on obese women. Med. Sci. Sports Exerc. 1997, 29, 992–998. [Google Scholar] [CrossRef]
- Hockney, R.A.; Montgomery, P.; Williams, C.; Geddes, J.R.; Cowen, P.J. Lack of effect of chromium supplementation on mental state and body weight in people with schizophrenia. J. Clin. Psychopharmacol. 2006, 26, 544–545. [Google Scholar] [CrossRef]
- Maleki, V.; Izadi, A.; Farsad-Naeimi, A.; Alizadeh, M. Chromium supplementation does not improve weight loss or metabolic and hormonal variables in patients with polycystic ovary syndrome: A systematic review. Nutr. Res. 2018, 56, 1–10. [Google Scholar] [CrossRef]
- Smith, A.R.; Shenvi, S.V.; Widlansky, M.; Suh, J.H.; Hagen, T.M. Lipoic acid as a potential therapy for chronic diseases associated with oxidative stress. Curr. Med. Chem. 2004, 11, 1135–1146. [Google Scholar] [CrossRef]
- Scott, B.C.; Aruoma, O.I.; Evans, P.J.; O’Neill, C.; Van der Vliet, A.; Cross, C.E.; Tritschler, H.; Halliwell, B. Lipoic and dihydrolipoic acids as antioxidants. A critical evaluation. Free Radic. Res. 1994, 20, 119–133. [Google Scholar] [CrossRef]
- Liu, J.; Head, E.; Gharib, A.M.; Yuan, W.; Ingersoll, R.T.; Hagen, T.M.; Cotman, C.W.; Ames, B.N. Memory loss in old rats is associated with brain mitochondrial decay and RNA/DNA oxidation: Partial reversal by feeding acetyl-L-carnitine and/or R-alpha -lipoic acid. Proc. Natl. Acad. Sci. USA 2002, 99, 2356–2361. [Google Scholar] [CrossRef] [PubMed]
- Wollin, S.D.; Jones, P.J. Alpha-lipoic acid and cardiovascular disease. J. Nutr. 2003, 133, 3327–3330. [Google Scholar] [CrossRef]
- Estrada, D.E.; Ewart, H.S.; Tsakiridis, T.; Volchuk, A.; Ramlal, T.; Tritschler, H.; Klip, A. Stimulation of glucose uptake by the natural coenzyme alpha-lipoic acid/thioctic acid: Participation of elements of the insulin signaling pathway. Diabetes 1996, 45, 1798–1804. [Google Scholar] [CrossRef] [PubMed]
- Streeper, R.S.; Henriksen, E.J.; Jacob, S.; Hokama, J.Y.; Fogt, D.L.; Tritschler, H.J. Differential effects of lipoic acid stereoisomers on glucose metabolism in insulin-resistant skeletal muscle. Am. J. Physiol. 1997, 273, E185–E191. [Google Scholar] [CrossRef] [PubMed]
- Saengsirisuwan, V.; Perez, F.R.; Sloniger, J.A.; Maier, T.; Henriksen, E.J. Interactions of exercise training and alpha-lipoic acid on insulin signaling in skeletal muscle of obese Zucker rats. Am. J. Physiol. Endocrinol. Metab. 2004, 287, E529–E536. [Google Scholar] [CrossRef] [PubMed]
- Hughes, V.A.; Fiatarone, M.A.; Fielding, R.A.; Kahn, B.B.; Ferrara, C.M.; Shepherd, P.; Fisher, E.C.; Wolfe, R.R.; Elahi, D.; Evans, W.J. Exercise increases muscle GLUT-4 levels and insulin action in subjects with impaired glucose tolerance. Am. J. Physiol. 1993, 264, E855–E862. [Google Scholar] [CrossRef]
- Jacob, S.; Streeper, R.S.; Fogt, D.L.; Hokama, J.Y.; Tritschler, H.J.; Dietze, G.J.; Henriksen, E.J. The antioxidant alpha-lipoic acid enhances insulin-stimulated glucose metabolism in insulin-resistant rat skeletal muscle. Diabetes 1996, 45, 1024–1029. [Google Scholar] [CrossRef]
- Konrad, T.; Vicini, P.; Kusterer, K.; Hoflich, A.; Assadkhani, A.; Bohles, H.J.; Sewell, A.; Tritschler, H.J.; Cobelli, C.; Usadel, K.H. alpha-Lipoic acid treatment decreases serum lactate and pyruvate concentrations and improves glucose effectiveness in lean and obese patients with type 2 diabetes. Diabetes Care 1999, 22, 280–287. [Google Scholar] [CrossRef]
- Kucukgoncu, S.; Zhou, E.; Lucas, K.B.; Tek, C. Alpha-lipoic acid (ALA) as a supplementation for weight loss: Results from a meta-analysis of randomized controlled trials. Obes. Rev. 2017, 18, 594–601. [Google Scholar] [CrossRef]
- Ziegler, D.; Hanefeld, M.; Ruhnau, K.J.; Meissner, H.P.; Lobisch, M.; Schutte, K.; Gries, F.A. Treatment of symptomatic diabetic peripheral neuropathy with the anti-oxidant alpha-lipoic acid. A 3-week multicentre randomized controlled trial (ALADIN Study). Diabetologia 1995, 38, 1425–1433. [Google Scholar] [CrossRef]
- Takeuchi, Y.; Miyamoto, T.; Kakizawa, T.; Shigematsu, S.; Hashizume, K. Insulin Autoimmune Syndrome possibly caused by alpha lipoic acid. Intern. Med. 2007, 46, 237–239. [Google Scholar] [CrossRef] [PubMed]
- Kass, D.A.; Duggal, P.; Cingolani, O. Obesity could shift severe COVID-19 disease to younger ages. Lancet 2020, 395, 1544–1545. [Google Scholar] [CrossRef]
- Watanabe, M.; Caruso, D.; Tuccinardi, D.; Risi, R.; Zerunian, M.; Polici, M.; Pucciarelli, F.; Tarallo, M.; Strigari, L.; Manfrini, S.; et al. Visceral fat shows the strongest association with the need of intensive Care in Patients with COVID-19. Metabolism 2020. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Risi, R.; Tuccinardi, D.; Baquero, C.J.; Manfrini, S.; Gnessi, L. Obesity and SARS-CoV-2: A population to safeguard. Diabetes Metab. Res. Rev. 2020, e3325. [Google Scholar] [CrossRef] [PubMed]
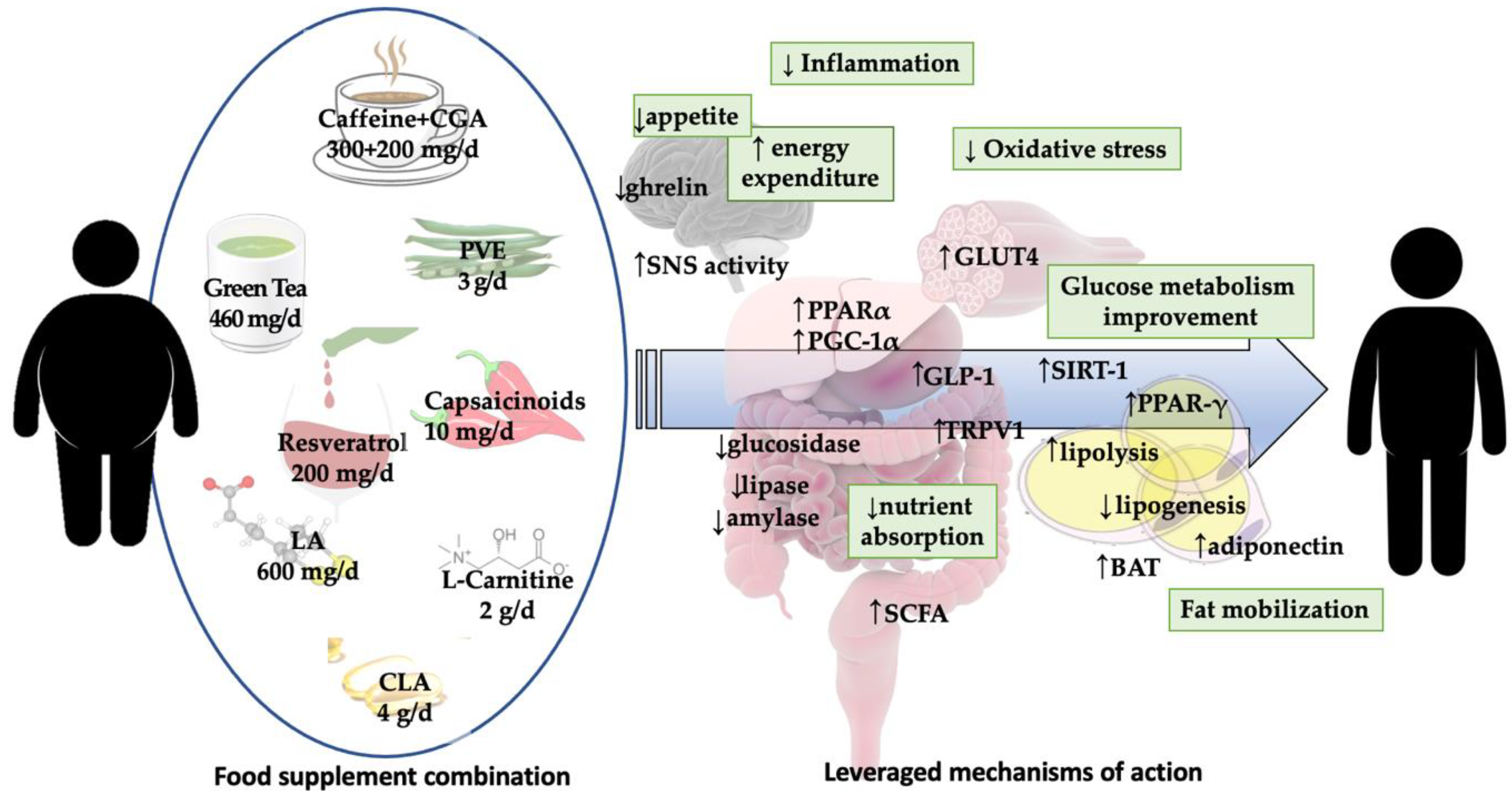
| Food Supplement | Dosage | Mechanisms of Action | Quality of Evidence | Side Effects | Recommendation |
|---|---|---|---|---|---|
| Green Tea | 100–460 mg/day | lipase, amylase, glucosidase inhibition *#; gut microbiota modification *# | moderate | None | A |
| Ginseng | 100 mg–18 g/day | lipase inhibition *#; appetite hormonwhie levels modification * | low | None | B |
| White Kidney Bean | 1–3 g/day | α-amylase inhibition *#; antioxidant *, anticarcinogenic *#, anti-inflammatory*#, glucose lowering *#, and cardioprotective properties *# | moderate | Lectin’s toxicity (abstent in phaseolus vulgaris extracts) | A |
| Chitosan | 1–4.5 g/day | absorption of dietary fats inhibition *#; decreased lipid peroxidation; adipogenesis inhibition * | low | Gastrointestinal discomfort and bloating | B |
| β-Glucans | 5–9 g/day | lipid and carbohydrate absorption inhibition *#; satiety induction *#; PYY-NPY axis activation *# | low | None | B |
| Psyllium | 3–10.5 g/day | carbohydrate absorption inhibition *#; decreased serum lipids *#; delayed gastric emptying *#; glucose control *#; satiety induction *# | low | Gastrointestinal discomfort and bloating | B |
| Glucomannan | 2–3 g/day | delayed gastric emptying *#, increased satiety *#; decreased post-prandial glucose concentration | low | None | B |
| Guar Gum | 9–30 g/day | increased postprandial fullness #, appetite and food intake reduction *# | low | Gastrointestinal discomfort, bloating, diarrhea | B |
| Agar | 180 g/day | delayed gastric emptying #; increased satiety *# | low | None | B |
| Inulin | 8–30 g/day | carbohydrate absorption inhibition *#; gut microbiota modulation *#; increased satiety *# | low | None | B |
| Food Supplement | Dosage | Mechanisms of Action | Quality of Evidence | Side Effects | Recommendation |
|---|---|---|---|---|---|
| Caralluma | 1 g/day | fatty acid synthesis inhibition and enhanced fatty acid oxidation *; ghrelin/neuropeptide Y expression inhibition *# | low | None | B |
| Spirulina | 1–4.5 g/day | decreased appetite *#; adipogenesis inhibition *; browning induction * | low | None | B |
| Whey protein | ∼100–600 g/week | increased satiety hormones; decreased appetite; enhanced fat mass oxidation; enhanced lean mass preservation *# | moderate | None | A |
| Coffee, caffeine and chlorogenic acids | 60 mg–1000 g/day | hunger suppression; energy expenditure stimulation; increased fat oxidation and brown adipose tissue activation *# | moderate | None | A |
| Bitter orange | 10–400 mg/day | suppressed appetite *#; energy expenditure and lipolysis increase *# | low | None | B |
| Guarana | 240–285 mg/day | decreased appetite #; increased energy expenditure and fat oxidation *#; adipogenesis inhibition *; browning induction * | low | Gastrointestinal discomfort; insomnia, migraine, tachycardia | B |
| Food Supplement | Dosage | Mechanisms of Action | Quality of Evidence | Recommendation | Side Effects |
|---|---|---|---|---|---|
| Capsaicin, capsaicinoids and capsinoids | 10–30 mg/day | browning, thermogenesis, fat oxidation *#, energy expenditure induction # | low | B | Gastrointestinal discomfort and diarrhea |
| Curcumin | 70 mg–12 g/day | adipogenesis inhibition *; insulin-sensitizing and anti-inflammatory properties *# | low | B | Gastrointestinal discomfort, headache, urticaria |
| L-Carnitine | 10 mg–4 g/day | increased energy expenditure and fat oxidation *#; improved insulin resistance *#; modulation of regulators of lipid catabolism or adipogenesis *; induction of satiety *# | low | B | None |
| Food Supplement | Dosage | Mechanisms of Action | Quality of Evidence | Side Effects | Recommendation |
|---|---|---|---|---|---|
| Pyruvate | 5–44 g/day | reduced insulin level and increased acetylCoA concentrations *# | low | None | B |
| Dyacilglycerol | 1.1–1.2 g/day | enhanced fat oxidation *#; reduced postprandial triglycerides *# | moderate | None | A |
| Licorice | 300–900 mg/day | reduced serum lipids *#; improved hepatic steatosis through beta-oxidation induction *# | low | Increased blood pressure, hypernatremia | C |
| Garcinia Gambogia | 400–2400 mg/day | decreased lipogenesis and increased lipolysis * | low | Hepatotoxicity, diarrhea | C |
| Resveratrol | 75–2000 mg/day | decreased adipogenesis; increased lipolysis;reduced lipogenesis *# | low | None | A |
| Conjugated linoleic acid | 1.5–6.8 g/day | decreased adipocytes size; inhibited adipogenesis; reducted lipogenesis; induced browining *; gut microbiota modification *#; | low | Occasional gastrointestinal discomfort | A |
| Aloe vera | 588–700 mg/day | Improved glucose and lipid metabolism *#; reduced oxidative stress * ; inhibited lipogenesis * | low | None | B |
| Flaxseed | 20–50 g/day | increased saxiety *# and lipolysis *; inhibited lipogenesis * | low | None | A |
| Grapefruit | 81–142 mg/day | improved glycemic control, enhanced insulin secretion and inhibited gluconeogenesis *#; increased fat oxidation and reduced lipogenesis * | low | Possible alteration of several drugs metabolism | B |
| Food Supplement | Dosage | Mechanisms of Action | Quality of Evidence | Side Effects | Recommendation |
|---|---|---|---|---|---|
| Mangosteen | 200–400 mg/day | inhibition of pancreatic lipase and fatty acid synthase *; improved glucose metabolism *# | Low | None | B |
| Chromium | 157–1000 μg/day | Energy expenditure increase *#; appetite suppression *#; improved glucose metabolism *# | Moderate | Diarrhea, vertigo, headache, urticaria | B |
| Lipoic Acid | 300–2400 mg/day | improved glucose metabolism *#; appetite suppression *#; increase of lipolysis and reduction of lipogenesis * | Low | Gastrointestinal disconfort, urticaria, hypoglycemia | B |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Watanabe, M.; Risi, R.; Masi, D.; Caputi, A.; Balena, A.; Rossini, G.; Tuccinardi, D.; Mariani, S.; Basciani, S.; Manfrini, S.; et al. Current Evidence to Propose Different Food Supplements for Weight Loss: A Comprehensive Review. Nutrients 2020, 12, 2873. https://doi.org/10.3390/nu12092873
Watanabe M, Risi R, Masi D, Caputi A, Balena A, Rossini G, Tuccinardi D, Mariani S, Basciani S, Manfrini S, et al. Current Evidence to Propose Different Food Supplements for Weight Loss: A Comprehensive Review. Nutrients. 2020; 12(9):2873. https://doi.org/10.3390/nu12092873
Chicago/Turabian StyleWatanabe, Mikiko, Renata Risi, Davide Masi, Alessandra Caputi, Angela Balena, Giovanni Rossini, Dario Tuccinardi, Stefania Mariani, Sabrina Basciani, Silvia Manfrini, and et al. 2020. "Current Evidence to Propose Different Food Supplements for Weight Loss: A Comprehensive Review" Nutrients 12, no. 9: 2873. https://doi.org/10.3390/nu12092873
APA StyleWatanabe, M., Risi, R., Masi, D., Caputi, A., Balena, A., Rossini, G., Tuccinardi, D., Mariani, S., Basciani, S., Manfrini, S., Gnessi, L., & Lubrano, C. (2020). Current Evidence to Propose Different Food Supplements for Weight Loss: A Comprehensive Review. Nutrients, 12(9), 2873. https://doi.org/10.3390/nu12092873










