Antiviral Properties of Flavonoids and Delivery Strategies
Abstract
1. Introduction
2. Antiviral Effects of Flavonoids
2.1. Flavonoids Targeting HCV, HBV and HAV Viruses
2.2. Antiviral Effects of Flavonoids Against Influenza Viruses
2.3. Antiviral Properties of Flavonoids Against Dengue and Zika Viruses
2.4. HSV, Respiratory Syncytial Virus (RSV), RRV: Antiviral Activities of Flavonoids
3. Insights into Flavonoid Molecular Targets and Antiviral Synergistic Effects
3.1. Influenza Virus Molecular Targets
3.2. Molecular Targets of HCV and HBV Viruses
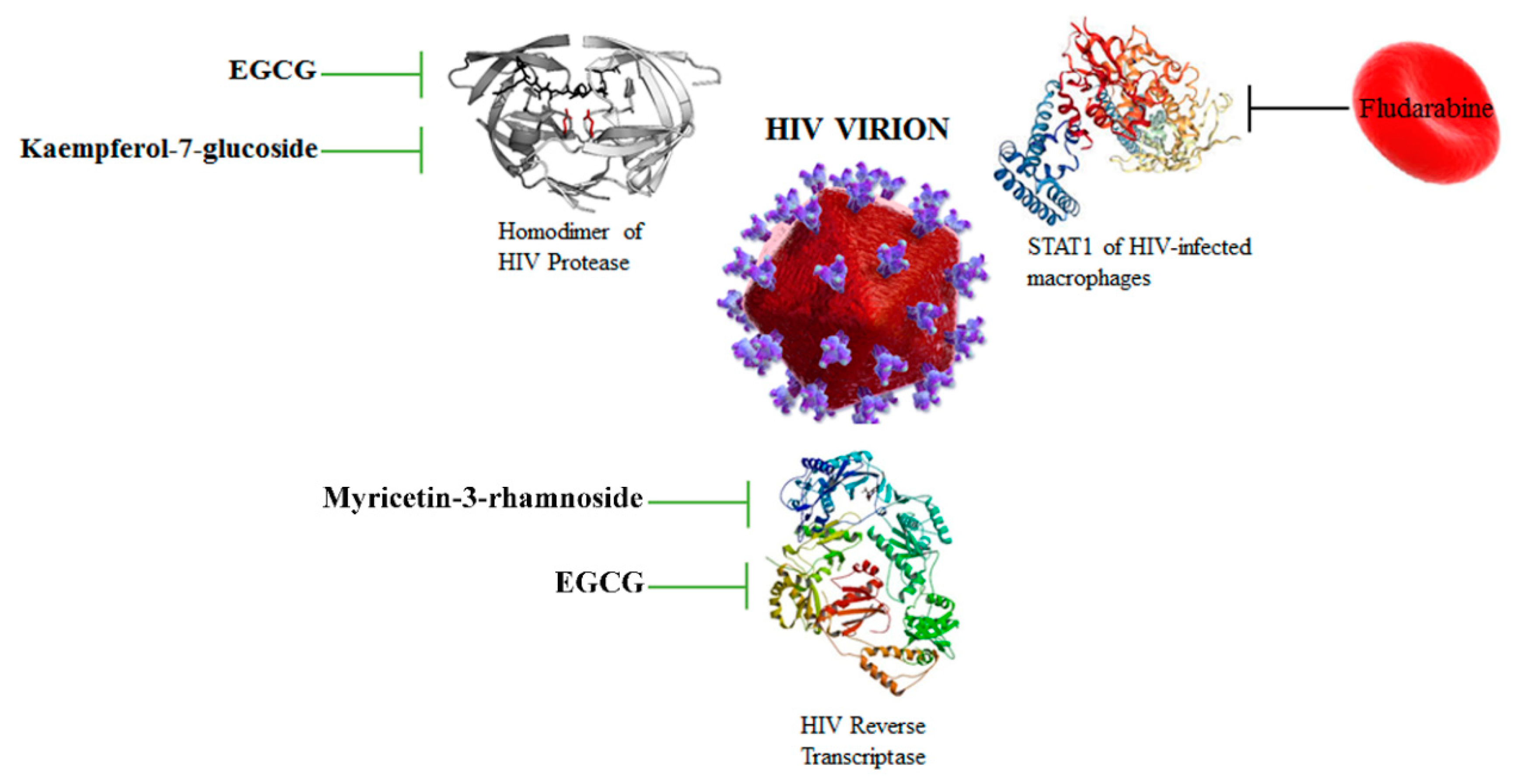
3.3. HIV Protease and Reverse Transcriptase Are Targeted by Flavonoids
3.4. HSV-1 and DENV-2: Decreasing ROS to Counteract Viral Infections
4. Delivery Strategies for Antiviral Drugs and Phytochemicals
5. Therapeutic Potential of Antiviral Dietary Flavonoids
6. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AhR | Aryl Hydrocarbon Receptor |
| DENV-2 | Dengue Virus-2 |
| EC50 | 50% Effective Concentration |
| EFSA | European Food Safety Authority |
| EGCG | Epigallocatechin-3-gallate |
| HAV | Human Hepatitis A Virus |
| HBV | Human Hepatitis B Virus |
| HCV | Human Hepatitis C Virus |
| HDV | Human Hepatitis D virus |
| HIV | Human Immunodeficiency Virus |
| HSP70 | Heat Shock Protein 70 |
| HSV-1 | Herpes Simplex Virus type-1 |
| IC50 | 50% Inhibitory Concentration |
| IFN-β | Interferon-β |
| IL-1 | Interleukin-1 |
| IL-6 | Interleukin-6 |
| IRES | Internal Ribosome Entry Site |
| IRF3 | Interferon regulatory factor 3 |
| IRF7 | Interferon regulatory factor 7 |
| MDCK | Madin-Darby Canine Kidney |
| NADPH | Nicotinamide Adenine Dinucleotide Phosphate |
| NF-kB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| NO | Nitric Oxide |
| NOX4 | NADPH Oxidase 4 |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| NS1 | Non Structural protein 1 |
| NS2 | Non Structural protein 2 |
| NS3 | Non Structural protein 3 |
| NS5A | Non Structural protein 5A |
| NS5B | Non Structural protein 5B |
| NTCP | sodium taurocholate cotransporting polypeptide |
| P protein | RSV phosphoprotein |
| PA | Polymerase Acidic subunit |
| PB1 | Protein Binding 1 subunit |
| PB2 | Protein Binding 2 subunit |
| PLA | Poly (d,l-Lactide) |
| Q-CNLC | Quercetin-loaded Cationic Nanostructure Lipid Carrier |
| RBCs | Red Blood Cells |
| RdDp | RNA-dependent DNA polymerase |
| RdRp | RNA-dependent RNA polymerase |
| ROS | Radical Oxygen Species |
| RRV | Rhesus rotavirus |
| RSV | Respiratory Syncytial Virus |
| SMDDS | Self-Micro-emulsifying Drug Delivery System |
| SNEDDS | Self-Nanoemulsifying Drug Delivery System |
| STAT1 | Signal transducer and activator of transcription 1 |
| SU.VI.MAX | Supplementation en Vitamines et Mineraux Antioxydants |
| TLR3 | Toll-Like Receptor 3 |
| TLR4 | Toll-Like Receptor 4 |
| TLR7 | Toll-Like Receptor 7 |
| TNF-α | Tumor Necrosis Factor-α |
References
- Ninfali, P.; Bacchiocca, M. Polyphenols and antioxidant capacity of vegetables under fresh and frozen conditions. J. Agric. Food Chem. 2003, 51, 2222–2226. [Google Scholar] [CrossRef] [PubMed]
- Ninfali, P.; Aluigi, G.; Bacchiocca, M.; Magnani, M. Antioxidant capacity of Extra-Virgin Olive Oils. JOACS 2001, 78, 243–247. [Google Scholar] [CrossRef]
- Ninfali, P.; Mea, G.; Giorgini, S.; Rocchi, M.; Bacchiocca, M. Antioxidant capacity of vegetables, spices and dressings relevant to nutrition. Br. J. Nutr. 2005, 93, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.R.; Kang, G.H.; Cho, S.G. Effect of flavonoids on human health: Old subjects but new challenges. Recent Pat. Biotechnol. 2007, 1, 139–150. [Google Scholar] [CrossRef]
- Papi, A.; Farabegoli, F.; Iori, R.; Orlandi, M.; De Nicola, G.R.; Bagatta, M.; Angelino, D.; Gennari, L.; Ninfali, P. Vitexin 2-O-xyloside, raphasatin and (-) eepigallocatechin-3-gallate synergistically affect cell growth and apoptosis of colon cancer cells. Food Chem. 2013, 138, 1521–1530. [Google Scholar] [CrossRef]
- Farabegoli, F.; Scarpa, E.S.; Frai, A.; Serafini, G.; Papi, A.; Spisni, E.; Antonini, E.; Benedetti, S.; Ninfali, P. Betalains increase vitexin-2-O-xyloside cytotoxicity in Caco-2 cancer cells. Food Chem. 2017, 218, 356–364. [Google Scholar] [CrossRef]
- Scarpa, E.S.; Antonini, E.; Palma, F.; Mari, M.; Ninfali, P. Antiproliferative activity of vitexin-2-O-xyloside and avenanthramides on CaCo-2 and HepG2 cancer cells occurs through apoptosis induction and reduction of pro-survival mechanisms. Eur. J. Nutr. 2018, 57, 1381–1395. [Google Scholar] [CrossRef]
- Scarpa, E.S.; Mari, M.; Antonini, E.; Palma, F.; Ninfali, P. Natural and synthetic avenathramides activate caspases 2,8,3 and downregulate hTERT, MDR1 and COX-2 genes in CaCo-2 and Hep3B cancer cells. Food Funct. 2018, 9, 2913–2921. [Google Scholar] [CrossRef]
- Antonini, E.; Iori, R.; Ninfali, P.; Scarpa, E.S. A Combination of Moringin and Avenanthramide 2f Inhibits the Proliferation of Hep3B Liver Cancer Cells Inducing Intrinsic and Extrinsic Apoptosis. Nutr. Cancer 2018, 70, 1159–1165. [Google Scholar] [CrossRef]
- Ninfali, P.; Antonini, E.; Frati, A.; Scarpa, E.-S. C-Glycosyl Flavonoids from Beta vulgaris Cicla and Betalains from Beta vulgais rubra: Antioxidant, Anticancer, Antiinflammatory Activities—A Review. Phytother. Res. 2017, 31, 871–884. [Google Scholar] [CrossRef]
- Ninfali, P.; Bacchiocca, M.; Antonelli, A.; Biagiotti, E.; Di Gioacchino, A.M.; Piccoli, G.; Stocchi, V.; Brandi, G. Characterization and biological activity of the main flaonoids from Swiss Chard (Beta vulgaris subspecies cycla). Phytomedicine 2007, 14, 216–221. [Google Scholar] [CrossRef] [PubMed]
- Moscona, A. Oseltamivir resistance-disabling our influenza defenses. N. Engl. J. Med. 2005, 22, 2633–2666. [Google Scholar] [CrossRef] [PubMed]
- Hostettmann, K.; Marston, A. Twenty years of research into medicinal plants: Results and perspectives. Phytochem. Rev. 2002, 1, 275–285. [Google Scholar] [CrossRef]
- Ni, L.; Zhou, L.; Zhou, M.; Zhao, J.; Wang, D.W. Combination of western medicine and Chinese traditional patent medicine in treating a family case of COVID-19 in Wuhan. Front. Med. 2020, 14, 210–214. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, R.; Sharma, B.; Kanwar, S.S. Antiviral Phytochemicals: An Overview. Biochem. Physiol. 2017, 6, 2–8. [Google Scholar] [CrossRef]
- Zakaryan, H.; Arabyan, E.; Oo, A.; Zandi, K. Flavonoids: Promising natural compounds against viral infections. Arch. Virol. 2017, 162, 2539–2551. [Google Scholar] [CrossRef]
- Ben-Shabat, S.; Yarmolinsky, L.; Porat, D.; Dahan, A. Antiviral effects of phytochemicals from medicinal plants: Applications and drug delivery strategies. Drug Deliv. Transl. Res. 2020, 10, 354–367. [Google Scholar] [CrossRef]
- Simoes, L.R.; Maciel, G.M.; Brandao, G.C.; Kroon, E.G.; Castilho, R.O.; Oliveira, A.B. Antiviral activity of Disticella elongata (Vahl) Urb. (Bignoniaceae), a potentially useful source of anti-dengue drugs from the state of Minas Gerais, Brazil. Lett. Appl. Microbiol. 2011, 53, 602–607. [Google Scholar] [CrossRef]
- Manvar, D.; Mishra, M.; Kumar, S.; Pandey, V.N. Identification and evaluation of anti hepatitis C virus phytochemicals from Eclipta alba. J. Ethnopharmacol. 2012, 144, 545–554. [Google Scholar] [CrossRef]
- Knipping, K.; Garssen, J.; van’t Land, B. An evaluation of the inhibitory effects against rotavirus infection of edible plant extracts. Virol. J. 2012, 9, 137–144. [Google Scholar] [CrossRef]
- Ding, F.; Liu, J.; Du, R.; Yu, Q.; Gong, L.; Jiang, H.; Rong, R. Qualitative and Quantitative Analysis for the Chemical Constituents of Tetrastigma hemsleyanum Diels et Gilg Using Ultra-High Performance Liquid Chromatography/Hybrid Quadrupole—Orbitrap Mass Spectrometry and Preliminary Screening for Anti-Influenza Virus Components. Evid. Based Complement. Alternat. Med. 2019, 2019, 9414926. [Google Scholar] [PubMed]
- Ji, S.; Li, R.; Wang, Q.; Miao, W.J.; Li, Z.W.; Si, L.L.; Qiao, X.; Yu, S.W.; Zhou, D.M.; Ye, M. Anti-H1N1 virus, cytotoxic and Nrf2 activation activities of chemical constituents from Scutellaria baicalensis. J. Ethnopharmacol. 2015, 176, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Qamar, M.T.; Mumtaz, A.; Naseem, R.; Ali, A.; Fatima, T.; Jabbar, T.; Ahmad, Z.; Ashfaq, U.A. Molecular Docking Based Screening of Plant Flavonoids as Dengue NS1 inhibitors. Bioinformation 2014, 10, 460–465. [Google Scholar] [CrossRef] [PubMed]
- Di Sotto, A.; Di Giacomo, S.; Amatore, D.; Locatelli, M.; Vitalone, A.; Toniolo, C.; Rotino, G.L.; Lo Scalzo, R.; Palamara, A.T.; Marcocci, M.E.; et al. A Polyphenol Rich Extract from Solanum melongena L. DR2 Peel Exhibits Antioxidant Properties and Anti-Herpes Simplex Virus Type 1 Activity In Vitro. Molecules 2018, 23, 2066. [Google Scholar] [CrossRef]
- Mirza, M.U.; Ghori, N.U.; Ikram, N.; Adil, A.R.; Manzoor, S. Pharmacoinformatics approach for investigation of alternative potential hepatitis C virus nonstructural protein 5B inhibitors. Drug Des. Devel. Ther. 2015, 9, 1825–1841. [Google Scholar] [CrossRef]
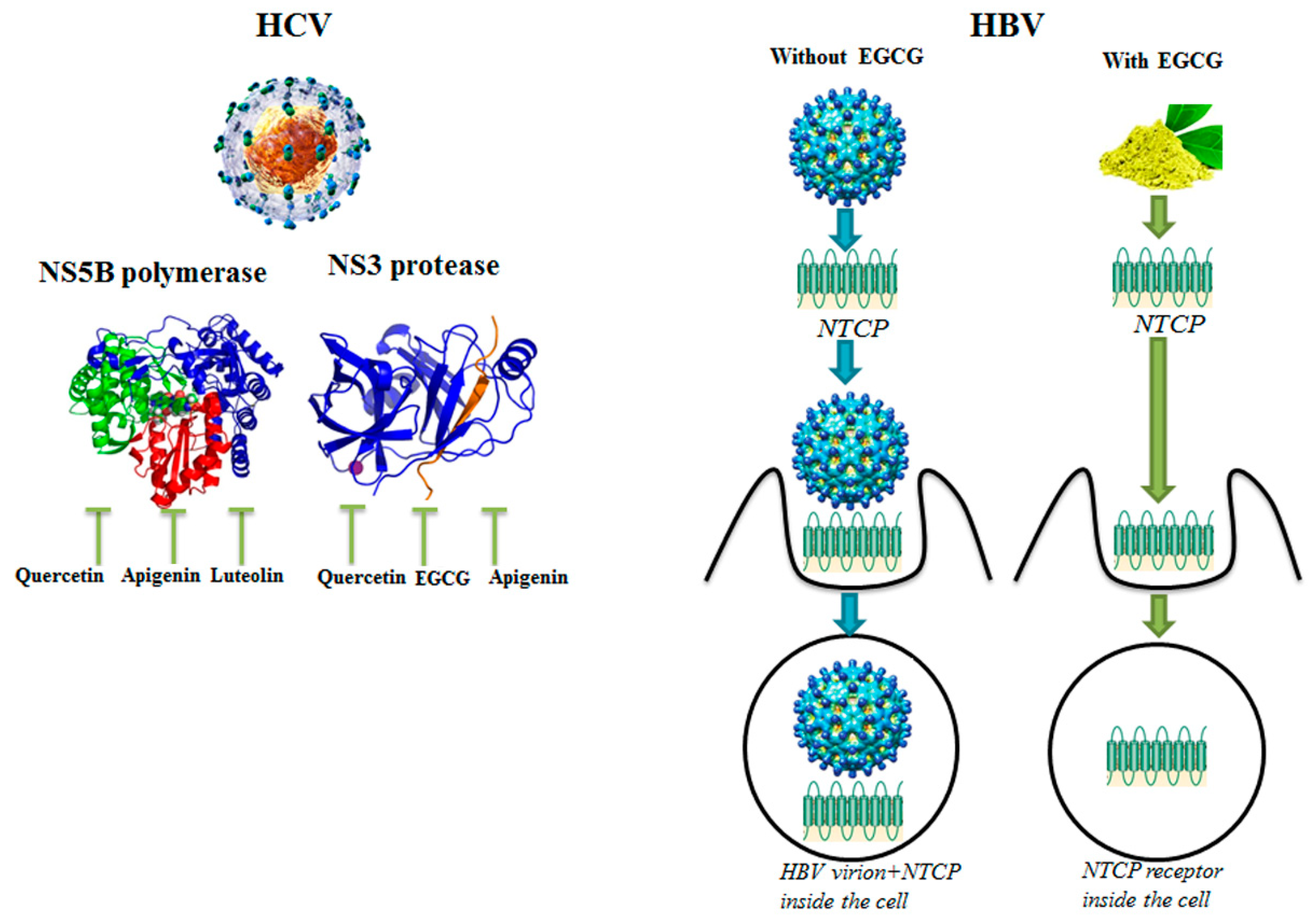
- Huang, H.C.; Tao, M.H.; Hung, T.M.; Chen, J.C.; Lin, Z.J.; Huang, C. (-)-Epigallocatechin-3-gallate inhibits entry of hepatitis B virus into hepatocytes. Antivir. Res. 2014, 111, 100–111. [Google Scholar] [CrossRef]
- Kehinde, I.; Ramharack, P.; Nlooto, M.; Gordon, M. The pharmacokinetic properties of HIV-1 protease inhibitors: A computational perspective on herbal phytochemicals. Heliyon 2019, 5, e02565. [Google Scholar] [CrossRef]
- Rehman, S.; Ashfaq, U.A.; Ijaz, B.; Riazuddin, S. Anti-hepatitis C virus activity and synergistic effect of Nymphaea alba extracts and bioactive constituents in liver infected cells. Microb. Pathog. 2018, 121, 198–209. [Google Scholar] [CrossRef]
- Anwar, F.; Latif, S.; Ashraf, M.; Gilani, A.H. Moringa oleifera: A food plant with multiple medicinal uses. Phytother. Res. 2007, 21, 17–25. [Google Scholar] [CrossRef]
- Haid, S.; Novodomskà, A.; Gentzsch, J.; Grethe, C.; Geuenich, S.; Bankwitz, D.; Chhatwal, P.; Jannack, B.; Hennebelle, T.; Bailleul, F.; et al. A Plant-Derived Flavonoid Inhibits Entry of All HCV Genotypes Into Human Hepatocytes. Gastroenterology 2012, 143, 213–222. [Google Scholar] [CrossRef]
- Ortega, J.T.; Suarez, A.I.; Serrano, M.L.; Baptista, J.; Pujol, F.H.; Rangel, H.R. The role of the glycosyl moiety of myricetin derivatives in anti-HIV-1 activity in vitro. AIDS Res. Ther. 2017, 14, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.J.; Wu, X.; Li, M.M.; Li, G.Q.; Yang, Y.T.; Luo, H.J.; Huang, W.H.; Chung, H.Y.; Ye, W.C.; Wang, G.C.; et al. Antiviral activity of polymethoxylated flavones from Guangchenpi, the edible and medicinal pericarps of Citrus reticulata “Chachi”. J. Agric. Food Chem. 2014, 62, 2182–2189. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.J.; Loe, M.W.; Lee, R.C.; Chu, J.J. Antiviral activity of pinocembrin against Zika virus replication. Antivir. Res. 2019, 167, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, R.; Wu, J.; Shen, Q. Characterization and evaluation of self-microemulsifying sustained-release pellet formulation of puerarin for oral delivery. Int. J. Pharm. 2012, 427, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Ghildiyal, R.; Prakash, V.; Chaudhary, V.K.; Gupta, V.; Gabrani, R. Phytochemicals as Antiviral Agents: Recent Updates. In Plant-Derived Bioactives, 1st ed.; Swany, M.K., Ed.; Springer Nature: Singapore, 2020; Volume 12, pp. 279–295. [Google Scholar]
- Bachmetov, L.; Gal-Tanamy, M.; Shapira, A.; Vorobeychik, M.; Giterman-Galam, T.; Sathiyamoorthy, P.; Golan-Goldhirsh, A.; Benhar, I.; Tur-Kaspa, R.; Zemel, R. Suppression of hepatitis C virus by the flavonoid quercetin is mediated by inhibition of NS3 protease activity. J. Viral Hepat. 2012, 19, 81–88. [Google Scholar] [CrossRef]
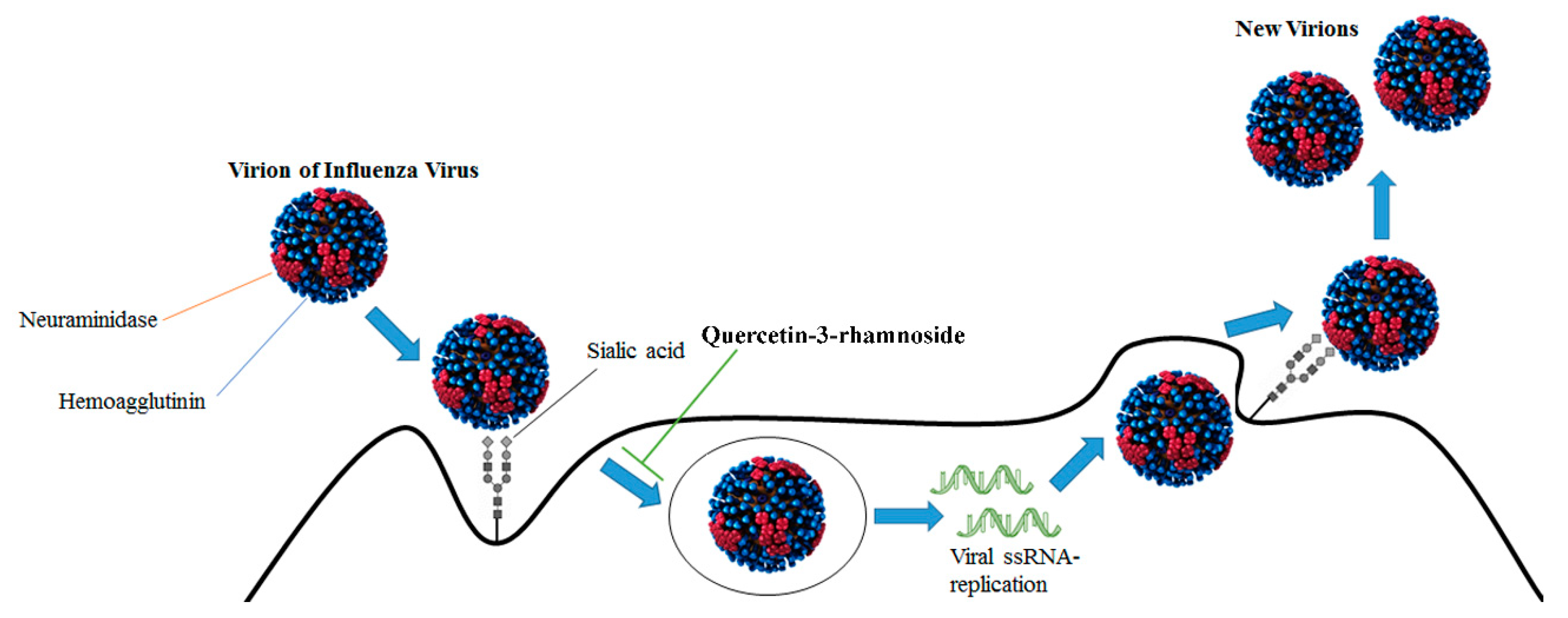
- Choi, H.J.; Song, J.H.; Park, K.S.; Kwon, D.H. Inhibitory effects of quercetin 3-rhamnoside on influenza A virus replication. Eur. J. Pharm. Sci. 2009, 37, 329–333. [Google Scholar] [CrossRef]
- Shi, D.; Chen, M.; Liu, L.; Wang, Q.; Liu, S.; Wang, L.; Wang, R. Anti-influenza A virus mechanism of three representative compounds from Flos Trollii via TLRs signaling pathways. J. Ethnopharmacol. 2020, 253, 112634. [Google Scholar] [CrossRef]
- Ahmed-Belkacem, A.; Guichou, J.F.; Brillet, R.; Ahnou, N.; Hernandez, E.; Pallier, C.; Pawlotsky, J.M. Inhibition of RNA binding to hepatitis C virus RNA-dependent RNA polymerase: A new mechanism for antiviral intervention. Nucleic Acid Res. 2014, 42, 9399–9409. [Google Scholar] [CrossRef]
- Rehman, S.; Ijaz, B.; Fatima, N.; Muhammad, S.A.; Riazuddin, S. Therapeutic potential of Taraxacum officinale against HCV NS5B polymerase: In-vitro and In silico study. Biomed. Pharmacother. 2016, 83, 881–891. [Google Scholar] [CrossRef]
- Khachatoorian, R.; Arumugaswani, V.; Raychauduri, S.; Yeh, G.K.; Maloney, E.M.; Wang, J.; Dasgupta, A.; French, S.W. Divergent antiviral effects of bioflavonoids on the hepatitis C virus life cycle. Virology 2012, 433, 346–355. [Google Scholar] [CrossRef]
- Fahmy, N.M.; Al-Sayed, E.; Moghannem, S.; Azam, F.; El-Shazly, M.; Singab, A.N. Breaking Down the Barriers to a Natural Antiviral Agent: Antiviral Activity and Molecular Docking of Erythrina speciosa Extract, Fractions, and the Major Compound. Chem. Biodivers. 2020, 17, e1900511. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Hattori, T.; Kodama, E.N. Epigallocatechin gallate inhibits the HIV reverse transcription step. Antivir. Chem. Chemother. 2011, 4, 239–243. [Google Scholar] [CrossRef]
- Moghaddam, E.; Teoh, B.T.; Sam, S.S.; Lani, R.; Hassandarvish, P.; Chik, Z.; Yueh, A.; Abubakar, S.; Zandi, K. Baicalin, a metabolite of baicalein with antiviral activity against dengue virus. Sci. Rep. 2014, 5, 5452–5459. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.J.; Song, J.H.; Kwon, D.H. Quercetin-3-rhamnoside exerts antiinfluenza A virus activity in mice. Phytother. Res. 2012, 26, 462–464. [Google Scholar] [CrossRef] [PubMed]
- Virology Blog: About Viruses and Viral Disease. Available online: https://www.virology.ws/2014/12/10/how-influenza-virus-infection-might-lead-to-gastrointestinal-symptoms/ (accessed on 1 June 2020).
- Glanz, V.Y.; Myasoedova, V.A.; Grechko, A.V.; Orekhov, A.N. Inhibition of sialidase activity as a therapeutic approach. Drug Des. Dev. Ther. 2018, 12, 3431–3437. [Google Scholar] [CrossRef]
- Iwai, Y.; Murakami, K.; Gomi, Y.; Hashimoto, T.; Asakawa, Y.; Okuno, Y.; Ishikawa, T.; Hatakeyama, D.; Echigo, N.; Kuzuhara, T. Anti-influenza activity of marchantins, macrocyclic bisbibenxyls contained in liverworts. PLoS ONE 2011, 6, e19825. [Google Scholar] [CrossRef]
- Hepatitis C Virus. Available online: https://en.wikipedia.org/wiki/Hepatitis_C_virus (accessed on 1 June 2020).
- HBV-GLUE: A Sequence Data Resource for Hepatitis B Virus. Available online: http://hbv-glue.cvr.gla.ac.uk/#/home (accessed on 1 June 2020).
- Corriere Nazionale: I 5 Virus Che Spaventano La Scienza. Available online: https://www.corrierenazionale.it/2020/02/09/i-5-virus-che-spaventano-la-scienza/ (accessed on 1 June 2020).
- De Chiara, G.; Marcocci, M.E.; Sgarbanti, R.; Civitelli, L.; Ripoli, C.; Piacentini, R.; Garaci, E.; Grassi, C.; Palamara, A.T. Infectious agents and neurodegeneration. Mol. Neurobiol. 2012, 46, 614–638. [Google Scholar] [CrossRef]
- Amatore, D.; Sgarbanti, R.; Aquilano, K.; Baldelli, S.; Limongi, D.; Civitelli, L.; Nencioni, L.; Garaci, E.; Ciriolo, M.R.; Palamara, A.T. Influenza virus replication in lung epithelial cells depends on redox-sensitive pathways activated by NOX4-derived ROS. Cell. Microbiol. 2015, 17, 131–145. [Google Scholar] [CrossRef]
- Sharifi-Rad, J.; Iriti, M.; Setzer, W.N.; Sharifi-Rad, M.; Roointan, A.; Salehi, B. Antiviral activity of Veronica Persica Poir. on herpes virus infection. Cell. Mol. Biol. 2018, 64, 11–17. [Google Scholar] [CrossRef]
- Lembo, D.; Cavalli, R. Nanoparticulate delivery systems for antiviral drugs. Antivir. Chem. Chemother. 2010, 21, 53–70. [Google Scholar] [CrossRef]
- Dube, A.; Nicolazzo, J.A.; Larson, I. Chitosan nanoparticles enhance the intestinal absorption of the green tea catechins (+)-catechin and (-)-epigallocatechin gallate. Eur. J. Pharm. Sci. 2010, 41, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Casettari, L.; Gennari, L.; Angelino, D.; Ninfali, P.; Castagnino, E. ORAC of chitosan and its derivatives. Food Hydrocoll. 2012, 28, 243–247. [Google Scholar] [CrossRef]
- Sims, K.R.; He, B.; Koo, H.; Benoit, D.S.W. Electrostatic Interactions Enable Nanoparticle Delivery of the Flavonoid Myricetin. ACS Omega 2020, 28, 12649–12659. [Google Scholar] [CrossRef] [PubMed]
- Kang, B.K.; Lee, J.S.; Chon, S.K.; Jeong, S.Y.; Yuk, S.H.; Khang, G.; Lee, H.B.; Cho, S.H. Development of self-microemulsifying drug delivery system (SMEDDS) for oral bioavailability enhancement of simvastatin in beagle dogs. Int. J. Pharm. 2004, 274, 65–73. [Google Scholar] [CrossRef]
- Koduru, J.R.; Kailasa, S.K.; Bhamore, J.R.; Kim, K.H.; Dutta, T.; Vellingiri, K. Phytochemical-assisted synthetic approaches for silver nanoparticles antimicrobial applications: A review. Adv. Colloid Interface Sci. 2018, 256, 326–339. [Google Scholar] [CrossRef]
- Kumari, A.; Yadav, S.K.; Pakade, Y.B.; Singh, B.; Yadav, S.C. Development of biodegradble nanoparticles for delivery of quercetin. Colloids Surf. B Biointerfaces 2010, 80, 184–192. [Google Scholar] [CrossRef]
- Zhai, Y.; Guo, S.; Liu, C.; Yang, C.; Dou, J.; Li, L.; Zhai, G. Preparation and in vitro evaluation of apigenin-loaded polymeric micelles. Colloids Surf. A 2013, 429, 24–30. [Google Scholar] [CrossRef]
- Kumari, A.; Kumar, V.; Yadav, S.K. Plant extract synthesized PLA nanoparticles for controlled and sustained release of quercetin: A green approach. PLoS ONE 2012, 7, e41230. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Ballesta, M.C.; Gil-Izquierdo, Á.; García-Viguera, C.; Domínguez-Perles, R. Nanoparticles and Controlled Delivery for Bioactive Compounds: Outlining Challenges for New “Smart-Foods” for Health. Foods 2018, 7, 72. [Google Scholar] [CrossRef]
- Magnani, M.; Balestra, E.; Fraternale, A.; Aquaro, S.; Paiardini, M.; Cervasi, B.; Casabianca, A.; Garaci, E.; Perno, C.F. Drug-loaded red blood cell-mediated clearance of HIV-1 macrophage reservoir by selective inhibition of STAT1 expression. J. Leukoc. Biol. 2003, 74, 764–771. [Google Scholar] [CrossRef]
- Jeewantha, H.M.A.; Slivkin, A.I. The terpene-indole alkaloids loaded erythrocytes as a drug carrier: Design and assessment. Clin. Pharmacol. 2018, 7, e0406. [Google Scholar] [CrossRef]
- Trineeva, O.V.; Khalahakun, A.D. Study of desorbtion and exemption of terpeno-indole alkaloids of vinkristin and vinblastin from erythrocitary cell carriers. Drug Dev. Regist. 2019, 8, 16–21. [Google Scholar]
- Blasa, M.; Angelino, D.; Gennari, L.; Ninfali, P. The cellular antioxidant activity in red blood cells (CAA-RBC): A new approach to bioavailability and synergy of phytochemicals and botanical extracts. Food Chem. 2011, 125, 685–691. [Google Scholar] [CrossRef]
- Asgary, S.; Naderi, G.H.; Askari, N. Protective effect of flavonoids against red blood cell hemolysis by free radicals. Exp. Clin. Cardiol. 2005, 2, 10. [Google Scholar]
- Hou, L.; Zhou, B.; Yang, L.; Liu, Z.L. Inhibition of free radical initiated peroxidation of human erythrocyte ghosts by flavonols and their glycosides. Org. Biomol. Chem. 2004, 2, 1419–1423. [Google Scholar] [CrossRef]
- Fiorani, M.; Accorsi, A.; Cantoni, O. Human Red Blood Cell sas a Natural Flavonoid Reservoir. Free Radic. Res. 2003, 37, 1331–1338. [Google Scholar] [CrossRef]
- Serafini, S.; Rossi, L.; Antonelli, A.; Fraternale, A.; Cerasi, A.; Crinelli, R.; Chiarantini, L.; Schiavano, G.F.; Magnani, M. Drug delivery through phagocytosis of red blood cells. Transfus. Med. Hemotherapy 2004, 31, 92–101. [Google Scholar] [CrossRef]
- Lalani, S.; Poh, C.L. Flavonoids as Antiviral Agents for Enterovirus A71 (EV-A71). Viruses 2020, 12, 184. [Google Scholar] [CrossRef]
- Xia, Q.; Zhang, Y.; Li, Z.; Hou, X.; Feng, N. Red blood cell membrane-camouflaged nanoparticles: A novel drug delivery system for antitumor application. Acta Pharm. Sin. B 2019, 9, 675–689. [Google Scholar] [CrossRef]
- Doshi, N.; Zahr, A.S.; Bhaskar, S.; Lahann, J.; Mitragotri, S. Red blood cell-mimicking synthetic biomaterial particles. Proc. Natl. Acad. Sci. USA 2009, 106, 21495–21499. [Google Scholar] [CrossRef]
- Merkel, T.J.; Jones, S.W.; Herlihy, K.P.; Kersey, F.R.; Shields, A.; Napier, M.; Luft, J.C.; Wu, H.; Zamboni, W.C.; Wang, A.; et al. Using mechanobiological mimicry of red blood cells to extend circulation times of hydrogel microparticles. Proc. Natl. Acad. Sci. USA 2011, 108, 586–591. [Google Scholar] [CrossRef] [PubMed]
- Antonelli, A.; Magnani, M. Engineering erythrocytes for the modulation of drugs’ and contrasting agents’ pharmacokinetics and biodistribution. Adv. Drug Deliv. Rev. 2016, 106, 73–87. [Google Scholar]
- Magnani, M.; Serafini, S.; Fraternale, A.; Antonelli, A.; Biagiotti, S.; Pierigè, F.; Sfara, C.; Rossi, L. Red blood cell-based delivery of drugs and nanomaterials for therapeutic and diagnostic applications. In Encyclopedia of Nanoscience and Nanotechnology, 1st ed.; Nalwa, H.S., Ed.; ASP: Brussels, Belgium, 2011; Volume 22, pp. 309–354. [Google Scholar]
- Villa, C.H.; Pan, D.C.; Zaitsev, S.; Cines, D.B.; Siegel, D.L.; Muzykantov, V.R. Delivery of drugs bound to erythrocytes: New avenues for an old intravascular carrier. Ther. Deliv. 2015, 6, 795–826. [Google Scholar] [CrossRef] [PubMed]
- Flower, R.; Peiretti, E.; Magnani, M.; Rossi, L.; Serafini, S.; Gryczynski, Z.; Gryczynski, I. Observation of erythrocyte dynamics in the retinal capillaries and choriocapillaris using ICG-loaded erythrocyte ghost cells. Investig. Ophthalmol. Vis. Sci. 2008, 49, 5510–5516. [Google Scholar] [CrossRef] [PubMed]
- Villa, C.H.; Cines, D.B.; Siegel, D.L.; Muzykantov, V. Erythrocytes as carriers for drug delivery in blood transfusion and beyond. Transfus. Med. Rev. 2017, 31, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Sirisinha, S. The potential impact of gut microbiota on your health: Current status and future challenges. Asian Pac. J. Allergy Immunol. 2016, 34, 429–464. [Google Scholar]
- Gibson, A.; Edgar, J.D.; Neville, C.E.; Gilchrist, S.; McKinley, M.C.; Patterson, C.; Young, I.S.; Woodside, J.V. Effect of fruit and vegetable consumption on immune function in older people: A randomized controlled trial. Am. J. Clin. Nutr. 2012, 96, 1429–1436. [Google Scholar] [CrossRef]
- Koper, J.E.B.; Loonen, L.M.P.; Wells, J.M.; Troise, A.D.; Capuano, E.; Fogliano, V. Polyphenols and Tryptophan Metabolites Activate the Aryl Hydrocarbon receptor in an in vitro Model of Colonic Fermentation. Mol. Nutr. Food Res. 2019, 63, e1800722. [Google Scholar] [CrossRef]
- Dai, W.; Bi, J.; Li, F.; Wang, S.; Huang, X.; Meng, X.; Sun, B.; Wang, D.; Kong, W.; Jiang, C.; et al. Antiviral Efficacy of Flavonoids Against Enterovirus 71 Infection in Vitro and in Newborn Mice. Viruses 2019, 11, 625. [Google Scholar] [CrossRef]
- Dayem, A.A.; Choi, H.Y.; Kim, Y.B.; Cho, S.G. Antiviral effect of methylated flavonol isorhamnetin against influenza. PLoS ONE 2015, 10, e0121610. [Google Scholar] [CrossRef]
- Guo, Q.; Zhao, L.; You, Q.; Yang, Y.; Gu, H.; Song, G.; Lu, N.; Xin, J. Anti-hepatitis B virus activity of wogonin in vitro and in vivo. Antivir. Res. 2007, 74, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Tang, Y.; Gao, C.; Li, Y.; Chen, S.; Xiong, T.; Li, J.; Du, M.; Gong, Z.; Chen, H.; et al. Characterization and biodistribution in vivo of quercetin-loaded cationic nanostructured lipid carriers. Colloids Surf. B Biointerfaces 2014, 115, 125–131. [Google Scholar] [CrossRef]
- EC 2017. EC Regulation 258/97. Safety of hydroxytyrosol as a novel food pursuant to Regulation (EC) No 258/97. EFSA J. 2017, 15, e04728. [Google Scholar] [CrossRef]
- Del Bo, C.; Bernardi, S.; Marino, M.; Porrini, M.; Tucci, M.; Guglielmetti, S.; Cherubini, A.; Carrieri, B.; Kirkup, B.; Kroon, P.; et al. Systematic Review on Polyphenol Intake and Health Outcomes: Is There Sufficient Evidence to Define a Health-Promoting Polyphenol-Rich Dietary Pattern? Nutrients 2019, 11, 1355. [Google Scholar]
- Perez-Jimenez, J.; Fezeu, L.; Touvier, M.; Arnault, N.; Manach, C.; Hercberg, S.; Galan, P.; Scalbert, A. Dietary Intake of 337 Polyphenols in French Adults. Am. J. Clin. Nutr. 2011, 93, 1220–1228. [Google Scholar] [CrossRef]
- Brickman, A.M.; Khan, U.A.; Provenzano, F.A.; Yeung, L.K.; Suzuki, W.; Schroeter, H.; Wall, M.; Sloan, R.P.; Small, S.A. Enhancing Dentate Gyrus Function With Dietary Flavanols Improves Cognition in Older Adults. Nat. Neurosci. 2014, 17, 1798–1803. [Google Scholar] [CrossRef]
- Ferri, P.; Angelino, D.; Gennari, L.; Benedetti, S.; Ambrogini, P.; Del Grande, P.; Ninfali, P. Enhancement of Flavonoid Ability to Cross the Blood-Brain Barrier of Rats by Co-Administration With α-tocopherol. Food Funct. 2015, 6, 394–400. [Google Scholar] [CrossRef]
- Galanakis, C.M. The Food Systems in the Era of the Coronavirus (COVID-19) Pandemic Crisis. Foods 2020, 9, 523. [Google Scholar] [CrossRef]
- Sanjuan, R.; Nebot, M.R.; Chirico, N.; Mansky, L.M.; Belshaw, R. Viral mutation rates. J. Virol. 2010, 84, 9733–9748. [Google Scholar] [CrossRef]
- Bilia, A.R.; Isacchi, B.; Righeschi, C.; Guccione, C.; Bergonzi, M.C. Flavonoids loaded in Nanocarriers: An Opportunity to Increase Oral Bioavailability and Bioefficacy. Food Nutr. Sci. 2014, 5, 1212–1227. [Google Scholar] [CrossRef]
- Cojocaru, F.D.; Botezat, D.; Gardikiotis, I.; Uritu, C.M.; Dodi, G.; Trandafir, L.; Rezus, C.; Rezus, E.; Tamba, B.I.; Mihai, C.T. Nanomaterials Designed for Antiviral Drug Delivery Transport across Biological Barriers. Pharmaceutics 2020, 12, 171. [Google Scholar] [CrossRef] [PubMed]
- EU 2015. Commission Regulation (EU) 2015/2283. Regulation (EU) 2015/2283 of the European Parliament and of the Council of 25 November 2015 on novel foods, amending Regulation (EU) No 1169/2011 of the European Parliament and of the Council and repealing Regulation (EC) No 258/97 of the European Parliament and of the Council and Commission Regulation (EC) No 1852/2001. OJL 2015, 327, 1–22. [Google Scholar]
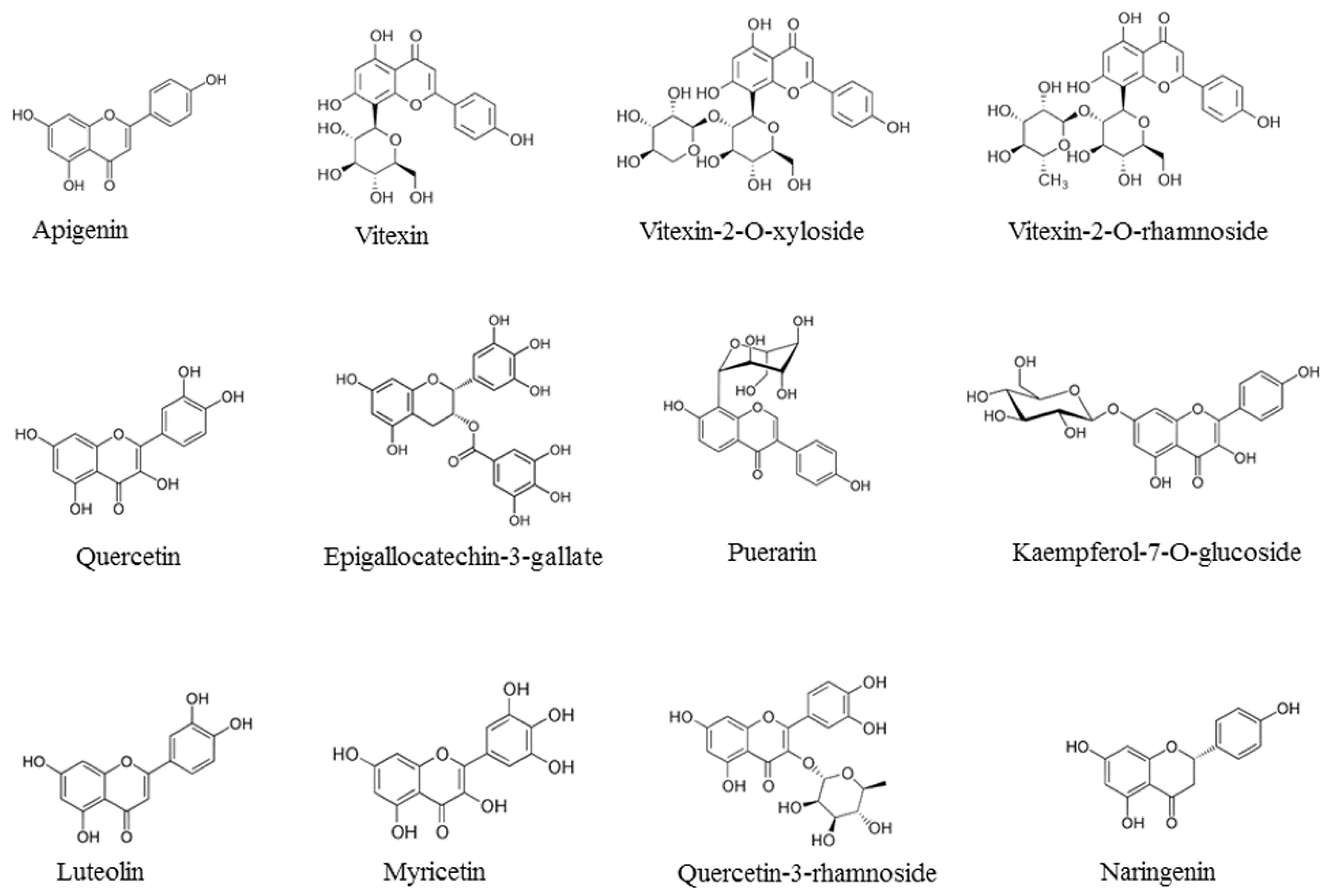
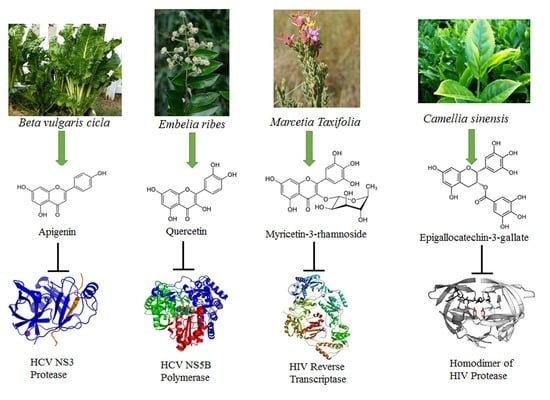
| Flavonoid | Class | Sources | Ref. |
|---|---|---|---|
| Acacetin-7-O-rutinoside | Flavone | Disticella elongata | [18] |
| Apigenin | Flavone | Eclipta alba | [19] |
| Apigenin-7-O-glucoside | Flavone | Melissa officinalis | [20] |
| Astragalin | Flavanol | Tetrastigma hemsleyanum | [21] |
| Baicalein | Flavone | Scutellaria baicalensis | [22] |
| Baicalin | Flavone | Scutellaria baicalensis | [22] |
| Chamaejasmin | Flavanone | Enkleia siamensis | [23] |
| Chrysin | Flavone | Scutellaria baicalensis | [22] |
| Delphinidin-3-rutinoside | Anthocyanidin | Solanum Melongena | [24] |
| Diosmetin | Flavone | Pistacia chinensis | [25] |
| Epigallocatechin-3-gallate | Flavin | Camellia sinensis | [26] |
| Fisetin | Flavanol | Carica papaya | [27] |
| Hyperoside | Flavanol | Nymphaea alba | [28] |
| Isokaempferide | Flavanol | Nymphaea alba | [28] |
| Isorhamnetin | Flavanol | Tetrastigma hemsleyanum | [21] |
| Isoquercetin | Flavanol | Tetrastigma hemsleyanum | [21] |
| Kaempferol | Flavanol | Moringa oleifera Lam | [29] |
| Kaempferol-7-O-glucoside | Flavanol | Carica papaya | [27] |
| Ladanein | Flavone | Marrubium peregrinum | [30] |
| Luteolin | Flavone | Eclipta alba | [19] |
| Myricetin | Flavanol | Marcetia taxifolia | [31] |
| Myricetin-3-rhamnoside | Flavanol | Marcetia taxifolia | [31] |
| Myricetin-3-(6-rhamnosylgalactoside) | Flavanol | Marcetia taxifolia | [31] |
| Naringenin | Flavanone | Citrus grandis Osbeck | [25] |
| Nobiletin | Flavone | Citrus reticulate | [32] |
| Oroxylin A | Flavone | Scutellaria baicalensis | [22] |
| Pectolinarin | Flavone | Disticella elongata | [18] |
| Pinocembrin | Flavanone | Pinus sibirica | [33] |
| Puerarin | Isoflavone | Pueraria lobata | [34] |
| Quercetagetin | Flavanol | Tagetes mandonii | [35] |
| Quercetin | Flavanol | Embelia ribes | [36] |
| Quercetin-3-rhamnoside | Flavanol | Houttuynia cordata | [37] |
| Quercitrin | Flavanol | Tetrastigma hemsleyanum | [21] |
| Reynoutrin | Flavanol | Nymphaea alba | [28] |
| Rutin | Flavanol | Tetrastigma hemsleyanum | [21] |
| Sanggenon O | Flavanone | Morus alba | [23] |
| Tangeretin | Flavone | Citrus reticulate | [32] |
| Vitexin | Flavone | Flos Trollii | [38] |
| Vitexin-2-O-rhamnoside | Flavone | Tetrastigma hemsleyanum | [21] |
| Vitexin-2-O-xyloside | Flavone | Beta vulgaris cicla | [10] |
| Wogonin | Flavone | Scutellaria baicalensis | [22] |
| Flavonoids | Inhibited Virus | Targets | Molecular Mechanisms | Ref. |
|---|---|---|---|---|
| Apigenin, Luteolin | HCV | HCV replicase | Inhibition of HCV replication | [19] |
| Vitexin, Apigenin-7-O-glucoside, | Rhesus Rotavirus | Rotavirus virions | Inhibition of viral replication | [20] |
| Vitexin | H1N1 influenza | TLR3, TLR4, | Decrease of inflammatory injury, | [38] |
| TLR7 pathways | Increase of IFN-β levels | |||
| Vitexin | HSV-1 and HAV | HSV-1 virions, | Inhibition of viral replication | [42] |
| HAV virions | ||||
| Apigenin, Isoquercetin, Quercetin | HCV | NS3 protease | Inhibition of HCV replication | [18] |
| Quercetin-3-rhamnoside | Influenza A/WS/33 | Influenza virions | Inhibition of virus infection | [37] |
| EGCG | HIV | Reverse transcriptase | Inhibition of HIV replication | [43] |
| Myricetin-3-rhamnoside | HIV | Reverse transcriptase | Inhibition of HIV replication | [31] |
| Quercetin, Catechin, Naringenin | HCV | NS5A, HSP70, | Inhibition of viral translation, | [41] |
| HCV virions | Inhibition of virion assembly | |||
| Delphinidin-3-rutinoside | HSV-1 | NOX4 | Inhibition of HSV-1 replication | [24] |
| EGCG | HBV | NTCP receptor | Inhibition of HBV entry into cells | [26] |
| Quercetin | HCV | NS3 protease | Inhibition of HCV replication, | [36] |
| Inhibition of virion production | ||||
| Luteolin, Quercetin | HCV | NS5B polymerase | Inhibition of HCV replication | [33] |
| EGCG, | Dengue | NS1 | Inhibition of NS1 glycosylation | [23] |
| Sanggenon O, Chamaejasmin | ||||
| Baicalin | Dengue Virus-2 (DENV-2) | DENV-2 virions | Inhibition of viral replication, | [40] |
| Viricidal activity | ||||
| Baicalin, Baicalein | H1N1 influenza | Nrf2 | Inhibition of viral replication | [44] |
| Luteolin | HCV | NS5B polymerase | Inhibition of HCV replication | [40] |
| Naringenin, Quercetin | HCV | Envelope 2 protein, | Inhibition of virion assembly, | [41] |
| NS5A, NS3 | Inhibition of HCV entry into cells | |||
| Tangeretin, Nobiletin | RSV | Phosphoprotein P | Inhibition of viral replication, | [32] |
| Inhibition of RSV entry into cells | ||||
| Kaempferol-7-O-glucoside, EGCG | HIV | HIV protease | Inhibition of virion production | [27] |
| Quercetagetin | HCV | NS5B polymerase | Inhibition of RNA binding to NS5B | [39] |
| Pinocembrin | Zika | Viral RNAs | Envelope protein synthesis inhibition | [33] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ninfali, P.; Antonelli, A.; Magnani, M.; Scarpa, E.S. Antiviral Properties of Flavonoids and Delivery Strategies. Nutrients 2020, 12, 2534. https://doi.org/10.3390/nu12092534
Ninfali P, Antonelli A, Magnani M, Scarpa ES. Antiviral Properties of Flavonoids and Delivery Strategies. Nutrients. 2020; 12(9):2534. https://doi.org/10.3390/nu12092534
Chicago/Turabian StyleNinfali, Paolino, Antonella Antonelli, Mauro Magnani, and Emanuele Salvatore Scarpa. 2020. "Antiviral Properties of Flavonoids and Delivery Strategies" Nutrients 12, no. 9: 2534. https://doi.org/10.3390/nu12092534
APA StyleNinfali, P., Antonelli, A., Magnani, M., & Scarpa, E. S. (2020). Antiviral Properties of Flavonoids and Delivery Strategies. Nutrients, 12(9), 2534. https://doi.org/10.3390/nu12092534