Vitamin D Doses from Solar Ultraviolet and Dietary Intakes in Patients with Depression: Results of a Case-Control Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Participants
2.3. Sample Size and Sampling Techniques
2.4. Data Collection Procedure
- Estimate vitamin D3 (IU) per day = Vitamin D Dose (VDD) × (4900 IU) × skin type factor × fraction of body exposed × age factor.
- Standard Vitamin D Dose (SVD) = Standard Erythemal Dose (SED/day) × Action Spectrum Conversion Factor (ASCF).
- Vitamin D Dose (VDD) = Standard Vitamin D Dose (SVD) × Geometric Conversion Factors (GCF).
2.5. Ethical Considerations
2.6. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Disclosure
References
- Lim, G.Y.; Tam, W.W.; Lu, Y.; Ho, C.S.; Zhang, M.W.; Ho, R.C. Prevalence of Depression in the Community from 30 Countries between 1994 and 2014. Sci. Rep. 2018, 8, 2861. [Google Scholar] [CrossRef]
- Shadrina, M.; Bondarenko, E.A.; Slominsky, P.A. Genetics Factors in Major Depression Disease. Front. Psychiatry 2018, 9, 334. [Google Scholar] [CrossRef]
- Kern, D.M.; Cepeda, M.S.; Defalco, F.; Etropolski, M. Treatment patterns and sequences of pharmacotherapy for patients diagnosed with depression in the United States: 2014 through 2019. BMC Psychiatry 2020, 20, 1–10. [Google Scholar] [CrossRef]
- Kirsch, I. Placebo Effect in the Treatment of Depression and Anxiety. Front. Psychiatry 2019, 10, 407. [Google Scholar] [CrossRef]
- Bracken, P.; Thomas, P.; Timimi, S.; Asen, E.; Behr, G.; Beuster, C.; Bhunnoo, S.; Browne, I.; Chhina, N.; Double, D.; et al. Psychiatry beyond the current paradigm. Br. J. Psychiatry 2012, 201, 430–434. [Google Scholar] [CrossRef]
- Jelovac, A.; Kolshus, E.; McLoughlin, D.M. Relapse Following Successful Electroconvulsive Therapy for Major Depression: A Meta-Analysis. Neuropsychopharmacology 2013, 38, 2467–2474. [Google Scholar] [CrossRef]
- Van Bronswijk, S.; Moopen, N.; Beijers, L.; Ruhe, H.G.; Peeters, F. Effectiveness of psychotherapy for treatment-resistant depression: A meta-analysis and meta-regression. Psychol. Med. 2019, 49, 366–379. [Google Scholar] [CrossRef] [PubMed]
- Cuijpers, P.; Noma, H.; Karyotaki, E.; Vinkers, C.H.; Cipriani, A.; Furukawa, T.A. A network meta-analysis of the effects of psychotherapies, pharmacotherapies and their combination in the treatment of adult depression. World Psychiatry 2020, 19, 92–107. [Google Scholar] [CrossRef] [PubMed]
- Firth, J.; Marx, W.; Dash, S.; Carney, R.; Teasdale, S.B.; Solmi, M.; Stubbs, B.; Schuch, F.B.; Carvalho, A.F.; Jacka, F.; et al. The Effects of Dietary Improvement on Symptoms of Depression and Anxiety: A Meta-Analysis of Randomized Controlled Trials. Psychosom. Med. 2019, 81, 265–280. [Google Scholar] [CrossRef] [PubMed]
- Spedding, S. Vitamin D and depression: A systematic review and meta-analysis comparing studies with and without biological flaws. Nutrients 2014, 6, 1501–1518. [Google Scholar] [CrossRef] [PubMed]
- Kjærgaard, M.; Joakimsen, R.; Jorde, R. Low serum 25-hydroxyvitamin D levels are associated with depression in an adult Norwegian population. Psychiatry Res. 2011, 190, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Umhau, J.C.; George, D.T.; Heaney, R.P.; Lewis, M.D.; Ursano, R.J.; Heilig, M.; Hibbeln, J.R.; Schwandt, M.L. Low vitamin D status and suicide: A case-control study of active duty military service members. PLoS ONE 2013, 8, e51543. [Google Scholar] [CrossRef]
- Milaneschi, Y.; Hoogendijk, W.; Lips, P.; Heijboer, A.C.; Schoevers, R.; van Hemert, A.M.; Beekman, A.T.F.; Smit, J.H.; Penninx, B.W.J.H. The association between low vitamin D and depressive disorders. Mol. Psychiatry 2014, 19, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Lardner, A.L. Vitamin D and hippocampal development-the story so far. Front. Mol. Neurosci. 2015, 8, 58. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, F.N.; Costa, A.P.; Ghisleni, G.; Diaz, A.P.; Rodrigues, A.L.S.; Peluffo, H.; Kaster, M.P. NLRP3 inflammasome-driven pathways in depression: Clinical and preclinical findings. Brain Behav. Immun. 2017, 64, 367–383. [Google Scholar] [CrossRef]
- Muthuramalingam, A.; Menon, V.; Rajkumar, R.P.; Negi, V.S. Is Depression an Inflammatory Disease? Findings from a Cross-Sectional Study at a Tertiary Care Center. Indian J. Psychol. Med. 2016, 38, 114–119. [Google Scholar] [CrossRef]
- Zhu, D.-M.; Zhao, W.; Zhang, B.; Zhang, Y.; Yang, Y.; Zhang, C.; Wang, Y.; Zhu, J.; Yu, Y. The Relationship Between Serum Concentration of Vitamin D, Total Intracranial Volume, and Severity of Depressive Symptoms in Patients With Major Depressive Disorder. Front. Psychiatry 2019, 10, 322. [Google Scholar] [CrossRef]
- Dana-Alamdari, L.; Kheirouri, S.; Noorazar, S.G. Serum 25-Hydroxyvitamin D in Patients with Major Depressive Disorder. Iran. J. Public Health 2015, 44, 690–697. [Google Scholar]
- Cuomo, A.; Maina, G.; Bolognesi, S.; Rosso, G.; Beccarini Crescenzi, B.; Zanobini, F.; Goracci, A.; Facchi, E.; Favaretto, E.; Baldini, I.; et al. Prevalence and Correlates of Vitamin D Deficiency in a Sample of 290 Inpatients With Mental Illness. Front. Psychiatry 2019, 10, 167. [Google Scholar] [CrossRef]
- Anglin, R.E.S.; Samaan, Z.; Walter, S.D.; McDonald, S.D. Vitamin D deficiency and depression in adults: Systematic review and meta-analysis. Br. J. Psychiatry 2013, 202, 100–107. [Google Scholar] [CrossRef]
- Sarris, J.; Murphy, J.; Mischoulon, D.; Papakostas, G.I.; Fava, M.; Berk, M.; Ng, C.H. Adjunctive Nutraceuticals for Depression: A Systematic Review and Meta-Analyses. Am. J. Psychiatry 2016, 173, 575–587. [Google Scholar] [CrossRef] [PubMed]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P.; Initiative, S. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. PLoS Med. 2007, 4, e040296. [Google Scholar] [CrossRef] [PubMed]
- Al-Haddad, F.A.; Al-Mahroos, F.T.; Al-Sahlawi, H.S.; Al-Amer, E. The impact of dietary intake and sun exposure on vitamin D deficiency among couples. Bahrain Med. Bull. 2014, 158, 1–5. [Google Scholar] [CrossRef][Green Version]
- WHO. Body Mass Index—BMI. Available online: https://www.euro.who.int/en/health-topics/disease-prevention/nutrition/a-healthy-lifestyle/body-mass-index-bmi (accessed on 1 July 2020).
- Upton, J. Beck Depression Inventory (BDI). In Encyclopedia of Behavioral Medicine; Gellman, M.D., Turner, J.R., Eds.; Springer: New York, NY, USA, 2013; pp. 178–179. [Google Scholar] [CrossRef]
- Naja, S.; Al-Kubaisi, N.; Chehab, M.; Al-Dahshan, A.; Abuhashem, N.; Bougmiza, I. Psychometric properties of the Arabic version of EPDS and BDI-II as a screening tool for antenatal depression: Evidence from Qatar. BMJ Open 2019, 9, e030365. [Google Scholar] [CrossRef] [PubMed]
- Jahrami, H.; Saif, Z.; AlHaddad, M.; Faris, M.e.A.-I.; Hammad, L.; Ali, B. Assessing dietary and lifestyle risk behaviours and their associations with disease comorbidities among patients with depression: A case-control study from Bahrain. Heliyon 2020, 6, e04323. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B.; Fakhoury, H.M.A.; Karras, S.N.; Al Anouti, F.; Bhattoa, H.P. Variations in 25-Hydroxyvitamin D in Countries from the Middle East and Europe: The Roles of UVB Exposure and Diet. Nutrients 2019, 11, 2065. [Google Scholar] [CrossRef]
- WHO. Physical Activity and Adults. Available online: https://www.who.int/dietphysicalactivity/publications/recommendations18_64yearsold/en/ (accessed on 1 July 2020).
- Godar, D.E.; Pope, S.J.; Grant, W.B.; Holick, M.F. Solar UV Doses of Young Americans and Vitamin D3 Production. Environ. Health Perspect. 2012, 120, 139–143. [Google Scholar] [CrossRef]
- Fitzpatrick, T.B. The Validity and Practicality of Sun-Reactive Skin Types I Through VI. Arch. Dermatol. 1988, 124, 869–871. [Google Scholar] [CrossRef]
- Silva, A.A. Erythemal dose rate under noon overcast skies. Photochem. Photobiol. Sci. 2013, 12, 777–786. [Google Scholar] [CrossRef]
- Godar, D.E.; Pope, S.J.; Grant, W.B.; Holick, M.F. Solar UV Doses of Adult Americans and Vitamin D3 Production. Derm. Endocrinol. 2011, 3, 243–250. [Google Scholar] [CrossRef]
- Pope, S.J.; Holick, M.F.; Mackin, S.; Godar, D.E. Action spectrum conversion factors that change erythemally weighted to previtamin D3-weighted UV doses. Photochem. Photobiol. 2008, 84, 1277–1283. [Google Scholar] [CrossRef] [PubMed]
- USDA. Dietary Supplement Ingredient Database. Available online: https://dietarysupplementdatabase.usda.nih.gov/ingredient_calculator/help.php (accessed on 1 July 2020).
- STATA, M.P. 16.1; StataCorp: College Station, TX, USA, 2020.
- Farid, E.; Jaradat, A.A.; Al-Segai, O.; Hassan, A.B. Prevalence of Vitamin D Deficiency in Adult Patients with Systemic Lupus Erythematosus in Kingdom of Bahrain. Egypt. J. Immunol. 2017, 24, 1–8. [Google Scholar] [PubMed]
- Voshaar, R.C.O.; Derks, W.J.; Comijs, H.C.; Schoevers, R.A.; de Borst, M.H.; Marijnissen, R.M. Antidepressants differentially related to 1,25-(OH)2 vitamin D3 and 25-(OH) vitamin D3 in late-life depression. Transl. Psychiatry 2014, 4, e383. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B.; Soles, C.M. Epidemiologic evidence supporting the role of maternal vitamin D deficiency as a risk factor for the development of infantile autism. Dermatoendocrinology 2009, 1, 223–228. [Google Scholar] [CrossRef]
- Itzhaky, D.; Amital, D.; Gorden, K.; Bogomolni, A.; Arnson, Y.; Amital, H. Low serum vitamin D concentrations in patients with schizophrenia. Isr. Med. Assoc. J. 2012, 14, 88–92. [Google Scholar]
- Annweiler, C.; Schott, A.M.; Rolland, Y.; Blain, H.; Herrmann, F.R.; Beauchet, O. Dietary intake of vitamin D and cognition in older women: A large population-based study. Neurology 2010, 75, 1810–1816. [Google Scholar] [CrossRef]
- Brouwer-Brolsma, E.M.; Feskens, E.J.; Steegenga, W.T.; de Groot, L.C. Associations of 25-hydroxyvitamin D with fasting glucose, fasting insulin, dementia and depression in European elderly: The SENECA study. Eur. J. Nutr. 2013, 52, 917–925. [Google Scholar] [CrossRef]
- Jaddou, H.Y.; Batieha, A.M.; Khader, Y.S.; Kanaan, S.H.; El-Khateeb, M.S.; Ajlouni, K.M. Depression is associated with low levels of 25-hydroxyvitamin D among Jordanian adults: Results from a national population survey. Eur. Arch. Psychiatry Clin. Neurosci. 2012, 262, 321–327. [Google Scholar] [CrossRef]
- Föcker, M.; Antel, J.; Ring, S.; Hahn, D.; Kanal, Ö.; Öztürk, D.; Hebebrand, J.; Libuda, L. Vitamin D and mental health in children and adolescents. Eur. Child Adolesc. Psychiatry 2017, 26, 1043–1066. [Google Scholar] [CrossRef]
- Schuch, F.; Vancampfort, D.; Firth, J.; Rosenbaum, S.; Ward, P.; Reichert, T.; Bagatini, N.C.; Bgeginski, R.; Stubbs, B. Physical activity and sedentary behavior in people with major depressive disorder: A systematic review and meta-analysis. J. Affect. Disord. 2017, 210, 139–150. [Google Scholar] [CrossRef]
- Vancampfort, D.; Firth, J.; Schuch, F.B.; Rosenbaum, S.; Mugisha, J.; Hallgren, M.; Probst, M.; Ward, P.B.; Gaughran, F.; De Hert, M.; et al. Sedentary behavior and physical activity levels in people with schizophrenia, bipolar disorder and major depressive disorder: A global systematic review and meta-analysis. World Psychiatry Off. J. World Psychiatric Assoc. WPA 2017, 16, 308–315. [Google Scholar] [CrossRef] [PubMed]
- McCann, J.C.; Ames, B.N. Is there convincing biological or behavioral evidence linking vitamin D deficiency to brain dysfunction? FASEB J. 2008, 22, 982–1001. [Google Scholar] [CrossRef] [PubMed]
- Irandoust, K.; Taheri, M. The Effect of Vitamin D supplement and Indoor Vs Outdoor Physical Activity on Depression of Obese Depressed Women. Asian J. Sports Med. 2017, 8, 1. [Google Scholar] [CrossRef]
| Variable * | Cases, n = 96 | Controls, n = 96 | p-Value ** |
|---|---|---|---|
| Sex | |||
| Male | 37 (39%) | 37 (39%) | 1.0 |
| Female | 59 (61%) | 59 (61%) | |
| Job-status | |||
| Employed | 27 (28%) | 69 (72%) | 0.001 |
| Unemployed | 69 (72%) | 27 (28%) | |
| Marital status | |||
| Single | 48 (50%) | 23 (24%) | 0.001 |
| Married | 48 (50%) | 73 (76%) | |
| BMI classification | |||
| Underweight | 4 (4%) | 2 (2%) | 0.25 |
| Normal | 26 (27%) | 34(35%) | |
| Overweight | 30 (31%) | 35 (37%) | |
| Obese | 36 (38%) | 25 (26%) | |
| Current tobacco smoker | 37% | 10% | 0.001 |
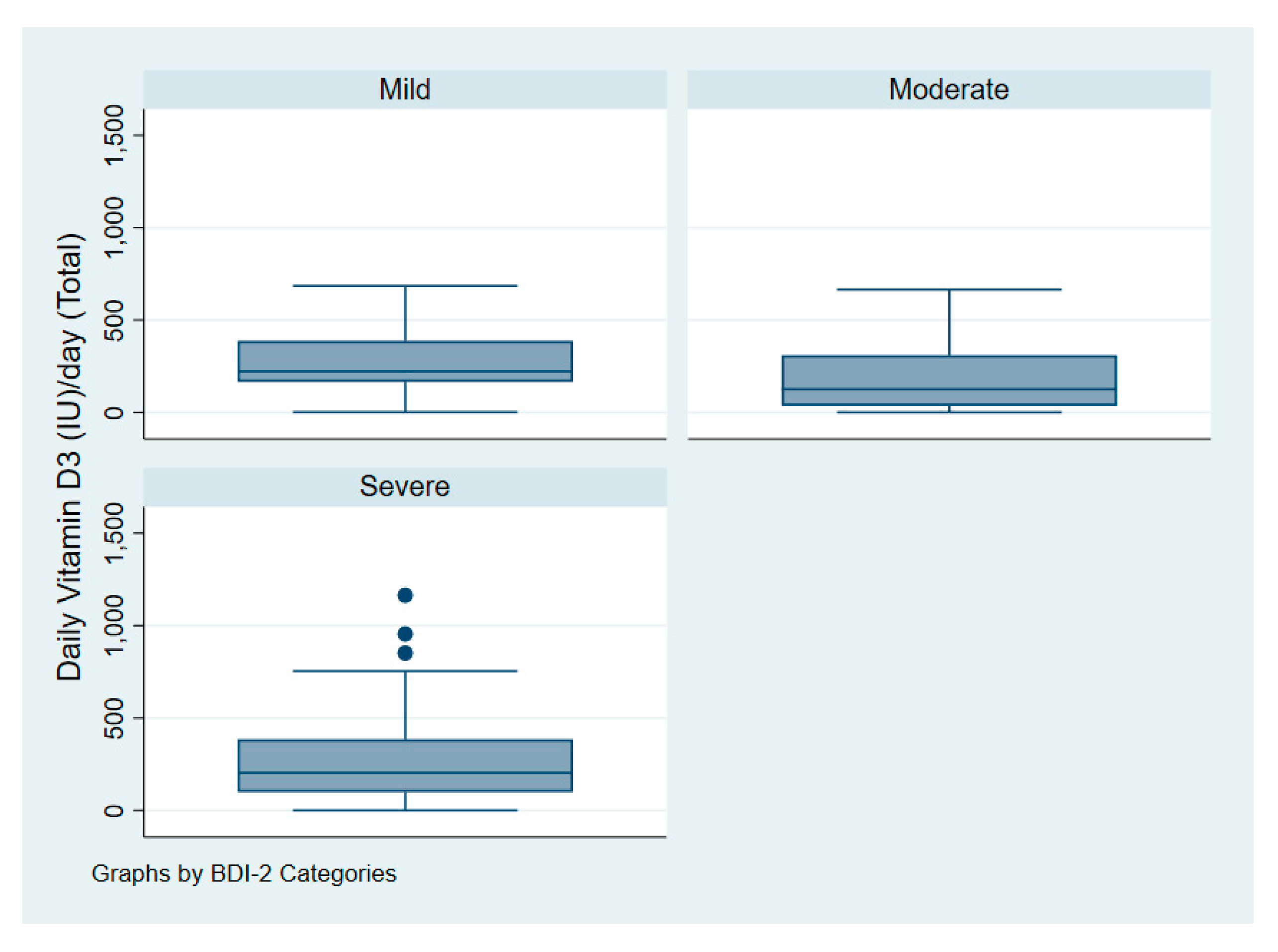
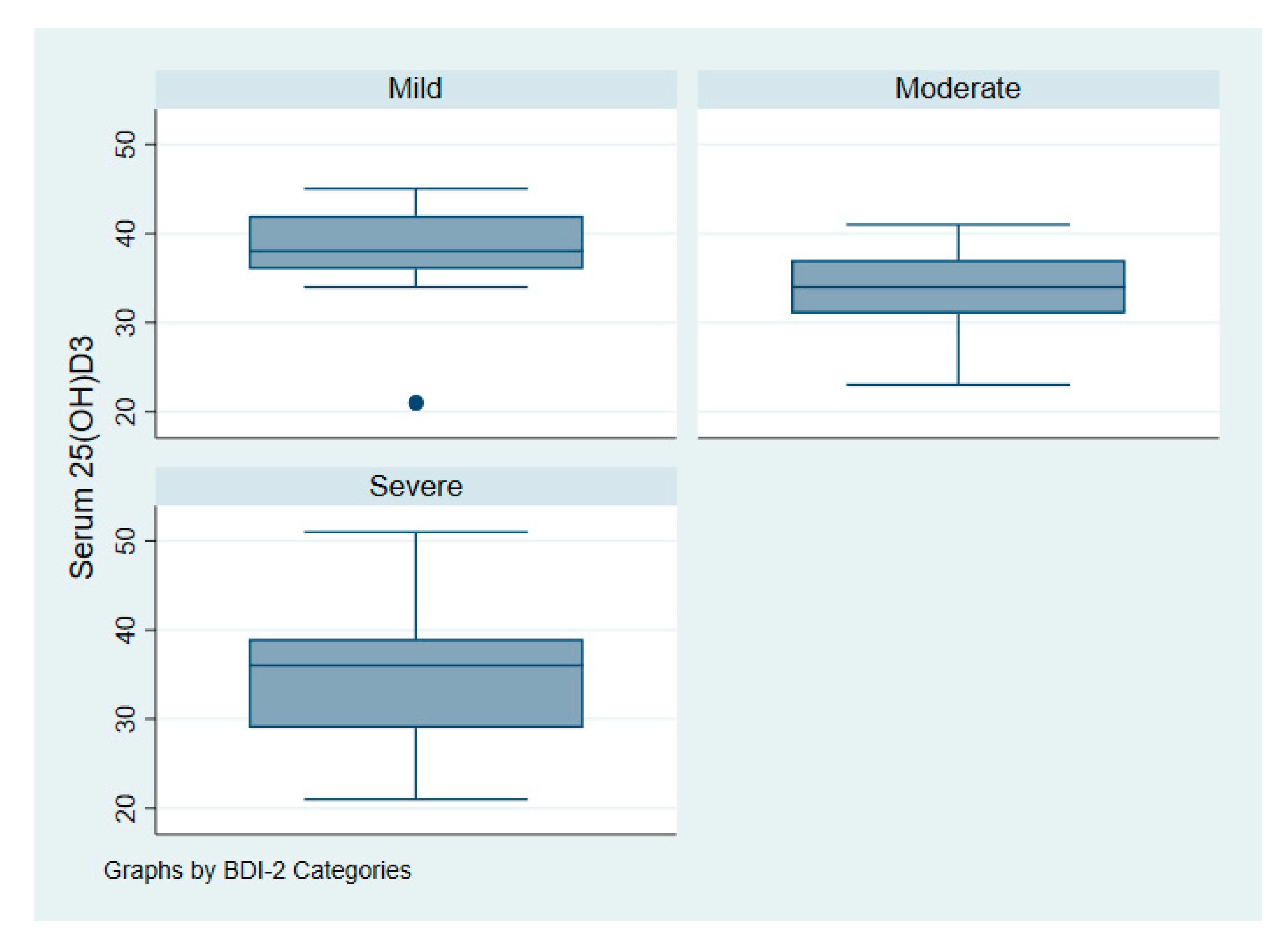
| Beck Depression Inventory-II (BDI-II) | |||
| Mild | 13 (13%) | Not applicable | - |
| Moderate | 40 (42%) | ||
| Severe | 43 (45%) | ||
| Age (year) | 44 ± 13 | 43 ± 15 | 0.4 |
| Weight (kg) | 76 ± 19 | 75 ± 17 | 0.63 |
| Height (cm) | 163 ± 10 | 165 ± 10 | 0.13 |
| BMI (kg/m2) | 29 ± 7 | 28 ± 6 | 0.11 |
| Body fat percentage (%) | 35 ± 12 | 33 ± 10 | 0.09 |
| Total body water percentage (%) | 36 ± 6 | 36 ± 7 | 0.91 |
| Body surface area (m2) | 2 ± 0.2 | 2 ± 0.2 | 0.98 |
| Lean mass (kg) | 49 ± 8 | 49 ± 8 | 0.68 |
| Fat mass (kg) | 28 ± 13 | 26 ± 10 | 0.31 |
| Serum 25(OH)D (nmol/L) *** a,b | 35 ± 7 | 38 ± 6 | 0.01 |
| * Variable | Cases, n = 96 | Controls, n = 96 | p-Value ** | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | SE | 95%CI | Mean | SD | SE | 95%CI | ||
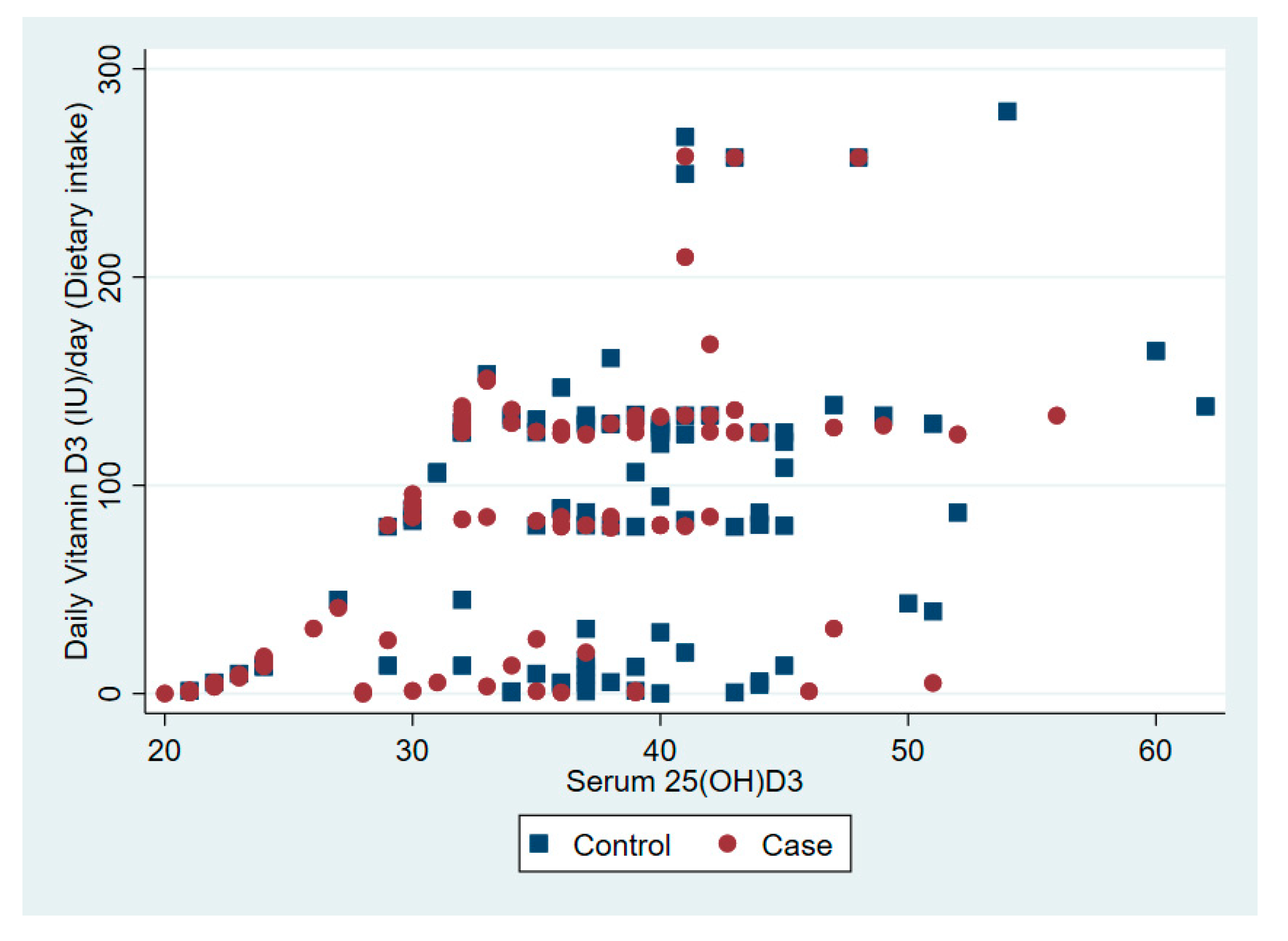
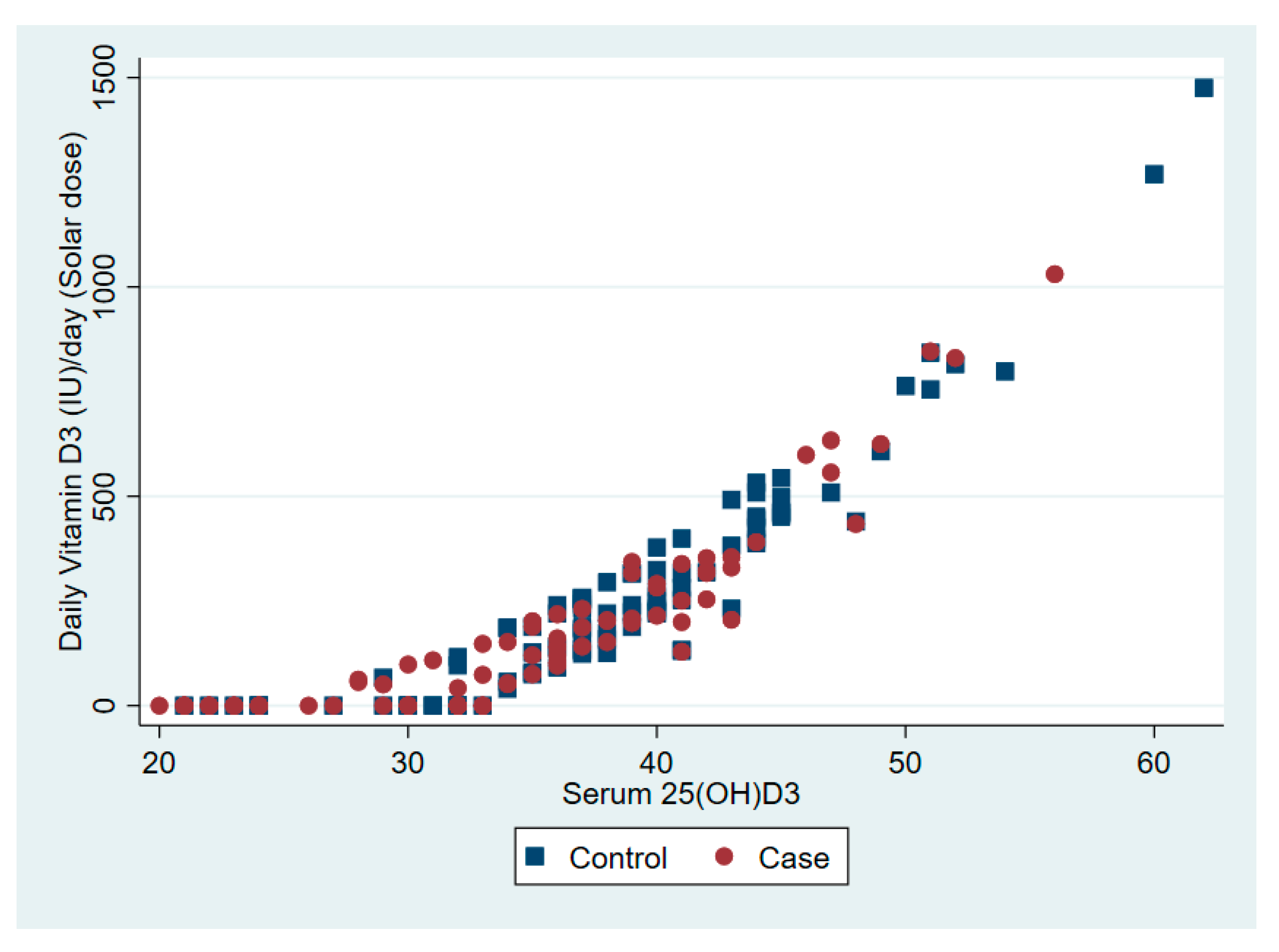
| Vitamin D intake from diet per day (IU) | 81 | 65 | 7 | 68–94 | 87 | 66 | 7 | 74–101 | 0.50 |
| Vitamin D synthesis from sunlight per day (IU) | 153 | 206 | 21 | 111–195 | 270 | 260 | 27 | 218–323 | 0.001 |
| Vitamin D per day (IU) | 234 | 275 | 23 | 189–280 | 357 | 275 | 28 | 301–413 | 0.001 |
| Share of Vitamin D from diet per day | 35% | 25% | 0.11 | ||||||
| Share of Vitamin D from sunlight exposure per day | 65% | 75% | 0.11 | ||||||
| Compliance with the recommended minimum daily intake (400 IU per day) | 18 (19%) | 29 (30%) | 0.048 | ||||||
| Vitamin D according to 25(OH)D Optimal—≥50 nmol/L Insufficient—≥30 ˂50 nmol/L Deficient—<30 nmol/L | 1 (2%) 34 (79%) 8 (19) | 3 (6%) 44 (88%) 3 (6%) | 0.13 | ||||||
| Serum 25(OH)D ≥ 30 nmol/L | 35 (83%) | 47 (94%) | 0.06 | ||||||
| Serum 25(OH)D ≥ 35 nmol/L | 24 (56%) | 38 (76%) | 0.04 | ||||||
| Serum 25(OH)D ≥ 40 nmol/L | 9 (21%) | 23 (46%) | 0.01 | ||||||
| Serum 25(OH)D ≥ 45 nmol/L | 3 (7%) | 4 (8%) | 0.90 | ||||||
| Serum 25(OH)D ≥ 50 nmol/L | 1 (2%) | 3 (6) | 0.40 | ||||||
| Cases with depression (n = 96) | ||
| Outcome variable: Daily vitamin D Dose | ||
| Explanatory Variables | β | p-Value |
| Education level | 60 | 0.12 |
| Smoking | 1 | 1 |
| Physical activity | 318 | 0.001 * |
| Controls (n = 96) | ||
| Outcome variable: Daily vitamin D Dose | ||
| Explanatory Variables | β | p-Value |
| Education level | 111 | 0.08 |
| Smoking | −52 | 0.60 |
| Physical activity | 267 | 0.001 * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jahrami, H.; Bragazzi, N.L.; Grant, W.B.; AlFarra, H.S.M.; AlFara, W.S.M.; Mashalla, S.; Saif, Z. Vitamin D Doses from Solar Ultraviolet and Dietary Intakes in Patients with Depression: Results of a Case-Control Study. Nutrients 2020, 12, 2587. https://doi.org/10.3390/nu12092587
Jahrami H, Bragazzi NL, Grant WB, AlFarra HSM, AlFara WSM, Mashalla S, Saif Z. Vitamin D Doses from Solar Ultraviolet and Dietary Intakes in Patients with Depression: Results of a Case-Control Study. Nutrients. 2020; 12(9):2587. https://doi.org/10.3390/nu12092587
Chicago/Turabian StyleJahrami, Haitham, Nicola Luigi Bragazzi, William Burgess Grant, Hala Shafeeq Mohamed AlFarra, Wafa Shafeeq Mohamed AlFara, Shahla Mashalla, and Zahra Saif. 2020. "Vitamin D Doses from Solar Ultraviolet and Dietary Intakes in Patients with Depression: Results of a Case-Control Study" Nutrients 12, no. 9: 2587. https://doi.org/10.3390/nu12092587
APA StyleJahrami, H., Bragazzi, N. L., Grant, W. B., AlFarra, H. S. M., AlFara, W. S. M., Mashalla, S., & Saif, Z. (2020). Vitamin D Doses from Solar Ultraviolet and Dietary Intakes in Patients with Depression: Results of a Case-Control Study. Nutrients, 12(9), 2587. https://doi.org/10.3390/nu12092587








