L-Carnitine’s Effect on the Biomarkers of Metabolic Syndrome: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy and Data Collection
2.2. Inclusion and Exclusion Criteria
2.3. Data Extraction and Quality Assessment
2.4. Data Analysis
3. Results
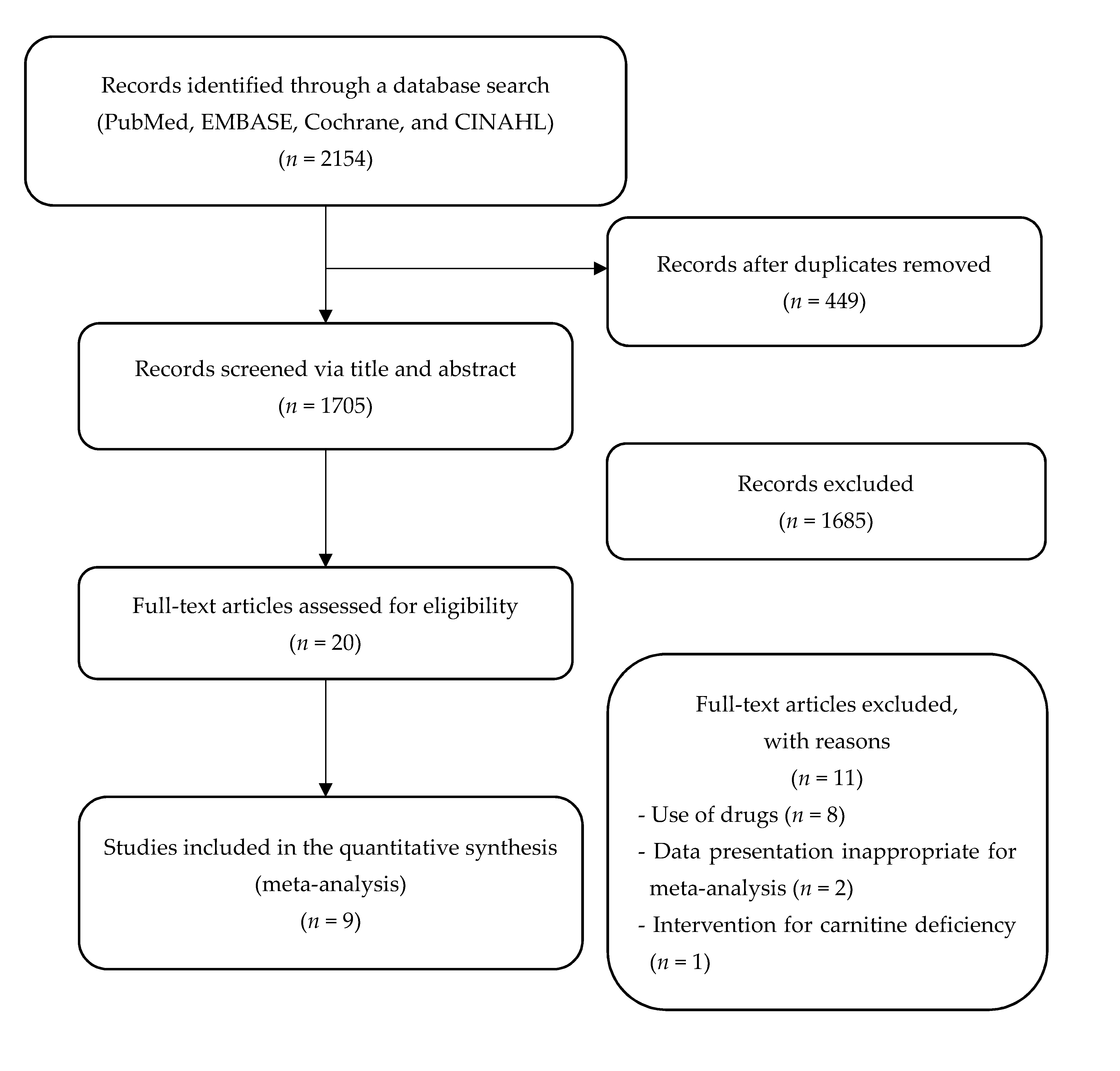
3.1. Study Selection
3.2. Characteristics of the Included RCT Studies
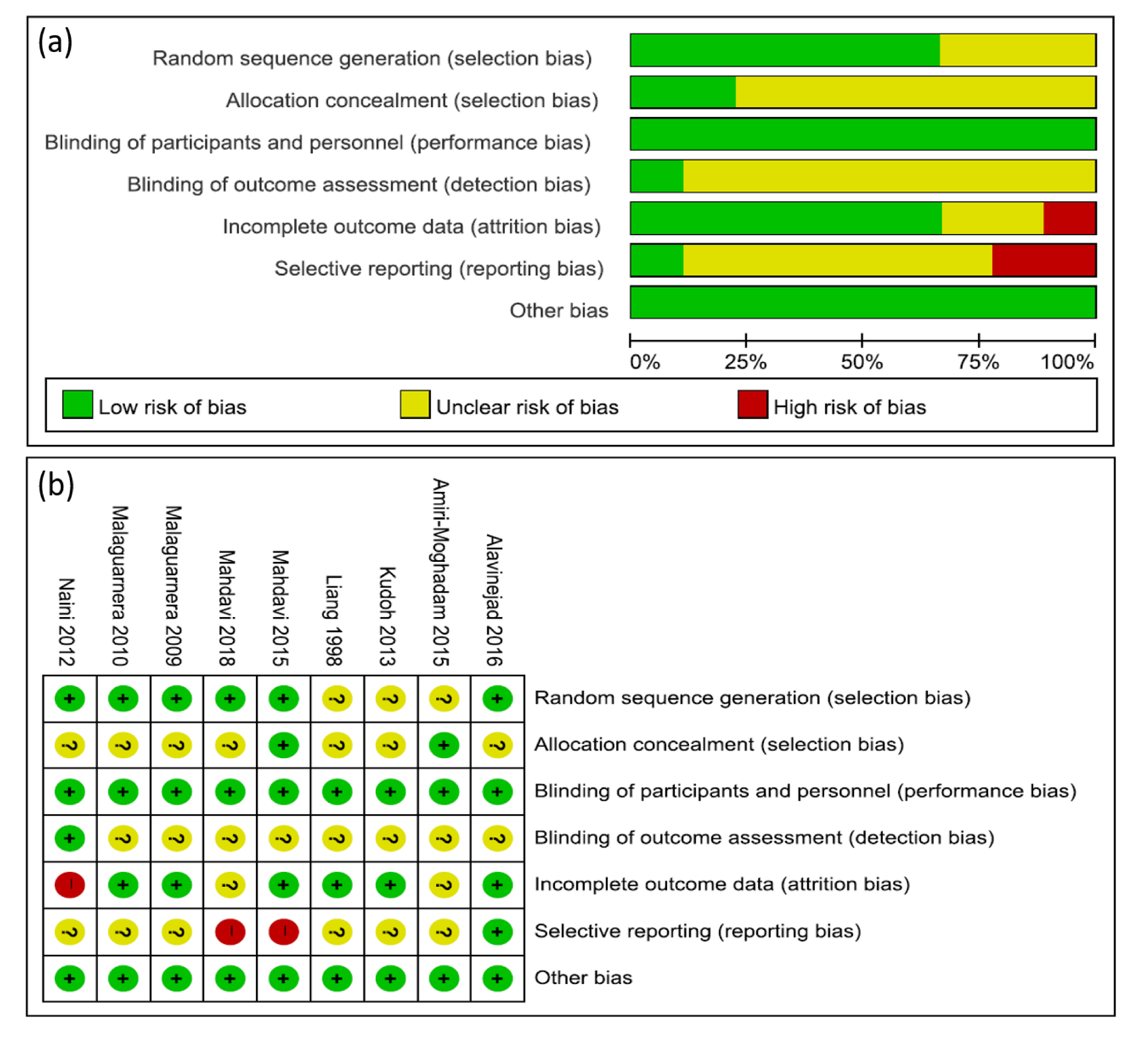
3.3. Risk of Bias in the Included RCT Studies
3.4. Effects of L-Carnitine Supplementation on Biomarkers of MetSyn
3.5. Subgroup Analysis and Heterogeneity
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Reaven, G.M. Role of Insulin Resistance in Human Disease. Diabetes 1988, 37, 1595–1607. [Google Scholar] [CrossRef]
- Grundy, S.M.; Brewer, H.B., Jr.; Cleeman, J.I.; Smith, S.C., Jr.; Lenfant, C. Definition of metabolic syndrome: Report of the National Heart, Lung, and Blood Institute/American Heart Association conference on scientific issues related to definition. Arterioscler. Thromb. Vasc. Biol. 2004, 109, 433–438. [Google Scholar] [CrossRef]
- Harmeyer, J. The physiological role of L-carnitine. Lohman Inf. 2002, 27, 15–21. [Google Scholar]
- Mingrone, G. Carnitine in type 2 diabetes. Ann. N. Y. Acad. Sci. 2004, 1033, 99–107. [Google Scholar] [CrossRef]
- Pooyandjoo, M.; Nouhi, M.; Shab-Bidar, S.; Djafarian, K.; Olyaeemanesh, A. The effect of (L-)carnitine on weight loss in adults: A systematic review and meta-analysis of randomized controlled trials. Obes. Rev. Off. J. Int. Assoc. Study Obes. 2016, 17, 970–976. [Google Scholar] [CrossRef]
- Talenezhad, N.; Mohammadi, M.; Ramezani-Jolfaie, N.; Mozaffari-Khosravi, H.; Salehi-Abargouei, A. Effects of l-carnitine supplementation on weight loss and body composition: A systematic review and meta-analysis of 37 randomized controlled clinical trials with dose-response analysis. Clin. Nutr. ESPEN 2020, 37, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Rajasekar, P.; Palanisamy, N.; Anuradha, C.V. Increase in nitric oxide and reductions in blood pressure, protein kinase C beta II and oxidative stress by L-carnitine: A study in the fructose-fed hypertensive rat. Clin. Exp. Hypertens. 2007, 29, 517–530. [Google Scholar] [CrossRef]
- Askarpour, M.; Hadi, A.; Dehghani Kari Bozorg, A.; Sadeghi, O.; Sheikhi, A.; Kazemi, M.; Ghaedi, E. Effects of L-carnitine supplementation on blood pressure: A systematic review and meta-analysis of randomized controlled trials. J. Hum. Hypertens. 2019, 33, 725–734. [Google Scholar] [CrossRef] [PubMed]
- Vidal-Casariego, A.; Burgos-Peláez, R.; Martínez-Faedo, C.; Calvo-Gracia, F.; Valero-Zanuy, M.; Luengo-Pérez, L.M.; Cuerda-Compés, C. Metabolic effects of L-carnitine on type 2 diabetes mellitus: Systematic review and meta-analysis. Exp. Clin. Endocrinol. 2013, 121, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Muoio, D.M.; Noland, R.C.; Kovalik, J.P.; Seiler, S.E.; Davies, M.N.; DeBalsi, K.L.; Ilkayeva, O.R.; Stevens, R.D.; Kheterpal, I.; Zhang, J.; et al. Muscle-specific deletion of carnitine acetyltransferase compromises glucose tolerance and metabolic flexibility. Cell Metab. 2012, 15, 764–777. [Google Scholar] [CrossRef]
- Malaguarnera, M.; Vacante, M.; Avitabile, T.; Malaguarnera, M.; Cammalleri, L.; Motta, M. L-Carnitine supplementation reduces oxidized LDL cholesterol in patients with diabetes. Am. J. Clin. Nutr. 2009, 89, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savović, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [PubMed]
- Otun, J.; Sahebkar, A.; Östlundh, L.; Atkin, S.L.; Sathyapalan, T. Systematic Review and Meta-analysis on the Effect of Soy on Thyroid Function. Sci. Rep. 2019, 9, 3964. [Google Scholar] [CrossRef]
- Alavinejad, P.; Zakerkish, M.; Eskandar, H.; Hashemi, s.J.; Chobineh, M.; Moghaddam, E. Evaluation of L-Carnitine Efficacy in the Treatment of Non- Alcoholic Fatty Liver Disease among Diabetic Patients: A Randomized Double Blind Pilot Study: Alavinejad P et al. L-Carnitine for management of NAFLD. J. Gastroenterol. Hepatol. Res. 2016, 5, 2191–2195. [Google Scholar] [CrossRef]
- Moghadam, S.A.; Nematy, M.; Eghtesadi, S.; Khalili, M.; Mojarrad, M.; Jazayeri, S.; Vosooghinia, H.; Khosravi, A.; Salehi, M.; Namdar, A. Effects of L-carnitine supplementation on body composition in patients with nonalcoholic steatohepatitis (NASH). Curr. Top. Nutraceut. Res. 2015, 13, 71–76. [Google Scholar] [CrossRef]
- Malek Mahdavi, A.; Mahdavi, R.; Kolahi, S.; Zemestani, M.; Vatankhah, A.M. L-Carnitine supplementation improved clinical status without changing oxidative stress and lipid profile in women with knee osteoarthritis. Nutr. Res. 2015, 35, 707–715. [Google Scholar] [CrossRef]
- Mahdavi, A.; Attari, R. Cardiometabolic responses to L-carnitine in obese women with knee osteoarthritis: A randomized, double-blind, placebo-controlled pilot study. Prog. Nutr. 2018, 20, 337–344. [Google Scholar]
- Emami Naini, A.; Moradi, M.; Mortazavi, M.; Amini Harandi, A.; Hadizadeh, M.; Shirani, F.; Basir Ghafoori, H.; Emami Naini, P. Effects of Oral L-Carnitine Supplementation on Lipid Profile, Anemia, and Quality of Life in Chronic Renal Disease Patients under Hemodialysis: A Randomized, Double-Blinded, Placebo-Controlled Trial. J. Nutr. Metab. 2012, 2012, 510483. [Google Scholar] [CrossRef]
- Malaguarnera, M.; Gargante, M.P.; Russo, C.; Antic, T.; Vacante, M.; Malaguarnera, M.; Avitabile, T.; Li Volti, G.; Galvano, F. L-carnitine supplementation to diet: A new tool in treatment of nonalcoholic steatohepatitis—A randomized and controlled clinical trial. Am. J. Gastroenterol. 2010, 105, 1338–1345. [Google Scholar] [CrossRef]
- Kudoh, Y.; Aoyama, S.; Torii, T.; Chen, Q.; Nagahara, D.; Sakata, H.; Nozawa, A. Hemodynamic stabilizing effects of L-carnitine in chronic hemodialysis patients. Cardiorenal Med. 2013, 3, 200–207. [Google Scholar] [CrossRef]
- Liang, Y.; Li, Y.; Shan, J.; Yu, B.; Ho, Z. The effects of oral L-carnitine treatment on blood lipid metabolism and the body fat content in the diabetic patient. Asia Pac. J. Clin. Nutr. 1998, 7, 192–195. [Google Scholar] [PubMed]
- Ruggenenti, P.; Cattaneo, D.; Loriga, G.; Ledda, F.; Motterlini, N.; Gherardi, G.; Orisio, S.; Remuzzi, G. Ameliorating hypertension and insulin resistance in subjects at increased cardiovascular risk: Effects of acetyl-L-carnitine therapy. Hypertension 2009, 54, 567–574. [Google Scholar] [CrossRef]
- González-Ortiz, M.; Hernández-González, S.O.; Hernández-Salazar, E.; Martínez-Abundis, E. Effect of oral L-carnitine administration on insulin sensitivity and lipid profile in type 2 diabetes mellitus patients. Ann. Nutr. Metab. 2008, 52, 335–338. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Liu, G.; Guo, G. Association of insulin resistance and autonomic tone in patients with pregnancy-induced hypertension. Clin. Exp. Hypertens. 2018, 40, 476–480. [Google Scholar] [CrossRef]
- Wu, X.; Han, T.; Gao, J.; Zhang, Y.; Zhao, S.; Sun, R.; Sun, C.; Niu, Y.; Li, Y. Association of Serum Calcium and Insulin Resistance With Hypertension Risk: A Prospective Population-Based Study. J. Am. Heart Assoc. 2019, 8, e009585. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Qu, H.; Yang, Z.; Rong, J.; Cai, W.; Zhou, H. Efficacy and Safety of L-Carnitine Treatment for Chronic Heart Failure: A Meta-Analysis of Randomized Controlled Trials. Biomed. Res. Int. 2017, 2017, 6274854. [Google Scholar] [CrossRef]
- Katz, A.M. Is the failing heart energy depleted? Cardiol. Clin. 1998, 16, 633–644. [Google Scholar] [CrossRef]
- Ringseis, R.; Keller, J.; Eder, K. Role of carnitine in the regulation of glucose homeostasis and insulin sensitivity: Evidence from in vivo and in vitro studies with carnitine supplementation and carnitine deficiency. Eur. J. Nutr. 2012, 51, 1–18. [Google Scholar] [CrossRef]
- Yang, S.K.; Xiao, L.; Song, P.A.; Xu, X.; Liu, F.Y.; Sun, L. Effect of L-carnitine therapy on patients in maintenance hemodialysis: A systematic review and meta-analysis. J. Nephrol. 2014, 27, 317–329. [Google Scholar] [CrossRef]
- Hurot, J.M.; Cucherat, M.; Haugh, M.; Fouque, D. Effects of L-carnitine supplementation in maintenance hemodialysis patients: A systematic review. J. Am. Soc. Nephrol. JASN 2002, 13, 708–714. [Google Scholar]
- Serban, M.C.; Sahebkar, A.; Mikhailidis, D.P.; Toth, P.P.; Jones, S.R.; Muntner, P.; Blaha, M.J.; Andrica, F.; Martin, S.S.; Borza, C.; et al. Impact of L-carnitine on plasma lipoprotein(a) concentrations: A systematic review and meta-analysis of randomized controlled trials. Sci. Rep. 2016, 6, 19188. [Google Scholar] [CrossRef] [PubMed]
- Bartel, L.L.; Hussey, J.L.; Shrago, E. Perturbation of serum carnitine levels in human adults by chronic renal disease and dialysis therapy. Am. J. Clin. Nutr. 1981, 34, 1314–1320. [Google Scholar] [CrossRef] [PubMed]
- Pekala, J.; Patkowska-Sokoła, B.; Bodkowski, R.; Jamroz, D.; Nowakowski, P.; Lochyński, S.; Librowski, T. L-carnitine-metabolic functions and meaning in humans life. Curr. Drug Metab. 2011, 12, 667–678. [Google Scholar] [CrossRef] [PubMed]
- Koeth, R.A.; Wang, Z.; Levison, B.S.; Buffa, J.A.; Org, E.; Sheehy, B.T.; Britt, E.B.; Fu, X.; Wu, Y.; Li, L.; et al. Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat. Med. 2013, 19, 576–585. [Google Scholar] [CrossRef]
- National Research Council Subcommittee on the Tenth Edition of the Recommended Dietary Allowances. The National Academies Collection: Reports funded by National Institutes of Health. In Recommended Dietary Allowances, 10th ed.; National Academies Press: Washington, DC, USA, 1989. [Google Scholar]
- Hathcock, J.N.; Shao, A. Risk assessment for carnitine. Regul. Toxicol. Pharmacol. RTP 2006, 46, 23–28. [Google Scholar] [CrossRef]
- Bruzell, E.; Steffensen, I.-L.; Granum, B.; Hetland, R.; Husøy, T.; Rohloff, J.; Wicklund, T. Risk Assessment of “Other Substances”-L-Carnitine and L-Carnitine-L-Tartrate Opinion of the Panel on Food Additives, Flavourings, Processing Aids, Materials in Contact with Food and Cosmetics of the Norwegian Scientific Committee for Food Safety; VKM: Oslo, Norway, 2015. [Google Scholar]

| Study | Population | Intervention | Outcomes of MetSyn | |||||
|---|---|---|---|---|---|---|---|---|
| Author (Country) | Year | Case | Mean Age (Int/Cont) | Sex (M/F) | Sample Size (Int/Cont) | Duration (Weeks) | Oral Dose (g/day) | |
| Alavinejad et al. [14] (Iran) | 2016 | T2DM and NASH | 60.0/59.0 | 38/16 | 28/26 | 12 | 0.75 | FBS, TG |
| Moghadam et al. [15] (Iran) | 2015 | NASH | 41.6/45.3 | 23/44 | 36/31 | 12 (+CR, EX) | 2 | WC |
| Kudoh et al. [20] (Japan) | 2013 | Under hemo-dialysis | 65.9/67.8 | 8/10 | 10/8 | 12 | 0.9 | SBP, DBP |
| Liang et al. [21] (China) | 1998 | T2DM | 59.4/57.9 | 16/30 | 23/23 | 12 | 3 | FBS, TG, HDLc |
| Mahdavi et al. [16] (Iran) | 2015 | Overweight or obese women with knee OA | 51.63/52.44 | 0/69 | 33/36 | 8 | 0.75 | TG, HDLc |
| Mahdavi et al. [17] (Iran) | 2018 | Obese women with knee OA | 51.56/52.60 | 0/48 | 23/25 | 8 | 0.75 | WC, FBS, SBP, DBP |
| Malaguarnera et al. [11] (Italy) | 2009 | T2DM | 49.0/48.0 | 58/23 | 41/40 | 12 (+CR) | 2 | FBS, TG, HDLc |
| Malaguarnera et al. [19] (Italy) | 2010 | NASH | 47.9/47.8 | 40/34 | 36/38 | 24 (+CR, EX) | 2 | FBS, TG, HDLc |
| Naini et al. [18] (Iran) | 2012 | Under hemo-dialysis | 53.9/51.8 | 26/25 | 24/27 | 16 | 1 | TG, HDLc |
| A Quantitative Synthesis of Data | Heterogeneity of Data | |||||
|---|---|---|---|---|---|---|
| Outcome | No. of RCTs | MD | 95% CI | p-Value | I2 | p-Value |
| FBS | ||||||
| Baseline FBS ≥ 100 mg/dL [11,14,19,21] | 4 | −10.74 | −15.90, −5.58 | <0.0001 | 0% | 0.46 |
| Baseline FBS < 100 mg/dL [17] | 1 | 0.99 | −6.43, 8.41 | 0.79 | - | - |
| TG | ||||||
| Baseline TG ≥ 150 mg/dL [11,14,19,21] | 4 | −21.48 | −50.56, 7.59 | 0.15 | 60% | 0.06 |
| Baseline TG < 150 mg/dL [16,18] | 2 | −20.39 | −54.88, 14.09 | 0.25 | 48% | 0.17 |
| HDL-C | ||||||
| Baseline HDLc < 40 mg/dL [18,19] | 2 | 3.5 | 1.50, 5.49 | 0.0006 | 0% | 0.49 |
| Baseline HDLc ≥ 40 mg/dL [11,16,21] | 3 | 0.94 | −0.12, 2.01 | 0.08 | 30% | 0.24 |
| A Quantitative Synthesis of Data | Heterogeneity of Data | |||||
|---|---|---|---|---|---|---|
| Outcome | No. of RCTs | MD | 95% CI | p-Value | I2 | p-Value |
| FBS | ||||||
| Iran [14,17] | 2 | 0.89 | −6.34, 8.12 | 0.81 | 0 | 0.91 |
| Italy [11,19] | 2 | −9.96 | −18.05, −1.87 | 0.02 | 54 | 0.14 |
| China [21] | 1 | −18.00 | −46.02, 10.02 | 0.21 | - | - |
| TG | ||||||
| Iran [14,16,18] | 3 | −17.76 | −40.69, 5.18 | 0.13 | 10 | 0.33 |
| Italy [11,19] | 2 | −13.50 | −40.63, 13.64 | 0.33 | 57 | 0.13 |
| China [21] | 1 | −115.14 | −207.57, −22.71 | 0.01 | - | - |
| HDL-C | ||||||
| Iran [16,18] | 2 | 0.86 | −7.11, 8.84 | 0.83 | 82 | 0.02 |
| Italy [11,19] | 2 | 1.85 | 0.04, 3.66 | 0.05 | 54 | 0.14 |
| China [21] | 1 | −1.16 | −8.91, 6.59 | 0.77 | - | - |
| A Quantitative Synthesis of Data | Heterogeneity of Data | |||||
|---|---|---|---|---|---|---|
| Outcome | No. of RCTs | MD | 95% CI | p-Value | I2 | p-Value |
| FBS | ||||||
| ≥1 g/d L-carnitine [11,19,21] | 3 | −11.41 | −16.10, −6.72 | <0.0001 | 16% | 0.31 |
| <1 g/d L-carnitine [14,17] | 2 | 0.89 | −6.34, 8.12 | 0.81 | 0% | 0.91 |
| TG | ||||||
| ≥1 g/d L-carnitine [11,19,21] | 3 | −29.85 | −60.08, 0.38 | 0.05 | 69% | 0.02 |
| <1 g/d L-carnitine [14,16] | 2 | −3.8 | −32.50, 24.90 | 0.08 | 0% | 0.85 |
| HDL-C | ||||||
| ≥1 g/d L-carnitine [11,18,19,21] | 4 | 1.66 | 0.70, 2.61 | 0.0007 | 40% | 0.17 |
| <1 g/d L-carnitine [16] | 1 | −3.45 | −8.98, 2.08 | 1.22 | - | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, M.; Park, S.; Lee, M. L-Carnitine’s Effect on the Biomarkers of Metabolic Syndrome: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2020, 12, 2795. https://doi.org/10.3390/nu12092795
Choi M, Park S, Lee M. L-Carnitine’s Effect on the Biomarkers of Metabolic Syndrome: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients. 2020; 12(9):2795. https://doi.org/10.3390/nu12092795
Chicago/Turabian StyleChoi, Munji, Seongmin Park, and Myoungsook Lee. 2020. "L-Carnitine’s Effect on the Biomarkers of Metabolic Syndrome: A Systematic Review and Meta-Analysis of Randomized Controlled Trials" Nutrients 12, no. 9: 2795. https://doi.org/10.3390/nu12092795
APA StyleChoi, M., Park, S., & Lee, M. (2020). L-Carnitine’s Effect on the Biomarkers of Metabolic Syndrome: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients, 12(9), 2795. https://doi.org/10.3390/nu12092795






